Characterization of Relevant Biomarkers for the Diagnosis of Food Allergies: An Overview of the 2S Albumin Family
Abstract
1. Introduction
2. Materials and Methods
2.1. 2S Albumins Isolation and Identification
2.2. Electrophoretic Procedures for Protein Characterization
2.3. Circular Dichroism
2.4. Computational Tools for the Analysis of 2S Albumins Structural Features
2.5. Western Blotting with Patients’ Sera
3. Results
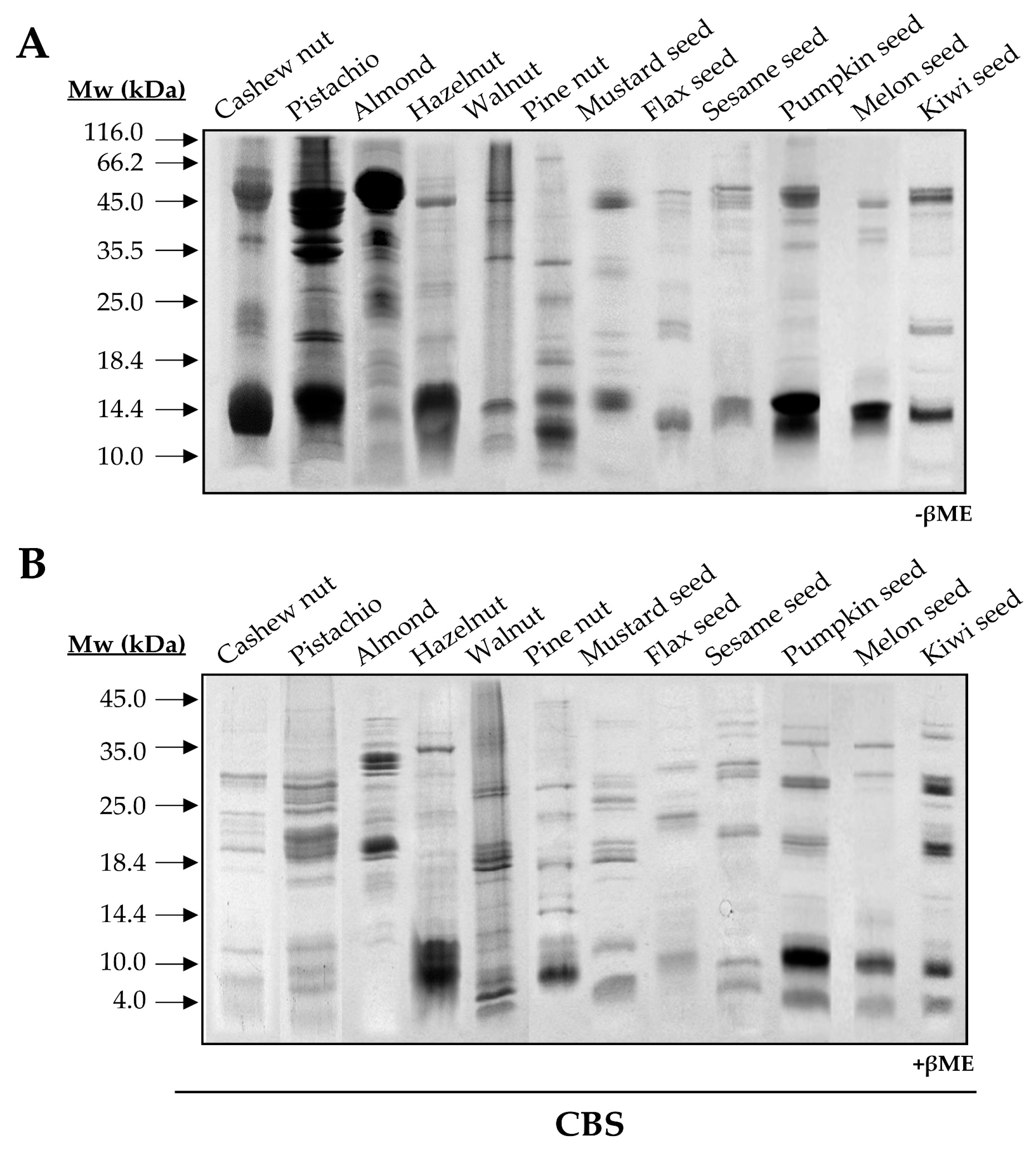
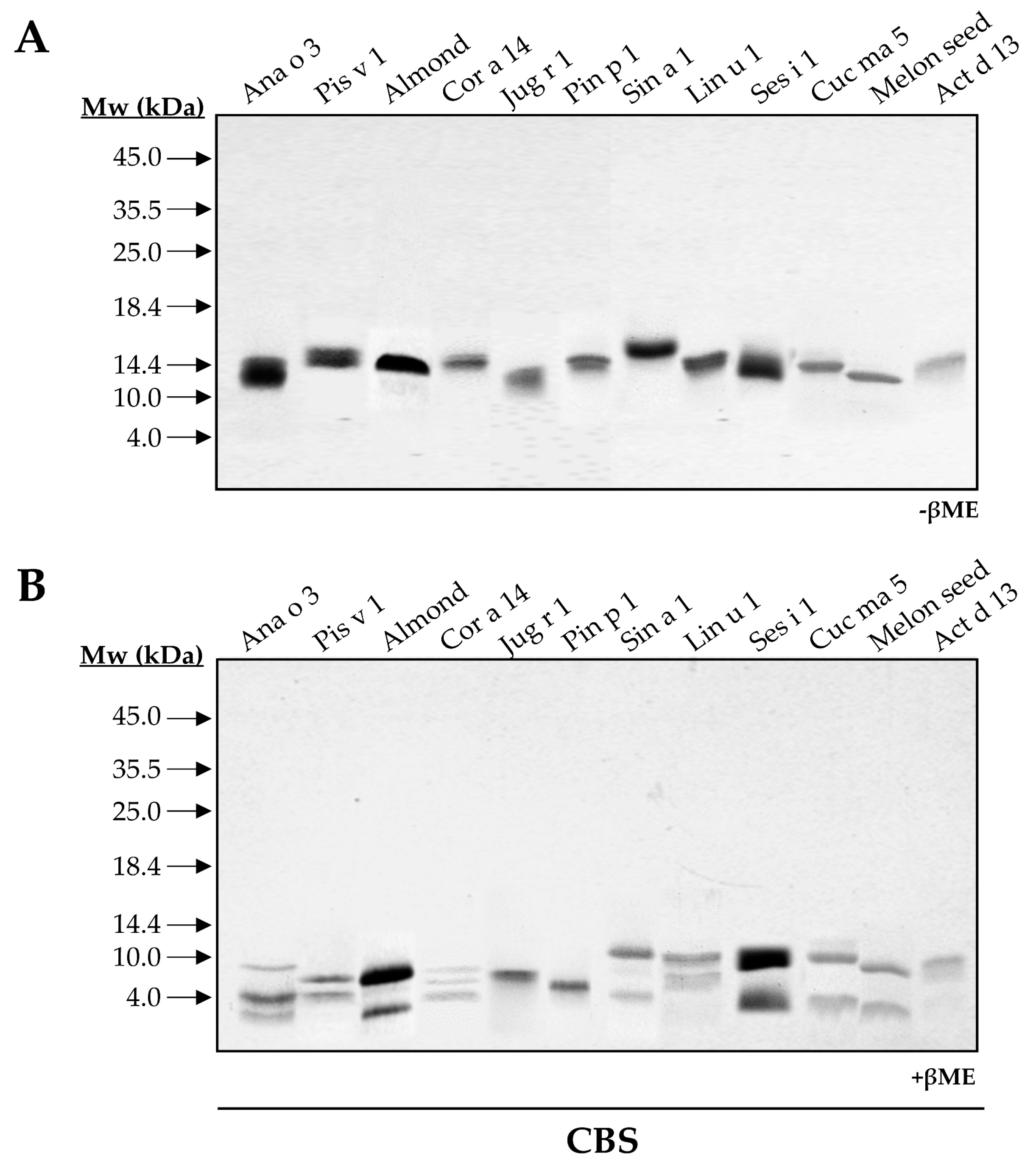
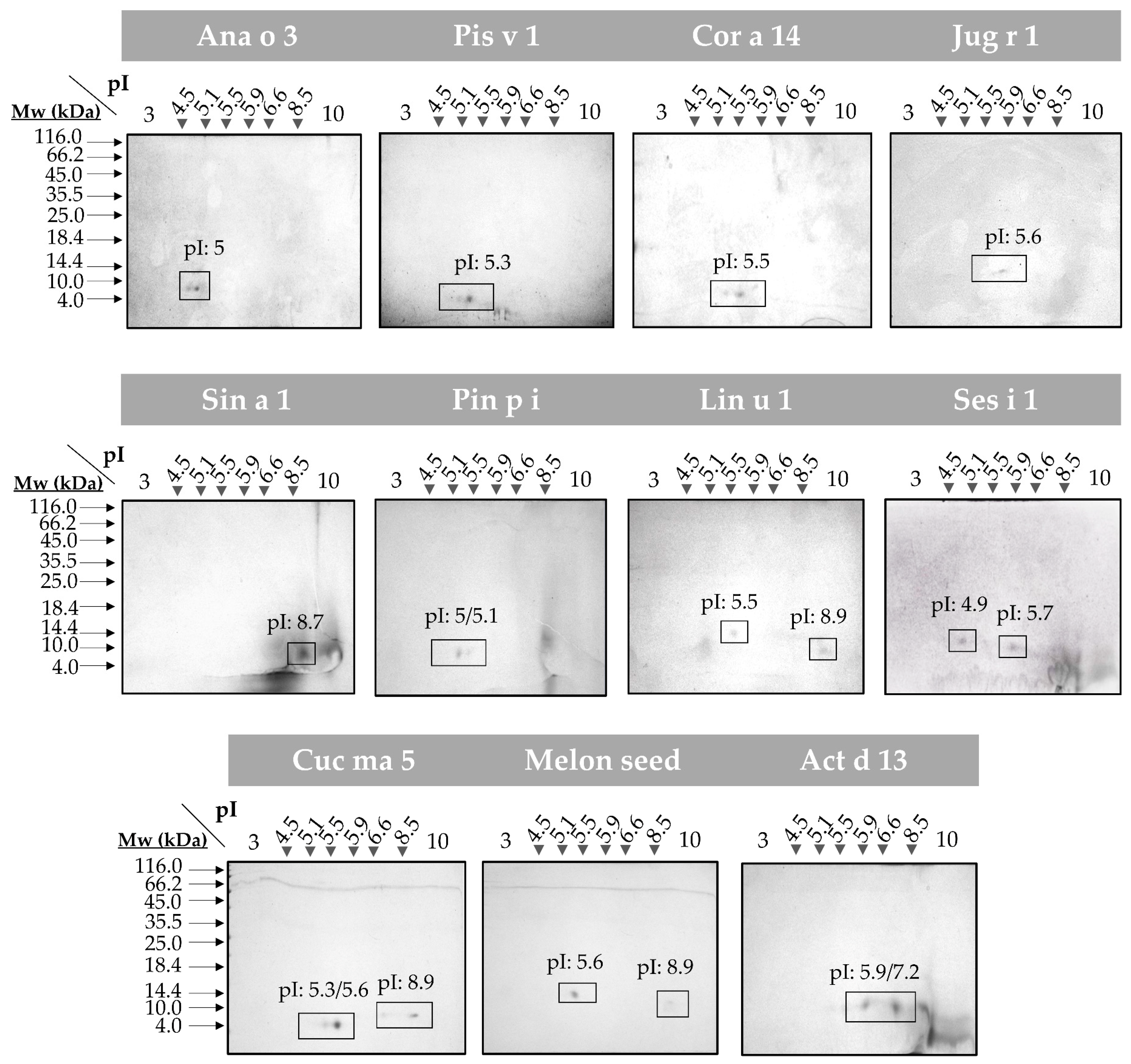
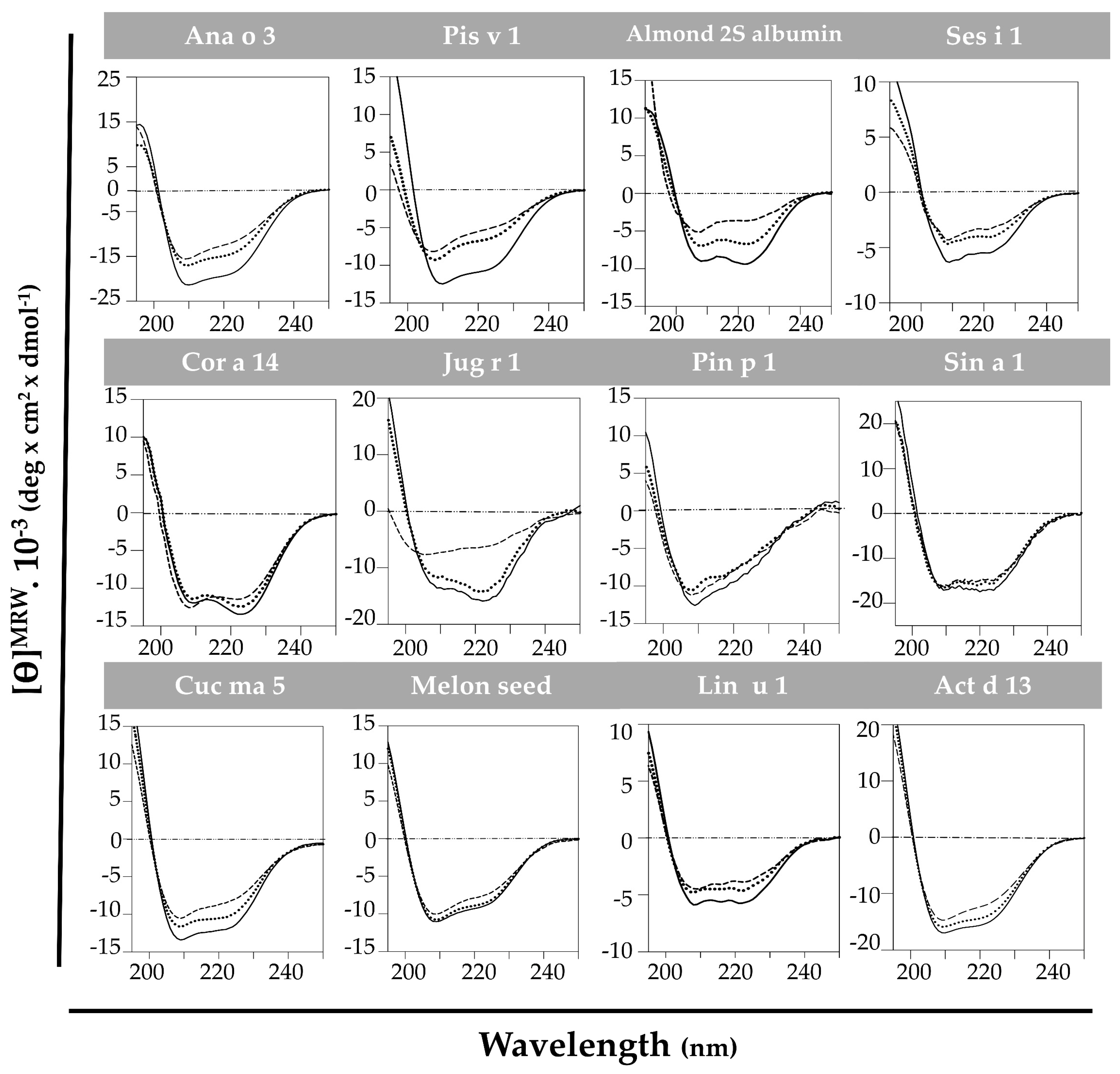
3.1. Experimental Characterization of Isolated 2S Albumins
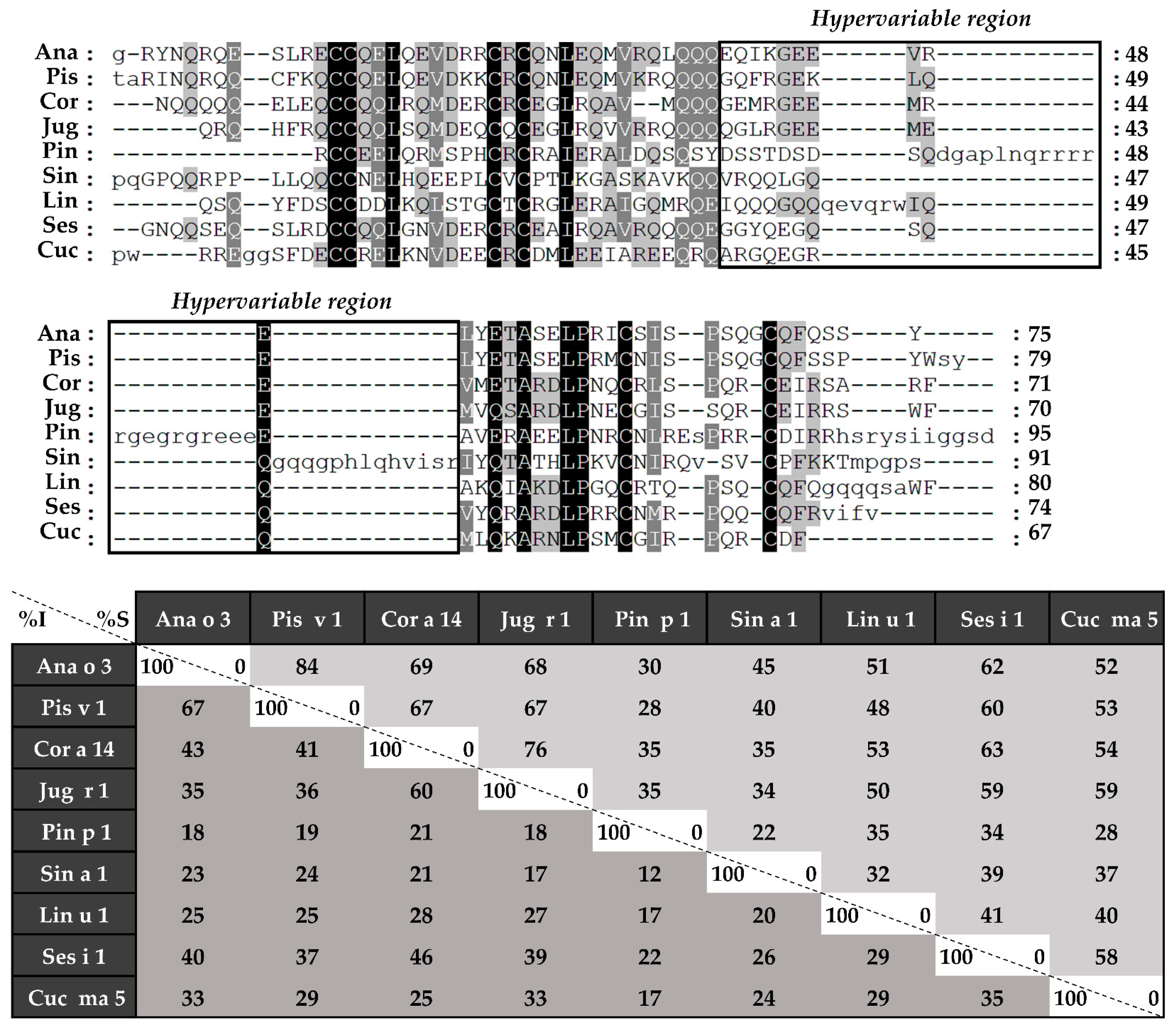
3.1.1. The Polymorphic Nature of 2S Albumins
3.1.2. Structural Behavior of 2S Albumins to Thermal Treatment
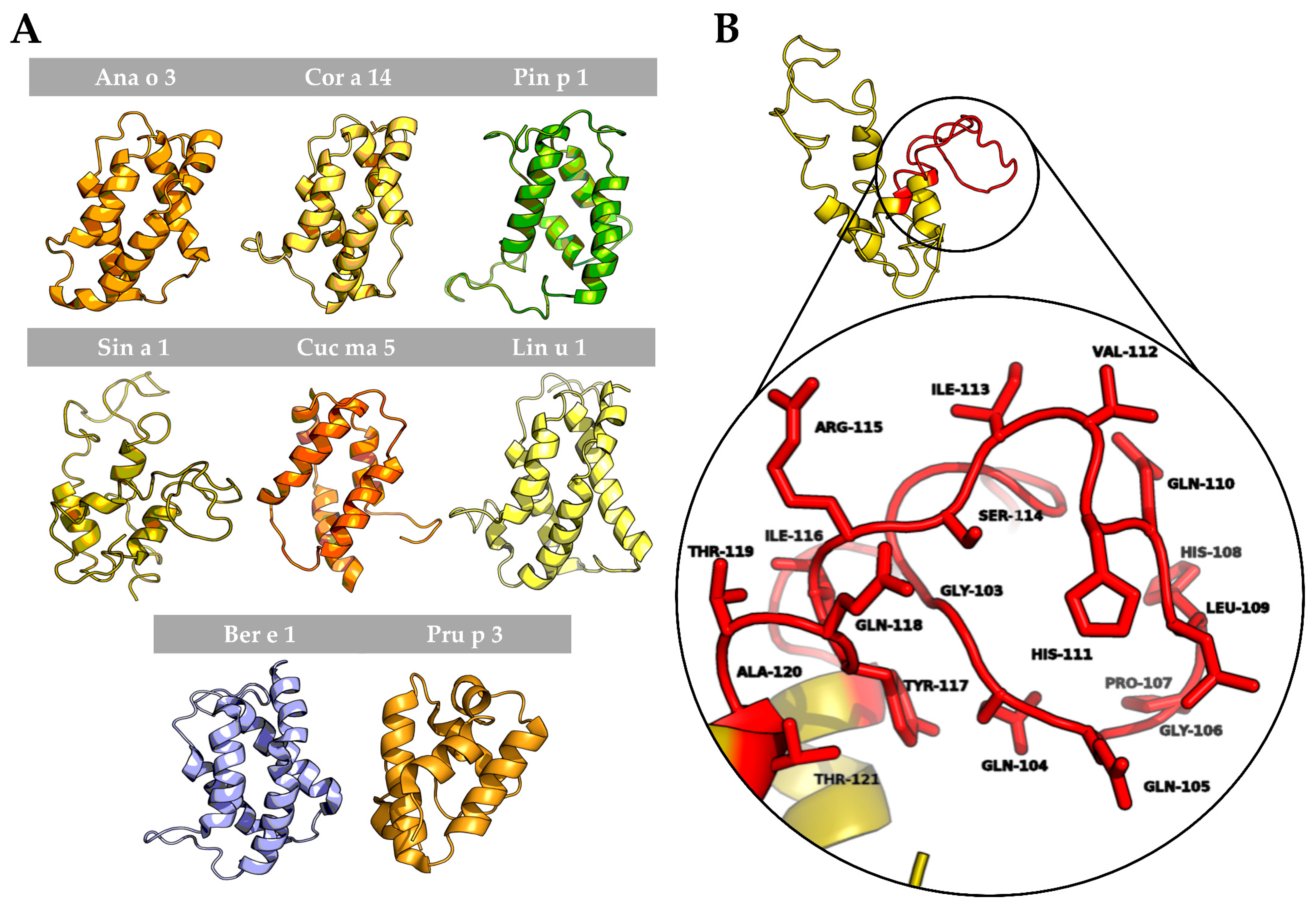
3.2. In Silico Analysis of the 2S Albumin Structures
3.3. The Link between the Structure and the Allergenicity of 2S Albumins
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
References
- Sicherer, S.H.; Sampson, H.A. Food allergy. J. Allergy Clin. Immunol. 2010, 125, S116–S125. [Google Scholar] [CrossRef]
- Weinberger, T.; Sicherer, S. Current perspectives on tree nut allergy: A review. J. Asthma Allergy 2018, 11, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, R.G.; Kleine-Tebbe, J. Molecular Allergy Diagnostics: Analytical Features That Support Clinical Decisions. Curr. Allergy Asthma Rep. 2015, 15, 57. [Google Scholar] [CrossRef] [PubMed]
- Scala, E.; Villalta, D.; Meneguzzi, G.; Giani, M.; Asero, R. Storage molecules from tree nuts, seeds and legumes: Relationships and amino acid identity among homologue molecules. Eur. Ann. Allergy Clin. Immunol. 2018, 50, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Masthoff, L.J.N.; Mattsson, L.; Zuidmeer-Jongejan, L.; Lidholm, J.; Andersson, K.; Akkerdaas, J.H.; Versteeg, S.A.; Garino, C.; Meijer, Y.; Kentie, P.; et al. Sensitization to Cor a 9 and Cor a 14 is highly specific for a hazelnut allergy with objective symptoms in Dutch children and adults. J. Allergy Clin. Immunol. 2013, 132, 393–399. [Google Scholar] [CrossRef]
- Andorf, S.; Borres, M.P.; Block, W.; Tupa, D.; Bollyky, J.B.; Sampath, V.; Elizur, A.; Lidholm, J.; Jones, J.E.; Galli, S.J.; et al. Association of Clinical Reactivity with Sensitization to Allergen Components in Multifood-Allergic Children. J. Allergy Clin. Immunol. Pract. 2017, 5, 1325–1334.e4. [Google Scholar] [CrossRef] [PubMed]
- Moreno, F.J.; Clemente, A. 2S Albumin Storage Proteins: What Makes them Food Allergens? Open Biochem. J. 2008, 2, 16–28. [Google Scholar] [CrossRef]
- Han, Y.; Lin, J.; Bardina, L.; Grishina, G.A.; Lee, C.; Seo, W.H.; Sampson, H.A. What characteristics confer proteins the ability to induce allergic responses? IgE epitope mapping and comparison of the structure of soybean 2S albumins and Ara h 2. Molecules 2016, 21, 622. [Google Scholar] [CrossRef]
- Vereda, A.; Sirvent, S.; Villalba, M.; Rodríguez, R.; Cuesta-Herranz, J.; Palomares, O. Improvement of mustard (Sinapis alba) allergy diagnosis and management by linking clinical features and component-resolved approaches. J. Allergy Clin. Immunol. 2011, 127, 1304–1307. [Google Scholar] [CrossRef]
- Faber, M.A.; De Graag, M.; Van Der Heijden, C.; Sabato, V.; Hagendorens, M.M.; Bridts, C.H.; De Clerck, L.S.; Ebo, D.G. Cor a 14: Missing link in the molecular diagnosis of hazelnut allergy? Int. Arch. Allergy Immunol. 2014, 164, 200–206. [Google Scholar] [CrossRef]
- Martín-Pedraza, L.; González, M.; Gómez, F.; Blanca-López, N.; Garrido-Arandia, M.; Rodríguez, R.; Torres, M.J.; Blanca, M.; Villalba, M.; Mayorga, C. Two nonspecific lipid transfer proteins (nsLTPs) from tomato seeds are associated to severe symptoms of tomato-allergic patients. Mol. Nutr. Food Res. 2016, 60, 1172–1182. [Google Scholar] [CrossRef]
- Bueno, C.; Martín-Pedraza, L.; Cuesta-Herranz, J.; Villalba, M. Is the Cross-Reactivity of Sin a 1, 2S Albumin from Mustard Seeds, Exclusively Restricted to Brassicaceae Members? JSM Allergy Asthma 2016, 1, 1001. [Google Scholar]
- Bueno-Díaz, C.; Martín-Pedraza, L.; Benedé, S.; Haroun-Díaz, E.; de las Heras, M.; Batanero, E.; Cuesta-Herranz, J.; Villalba, M. Seed storage 2S albumins are predictive indicators of exclusive Anacardiaceae cross-reactivity. Clin. Exp. Allergy 2019, 49, 545–549. [Google Scholar] [CrossRef] [PubMed]
- Bueno-Díaz, C.; Martín-Pedraza, L.; León, L.; Haroun-Díaz, E.; Pastor-Vargas, C.; Muñoz-García, E.; de las Heras, M.; Batanero, E.; Cuesta-Herranz, J.; Villalba, M.; et al. 2S albumins and 11S globulins, two storage proteins involved in pumpkin seeds allergy. Allergy 2021, 76, 383–386. [Google Scholar] [CrossRef] [PubMed]
- Cabanillas, B.; Crespo, J.F.; Maleki, S.J.; Rodriguez, J.; Novak, N. Pin p 1 is a major allergen in pine nut and the first food allergen described in the plant group of gymnosperms. Food Chem. 2016, 210, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, K.; Schweimer, K.; Reese, G.; Randow, S.; Suhr, M.; Becker, W.M.; Vieths, S.; Rösch, P. Structure and stability of 2S albumin-type peanut allergens: Implications for the severity of peanut allergic reactions. Biochem. J. 2006, 395, 463–472. [Google Scholar] [CrossRef] [PubMed]
- Starkl, P.; Krishnamurthy, D.; Szalai, K.; Felix, F.; Oberthuer, D.; Sampson, H.A.; Swoboda, I.; Betzel, C. Heating affects structure, enterocyte adsorption and signaling, as well as allergenicity of peanut allergen Ara h 2. Open Allergy J. 2011, 4, 24–34. [Google Scholar] [CrossRef] [PubMed]
- Dall’Antonia, F.; Pavkov-Keller, T.; Zangger, K.; Keller, W. Structure of allergens and structure based epitope predictions. Methods 2014, 66, 3–21. [Google Scholar] [CrossRef]
- Brosseron, L.; Lopes, J.; Mesquita, M.; Franco-Leandro, P.; Sousa, M.J.; Ferreira, A.R.; Lopes, I. Sensitization to Ana o 3 in tree nut and peanut allergy. Allergy Eur. J. Allergy Clin. Immunol. 2019, 74, 854–915. [Google Scholar] [CrossRef]
- Puumalainen, T.J.; Poikonen, S.; Kotovuori, A.; Vaali, K.; Kalkkinen, N.; Reunala, T.; Turjanmaa, K.; Palosuo, T. Napins, 2S albumins, are major allergens in oilseed rape and turnip rape. J. Allergy Clin. Immunol. 2006, 117, 426–432. [Google Scholar] [CrossRef]
- Asero, R.; Mistrello, G.; Roncarolo, D.; Amato, S. Allergenic similarities of 2S albumins. Allergy 2002, 57, 61–62. [Google Scholar] [CrossRef] [PubMed]
- Masilamani, M.; Commins, S.; Shreffler, W. Determinants of Food Allergy. Immunol. Allergy Clin. N. Am. 2012, 32, 11–33. [Google Scholar] [CrossRef]
- Breiteneder, H.; Radauer, C. A classification of plant food allergens. J. Allergy Clin. Immunol. 2004, 113, 821–830. [Google Scholar] [CrossRef]
- Breiteneder, H.; Mills, E.N.C. Molecular properties of food allergens. J. Allergy Clin. Immunol. 2005, 115, 14–23. [Google Scholar] [CrossRef]
- Starkl, P.; Felix, F.; Krishnamurthy, D.; Stremnitzer, C.; Roth-Walter, F.; Prickett, S.R.; Voskamp, A.L.; Willensdorfer, A.; Szalai, K.; Weichselbaumer, M.; et al. An unfolded variant of the major peanut allergen Ara h 2 with decreased anaphylactic potential. Clin. Exp. Allergy 2012, 42, 1801–1812. [Google Scholar] [CrossRef]
- Masthoff, L.J.; Hoff, R.; Verhoeckx, K.C.M.; Van Os-Medendorp, H.; Michelsen-Huisman, A.; Baumert, J.L.; Pasmans, S.G.; Meijer, Y.; Knulst, A.C. A systematic review of the effect of thermal processing on the allergenicity of tree nuts. Allergy Eur. J. Allergy Clin. Immunol. 2013, 68, 983–993. [Google Scholar] [CrossRef]
- Maleki, S.J.; Chung, S.Y.; Champagne, E.T.; Raufman, J.P. The effects of roasting on the allergenic properties of peanut proteins. J. Allergy Clin. Immunol. 2000, 106, 763–768. [Google Scholar] [CrossRef]
- Vissers, Y.M.; Blanc, F.; Skov, P.S.; Johnson, P.E.; Rigby, N.M.; Przybylski-Nicaise, L.; Bernard, H.; Wal, J.M.; Ballmer-Weber, B.; Zuidmeer-Jongejan, L.; et al. Effect of heating and glycation on the allergenicity of 2S albumins (Ara h 2/6) from peanut. PLoS ONE 2011, 6, e23998. [Google Scholar] [CrossRef] [PubMed]
- Gruber, P.; Becker, W.M.; Hofmann, T. Influence of the maillard reaction on the allergenicity of rAra h 2, a recombinant major allergen from peanut (Arachis hypogaea), its major epitopes, and peanut agglutinin. J. Agric. Food Chem. 2005, 53, 2289–2296. [Google Scholar] [CrossRef] [PubMed]
- Martín-Pedraza, L.; Wangorsch, A.; Bueno-Diaz, C.; de las Heras, M.; Scheurer, S.; Cuesta-Herranz, J.; Villalba, M. 2S albumins and nsLTP are involved in anaphylaxis to pizza sauce: IgE recognition before and after allergen processing. Food Chem. 2020, 321, 126679. [Google Scholar] [CrossRef] [PubMed]
- Dreskin, S.C.; Koppelman, S.J.; Andorf, S.; Nadeau, K.C.; Kalra, A.; Braun, W.; Negi, S.S.; Chen, X.; Schein, C.H. The importance of the 2S albumins for allergenicity and cross-reactivity of peanuts, tree nuts, and sesame seeds. J. Allergy Clin. Immunol. 2021, 147, 1154–1163. [Google Scholar] [CrossRef]
- Pantoja-Uceda, D.; Bruix, M.; Giménez-Gallego, G.; Rico, M.; Santoro, J. Solution Structure of RicC3, a 2S Albumin Storage Protein from Ricinus communis. Biochemistry 2003, 42, 13839–13847. [Google Scholar] [CrossRef] [PubMed]
- Menéndez-Arias, L.; Domínguez, J.; Moneo, I.; Rodríguez, R. Epitope mapping of the major allergen from yellow mustard seeds, Sin a I. Mol. Immunol. 1990, 27, 143–150. [Google Scholar] [CrossRef]
- Monsalve, R.I.; Villalba, M.; Rodríguez, R.; Monsalve, R.I. Allergy to Mustard Seeds: The Importance of 2S Albumins as Food Allergens. Internet Symp. Food Allerg. 2001, 3, 57–69. [Google Scholar]
- Monsalve, R.I.; Gonzalez de la Pena, M.A.; Menendez-Arias, L.; Lopez-Otin, C.; Villalba, M.; Rodriguez, R. Characterization of a new oriental-mustard (Brassica juncea) allergen, Bra j IE: Detection of an allergenic epitope. Biochem. J. 1993, 293, 625–632. [Google Scholar] [CrossRef]
- Maleki, S.J.; Teuber, S.S.; Cheng, H.; Chen, D.; Comstock, S.S.; Ruan, S.; Schein, C.H. Computationally predicted IgE epitopes of walnut allergens contribute to cross-reactivity with peanuts. Allergy Eur. J. Allergy Clin. Immunol. 2011, 66, 1522–1529. [Google Scholar] [CrossRef]
- Salminen, T.A.; Blomqvist, K.; Edqvist, J. Lipid transfer proteins: Classification, nomenclature, structure, and function. Planta 2016, 244, 971–997. [Google Scholar] [CrossRef] [PubMed]
- Berecz, B.; Clare Mills, E.N.; Parádi, I.; Láng, F.; Tamás, L.; Shewry, P.R.; MacKie, A.R. Stability of sunflower 2S albumins and LTP to physiologically relevant in vitro gastrointestinal digestion. Food Chem. 2013, 138, 2374–2381. [Google Scholar] [CrossRef] [PubMed]
- Oñaderra, M.; Monsalve, R.I.; Mancheño, J.M.; Villalba, M.; Del Pozo, A.M.; Gavilanes, J.G.; Rodriguez, R. Food mustard allergen interaction with phospholipid vesicles. Eur. J. Biochem. 1994, 225, 609–615. [Google Scholar] [CrossRef]
- Pantoja-Uceda, D.; Palomares, O.; Bruix, M.; Villalba, M.; Rodríguez, R.; Rico, M.; Santoro, J. Solution structure and stability against digestion of rproBnIb, a recombinant 2S albumin from rapeseed: Relationship to its allergenic properties. Biochemistry 2004, 43, 16036–16045. [Google Scholar] [CrossRef]
- Mirotti, L.; Florsheim, E.; Rundqvist, L.; Larsson, G.; Spinozzi, F.; Leite-De-Moraes, M.; Russo, M.; Alcocer, M. Lipids are required for the development of Brazil nut allergy: The role of mouse and human iNKT cells. Allergy Eur. J. Allergy Clin. Immunol. 2013, 68, 74–83. [Google Scholar] [CrossRef]
- Dearman, R.J.; Alcocer, M.J.C.; Kimber, I. Influence of plant lipids on immune responses in mice to the major Brazil nut allergen Ber e 1. Clin. Exp. Allergy 2007, 37, 582–591. [Google Scholar] [CrossRef] [PubMed]
- Taylor, S.L.; Baumert, J.L.; Kruizinga, A.G.; Remington, B.C.; Crevel, R.W.R.; Brooke-Taylor, S.; Allen, K.J.; Houben, G. Establishment of Reference Doses for residues of allergenic foods: Report of the VITAL Expert Panel. Food Chem. Toxicol. 2014, 63, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Crespo, J.F.; Bueno, C.; Villalba, M.; Monaci, L.; Cuadrado, C.; Novak, N.; Cabanillas, B. Epitope mapping of the major allergen 2S albumin from pine nut. Food Chem. 2021, 339, 127895. [Google Scholar] [CrossRef] [PubMed]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bueno-Díaz, C.; Martín-Pedraza, L.; Parrón, J.; Cuesta-Herranz, J.; Cabanillas, B.; Pastor-Vargas, C.; Batanero, E.; Villalba, M. Characterization of Relevant Biomarkers for the Diagnosis of Food Allergies: An Overview of the 2S Albumin Family. Foods 2021, 10, 1235. https://doi.org/10.3390/foods10061235
Bueno-Díaz C, Martín-Pedraza L, Parrón J, Cuesta-Herranz J, Cabanillas B, Pastor-Vargas C, Batanero E, Villalba M. Characterization of Relevant Biomarkers for the Diagnosis of Food Allergies: An Overview of the 2S Albumin Family. Foods. 2021; 10(6):1235. https://doi.org/10.3390/foods10061235
Chicago/Turabian StyleBueno-Díaz, Cristina, Laura Martín-Pedraza, Jorge Parrón, Javier Cuesta-Herranz, Beatriz Cabanillas, Carlos Pastor-Vargas, Eva Batanero, and Mayte Villalba. 2021. "Characterization of Relevant Biomarkers for the Diagnosis of Food Allergies: An Overview of the 2S Albumin Family" Foods 10, no. 6: 1235. https://doi.org/10.3390/foods10061235
APA StyleBueno-Díaz, C., Martín-Pedraza, L., Parrón, J., Cuesta-Herranz, J., Cabanillas, B., Pastor-Vargas, C., Batanero, E., & Villalba, M. (2021). Characterization of Relevant Biomarkers for the Diagnosis of Food Allergies: An Overview of the 2S Albumin Family. Foods, 10(6), 1235. https://doi.org/10.3390/foods10061235





