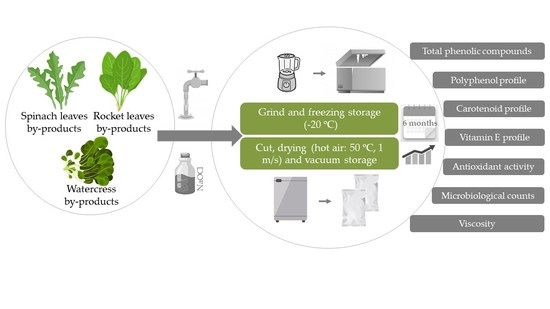
Impact of Processing Approach and Storage Time on Bioactive and Biological Properties of Rocket, Spinach and Watercress Byproducts
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Processing Byproducts
2.3. Microbiological Evaluation
2.4. Pulps Viscosity Monitorization
2.5. Preparation of Methanolic Extracts
2.6. Total Phenolic Content (TPC) Assay
2.7. Antioxidant Activity
2.7.1. 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulphonic acid (ABTS) Assay
2.7.2. Oxygen Radical Absorbance Capacity Assay (ORAC) Assay
2.7.3. 2-diphenyl-1-picrylhydrazyl (DPPH) Assay
2.8. Identification and Quantification of Phenolics by High-Performance Liquid Chromatography (HPLC)
2.9. Monitorization of Carotenoid and Vitamin E Content
2.9.1. Carotenoids and Vitamin E Extraction
2.9.2. Identification and Quantification of Carotenoids by HPLC
2.9.3. Identification and Quantification of Vitamin E by HPLC
2.10. Statistical Analysis
3. Results and Discussion
3.1. Microbial Counts Evaluation
3.2. Pulps Viscosity Assessment
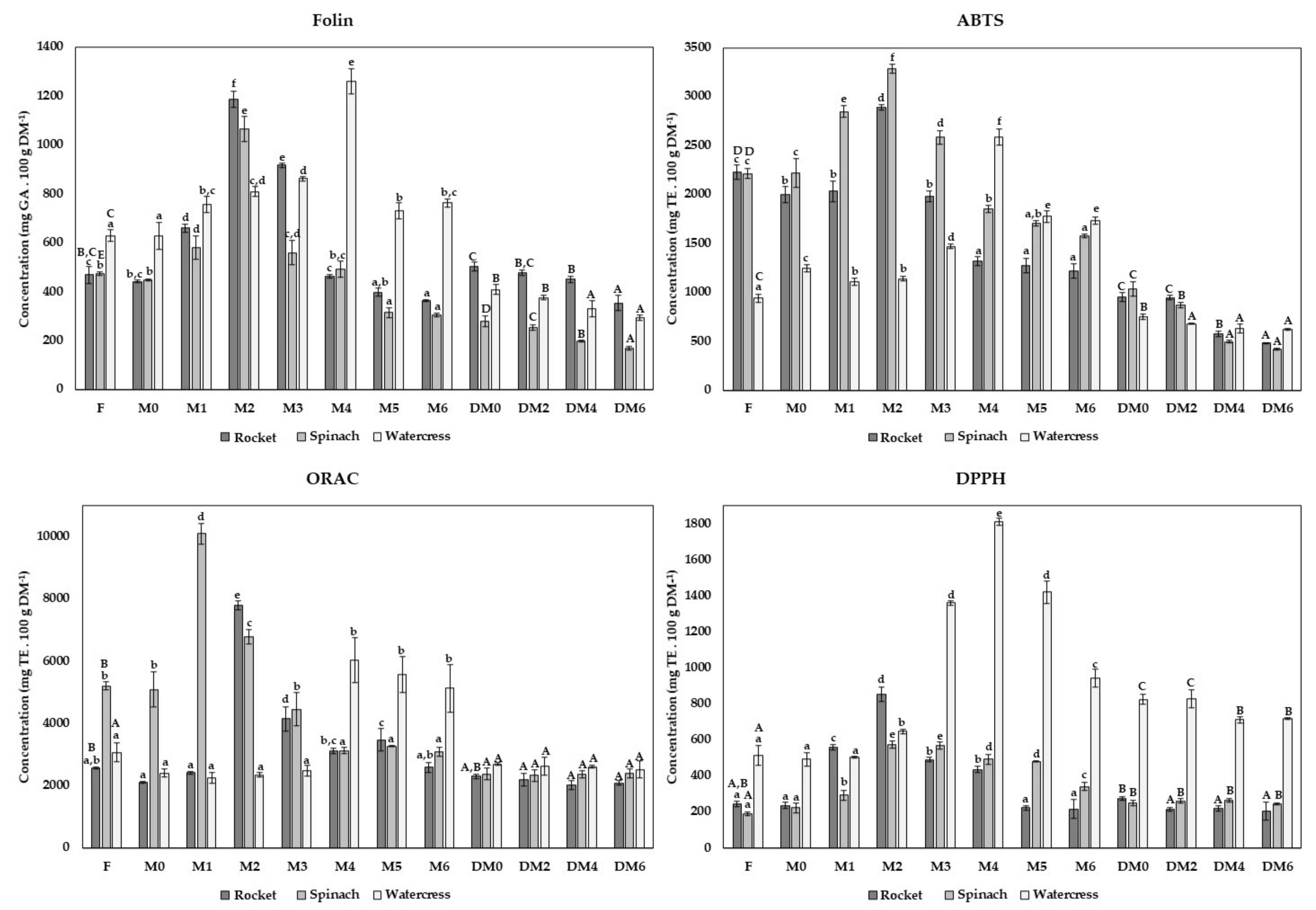
3.3. Monitorization of TPC and Antioxidant Activity
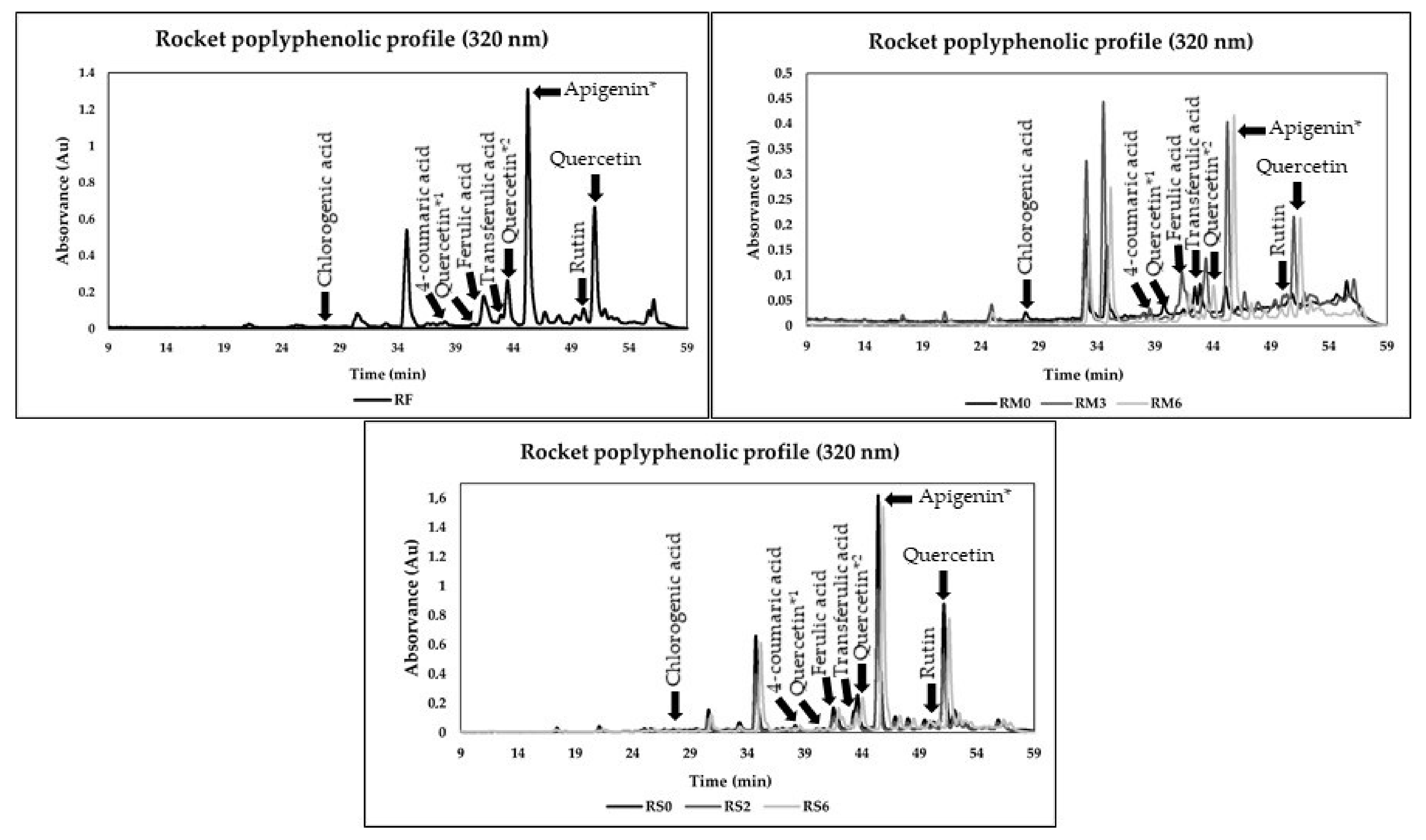
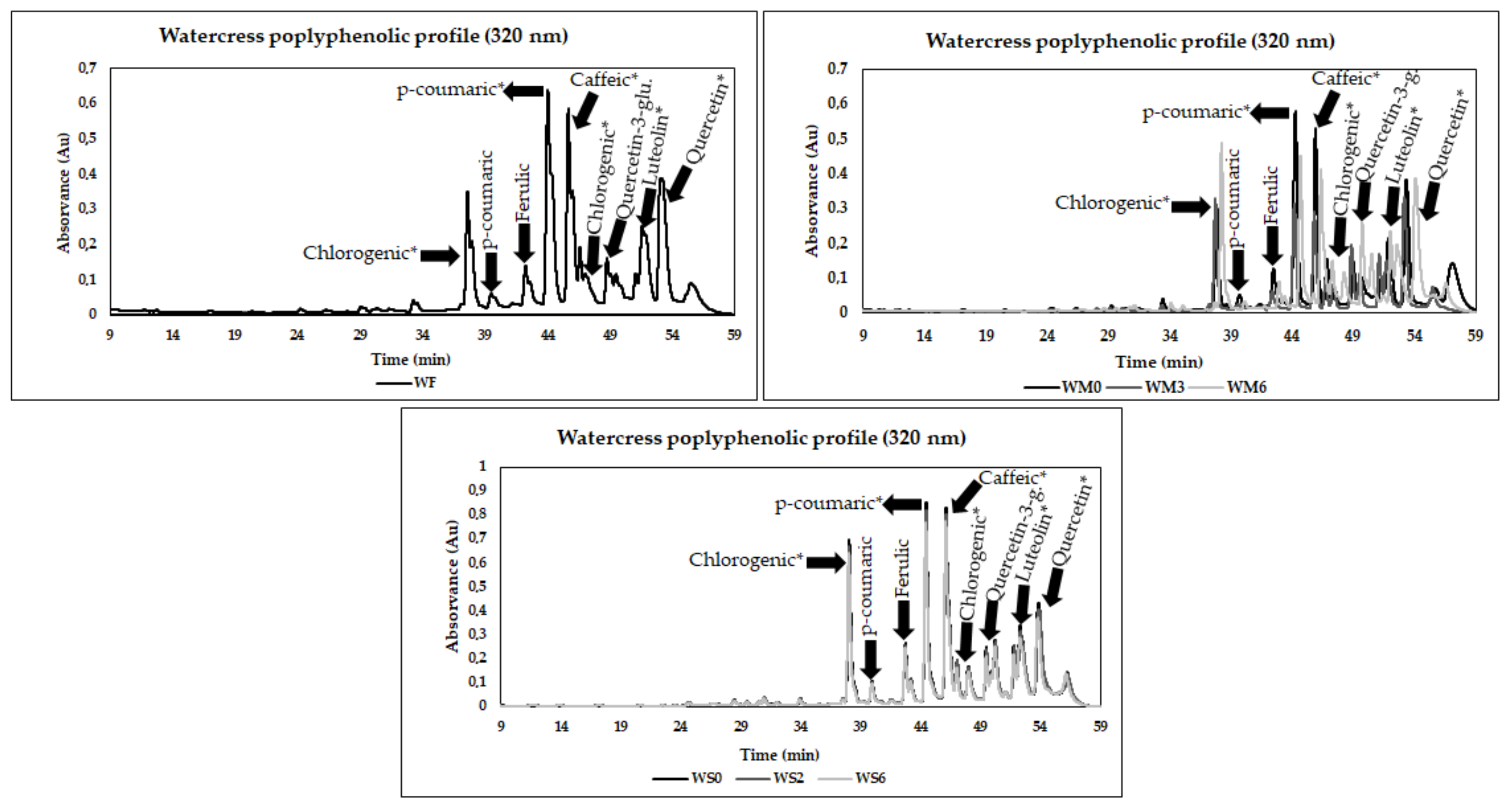
3.4. Phenolic Composition
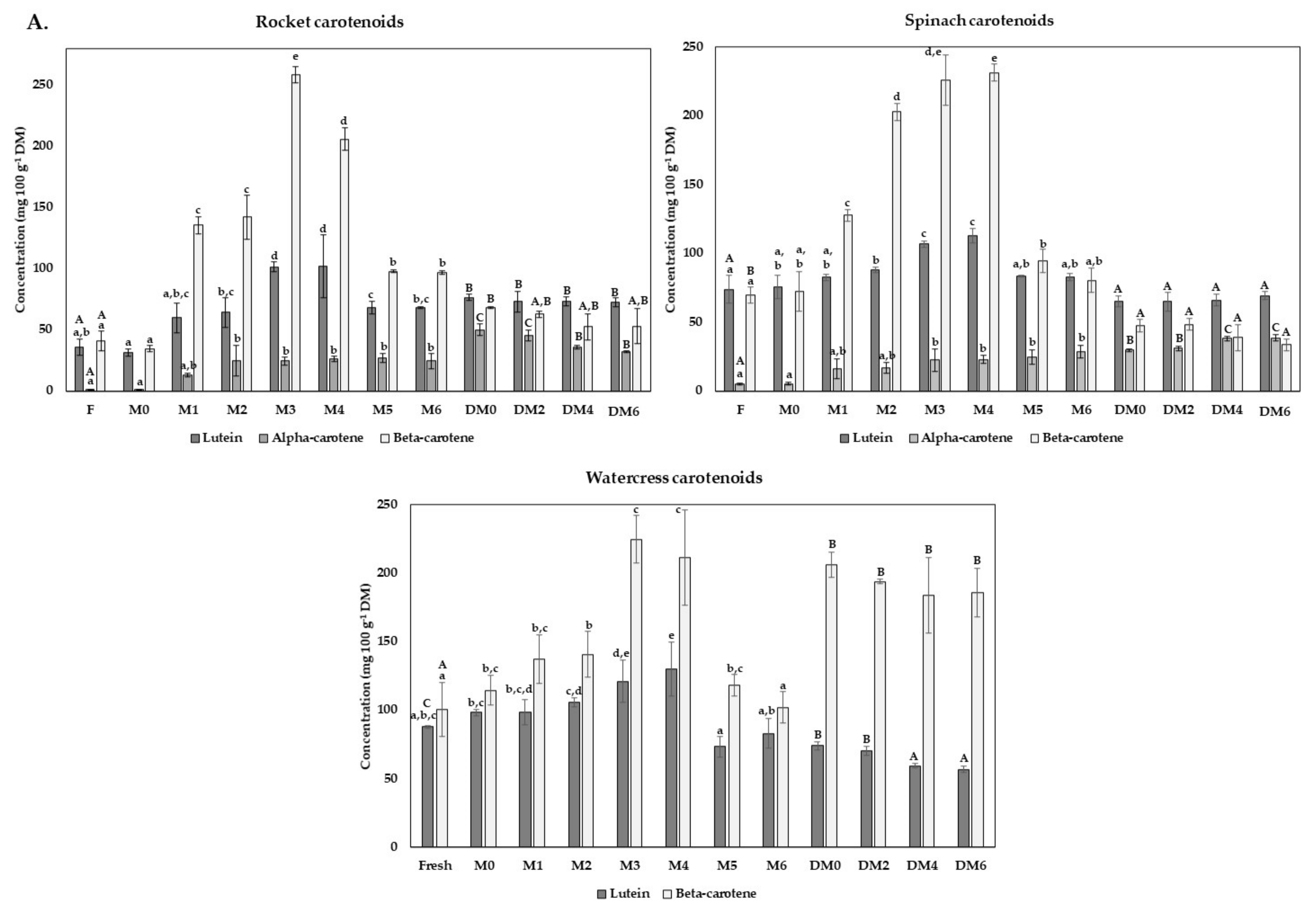
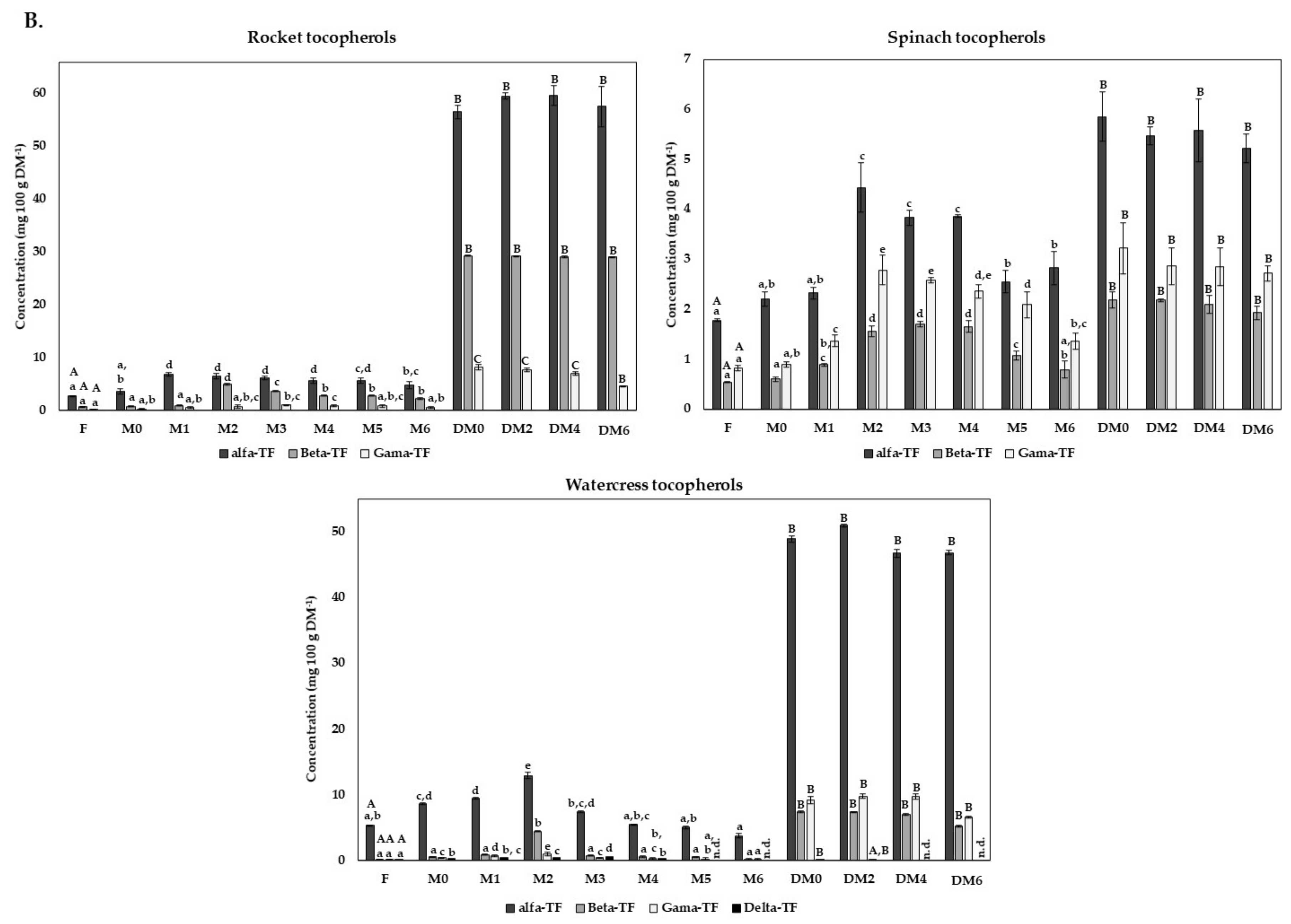
3.5. Carotenoids and Vitamine E Content
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kamiloglu, S.; Toydemir, G.; Boyacioglu, D.; Beekwilder, J.; Hall, R.D.; Capanoglu, E. A Review on the Effect of Drying on Antioxidant Potential of Fruits and Vegetables. Crit. Rev. Food Sci. Nutr. 2016, 56, S110–S129. [Google Scholar] [CrossRef]
- Neri, L.; Faieta, M.; Di Mattia, C.; Sacchetti, G.; Mastrocola, D.; Pittia, P. Antioxidant Activity in Frozen Plant Foods: Effect of Cryoprotectants, Freezing Process and Frozen Storage. Foods 2020, 9, 1886. [Google Scholar] [CrossRef]
- Jenkins, W.; Tucker, M.E.; Grim, J. Routledge Handbook of Religion and Ecology; Routledge International Handbooks: London, UK, 2016; ISBN 9781315764788. [Google Scholar]
- Corrado, S.; Sala, S. Food waste accounting along global and European food supply chains: State of the art and outlook. Waste Manag. 2018, 79, 120–131. [Google Scholar] [CrossRef]
- Martínez, L.; Bastida, P.; Castillo, J.; Ros, G.; Nieto, G. Green alternatives to synthetic antioxidants, antimicrobials, nitrates, and nitrites in clean label Spanish Chorizo. Antioxidants 2019, 8, 184. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- European Commission. Closing the Loop—An EU Action Plan for the Circular Economy; Procedia Cirp, Elsevier: Brussels, Belgium, 2015. [Google Scholar]
- Faustino, M.; Veiga, M.; Sousa, P.; Costa, E.M.; Silva, S.; Pintado, M. Agro-food byproducts as a new source of natural food additives. Molecules 2019, 24, 1056. [Google Scholar] [CrossRef] [Green Version]
- Nayak, B.; Liu, R.H.; Tang, J. Effect of Processing on Phenolic Antioxidants of Fruits, Vegetables, and Grains—A Review. Crit. Rev. Food Sci. Nutr. 2015, 55, 887–918. [Google Scholar] [CrossRef] [PubMed]
- Santos, J.; Oliveira, M.B.P.P.; Ibáñez, E.; Herrero, M. Phenolic profile evolution of different ready-to-eat baby-leaf vegetables during storage. J. Chromatogr. A 2014, 1327, 118–131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saini, R.K.; Ko, E.Y.; Keum, Y.S. Minimally processed ready-to-eat baby-leaf vegetables: Production, processing, storage, microbial safety, and nutritional potential. Food Rev. Int. 2017, 33, 644–663. [Google Scholar] [CrossRef]
- Pinela, J.; Barros, L.; Barreira, J.C.M.; Carvalho, A.M.; Oliveira, M.B.P.P.; Santos-Buelga, C.; Ferreira, I.C.F.R. Postharvest changes in the phenolic profile of watercress induced by post-packaging irradiation and modified atmosphere packaging. Food Chem. 2018, 254, 70–77. [Google Scholar] [CrossRef] [Green Version]
- Hung, P.V.; Duy, T.L. Effects of drying methods on bioactive compounds of vegetables and correlation between bioactive compounds and their antioxidants. Int. Food Res. J. 2012, 19, 327–332. [Google Scholar]
- Ek, P.; Araújo, A.C.; Oliveira, S.M.; Ramos, I.N.; Brandão, T.R.S.; Silva, C.L.M. Assessment of nutritional quality and color parameters of convective dried watercress (Nasturtium officinale). J. Food Process. Preserv. 2018, 42, 1–9. [Google Scholar] [CrossRef]
- Gonçalves, E.M.; Cruz, R.M.S.; Abreu, M.; Brandão, T.R.S.; Silva, C.L.M. Biochemical and colour changes of watercress (Nasturtium officinale R. Br.) during freezing and frozen storage. J. Food Eng. 2009, 93, 32–39. [Google Scholar] [CrossRef] [Green Version]
- Minatel, I.O.; Borges, C.V.; Ferreira, M.I.; Gomez, H.A.G.; Chen, C.-Y.O.; Lima, G.P.P. Phenolic Compounds: Functional Properties, Impact of Processing and Bioavailability. In Phenolic Compounds—Biological Activity; InTechOpen: Zagreb, Croatia, 2017. [Google Scholar]
- Araújo-Rodrigues, H.; Santos, D.; Campos, D.A.; Ratinho, M.; Rodrigues, I.M.; Pintado, M.E. Development of Frozen Pulps and Powders from Carrot and Tomato by-Products: Impact of Processing and Storage Time on Bioactive and Biological Properties. Horticulturae 2021, 7, 185. [Google Scholar] [CrossRef]
- Ijabadeniyi, O.A.; Pillay, Y. Microbial safety of low water activity foods: Study of simulated and durban household samples. J. Food Qual. 2017, 2017, 4931521. [Google Scholar] [CrossRef] [Green Version]
- Ainsworth, E.A.; Gillespie, K.M. Estimation of total phenolic content and other oxidation substrates in plant tissues using Folin-Ciocalteu reagent. Nat. Protoc. 2007, 2, 875–877. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, B.; Falco, V.; Moutinho-Pereira, J.; Bacelar, E.; Peixoto, F.; Correia, C. Effects of elevated CO2 on grapevine (Vitis vinifera L.): Volatile composition, phenolic content, and in vitro antioxidant activity of red wine. J. Agric. Food Chem. 2009, 57, 265–273. [Google Scholar] [CrossRef] [PubMed]
- Coscueta, E.R.; Campos, D.A.; Osório, H.; Nerli, B.B.; Pintado, M. Enzymatic soy protein hydrolysis: A tool for biofunctional food ingredient production. Food Chem. X 2019, 1, 100006. [Google Scholar] [CrossRef] [PubMed]
- Schaich, K.M.; Tian, X.; Xie, J. Hurdles and pitfalls in measuring antioxidant efficacy: A critical evaluation of ABTS, DPPH, and ORAC assays. J. Funct. Foods 2015, 14, 111–125. [Google Scholar] [CrossRef]
- Campos, D.A.; Ribeiro, T.B.; Teixeira, J.A.; Pastrana, L.; Pintado, M.M. Integral valorization of pineapple (Ananas comosus L.) By-products through a green chemistry approach towards Added Value Ingredients. Foods 2020, 9, 60. [Google Scholar] [CrossRef] [Green Version]
- Oliveira, A.; Pintado, M.; Almeida, D.P.F. Phytochemical composition and antioxidant activity of peach as affected by pasteurization and storage duration. LWT- Food Sci. Technol. 2012, 49, 202–207. [Google Scholar] [CrossRef]
- Slavin, M.; Yu, L. A single extraction and HPLC procedure for simultaneous analysis of phytosterols, tocopherols and lutein in soybeans. Food Chem. 2012, 135, 2789–2795. [Google Scholar] [CrossRef]
- Saraiva, M.; Correia, C.; Cunha, I.; Maia, C.; Bonito, C.; Furtado, R.; Calhau, M. Interpretação de Resultados de Ensaios Microbiológicos em Alimentos Prontos Para Consumo e em Superfícies do Ambiente de Preparação e Distribuição Alimentar: Valores-Guia; INSA, IP—Instituto Nacional de Saúde Doutor Ricardo Jorge: Lisboa, Portugal, 2019; ISBN 9789898794529.
- Ntuli, V.; Chatanga, P.; Kwiri, R.; Gadaga, H.T.; Gere, J.; Matsepo, T.; Potloane, R.P. Microbiological quality of selected dried fruits and vegetables in Maseru, Lesotho. African J. Microbiol. Res. 2017, 11, 185–193. [Google Scholar] [CrossRef] [Green Version]
- Bonat Celli, G.; Ghanem, A.; Su-Ling Brooks, M. Influence of freezing process and frozen storage on the quality of fruits and fruit products. Food Rev. Int. 2016, 32, 280–304. [Google Scholar] [CrossRef]
- Thaipong, K.; Boonprakob, U.; Crosby, K.; Cisneros-Zevallos, L.; Hawkins Byrne, D. Comparison of ABTS, DPPH, FRAP, and ORAC assays for estimating antioxidant activity from guava fruit extracts. J. Food Compos. Anal. 2006, 19, 669–675. [Google Scholar] [CrossRef]
- Coscueta, E.R.; Reis, C.A.; Pintado, M. Phenylethyl isothiocyanate extracted from watercress by-products with aqueous micellar systems: Development and optimisation. Antioxidants 2020, 9, 698. [Google Scholar] [CrossRef]
- Ishaq, S.; Arif, S. Quality assessment of stored watercress (Nasturtium oficinale R.Br.) vegetable at freezing temperature. Int. J. Biosci. 2019, 14, 121–133. [Google Scholar] [CrossRef]
- Ribas-Agustí, A.; Martín-Belloso, O.; Soliva-Fortuny, R.; Elez-Martínez, P. Food processing strategies to enhance phenolic compounds bioaccessibility and bioavailability in plant-based foods. Crit. Rev. Food Sci. Nutr. 2018, 58, 2531–2548. [Google Scholar] [CrossRef] [Green Version]
- Frati, A.; Antonini, E.; Ninfali, P. Industrial Freezing, Cooking, and Storage Differently Affect Antioxidant Nutrients in Vegetables. In Fruits, Vegetables, and Herbs; Academic Press: Cambridge, MA, USA, 2016; ISBN 9780128029893. [Google Scholar]
- Kamiloglu, S. Industrial freezing effects on the content and bioaccessibility of spinach (Spinacia oleracea L.) polyphenols. J. Sci. Food Agric. 2020, 100, 4190–4198. [Google Scholar] [CrossRef]
- Ninfali, P.; Bacchiocca, M. Polyphenols and Antioxidant Capacity of Vegetables under Fresh and Frozen Conditions. J. Agric. Food Chem. 2003, 51, 2222–2226. [Google Scholar] [CrossRef]
- Ellong, E.N.; Billard, C.; Adenet, S.; Rochefort, K. Polyphenols, Carotenoids, Vitamin C Content in Tropical Fruits and Vegetables and Impact of Processing Methods. Food Nutr. Sci. 2015, 06, 299–313. [Google Scholar] [CrossRef] [Green Version]
- Lutz, M.; Hernández, J.; Henríquez, C. Phenolic content and antioxidant capacity in fresh and dry fruits and vegetables grown in Chile. CYTA - J. Food 2015, 13, 541–547. [Google Scholar] [CrossRef]
- Pasini, F.; Verardo, V.; Caboni, M.F.; D’Antuono, L.F. Determination of glucosinolates and phenolic compounds in rocket salad by HPLC-DAD-MS: Evaluation of Eruca sativa Mill. and Diplotaxis tenuifolia L. genetic resources. Food Chem. 2012, 133, 1025–1033. [Google Scholar] [CrossRef]
- Li, Z.; Lee, H.W.; Liang, X.; Liang, D.; Wang, Q.; Huang, D.; Ong, C.N. Profiling of phenolic compounds and antioxidant activity of 12 cruciferous vegetables. Molecules 2018, 23, 1139. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alarcón-Flores, M.I.; Romero-González, R.; Martínez Vidal, J.L.; Garrido Frenich, A. Determination of Phenolic Compounds in Artichoke, Garlic and Spinach by Ultra-High-Performance Liquid Chromatography Coupled to Tandem Mass Spectrometry. Food Anal. Methods 2014, 7, 2095–2106. [Google Scholar] [CrossRef]
- Rashmi, H.B.; Negi, P.S. Phenolic acids from vegetables: A review on processing stability and health benefits. Food Res. Int. 2020, 136, 109298. [Google Scholar] [CrossRef]
- Gómez-García, R.; Campos, D.A.; Oliveira, A.; Aguilar, C.N.; Madureira, A.R.; Pintado, M. A chemical valorisation of melon peels towards functional food ingredients: Bioactives profile and antioxidant properties. Food Chem. 2021, 335, 127579. [Google Scholar] [CrossRef]
- O’Neill, M.E.; Carroll, Y.; Corridan, B.; Olmedilla, B.; Granado, F.; Blanco, I.; Van den Berg, H.; Hininger, I.; Rousell, A.M.; Chopra, M.; et al. A European carotenoid database to assess carotenoid intakes and its use in a five-country comparative study. Br. J. Nutr. 2001, 85, 499–507. [Google Scholar] [CrossRef]
- Saini, R.K.; Shang, X.M.; Ko, E.Y.; Choi, J.H.; Kim, D.; Keum, Y.S. Characterization of nutritionally important phytoconstituents in minimally processed ready-to-eat baby-leaf vegetables using HPLC–DAD and GC–MS. J. Food Meas. Charact. 2016, 10, 341–349. [Google Scholar] [CrossRef]
- Syamila, M.; Gedi, M.A.; Briars, R.; Ayed, C.; Gray, D.A. Effect of temperature, oxygen and light on the degradation of β-carotene, lutein and α-tocopherol in spray-dried spinach juice powder during storage. Food Chem. 2019, 284, 188–197. [Google Scholar] [CrossRef]
- Villatoro-Pulido, M.; Priego-Capote, F.; Álvarez-Sánchez, B.; Saha, S.; Philo, M.; Obregón-Cano, S.; De Haro-Bailón, A.; Font, R.; Del Río-Celestino, M. An approach to the phytochemical profiling of rocket [Eruca sativa (Mill.) Thell]. J. Sci. Food Agric. 2013, 93, 3809–3819. [Google Scholar] [CrossRef]
- Kopsell, D.A.; Barickman, T.C.; Sams, C.E.; McElroy, J.S. Influence of nitrogen and sulfur on biomass production and carotenoid and glucosinolate concentrations in watercress (Nasturtium officinale R. Br.). J. Agric. Food Chem. 2007, 55, 10628–10634. [Google Scholar] [CrossRef] [PubMed]
- Kidmose, U.; Knuthsen, P.; Edelenbos, M.; Justesen, U.; Hegelund, E. Carotenoids and flavonoids in organically grown spinach (Spinacia oleracea L) genotypes after deep frozen storage. J. Sci. Food Agric. 2001, 81, 918–923. [Google Scholar] [CrossRef]
- Delchier, N.; Reich, M.; Renard, C.M.G.C. Impact of cooking methods on folates, ascorbic acid and lutein in green beans (Phaseolus vulgaris) and spinach (Spinacea oleracea). LWT- Food Sci. Technol. 2012, 49, 197–201. [Google Scholar] [CrossRef]
- Esposito, F.; Arlotti, G.; Bonifati, A.; Napolitano, A.; Vitale, D.; Fogliano, V. Antioxidant activity and dietary Wbre in durum wheat bran by-products. Food Res. Int. 2005, 38, 1167–1173. [Google Scholar] [CrossRef]
- Ramachandraiah, K.; Chin, K.B. Effect of particle size of persimmon by-product powders on their physicochemical properties and antioxidant activities in porcine patties. J. Food Process Eng. 2018, 41, e12610. [Google Scholar] [CrossRef]
- Char, C. Carrots (Daucus carota L.). In Fruit and Vegetable Phytochemicals; John Wiley & Son: Hoboken, NJ, USA, 2017; pp. 969–978. [Google Scholar]
- Bermúdez, L.; Del Pozo, T.; Silvestre Lira, B.; de Godoy, F.; Boos, I.; Romanò, C.; Previtali, V.; Almeida, J.; Bréhélin, C.; Asis, R.; et al. A Tomato Tocopherol-Binding Protein Sheds Light on Intracellular α-Tocopherol Metabolism in Plants. Plant Cell Physiol. 2018, 59, 2188–2203. [Google Scholar] [CrossRef] [Green Version]
- Lee, H.W.; Zhang, H.; Liang, X.; Ong, C.N. Simultaneous determination of carotenoids, tocopherols and phylloquinone in 12 Brassicaceae vegetables. Lwt 2020, 130, 109649. [Google Scholar] [CrossRef]
- Pereira, C.; Dias, M.I.; Petropoulos, S.A.; Plexida, S.; Chrysargyris, A.; Tzortzakis, N.; Calhelha, R.C.; Ivanov, M.; Stojković, D.; Soković, M.; et al. The effects of biostimulants, biofertilizers and water-stress on nutritional value and chemical composition of two spinach genotypes (Spinacia oleracea L.). Molecules 2019, 24, 4494. [Google Scholar] [CrossRef] [Green Version]
- Saini, R.K.; Shang, X.M.; Ko, E.Y.; Choi, J.H.; Keum, Y.-S. Stability of carotenoids and tocopherols in ready-to-eat baby-leaf lettuce and salad rocket during low-temperature storage. Int. J. Food Sci. Nutr. 2016, 67, 489–495. [Google Scholar] [CrossRef]

| Vegetables | Sample | TAB | Enterobacteriacea | Yeasts and Molds | Bacillus cereus spp. | Aerobic Spore-Forms | Anaerobic Spore-Forms |
|---|---|---|---|---|---|---|---|
| Rocket | Non washed | 8.20 ± 0.18 | 7.00 ± 0.07 | 5.95 ± 0.10 | 4.80 ± 0.09 | - | - |
| Washed | 4.95 ± 0.10 | 3.81 ± 0.08 | 3.70 ± 0.09 | 2.84 ± 0.10 | - | - | |
| Fresh pulp | 5.04 ± 0.03 | 3.92 ± 0.03 | 3.75 ± 0.15 | 2.92 ± 0.05 | - | - | |
| M0 | 5.00 ± 0.09 | 3.84 ± 0.08 | 3.89 ± 0.13 | 2.90 ± 0.03 | - | - | |
| M1 | 5.02 ± 0.09 | 3.85 ± 00.08 | 3.82 ± 0.11 | 2.83 ± 0.13 | - | - | |
| M2 | 5.05 ± 0.10 | 3.89 ± 0.02 | 3.74 ± 0.29 | 2.70 ± 0.05 | - | - | |
| M3 | 5.02 ± 0.04 | 3.86 ± 0.05 | 3.73 ± 0.03 | 2.67 ± 0.09 | - | - | |
| M4 | 4.92 ± 0.04 | 3.84 ± 0.15 | 3.66 ± 0.11 | 2.71 ± 0.08 | - | - | |
| M5 | 4.99 ± 0.10 | 3.81 ± 0.09 | 3.71 ± 0.15 | 2.69 ± 0.15 | - | - | |
| M6 | 4.96 ± 0.14 | 3.84 ± 0.03 | 3.69 ± 0.08 | 2.72 ± 0.08 | - | - | |
| DM0 | 6.07 ± 0.84 | 6.16 ± 0.17 | - | - | 4.95 ± 0.61 | 3.92 ± 0.11 | |
| DM6 | 4.99 ± 0.13 | 4.08 ± 0.13 | - | - | 4.47 ± 0.92 | 3.52 ± 0.74 | |
| Spinach | Non washed | 8.09 ± 0.09 | 6.58 ± 0.04 | 5.59 ± 0.05 | 4.43 ± 0.05 | - | - |
| Washed | 4.54 ± 0.10 | 3.98 ± 0.05 | 3.58 ± 0.13 | 2.49 ± 0.09 | - | - | |
| Fresh pulp | 4.62 ± 0.09 | 4.03 ± 0.04 | 3.61 ± 0.16 | 2.51 ± 0.16 | - | - | |
| M0 | 4.65 ± 0.10 | 4.01 ± 0.10 | 3.59 ± 0.14 | 2.56 ± 0.10 | - | - | |
| M1 | 4.67 ± 0.11 | 4.00 ± 0.08 | 3.61 ± 0.19 | 2.55 ± 0.14 | - | - | |
| M2 | 4.67 ± 0.05 | 4.03 ± 0.07 | 3.67 ± 0.05 | 2.58 ± 0.13 | - | - | |
| M3 | 4.68 ± 0.16 | 4.04 ± 0.11 | 3.75 ± 0.09 | 2.61 ± 0.17 | - | - | |
| M4 | 4.60 ± 0.20 | 4.02 ± 0.11 | 3.73 ± 0.05 | 2.69 ± 0.02 | - | - | |
| M5 | 4.72 ± 0.18 | 4.04 ± 0.09 | 3.76 ± 0.06 | 2.64 ± 0.15 | - | - | |
| M6 | 4.65 ± 0.16 | 4.02 ± 0.15 | 3.68 ± 0.03 | 2.66 ± 0.05 | - | - | |
| DM0 | >5.48 ± 0.00 | 5.46 ± 0.49 | - | - | 5.85 ± 0.27 | 4.74 ± 0.06 | |
| DM6 | 6.93 ± 0.49 | 6.31 ± 0.56 | - | - | 5.33 ± 0.22 | 4.48 ± 0.43 | |
| Watercress | Non washed | 8.30 ± 0.18 | 7.23 ± 0.11 | 6.67 ± 0.08 | 4.45 ± 0.05 | - | - |
| Washed | 4.80 ± 0.15 | 4.06 ± 0.06 | 3.82 ± 0.12 | 2.51 ± 0.11 | - | - | |
| Fresh pulp | 4.96 ± 0.10 | 4.12 ± 0.13 | 3.87 ± 0.16 | 2.74 ± 0.11 | - | - | |
| M0 | 4.99 ± 0.10 | 4.14 ± 0.12 | 3.81 ± 0.18 | 2.69 ± 0.17 | - | - | |
| M1 | 4.94 ± 0.18 | 4.11 ± 0.06 | 3.82 ± 0.10 | 2.72 ± 0.16 | - | - | |
| M2 | 4.96 ± 0.15 | 4.10 ± 0.04 | 3.87 ± 0.16 | 2.77 ± 0.09 | - | - | |
| M3 | 4.98 ± 0.06 | 4.12 ± 0.06 | 3.88 ± 0.11 | 2.67 ± 0.23 | - | - | |
| M4 | 5.00 ± 0.05 | 4.14 ± 0.08 | 3.91 ± 0.10 | 2.75 ± 0.16 | - | - | |
| M5 | 4.96 ± 0.05 | 4.17 ± 0.06 | 3.85 ± 0.13 | 2.64 ± 0.08 | - | - | |
| M6 | 4.94 ± 0.07 | 4.11 ± 0.08 | 3.86 ± 0.13 | 2.65 ± 0.14 | - | - | |
| DM0 | >6.48 ± 0.00 | 7.00 ± 0.05 | - | - | 6.15 ± 0.35 | 5.24 ± 0.29 | |
| DM6 | 7.49 ± 0.53 | 6.42 ± 0.08 | - | - | 6.06 ± 0.01 | 5.14 ± 1.04 |
| Vegetable | Sample | Viscosity (mPa s−1) |
|---|---|---|
| Rocket | Fresh pulp | 364.24 ± 3.22 g |
| M0 | 311.02 ± 3.93 f | |
| M1 | 299.12 ± 2.79 e | |
| M2 | 290.76 ± 1.97 d | |
| M3 | 282.68 ± 0.81 c | |
| M4 | 273.20 ± 2.46 b | |
| M5 | 264.41 ± 3.85 a | |
| M6 | 257.00 ± 1.93 a | |
| Spinach | Fresh pulp | 155.70 ± 2.12 f |
| M0 | 121.24 ± 1.10 e | |
| M1 | 115.18 ± 1.71 d | |
| M2 | 107.10 ± 0.90 c | |
| M3 | 103.66 ± 0.89 b,c | |
| M4 | 100.23 ± 3.60 b | |
| M5 | 96.22 ± 1.99 a | |
| M6 | 92.46 ± 1.63 a | |
| Watercress | Fresh pulp | 253.68 ± 7.97 h |
| M0 | 203.82 ± 1.69 g | |
| M1 | 194.42 ± 3.54 f | |
| M2 | 180.78 ± 1.99 e | |
| M3 | 169.22 ± 1.17 d | |
| M4 | 152.12 ± 3.40 c | |
| M5 | 140.80 ± 1.07 b | |
| M6 | 135.80 ± 3.96 a |
| R | Chlorogenic Acid | p-Coumaric Acid | Quercetin *1 | Ferulic Acid | Transferulic Acid | Quercetin *2 | Apigenin * | Rutin | Quercetin-3-glucoside |
| F | 6.0 ± 0.47 d,B | 1.3 ± 0.6 c,d,e,A | 4.1 ± 0.1 a,A | 6.0 ± 0.1 c,B | 15.8 ± 0.3 f,C | 12.2 ± 0.4 a,A | 137.31 ± 0.74 a,A | 36.4 ± 0.3 h,B | 48.3 ± 1.2 f,C |
| M0 | 1.3 ± 0.03 a,b | 0.6 ± 0.0 a,b | 5.6 ± 0.8 a | 3.4 ± 0.1 a | 4.6 ± 0.0 e | 19.1 ± 1.5 b | 88.17 ± 0.41 b | 15.6 ± 0.2 f | 38.9 ± 3.8 e |
| M1 | 1.5 ± 0.09 a | 1.7 ± 0.3 e | 15.2 ± 0.1 c | 5.1 ± 0.0 b | 2.4 ± 0.1 c | 49.7 ± 0.4 d | 105.81 ± 1.66 c | 12.3 ± 0.9 e | 22.9 ± 2.5 b |
| M2 | 7.5 ± 1.09 e | 3.3 ± 0.3 f | 48.7 ± 1.8 g | 12.7 ± 0.1 e | 0.8 ± 0.0 a | 131.3 ± 4.8 g | 102.49 ± 0.77 c | 2.3 ± 0.2 a | 27.2 ± 1.5 c |
| M3 | 2.9 ± 0.10 c | 1.6 ± 0.1 d,e | 31.8 ± 0.8 f | 25.2 ± 0.4 g | 2.3 ± 0.2 b,c | 65.8 ± 3.2 e | 205.04 ± 2.53 d | 6.4 ± 0.6 c | 18.1 ± 1.5 b |
| M4 | 2.0 ± 0.00 a,b,c | 0.9 ± 0.1 a,b,c | 17.2 ± 0.3 d | 17.3 ± 0.0 f | 1.9 ± 0.2 b | 87.4 ± 1.8 f | 232.71 ± 4.76 e | 22.9 ± 1.1 g | 34.3 ± 1.8 d |
| M5 | 2.6 ± 0.03 b,c | 1.0 ± 0.1 b,c,d | 24.3 ± 0.1 e | 6.7 ± 0.4 d | 3.8 ± 0.3 d | 34.7 ± 0.3 c | 314.35 ± 0.19 f | 8.2 ± 0.5 d | 20.5 ± 0.6 b |
| M6 | 1.3 ± 0.00 a | 0.4 ± 0.0 a | 11.6 ± 0.0 b | 3.2 ± 0.0 a | 1.9 ± 0.0 b,c | 16.5 ± 0.0 a,b | 149.82 ± 1.22 g | 4.0 ± 0.0 b | 9.8 ± 0.0 a |
| DM0 | 2.3 ± 0.07 A | 1.6 ± 0.1 A | 31.7 ± 0.9 C | 5.5 ± 0.4 B | 3.1 ± 0.0 B | 45.9 ± 0.5 D | 444.75 ± 5.23 B | 14.0 ± 0.9 A | 14.9 ± 0.5 B |
| DM2 | 2.1 ± 0.00 A | 1.4 ± 0.1 A | 29.1 ± 1.7 C | 5.5 ± 0.1 B | 2.9 ± 0.1 B | 39.5 ± 1.1 C | 429.89 ± 3.24 C | 13.6 ± 0.6 A | 14.1 ± 0.1 A,B |
| DM4 | 2.1 ± 0.01 A | 1.5 ± 0.1 A | 22.0 ± 0.0 B | 4.2 ± 0.0 A | 3.0 ± 0.1 A | 34.4 ± 0.1 B | 400.60 ± 0.43 D | 13.4 ± 0.9 A | 13.8 ± 0.2 A,B |
| DM6 | 2.0 ± 0.16 A | 1.5 ± 0.0 A | 22.3 ± 1.3 B | 4.4 ± 0.3 A | 2.0 ± 0.3 A | 35.6 ± 1.4 B | 407.05 ± 1.35 D | 13.7 ± 0.8 A | 12.4 ± 0.8 A |
| S | Epigallocatechin Gallate * | Caffeic Acid * | p-Coumaric Acid | Lutein *1 | Rutin *1 | Rutin *2 | Lutein *2 | - | - |
| F | 10.9 ± 0.1 b,A | 8.5 ± 0.7 d,B | N.D. | 4.3 ± 0.0 a,A | 2.4 ± 0.1 a,A | 2.7 ± 0.0 a,A | 21.2 ± 0.2 b,A | - | - |
| M0 | 3.1 ± 0.2 a | 5.9 ± 0.0 b,c | 0.1 ± 0.0 a,b | 16.2 ± 0.4 b | 3.1 ± 0.1 a,b | 7.9 ± 0.5 b | 34.3 ± 0.2 c | - | - |
| M1 | 5.6 ± 0.1 a | 4.1 ± 0.0 a | 0.4 ± 0.0 c | 31.5 ± 0.2 c | 6.5 ± 0.2 b,c | 12.9 ± 0.0 c | 80.6 ± 1.2 d,e | - | - |
| M2 | 18.1 ± 0.1 c | 3.9 ± 0.2 a | 1.2 ± 0.1 d | 39.0 ± 0.2 d | 25.6 ± 2.8 d | 32.1 ± 0.0 f | 82.2 ± 4.2 d,e | - | - |
| M3 | 27.6 ± 1.3 d | 6.7 ± 0.4 c | 1.9 ± 0.0 e | 47.0 ± 1.3 e | 27.2 ± 2.1 d | 25.9 ± 0.5 e | 149.0 ± 7.0 f | - | - |
| M4 | 30.0 ± 2.4 d | 5.4 ± 0.1 b | 2.0 ± 0.1 e | 56.0 ± 1.7 f | 5.1 ± 0.3 a,b | 16.9 ± 0.5 d | 87.9 ± 1.1 e | - | - |
| M5 | 28.9 ± 1.2 d | 3.7 ± 0.4 a | 0.2 ± 0.0 b | 39.8 ± 2.5 d | 5.1 ± 0.3 a,b | 10.4 ± 2.2 b,c | 74.1 ± 3.5 d | - | - |
| M6 | 38.9 ± 3.9 e | 10.6 ± 1.0 e | N.D. | 2.5 ± 0.1 a | 4.8 ± 0.9 c | 38.2 ± 2.3 g | 9.8 ± 0.7 a | - | - |
| DM0 | 47.6 ± 0.9 C | 4.2 ± 0.2 A | 1.6 ± 0.1 C | 9.5 ± 0.2 B | 3.9 ± 0.2 C | 14.8 ± 0.2 C | 78.6 ± 8.2 D | - | - |
| DM2 | 47.1 ± 0.9 C | 3.7 ± 0.5 A | 1.6 ± 0.1 C | 9.5 ± 0.5 B | 3.4 ± 0.1 B | 16.5 ± 0.6 D | 63.2 ± 5.2 C | - | - |
| DM4 | 47.3 ± 0.0 C | 3.6 ± 0.5 A | 1.4 ± 0.0 B | 9.5 ± 0.1 B | 3.4 ± 0.0 B | 10.9 ± 0.1 B | 51.2 ± 3.5 B,C | - | - |
| DM6 | 45.2 ± 0.1 B | 3.5 ± 0.2 A | 0.9 ± 0.0 A | 9.5 ± 0.5 B | 2.5 ± 0.1 A | 10.2 ± 0.1 B | 50.1 ± 0.9 B | - | - |
| W | Chlorogenic Acid *1 | p-Coumaric Acid | Ferulic Acid | p-Coumaric Acid * | Caffeic Acid * | p-Coumaric Acid *2 | Quercetin-3-glucoside | Luteolin * | Quercetin * |
| F | 58.0 ± 2.7 g,B | 5.3 ± 1.3 d,B | 14.5 ± 1.8 d,B | 54.0 ± 0.6 f,B | 214.9 ± 1.6 h,C | 12.4 ± 0.6 e,B | 37.8 ± 1.1 c,B | 30.9 ± 0.1 d,D | 92.2 ± 2.5 e,B |
| M0 | 36.1 ± 0.7 e | 5.0 ± 0.5 d | 15.3 ± 0.6 d | 34.7 ± 0.4 d | 174.6 ± 2.0 g | 7.9 ± 0.5 c | 55.7 ± 0.9 d | 48.0 ± 0.9 f | 69.0 ± 1.7 d |
| M1 | 49.0 ± 0.6 f | 3.6 ± 0.1 c | 8.4 ± 0.3 c | 30.1 ± 0.2 c | 154.6 ± 1.0 f | 8.2 ± 0.1 c | 41.3 ± 0.8 c | 36.4 ± 0.6 e | 64.6 ± 0.9 d |
| M2 | 22.3 ± 1.9 d | 1.4 ± 0.1 a,b | 5.0 ± 0.1 b | 10.6 ± 1.4 b | 52.5 ± 0.3 e | 3.0 ± 0.2 b | 23.1 ± 0.5 b | 24.2 ± 3.3 c | 29.7 ± 1.4 b |
| M3 | 12.0 ± 0.7 c | 0.3 ± 0.1 a | 1.2 ± 0.3 a | 5.6 ± 0.3 a | 26.9 ± 0.1 c | 1.3 ± 0.2 a | 13.6 ± 0.3 a | 14.8 ± 0.3 b | 19.3 ± 1.4 a |
| M4 | 5.2 ± 0.4 a | 1.0 ± 0.0 a,b | 1.7 ± 0.1 a | 10.5 ± 0.4 b | 8.0 ± 0.7 a | 10.5 ± 0.5 d | 23.7 ± 2.0 b | 4.9 ± 0.7 a | 45.2 ± 0.7 c |
| M5 | 6.7 ± 0.2 a,b | 1.9 ± 0.1 b | 6.2 ± 0.6 b | 47.9 ± 0.5 e | 13.2 ± 0.2 b | 48.3 ± 0.7 f | 86.3 ± 0.6 e | 18.6 ± 0.2 b | 140.9 ± 0.8 f |
| M6 | 8.1 ± 0.1 b | 1.1 ± 0.1 a,b | 6.5 ± 0.5 b | 58.1 ± 0.2 g | 30.9 ± 0.2 d | 58.2 ± 0.4 g | 104.8 ± 6.6 f | 25.6 ± 0.4 c | 168.6 ± 8.3 g |
| DM0 | 2.5 ± 0.1 A | 3.5 ± 0.1 A | 7.9 ± 0.1 A | 27.4 ± 0.4 A | 142.8 ± 0.2 A,B | 6.7 ± 0.2 A | 43.2 ± 0.2 D | 10.3 ± 0.4 C | 63.4 ± 0.4 A |
| DM2 | 2.1 ± 0.2 A | 3.3 ± 0.0 A | 7.3 ± 0.0 A | 26.3 ± 0.5 A | 143.3 ± 0.7 B | 6.5 ± 0.2 A | 41.7 ± 0.4 C | 7.3 ± 0.2 A,B | 61.2 ± 0.6 A |
| DM4 | 2.1 ± 0.0 A | 3.2 ± 0.0 A | 7.0 ± 0.3 A | 26.4 ± 0.7 A | 141.5 ± 1.4 A,B | 6.2 ± 0.1 A | 36.5 ± 0.4 A | 6.8 ± 0.1 A | 62.6 ± 0.6 A |
| DM6 | 2.0 ± 0.2 A | 3.4 ± 0.1 A | 8.3 ± 0.3 A | 27.1 ± 0.6 A | 140.3 ± 0.7 A | 6.5 ± 0.3 A | 42.9 ± 0.4 D | 8.1 ± 0.8 B | 63.6 ± 0.9 A |
| Group of Phenolic Antioxidants | Phenolic Antioxidants | Chemical Formula | Retention Time (min) | λ max (nm) |
|---|---|---|---|---|
| Hydroxycinnamic acid | ||||
| Phenolic acids | Chlorogenic acid | C16H18O9 | 27.2 | 326.0 |
| Caffeic acid | C9H8O4 | 31.0 | 323.6 | |
| p-coumaric acid | C9H8O3 | 39.9 | 309.3 | |
| Ferulic acid | C10H10O4 | 42.4 | 322.4 | |
| Trans-ferulic acid | C10H10O4 | 42.6 | 322.4 | |
| Flavonols | ||||
| Quercetin-3-glucoside | C15H10O7 | 49.9 | 343.9 | |
| Flavones | ||||
| Flavanoids | Luteolin-7-O-glucoside | C15H10O6 | 48.3 | 354.7 |
| Rutin | C27H30O16 | 49.8 | 353.5 | |
| Apigenin | C15H10O5 | 53.7 | 268.0 | |
| Flavanols (cathecin) | ||||
| Epigallocatechin gallate | C22H18O11 | 29.4 | 276.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Araújo-Rodrigues, H.; Santos, D.; Campos, D.A.; Guerreiro, S.; Ratinho, M.; Rodrigues, I.M.; Pintado, M.E. Impact of Processing Approach and Storage Time on Bioactive and Biological Properties of Rocket, Spinach and Watercress Byproducts. Foods 2021, 10, 2301. https://doi.org/10.3390/foods10102301
Araújo-Rodrigues H, Santos D, Campos DA, Guerreiro S, Ratinho M, Rodrigues IM, Pintado ME. Impact of Processing Approach and Storage Time on Bioactive and Biological Properties of Rocket, Spinach and Watercress Byproducts. Foods. 2021; 10(10):2301. https://doi.org/10.3390/foods10102301
Chicago/Turabian StyleAraújo-Rodrigues, Helena, Diva Santos, Débora A. Campos, Suse Guerreiro, Modesta Ratinho, Ivo M. Rodrigues, and Manuela E. Pintado. 2021. "Impact of Processing Approach and Storage Time on Bioactive and Biological Properties of Rocket, Spinach and Watercress Byproducts" Foods 10, no. 10: 2301. https://doi.org/10.3390/foods10102301
APA StyleAraújo-Rodrigues, H., Santos, D., Campos, D. A., Guerreiro, S., Ratinho, M., Rodrigues, I. M., & Pintado, M. E. (2021). Impact of Processing Approach and Storage Time on Bioactive and Biological Properties of Rocket, Spinach and Watercress Byproducts. Foods, 10(10), 2301. https://doi.org/10.3390/foods10102301