Application of White Mustard Bran and Flour on Bread as Natural Preservative Agents
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Fungal Strains and Culture Conditions
2.3. Extraction of Water-Soluble Mustard Components
2.4. Antifungal Activity of Freeze-Dried Mustard Extracts in Agar Diffusion Method
2.5. Antifungal Properties of MB over Time in Agar Diffusion Method
2.6. Determination of the Minimum Inhibitory Concentration (MIC) and the Minimum Fungicidal Concentration (MFC) of MB
2.7. Application of MB in Bread Formulation
2.8. Determination of the Fungal Population and Shelf Life
2.9. Statistical Analysis
3. Results and Discussion
3.1. In Vitro Antifungal Activity of MB and Antifungal Stability over Time
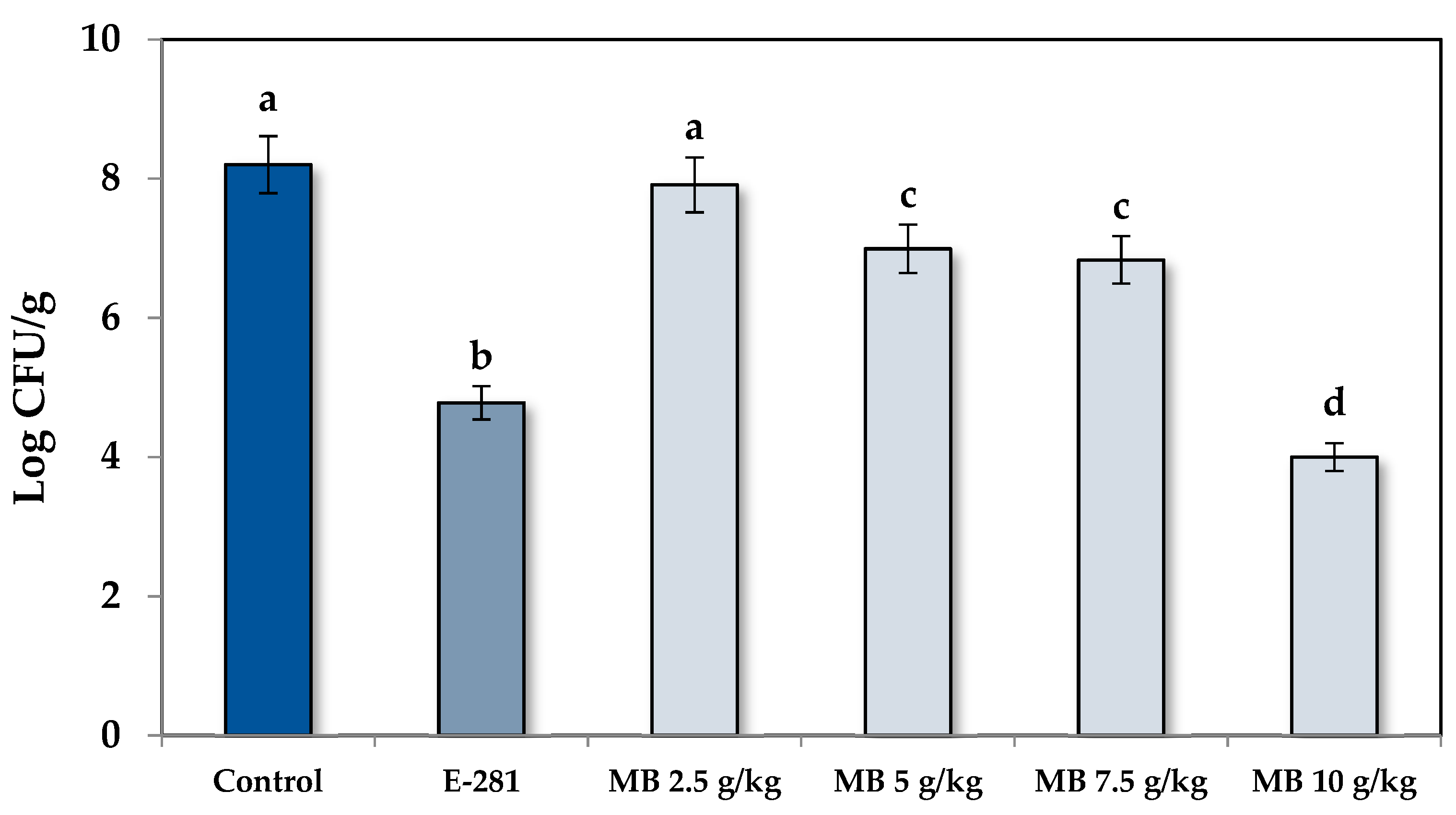
3.2. Antifungal Properties of White Mustard Bran on Bread Formulations
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Smith, J.P.; Daifas, D.P.; El-Khoury, W.; Koukoutsis, J.; El-Khoury, A. Shelf Life and Safety Concerns of Bakery Products—A Review. Crit. Rev. Food Sci. Nutr. 2004, 44, 19–55. [Google Scholar] [CrossRef]
- Axel, C.; Zannini, E.; Arendt, E.K. Mold spoilage of bread and its biopreservation: A review of current strategies for bread shelf life extension. Crit. Rev. Food Sci. Nutr. 2017, 57, 3528–3542. [Google Scholar] [CrossRef]
- Garcia, M.V.; Bernardi, A.O.; Copetti, M.V. The fungal problem in bread production: Insights of causes, consequences, and control methods. Curr. Opin. Food Sci. 2019, 29, 1–6. [Google Scholar] [CrossRef]
- Lee, H.J.; Ryu, D. Worldwide Occurrence of Mycotoxins in Cereals and Cereal-Derived Food Products: Public Health Perspectives of Their Co-occurrence. J. Agric. Food Chem. 2017, 65, 7034–7051. [Google Scholar] [CrossRef]
- Russo, P.; Fares, C.; Longo, A.; Spano, G.; Capozzi, V. Lactobacillus plantarum with Broad Antifungal Activity as a Protective Starter Culture for Bread Production. Foods 2017, 6, 110. [Google Scholar] [CrossRef] [PubMed]
- Vaclavikova, M.; Malachova, A.; Veprikova, Z.; Dzuman, Z.; Zachariasova, M.; Hajslova, J. “Emerging” mycotoxins in cereals processing chains: Changes of enniatins during beer and bread making. Food Chem. 2013, 136, 750–757. [Google Scholar] [CrossRef] [PubMed]
- Vidal, A.; Marín, S.; Ramos, A.J.; Cano-Sancho, G.; Sanchis, V. Determination of aflatoxins, deoxynivalenol, ochratoxin A and zearalenone in wheat and oat based bran supplements sold in the Spanish market. Food Chem. Toxicol. 2013, 53, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Škrbić, B.; Živančev, J.; Durišić-Mladenović, N.; Godula, M. Principal mycotoxins in wheat flour from the Serbian market: Levels and assessment of the exposure by wheat-based products. Food Control 2012, 25, 389–396. [Google Scholar] [CrossRef]
- Almeida-Ferreira, G.C.; Barbosa-Tessmann, I.P.; Sega, R.; Machinski Junior, M. Occurrence of zearalenone in wheat- and corn-based products commercialized in the state of paraná, Brazil. Braz. J. Microbiol. 2013, 44, 371–375. [Google Scholar] [CrossRef]
- Alexa, E.; Dehelean, C.A.; Poiana, M.A.; Radulov, I.; Cimpean, A.M.; Bordean, D.M.; Tulcan, C.; Pop, G. The occurrence of mycotoxins in wheat from western Romania and histopathological impact as effect of feed intake. Chem. Cent. J. 2013, 7, 1–11. [Google Scholar] [CrossRef]
- Iqbal, S.Z.; Asi, M.R.; Jinap, S.; Rashid, U. Detection of aflatoxins and zearalenone contamination in wheat derived products. Food Control 2014, 35, 223–226. [Google Scholar] [CrossRef]
- Thanushree, M.P.; Sailendri, D.; Yoha, K.S.; Moses, J.A.; Anandharamakrishnan, C. Mycotoxin contamination in food: An exposition on spices. Trends Food Sci. Technol. 2019, 93, 69–80. [Google Scholar] [CrossRef]
- Edite Bezerra da Rocha, M.; Freire, F.d.C.O.; Erlan Feitosa Maia, F.; Izabel Florindo Guedes, M.; Rondina, D. Mycotoxins and their effects on human and animal health. Food Control 2014, 36, 159–165. [Google Scholar] [CrossRef]
- de Melo Nazareth, T.; Luz, C.; Torrijos, R.; Quiles, J.M.; Luciano, F.B.; Mañes, J.; Meca, G. Potential application of lactic acid bacteria to reduce aflatoxin B1 and fumonisin B1 occurrence on corn kernels and corn ears. Toxins 2019, 12, 21. [Google Scholar] [CrossRef]
- Elsanhoty, R.M.; Ramadan, M.F.; El-Gohery, S.S.; Abol-Ela, M.F.; Azeke, M.A. Ability of selected microorganisms for removing aflatoxins invitro and fate of aflatoxins in contaminated wheat during baladi bread baking. Food Control 2013, 33, 287–292. [Google Scholar] [CrossRef]
- Gavahian, M.; Chu, Y.H.; Lorenzo, J.M.; Mousavi Khaneghah, A.; Barba, F.J. Essential oils as natural preservatives for bakery products: Understanding the mechanisms of action, recent findings, and applications. Crit. Rev. Food Sci. Nutr. 2020, 60, 310–321. [Google Scholar] [CrossRef]
- Le Lay, C.; Mounier, J.; Vasseur, V.; Weill, A.; Le Blay, G.; Barbier, G.; Coton, E. In vitro and in situ screening of lactic acid bacteria and propionibacteria antifungal activities against bakery product spoilage molds. Food Control 2016, 60, 247–255. [Google Scholar] [CrossRef]
- Grubb, C.D.; Abel, S. Glucosinolate metabolism and its control. Trends Plant Sci. 2006, 11, 89–100. [Google Scholar] [CrossRef]
- Nazareth, T.d.M.; Quiles, J.M.; Torrijos, R.; Luciano, F.B.; Mañes, J.; Meca, G. Antifungal and antimycotoxigenic activity of allyl isothiocyanate on barley under different storage conditions. LWT 2019, 112, 108237. [Google Scholar] [CrossRef]
- Bahmid, N.A.; Heising, J.; Fogliano, V.; Dekker, M. Packaging design using mustard seeds as a natural antimicrobial: A study on inhibition of Pseudomonas fragi in liquid medium. Foods 2020, 9, 789. [Google Scholar] [CrossRef]
- Vig, A.P.; Rampal, G.; Thind, T.S.; Arora, S. Bio-protective effects of glucosinolates—A review. LWT-Food Sci. Technol. 2009, 42, 1561–1572. [Google Scholar] [CrossRef]
- Quiles, J.M.; Nazareth, T.d.M.; Luz, C.; Luciano, F.B.; Mañes, J.; Meca, G. Development of an antifungal and antimycotoxigenic device containing allyl isothiocyanate for silo fumigation. Toxins 2019, 11, 137. [Google Scholar] [CrossRef] [PubMed]
- Ekanayake, A.; Kester, J.J.; Li, J.J.; Zehentbauer, G.N.; Bunke, P.R.; Zent, J.B. IsoGardTM: A natural antimicrobial agent derived from white mustard seed. Acta Hortic. 2006, 709, 101–108. [Google Scholar] [CrossRef]
- Donsì, F.; Velikov, K.P. Mechanical cell disruption of mustard bran suspensions for improved dispersion properties and protein release. Food Funct. 2020, 11, 6273–6284. [Google Scholar] [CrossRef]
- Espinel-Ingroff, A.; Cantón, E.; Pemán, J. Antifungal susceptibility testing of filamentous fungi. Curr. Fungal Infect. Rep. 2012, 6, 41–50. [Google Scholar] [CrossRef]
- Doulia, D.; Katsinis, G.; Mougin, B. Prolongation of the microbial shelf life of wrapped part baked baguettes. Int. J. Food Prop. 2000, 3, 447–457. [Google Scholar] [CrossRef]
- Pitt, J.I.; Hocking, A.D. Methods for Isolation, Enumeration and Identification. In Fungi and Food Spoilage; Springer US: Boston, MA, USA, 2009; pp. 19–52. ISBN 9780387922065. [Google Scholar]
- Okunade, O.A.; Ghawi, S.K.; Methven, L.; Niranjan, K. Thermal and pressure stability of myrosinase enzymes from black mustard (Brassica nigra L. W.D.J. Koch. var. nigra), brown mustard (Brassica juncea L. Czern. var. juncea) and yellow mustard (Sinapsis alba L. subsp. maire) seeds. Food Chem. 2015, 187, 485–490. [Google Scholar] [CrossRef] [PubMed]
- Van Eylen, D.; Indrawati; Hendrickx, M.; Van Loey, A. Temperature and pressure stability of mustard seed (Sinapis alba L.) myrosinase. Food Chem. 2006, 97, 263–271. [Google Scholar] [CrossRef]
- Tsao, R.; Yu, Q.; Friesen, I.; Potter, J.; Chiba, M. Factors affecting the dissolution and degradation of oriental mustard- derived sinigrin and allyl isothiocyanate in aqueous media. J. Agric. Food Chem. 2000, 48, 1898–1902. [Google Scholar] [CrossRef]
- Saladino, F.; Bordin, K.; Luciano, F.B.; Franzón, M.F.; Mañes, J.; Meca, G. Antimicrobial Activity of the Glucosinolates. In Glucosinolates; Springer: Cham, Switzerland, 2017; pp. 249–274. ISBN 9783319254616. [Google Scholar]
- Sotelo, T.; Lema, M.; Soengas, P.; Cartea, M.E.; Velasco, P. In vitro activity of Glucosinolates and their degradation products against Brassica-pathogenic bacteria and fungi. Appl. Environ. Microbiol. 2015, 81, 432–440. [Google Scholar] [CrossRef]
- Wang, T.; Li, Y.; Bi, Y.; Zhang, M.; Zhang, T.; Zheng, X.; Dong, Y.; Huang, Y. Benzyl isothiocyanate fumigation inhibits growth, membrane integrity and mycotoxin production in: Alternaria alternata. RSC Adv. 2020, 10, 1829–1837. [Google Scholar] [CrossRef]
- Azaiez, I.; Meca, G.; Manyes, L.; Fernández-Franzón, M. Antifungal activity of gaseous allyl, benzyl and phenyl isothiocyanate in vitro and their use for fumonisins reduction in bread. Food Control 2013, 32, 428–434. [Google Scholar] [CrossRef]
- Ribeiro-Santos, R.; Andrade, M.; Melo, N.R.d.; Sanches-Silva, A. Use of essential oils in active food packaging: Recent advances and future trends. Trends Food Sci. Technol. 2017, 61, 132–140. [Google Scholar] [CrossRef]
- Nielsen, P.V.; Rios, R. Inhibition of fungal growth on bread by volatile components from spices and herbs, and the possible application in active packaging, with special emphasis on mustard essential oil. Int. J. Food Microbiol. 2000, 60, 219–229. [Google Scholar] [CrossRef]
- Saladino, F.; Manyes, L.; Luciano, F.B.; Mañes, J.; Fernandez-Franzon, M.; Meca, G. Bioactive compounds from mustard flours for the control of patulin production in wheat tortillas. LWT-Food Sci. Technol. 2016, 66, 101–107. [Google Scholar] [CrossRef]
- Quiles, J.M.; Manyes, L.; Luciano, F.; Mañes, J.; Meca, G. Influence of the antimicrobial compound allyl isothiocyanate against the Aspergillus parasiticus growth and its aflatoxins production in pizza crust. Food Chem. Toxicol. 2015, 83, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Clemente, I.; Aznar, M.; Nerín, C. Synergistic properties of mustard and cinnamon essential oils for the inactivation of foodborne moulds in vitro and on Spanish bread. Int. J. Food Microbiol. 2019, 298, 44–50. [Google Scholar] [CrossRef]

| Fungi Strain | MF | MB |
|---|---|---|
| P. camemberti CECT 2267 | − | +++ |
| P. expansum CECT 2278 | − | +++ |
| P. roqueforti CECT 2905 | − | +++ |
| P. digitatum CECT 2954 | − | ++ |
| P. commune CECT 20767 | − | +++ |
| P. solitum CECT 20818 | − | ++ |
| P. verrucosum VTT D-01847 | − | +++ |
| A. flavus ITEM 8111 | − | + |
| A. parasiticus CECT 2681 | − | + |
| A. ochraceus CECT 2093 | − | + |
| A. lacticoffeatus CECT 20581 | − | + |
| A. steynii CECT 20510 | − | + |
| A. tubingensis CECT 20543 | − | + |
| A. tubingensis CECT 20544 | − | + |
| F. proliferatum ITEM 12072 | − | ++ |
| F. verticillioides ITEM 12052 | − | +++ |
| F. verticillioides ITEM 12044 | + | +++ |
| F. graminearum ITEM 126 | − | ++ |
| F. sporotrichioides ITEM 12168 | − | + |
| F. poae ITEM 9151 | + | ++ |
| Fungi Strain | 4 °C | 25 °C | 50 °C | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 24 h | 48 h | 72 h | 168 h | 24 h | 48 h | 72 h | 168 h | 24–168 h | |
| P. camemberti CECT 2267 | +++ | +++ | +++ | ++ | ++ | ++ | ++ | + | − |
| P. expansum CECT 2278 | +++ | +++ | +++ | ++ | ++ | ++ | ++ | + | − |
| P. roqueforti CECT 2905 | +++ | +++ | +++ | ++ | ++ | ++ | ++ | + | − |
| P. digitatum CECT 2954 | +++ | +++ | +++ | ++ | ++ | ++ | + | + | − |
| P. commune CECT 20767 | +++ | +++ | +++ | ++ | ++ | ++ | ++ | + | − |
| P. solitum CECT 20818 | +++ | +++ | +++ | ++ | ++ | ++ | ++ | + | − |
| P. verrucosum VTT D-01847 | +++ | +++ | +++ | ++ | ++ | ++ | ++ | ++ | − |
| A. flavus ITEM 8111 | + | + | + | − | + | + | − | − | − |
| A. parasiticus CECT 2681 | + | + | + | − | + | + | − | − | − |
| A. ochraceus CECT 2093 | + | + | + | − | + | + | − | − | − |
| A. lacticoffeatus CECT 20581 | + | + | + | − | + | + | − | − | − |
| A. steynii CECT 20510 | + | + | + | − | + | + | − | − | − |
| A. tubingensis CECT 20543 | + | + | + | − | + | + | − | − | − |
| A. tubingensis CECT 20544 | + | + | + | − | + | + | − | − | − |
| F. proliferatum ITEM 12072 | ++ | ++ | ++ | + | ++ | ++ | ++ | + | − |
| F. verticillioides ITEM 12052 | +++ | +++ | +++ | ++ | ++ | ++ | ++ | + | − |
| F. verticillioides ITEM 12044 | +++ | +++ | ++ | + | ++ | ++ | + | + | − |
| F. graminearum ITEM 126 | ++ | ++ | ++ | + | + | + | + | + | − |
| F. sporotrichioides ITEM 12168 | + | + | + | + | + | + | + | + | − |
| F. poae ITEM 9151 | ++ | ++ | ++ | + | ++ | ++ | ++ | + | − |
| Fungi Strain | MIC | MFC |
|---|---|---|
| P. camemberti CECT 2267 | 0.3 | 0.6 |
| P. expansum CECT 2278 | 0.6 | 1.2 |
| P. roqueforti CECT 2905 | 0.3 | 0.6 |
| P. digitatum CECT 2954 | 1.2 | 2.3 |
| P. commune CECT 20767 | 0.6 | 1.2 |
| P. solitum CECT 20818 | 1.2 | 4.7 |
| P. verrucosum VTT D-01847 | 0.6 | 1.2 |
| A. flavus ITEM 8111 | 1.2 | 4.7 |
| A. parasiticus CECT 2681 | 1.2 | 4.7 |
| A. ochraceus CECT 2093 | 1.2 | 4.7 |
| A. lacticoffeatus CECT 20581 | 1.2 | 2.3 |
| A. steynii CECT 20510 | 2.3 | 9.4 |
| A. tubingensis CECT 20543 | 1.2 | 4.7 |
| A. tubingensis CECT 20544 | 1.2 | 18.8 |
| F. proliferatum ITEM 12072 | 0.6 | 9.4 |
| F. verticillioides ITEM 12052 | 0.6 | 2.3 |
| F. verticillioides ITEM 12044 | 1.2 | 2.3 |
| F. graminearum ITEM 126 | 4.7 | 9.4 |
| F. sporotrichioides ITEM 12168 | 4.7 | 9.4 |
| F. poae ITEM 9151 | 2.3 | 4.7 |
| Treatment | Days | ||||||
|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | |
| Control | − | − | − | + | ++ | + + | ++ |
| Commercial | − | − | − | − | − | − | + |
| MB 2.5 g/kg | − | − | − | + | + + | ++ | ++ |
| MB 5 g/kg | − | − | − | − | − | + | ++ |
| MB 7.5 g/kg | − | − | − | − | − | + | ++ |
| MB 10 g/kg | − | − | − | − | − | − | + |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Torrijos, R.; Nazareth, T.d.M.; Quiles, J.M.; Mañes, J.; Meca, G. Application of White Mustard Bran and Flour on Bread as Natural Preservative Agents. Foods 2021, 10, 431. https://doi.org/10.3390/foods10020431
Torrijos R, Nazareth TdM, Quiles JM, Mañes J, Meca G. Application of White Mustard Bran and Flour on Bread as Natural Preservative Agents. Foods. 2021; 10(2):431. https://doi.org/10.3390/foods10020431
Chicago/Turabian StyleTorrijos, Raquel, Tiago de Melo Nazareth, Juan Manuel Quiles, Jordi Mañes, and Giuseppe Meca. 2021. "Application of White Mustard Bran and Flour on Bread as Natural Preservative Agents" Foods 10, no. 2: 431. https://doi.org/10.3390/foods10020431
APA StyleTorrijos, R., Nazareth, T. d. M., Quiles, J. M., Mañes, J., & Meca, G. (2021). Application of White Mustard Bran and Flour on Bread as Natural Preservative Agents. Foods, 10(2), 431. https://doi.org/10.3390/foods10020431