Carotenoids from Foods of Plant, Animal and Marine Origin: An Efficient HPLC-DAD Separation Method
Abstract
:1. Introduction
2. Experimental Section
2.1. Reagents and Standards
2.2. Instrumentation
2.3. Extraction of Carotenoids from Various Matrices
2.3.1. Tomato Waste
2.3.2. Foods of Animal (Duck and Goose Egg Yolks) and Marine (Penaeus kerathurus Prawn) Origin
2.4. HPLC Analysis of Carotenoids
2.5. Identification and Quantification of Carotenoids
2.6. Determination of Limits of Detection (LOD) and Limits of Quantification (LOQ)
2.7. Statistical Analysis
3. Results and Discussion
3.1. HPLC Analysis of Carotenoids in Tomato Waste Extract
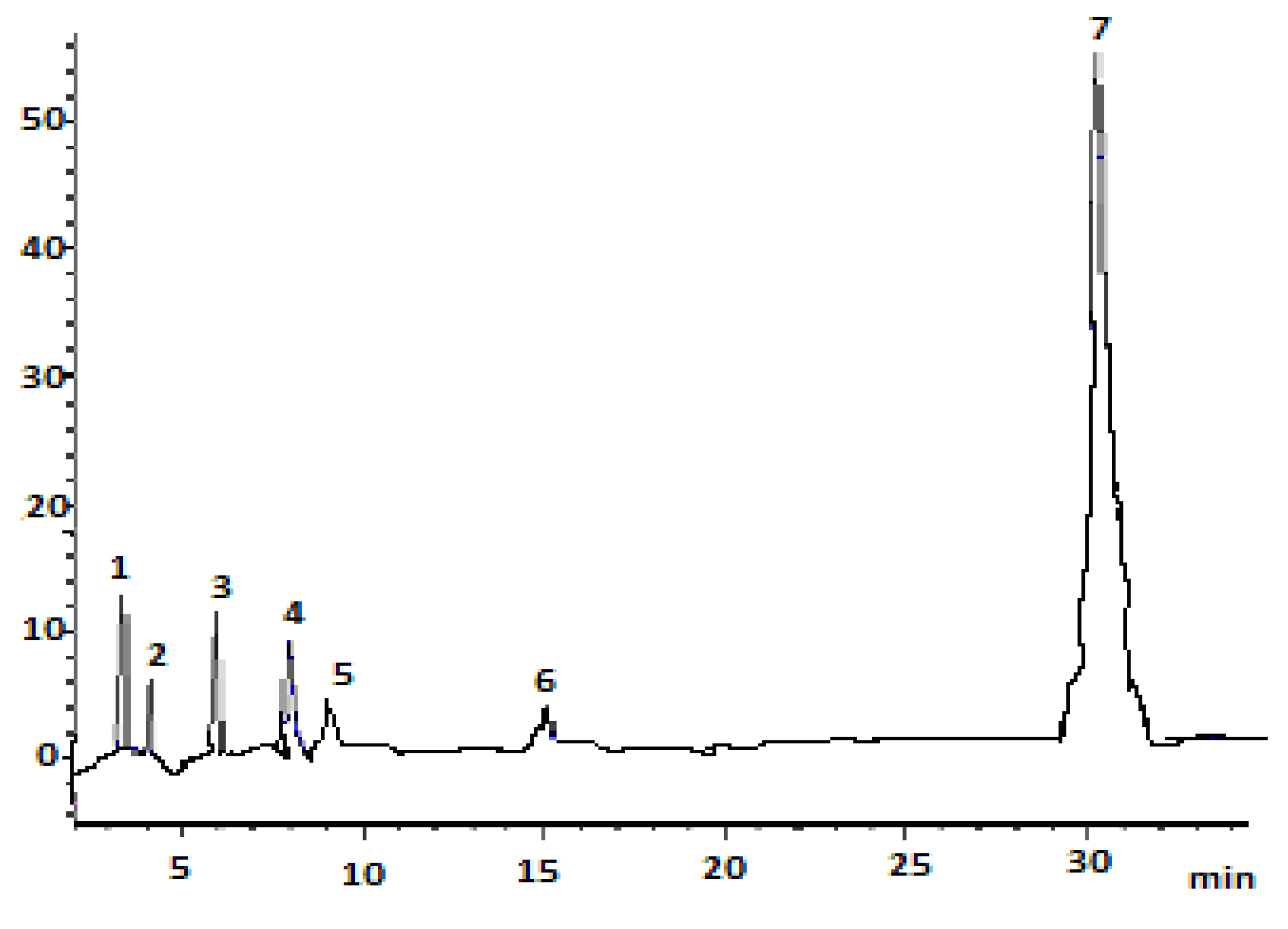
| Peak No. | Compound | RT(min) | λ (nm)Found | λ (nm) Reported | Q-Ratio Found | Q-Ratio Reported | k | α | Content (μg/100 g Dry Basis ) |
|---|---|---|---|---|---|---|---|---|---|
| [7,13,14,16,18,19] | [7,13,14,16,18,19] | ||||||||
| 1 | All- trans-lutein | 3.37 ± 0.04 | 423, 447, 477 | 422, 446, 476 | 0.04 | 0.06 | 0.53 | 2.06 | 39.14 ± 0.21 |
| 2 | 9- cis-lutein | 4.94 ± 0.06 | 350, 420, 442, 474 | 356, 428, 446, 476 | 0.10 | 0.12 | 1.24 | 1.32 | 17.59 ± 0.05 |
| 3 | 13 -cis-lutein | 5.91 ±0.09 | 376, 437, 458, 485 | 374, 434, 458, 488 | 0.31 | 0.33 | 1.69 | 1.51 | 42.69 ± 0.03 |
| 4 | All- trans-β-carotene | 7.97 ± 0.03 | 428, 454, 482 | 458, 482 | - | 0.12 | 2.62 | 1.15 | 48.48 ± 0.92 |
| 5 | 9 -cis-β-carotene | 8.90 ± 0.08 | 340, 449, 480 | 344, 452, 476 | 0.11 | 0.12 | 3.04 | 1.88 | 4.24 ± 0.01 |
| 6 | 13- cis-β-carotene | 15.09 ± 0.05 | 345, 451, 479 | 344, 422, 458, 476 | 0.34 | 0.35 | 5.86 | 2.15 | 4.42 ± 0.01 |
| 7 | All- trans-lycopene | 30.23 ± 0.20 | 450, 476, 507 | 452, 476, 506 | - | 0.06 | 12.74 | 2.15 | 64.84 ± 0.87 |
3.2. HPLC Analysis of Carotenoids in Avian (Duck and Goose) Egg Yolks
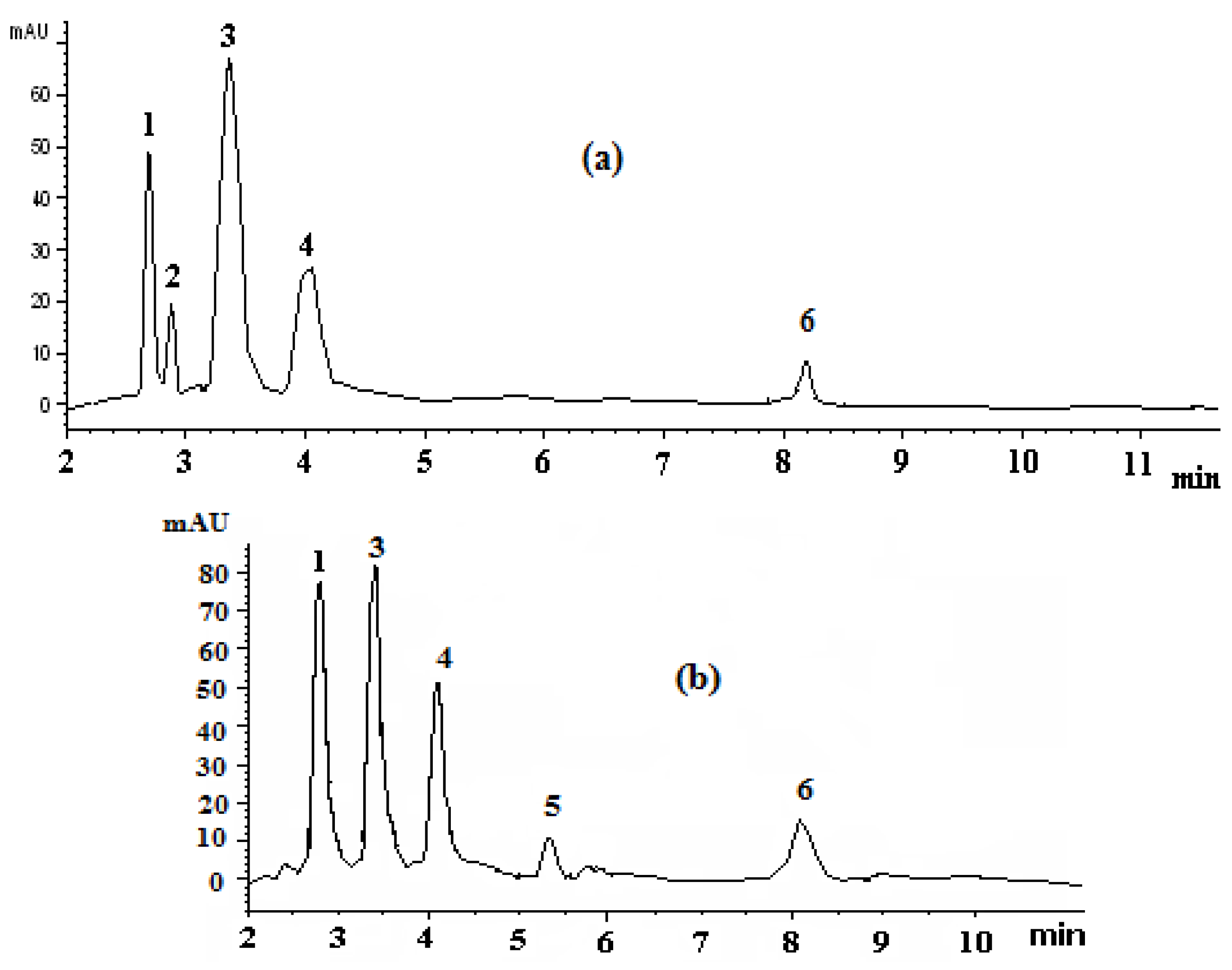
| Peak No. | Compound | RT(min) | λ (nm) found | λ (nm) reported | Q-Ratio Found | Q-Ratio Reported | k | α | Content (mg/100 g Wet Weight) [ 11] | |
|---|---|---|---|---|---|---|---|---|---|---|
| [7,13,16,20,21,22] | [7,13,16,20,21,22] | Duck | Goose | |||||||
| 1 | All- trans zeaxanthin | 2.75 ± 0.08 | 429, 450, 478 | 424, 454, 478 | 0.08 | 0.06 | 0.60 | 1.11 | 3.52 ± 0.80 | 6.22 ± 1.53 |
| 2 | Neolutein ( cis isomer of lutein) | 2.82 ± 0.02 | 330, 422, 443, 471 | 332, 442 | 0.24 | - | 0.63 | 1.77 | 1.50 ± 0.49 | - |
| 3 | All- trans lutein | 3.36 ± 0.05 | 425, 448, 476 | 426, 448, 472 | 0.08 | 0.06 | 0.89 | 1.57 | 9.88 ± 1.10 | 5.16 ± 0.61 |
| 4 | All- trans canthaxanthin | 4.07 ± 0.06 | 428, 454, 480 | 428, 452, 478 | 0.10 | 0.08 | 1.02 | 1.60 | 4.76 ± 3.41 | 3.84 ± 2.64 |
| 5 | β-Cryptoxanthin | 5.24 ± 0.02 | 428, 450, 477 | 428, 454, 480 | 0.17 | 0.16 | 1.47 | 1.92 | - | 1.17 ± 0.17 * |
| 6 | All- trans-β-carotene | 8.12 ± 0.02 | 433, 455, 481 | 426, 454, 478 | 0.08 | 0.12 | 2.83 | 1.92 | ** | 0.27 ± 0.02 |
3.3. HPLC Analysis of Shrimp Carotenoids

| Peak No. | Compound | RT(min) | λ (nm) Found | λ (nm) Reported | Q-Ratio Found | Q-Ratio Reported | k | α | mg/100 g Cephalothorax | mg/100 g Muscle |
|---|---|---|---|---|---|---|---|---|---|---|
| [20,21,23,24,25,26] | [20,21,23,24,25,26] | |||||||||
| 1 | All- trans zeaxanthin | 2.53 ± 0.03 | 429, 450, 478 | 424, 454, 478 | 0.09 | 0.06 | 0.37 | 1.0 | 2.29 ± 0.06 | 0.52 ± 0.03 |
| 2 | All- trans lutein | 2.96 ± 0.06 | 425, 448, 476 | 426, 448, 472 | 0.08 | 0.06 | 0.60 | 1.63 | 2.65 ± 0.05 | 0.32 ± 0.02 |
| 3 | All- trans canthaxanthin | 4.55 ± 0.09 | 428, 454, 480 | 428, 452, 478 | 0.11 | 0.08 | 1.45 | 2.43 | 4.12 ± 0.07 | 0.73 ± 0.03 |
| 4 | β-Cryptoxanthin | 5.26 ± 0.03 | 428, 450, 477 | 425, 454, 478 | 0.04 | 0.05 | 1.83 | 1.26 | 0.37 ± 0.02 a | - |
| 5 | α -Cryptoxanthin | 5.95 ± 0.05 | 427, 447, 472 | 423, 446, 473 | - | 0.06 | 2.21 | 1.20 | ||
| 6 | Unidentified astaxanthin cis-isomer | 6.74 ± 0.05 | 474 | - | 0.18 | - | 2.63 | 1.19 | 2.70 ± 0.05 b | 0.42 ± 0.02 b |
| 7 | Unidentified astaxanthin cis-isomer | 7.21 ± 0.07 | 475 | - | 0.11 | - | 2.89 | 1.10 | ||
| 8 | Unidentified astaxanthin cis-isomer | 7.84 ± 0.04 | 475 | - | 0.07 | - | 3.23 | 1.12 | ||
| 9 | (3 R,3′R)-trans-astaxanthin | 8.83 ± 0.09 | 478 | 477.6 | - | - | 3.76 | 1.16 | ||
| 10 | Unidentified astaxanthin cis-isomer | 11.39 ± 0.06 | 474 | - | 0.11 | - | 5.14 | 1.37 | ||
| 11 | (3 S,3′S)-trans-astaxanthin | 11.98 ± 0.03 | 479 | 478.8 | - | - | 5.46 | 1.06 | ||
| 12 | Unidentified astaxanthin ester | 16.07 ± 0.07 | 481 | 482.5 | - | - | 7.67 | 1.40 | 0.80 ± 0.05 | 0.14 ± 0.01 |
| 13 | Unidentified astaxanthin ester | 20.05 ± 0.05 | 483 | 482.5 | - | - | 9.82 | 1.28 | 0.37 ± 0.03 | 0.07 ± 0.01 |
4. Conclusions
Conflicts of Interest
References
- Babu, C.M.; Chakrabarti, R.; Sambasivarao, K.R.S. Enzymatic isolation of carotenoid-protein complex from shrimp head waste and its use as a source of carotenoids. LWT Food Sci. Technol. 2008, 41, 227–235. [Google Scholar] [CrossRef]
- Bernal, J.; Mendiola, J.A.; Ibáñez, E.; Cifuentes, A. Advanced analysis of nutraceuticals. J. Pharm. Biomed. Anal. 2011, 55, 758–774. [Google Scholar] [CrossRef]
- Rao, A.V.; Agarwal, S. Role of lycopene as antioxidant carotenoid in the prevention of chronic diseases: A review. Nutr. Res. 1999, 19, 305–323. [Google Scholar] [CrossRef]
- Miller, N.J.; Sampson, J.; Candeias, L.P.; Bramley, P.M.; Rice-Evans, C.A. Antioxidant activities of carotenes and xanthophylls. FEBS Lett. 1996, 384, 240–242. [Google Scholar] [CrossRef]
- McNamara, D.J.; Thesmar, H.S. Eggs; Egg Nutrition Center: Washington, DC, USA, 2005. [Google Scholar]
- Sánchez-Camargo, A.P.; Meireles, M.A.A.; Lopes, B.L.F.; Cabral, F.A. Proximate composition and extraction of carotenoids and lipids from Brazilian red spotted shrimp waste (Farfantepenaeus paulensis). J. Food Eng. 2011, 102, 87–93. [Google Scholar] [CrossRef]
- Lin, C.H.; Chen, B.H. Determination of carotenoids in tomato juice by liquid chromatography. J. Chromatogr. A 2003, 1012, 103–109. [Google Scholar] [CrossRef]
- Chen, J.P.; Tai, C.Y.; Chen, B.H. Improved liquid chromatographic method for determination of carotenoids in Taiwanese mango (Mangifera indica L.). J. Chromatogr. A 2004, 1054, 261–268. [Google Scholar]
- Melėndez-Martínez, A.J.; Vicario, I.M.; Heredia, F.J. Review: Analysis of carotenoids in orange juice. J. Food Compos. Anal. 2007, 20, 638–649. [Google Scholar] [CrossRef]
- Strati, I.F.; Oreopoulou, V. Process optimisation for recovery of carotenoids from tomato waste. Food Chem. 2011, 129, 747–752. [Google Scholar]
- Sinanoglou, V.J.; Strati, I.F.; Miniadis-Meimaroglou, S. Lipid, fatty acid and carotenoid content of edible egg yolks from avian species: A comparative study. Food Chem. 2011, 124, 971–977. [Google Scholar] [CrossRef]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar]
- Lee, M.T.; Chen, B.H. Separation of lycopene and its cis isomers by liquid chromatography. Chromatographia 2001, 54, 613–617. [Google Scholar] [CrossRef]
- Chen, B.H.; Peng, H.Y.; Chen, H.E. Changes of carotenoids, color and vitamin A contents during processing of carrot juice. J. Agric. Food Chem. 1995, 43, 1912–1918. [Google Scholar]
- Champion, B. Tips and Tricks of HPLC Separations; Agilent Technologies, Inc.: Santa Clara, CA, USA, 2011. [Google Scholar]
- Tai, C.Y.; Chen, B.H. Analysis and stability of carotenoids in the flowers of daylily (Hemerocallis disticha) as affected by various treatments. J. Agric. Food Chem. 2000, 48, 5962–5968. [Google Scholar] [CrossRef]
- U.S. Department of Health and Human ServicesFood and Drug AdministrationCenter for Drug Evaluation and Research (CDER)Center for Biologics Evaluation and Research (CBER)Guidance for Industry: Q2B Validation of Analytical Procedures: Methodology; Center for Drug Evaluation and Research: Rockville, MD, USA, 1996.
- Saleh, M.H.; Tan, B. Separation and identification of cis/trans carotenoid isomers. J. Agric. Food Chem. 1991, 39, 1438–1443. [Google Scholar] [CrossRef]
- Schierle, J.; Bretzel, W.; Bühler, I.; Faccin, N.; Hess, D.; Steiner, K.; Schüep, W. Content and isomeric ratio of lycopene in food and human blood plasma. Food Chem. 1997, 59, 459–465. [Google Scholar] [CrossRef]
- Hashimoto, H.; Miki, Y.; Kuki, M.; Shimamura, T.; Utsumi, H.; Koyama, Y. Isolation by high-pressure liquid chromatography of the cis-trans isomers of β-apo-8′-carotenal, determination of their S0-state configuration by NMR spectroscopy, and prediction of their S1- and T1-state configuration by transient Raman spectroscopy. J. Am. Chem. Soc. 1993, 115, 9216–9225. [Google Scholar] [CrossRef]
- Inbaraj, B.S.; Lu, H.; Hung, C.F.; Wu, W.B.; Lin, C.L.; Chen, B.H. Determination of carotenoids and their esters in fruits of Lycium barbarum Linnaeus by HPLC-DAD-APCI-MS. J. Pharm. Biomed. Anal. 2008, 47, 812–818. [Google Scholar] [CrossRef]
- Khachik, F.; Spangler, C.J.; Smith, J.C., Jr.; Canfield, L.M.; Steck, A.; Pfander, H. Identification, quantification, and relative concentrations of carotenoids and their metabolites in human milk and serum. Anal. Chem. 1997, 69, 1873–1881. [Google Scholar] [CrossRef]
- Liu, H.L.; Kao, T.H.; Chen, B.H. Determination of carotenoids in the chinese medical herb Jiao-Gu-Lan (Gynostemma Pentaphyllum MAKINO) by liquid chromatography. Chromatographia 2004, 60, 411–417. [Google Scholar] [CrossRef]
- Gentili, A.; Caretti, F. Evaluation of a method based on liquid chromatography-diode array detector-tandem mass spectrometry for a rapid and comprehensive characterization of the fat-soluble vitamin and carotenoid profile of selected plant foods. J. Chromatogr. A 2011, 1218, 684–697. [Google Scholar] [CrossRef]
- Rajendran, V.; Pu, Y.S.; Chen, B.H. An improved HPLC method for determination of carotenoids in human serum. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2005, 824, 99–106. [Google Scholar] [CrossRef]
- Yuan, J.-P.; Chen, F. Identification of astaxanthin isomers in Haematococcus lacustris by HPLC-photodiode array detection. Biotechnol. Tech. 1997, 11, 455–459. [Google Scholar] [CrossRef]
- Turujman, S.A. Rapid direct resolution of the stereoisomers of all-trans astaxanthin on a Pirkle covalent L-leucine column. J. Chromatogr. 1993, 631, 197–199. [Google Scholar] [CrossRef]
- Carotenoid Pigmentation of Salmonid Fishes—Recent Progress. Available online: http://www.aces.edu/dept/fisheries/education/pond_to_plate/documents/CaroteniodPigmentationinTroutNorway.pdf (accessed on 15 October 2012).
- Sachindra, N.M.; Bhaskar, N.; Mahendrakar, N.S. Carotenoids in different body components of Indian shrimps. J. Sci. Food Agric. 2005, 85, 167–172. [Google Scholar] [CrossRef]
- Howell, B.K.; Matthews, A.D. The carotenoids of wild and blue disease affected farmed tiger shrimp (Penaeus monodon, Fabricius). Comp. Biochem. Physiol. B Comp. Biochem. 1991, 98, 375–379. [Google Scholar] [CrossRef]
- Yanar, Y.; Çelik, M.; Yanar, M. Seasonal changes in total carotenoid contents of wild marine shrimps (Penaeus semisulcatus and Metapenaeus monoceros) inhabitating the eastern Mediterranean. Food Chem. 2004, 88, 267–269. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Strati, I.F.; Sinanoglou, V.J.; Kora, L.; Miniadis-Meimaroglou, S.; Oreopoulou, V. Carotenoids from Foods of Plant, Animal and Marine Origin: An Efficient HPLC-DAD Separation Method. Foods 2012, 1, 52-65. https://doi.org/10.3390/foods1010052
Strati IF, Sinanoglou VJ, Kora L, Miniadis-Meimaroglou S, Oreopoulou V. Carotenoids from Foods of Plant, Animal and Marine Origin: An Efficient HPLC-DAD Separation Method. Foods. 2012; 1(1):52-65. https://doi.org/10.3390/foods1010052
Chicago/Turabian StyleStrati, Irini F., Vassilia J. Sinanoglou, Lintita Kora, Sofia Miniadis-Meimaroglou, and Vassiliki Oreopoulou. 2012. "Carotenoids from Foods of Plant, Animal and Marine Origin: An Efficient HPLC-DAD Separation Method" Foods 1, no. 1: 52-65. https://doi.org/10.3390/foods1010052






