The Investigation of Mycotoxins and Enterobacteriaceae of Cereal-Based Baby Foods Marketed in Turkey
Abstract
:1. Introduction
2. Materials and Methods
2.1. Samples Collection
2.2. Sample Preparation
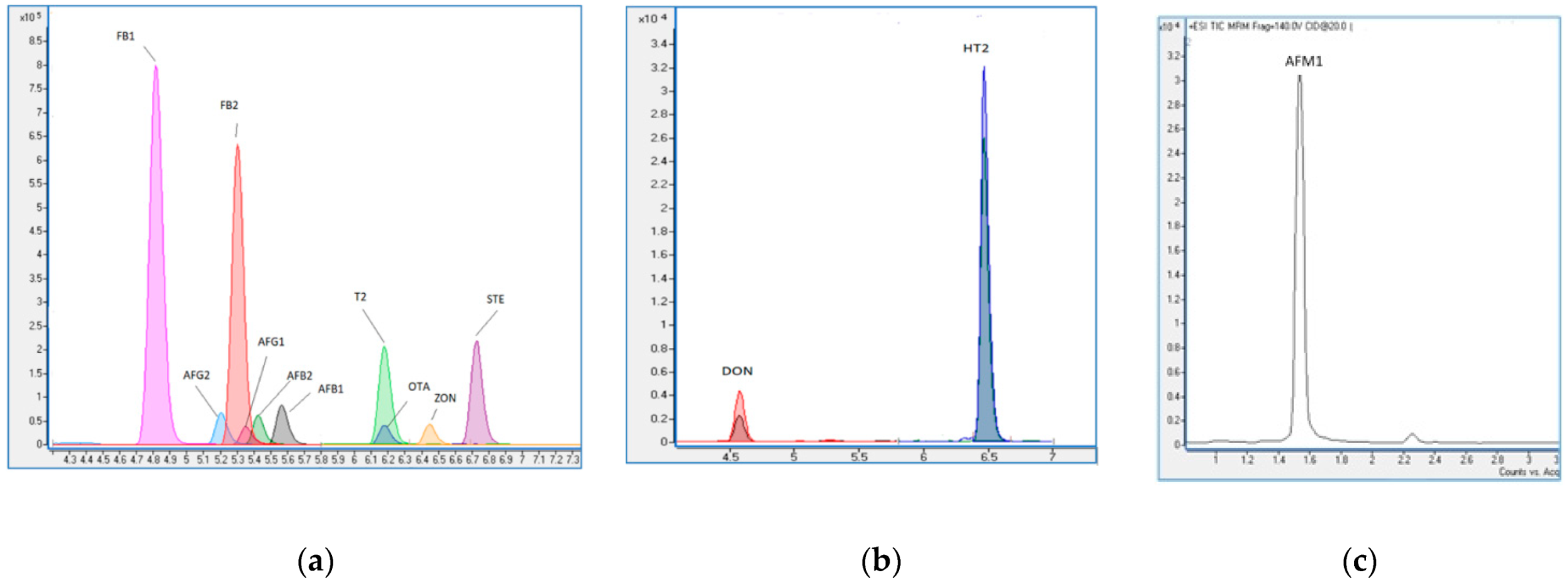
2.3. LC–MS/MS Procedure
2.4. Estimation of Dietary Intake of Mycotoxins
2.5. Bacteriological Analysis
2.6. Statistics
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Redmond, E.C.; Griffith, C.J. The importance of hygiene in the domestic kitchen: Implications for preparation and storage of food and infant formula. Perspect. Public Health 2009, 129, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Kurtz, A.; Thomopoulos, R. Safety vs. Sustainability concerns of infant food users: French results and european perspectives. Sustainability 2021, 13, 10074. [Google Scholar] [CrossRef]
- Portela, J.B.; Coimbra, P.T.; Cappato, L.P.; Alvarenga, V.O.; Oliveira, R.B.; Pereira, K.S.; Azeredo, D.R.P.; Sant’Ana, A.S.; Nascimento, J.S.; Cruz, A.G. Predictive model for inactivation of Salmonella in infant formula during microwave heating processing. Food Control 2019, 104, 308–312. [Google Scholar] [CrossRef]
- Wang, X.; Meng, J.; Zhang, J.; Zhou, T.; Zhang, Y.; Yang, B.; Xi, M.; Xia, X. Characterization of Staphylococcus aureus isolated from powdered infant formula milk and infant rice cereal in China. Int. J. Food Microbiol. 2012, 153, 142–147. [Google Scholar] [CrossRef]
- Francisquini, J.D.A.; Nunes, L.; Martins, E.; Stephani, R.; Perrone, Í.T.; Carvalho, A.F.D. How the heat treatment affects the constituents of infant formulas: A review. Braz. J. Food Technol. 2020, 23, e2019272. [Google Scholar] [CrossRef]
- Briefel, R.R.; Reidy, K.; Karwe, V.; Devaney, B. Feeding infants and toddlers study: Improvements needed in meeting infant feeding recommendations. J. Am. Diet Assoc. 2004, 104 (Suppl. 1), 31–37. [Google Scholar] [CrossRef] [PubMed]
- Er Demirhan, B.; Demirhan, B.; Yentür, G. Importance of aflatoxins presence in baby foods. Gazi Sağlık Bilim. Derg. 2017, 2, 01–08. (In Turkish) [Google Scholar]
- Buchanan, R.L.; Oni, R. Use of microbiological indicators for assessing hygiene controls for the manufacture of powdered infant formula. J. Food Prot. 2012, 75, 989–997. [Google Scholar] [CrossRef]
- Aidoo, K.E.; Mohamed, S.M.; Candlish, A.A.; Tester, R.F.; Elgerbi, A.M. Occurrence of fungi and mycotoxins in some commercial baby foods in North Africa. Food Nutr. Sci. 2011, 2, 751–758. [Google Scholar] [CrossRef] [Green Version]
- Tonon, K.M.; Savi, G.D.; Scussel, V.M. Application of a LC–MS/MS method for multi-mycotoxin analysis in infant formula and milk-based products for young children commercialized in Southern Brazil. J. Environ. Sci. Health B 2018, 53, 685–691. [Google Scholar] [CrossRef] [PubMed]
- Hernández, M.; Juan-García, A.; Moltó, J.C.; Mañes, J.; Juan, C. Evaluation of mycotoxins in infant breast milk and infant food, reviewing the literature data. Toxins 2021, 13, 535. [Google Scholar] [CrossRef] [PubMed]
- Bullerman, L.B.; Bianchini, A. Stability of mycotoxins during food processing. Int. J. Food Microbiol. 2007, 119, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Sherif, S.O.; Salama, E.E.; Abdel-Wahhab, M.A. Mycotoxins and child health: The need for health risk assessment. Int. J. Hyg. Environ. Health 2009, 212, 347–368. [Google Scholar] [CrossRef] [PubMed]
- Beltrán, E.; Ibáñez, M.; Sancho, J.V.; Cortés, M.Á.; Yusà, V.; Hernández, F. UHPLC–MS/MS highly sensitive determination of aflatoxins, the aflatoxin metabolite M1 and ochratoxin A in baby food and milk. Food Chem. 2011, 126, 737–744. [Google Scholar] [CrossRef]
- Yentür, G.; Er, B. The evaluation of the aflatoxin presence in foods. Turk Hij. Den. Biyol. Derg. 2012, 69, 41–52. (In Turkish) [Google Scholar] [CrossRef]
- Yentür, G.; Er, B.; Özkan, M.G.; Öktem, A.B. Determination of aflatoxins in peanut butter and sesame samples using high-performance liquid chromatography method. Eur. Food Res. Technol. 2006, 224, 167–170. [Google Scholar] [CrossRef]
- Yentür, G.; Onurdağ, F.K.; Er, B.; Demirhan, B. Investigation of aflatoxin B1 levels in red pepper and products consumed in Ankara. Turk. J. Pharm. Sci. 2012, 9, 293–300. [Google Scholar]
- Juan, C.; Raiola, A.; Mañes, J.; Ritieni, A. Presence of mycotoxin in commercial infant formulas and baby foods from Italian market. Food Control 2014, 39, 227–236. [Google Scholar] [CrossRef]
- Min, L.; Fink-Gremmels, J.; Li, D.; Tong, X.; Tang, J.; Nan, X.; Yu, Z.; Chen, W.; Wang, G. An overview of aflatoxin B1 biotransformation and aflatoxin M1 secretion in lactating dairy cows. Anim. Nutr. 2021, 7, 42–48. [Google Scholar] [CrossRef]
- Songsermsakul, P.; Razzazi-Fazeli, E. A review of recent trends in applications of liquid chromatography-mass spectrometry for determination of mycotoxins. J. Liq. Chromatogr. Relat. Technol. 2008, 31, 1641–1686. [Google Scholar] [CrossRef]
- Xu, L.; Zhang, Z.; Zhang, Q.; Li, P. Mycotoxin determination in foods using advanced sensors based on antibodies or aptamers. Toxins 2016, 8, 239. [Google Scholar] [CrossRef] [Green Version]
- Cappozzo, J.; Jackson, L.; Lee, H.J.; Zhou, W.; Al-Taher, F.; Zweigenbaum, J.; Ryu, D. Occurrence of ochratoxin A in infant foods in the United States. J. Food Prot. 2017, 80, 251–256. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Leal, M.R.; Míguez, M.P.; Fernández-Franzón, M. Detection of seven trichothecene mycotoxins in infant cereal foods by QuEChERS extraction and liquid chromatography coupled to tandem mass spectrometry. Rev. Toxicol. 2013, 30, 203–208. [Google Scholar]
- Rai, A.; Das, M.; Tripathi, A. Occurrence and toxicity of a fusarium mycotoxin, zearalenone. Crit. Rev. Food Sci. Nutr. 2020, 60, 2710–2729. [Google Scholar] [CrossRef] [PubMed]
- Alvito, P.C.; Sizoo, E.A.; Almeida, C.M.M.; Egmond, H.P.V. Occurrence of aflatoxins and ochratoxin A in baby foods in Portugal. Food Anal. Methods 2010, 3, 22–30. [Google Scholar] [CrossRef]
- Jasem Kit. Food Safety & Quality. 2021. Available online: https://jasem.com.tr/en/products/food-safety-quality/ (accessed on 9 November 2021).
- Piacentini, K.C.; Ferranti, L.S.; Pinheiro, M.; Bertozzi, B.G.; Rocha, L.O. Mycotoxin contamination in cereal-based baby foods. Curr. Opin. Food Sci. 2019, 30, 73–78. [Google Scholar] [CrossRef]
- EFSA Scientific Committee. Guidance on selected default values to be used by the EFSA Scientific Committee, Scientific Panels and Units in the absence of actual measured data. EFSA J. 2012, 10, 2579. [Google Scholar]
- Halkman, A.K. Merck Applications of Food Microbiology; Başak Matbaacılık ve Tanıtım Hizmetleri Ltd. Şti.: Ankara, Turkey, 2005; pp. 135–238. [Google Scholar]
- Daniel, N.W. Bioistatistic: A Foundation for Analysis in the Health Sciences; Wiley: New York, NY, USA, 1991; p. 5. [Google Scholar]
- Ishikawa, A.T.; Takabayashi-Yamashita, C.R.; Ono, E.; Bagatin, A.K.; Rigobello, F.F.; Kawamura, O.; Hirooka, E.Y.; Itano, E.N. Exposure assessment of infants to aflatoxin M1 through consumption of breast milk and infant powdered milk in Brazil. Toxins 2016, 8, 246. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oueslati, S.; Berrada, H.; Mañes, J.; Juan, C. Presence of mycotoxins in Tunisian infant foods samples and subsequent risk assessment. Food Control 2018, 84, 362–369. [Google Scholar] [CrossRef]
- Rodríguez-Carrasco, Y.; Ruiz, M.J.; Font, G.; Berrada, H. Exposure estimates to Fusarium mycotoxins through cereals intake. Chemosphere 2013, 93, 2297–2303. [Google Scholar] [CrossRef] [PubMed]
- Adetunji, M.C.; Atanda, O.O.; Ezekiel, C.N. Risk assessment of mycotoxins in stored maize grains consumed by infants and young children in Nigeria. Children 2017, 4, 58. [Google Scholar] [CrossRef] [PubMed]
- Turkish Food Codex. Türk Gıda Kodeksi Bulaşanlar Yönetmeliği. Resmî Gazete Tarihi: 29.12.2011 Resmî Gazete Sayısı: 28157 (3.Mükerrer). Available online: https://www.mevzuat.gov.tr/mevzuat?MevzuatNo=15692&MevzuatTur=7&MevzuatTertip=5 (accessed on 9 November 2021).
- Baydar, T.; Erkekoglu, P.; Sipahi, H.; Sahin, G. Aflatoxin B1, M1 and ochratoxin A levels in infant formulae and baby foods marketed in Ankara, Turkey. J. Food Drug Anal. 2007, 15, 89–92. [Google Scholar] [CrossRef]
- Er, B.; Demirhan, B.; Yentür, G. Investigation of aflatoxin M1 levels in infant follow-on milks and infant formulas sold in the markets of Ankara, Turkey. J. Dairy Sci. 2014, 97, 3328–3331. [Google Scholar] [CrossRef]
- Hampikyan, H.; Bingol, E.B.; Colak, H.; Cetin, O.; Bingol, B. Determination of ochratoxin A in baby foods by ELISA and HPLC. Acta Aliment. 2015, 44, 578–584. [Google Scholar] [CrossRef] [Green Version]
- Kabak, B. Aflatoxin M1 and ochratoxin A in baby formulae in Turkey: Occurrence and safety evaluation. Food Control 2012, 26, 182–187. [Google Scholar] [CrossRef]
- Lombaert, G.A.; Pellaers, P.; Roscoe, V.; Mankotia, M.; Neil, R.; Scott, P.M. Mycotoxins in infant cereal foods from the Canadian retail market. Food Addit. Contam. 2003, 20, 494–504. [Google Scholar] [CrossRef] [PubMed]
- Kalantari, H.; Kalantari, G.H.; Nazari, K.Z. Evaluation of aflatoxins contamination in baby food supplements (Mamana & Ghoncheh). Jundishapur. J. Nat. Pharm. Prod. 2011, 6, 42–50. [Google Scholar]
- Rubert, J.; Soler, C.; Mañes, J. Application of an HPLC–MS/MS method for mycotoxin analysis in commercial baby foods. Food Chem. 2012, 133, 176–183. [Google Scholar] [CrossRef]
- Assunção, R.; Martins, C.; Vasco, E.; Jager, A.; Oliveira, C.; Cunha, S.C.; Fernandes, J.O.; Nunes, B.; Loureiro, S.; Alvito, P. Portuguese children dietary exposure to multiple mycotoxins–an overview of risk assessment under MYCOMIX project. Food Chem. Toxicol. 2018, 118, 399–408. [Google Scholar] [CrossRef]
- Al-Taher, F.; Cappozzo, J.; Zweigenbaum, J.; Lee, H.J.; Jackson, L.; Ryu, D. Detection and quantitation of mycotoxins in infant cereals in the US market by LC-MS/MS using a stable isotope dilution assay. Food Control 2017, 72, 27–35. [Google Scholar] [CrossRef] [Green Version]
- Sezer, C.; Vatansever, L.; Bilge, N. The microbiological quality of infant milk and follow-on formula. Van Vet. J. 2015, 26, 31–34. [Google Scholar]
- Heperkan, D.; Dalkilic-Kaya, G.; Juneja, V.K. Cronobacter sakazakii in baby foods and baby food ingredients of dairy origin and microbiological profile of positive samples. LWT 2017, 75, 402–407. [Google Scholar] [CrossRef]
- Kim, S.A.; Oh, S.W.; Lee, Y.M.; Imm, J.Y.; Hwang, I.G.; Kang, D.H.; Rhee, M.S. Microbial contamination of food products consumed by infants and babies in Korea. Lett. Appl. Microbiol. 2011, 53, 532–538. [Google Scholar] [CrossRef] [PubMed]
- Sani, N.A.; Hartantyo, S.H.P.; Forsythe, S.J. Microbiological assessment and evaluation of rehydration instructions on powdered infant formulas, follow-up formulas, and infant foods in Malaysia. J. Dairy Sci. 2013, 96, 1–8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sadek, Z.I.; Abdel-Rahman, M.A.; Azab, M.S.; Darwesh, O.M.; Hassan, M.S. Microbiological evaluation of infant foods quality and molecular detection of Bacillus cereus toxins relating genes. Toxicol. Rep. 2018, 5, 871–877. [Google Scholar] [CrossRef] [PubMed]
| Mycotoxins | R2 | LOD | LOQ | RSD (%) | % REC. |
|---|---|---|---|---|---|
| AFB1 | 0.997 | 0.01 | 0.02 | 7.27 | 98.11 |
| AFB2 | 0.998 | 0.02 | 0.07 | 4.9 | 102 |
| AFG1 | 0.998 | 0.04 | 0.12 | 3.41 | 102.34 |
| AFG2 | 0.997 | 0.04 | 0.15 | 6.71 | 96.3 |
| AFM1 | 0.996 | 0.03 | 0.01 | 4.18 | 106 |
| FB1 | 0.998 | 0.58 | 1.93 | 3.27 | 82.53 |
| FB2 | 0.998 | 0.24 | 0.80 | 2.96 | 92.97 |
| OTA | 0.997 | 0.01 | 0.03 | 3.65 | 91.17 |
| STE | 0.999 | 0.02 | 0.07 | 3.53 | 100.27 |
| DON | 0.997 | 2.40 | 8.01 | 5.52 | 108.67 |
| ZON | 0.997 | 0.13 | 0.45 | 3.93 | 107.61 |
| HT2 | 0.997 | 0.40 | 1.35 | 8.00 | 97.94 |
| T2 | 0.998 | 0.15 | 0.49 | 4.78 | 96.47 |
| Mycotoxins | Positive Samples (%) | Concentration (μg/kg) | Estimation of Daily Exposure of Aged 6–12 Months (μg/kg/bw) | Estimation of Daily Exposure of Aged 1–3 Years (μg/kg/bw) | |
|---|---|---|---|---|---|
| Minimum and Maximum | Mean ± S.E | ||||
| AFB1 | 12.94 | 0.01–0.08 | 0.036 ± 0.008 | 0.001 | 0.0008 |
| AFB2 | 3.53 | 0.03–0.06 | 0.05 ± 0.01 | 0.002 | 0.001 |
| AFG1 | nd | nd | nd | nd | nd |
| AFG2 | nd | nd | nd | nd | nd |
| AFM1 * | nd | nd | nd | nd | nd |
| FB1 | 12.94 | 2.01–14.09 | 4.70 ± 1.025 | 0.15 | 0.11 |
| FB2 | 10.59 | 0.04–0.20 | 0.071 ± 0.017 | 0.002 | 0.002 |
| OTA | 2.35 | 0.06–0.07 | 0.065 ± 0.005 | 0.002 | 0.002 |
| STE | 34.12 | 0.01–0.50 | 0.063 ± 0.018 | 0.002 | 0.002 |
| DON | 14.12 | 6.32–37.52 | 17.82 ± 2.85 | 0.57 | 0.42 |
| ZON | nd | nd | nd | nd | nd |
| T2 | nd | nd | nd | nd | nd |
| HT2 | 34.12 | 0.02–3.31 | 1.76 ± 0.15 | 0.06 | 0.04 |
| Brands | |||||
|---|---|---|---|---|---|
| A | B | C | D | ||
| AFB1 | N | 10 | 24 | 20 | 31 |
| Mean ± S.E (μg/kg) | nd | 0.036 ± 0.008 | nd | nd | |
| Min (μg/kg) | nd | 0.01 | nd | nd | |
| Max (μg/kg) | nd | 0.08 | nd | nd | |
| AFB2 | N | 10 | 24 | 20 | 31 |
| Mean ± S.E (μg/kg) | nd | 0.03 | nd | 0.06 | |
| Min (μg/kg) | nd | nd | nd | 0.06 | |
| Max (μg/kg) | nd | 0.03 | nd | 0.06 | |
| FB1 | N | 10 | 24 | 20 | 31 |
| Mean ± S.E (μg/kg) | nd | 4.70 ± 1.03 | nd | nd | |
| Min (μg/kg) | nd | 2.01 | nd | nd | |
| Max (μg/kg) | nd | 14.09 | nd | nd | |
| FB2 | N | 10 | 24 | 20 | 31 |
| Mean ± S.E (μg/kg) | nd | 0.07 ± 0.02 | nd | nd | |
| Min (μg/kg) | nd | 0.04 | nd | nd | |
| Max (μg/kg) | nd | 0.20 | nd | nd | |
| OTA | N | 10 | 24 | 20 | 31 |
| Mean ± S.E (μg/kg) | nd | 0.07 ± 0.005 | nd | nd | |
| Min (μg/kg) | nd | 0.06 | nd | nd | |
| Max (μg/kg) | nd | 0.07 | nd | nd | |
| STE | N | 10 | 24 | 20 | 31 |
| Mean ± S.E (μg/kg) | 0.084 ± 0.031 | 0.07 ± 0.03 | 0.02 ± 0.003 | 0.11 ± 0.02 | |
| Min (μg/kg) | 0.01 | 0.01 | 0.01 | 0.08 | |
| Max (μg/kg) | 0.16 | 0.50 | 0.03 | 0.13 | |
| DON | N | 10 | 24 | 20 | 31 |
| Mean ± S.E (μg/kg) | nd | nd | nd | 17.82 ± 2.85 | |
| Min (μg/kg) | nd | nd | nd | 6.32 | |
| Max (μg/kg) | nd | nd | nd | 37.52 | |
| HT2 | N | 10 | 24 | 20 | 31 |
| Mean ± S.E (μg/kg) | nd | 1.88 ± 0.07 | 2.15 ± 0.22 | 1.64 ± 0.25 | |
| Min (μg/kg) | nd | 1.53 | 1.93 | 0.02 | |
| Max (μg/kg) | nd | 2.19 | 2.37 | 3.31 | |
| Positive Samples (%) | Number of Positive Samples | Mean ± SD (log CFU/g) | |
|---|---|---|---|
| Enterobacteriaceae | 10.59 | 9 | 3.14 ± 0.38 |
| TAMB | 30.59 | 26 | 3.09 ± 0.50 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Er Demirhan, B.; Demirhan, B. The Investigation of Mycotoxins and Enterobacteriaceae of Cereal-Based Baby Foods Marketed in Turkey. Foods 2021, 10, 3040. https://doi.org/10.3390/foods10123040
Er Demirhan B, Demirhan B. The Investigation of Mycotoxins and Enterobacteriaceae of Cereal-Based Baby Foods Marketed in Turkey. Foods. 2021; 10(12):3040. https://doi.org/10.3390/foods10123040
Chicago/Turabian StyleEr Demirhan, Buket, and Burak Demirhan. 2021. "The Investigation of Mycotoxins and Enterobacteriaceae of Cereal-Based Baby Foods Marketed in Turkey" Foods 10, no. 12: 3040. https://doi.org/10.3390/foods10123040
APA StyleEr Demirhan, B., & Demirhan, B. (2021). The Investigation of Mycotoxins and Enterobacteriaceae of Cereal-Based Baby Foods Marketed in Turkey. Foods, 10(12), 3040. https://doi.org/10.3390/foods10123040






