Analytical Characterization of the Widely Consumed Commercialized Fermented Beverages from Russia (Kefir and Ryazhenka) and South Africa (Amasi and Mahewu): Potential Functional Properties and Profiles of Volatile Organic Compounds
Abstract
:1. Introduction
2. Materials and Methods
2.1. Studied Beverages
2.2. Measurments of pH, Antioxidant Capacity, ACE Inhibitory Activity and Degree of Proteolysis
2.3. Fatty Acid Analysis
2.4. Volatile Organic Compounds Analysis
2.5. Statistical Analysis
3. Results and Discussion
3.1. Antioxidant and Antihypertensive Properties
3.2. Profile of Fatty Acids
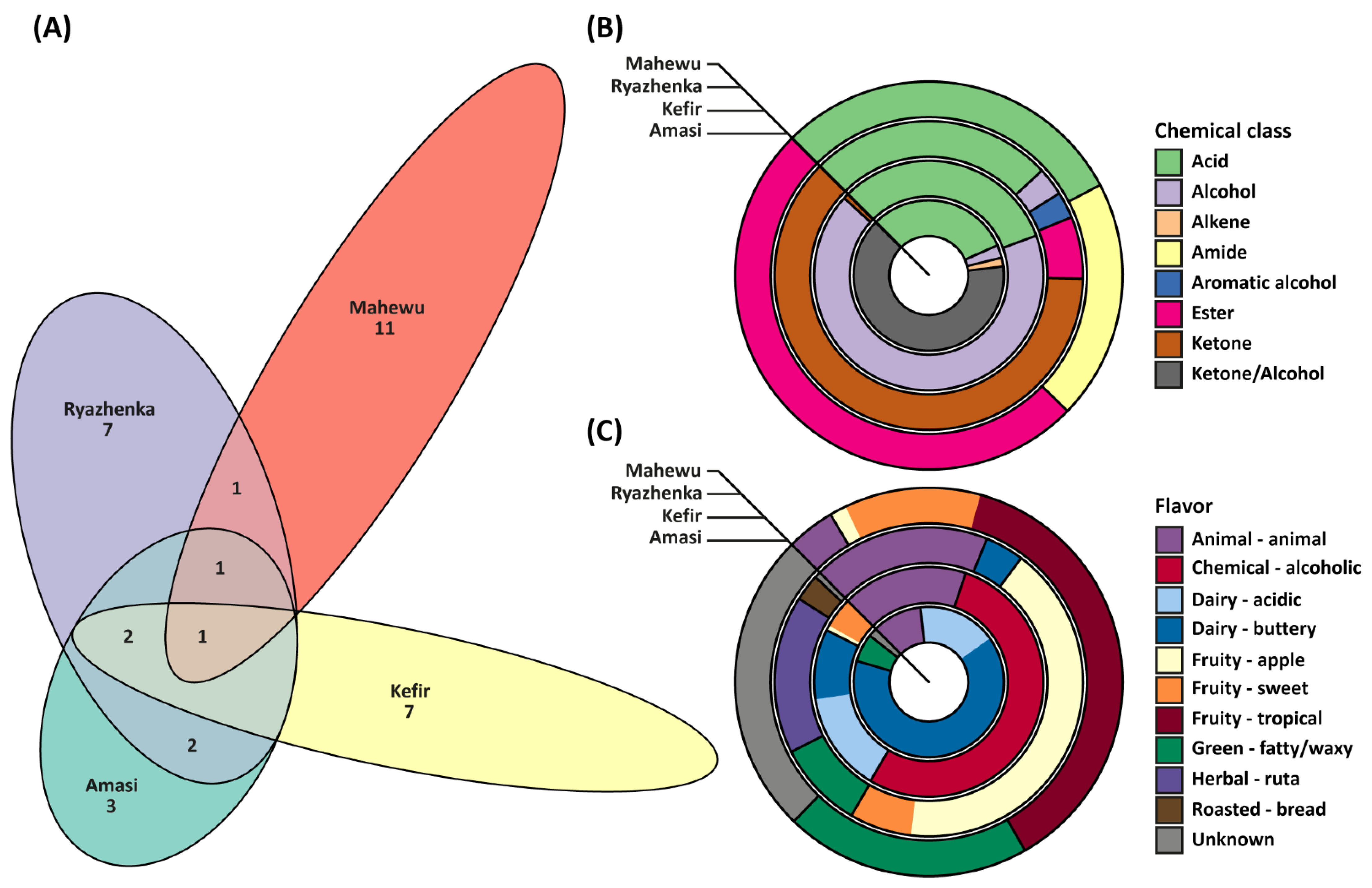
3.3. Volatile Organic Compounds
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Holzapfel, W. Appropriate starter culture technologies for small-scale fermentation in developing countries. Int. J. Food Microbiol. 2002, 75, 197–212. [Google Scholar] [CrossRef]
- Tamang, J.P.; Cotter, P.D.; Endo, A.; Han, N.S.; Kort, R.; Liu, S.Q.; Mayo, B.; Westerik, N.; Hutkins, R. Fermented foods in a global age: East meets West. Compr. Rev. Food Sci. Food Saf. 2019, 19, 184–217. [Google Scholar] [CrossRef] [Green Version]
- Kandasamy, S.; Kavitake, D.; Shetty, P.H. Lactic Acid Bacteria and Yeasts as Starter Cultures for Fermented Foods. In Innovations in Technologies for Fermented Food and Beverage Industries; Springer: Singapore, 2018; pp. 25–52. [Google Scholar]
- Rozhkova, I.V.; Moiseenko, K.V.; Glazunova, O.A.; Begunova, A.V.; Fedorova, T.V. 9 Russia and Commonwealth of Independent States (CIS) Domestic Fermented Milk Products; De Gruyter: Berlin, Germany, 2020; pp. 215–234. [Google Scholar]
- Chelule, P.K.; Mokoena, M.P.; Gqaleni, N. Advantages of traditional lactic acid bacteria fermentation of food in Africa. In Current Research, Technology and Education Topics in Applied Microbiology and Microbial Biotechnology; Mendez-Vilas, A., Ed.; Formatex: Badajoz, Spain, 2010; pp. 1160–1167. [Google Scholar]
- Ignat, M.V.; Salanţă, L.C.; Pop, O.L.; Pop, C.R.; Tofană, M.; Mudura, E.; Coldea, T.E.; Borşa, A.; Pasqualone, A. Current functionality and potential improvements of non-alcoholic fermented cereal beverages. Foods 2020, 9, 1031. [Google Scholar] [CrossRef]
- Maleke, M.S.; Adefisoye, M.A.; Doorsamy, W.; Adebo, O.A. Processing, nutritional composition and microbiology of amasi: A Southern African fermented milk product. Sci. Afr. 2021, 12, e00795. [Google Scholar] [CrossRef]
- Holzapfel, W.; Taljaard, J.L. Industrialization of Mageu Fermentation in South Africa. In Industrialization of Indigenous Fermented Foods; Steinkraus, K., Ed.; CRC Press: Boca Raton, FL, USA, 2004. [Google Scholar]
- Moyane, N.; Jideani, A.I.O. The physicochemical and sensory evaluation of commercial sour milk (amasi) products. African J. Food Sci. 2013, 7, 56–62. [Google Scholar] [CrossRef]
- Mashau, M.E.; Jideani, A.I.O.; Maliwichi, L.L. Evaluation of the shelf-life extension and sensory properties of mahewu–A non-alcoholic fermented beverage by adding Aloe vera (Aloe barbadensis) powder. Br. Food J. 2020, 122, 3419–3432. [Google Scholar] [CrossRef]
- Farag, M.A.; Jomaa, S.A.; El-Wahed, A.A.; El-Seedi, A.H.R. The Many Faces of Kefir Fermented Dairy Products: Quality Characteristics, Flavour Chemistry, Nutritional Value, Health Benefits, and Safety. Nutrients 2020, 12, 346. [Google Scholar] [CrossRef] [Green Version]
- Chrysochou, P.; Askegaard, S.; Grunert, K.G.; Kristensen, D.B. Social discourses of healthy eating. A market segmentation approach. Appetite 2010, 55, 288–297. [Google Scholar] [CrossRef]
- Chen, L.A.; House, L. Food lifestyle patterns among contemporary food shoppers. Int. J. Consum. Stud. 2021, 12739. [Google Scholar] [CrossRef]
- Romaniw, O.C.; Rajpal, R.; Duncan, A.M.; Keller, H.H.; Duizer, L.M. Nutrition in Disguise: Effects of Food Neophobia, Healthy Eating Interests and Provision of Health Information on Liking and Perceptions of Nutrient-Dense Foods in Older Adults. Foods 2020, 10, 60. [Google Scholar] [CrossRef] [PubMed]
- Herrero, M.; Simo, C.; Garcia-Cañas, V.; Ibanez, E.; Cifuentes, A. Foodomics: MS-based strategies in modern food science and nutrition. Mass Spectrom. Rev. 2011, 31, 49–69. [Google Scholar] [CrossRef] [Green Version]
- El Sohaimy, S. Functional foods and nutraceuticals-modern approach to food science. World Appl. Sci. J. 2012, 20, 691–708. [Google Scholar]
- Adebo, O.A.; Njobeh, P.B.; Adebiyi, J.A.; Gbashi, S.; Kayitesi, E. Food Metabolomics: A New Frontier in Food Analysis and its Application to Understanding Fermented Foods. In Functional Food—Improve Health through Adequate Food; IntechOpen: London, UK, 2017. [Google Scholar]
- Melini, F.; Melini, V.; Luziatelli, F.; Ficca, A.G.; Ruzzi, M. Health-Promoting Components in Fermented Foods: An Up-to-Date Systematic Review. Nutrients 2019, 11, 1189. [Google Scholar] [CrossRef] [Green Version]
- Cheng, H. Volatile Flavor Compounds in Yogurt: A Review. Crit. Rev. Food Sci. Nutr. 2010, 50, 938–950. [Google Scholar] [CrossRef] [PubMed]
- Spence, C. What Is the Relationship between the Presence of Volatile Organic Compounds in Food and Drink Products and Multisensory Flavour Perception? Foods 2021, 10, 1570. [Google Scholar] [CrossRef] [PubMed]
- Ahn, Y.-Y.; Ahnert, S.E.; Bagrow, J.; Barabasi, A. Flavor network and the principles of food pairing. Sci. Rep. 2011, 1, 196. [Google Scholar] [CrossRef] [PubMed]
- Spence, C. Food and beverage flavour pairing: A critical review of the literature. Food Res. Int. 2020, 133, 109124. [Google Scholar] [CrossRef] [PubMed]
- Begunova, A.; Rozhkova, I.; Glazunova, O.; Moiseenko, K.; Savinova, O.; Fedorova, T. Fermentation Profile and Probiotic-Related Characteristics of Bifidobacterium longum MC-42. Ferment 2021, 7, 101. [Google Scholar] [CrossRef]
- Chen, J.; Liu, H. Nutritional Indices for Assessing Fatty Acids: A Mini-Review. Int. J. Mol. Sci. 2020, 21, 5695. [Google Scholar] [CrossRef]
- Garg, N.; Sethupathy, A.; Tuwani, R.; Nk, R.; Dokania, S.; Iyer, A.; Gupta, A.; Agrawal, S.; Singh, N.; Shukla, S.; et al. FlavorDB: A database of flavor molecules. Nucleic Acids Res. 2018, 46, D1210–D1216. [Google Scholar] [CrossRef]
- Foodpairing Team How to Interpret an Aroma Profile. Available online: https://www.foodpairing.com/how-to-interpret-an-aroma-profile/ (accessed on 18 September 2021).
- Brown, L.; Pingitore, E.; Mozzi, F.; Saavedra, L.; Villegas, J.M.; Hebert, E.M. Lactic Acid Bacteria as Cell Factories for the Generation of Bioactive Peptides. Protein Pept. Lett. 2017, 24, 146–155. [Google Scholar] [CrossRef] [PubMed]
- Séverin, S.; Wenshui, X. Milk Biologically Active Components as Nutraceuticals: Review. Crit. Rev. Food Sci. Nutr. 2005, 45, 645–656. [Google Scholar] [CrossRef]
- Li, G.; Liu, W.; Wang, Y.; Jia, F.; Wang, Y.; Ma, Y.; Gu, R.; Lu, J. Functions and Applications of Bioactive Peptides from Corn Gluten Meal. Adv. Food Nutr. Res. 2019, 87, 1–41. [Google Scholar] [CrossRef]
- Udenigwe, C.; Aluko, R.E. Food Protein-Derived Bioactive Peptides: Production, Processing, and Potential Health Benefits. J. Food Sci. 2011, 77, R11–R24. [Google Scholar] [CrossRef]
- Rosa, D.D.; Dias, M.M.; Grześkowiak, Ł.M.; Reis, S.A.; Conceição, L.L.; Maria do Carmo, G.P. Milk kefir: Nutritional, microbiological and health benefits. Nutr. Res. Rev. 2017, 30, 82–96. [Google Scholar] [CrossRef]
- Bansal, S.; Mangal, M.; Sharma, S.K.; Gupta, R.K. Non-dairy Based Probiotics: A Healthy Treat for Intestine. Crit. Rev. Food Sci. Nutr. 2014, 56, 1856–1867. [Google Scholar] [CrossRef]
- Du Plooy, Z.; Schönfeldt, H.; Hall, N. The role of traditional foods in food-based dietary guidelines—A South African case study on maas (cultured milk). Food Chem. 2018, 238, 22–28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gómez-Cortés, P.; Juárez, M.; de la Fuente, M.A. Milk fatty acids and potential health benefits: An updated vision. Trends Food Sci. Technol. 2018, 81, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Calder, P.C. Functional Roles of Fatty Acids and Their Effects on Human Health. J. Parenter. Enter. Nutr. 2015, 39 (Suppl. S1), 18S–32S. [Google Scholar] [CrossRef] [PubMed]
- Williams, C.M. Dietary fatty acids and human health. Anim. Res. 2000, 49, 165–180. [Google Scholar] [CrossRef] [Green Version]
- Vieira, C.P.; Alvares, T.S.; Gomes, L.S.; Torres, A.; Paschoalin, V.; Conte-Junior, C.A. Kefir Grains Change Fatty Acid Profile of Milk during Fermentation and Storage. PLoS ONE 2015, 10, e0139910. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.-H.; Jeong, D.; Song, K.-Y.; Seo, K.-H. Comparison of traditional and backslopping methods for kefir fermentation based on physicochemical and microbiological characteristics. LWT 2018, 97, 503–507. [Google Scholar] [CrossRef]
- Jenske, R.; Vetter, W. Enantioselective Analysis of 2- and 3-Hydroxy Fatty Acids in Food Samples. J. Agric. Food Chem. 2008, 56, 11578–11583. [Google Scholar] [CrossRef] [PubMed]
- Jenske, R.; Vetter, W. Concentrations of medium-chain 2- and 3-hydroxy fatty acids in foodstuffs. Food Chem. 2009, 114, 1122–1129. [Google Scholar] [CrossRef]
- Abdoul-Aziz, S.K.A.; Zhang, Y.; Wang, J. Milk Odd and Branched Chain Fatty Acids in Dairy Cows: A Review on Dietary Factors and Its Consequences on Human Health. Animals 2021, 11, 3210. [Google Scholar] [CrossRef]
- Taormina, V.M.; Unger, A.L.; Schiksnis, M.R.; Torres-Gonzalez, M.; Kraft, J. Branched-Chain Fatty Acids—An Underexplored Class of Dairy-Derived Fatty Acids. Nutrients 2020, 12, 2875. [Google Scholar] [CrossRef]
- Blandino, A.; Al-Aseeri, M.E.; Pandiella, S.S.; Cantero, D.; Webb, C. Cereal-based fermented foods and beverages. Food Res. Int. 2003, 36, 527–543. [Google Scholar] [CrossRef]
- Dan, T.; Wang, D.; Wu, S.; Jin, R.; Ren, W.; Sun, T. Profiles of Volatile Flavor Compounds in Milk Fermented with Different Proportional Combinations of Lactobacillus delbrueckii subsp. bulgaricus and Streptococcus thermophilus. Molecules 2017, 22, 1633. [Google Scholar] [CrossRef] [Green Version]
- Dan, T.; Wang, D.; Jin, R.; Zhang, H.; Zhou, T.; Sun, T. Characterization of volatile compounds in fermented milk using solid-phase microextraction methods coupled with gas chromatography-mass spectrometry. J. Dairy Sci. 2017, 100, 2488–2500. [Google Scholar] [CrossRef] [Green Version]
- Chen, C.; Zhao, S.; Hao, G.; Yu, H.; Tian, H.; Zhao, G. Role of lactic acid bacteria on the yogurt flavour: A review. Int. J. Food Prop. 2017, 20, S316–S330. [Google Scholar] [CrossRef] [Green Version]
- Preininger, M.; Gimelfarb, L.; Li, H.-C.; Dias, B.E.; Fahmy, F.; White, J. Identification of Dihydromaltol (2,3-Dihydro-5-hydroxy-6-methyl-4H-pyran-4-one) in Ryazhenka Kefir and Comparative Sensory Impact Assessment of Related Cycloenolones. J. Agric. Food Chem. 2009, 57, 9902–9908. [Google Scholar] [CrossRef]
- Dragone, G.; Mussatto, S.I.; Oliveira, J.; Teixeira, J.A. Characterisation of volatile compounds in an alcoholic beverage produced by whey fermentation. Food Chem. 2009, 112, 929–935. [Google Scholar] [CrossRef] [Green Version]
- Dertli, E.; Çon, A.H. Microbial diversity of traditional kefir grains and their role on kefir aroma. LWT 2017, 85, 151–157. [Google Scholar] [CrossRef]
- Wang, H.; Sun, X.; Song, X.; Guo, M. Effects of kefir grains from different origins on proteolysis and volatile profile of goat milk kefir. Food Chem. 2021, 339, 128099. [Google Scholar] [CrossRef]
- Xiang, J.; Liu, F.; Wang, B.; Chen, L.; Liu, W.; Tan, S. A Literature Review on Maillard Reaction Based on Milk Proteins and Carbohydrates in Food and Pharmaceutical Products: Advantages, Disadvantages, and Avoidance Strategies. Foods 2021, 10, 1998. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.-Q.; Holland, R.; Crow, V.L. Esters and their biosynthesis in fermented dairy products: A review. Int. Dairy J. 2004, 14, 923–945. [Google Scholar] [CrossRef]
| Product Name, Type of Packaging, Net Weight | Manufacturer, Address | Nutritional Information per 100 g of Product | Ingredients: |
|---|---|---|---|
| «Ruzskyi Kefir» fat 3.2–4.0%. Hermetically packed in a Pure-Pak, m = 1000 g | JSC “RUZKE MILK”, Russia, Moscow Region. | Fat—3.8 g Protein—3.0 g Carbohydrate—4.0 g Energy—from 238 to 268 kJ/ from 57 to 64 kcal | Whole milk, culture on kefir grains. The amount of yeast at the end of the shelf life is at least 104 CFU·g–1. The number of lactic acid microorganisms is not less than 107 CFU·g–1. |
| Kefir «Asenievskaya ferma» fat 3.2%. Hermetically packed in a polypropylene bottle, m = 450 g | Agricultural production cooperative “Agricultural artel (collective farm) “Pervomaisky”. Russia, Kaluga region. | Fat—3.2 g Protein—3.0 g Carbohydrate—4.0 g Energy—238 kJ/57 kcal | Normalized milk, culture on kefir grains. The number of lactic acid producing microorganisms is not less than 107 CFU·g–1. The amount of yeast at the end of the shelf life is at least 104 CFU·g–1. |
| Kefir «Molochnaya kultura» fat from 3.5 to 4.5%. Hermetically packed in a polypropylene glass, m = 500 g | Milk Culture LLC, Russia, Leningrad Region. | Fat—4.1 g Protein—3.0 g Carbohydrate—4.0 g Energy—60–70 kcal/ 250–290 kJ | Whole milk, culture on kefir grains. The number of lactic acid producing microorganisms is not less than 1 × 107 CFU·g–1. The amount of yeast at the end of the shelf life is at least 1 × 104 CFU·g–1. |
| Ryazhenka «Ruzskaya» fat 2.5%. Hermetically packed in a Pure-Pak. m = 330 g | JSC “RUZKE MILK”, Russia, Moscow Region. | Fat—2.5 g Protein—3.0 g Carbohydrate—4.0 g Energy—from 211 kJ/50 kcal | Normalized baked milk, fermentation of lactic acid producing microorganisms. The number of lactic acid microorganisms is not less than 1 × 107 CFU·g–1. |
| Ryazhenka «Molochnaya kultura» fat from 3.5 to 4.5%. Hermetically packed in a polypropylene glass, m = 500 g | Milk Culture LLC, Russia, Leningrad Region. | Fat—3.8 gProtein—3.0 g Carbohydrate—4.1 g Energy—60–70 kcal/ 250–290 kJ | Whole milk, fermentation of lactic acid producing microorganisms. The number of lactic acid microorganisms is not less 1 × 107 CFU·g–1. |
| «Full cream MAAS PASTEURISED» fat 3.7%. Hermetically packed in a polypropylene bottle, m = 500 g | Specially packaged for Pick n Pay Ltd. «PnP», South Africa, Kensington. | Fat—3.7 g Protein—3.3 g Carbohydrate—5.0 g Energy—270 kJ | Full Cream milk, Starter Culture. |
| Full cream MAAS «AMASI OTHANDO» fat 3.3%. Hermetically packed in a Pure-Pak, m = 500 g | Clover S.A. (PTY) LTD Clover Park, South Africa, Roodepoort. | Fat—3.3 g Protein—3.3 g Carbohydrate—4.0 g Energy—241 kJ | Milk and/or Recombined Milk, Culture. |
| Full cream MAAS «INKOMAZI Rich and Creamy» fat 3.4%. Hermetically packed in a cardboard bag (Nampak Liquid Cartons), m = 500 g | Manufactured for Danone Southern Africa (PTY) LTD, 99 Skew Road, Boksburg North, 1459, South Africa | Fat—3.4 g Protein—3.2 g Carbohydrate—5.0 g Energy—265 kJ | Full fat milk, MAAS cultures. |
| «MNANDI Amahewu Creamy flavour» fat < 0.1%. Hermetically packed in a Pure-Pak, m = 1 L | «RcL Foods», South Africa | Fat < 0.1 g Protein—0.7 g Carbohydrate—6.0 g Energy—122 kJ | Water, Maize (7%), Lactic Acid producing culture, Sugar, Flavouring, Preservative (Potassium Sorbate), Non- nutritive sweeteners (Sodium cyclamate, Sodium saccharin, Aspartame), Contains Phenylalanine. |
| Product | pH | ORAC, µmol(TE)·mL−1 | ACE-I Activity (IC50), mg(Protein)·mL−1 | mM(Leu) |
|---|---|---|---|---|
| Kefir | 4.44 ± 0.03 | 430 a ± 92 | 2.75 a ± 0.61 | 5.71 a ± 0.56 |
| Ryazhenka | 4.46 ± 0.06 | 414 a ± 43 | 3.94 a ± 1.92 | 3.72 b ± 0.67 |
| Amasi | 4.47 ± 0.04 | 682 b ± 82 | 1.72 b ± 0.39 | 8.32 c ± 0.72 |
| Mahewu | 3.54 ± 0.02 | 326 c ± 52 | 0.92 c ± 0.67 | 0.68 d ± 0.12 |
| Fatty Acid | Relative Abundance, % | ||||||
|---|---|---|---|---|---|---|---|
| Name | Abbreviation | Kefir | Ryazhenka | Amasi | |||
| Mean | SD | Mean | SD | Mean | SD | ||
| Saturated Fatty Acids (SFA) | |||||||
| Butanoic acid | C4:0 | 1.82 a | 0.054 | 1.64 b | 0.092 | ND | - |
| Pentanoic acid | C5:0 | 0.04 | 0.011 | 0.03 | 0.009 | ND | - |
| Hexanoic acid | C6:0 | 1.77 | 0.126 | 1.65 | 1.002 | 1.81 | 0.254 |
| Heptanoic acid | C7:0 | 0.05 | 0.017 | 0.04 | 0.010 | ND | - |
| Octanoic acid | C8:0 | 1.46 a | 0.025 | 1.30 b | 0.102 | 1.49 a | 0.132 |
| Nonanoic acid | C9:0 | 0.07 | 0.008 | 0.05 | 0.003 | ND | - |
| Decanoic acid | C10:0 | 3.63 a | 0.074 | 3.26 b | 0.021 | 4.00 c | 0.082 |
| Undecanoic acid | C11:0 | 0.13 | 0.014 | 0.09 | 0.012 | 0.08 | 0.008 |
| Dodecanoic acid | C12:0 | 4.41 a | 0.220 | 3.91 b | 0.081 | 4.07 c | 0.051 |
| Tridecanoic acid | C13:0 | 0.16 | 0.022 | 0.14 | 0.021 | 0.09 | 0.030 |
| Tetradecanoic acid | C14:0 | 10.48 | 0.682 | 10.84 | 0.321 | 10.38 | 0.260 |
| Pentadecanoic acid | C15:0 | 1.50 a | 0.041 | 1.61 b | 0.021 | 1.22 c | 0.018 |
| Hexadecanoic acid | C16:0 | 25.07 a | 0.173 | 24.32 b | 0.210 | 25.66 c | 0.050 |
| Heptadecanoic acid | C17:0 | 0.63 | 0.010 | 1.24 | 0.021 | 0.52 | 0.010 |
| Octadecanoic acid | C18:0 | 11.90 a | 0.193 | 13.53 b | 0.224 | 15.83 c | 0.150 |
| Total SFA | 63.11 a | 0.780 | 63.65 a | 1.109 | 65.15 b | 0.430 | |
| Monounsaturated fatty acids (MUFA) | |||||||
| 4-Decenoic acid | C10:1 (n − 6) | 0.38 | 0.021 | 0.34 | 0.008 | 0.35 | 0.006 |
| 9-Tetradecenoic acid | C14:1 (n − 5) | 1.21 | 0.012 | 1.16 | 0.026 | 0.85 | 0.041 |
| 9-Hexadecenoic acid | C16:1 (n − 7) | 1.92 | 0.081 | 1.72 | 0.102 | 1.28 | 0.026 |
| 9-Octadecenoic acid | C18:1 (n − 9) | 20.45 a | 0.410 | 21.72 b | 0.320 | 28.53 c | 0.970 |
| 12-Octadecenoic acid | C18:1 (n − 6) | 6.06 | 1.021 | 5.21 | 0.524 | ND | - |
| Total MUFA | 30.02 | 1.103 | 30.15 | 0.623 | 31.02 | 0.971 | |
| Polyunsaturated fatty acids (PUFA) | |||||||
| 9,12-Octadecadienoic acid | C18:2 (n − 6) | 4.04 a | 0.140 | 3.50 b | 0.250 | 2.04 c | 0.134 |
| 10,12-Octadecadienoic acid | C18:2 (n − 6) | 1.06 a | 0.014 | 1.29 b | 0.008 | ND | - |
| 8,11,14-Eicosatrienoic acid | C20:3 (n − 6) | 0.23 | 0.023 | ND | - | ND | - |
| 5,8,11,14-Eicosatetraenoic acid | C20:4 (n − 6) | 0.19 | 0.025 | 0.16 | 0.016 | ND | - |
| Total PUFA | 5.53 a | 0.145 | 4.96 b | 0.251 | 2.04 c | 0.134 | |
| Branched chain fatty acids (BCFA) | |||||||
| Tridecanoic acid, 12-methyl | iso-C14:0 | 0.26 a | 0.021 | 0.33 b | 0.024 | ND | - |
| Tetradecanoic acid, 12-methyl | anteiso-C15:0 | 0.45 a | 0.010 | 0.53 b | 0.016 | 0.59 c | 0.023 |
| 14-methylhexadecanoic acid | anteiso-C17:0 | ND | - | ND | - | 0.57 | 0.071 |
| 15-methylhexadecanoic acid | iso-C17:0 | 0.45 a | 0.010 | 0.34 b | 0.016 | 0.62 c | 0.052 |
| Total BCFA | 1.17 a | 0.025 | 1.19 a | 0.033 | 1.79 b | 0.091 | |
| 2-hydroxy fatty acids (2OH-FA) | |||||||
| Hexanoic acid, 2-hydroxy | 2OH-C6:0 | 0.08 a | 0.007 | 0.05 b | 0.002 | ND | - |
| Butyric acid, 2-hydroxy-3-methyl | 2OH-iso-C5:0 | 0.03 | 0.001 | ND | - | ND | - |
| Pentanoic acid, 2-hydroxy-3-methyl | 2OH-anteiso-C6:0 | 0.06 | 0.005 | ND | - | ND | - |
| Total 2OH-FA | 0.17 a | 0.009 | 0.05 b | 0.002 | ND | - | |
| Index | Kefir | Ryazhenka | Amasi | |||
|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | |
| PUFA/SFA | 0.088 a | 0.003 | 0.078 a | 0.004 | 0.031 b | 0.002 |
| IA | 2.008 a | 0.021 | 2.039 a | 0.011 | 2.155 b | 0.008 |
| HPI | 0.498 a | 0.005 | 0.490 a | 0.003 | 0.464 b | 0.002 |
| IT | 2.670 a | 0.172 | 2.774 a | 0.109 | 3.138 b | 0.187 |
| HH | 0.802 a | 0.031 | 0.816 a | 0.019 | 0.762 b | 0.025 |
| UI | 41.686 a | 1.113 | 40.391 a | 0.672 | 35.096 b | 0.980 |
| IUPAC Name | Chemical Class | Concise Flavor Description | Relative Abundance, % |
|---|---|---|---|
| Kefir | |||
| 3-methylbutan-1-ol | Alcohol | Chemical—alcoholic | 35.75 |
| ethanol | Alcohol | Chemical—alcoholic | 16.41 |
| acetic acid | Acid | Dairy—acidic | 14.11 |
| butane-2,3-diol | Alcohol | Dairy—buttery | 9.87 |
| hexanoic acid | Acid | Animal—animal | 8.89 |
| octanoic acid | Acid | Animal—animal | 7.24 |
| 2,7-dimethyloctane-4,5-diol | Alcohol | Fruity—sweet | 4.31 |
| decanoic acid | Acid | Animal—animal | 1.59 |
| hexan-1-ol | Alcohol | Chemical—alcoholic | 1.11 |
| octan-2-one | Ketone | Fruity—apple | 0.72 |
| Ryazhenka | |||
| heptan-2-one | Ketone | Fruity—apple | 41.64 |
| nonan-2-one | Ketone | Herbal—ruta | 16.55 |
| hexanoic acid | Acid | Animal—animal | 11.23 |
| ethyl methyl carbonate | Ester | Fruity—sweet | 6.49 |
| octanoic acid | Acid | Animal—animal | 5.57 |
| undecan-2-one | Ketone | Green—fatty/waxy | 3.33 |
| butanoic acid | Acid | Dairy—buttery | 3.17 |
| hexadecan-1-ol | Alcohol | Green—fatty/waxy | 2.95 |
| furan-2-ylmethanol | Aromatic alcohol | Roasted—bread | 2.77 |
| octadecanoic acid | Acid | Green—Fatty/Waxy | 2.48 |
| decanoic acid | Acid | Animal—animal | 1.86 |
| nonanoic acid | Acid | Dairy—buttery | 0.91 |
| tetradecan-2-one | Ketone | Unknown | 0.65 |
| hexadecanoic acid | Acid | Green—fatty/waxy | 0.39 |
| Amasi | |||
| 3-hydroxybutan-2-one (acetoin) | Ketone/Alcohol | Dairy—buttery | 64.44 |
| 3-hydroxypropanoic acid | Acid | Dairy—acidic | 16.90 |
| hexanoic acid | Acid | Animal—animal | 6.76 |
| octanoic acid | Acid | Animal—animal | 3.34 |
| hexadecan-1-ol | Alcohol | Green—fatty/waxy | 2.74 |
| octadecanoic acid | Acid | Green—Fatty/Waxy | 2.45 |
| dec-1-ene | Alkene | Unknown | 1.90 |
| hexadecanoic acid | Acid | Green—fatty/waxy | 0.89 |
| decanoic acid | Acid | Animal—animal | 0.59 |
| Mahewu | |||
| pentyl acetate | Ester | Fruity—tropical | 20.20 |
| 2-hydroxypropanamide | Amide | Unknown | 20.04 |
| hexadecanoic acid | Acid | Green—fatty/waxy | 15.16 |
| prop-2-enyl hexanoate | Ester | Fruity—tropical | 6.85 |
| ethyl methyl carbonate | Ester | Fruity—sweet | 5.95 |
| pentyl 2-methylpropanoate | Ester | Fruity—sweet | 5.44 |
| tetradecanoic acid | Acid | Green–fatty/waxy | 5.36 |
| (E)-octadec-11-enoic acid | Acid | Unknown | 5.21 |
| pentyl butanoate | Ester | Fruity—tropical | 4.64 |
| ethyl butanoate | Ester | Fruity—tropical | 2.98 |
| ethyl nonanoate | Ester | Fruity—tropical | 2.76 |
| hexanoic acid | Acid | Animal—animal | 2.27 |
| (E)-hex-2-enoic acid | Acid | Animal—animal | 1.77 |
| pentyl 2-methylbutanoate | Ester | Fruity—apple | 1.38 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moiseenko, K.V.; Glazunova, O.A.; Savinova, O.S.; Ajibade, B.O.; Ijabadeniyi, O.A.; Fedorova, T.V. Analytical Characterization of the Widely Consumed Commercialized Fermented Beverages from Russia (Kefir and Ryazhenka) and South Africa (Amasi and Mahewu): Potential Functional Properties and Profiles of Volatile Organic Compounds. Foods 2021, 10, 3082. https://doi.org/10.3390/foods10123082
Moiseenko KV, Glazunova OA, Savinova OS, Ajibade BO, Ijabadeniyi OA, Fedorova TV. Analytical Characterization of the Widely Consumed Commercialized Fermented Beverages from Russia (Kefir and Ryazhenka) and South Africa (Amasi and Mahewu): Potential Functional Properties and Profiles of Volatile Organic Compounds. Foods. 2021; 10(12):3082. https://doi.org/10.3390/foods10123082
Chicago/Turabian StyleMoiseenko, Konstantin V., Olga A. Glazunova, Olga S. Savinova, Betty O. Ajibade, Oluwatosin A. Ijabadeniyi, and Tatyana V. Fedorova. 2021. "Analytical Characterization of the Widely Consumed Commercialized Fermented Beverages from Russia (Kefir and Ryazhenka) and South Africa (Amasi and Mahewu): Potential Functional Properties and Profiles of Volatile Organic Compounds" Foods 10, no. 12: 3082. https://doi.org/10.3390/foods10123082
APA StyleMoiseenko, K. V., Glazunova, O. A., Savinova, O. S., Ajibade, B. O., Ijabadeniyi, O. A., & Fedorova, T. V. (2021). Analytical Characterization of the Widely Consumed Commercialized Fermented Beverages from Russia (Kefir and Ryazhenka) and South Africa (Amasi and Mahewu): Potential Functional Properties and Profiles of Volatile Organic Compounds. Foods, 10(12), 3082. https://doi.org/10.3390/foods10123082







