Effect of Addition of Green Coffee Parchment on Structural, Qualitative and Chemical Properties of Gluten-Free Bread
Abstract
1. Introduction
2. Materials and Methods
2.1. Green Coffee Parchment Characteristics
2.2. Bread Samples Preparation and Storage
2.3. Bread Physical Analyses
2.4. Bread Chemical Analyses
Chemicals
2.5. Total Phenolic Content (TPC), Total Antioxidant Capacity (TAC), and Oxitest
2.6. Determination of 5-HMF
2.7. Sensory Analysis
2.8. Statistical Analysis
3. Results and Discussion
3.1. Green Coffee Parchment Characteristics
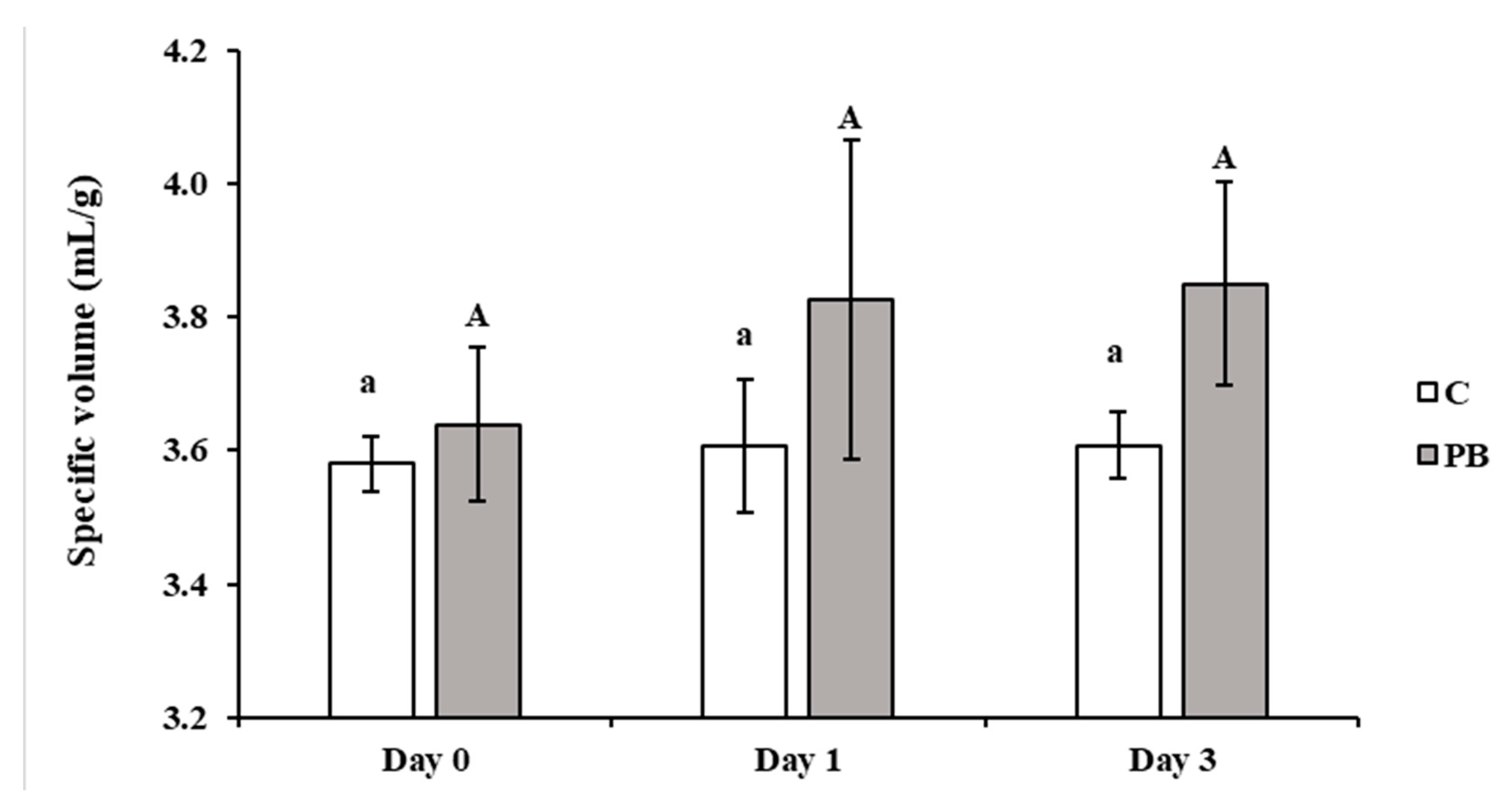
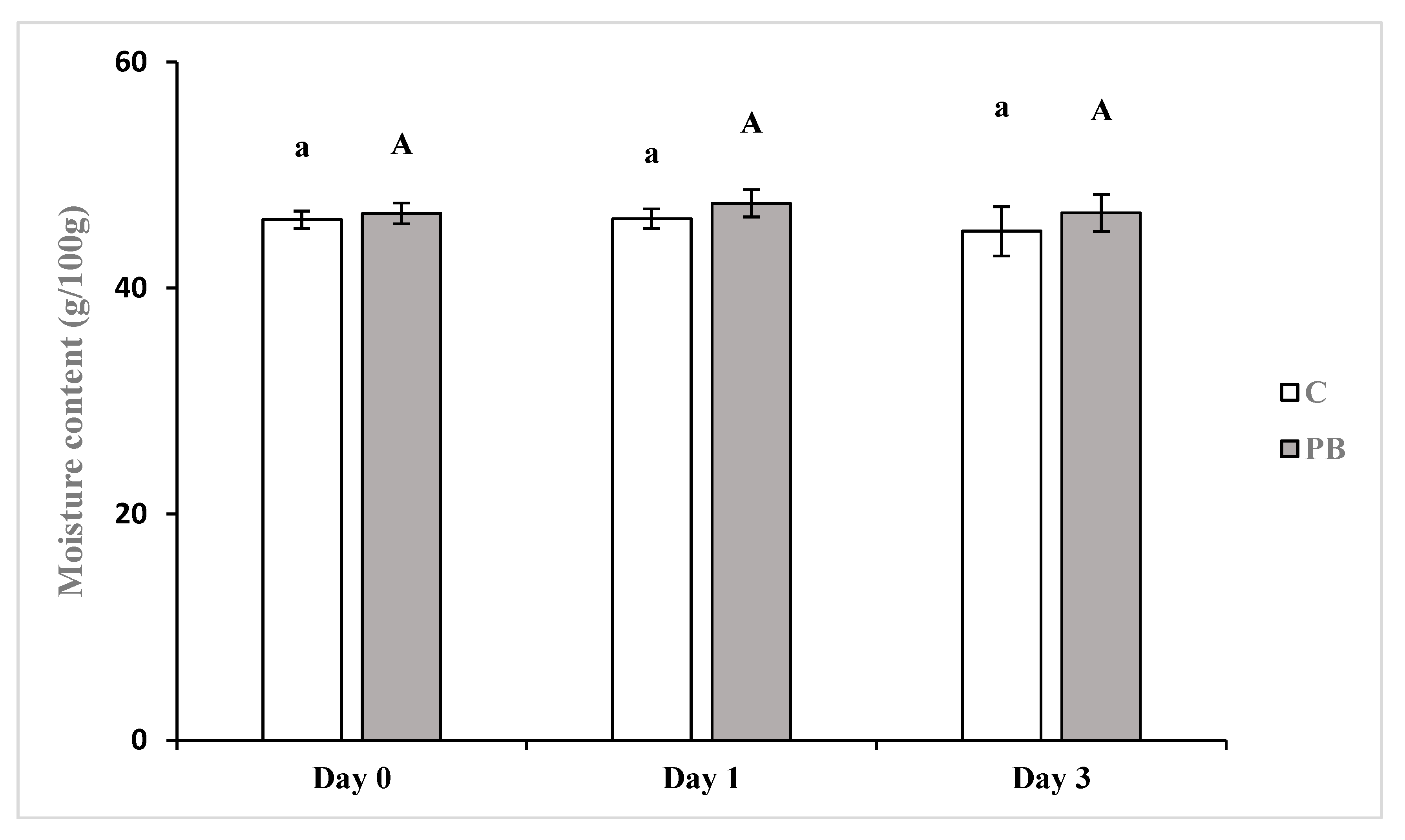
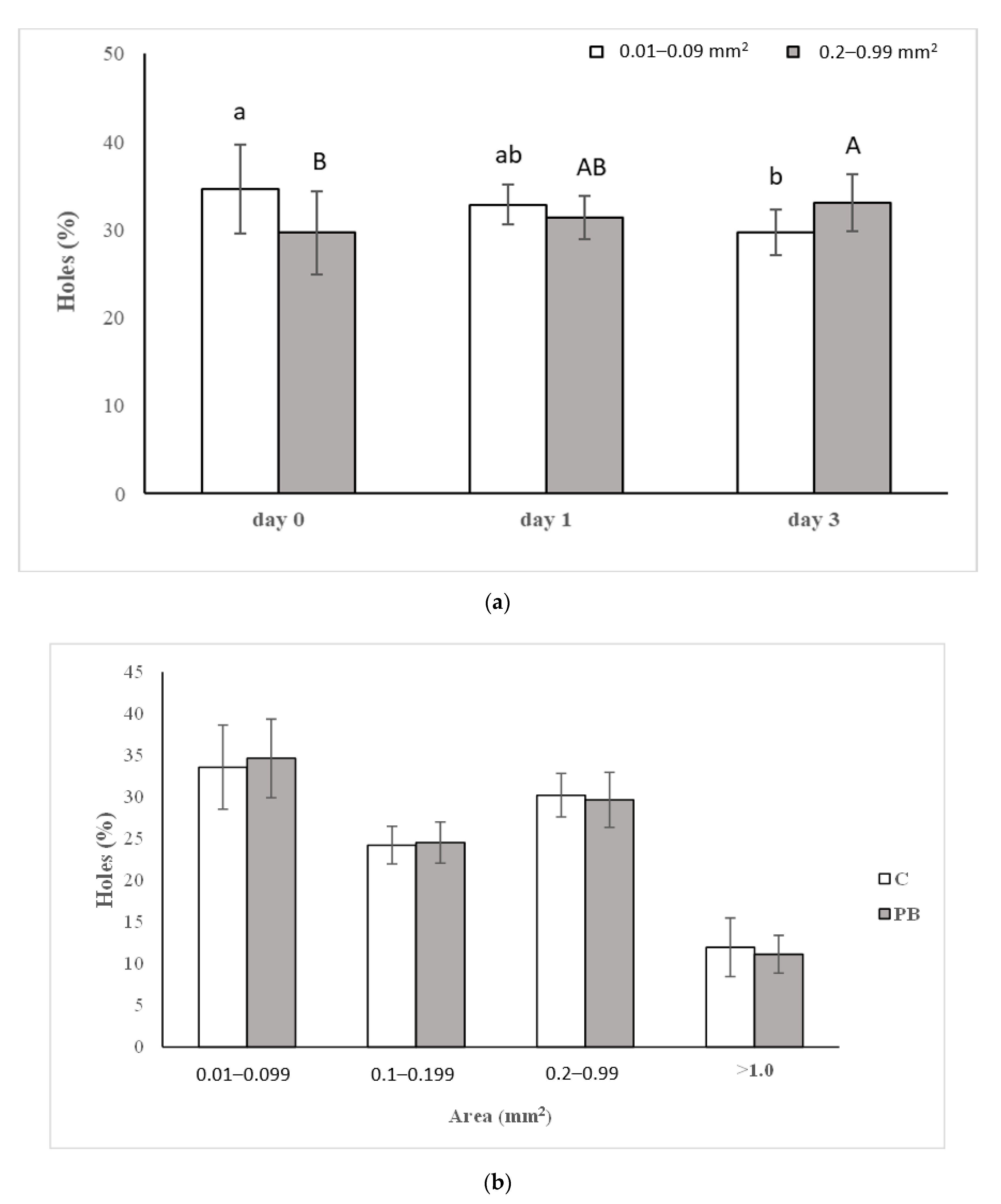
3.2. Bread Physical Characterization
3.3. Bread Chemical Characterization
3.4. Sensory Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- FAOSTAT. Food and Agriculture Organization of the United Nations. 2008. Available online: http://www.fao.org/faostat/en/#data/QC (accessed on 17 October 2020).
- Iriondo-De Hond, A.; Aparicio García, N.; Fernandez-Gomez, B.; Guisantes-Batan, E.; Velázquez Escobar, F.; Blanch, G.P.; San Andres, M.I.; Sanchez-Fortun, S.; del Castillo, M.D. Validation of coffee by-products as novel food ingredients. Innov. Food Sci. Emerg. 2019, 51, 194–204. [Google Scholar] [CrossRef]
- Esquivel, P.; Jiménez, V.M. Functional properties of coffee and coffee by-products. Food Res. Int. 2012, 46, 488–495. [Google Scholar] [CrossRef]
- Pourfarzad, A.; Mahdavian-Mehr, H.; Sedaghat, N. Coffee silverskin as a source of dietary fiber in bread-making: Optimization of chemical treatment using response surface methodology. LWT—Food Sci. Technol. 2013, 50, 599–606. [Google Scholar] [CrossRef]
- Gocmen, D.; Sahan, Y.; Yildiz, E.; Coskun, M.; Aroufai, İ.A. Use of coffee silverskin to improve the functional properties of cookies. J. Food Sci. Technol. 2019, 56, 2979–2988. [Google Scholar] [CrossRef] [PubMed]
- Cubero-Castillo, E.; Bonilla-Leiva, A.; García-Velazques, E. Coffee berry processing byproduct valorization: Coffee parchment as a potential fiber source to enrich bakery goods. J. Health Popul. Nutr. 2017, 1, 1–7. [Google Scholar]
- Benitez, V.; Rebollo-Hernanz, M.; Hernanz, S.; Chantres, S.; Aguilera, Y.; Martin-Cabrejas, M.A. Coffee parchment as a new dietary fiber ingredient: Functional and physiological characterization. Food Res. Int. 2019, 122, 105–113. [Google Scholar] [CrossRef]
- Klingel, T.; Kremer, J.I.; Gottstein, V.; Rajcic de Rezende, T.; Schwarz, S.; Lachenmeier, D.W. A Review of Coffee By-Products Including Leaf, Flower, Cherry, Husk, Silver Skin, and Spent Grounds as Novel Foods within the European Union. Foods 2020, 9, 665. [Google Scholar] [CrossRef]
- Iriondo-DeHond, A.; Iriondo-DeHond, M.; Del Castillo, M.D. Applications of compounds from coffee processing by-products. Biomolecules 2020, 10, 1219. [Google Scholar] [CrossRef]
- Dadi, D.; Mengistie, E.; Terefe, G.; Getahun, T.; Haddis, A.; Birke, W.; Van der Bruggen, B. Assessment of the effluent quality of wet coffee processing wastewater and its influence on downstream water quality. Ecohydrol. Hydrobiol. 2018, 18, 201–211. [Google Scholar] [CrossRef]
- Arslan, M.; Rakha, A.; Xiaobo, Z.; Mahmood, M.A. Complimenting gluten free bakery products with dietary fiber: Opportunities and constraints. Trends Food Sci. Tech. 2019, 83, 194–202. [Google Scholar] [CrossRef]
- Guglielmetti, A.; Fernandez-Gomez, B.; Zeppa, G.; Castillo, M. Nutritional quality, potential health promoting properties and sensory perception of an improved gluten-free bread formulation containing inulin, rice protein and bioactive compounds extracted from coffee byproducts. Pol. J. Food Nutr. Sci. 2019, 51, 157–166. [Google Scholar] [CrossRef]
- Rinaldi, M.; Littardi, P.; Paciulli, M.; Caligiani, A.; Chiavaro, E. Effect of cocoa bean shells granulometries on qualitative properties of gluten-free bread during storage. Eur. Food Res. Technol. 2020, 246, 1583–1590. [Google Scholar] [CrossRef]
- AOAC, Association of Official Analytical Chemists. Official Method of Analysis, 16th ed.; Association of Official Analytical: Washington, DC, USA, 2002. [Google Scholar]
- Sarangapani, C.; Thirumdas, R.; Devi, Y.; Trimukhe, A.; Deshmukh, R.R.; Annapure, U.S. Effect of low-pressure plasma on physico–chemical and functional properties of parboiled rice flour. LWT Food Sci. Technol. 2016, 69, 482–489. [Google Scholar] [CrossRef]
- AACC, American Association of Cereal Chemists. Approved Methods of the AACC, 10th ed.; American Association of Cereal Chemists: St. Paul, MN, USA, 2000. [Google Scholar]
- Rinaldi, M.; Paciulli, M.; Caligiani, A.; Scazzina, F.; Chiavaro, E. Sourdough fermentation and chestnut flour in gluten-free bread: A shelf-life evaluation. Food Chem. 2017, 224, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Bourne, M.C. Texture profile analysis. Food Technol. 1978, 32, 62–66. [Google Scholar]
- Pasqualone, A.; Delvecchio, L.N.; Mangini, G.; Taranto, F.; Blanco, A. Variability of total soluble phenolic compounds and antioxidant activity in a collection of tetraploid wheat. Agric. Food Sci. 2014, 23, 307–316. [Google Scholar] [CrossRef]
- Raghavendra, S.N.; Rastogi, N.K.; Raghavarao, K.S.M.S.; Tharanathan, R.N. Dietary fiber from coconut residue: Effects of different treatments and particle size on the hydration properties. Eur. Food Res. Technol. 2004, 218, 563–567. [Google Scholar] [CrossRef]
- Esposito, F.; Arlotti, G.; Bonifati, A.M.; Napolitano, A.; Vitale, D.; Fogliano, V. Antioxidant activity and dietary fibre in durum wheat bran by-products. Food Res. Int. 2005, 38, 1167–1173. [Google Scholar] [CrossRef]
- Sabanis, D.; Lebesi, D.; Tzia, C. Effect of dietary fibre enrichment on selected properties of gluten-free bread. LWT—Food Sci. Technol. 2009, 42, 1380–1389. [Google Scholar] [CrossRef]
- Gómez, M.; Ronda, F.; Blanco, C.A.; Caballero, P.A.; Apesteguía, A. Effect of dietary fibre on dough rheology and bread quality. Eur. Food Res. Technol. 2003, 216, 51–56. [Google Scholar] [CrossRef]
- Cauvain, S.P. Breadmaking: Improving Quality; Woodhead Publishing: Cambridge, UK, 2012; pp. 299–329. [Google Scholar]
- Gray, J.A.; Bemiller, J.N. Bread staling: Molecular basis and control. Compr. Rev. Food Sci. Food Saf. 2003, 2, 1–21. [Google Scholar] [CrossRef]
- Masure, H.G.; Fierens, E.; Delcour, J.A. Current and forward looking experimental approaches in gluten-free bread making research. J. Cereal Sci. 2016, 67, 92–111. [Google Scholar] [CrossRef]
- Gallagher, E.; Gormley, T.R.; Arendt, E.K. Crust and crumb characteristics of gluten free breads. J. Food Eng. 2003, 56, 153–161. [Google Scholar] [CrossRef]
- Cappa, C.; Lucisano, M.; Mariotti, M. Influence of Psyllium, sugar beet fibre and water on gluten-free dough properties and bread quality. Carbohydr. Polym. 2013, 98, 1657–1666. [Google Scholar] [CrossRef]
- Conte, P.; Fadda, C.; Drabińska, N.; Krupa-Kozak, U. Technological and nutritional challenges, and novelty in gluten-free breadmaking: A review. Pol. J. Food Nutr. Sci. 2019, 69, 5–21. [Google Scholar] [CrossRef]
- Saeed, N.; Khan, M.R.; Shabbir, M. Antioxidant activity, total phenolic and total flavonoid contents of whole plant extracts Torilis leptophylla L. BMC Complement. Altern. Med. 2012, 12, 221. [Google Scholar] [CrossRef]
- Zhang, Y.; An, X. Inhibitory mechanism of quercetin against the formation of 5- (hydroxymethyl)-2-furaldehyde in buckwheat flour bread by ultra-performance liquid chromatography coupled with high-resolution tandem mass spectrometry. Food Res. Int. 2017, 95, 68–81. [Google Scholar] [CrossRef]
- Favreau-Farhadi, N.; Pecukonis, L.; Barrett, A. The inhibition of maillard browning by different concentrations of rosmarinic acid and epigallocatechin-3-gallate in model, bakery, and fruit systems. J. Food Sci. 2015, 80, C2140–C2146. [Google Scholar] [CrossRef]


| L * | a * | b * | ||||||||
| C | PB | C | PB | C | PB | |||||
| Crust | Day 0 | 86.88 ± 0.69 a | 77.14 ± 1.03 a | *** | 2.48 ± 0.29 b | 3.05 ± 0.28 b | *** | 14.33 ± 0.93 a | 15.19 ± 0.85 ab | * |
| Day 1 | 85.29 ± 0.72 b | 77.24 ± 1.23 a | *** | 2.93 ± 0.32 a | 3.22 ± 0.27 ab | * | 15.12 ± 0.95 a | 15.50 ± 0.81 a | ||
| Day 3 | 85.59 ± 1.13 b | 77.04 ± 1.08 a | *** | 2.96 ± 0.45 a | 3.27 ± 0.20 a | * | 14.19 ± 0.86 a | 14.88 ± 0.65 b | * | |
| L * | a * | b * | ||||||||
| C | PB | C | PB | C | PB | |||||
| Crumb | Day 0 | 85.86 ± 0.74 a | 69.11 ± 1.96 a | *** | −0.66 ± 0.08 b | 3.38 ± 0.24 b | *** | 11.68 ± 1.08 a | 16.20 ± 0.74 a | *** |
| Day 1 | 83.74 ± 2.06 b | 69.60 ± 1.22 a | *** | −0.55 ± 0.09 a | 3.32 ± 0.19 b | *** | 10.91 ± 0.81 a | 15.91 ± 0.63 a | *** | |
| Day 3 | 86.34 ± 1.28 a | 70.12 ± 1.56 a | *** | −0.63 ± 0.10 ab | 3.67 ± 0.24 a | *** | 11.45 ± 0.46 a | 16.27 ± 0.50 a | *** | |
| CRUST | CRUMB | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hardness (N) | Hardness (N) | Resilience (%) | Cohesiveness (-) | Springiness (-) | Chewiness (N) | |||||||
| C | PB | C | PB | C | PB | C | PB | C | PB | C | PB | |
| Day 0 | 3.20 ± 0.89 a | 3.80 ± 1.11 a | 1.83 ± 0.23 a | 1.79 ± 0.35 b | 23.13 ± 1.59 a | 25.52 ± 2.46 a | 0.60 ± 0.01 a | 0.62 ± 0.03 a | 87.24 ± 3.55 b | 87.36 ± 3.11 a | 0.98 ± 0.17 a | 0.96 ± 0.19 a |
| Day 1 | 1.83 ± 0.27 b | 2.23 ± 0.57 b | 2.01 ± 0.15 a | 1.80 ± 0.38 b | 22.02 ± 2.63 a | 19.87 ± 3.42 b | 0.56 ± 0.04 ab | 0.52 ± 0.05 b | 90.05 ± 2.76 b | 84.55 ± 7.27 a | 1.07 ± 0.20 a | 0.80 ± 0.24 b |
| Day 3 | 1.54 ± 0.26 b | 1.83 ± 0.51 b | 1.88 ± 0.18 a | 2.09 ± 0.72 a | 24.52 ± 4.30 a | 19.88 ± 2.87 b | 0.56 ± 0.06 b | 0.52 ± 0.05 b | 95.62 ± 2.19 a | 85.54 ± 3.59 a | 0.96 ± 0.09 a | 0.91 ± 0.27 ab |
| TPC (mg GAE/g) | Antioxidant Capacity (%) | Oxidative Stability (min) | |||
|---|---|---|---|---|---|
| C | PB | C | PB | C | PB |
| 0.98 ± 0.07 | 1.07 ± 1.16 | 10.9 ± 0.2 | 65.6 ± 1.6 * | 493.0 ± 5.7 | 748.5 ± 12.0 ** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Littardi, P.; Rinaldi, M.; Grimaldi, M.; Cavazza, A.; Chiavaro, E. Effect of Addition of Green Coffee Parchment on Structural, Qualitative and Chemical Properties of Gluten-Free Bread. Foods 2021, 10, 5. https://doi.org/10.3390/foods10010005
Littardi P, Rinaldi M, Grimaldi M, Cavazza A, Chiavaro E. Effect of Addition of Green Coffee Parchment on Structural, Qualitative and Chemical Properties of Gluten-Free Bread. Foods. 2021; 10(1):5. https://doi.org/10.3390/foods10010005
Chicago/Turabian StyleLittardi, Paola, Massimiliano Rinaldi, Maria Grimaldi, Antonella Cavazza, and Emma Chiavaro. 2021. "Effect of Addition of Green Coffee Parchment on Structural, Qualitative and Chemical Properties of Gluten-Free Bread" Foods 10, no. 1: 5. https://doi.org/10.3390/foods10010005
APA StyleLittardi, P., Rinaldi, M., Grimaldi, M., Cavazza, A., & Chiavaro, E. (2021). Effect of Addition of Green Coffee Parchment on Structural, Qualitative and Chemical Properties of Gluten-Free Bread. Foods, 10(1), 5. https://doi.org/10.3390/foods10010005







