A Luminescence-Based Human TRPV1 Assay System for Quantifying Pungency in Spicy Foods
Abstract
1. Introduction
2. Experimental Section
2.1. Materials
2.2. Analysis of the Fluorescence Spectrum of Foods
2.3. Culture of Cells Expressing hTRPV1
2.4. Fluorescence-Based hTRPV1 Assay
2.5. Luminescence-Based Assay of hTRPV1 Activity
2.6. Sensory Evaluation of Spicy Instant Noodle Pungency
2.7. Statistical Analysis
3. Results and Discussion
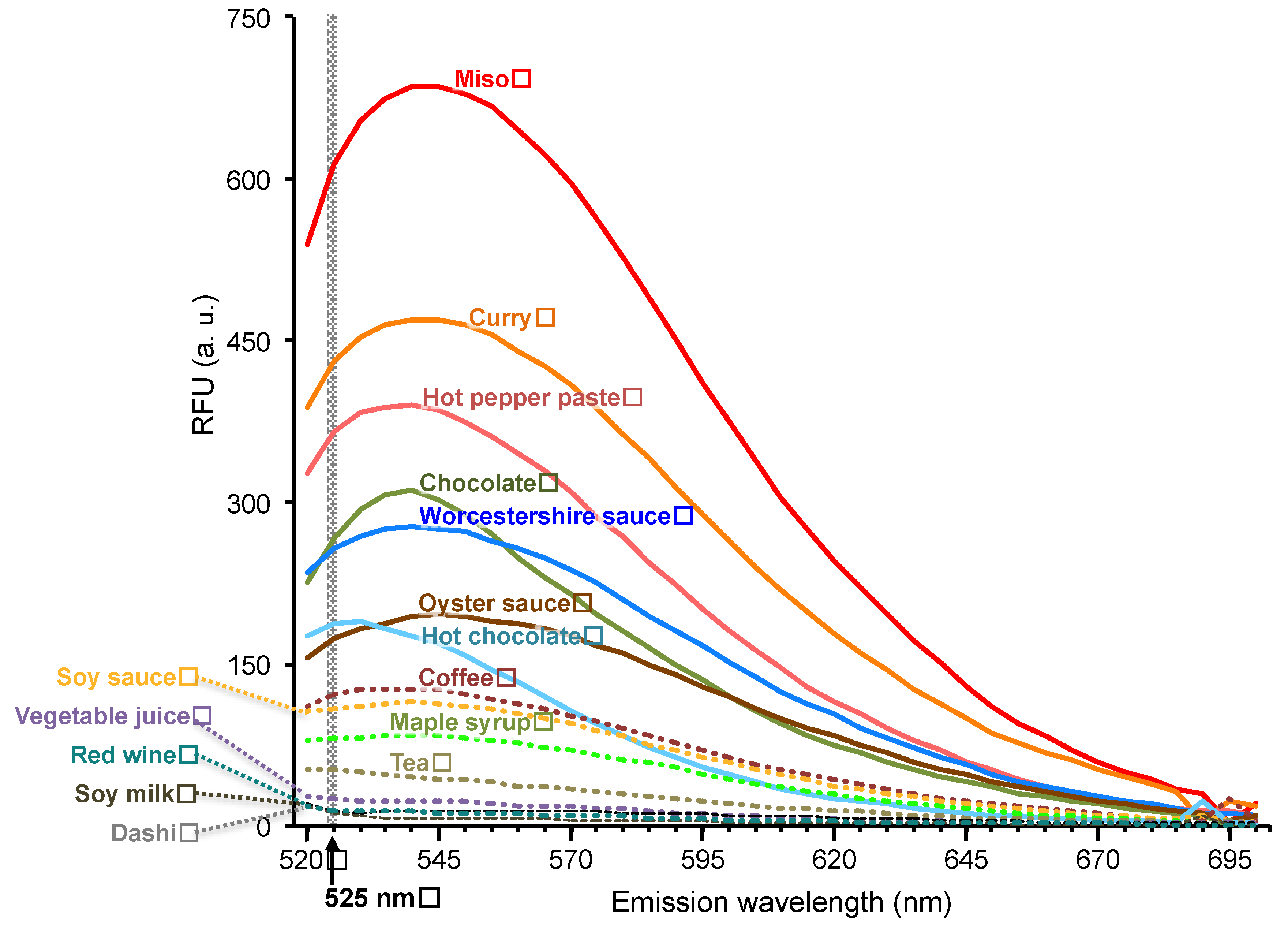
3.1. Fluorescence Spectrum of Tested Foods
3.2. Evaluation of hTRPV1 Activity Using Fluorescence and Luminescence-Based Assay Systems
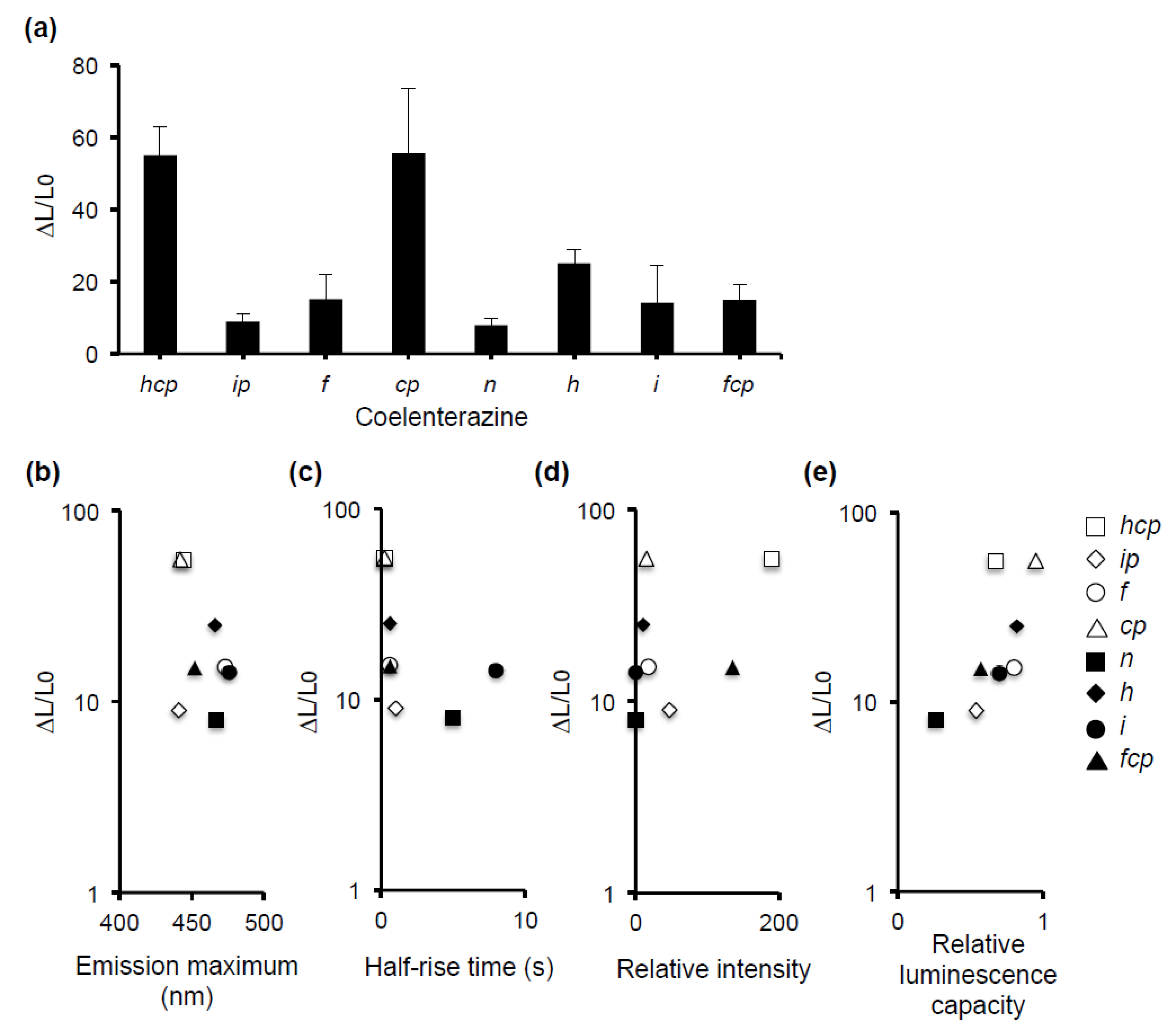
3.3. Comparison of the hTRPV1 Activity of Coelenterazines with Different Luminescence Characteristics
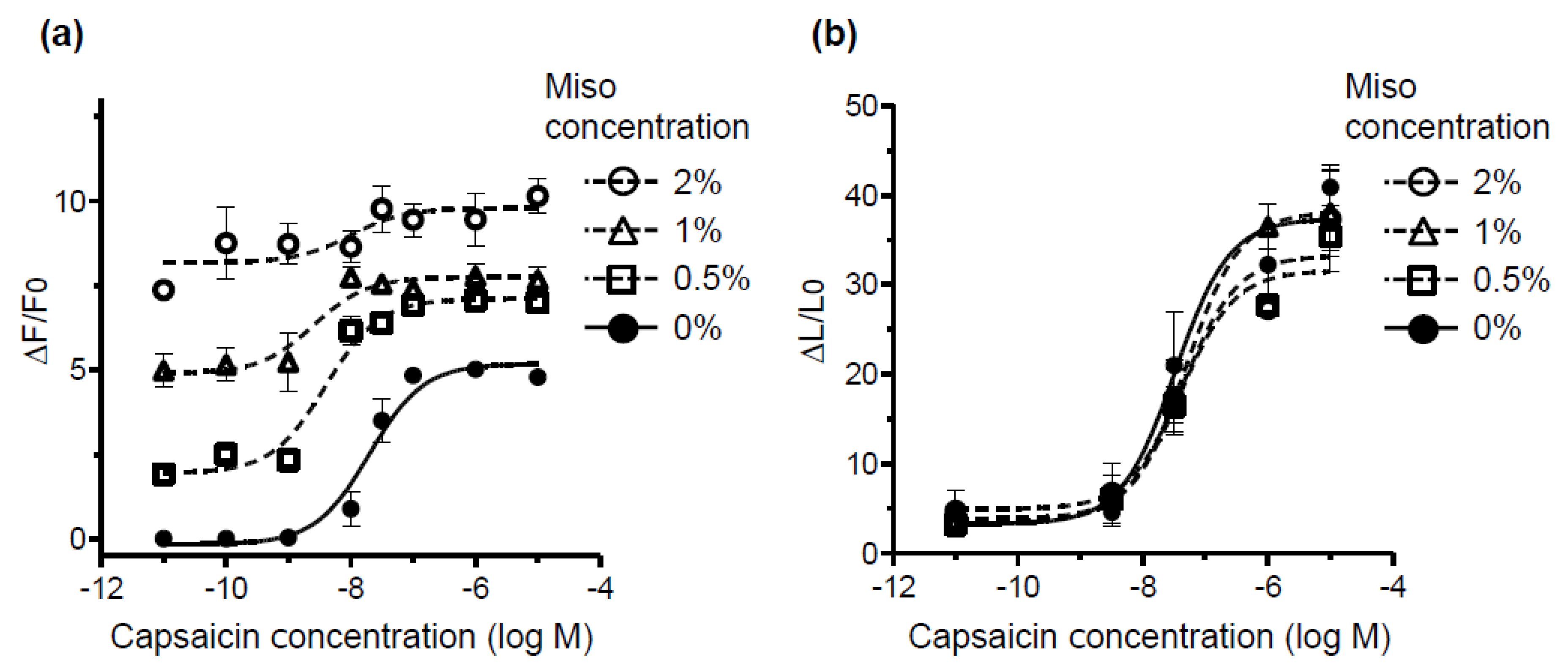
3.4. Evaluation of Capsaicin hTRPV1 Activity in the Presence of Fluorescent Substances
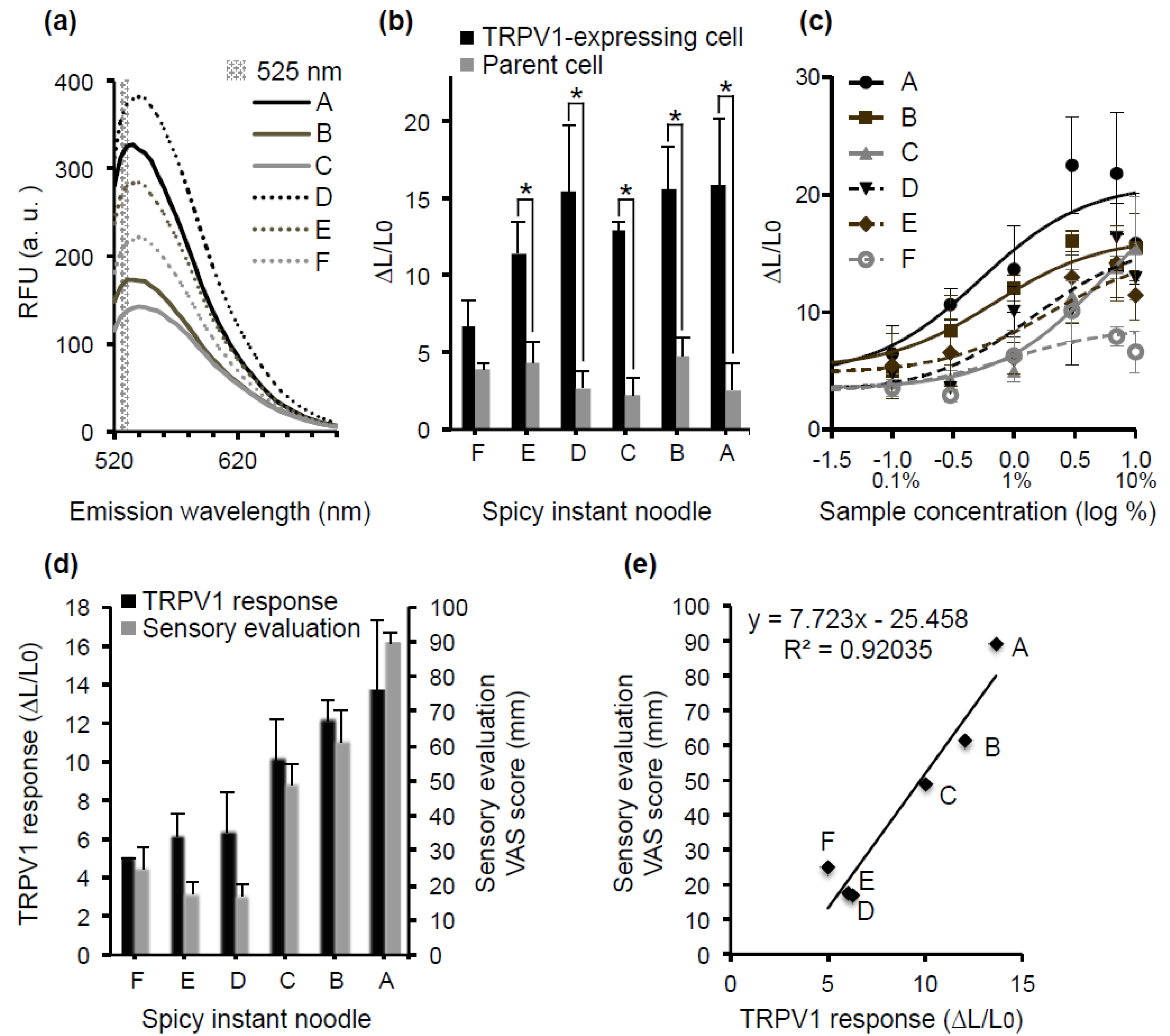
3.5. Relationship between hTRPV1 Activity and the Sensory Evaluation of Spicy Instant Noodles
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Prescott, J.; Stevenson, R.J. Pungency in food perception and preference. Food Rev. Int. 1995, 11, 665–698. [Google Scholar] [CrossRef]
- Scoville, W.L. Note on Capsicums. J. Am. Pharm. Assoc. 1912, 1, 453–454. [Google Scholar] [CrossRef]
- Aroke, E.N.; Powell-Roach, K.L.; Jaime-Lara, R.B.; Tesfaye, M.; Roy, A.; Jackson, P.; Joseph, P.V. Taste the Pain: The Role of TRP Channels in Pain and Taste Perception. Int. J. Mol. Sci. 2020, 21, 5929. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.M.; Wu, X.L.; Zhang, G.Y.; Ma, X.; He, D.X. Functional food development: Insights from TRP channels. J. Funct. Foods 2019, 56, 384–394. [Google Scholar] [CrossRef]
- Meotti, F.C.; Lemos de Andrade, E.; Calixto, J.B. TRP modulation by natural compounds. Handb. Exp. Pharmacol. 2014, 223, 1177–1238. [Google Scholar]
- McNamara, F.N.; Randall, A.; Gunthorpe, M.J. Effects of piperine, the pungent component of black pepper, at the human vanilloid receptor (TRPV1). Br. J. Pharmacol. 2005, 144, 781–790. [Google Scholar] [CrossRef]
- Okumura, Y.; Narukawa, M.; Iwasaki, Y.; Ishikawa, A.; Matsuda, H.; Yoshikawa, M.; Watanabe, T. Activation of TRPV1 and TRPA1 by black pepper components. Biosci. Biotechnol. Biochem. 2010, 74, 1068–1072. [Google Scholar] [CrossRef]
- Iwasaki, Y.; Morita, A.; Iwasawa, T.; Kobata, K.; Sekiwa, Y.; Morimitsu, Y.; Kubota, K.; Watanabe, T. A nonpungent component of steamed ginger--[10]-shogaol--increases adrenaline secretion via the activation of TRPV1. Nutr. Neurosci. 2006, 9, 169–178. [Google Scholar]
- Sugai, E.; Morimitsu, Y.; Iwasaki, Y.; Morita, A.; Watanabe, T.; Kubota, K. Pungent qualities of sanshool-related compounds evaluated by a sensory test and activation of rat TRPV1. Biosci. Biotechnol. Biochem. 2005, 69, 1951–1957. [Google Scholar] [CrossRef]
- Caterina, M.J.; Schumacher, M.A.; Tominaga, M.; Rosen, T.A.; Levine, J.D.; Julius, D. The capsaicin receptor: A heat-activated ion channel in the pain pathway. Nature 1997, 389, 816–824. [Google Scholar] [CrossRef]
- Iwasaki, Y.; Saito, O.; Tanabe, M.; Inayoshi, K.; Kobata, K.; Uno, S.; Morita, A.; Watanabe, T. Monoacylglycerols activate capsaicin receptor, TRPV1. Lipids 2008, 43, 471–483. [Google Scholar] [CrossRef]
- Su, K.H.; Lin, S.J.; Wei, J.; Lee, K.I.; Zhao, J.F.; Shyue, S.K.; Lee, T.S. The essential role of transient receptor potential vanilloid 1 in simvastatin-induced activation of endothelial nitric oxide synthase and angiogenesis. Acta. Physiol. 2014, 212, 191–204. [Google Scholar] [CrossRef] [PubMed]
- Terada, Y.; Yamashita, R.; Ihara, N.; Yamazaki-Ito, T.; Takahashi, Y.; Masuda, H.; Sakuragawa, S.; Ito, S.; Ito, K.; Watanabe, T. Human TRPA1 activation by terpenes derived from the essential oil of daidai, Citrus aurantium L. var. daidai Makino. Biosci. Biotechnol. Biochem. 2019, 83, 1721–1728. [Google Scholar] [CrossRef] [PubMed]
- Christensen, J.; Nørgaard, L.; Bro, R.; Engelsen, S.B. Multivariate autofluorescence of intact food systems. Chem. Rev. 2006, 106, 1979–1994. [Google Scholar] [CrossRef]
- Andersen, C.M.; Mortensen, G. Fluorescence spectroscopy: A rapid tool for analyzing dairy products. J. Agric. Food Chem. 2008, 56, 720–729. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Lee, B.H.; Mao, R.; Cai, A.; Jia, Y.; Clifton, H.; Schaefer, S.; Xu, L.; Zheng, J. Nicotinic acid activates the capsaicin receptor TRPV1: Potential mechanism for cutaneous flushing. Arterioscler. Thromb. Vasc. Biol. 2014, 34, 1272–1280. [Google Scholar] [CrossRef]
- Tominaga, M.; Caterina, M.J.; Malmberg, A.B.; Rosen, T.A.; Gilbert, H.; Skinner, K.; Raumann, B.E.; Basbaum, A.I.; Julius, D. The cloned capsaicin receptor integrates multiple pain-producing stimuli. Neuron 1998, 21, 531–543. [Google Scholar] [CrossRef]
- Shimomura, O.; Johnson, F.H.; Saiga, Y. Extraction, purification and properties of aequorin, a bioluminescent protein from the luminous hydromedusan, Aequorea. J. Cell Comp. Physiol. 1962, 59, 223–239. [Google Scholar] [CrossRef]
- Shimomura, O.; Johnson, F.H. Regeneration of the photoprotein aequorin. Nature 1975, 256, 236–238. [Google Scholar] [CrossRef]
- Toda, Y.; Okada, S.; Misaka, T. Establishment of a new cell-based assay to measure the activity of sweeteners in fluorescent food extracts. J. Agric. Food Chem. 2011, 59, 12131–12138. [Google Scholar] [CrossRef]
- Knight, P.J.K.; Pfeifer, T.A.; Grigliatti, T.A. A functional assay for G-protein-coupled receptors using stably transformed insect tissue culture cell lines. Anal. Biochem. 2003, 320, 88–103. [Google Scholar] [CrossRef]
- Chubanov, V.; Mederos y Schnitzler, M.; Meissner, M.; Schafer, S.; Abstiens, K.; Hofmann, T.; Gudermann, T. Natural and synthetic modulators of SK (Kca2) potassium channels inhibit magnesium-dependent activity of the kinase-coupled cation channel TRPM7. Br. J. Pharmacol. 2012, 166, 1357–1376. [Google Scholar] [CrossRef] [PubMed]
- T-REx System. Available online: https://assets.thermofisher.com/TFS-Assets/LSG/manuals/trexsystem_man.pdf (accessed on 25 November 2020).
- Terada, Y.; Horie, S.; Takayama, H.; Uchida, K.; Tominaga, M.; Watanabe, T. Activation and inhibition of thermosensitive TRP channels by voacangine, an alkaloid present in Voacanga africana, an African tree. J. Nat. Prod. 2014, 77, 285–297. [Google Scholar] [CrossRef] [PubMed]
- Gould, D.; Kelly, D.; Goldstone, L.; Gammon, J. Examining the validity of pressure ulcer risk assessment scales: Developing and using illustrated patient simulations to collect the data. J. Clin. Nurs. 2001, 10, 697–706. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.W.; Monro, J.; Lu, J.; Rush, E. The Effect of Cold Treatment of Parboiled Rice with Lowered Glycaemic Potency on Consumer Liking and Acceptability. Foods 2018, 7, 207. [Google Scholar] [CrossRef]
- Lam, P.M.; Hainsworth, A.H.; Smith, G.D.; Owen, D.E.; Davies, J.; Lambert, D.G. Activation of recombinant human TRPV1 receptors expressed in SH-SY5Y human neuroblastoma cells increases [Ca2+]i, initiates neurotransmitter release and promotes delayed cell death. J. Neurochem. 2007, 102, 801–811. [Google Scholar] [CrossRef]
- Lee, K.I.; Lee, H.T.; Lin, H.C.; Tsay, H.J.; Tsai, F.C.; Shyue, S.K.; Lee, T.S. Role of transient receptor potential ankyrin 1 channels in Alzheimer’s disease. J. Neuroinflammation 2016, 13, 92. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Zhu, Y.; Zhu, M.X.; Hu, H. Cell-based calcium assay for medium to high throughput screening of TRP channel functions using FlexStation 3. J. Vis.Exp. 2011, e3149. [Google Scholar] [CrossRef]
- Ogasawara, M.; Yamada, Y.; Egi, M. Taste enhancer from the long-term ripening of miso (soybean paste). Food Chem. 2006, 99, 736–741. [Google Scholar] [CrossRef]
- Devi, W.S.; Sarojnalini, C. Effect of Cooking on the Polyunsaturated Fatty Acid and Antioxidant Properties of Small Indigenous Fish Species of the Eastern Himalayas. Int. J. Eng. Res. Appl. 2014, 4, 146–151. [Google Scholar]
- Bozkurt, H.; Erkmen, O. Effects of production techniques on the quality of hot pepper paste. J. Food Eng. 2004, 64, 173–178. [Google Scholar] [CrossRef]
- Terasawa, N.; Murata, M.; Homma, S. Comparison of Brown Pigments in Foods by Microbial Decolorization. J. Food Sci. 1996, 61, 669–672. [Google Scholar] [CrossRef]
- Liu, D.; He, Y.; Xiao, J.; Zhou, Q.; Wang, M. The occurrence and stability of Maillard reaction products in various traditional Chinese sauces. Food Chem. 2020, in press. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Shi, Y.; Li, M.; Xiong, L.; Sun, Q. Characterization of Maillard reaction products micro/nano-particles present in fermented soybean sauce and vinegar. Sci. Rep. 2019, 9, 11285. [Google Scholar] [CrossRef]
- Havsteen, B.H. The biochemistry and medical significance of the flavonoids. Pharmacol. Ther. 2002, 96, 67–202. [Google Scholar] [CrossRef]
- Gonzalez-Barrio, R.; Nunez-Gomez, V.; Cienfuegos-Jovellanos, E.; Garcia-Alonso, F.J.; Periago-Caston, M.J. Improvement of the Flavanol Profile and the Antioxidant Capacity of Chocolate Using a Phenolic Rich Cocoa Powder. Foods 2020, 9, 189. [Google Scholar] [CrossRef]
- Wang, S.; Yamamoto, S.; Kogure, Y.; Zhang, W.; Noguchi, K.; Dai, Y. Partial Activation and Inhibition of TRPV1 Channels by Evodiamine and Rutaecarpine, Two Major Components of the Fruits of Evodia rutaecarpa. J. Nat. Prod. 2016, 79, 1225–1230. [Google Scholar] [CrossRef]
- Shimomura, O.; Musicki, B.; Kishi, Y. Semi-synthetic aequorins with improved sensitivity to Ca2+ ions. Biochem. J. 1989, 261, 913–920. [Google Scholar] [CrossRef]
- Physical Properties of Coelenterazine Analogs. Available online: https://search.cosmobio.co.jp/cosmo_search_p/search_gate2/docs/BTI_/10110.20150327.pdf (accessed on 25 November 2020).
- Dierkes, G.; Krieger, S.; Duck, R.; Bongartz, A.; Schmitz, O.J.; Hayen, H. High-performance liquid chromatography-mass spectrometry profiling of phenolic compounds for evaluation of olive oil bitterness and pungency. J. Agric. Food Chem. 2012, 60, 7597–7606. [Google Scholar] [CrossRef]
- Oi-Kano, Y.; Iwasaki, Y.; Nakamura, T.; Watanabe, T.; Goto, T.; Kawada, T.; Watanabe, K.; Iwai, K. Oleuropein aglycone enhances UCP1 expression in brown adipose tissue in high-fat-diet-induced obese rats by activating beta-adrenergic signaling. J. Nutr. Biochem. 2017, 40, 209–218. [Google Scholar] [CrossRef]
- Schneider, D.J.; Seuß-Baum, I.; Schlich, E. Relationship between pungency and food components—A comparison of chemical and sensory evaluations. Food Qual. Prefer. 2014, 38, 98–106. [Google Scholar] [CrossRef]
- Perkins, B.; Bushway, R.; Guthrie, K.; Fan, T.; Stewart, B.; Prince, A.; Williams, M. Determination of capsaicinoids in salsa by liquid chromatography and enzyme immunoassay. J. AOAC Int. 2002, 85, 82–85. [Google Scholar] [PubMed]
- Salazar, H.; Llorente, I.; Jara-Oseguera, A.; Garcia-Villegas, R.; Munari, M.; Gordon, S.E.; Islas, L.D.; Rosenbaum, T. A single N-terminal cysteine in TRPV1 determines activation by pungent compounds from onion and garlic. Nat. Neurosci. 2008, 11, 255–261. [Google Scholar] [CrossRef] [PubMed]
- Koizumi, K.; Iwasaki, Y.; Narukawa, M.; Iitsuka, Y.; Fukao, T.; Seki, T.; Ariga, T.; Watanabe, T. Diallyl sulfides in garlic activate both TRPA1 and TRPV1. Biochem. Biophys. Res. Commun. 2009, 382, 545–548. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matsuyama, M.; Terada, Y.; Yamazaki-Ito, T.; Ito, K. A Luminescence-Based Human TRPV1 Assay System for Quantifying Pungency in Spicy Foods. Foods 2021, 10, 151. https://doi.org/10.3390/foods10010151
Matsuyama M, Terada Y, Yamazaki-Ito T, Ito K. A Luminescence-Based Human TRPV1 Assay System for Quantifying Pungency in Spicy Foods. Foods. 2021; 10(1):151. https://doi.org/10.3390/foods10010151
Chicago/Turabian StyleMatsuyama, Minami, Yuko Terada, Toyomi Yamazaki-Ito, and Keisuke Ito. 2021. "A Luminescence-Based Human TRPV1 Assay System for Quantifying Pungency in Spicy Foods" Foods 10, no. 1: 151. https://doi.org/10.3390/foods10010151
APA StyleMatsuyama, M., Terada, Y., Yamazaki-Ito, T., & Ito, K. (2021). A Luminescence-Based Human TRPV1 Assay System for Quantifying Pungency in Spicy Foods. Foods, 10(1), 151. https://doi.org/10.3390/foods10010151





