Dairy Lactic Acid Bacteria and Their Potential Function in Dietetics: The Food–Gut-Health Axis
Abstract
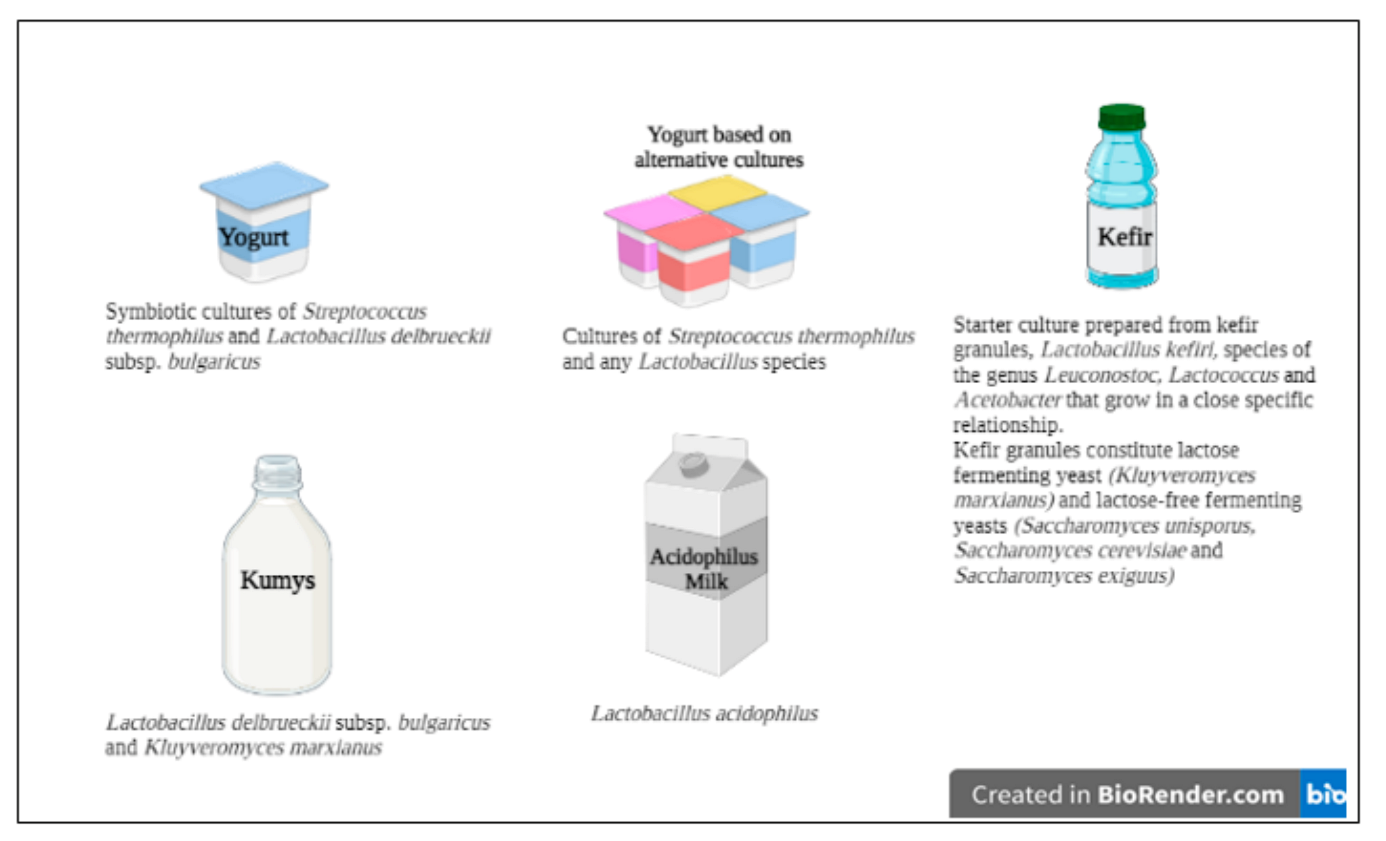
:1. Introduction
2. Definitions and Some Characteristics of LABs
3. Importance of LABs in Dairy Foods in Terms of Health
4. Involvement of Dairy LABs in the Modulation of Non-Communicable Diseases (NCDs)
4.1. Obesity
4.2. Cardiovascular Diseases
4.3. Diabetes Mellitus
4.4. Cancer
4.5. Inflammatory Bowel Diseases (IBD)
5. Conclusions
Author Contributions
Funding
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- García-Burgos, M.; Moreno-Fernández, J.; Alférez, M.J.M.; Díaz-Castro, J.; López-Aliaga, I. New perspectives in fermented dairy products and their health relevance. J. Funct. Foods 2020, 72, 104059. [Google Scholar] [CrossRef]
- Rezac, S.; Kok, C.R.; Heermann, M.; Hutkins, R. Fermented foods as a dietary source of live organisms. Front. Microbiol. 2018, 9, 1785. [Google Scholar] [CrossRef] [PubMed]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. The international scientific association for probiotics and prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zoumpopoulou, G.; Pot, B.; Tsakalidou, E.; Papadimitriou, K. Dairy probiotics: Beyond the role of promoting gut and immune health. Int. Dairy J. 2017, 67, 46–60. [Google Scholar] [CrossRef]
- Hardy, H.; Harris, J.; Lyon, E.; Beal, J.; Foey, A. Probiotics, prebiotics and immunomodulation of gut mucosal defences: Homeostasis and immunopathology. Nutrients 2013, 5, 1869–1912. [Google Scholar] [CrossRef]
- Rooks, M.G.; Garrett, W.S. Gut microbiota, metabolites and host immunity. Nat. Rev. Immunol. 2016, 16, 341–352. [Google Scholar] [CrossRef]
- Pessione, E.; Cirrincione, S. Bioactive molecules released in food by lactic acid bacteria: Encrypted peptides and biogenic amines. Front. Microbiol. 2016, 7, 876. [Google Scholar] [CrossRef] [Green Version]
- Bourrie, B.C.T.; Willing, B.P.; Cotter, P.D. The microbiota and health promoting characteristics of the fermented beverage kefir. Front. Microbiol. 2016, 7, 647. [Google Scholar] [CrossRef] [Green Version]
- Savaiano, D.A.; Hutkins, R.W. Yogurt, cultured fermented milk, and health: A systematic review. Nutr. Rev. 2021, 79, 599–614. [Google Scholar] [CrossRef]
- Eor, J.Y.; Tan, P.L.; Son, Y.J.; Lee, C.S.; Kim, S.H. Milk products fermented by lactobacillus strains modulate the gut–bone axis in an ovariectomised murine model. Int. J. Dairy Technol. 2020, 73, 743–756. [Google Scholar] [CrossRef]
- Carabotti, M.; Scirocco, A.; Maselli, M.A.; Severi, C. The gut-brain axis: Interactions between enteric microbiota, central and enteric nervous systems. Ann. Gastroenterol. 2015, 28, 203–209. [Google Scholar]
- Butler, M.I.; Bastiaanssen, T.F.S.; Long-Smith, C.; Berding, K.; Morkl, S.; Cusack, A.-M.; Strain, C.; Busca, K.; Porteous-Allen, P.; Claesson, M.J.; et al. Recipe for a healthy gut: Intake of unpasteurised milk is associated with increased lactobacillus abundance in the human gut microbiome. Nutrients 2020, 12, 1468. [Google Scholar] [CrossRef]
- Aslam, H.; Marx, W.; Rocks, T.; Loughman, A.; Chandrasekaran, V.; Ruusunen, A.; Dawson, S.L.; West, M.; Mullarkey, E.; Pasco, J.A.; et al. The effects of dairy and dairy derivatives on the gut microbiota: A systematic literature review. Gut Microbes 2020, 12, 1799533. [Google Scholar] [CrossRef]
- Guillemard, E.; Tondu, F.; Lacoin, F.; Schrezenmeir, J. Consumption of a fermented dairy product containing the probiotic Lactobacillus casei DN-114 001 reduces the duration of respiratory infections in the elderly in a randomised controlled trial. Br. J. Nutr. 2010, 103, 58–68. [Google Scholar] [CrossRef] [Green Version]
- Yu, A.O.; Leveau, J.H.J.; Marco, M.L. Abundance, diversity and plant-specific adaptations of plant-associated lactic acid bacteria. Environ. Microbiol. Rep. 2020, 12, 16–29. [Google Scholar] [CrossRef]
- He, L.; Li, J.; Sun, Z. More critical consideration on enhancing micronutrient bioavailability of phytate rich foods by phytase-producing lactic acid bacteria. Trends Food Sci. Technol. 2020, 102, 37–38. [Google Scholar] [CrossRef]
- Levit, R.; Savoy de Giori, G.; Moreno de LeBlanc, A.; LeBlanc, J.G. Recent update on lactic acid bacteria producing riboflavin and folates: Application for food fortification and treatment of intestinal inflammation. J. Appl. Microbiol. 2021, 130, 1412–1424. [Google Scholar] [CrossRef]
- Ferreira, C.L.L. Prebióticos e Probióticos: Atualização e Prospecção; Editora Rubio: Rio de Janeiro, Brazil, 2012; ISBN 8564956020. [Google Scholar]
- Börner, R.A.; Kandasamy, V.; Axelsen, A.M.; Nielsen, A.T.; Bosma, E.F. Genome editing of lactic acid bacteria: Opportunities for food, feed, pharma and biotech. FEMS Microbiol. Lett. 2019, 366, fny291. [Google Scholar] [CrossRef]
- Castillo Martinez, F.A.; Balciunas, E.M.; Salgado, J.M.; Domínguez González, J.M.; Converti, A.; de Souza Oliveira, R.P. Lactic acid properties, applications and production: A review. Trends Food Sci. Technol. 2013, 30, 70–83. [Google Scholar] [CrossRef]
- Pessione, E. Lactic acid bacteria contribution to gut microbiota complexity: Lights and shadows. Front. Cell. Infect. Microbiol. 2012, 2, 86. [Google Scholar] [CrossRef] [Green Version]
- Mulaw, G.; Sisay Tessema, T.; Muleta, D.; Tesfaye, A. In vitro evaluation of probiotic properties of lactic acid bacteria isolated from some traditionally fermented ethiopian food products. Int. J. Microbiol. 2019, 2019, 7179514. [Google Scholar] [CrossRef] [Green Version]
- Marcial-Coba, M.S.; Knøchel, S.; Nielsen, D.S. Low-moisture food matrices as probiotic carriers. FEMS Microbiol. Lett. 2019, 366, fnz006. [Google Scholar] [CrossRef]
- Ershidat, O.T.M.; Mazahreh, A.S. Probiotics bacteria in fermented dairy products. Pakistan J. Nutr. 2009, 8, 1107–1113. [Google Scholar] [CrossRef] [Green Version]
- Gao, J.; Li, X.; Zhang, G.; Sadiq, F.A.; Simal-Gandara, J.; Xiao, J.; Sang, Y. Probiotics in the dairy industry—Advances and opportunities. Compr. Rev. Food Sci. Food Saf. 2021, 20, 3937–3982. [Google Scholar] [CrossRef]
- Oh, S. Probiotics in Dairy Products. In Beneficial Microorganisms in Food and Nutraceuticals; Liong, M.-T., Ed.; Microbiology Monographs; Springer International Publishing: Cham, Switzerland, 2015; Volume 27, pp. 203–219. ISBN 978-3-319-23176-1. [Google Scholar]
- Reuben, R.C.; Roy, P.C.; Sarkar, S.L.; Alam, A.S.M.R.U.; Jahid, I.K.; Rubayet Ul Alam, A.S.M.; Jahid, I.K. Characterization and evaluation of lactic acid bacteria from indigenous raw milk for potential probiotic properties. J. Dairy Sci. 2020, 103, 1223–1237. [Google Scholar] [CrossRef]
- Meybodi, N.M.; Mortazavian, A.M.; Arab, M.; Nematollahi, A. Probiotic viability in yoghurt: A review of influential factors. Int. Dairy J. 2020, 109, 104793. [Google Scholar] [CrossRef]
- Terpou, A.; Papadaki, A.; Bosnea, L.; Kanellaki, M.; Kopsahelis, N. Novel frozen yogurt production fortified with sea buckthorn berries and probiotics. LWT 2019, 105, 242–249. [Google Scholar] [CrossRef]
- Ghosh, T.; Beniwal, A.; Semwal, A.; Navani, N.K. Mechanistic insights into probiotic properties of lactic acid bacteria associated with ethnic fermented dairy products. Front. Microbiol. 2019, 10, 502. [Google Scholar] [CrossRef] [Green Version]
- Azaïs-Braesco, V.; Bresson, J.L.; Guarner, F.; Corthier, G. Not all lactic acid bacteria are probiotics, …but some are. Br. J. Nutr. 2010, 103, 1079–1081. [Google Scholar] [CrossRef] [Green Version]
- Davoren, M.J.; Liu, J.; Castellanos, J.; Rodríguez-Malavé, N.I.; Schiestl, R.H. A novel probiotic, lactobacillus johnsonii 456, resists acid and can persist in the human gut beyond the initial ingestion period. Gut Microbes 2019, 10, 458–480. [Google Scholar] [CrossRef]
- Milk and Milk Products. Joint FAO/WHO Codex Alimentarius Commission, 2nd ed.; Food & Agriculture Organization: Rome, Italy, 2011. [Google Scholar]
- Campana, R.; van Hemert, S.; Baffone, W. Strain-specific probiotic properties of lactic acid bacteria and their interference with human intestinal pathogens invasion. Gut Pathog. 2017, 9, 12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharifi Yazdi, M.K.; Davoodabadi, A.; Khesht Zarin, H.R.; Tajabadi Ebrahimi, M.; Soltan Dallal, M.M. Characterisation and probiotic potential of lactic acid bacteria isolated from Iranian traditional yogurts. Ital. J. Anim. Sci. 2017, 16, 185–188. [Google Scholar] [CrossRef] [Green Version]
- Uymaz Tezel, B. Preliminary in vitro evaluation of the probiotic potential of the bacteriocinogenic strain Enterococcus lactis PMD74 isolated from ezine cheese. J. Food Qual. 2019, 2019, 4693513. [Google Scholar] [CrossRef] [Green Version]
- Bhagat, D.; Raina, N.; Kumar, A.; Katoch, M.; Khajuria, Y.; Slathia, P.S.; Sharma, P. Probiotic properties of a phytase producing pediococcus acidilactici strain SMVDUDB2 isolated from traditional fermented cheese product, kalarei. Sci. Rep. 2020, 10, 1926. [Google Scholar] [CrossRef] [PubMed]
- Rong, J.; Zheng, H.; Liu, M.; Hu, X.; Wang, T.; Zhang, X.; Jin, F.; Wang, L. Probiotic and anti-inflammatory attributes of an isolate Lactobacillus helveticus NS8 from Mongolian fermented koumiss. BMC Microbiol. 2015, 15, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Tulumoğlu, Ş.; Kaya, H.İ.; Şimşek, Ö. Probiotic characteristics of Lactobacillus fermentum strains isolated from tulum cheese. Anaerobe 2014, 30, 120–125. [Google Scholar] [CrossRef]
- Soni, R.; Jain, N.K.; Shah, V.; Soni, J.; Suthar, D.; Gohel, P. Development of probiotic yogurt: Effect of strain combination on nutritional, rheological, organoleptic and probiotic properties. J. Food Sci. Technol. 2020, 57, 2038–2050. [Google Scholar] [CrossRef]
- Popović, N.; Brdarić, E.; Đokić, J.; Dinić, M.; Veljović, K.; Golić, N.; Terzić-Vidojević, A. Yogurt produced by novel natural starter cultures improves gut epithelial barrier in vitro. Microorganisms 2020, 8, 1586. [Google Scholar] [CrossRef]
- Karami, S.; Roayaei, M.; Hamzavi, H.; Bahmani, M.; Hassanzad-Azar, H.; Leila, M.; Rafieian-Kopaei, M. Isolation and identification of probiotic Lactobacillus from local dairy and evaluating their antagonistic effect on pathogens. Int. J. Pharm. Investig. 2017, 7, 137. [Google Scholar] [CrossRef]
- Kumar, A.; Kumar, D. Characterization of Lactobacillus isolated from dairy samples for probiotic properties. Anaerobe 2015, 33, 117–123. [Google Scholar] [CrossRef]
- Zoumpopoulou, G.; Tzouvanou, A.; Mavrogonatou, E.; Alexandraki, V.; Georgalaki, M.; Anastasiou, R.; Papadelli, M.; Manolopoulou, E.; Kazou, M.; Kletsas, D.; et al. Probiotic features of lactic acid bacteria isolated from a diverse pool of traditional greek dairy products regarding specific strain-host interactions. Probiotics Antimicrob. Proteins 2018, 10, 313–322. [Google Scholar] [CrossRef]
- Xing, Z.; Tang, W.; Geng, W.; Zheng, Y.; Wang, Y. In vitro and in vivo evaluation of the probiotic attributes of Lactobacillus kefiranofaciens XL10 isolated from Tibetan kefir grain. Appl. Microbiol. Biotechnol. 2017, 101, 2467–2477. [Google Scholar] [CrossRef]
- Ochoa-Repáraz, J.; Kasper, L.H. The Second Brain: Is the gut microbiota a link between obesity and central nervous system disorders? Curr. Obes. Rep. 2016, 5, 51–64. [Google Scholar] [CrossRef] [Green Version]
- Appleton, J. The gut-brain axis: Influence of microbiota on mood and mental health. Integr. Med. 2018, 17, 28–32. [Google Scholar]
- De Filippis, F.; Pasolli, E.; Ercolini, D. The food-gut axis: Lactic acid bacteria and their link to food, the gut microbiome and human health. FEMS Microbiol. Rev. 2020, 44, 454–489. [Google Scholar] [CrossRef]
- Karakan, T.; Ozkul, C.; Küpeli Akkol, E.; Bilici, S.; Sobarzo-Sánchez, E.; Capasso, R. Gut-brain-microbiota axis: Antibiotics and functional gastrointestinal disorders. Nutrients 2021, 13, 389. [Google Scholar] [CrossRef]
- Asano, Y.; Hiramoto, T.; Nishino, R.; Aiba, Y.; Kimura, T.; Yoshihara, K.; Koga, Y.; Sudo, N. Critical role of gut microbiota in the production of biologically active, free catecholamines in the gut lumen of mice. Am. J. Physiol. Liver Physiol. 2012, 303, G1288–G1295. [Google Scholar] [CrossRef] [Green Version]
- Iyer, L.M.; Aravind, L.; Coon, S.L.; Klein, D.C.; Koonin, E. V Evolution of cell–cell signaling in animals: Did late horizontal gene transfer from bacteria have a role? Trends Genet. 2004, 20, 292–299. [Google Scholar] [CrossRef]
- Morais, L.H.; Schreiber, H.L.; Mazmanian, S.K. The gut microbiota–brain axis in behaviour and brain disorders. Nat. Rev. Microbiol. 2021, 19, 241–255. [Google Scholar] [CrossRef]
- Cryan, J.F.; O’Riordan, K.J.; Cowan, C.S.M.; Sandhu, K.V.; Bastiaanssen, T.F.S.; Boehme, M.; Codagnone, M.G.; Cussotto, S.; Fulling, C.; Golubeva, A.V.; et al. The microbiota-gut-brain axis. Physiol. Rev. 2019, 99, 1877–2013. [Google Scholar] [CrossRef]
- Sobko, T.; Huang, L.; Midtvedt, T.; Norin, E.; Gustafsson, L.E.; Norman, M.; Jansson, E.Å.; Lundberg, J.O. Generation of NO by probiotic bacteria in the gastrointestinal tract. Free Radic. Biol. Med. 2006, 41, 985–991. [Google Scholar] [CrossRef]
- De Angelis, M.; Garruti, G.; Minervini, F.; Bonfrate, L.; Portincasa, P.; Gobbetti, M. The food-gut human axis: The effects of diet on gut microbiota and metabolome. Curr. Med. Chem. 2019, 26, 3567–3583. [Google Scholar] [CrossRef]
- Chung, Y.-C.; Jin, H.-M.; Cui, Y.; Kim, D.S.; Jung, J.M.; Park, J.-I.; Jung, E.-S.; Choi, E.-K.; Chae, S.-W. Fermented milk of Lactobacillus helveticus IDCC3801 improves cognitive functioning during cognitive fatigue tests in healthy older adults. J. Funct. Foods 2014, 10, 465–474. [Google Scholar] [CrossRef]
- Marcos, A.; Wärnberg, J.; Nova, E.; Gómez, S.; Alvarez, A.; Alvarez, R.; Mateos, J.A.; Cobo, J.M. The effect of milk fermented by yogurt cultures plus Lactobacillus casei DN-114001 on the immune response of subjects under academic examination stress. Eur. J. Nutr. 2004, 43, 381–389. [Google Scholar] [CrossRef]
- Perazza, L.R.; Daniel, N.; Dubois, M.; Pilon, G.; Varin, T.V.; Blais, M.; Martinez Gonzales, J.L.; Bouchard, M.; Asselin, C.; Lessard, M.; et al. Distinct effects of milk-derived and fermented dairy protein on gut microbiota and cardiometabolic markers in diet-induced obese mice. J. Nutr. 2020, 150, 2673–2686. [Google Scholar] [CrossRef]
- Buziau, A.M.; Soedamah-Muthu, S.S.; Geleijnse, J.M.; Mishra, G.D. Total fermented dairy food intake is inversely associated with cardiovascular disease risk in women. J. Nutr. 2019, 149, 1797–1804. [Google Scholar] [CrossRef] [Green Version]
- Godos, J.; Tieri, M.; Ghelfi, F.; Titta, L.; Marventano, S.; Lafranconi, A.; Gambera, A.; Alonzo, E.; Sciacca, S.; Buscemi, S.; et al. Dairy foods and health: An umbrella review of observational studies. Int. J. Food Sci. Nutr. 2020, 71, 138–151. [Google Scholar] [CrossRef]
- Jakobsen, M.U.; Trolle, E.; Outzen, M.; Mejborn, H.; Grønberg, M.G.; Lyndgaard, C.B.; Stockmarr, A.; Venø, S.K.; Bysted, A. Intake of dairy products and associations with major atherosclerotic cardiovascular diseases: A systematic review and meta-analysis of cohort studies. Sci. Rep. 2021, 11, 1303. [Google Scholar] [CrossRef]
- Zhang, K.; Chen, X.; Zhang, L.; Deng, Z. Fermented dairy foods intake and risk of cardiovascular diseases: A meta-analysis of cohort studies. Crit. Rev. Food Sci. Nutr. 2020, 60, 1189–1194. [Google Scholar] [CrossRef]
- Rodríguez-Sánchez, S.; Ramos, I.M.; Seseña, S.; Poveda, J.M.; Palop, M.L. Potential of Lactobacillus strains for health-promotion and flavouring of fermented dairy foods. LWT 2021, 143, 111102. [Google Scholar] [CrossRef]
- Ebringer, L.; Ferenčík, M.; Krajčovič, J. Beneficial health effects of milk and fermented dairy products—Review. Folia Microbiol. 2008, 53, 378–394. [Google Scholar] [CrossRef] [PubMed]
- Linares, D.M.; Gómez, C.; Renes, E.; Fresno, J.M.; Tornadijo, M.E.; Ross, R.P.; Stanton, C. Lactic acid bacteria and bifidobacteria with potential to design natural biofunctional health-promoting dairy foods. Front. Microbiol. 2017, 8, 846. [Google Scholar] [CrossRef] [PubMed]
- Mathur, H.; Beresford, T.P.; Cotter, P.D. Health benefits of lactic acid bacteria (LAB) fermentates. Nutrients 2020, 12, 1679. [Google Scholar] [CrossRef] [PubMed]
- Nampoothiri, K.M.; Beena, D.J.; Vasanthakumari, D.S.; Ismail, B. Health benefits of exopolysaccharides in fermented foods. In Fermented Foods in Health and Disease Prevention; Elsevier: Amsterdam, The Netherlands, 2017; pp. 49–62. [Google Scholar]
- Nongonierma, A.B.; FitzGerald, R.J. The scientific evidence for the role of milk protein-derived bioactive peptides in humans: A Review. J. Funct. Foods 2015, 17, 640–656. [Google Scholar] [CrossRef] [Green Version]
- Evivie, S.E.; Huo, G.-C.; Igene, J.O.; Bian, X. Some current applications, limitations and future perspectives of lactic acid bacteria as probiotics. Food Nutr. Res. 2017, 61, 1318034. [Google Scholar] [CrossRef] [Green Version]
- World Health Organization Obesity and Overweight Fact Sheet. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 18 October 2021).
- Sun, N.-N.; Wu, T.-Y.; Chau, C.-F. Natural dietary and herbal products in anti-obesity treatment. Molecules 2016, 21, 1351. [Google Scholar] [CrossRef]
- Hong, S.-M.; Chung, E.-C.; Kim, C.-H. Anti-obesity effect of fermented whey beverage using lactic acid bacteria in diet-induced obese rats. Korean J. Food Sci. Anim. Resour. 2015, 35, 653–659. [Google Scholar] [CrossRef] [Green Version]
- Chaudhari, D.D.; Singh, R.; Mallappa, R.H.; Rokana, N.; Kaushik, J.K.; Bajaj, R.; Batish, V.K.; Grover, S. Evaluation of casein & whey protein hydrolysates as well as milk fermentates from Lactobacillus helveticus for expression of gut hormones. Indian J. Med. Res. 2017, 146, 409–419. [Google Scholar] [CrossRef]
- Fernandez, M.A.; Panahi, S.; Daniel, N.; Tremblay, A.; Marette, A. Yogurt and cardiometabolic diseases: A critical review of potential mechanisms. Adv. Nutr. 2017, 8, 812–829. [Google Scholar] [CrossRef] [Green Version]
- Pothuraju, R.; Sharma, R.K.; Kavadi, P.K.; Chagalamarri, J.; Jangra, S.; Bhakri, G.; De, S. Anti-obesity effect of milk fermented by Lactobacillus plantarum NCDC 625 alone and in combination with herbs on high fat diet fed C57BL/6J mice. Benef. Microbes 2016, 7, 375–385. [Google Scholar] [CrossRef]
- Stojanov, S.; Berlec, A.; Štrukelj, B. The influence of probiotics on the firmicutes/bacteroidetes ratio in the treatment of obesity and inflammatory bowel disease. Microorganisms 2020, 8, 1715. [Google Scholar] [CrossRef]
- Kadooka, Y.; Sato, M.; Ogawa, A.; Miyoshi, M.; Uenishi, H.; Ogawa, H.; Ikuyama, K.; Kagoshima, M.; Tsuchida, T. Effect of Lactobacillus gasseri SBT2055 in fermented milk on abdominal adiposity in adults in a randomised controlled trial. Br. J. Nutr. 2013, 110, 1696–1703. [Google Scholar] [CrossRef] [Green Version]
- Kadooka, Y.; Ogawa, A.; Ikuyama, K.; Sato, M. The probiotic Lactobacillus gasseri SBT2055 inhibits enlargement of visceral adipocytes and upregulation of serum soluble adhesion molecule (sICAM-1) in rats. Int. Dairy J. 2011, 21, 623–627. [Google Scholar] [CrossRef]
- Hill, D.; Sugrue, I.; Arendt, E.; Hill, C.; Stanton, C.; Ross, R.P. Recent advances in microbial fermentation for dairy and health. F1000Research 2017, 6, 751. [Google Scholar] [CrossRef]
- Kim, D.-H.; Jeong, D.; Kang, I.-B.; Kim, H.; Song, K.-Y.; Seo, K.-H. Dual function of Lactobacillus kefiri DH5 in preventing high-fat-diet-induced obesity: Direct reduction of cholesterol and upregulation of PPAR-α in adipose tissue. Mol. Nutr. Food Res. 2017, 61, 1700252. [Google Scholar] [CrossRef]
- Wakil, S.J.; Abu-Elheiga, L.A. Fatty acid metabolism: Target for metabolic syndrome. J. Lipid Res. 2009, 50, S138–S143. [Google Scholar] [CrossRef] [Green Version]
- Kinariwala, D.; Panchal, G.; Sakure, A.; Hati, S. Exploring the potentiality of lactobacillus cultures on the production of milk-derived bioactive peptides with antidiabetic activity. Int. J. Pept. Res. Ther. 2020, 26, 1613–1627. [Google Scholar] [CrossRef]
- Lim, J.; Kale, M.; Kim, D.-H.; Kim, H.-S.; Chon, J.-W.; Seo, K.-H.; Lee, H.G.; Yokoyama, W.; Kim, H. Antiobesity effect of exopolysaccharides isolated from kefir grains. J. Agric. Food Chem. 2017, 65, 10011–10019. [Google Scholar] [CrossRef]
- Dao, M.C.; Everard, A.; Aron-Wisnewsky, J.; Sokolovska, N.; Prifti, E.; Verger, E.O.; Kayser, B.D.; Levenez, F.; Chilloux, J.; Hoyles, L.; et al. Akkermansia muciniphila and improved metabolic health during a dietary intervention in obesity: Relationship with gut microbiome richness and ecology. Gut 2016, 65, 426–436. [Google Scholar] [CrossRef] [Green Version]
- Medrano, M.; Gangoiti, M.V.; Simonelli, N.; Abraham, A.G. Kefiran fermentation by human faecal microbiota: Organic acids production and in vitro biological activity. Bioact. Carbohydr. Diet. Fibre 2020, 24, 100229. [Google Scholar] [CrossRef]
- Nabavi, S.; Rafraf, M.; Somi, M.; Homayouni-Rad, A.; Asghari-Jafarabadi, M. Probiotic yogurt improves body mass index and fasting insulin levels without affecting serum leptin and adiponectin levels in non-alcoholic fatty liver disease (NAFLD). J. Funct. Foods 2015, 18, 684–691. [Google Scholar] [CrossRef]
- World Health Organisation Cardiovascular Diseases (CVDs). Available online: https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds) (accessed on 18 October 2021).
- Bays, H.E.; Taub, P.R.; Epstein, E.; Michos, E.D.; Ferraro, R.A.; Bailey, A.L.; Kelli, H.M.; Ferdinand, K.C.; Echols, M.R.; Weintraub, H.; et al. Ten things to know about ten cardiovascular disease risk factors. Am. J. Prev. Cardiol. 2021, 5, 100149. [Google Scholar] [CrossRef]
- Cruijsen, E.; Jacobo Cejudo, M.G.; Küpers, L.K.; Busstra, M.C.; Geleijnse, J.M. Dairy consumption and mortality after myocardial infarction: A prospective analysis in the Alpha Omega Cohort. Am. J. Clin. Nutr. 2021, 114, 59–69. [Google Scholar] [CrossRef]
- Wu, Y.; Zhang, Q.; Ren, Y.; Ruan, Z. Effect of probiotic Lactobacillus on lipid profile: A systematic review and meta-analysis of randomized, controlled trials. PLoS ONE 2017, 12, e0178868. [Google Scholar] [CrossRef] [PubMed]
- Pereira, D.I.A.; Gibson, G.R. Cholesterol assimilation by lactic acid bacteria and bifidobacteria isolated from the human gut. Appl. Environ. Microbiol. 2002, 68, 4689–4693. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anderson, J.W.; Gilliland, S.E. Effect of fermented milk (yogurt) containing lactobacillus acidophilus L1 on serum cholesterol in hypercholesterolemic humans. J. Am. Coll. Nutr. 1999, 18, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Chiu, C.-H.; Lu, T.-Y.; Tseng, Y.-Y.; Pan, T.-M. The effects of Lactobacillus-fermented milk on lipid metabolism in hamsters fed on high-cholesterol diet. Appl. Microbiol. Biotechnol. 2006, 71, 238–245. [Google Scholar] [CrossRef] [PubMed]
- Akbarzadeh, F.; Homayouni, A. Dairy probiotic foods and coronary heart disease: A review on mechanism of action. In Probiotics; Rigobelo, E., Ed.; InTech: London, UK, 2012; pp. 121–128. [Google Scholar]
- Tomaro-Duchesneau, C.; Jones, M.L.; Shah, D.; Jain, P.; Saha, S.; Prakash, S. Cholesterol assimilation by lactobacillus probiotic bacteria: An in vitro investigation. Biomed Res. Int. 2014, 2014, 380316. [Google Scholar] [CrossRef] [Green Version]
- Agerholm-Larsen, L.; Raben, A.; Haulrik, N.; Hansen, A.; Manders, M.; Astrup, A. Effect of 8 week intake of probiotic milk products on risk factors for cardiovascular diseases. Eur. J. Clin. Nutr. 2000, 54, 288–297. [Google Scholar] [CrossRef]
- Albano, C.; Morandi, S.; Silvetti, T.; Casiraghi, M.C.; Manini, F.; Brasca, M. Lactic acid bacteria with cholesterol-lowering properties for dairy applications: In vitro and in situ activity. J. Dairy Sci. 2018, 101, 10807–10818. [Google Scholar] [CrossRef] [Green Version]
- Fabian, E.; Elmadfa, I. Influence of daily consumption of probiotic and conventional yoghurt on the plasma lipid profile in young healthy women. Ann. Nutr. Metab. 2006, 50, 387–393. [Google Scholar] [CrossRef]
- Matsuzaki, T.; Yamazaki, R.; Hashimoto, S.; Yokokura, T. Antidiabetic effects of an oral administration of lactobacillus casei in a non-insulin-dependent diabetes mellitus (niddm) model using kk-ay mice. Endocr. J. 1997, 44, 357–365. [Google Scholar] [CrossRef] [Green Version]
- Naito, E.; Yoshida, Y.; Makino, K.; Kounoshi, Y.; Kunihiro, S.; Takahashi, R.; Matsuzaki, T.; Miyazaki, K.; Ishikawa, F. Beneficial effect of oral administration of Lactobacillus casei strain shirota on insulin resistance in diet-induced obesity mice. J. Appl. Microbiol. 2011, 110, 650–657. [Google Scholar] [CrossRef]
- Okubo, H.; Sakoda, H.; Kushiyama, A.; Fujishiro, M.; Nakatsu, Y.; Fukushima, T.; Matsunaga, Y.; Kamata, H.; Asahara, T.; Yoshida, Y.; et al. Lactobacillus casei strain shirota protects against nonalcoholic steatohepatitis development in a rodent model. Am. J. Physiol. Liver Physiol. 2013, 305, G911–G918. [Google Scholar] [CrossRef]
- Naito, E.; Yoshida, Y.; Kunihiro, S.; Makino, K.; Kasahara, K.; Kounoshi, Y.; Aida, M.; Hoshi, R.; Watanabe, O.; Igarashi, T.; et al. Effect of lactobacillus casei strain shirota-fermented milk on metabolic abnormalities in obese prediabetic japanese men: A randomised, double-blind, placebo-controlled trial. Biosci. Microbiota Food Health 2018, 37, 9–18. [Google Scholar] [CrossRef] [Green Version]
- Gil-Rodríguez, A.M.; Beresford, T. Bile salt hydrolase and lipase inhibitory activity in reconstituted skim milk fermented with lactic acid bacteria. J. Funct. Foods 2021, 77, 104342. [Google Scholar] [CrossRef]
- Liong, M.T.; Shah, N.P. Bile salt deconjugation ability, bile salt hydrolase activity and cholesterol co-precipitation ability of lactobacilli strains. Int. Dairy J. 2005, 15, 391–398. [Google Scholar] [CrossRef]
- Hsu, C.; Hou, Y.; Wang, C.; Lin, S.; Jhou, B.; Chen, C.-C.; Chen, Y. Antiobesity and uric acid-lowering effect of lactobacillus plantarum GKM3 in high-fat-diet-induced obese rats. J. Am. Coll. Nutr. 2019, 38, 623–632. [Google Scholar] [CrossRef]
- Ostadrahimi, A.; Taghizadeh, A.; Mobasseri, M.; Farrin, N.; Payahoo, L.; Beyramalipoor Gheshlaghi, Z.; Vahedjabbari, M. Effect of probiotic fermented milk (kefir) on glycemic control and lipid profile in type 2 diabetic patients: A randomized double-blind placebo-controlled clinical trial. Iran. J. Public Health 2015, 44, 228–237. [Google Scholar]
- Yusuf, D.; Nuraida, L.; Dewanti-Hariyadi, R.; Hunaefi, D. In Vitro Characterization of Lactic Acid Bacteria from Indonesian Kefir Grains as Probiotics with Cholesterol-Lowering Effect. J. Microbiol. Biotechnol. 2020, 30, 726–732. [Google Scholar] [CrossRef]
- Ito, M.; Kusuhara, S.; Yokoi, W.; Sato, T.; Ishiki, H.; Miida, S.; Matsui, A.; Nakamori, K.; Nonaka, C.; Miyazaki, K. Streptococcus thermophilus fermented milk reduces serum MDA-LDL and blood pressure in healthy and mildly hypercholesterolaemic adults. Benef. Microbes 2017, 8, 171–178. [Google Scholar] [CrossRef]
- Fujihara, K.; Suzuki, H.; Sato, A.; Kodama, S.; Heianza, Y.; Saito, K.; Iwasaki, H.; Kobayashi, K.; Yatoh, S.; Takahashi, A.; et al. Circulating malondialdehyde-modified LDL-related variables and coronary artery stenosis in asymptomatic patients with type 2 diabetes. J. Diabetes Res. 2015, 2015, 507245. [Google Scholar] [CrossRef]
- Ono, M.; Takebe, N.; Oda, T.; Nakagawa, R.; Matsui, M.; Sasai, T.; Nagasawa, K.; Honma, H.; Kajiwara, T.; Taneichi, H.; et al. Association of coronary artery calcification with MDA-LDL-C/LDL-C and urinary 8-isoprostane in japanese patients with type 2 diabetes. Intern. Med. 2014, 53, 391–396. [Google Scholar] [CrossRef] [Green Version]
- Nakajima, H.; Suzuki, Y.; Hirota, T. Cholesterol lowering activity of ropy fermented milk. J. Food Sci. 1992, 57, 1327–1329. [Google Scholar] [CrossRef]
- World Health Organisation Noncommunicable Diseases: Hypertension. Available online: https://www.who.int/news-room/q-a-detail/noncommunicable-diseases-hypertension (accessed on 23 August 2021).
- Beltrán-Barrientos, L.M.; Hernández-Mendoza, A.; Torres-Llanez, M.J.; González-Córdova, A.F.; Vallejo-Córdoba, B. Invited review: Fermented milk as antihypertensive functional food. J. Dairy Sci. 2016, 99, 4099–4110. [Google Scholar] [CrossRef] [Green Version]
- Cook, N.R.; Cohen, J.; Hebert, P.R.; Taylor, J.O.; Hennekens, C.H. Implications of small reductions in diastolic blood pressure for primary prevention. Arch. Intern. Med. 1995, 155, 701–709. [Google Scholar] [CrossRef]
- Beltrán-Barrientos, L.; Hernández-Mendoza, A.; González-Córdova, A.; Astiazarán-García, H.; Esparza-Romero, J.; Vallejo-Córdoba, B. Mechanistic pathways underlying the antihypertensive effect of fermented milk with lactococcus lactis NRRL B-50571 in spontaneously hypertensive rats. Nutrients 2018, 10, 262. [Google Scholar] [CrossRef] [Green Version]
- Vasquez, E.C.; Pereira, T.M.C.; Peotta, V.A.; Baldo, M.P.; Campos-Toimil, M. Probiotics as Beneficial Dietary Supplements to Prevent and Treat Cardiovascular Diseases: Uncovering Their Impact on Oxidative Stress. Oxid. Med. Cell. Longev. 2019, 2019, 3086270. [Google Scholar] [CrossRef] [Green Version]
- Yamaguchi, N.; Kawaguchi, K.; Yamamoto, N. Study of the mechanism of antihypertensive peptides VPP and IPP in spontaneously hypertensive rats by DNA microarray analysis. Eur. J. Pharmacol. 2009, 620, 71–77. [Google Scholar] [CrossRef]
- Beltrán-Barrientos, L.M.; González-Córdova, A.F.; Hernández-Mendoza, A.; Torres-Inguanzo, E.H.; Astiazarán-García, H.; Esparza-Romero, J.; Vallejo-Cordoba, B. Randomized double-blind controlled clinical trial of the blood pressure–lowering effect of fermented milk with Lactococcus lactis: A pilot study. J. Dairy Sci. 2018, 101, 2819–2825. [Google Scholar] [CrossRef] [Green Version]
- Rodríguez-Figueroa, J.C.; González-Córdova, A.F.; Astiazaran-García, H.; Hernández-Mendoza, A.; Vallejo-Cordoba, B. Antihypertensive and hypolipidemic effect of milk fermented by specific Lactococcus lactis strains. J. Dairy Sci. 2013, 96, 4094–4099. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moreno-Montoro, M.; Olalla-Herrera, M.; Rufián-Henares, J.Á.; Martínez, R.G.; Miralles, B.; Bergillos, T.; Navarro-Alarcón, M.; Jauregi, P. Antioxidant, ACE-inhibitory and antimicrobial activity of fermented goat milk: Activity and physicochemical property relationship of the peptide components. Food Funct. 2017, 8, 2783–2791. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wasilewska, E.; Zlotkowska, D.; Wroblewska, B. Yogurt starter cultures of Streptococcus thermophilus and Lactobacillus bulgaricus ameliorate symptoms and modulate the immune response in a mouse model of dextran sulfate sodium-induced colitis. J. Dairy Sci. 2019, 102, 37–53. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Makino, S.; Sato, A.; Goto, A.; Nakamura, M.; Ogawa, M.; Chiba, Y.; Hemmi, J.; Kano, H.; Takeda, K.; Okumura, K.; et al. Enhanced natural killer cell activation by exopolysaccharides derived from yogurt fermented with Lactobacillus delbrueckii ssp. bulgaricus OLL1073R-1. J. Dairy Sci. 2016, 99, 915–923. [Google Scholar] [CrossRef] [Green Version]
- de Moreno de LeBlanc, A.; Perdigón, G. Reduction of beta-glucuronidase and nitroreductase activity by yoghurt in a murine colon cancer model. Biocell 2005, 29, 15–24. [Google Scholar] [PubMed]
- Qian, Y.; Li, M.; Wang, W.; Wang, H.; Zhang, Y.; Hu, Q.; Zhao, X.; Suo, H. Effects of lactobacillus casei YBJ02 on lipid metabolism in hyperlipidemic mice. J. Food Sci. 2019, 84, 3793–3803. [Google Scholar] [CrossRef] [PubMed]
- Daliri, E.B.-M.; Lee, B.H.; Park, B.-J.; Kim, S.-H.; Oh, D.-H. Antihypertensive peptides from whey proteins fermented by lactic acid bacteria. Food Sci. Biotechnol. 2018, 27, 1781–1789. [Google Scholar] [CrossRef]
- Zhao, X.; Zhang, J.; Yi, S.; Li, X.; Guo, Z.; Zhou, X.; Mu, J.; Yi, R. Lactobacillus plantarum CQPC02 Prevents Obesity in Mice through the PPAR-α Signaling Pathway. Biomolecules 2019, 9, 407. [Google Scholar]
- Cho, W.-Y.; Hong, G.-E.; Lee, H.-J.; Yeon, S.-J.; Paik, H.-D.; Hosaka, Y.; Lee, C.-H. Effect of yogurt fermented by lactobacillus fermentum TSI and l. Fermentum S2 derived from a mongolian traditional dairy product on rats with high-fat-diet-induced obesity. Foods 2020, 9, 594. [Google Scholar] [CrossRef]
- Aragón, F.; Carino, S.; Perdigón, G.; de Moreno de LeBlanc, A. Inhibition of growth and metastasis of breast cancer in mice by milk fermented with Lactobacillus casei CRL 431. J. Immunother. 2015, 38, 185–196. [Google Scholar] [CrossRef]
- Méndez Utz, V.E.; Pérez Visñuk, D.; Perdigón, G.; de Moreno de LeBlanc, A. Milk fermented by Lactobacillus casei CRL431 administered as an immune adjuvant in models of breast cancer and metastasis under chemotherapy. Appl. Microbiol. Biotechnol. 2021, 105, 327–340. [Google Scholar] [CrossRef]
- Mendez Utz, V.E.; Perdigón, G.; de Moreno de LeBlanc, A. Oral administration of milk fermented by Lactobacillus casei CRL431 was able to decrease metastasis from breast cancer in a murine model by modulating immune response locally in the lungs. J. Funct. Foods 2019, 54, 263–270. [Google Scholar] [CrossRef]
- Aragón, F.; Carino, S.; Perdigón, G.; de Moreno de LeBlanc, A. The administration of milk fermented by the probiotic Lactobacillus casei CRL 431 exerts an immunomodulatory effect against a breast tumour in a mouse model. Immunobiology 2014, 219, 457–464. [Google Scholar] [CrossRef]
- Donkor, O.N.; Henriksson, A.; Singh, T.K.; Vasiljevic, T.; Shah, N.P. ACE-inhibitory activity of probiotic yoghurt. Int. Dairy J. 2007, 17, 1321–1331. [Google Scholar] [CrossRef]
- Kalergis, M.; Leung Yinko, S.S.L.; Nedelcu, R. Dairy products and prevention of type 2 diabetes: Implications for research and practice. Front. Endocrinol. 2013, 4, 90. [Google Scholar] [CrossRef] [Green Version]
- Saeedi, P.; Petersohn, I.; Salpea, P.; Malanda, B.; Karuranga, S.; Unwin, N.; Colagiuri, S.; Guariguata, L.; Motala, A.A.; Ogurtsova, K.; et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas. Diabetes Res. Clin. Pract. 2019, 157, 107843. [Google Scholar] [CrossRef] [Green Version]
- Das, S.K.; Elbein, S.C. The Genetic Basis of Type 2 Diabetes. Cellscience 2006, 2, 100–131. [Google Scholar] [CrossRef]
- International Diabetes Federation Type 2 Diabetes. Available online: https://www.idf.org/aboutdiabetes/type-2-diabetes.html (accessed on 23 August 2021).
- Choi, H.K. Dairy consumption and risk of type 2 diabetes mellitus in men. Arch. Intern. Med. 2005, 165, 997. [Google Scholar] [CrossRef] [Green Version]
- Malik, V.S.; Sun, Q.; van Dam, R.M.; Rimm, E.B.; Willett, W.C.; Rosner, B.; Hu, F.B. Adolescent dairy product consumption and risk of type 2 diabetes in middle-aged women. Am. J. Clin. Nutr. 2011, 94, 854–861. [Google Scholar] [CrossRef] [Green Version]
- Sluijs, I.; Forouhi, N.G.; Beulens, J.W.J.; van der Schouw, Y.T.; Agnoli, C.; Arriola, L.; Balkau, B.; Barricarte, A.; Boeing, H.; Bueno-de-Mesquita, H.B.; et al. The amount and type of dairy product intake and incident type 2 diabetes: Results from the EPIC-InterAct Study. Am. J. Clin. Nutr. 2012, 96, 382–390. [Google Scholar] [CrossRef] [Green Version]
- Tong, X.; Dong, J.-Y.; Wu, Z.-W.; Li, W.; Qin, L.-Q. Dairy consumption and risk of type 2 diabetes mellitus: A meta-analysis of cohort studies. Eur. J. Clin. Nutr. 2011, 65, 1027–1031. [Google Scholar] [CrossRef] [Green Version]
- Ejtahed, H.S.; Mohtadi-Nia, J.; Homayouni-Rad, A.; Niafar, M.; Asghari-Jafarabadi, M.; Mofid, V. Probiotic yogurt improves antioxidant status in type 2 diabetic patients. Nutrition 2012, 28, 539–543. [Google Scholar] [CrossRef]
- Pei, R.; Martin, D.A.; DiMarco, D.M.; Bolling, B.W. Evidence for the effects of yogurt on gut health and obesity. Crit. Rev. Food Sci. Nutr. 2017, 57, 1569–1583. [Google Scholar] [CrossRef] [PubMed]
- Yadav, H.; Jain, S.; Sinha, P.R. Antidiabetic effect of probiotic dahi containing Lactobacillus acidophilus and Lactobacillus casei in high fructose fed rats. Nutrition 2007, 23, 62–68. [Google Scholar] [CrossRef] [PubMed]
- Salguero, M.; Al-Obaide, M.; Singh, R.; Siepmann, T.; Vasylyeva, T. Dysbiosis of gram-negative gut microbiota and the associated serum lipopolysaccharide exacerbates inflammation in type 2 diabetic patients with chronic kidney disease. Exp. Ther. Med. 2019, 18, 3461–3469. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schiffrin, E.J.; Parlesak, A.; Bode, C.; Christian Bode, J.; van’t Hof, M.A.; Grathwohl, D.; Guigoz, Y. Probiotic yogurt in the elderly with intestinal bacterial overgrowth: Endotoxaemia and innate immune functions. Br. J. Nutr. 2008, 101, 961–966. [Google Scholar] [CrossRef] [Green Version]
- Mohamadshahi, M.; Veissi, M.; Haidari, F.; Shahbazian, H.; Kaydani, G.-A.; Mohammadi, F. Effects of probiotic yogurt consumption on inflammatory biomarkers in patients with type 2 diabetes. Bioimpacts 2014, 4, 83–88. [Google Scholar] [CrossRef]
- Toshimitsu, T.; Gotou, A.; Sashihara, T.; Furuichi, K.; Hachimura, S.; Shioya, N.; Suzuki, S.; Asami, Y. Ingesting yogurt containing Lactobacillus plantarum OLL2712 reduces abdominal fat accumulation and chronic inflammation in overweight adults in a randomized placebo-controlled trial. Curr. Dev. Nutr. 2021, 5, nzab006. [Google Scholar] [CrossRef]
- Chen, Y.-T.; Lin, Y.-C.; Lin, J.-S.; Yang, N.-S.; Chen, M.-J. Sugary kefir strain Lactobacillus mali APS1 ameliorated hepatic steatosis by regulation of SIRT-1/Nrf-2 and gut microbiota in rats. Mol. Nutr. Food Res. 2018, 62, 1700903. [Google Scholar] [CrossRef]
- Pugliero, S.; Lima, D.Y.; Rodrigues, A.M.; Bogsan, C.S.B.; Rogero, M.M.; Punaro, G.R.; Higa, E.M.S. Kefir reduces nitrosative stress and upregulates Nrf2 in the kidney of diabetic rats. Int. Dairy J. 2021, 114, 104909. [Google Scholar] [CrossRef]
- Virtanen, T.; Pihlanto, A.; Akkanen, S.; Korhonen, H. Development of antioxidant activity in milk whey during fermentation with lactic acid bacteria. J. Appl. Microbiol. 2007, 102, 106–115. [Google Scholar] [CrossRef]
- Apostolidis, E.; Kwon, Y.I.; Shetty, K. Potential of select yogurts for diabetes and hypertension management. J. Food Biochem. 2006, 30, 699–717. [Google Scholar] [CrossRef]
- Chen, H.L.; Tsai, T.C.; Tsai, Y.C.; Liao, J.W.; Yen, C.C.; Chen, C.M. Kefir peptides prevent high-fructose corn syrup-induced non-alcoholic fatty liver disease in a murine model by modulation of inflammation and the JAK2 signaling pathway. Nutr. Diabetes 2016, 6, e237. [Google Scholar] [CrossRef]
- Kadooka, Y.; Sato, M.; Imaizumi, K.; Ogawa, A.; Ikuyama, K.; Akai, Y.; Okano, M.; Kagoshima, M.; Tsuchida, T. Regulation of abdominal adiposity by probiotics (Lactobacillus gasseri SBT2055) in adults with obese tendencies in a randomized controlled trial. Eur. J. Clin. Nutr. 2010, 64, 636–643. [Google Scholar] [CrossRef] [Green Version]
- World Health Organisation Cancer. Available online: https://www.who.int/news-room/fact-sheets/detail/cancer (accessed on 23 August 2021).
- Lampe, J.W. Dairy products and cancer. J. Am. Coll. Nutr. 2011, 30, 464S–470S. [Google Scholar] [CrossRef]
- Marmot, M.; Atinmo, T.; Byers, T.; Chen, J.; Hirohata, T.; Jackson, A.; James, W.; Kolonel, L.; Kumanyika, S.; Leitzmann, C.; et al. Food, Nutrition, Physical Activity, and the Prevention of Cancer: A Global Perspective (WCRF/AICR Expert Report); World Cancer Research Fund/American Institute for Cancer Research: Washington, DC, WA, USA, 2007. [Google Scholar]
- Davoodi, H.; Esmaeili, S.; Mortazavian, A.M. Effects of milk and milk products consumption on cancer: A review. Compr. Rev. Food Sci. Food Saf. 2013, 12, 249–264. [Google Scholar] [CrossRef]
- Tasdemir, S.S.; Sanlier, N. An insight into the anticancer effects of fermented foods: A review. J. Funct. Foods 2020, 75, 104281. [Google Scholar] [CrossRef]
- de Moreno de LeBlanc, A.; Matar, C.; Perdigón, G. The application of probiotics in cancer. Br. J. Nutr. 2007, 98 (Suppl. 1), S105–S110. [Google Scholar] [CrossRef] [Green Version]
- De Moreno De Leblanc, A.; Perdigón, G. The application of probiotic fermented milks in cancer and intestinal inflammation. Proc. Nutr. Soc. 2010, 69, 421–428. [Google Scholar] [CrossRef] [Green Version]
- Ghoneum, M.; Gimzewski, J. Apoptotic effect of a novel kefir product, PFT, on multidrug-resistant myeloid leukemia cells via a hole-piercing mechanism. Int. J. Oncol. 2014, 44, 830–837. [Google Scholar] [CrossRef] [Green Version]
- Lavelle, A.; Sokol, H. Gut microbiota-derived metabolites as key actors in inflammatory bowel disease. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 223–237. [Google Scholar] [CrossRef]
- Guan, Q. A comprehensive review and update on the pathogenesis of inflammatory bowel disease. J. Immunol. Res. 2019, 2019, 7247238. [Google Scholar] [CrossRef] [Green Version]
- Ng, S.C.; Shi, H.Y.; Hamidi, N.; Underwood, F.E.; Tang, W.; Benchimol, E.I.; Panaccione, R.; Ghosh, S.; Wu, J.C.Y.; Chan, F.K.L.; et al. Worldwide incidence and prevalence of inflammatory bowel disease in the 21st century: A systematic review of population-based studies. Lancet 2017, 390, 2769–2778. [Google Scholar] [CrossRef]
- Lorea Baroja, M.; Kirjavainen, P.V.; Hekmat, S.; Reid, G. Anti-inflammatory effects of probiotic yogurt in inflammatory bowel disease patients. Clin. Exp. Immunol. 2007, 149, 470–479. [Google Scholar] [CrossRef]
- Saez-Lara, M.J.; Gomez-Llorente, C.; Plaza-Diaz, J.; Gil, A. The role of probiotic lactic acid bacteria and bifidobacteria in the prevention and treatment of inflammatory bowel disease and other related diseases: A systematic review of randomized human clinical trials. Biomed Res. Int. 2015, 2015, 1–15. [Google Scholar] [CrossRef]
- Santiago-López, L.; Hernández-Mendoza, A.; Mata-Haro, V.; Vallejo-Córdoba, B.; Wall-Medrano, A.; Astiazarán-García, H.; Estrada-Montoya, M.; González-Córdova, A. Effect of milk fermented with lactobacillus fermentum on the inflammatory response in mice. Nutrients 2018, 10, 1039. [Google Scholar] [CrossRef] [Green Version]
- Owaga, E.; Hsieh, R.-H.; Mugendi, B.; Masuku, S.; Shih, C.-K.; Chang, J.-S. Th17 cells as potential probiotic therapeutic targets in inflammatory bowel diseases. Int. J. Mol. Sci. 2015, 16, 20841–20858. [Google Scholar] [CrossRef] [Green Version]
- Lee, H.-S.; Han, S.-Y.; Bae, E.-A.; Huh, C.-S.; Ahn, Y.-T.; Lee, J.-H.; Kim, D.-H. Lactic acid bacteria inhibit proinflammatory cytokine expression and bacterial glycosaminoglycan degradation activity in dextran sulfate sodium-induced colitic mice. Int. Immunopharmacol. 2008, 8, 574–580. [Google Scholar] [CrossRef]
- Håversen, L.; Ohlsson, B.G.; Hahn-Zoric, M.; Hanson, L.Å.; Mattsby-Baltzer, I. Lactoferrin down-regulates the LPS-induced cytokine production in monocytic cells via NF-κB. Cell. Immunol. 2002, 220, 83–95. [Google Scholar] [CrossRef]
- Senol, A.; Isler, M.; Sutcu, R.; Akin, M.; Cakir, E.; Ceyhan, B.M.; Kockar, M.C. Kefir treatment ameliorates dextran sulfate sodium-induced colitis in rats. World J. Gastroenterol. 2015, 21, 13020–13029. [Google Scholar] [CrossRef] [PubMed]
- Sevencan, N.O.; Isler, M.; Kapucuoglu, F.N.; Senol, A.; Kayhan, B.; Kiztanir, S.; Kockar, M.C. Dose-dependent effects of kefir on colitis induced by trinitrobenzene sulfonic acid in rats. Food Sci. Nutr. 2019, 7, 3110–3118. [Google Scholar] [CrossRef] [PubMed]
- Curciarello, R.; Canziani, K.E.; Salto, I.; Barbiera Romero, E.; Rocca, A.; Doldan, I.; Peton, E.; Brayer, S.; Sambuelli, A.M.; Goncalves, S.; et al. Probiotic lactobacilli isolated from kefir promote down-regulation of inflammatory lamina propria t cells from patients with active IBD. Front. Pharmacol. 2021, 12, 658026. [Google Scholar] [CrossRef] [PubMed]

| LAB | Family | Genus | Gram −/+ | Growth Conditions | Type of Lactic Acid | ||
|---|---|---|---|---|---|---|---|
| Heat-Stable (45 °C) | Salt-Tolerant (18% NaCl) | Acid-Resistant (pH 4.4) | |||||
| Dairy | |||||||
| Lactobacillaceae | Lactobacillus | + | Changeable | - | Changeable | D, L, DL | |
| Pediococcus | + | Changeable | - | + | L, DL | ||
| Streptococcaceae | Streptococcus | + | Changeable | - | - | L | |
| Lactococcus | + | - | - | Changeable | L | ||
| Propionibacteriaceae | Propionibacterium | + | - | - | - | ||
| Enterococcaceae | Enterococcus | + | + | - | + | L | |
| Leuconostocaecae | Leuconostoc | + | - | - | Changeable | D | |
| Nondairy | |||||||
| Aerococcaceae | Aerococcus | + | - | - | - | L | |
| Carnobacteriaceae | Carnobacterium | + | - | - | NA | L | |
| Enterococcaceae | Tetragenococcus | + | - | + | Changeable | L | |
| Enterococcaceae | Vagococcus | + | - | - | NA | L | |
| Fructobacillus | + | NA | - | NA | D | ||
| Leuconostocaecae | Oenococcus | + | - | - | Changeable | D | |
| Weissella | + | - | - | Changeable | D, L | ||
| Dairy Products | Isolated Probiotic Strains | Their Bioactivities and Stability Issues | Reference(s) |
|---|---|---|---|
| Kalarei, a traditional fermented cheese product | Pediococcus acidilactici SMVDUDB2 | * An 80% survival rate at low pH (2.0 and 3.0) and high bile salt concentration (0.3 and 0.5%) * High hydrophobicity affinity (33.3%) with ethyl acetate * Autoaggregation (77.68 ± 0.68%) and coaggregation (73.57 ± 0.47%) with Staphylococcus aureus (MTCC 3160) * Antibacterial activity against Bacillus subtilis (MTCC 121), Mycobacterium smegmatis (MTCC 994), Staphylococcus aureus (MTCC 3160), Proteus vulgaris (MTCC 426), Escherichia coli (MTCC 1652), and Lactocaseibacillus rhamnosus (MTCC 1408) | [37] |
| Ezine cheese (a Turkish cheese) | Enterococcus lactis PMD74 | * The strain showed autoaggregative (41%) and coaggregative properties along with high viability at acidic pH (3.0) and in the presence of pepsin, pancreatin, and bile salts (0.3% and 0.5%). * The strain PMD74 inhibited the growth of a number of Gram-positive bacteria (Listeria monocytogenes, Lactobacillus sake, Staphylococcus aureus, and Enterococcus faecalis). | [36] |
| Tulum cheese (a Turkish cheese) | Seven Limosilactobacillus fermentum strains | * Limosilactobacillus fermentum LP3 and LP4 were able to tolerate acidic pH (2.5) and 1% bile salt. * Although all strains had similar enzymatic activity and antibiotic resistance patterns, the highest antagonistic effect belonged to LP3, LP4, and LP6 and the highest cholesterol assimilation belonged to LP3 and LP4, respectively. | [39] |
| Probiotic yogurt | Lactobacillus acidophilus, Bifidobacterium bifidum, Lactiplantibacillus plantarum, Lacticaseibacillus casei | * A combination of Lactobacillus acidophilus and Bifidobacterium bifidum survived at pH 1.5 during an incubation period of 1.5 h and also showed good survivability at 0.3% bile salt concentration. * At pH 2.0, 3.0, and 4.0, the survivability rate for Lactobacillus acidophilus and Bifidobacterium bifidum was 54, 66, and 64%, respectively. | [40] |
| Yogurt | Streptococcus thermophilus BGKMJ1-36 and Lactobacillus bulgaricus BGVLJ1-21 | * Both strains grew at 37 and 45 °C in GM17 broth, while they did not grow in GM17 broth with 2% NaCl. * Both strains showed antimicrobial activity toward Listeria monocytogenes, while the BGKMJ1-36 strain produced EPS. * The colonies of BGKMJ1-36 and BGVLJ1-21 strains that successfully survived transit of the yogurt via simulated gastrointestinal tract conditions have been examined for adhesion to intestinal epithelial Caco-2 cells. | [41] |
| Iranian traditional yogurts | 12 LAB isolates from two genera (Pediococcus; 6 P. acidilacticii isolates and Lactobacillus; 2 Lactiplantibacillus plantarum, 2 Levilactobacillus brevis, 1 Limosilactobacillus fermentum and 1 Lactobacillus kefiri isolates). | * Limosilactobacillus fermentum 27 had the highest acid tolerance, while Levilactobacillus brevis 25 had the highest bile salt tolerance. * Pediococcus acidilactici 23 showed a lower acid tolerance as well as Levilactobacillus Brevis 86 exhibited a lower bile salt tolerance than others. | [35] |
| Local dairy (cow milk, buffalo milk, cheese, and yogurt) | Lactobacillus alimentarius, Lactobacillus sake, and Lactobacillus collinoides | * The Lactobacillus strains inhibited pathogens’ growth. * All three isolates showed moderate activity apart from Lactobacillus collinoides and Lactobacillus alimentarius, which had relatively strong activity against Pseudomonas aeruginosa and Bacillus subtilis. | [42] |
| 30 dairy samples (household milk and curd) | 12 Lactobacillus isolates (LBS 1-LBS 12) | * Eight isolates (LBS 1-6, 8 and 11) were bile resistant (survival >50% at 0.3% bile salt w/v) and five isolates (LBS 1, 2, 5, 6 and 11) were resistant at acidic pH (survival >50% at pH 3). * All isolates inhibited the growth of Staphylococcus aureus. * LBS 2 also inhibited the growth of Escherichia coli and Salmonella typhimurium. * Isolate LBS 2 was resistant to five antibiotics as well as Lactocaseibacillus rhamnosus LBS2 successfully adhered to rat epithelial cells in in vitro conditions. | [43] |
| Traditional Greek dairy products (Feta, Kasseri, Xynotyri, Graviera, Formaela, Galotyri, and Kefalotyri cheeses as well as yogurt and milk) | 25 LAB strains | * Only Streptococcus thermophilus ACA-DC 26 (Greek yogurt isolate) had antimicrobial activity (against Streptococcus mutans LMG 14558T). * Two Lactiplantibacillus plantarum strains (ACA-DC 2640 and ACA-DC 4039) showed the highest adhesion according to a collagen-based microplate assay and by using HΤ-29 and Caco-2 cells. * Milk cell-free supernatants of Lactiplantibacillus plantarum ACA-DC 2640 and ACA-DC 4039 showed strong angiotensin I-converting enzyme inhibition. * Lactiplantibacillus plantarum ACA-DC 2640, Streptococcus thermophilus ACA-DC 26, and ACA-DC 170 had anti-inflammatory activity. | [44] |
| Tibetan kefir | Lactobacillus kefiranofaciens XL10 | * XL10 survived 3-h incubation at pH 3.5 and exhibited cell surface hydrophobicity of ~79.9% and autoaggregation of ~27.8%. * XL10 successfully adhered to the mucous tissue and colonized the ileum of the mice. * XL10 modulated gut microbiota by increasing the Bifidobacteriaceae family and decreasing in Proteobacteria phyla. | [45] |
| Mongolian fermented koumiss | Lactobacillus helveticus NS8 | * Although NS8 exhibited a moderate survival ability in the gastrointestinal tract environment in vitro, an excellent adhesion ability to human intestinal cells and significant autoaggregation and cell-surface hydrophobicity were reported. * NS8 was able to decline the proinflammatory effects of lipopolysaccharide by inducing higher levels of IL-10. | [38] |
| References | Health Effect | Study Design | Intervention | Intervention Duration | Dairy Product Type | Milk Heat Treatment | LAB Species | Main Results |
|---|---|---|---|---|---|---|---|---|
| ANIMAL MODELS, IN VITRO AND IN VIVO STUDIES | ||||||||
| [58] | Cardiometabolic markers and intestinal microbiota | 8-weeks-old C57BL/6J wild-type (WT) and atherosclerotic (LRKO) male mice | HFD/high-sucrose diet [66% kcal lipids, 22% kcal carbohydrates (100% sucrose), 12% kcal proteins]. Protein sources groups: 1. 100% non-dairy protein (NDP) 2. 50% of the NDP energy replaced by milk protein (MP) 3. Lactobacillus helveticus fermented milk protein (FMP) 4. Greek-style yogurt protein (YP) | 12 weeks (WT mice) 24 weeks (LRKO mice) | Yogurt and fermented milk | 90 °C |
|
|
| [121] | Dextran sulfate sodium-induced colitis and intestinal microbiota | 8-weeks-old specific-pathogen-free BALB/c female mice | 1. Interventional colitis group (DSS-YC group) was administered 200 μL/d per mouse of the YC mixture (~5 × 109 cfu) intragastrically. 2. The control colitis group (DSS group) received saline intragastrically as a vehicle. 3. Healthy control group received normal drinking water ad libitum (without DSS) | 8 days | Yogurt culture (YC) bacteria | Not applied |
|
|
| [122] | Immunostimulatory effects |
| EPS and bacteria isolated from 3 yogurt varieties fermented with different starter cultures administered to mice at doses of 100 μg/mouse (EPS) and 109 cfu/mouse (bacteria) Yogurt types 1. Lactobacillus delbrueckii ssp. bulgaricus OLL1073R-1 and Streptococcus thermophilus OLS3059 (OLL1073R-1 yogurt) 2. Lactobacillus delbrueckii ssp. bulgaricus OLL1245 and Streptococcus thermophilus OLS3059 (yogurt A) 3. Lactobacillus delbrueckii ssp.bulgaricus OLL1256 and Streptococcus thermophilus OLS3295 (yogurt B). | 3 weeks | Yogurt | 90 °C |
|
|
| [123] | Colon cancer | BALB/c mice |
| 6 months | Yogurt | Commercial yogurt process |
|
|
| [124] | Hyperlipidemia and obesity | Swiss mice |
| 6 weeks | Yak yogurt- a Tibetan dairy product | Not reported |
|
|
| [125] | Hypertension | Rabbit lung cultures | Whey proteins were fermented with 34 LAB and their ability to inhibit ACE activity was compared. | 48 h | Whey proteins | 37 °C |
|
|
| [126] | Obesity, hyperlipidemia, and inflammation | 6-weeks-old specific- pathogen-free C57BL/6J mice |
| 8 weeks | Yogurt culture bacteria | Not reported |
|
|
| [127] | Obesity and hyperlipidemia | 6-weeks-old male Sprague-Dawley rats |
| 8 weeks | Yogurt | 85 °C |
|
|
| [128] | Breast cancer | 6-weeks-old female BALB/c mice |
| 50 days | Fermented milk | Sterilized |
|
|
| [129] | Breast cancer | 7–8 weeks-old female BALB/c mice |
| 36 days | Fermented milk | Sterilized |
|
|
| [130] | Breast cancer | 7–8 weeks-old female BALB/c mice |
| 60 days | Fermented milk | Sterilized |
|
|
| [131] | Breast cancer | BALB/c mice |
| 28 days | Fermented milk | Sterilized |
|
|
| [132] | ACE inhibitory activity and hypertension | -ACE inhibitory activity-in vitro |
| 28 days | Yogurt | 85 °C | Lactobacillus delbrueckii ssp. bulgaricus Lb1466, Streptococcus thermophilus St1342, Lactobacillus acidophilus L10, Lacticaseibacillus casei and Bifidobacterium lactis B94 |
|
| [72] | Obesity and hyperlipidemia | 4-week-old male Sprague-Dawley (SD) rats |
| 4 weeks | Whey beverage | 70 °C |
|
|
| [80] | Obesity and hyperlipidemia | 4-week-old male C57BL/6 mice |
| 6 weeks | Kefir culture | 25 °C |
|
|
| [83] | Obesity | 4-week-old male C57BL/6J mice and 3T3-L1 preadipocyte cells |
| 4 weeks | Kefir grain | 30 °C | Kefir grain culture |
|
| HUMAN STUDIES (RCTs) | ||||||||
| [77] | Obesity | 210 Japanese adults with large visceral fat areas (80.2–187.8 cm2) 35–60 years | Fermented milk groups: Containing 107 cfu LG2055/g, 200 g FM/d Containing 106 cfu LG2055/g, 200 g FM/d Containing 0 cfu LG2055/g (control), 200 g FM/d | 12 weeks | Fermented milk | 40 °C |
|
|
| [102] | T2DM | 100 obese prediabetic males (BMI ≥ 25), (1-h post-load plasma glucose (PG) levels ≥180 mg/dL) ears |
| 8 weeks | Fermented milk | Not reported | Lacticaseibacillus casei strain Shirota (LcS) |
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ağagündüz, D.; Yılmaz, B.; Şahin, T.Ö.; Güneşliol, B.E.; Ayten, Ş.; Russo, P.; Spano, G.; Rocha, J.M.; Bartkiene, E.; Özogul, F. Dairy Lactic Acid Bacteria and Their Potential Function in Dietetics: The Food–Gut-Health Axis. Foods 2021, 10, 3099. https://doi.org/10.3390/foods10123099
Ağagündüz D, Yılmaz B, Şahin TÖ, Güneşliol BE, Ayten Ş, Russo P, Spano G, Rocha JM, Bartkiene E, Özogul F. Dairy Lactic Acid Bacteria and Their Potential Function in Dietetics: The Food–Gut-Health Axis. Foods. 2021; 10(12):3099. https://doi.org/10.3390/foods10123099
Chicago/Turabian StyleAğagündüz, Duygu, Birsen Yılmaz, Teslime Özge Şahin, Bartu Eren Güneşliol, Şerife Ayten, Pasquale Russo, Giuseppe Spano, João Miguel Rocha, Elena Bartkiene, and Fatih Özogul. 2021. "Dairy Lactic Acid Bacteria and Their Potential Function in Dietetics: The Food–Gut-Health Axis" Foods 10, no. 12: 3099. https://doi.org/10.3390/foods10123099
APA StyleAğagündüz, D., Yılmaz, B., Şahin, T. Ö., Güneşliol, B. E., Ayten, Ş., Russo, P., Spano, G., Rocha, J. M., Bartkiene, E., & Özogul, F. (2021). Dairy Lactic Acid Bacteria and Their Potential Function in Dietetics: The Food–Gut-Health Axis. Foods, 10(12), 3099. https://doi.org/10.3390/foods10123099










