Cannabidiol in Dentistry: A Scoping Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Eligibility Criteria
2.2. Information Sources and Search
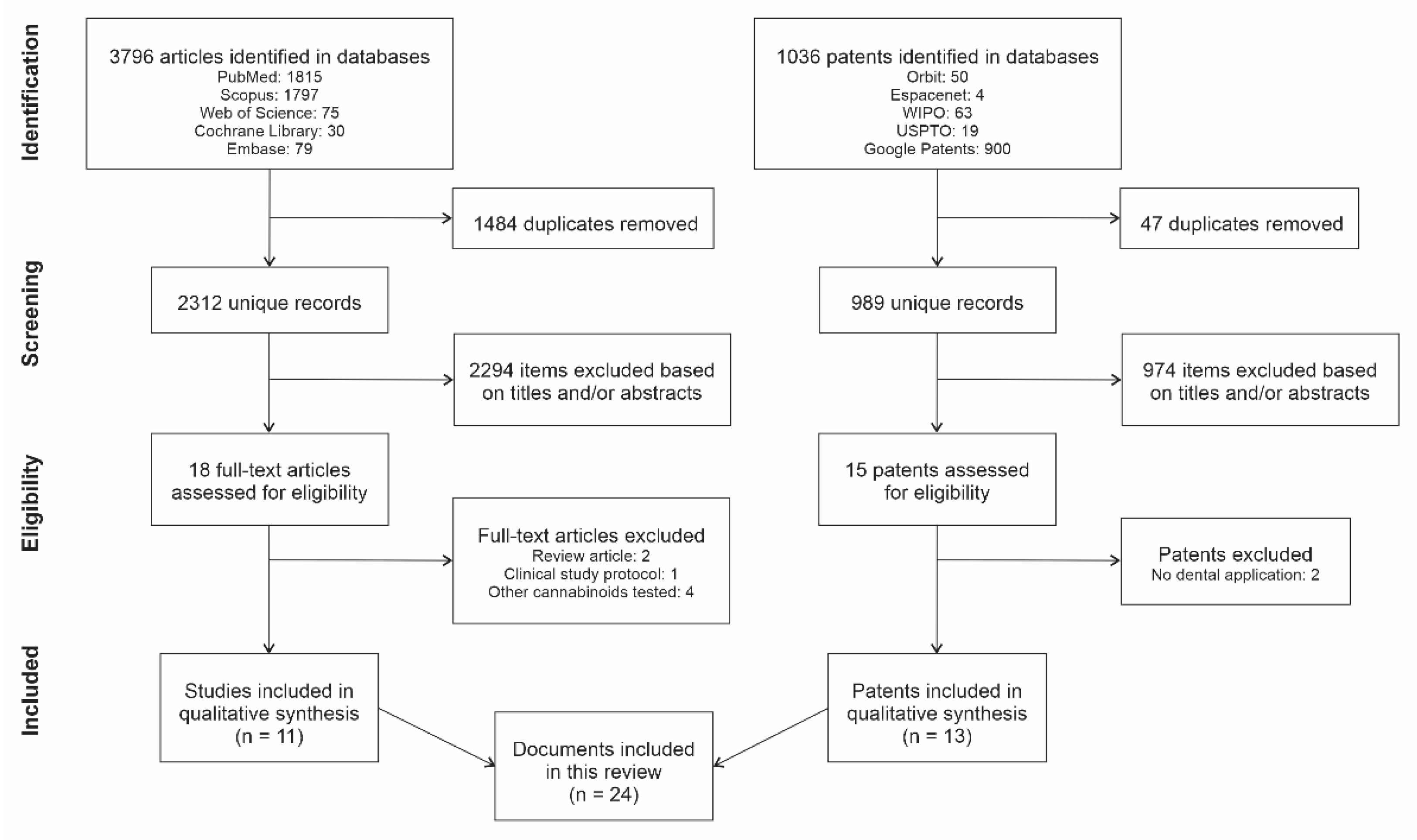
2.3. Selection of Sources of Evidence
2.4. Data Charting, Data Items, and Analysis
3. Results
3.1. Characteristics of Studies
3.2. Characteristics of Patents
3.3. Synthesis of Results and Summary of Evidence
4. Discussion
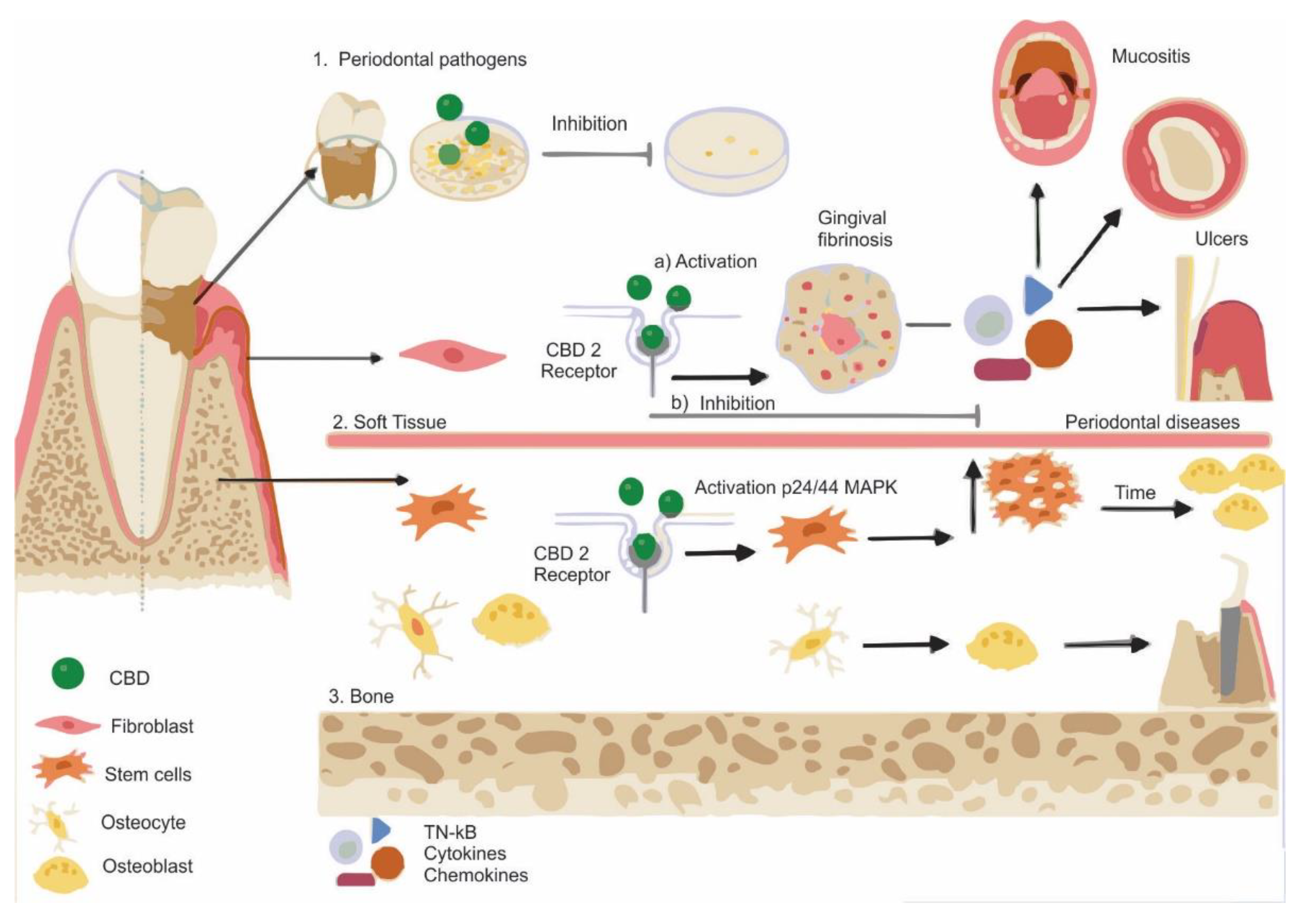
4.1. Periodontal Therapy
4.2. Oral Medicine
4.3. Adjuncts to Oral Surgery and Traumatology
4.4. The Scientific and Technological Scenario
4.5. Limitations
4.6. Future Challenges
- Studying of the behavior, dosage, and mechanisms of action of CDB used in dentistry-related clinical conditions;
- Acceptability of CBD-based treatments among dental patients;
- Standardization of the methods used for extraction of CBD and standardization of in vitro and in vivo tests with this compound [46];
- Evaluation of the cytocompatibility of CBD in order to develop safe products;
- More basic studies are needed to increase safety of CBD for use in dental patients;
- Since CBD has been approved for use in patients in many countries, clinical studies with patients are also needed in dentistry, which means that the available evidence is of low quality, low certainty, and prone to risk of bias.
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Rossi, F.; Siniscalco, D.; Luongo, L.; De Petrocellis, L.; Bellini, G.; Petrosino, S.; Torella, M.; Santoro, C.; Nobili, B.; Perrotta, S.; et al. The endovanilloid/endocannabinoid system in human osteoclasts: Possible involvement in bone formation and resorption. Bone 2009, 44, 476–484. [Google Scholar] [CrossRef] [PubMed]
- Pisanti, S.; Malfitano, A.M.; Ciaglia, E.; Lamberti, A.; Ranieri, R.; Cuomo, G.; Abate, M.; Faggiana, G.; Proto, M.C.; Fiore, D.; et al. Cannabidiol: State of the art and new challenges for therapeutic applications. Pharmacol. Ther. 2017, 175, 133–150. [Google Scholar] [CrossRef] [PubMed]
- Apostu, D.; Lucaciu, O.; Mester, A.; Benea, H.; Oltean-Dan, D.; Onisor, F.; Baciut, M.; Bran, S. Cannabinoids and bone regeneration. Drug Metab. Rev. 2019, 51, 65–75. [Google Scholar] [CrossRef]
- Amin, M.R.; Ali, D.W. Pharmacology of Medical Cannabis. Adv. Exp. Med. Biol. 2019, 1162, 151–165. [Google Scholar]
- Burstein, S. Cannabidiol (CBD) and its analogs: A review of their effects on inflammation. Bioorg. Med. Chem. 2015, 23, 1377–1385. [Google Scholar] [CrossRef] [PubMed]
- Pavanini, S.; Lauriola, S.; Mottola, A.; Cocchi, R. Il sistema endocannabinoide: Attuali conoscenze e potenziali applicazioni in odontostomatologia. Ital. Oral Surg. 2011, 10, 66–86. [Google Scholar] [CrossRef]
- Kamali, A.; Oryan, A.; Hosseini, S.; Ghanian, M.H.; Alizadeh, M.; Eslaminejad, M.B.; Baharvand, H. Cannabidiol-loaded microspheres incorporated into osteoconductive scaffold enhance mesenchymal stem cell recruitment and regeneration of critical-sized bone defects. Mater. Sci. Eng. C 2019, 101, 64–75. [Google Scholar] [CrossRef] [PubMed]
- Kogan, N.M.; Melamed, E.; Wasserman, E.; Raphael, B.; Breuer, A.; Stok, K.S.; Sondergaard, R.; Escudero, A.V.; Baraghithy, S.; Attar-Namdar, M.; et al. Cannabidiol, a major non-psychotropic cannabis constituent enhances fracture healing and stimulates lysyl hydroxylase activity in osteoblasts. J. Bone Miner. Res. 2015, 30, 1905–1913. [Google Scholar] [CrossRef] [PubMed]
- Whyte, L.S.; Ford, L.; Ridge, S.; Cameron, G.; Rogers, M.; Ross, R. Cannabinoids and bone: Endocannabinoids modulate human osteoclast function in vitro. Br. J. Pharmacol. 2012, 165, 2584–2597. [Google Scholar] [CrossRef]
- Jung, B.; Lee, J.K.; Kim, J.; Kang, E.K.; Han, S.Y.; Lee, H.Y.; Choi, I.S. Synthetic strategies for (-)-cannabidiol and its structural analogs. Chem. Asian J. 2019, 14, 3749–3762. [Google Scholar] [CrossRef]
- Technavio. Cannabidiol Market by Source and Geography-Forecast and Analysis 2021-025. 27 October 2021. Available online: https://www.technavio.com/report/cbd-oil-market-industry-analysis (accessed on 14 December 2021).
- Cuba, L.F.; Salum, F.G.; Guimarães, F.S.; Cherubini, K.; Borghetti, R.L.; de Figueiredo, M.A. Cannabidiol on 5-FU-induced oral mucositis in mice. Oral Dis. 2020, 26, 1483–1493. [Google Scholar] [CrossRef] [PubMed]
- Konermann, A.; Jäger, A.; Held, S.A.; Brossart, P.; Schmöle, A. In vivo and in vitro identification of endocannabinoid signaling in periodontal tissues and their potential role in local pathophysiology. Cell. Mol. Neurobiol. 2017, 37, 1511–1520. [Google Scholar] [CrossRef] [PubMed]
- Miyashita, K.; Oyama, T.; Sakuta, T.; Tokuda, M.; Torii, M. Anandamide induces matrix metalloproteinase-2 production through cannabinoid-1 receptor and transient receptor potential vanilloid-1 in human dental pulp cells in culture. J. Endod. 2012, 38, 786–790. [Google Scholar] [CrossRef] [PubMed]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; Moher, D.; Peters, M.D.J.; Horsley, T.; Weeks, L.; et al. PRISMA extension for scoping reviews (PRISMA-ScR): Checklist and explanation. Ann. Intern. Med. 2018, 169, 467–473. [Google Scholar] [CrossRef]
- Peters, M.D.; Godfrey, C.M.; Khalil, H.; McInerney, P.; Parker, D.; Soares, C.B. Guidance for conducting systematic scoping reviews. Int. J. Evid. Based Healthc. 2015, 13, 141–146. [Google Scholar] [CrossRef]
- Levac, D.; Colquhoun, H.; O’Brien, K.K. Scoping studies: Advancing the methodology. Implement. Sci. 2010, 5, 69. [Google Scholar] [CrossRef]
- Napimoga, M.H.; Benatti, B.B.; Lima, F.O.; Alves, P.M.; Campos, A.C.; Pena-Dos-Santos, D.R.; Severino, F.P.; Cunha, F.Q.; Guimarães, F.S. Cannabidiol decreases bone resorption by inhibiting RANK/RANKL expression and pro-inflammatory cytokines during experimental periodontitis in rats. Int. Immunopharmacol. 2009, 9, 216–222. [Google Scholar] [CrossRef]
- Klein, M.; de Quadros De Bortolli, J.; Guimarães, F.S.; Salum, F.G.; Cherubini, K.; de Figueiredo, M.A. Effects of cannabidiol, a Cannabis sativa constituent, on oral wound healing process in rats: Clinical and histological evaluation. Phytother. Res. 2018, 32, 2275–2281. [Google Scholar] [CrossRef]
- Cuba, L.F.; Salum, F.; Cherubini, K.; Figueiredo, M. Cannabidiol: An alternative therapeutic agent for oral mucositis? J. Clin. Pharm. Ther. 2017, 42, 245–250. [Google Scholar] [CrossRef]
- Scionti, D.; Diomede, F.; Marchisio, M.; Grassi, G.; Pollastro, F.; Piattelli, A.; Piattelli, A.; Bramanti, P.; Mazzon, E.; Trubiani, O. Cannabidiol modulates the immunophenotype and inhibits the activation of the inflammasome in human gingival mesenchymal stem cells. Front. Physiol. 2016, 7, 559. [Google Scholar]
- Gu, Z.; Singh, S.; Niyogi, R.G.; Lamont, G.J.; Wang, H.; Lamont, R.J.; Scott, D.A. Marijuana-derived cannabinoids trigger a CB2/PI3K axis of suppression of the innate response to oral pathogens. Front. Immunol. 2019, 10, 2288. [Google Scholar] [CrossRef] [PubMed]
- Ossola, C.A.; Surkin, P.N.; Mohn, C.E.; Elverdin, J.C.; Fernández-Solari, J. Anti-inflammatory and osteoprotective effects of cannabinoid-2 receptor agonist HU-308 in a rat model of lipopolysaccharide-induced periodontitis. J. Periodontol. 2016, 87, 725–734. [Google Scholar] [CrossRef] [PubMed]
- Rawal, S.; Dabbous, M.K.; Tipton, D. Effect of cannabidiol on human gingival fibroblast extracellular matrix metabolism: MMP production and activity, and production of fibronectin and transforming growth factor β. J. Periodontal Res. 2012, 47, 320–329. [Google Scholar] [CrossRef] [PubMed]
- Stahl, V.; Vasudevan, K. Comparison of efficacy of cannabinoids versus commercial oral care products in reducing bacterial content from dental plaque: A preliminary observation. Cureus 2020, 12, e6809. [Google Scholar] [CrossRef]
- Petrescu, N.B.; Jurj, A.; Sorițău, O.; Lucaciu, O.P.; Dirzu, N.; Raduly, L.; Berindan-Neagoe, I.; Cenariu, M.; Boșca, B.A.; Campian, R.S.; et al. Cannabidiol and vitamin D3 impact on osteogenic differentiation of human dental mesenchymal stem cells. Medicina 2020, 56, 607. [Google Scholar] [CrossRef]
- Anastassov, G.; Lekhram, C. Oral Care Composition Comprising Cannabinoids. U.S.Patent US20190076349, US10172786, EP3233021, EP3233021, WO2016/100516, US20160166498, 15 December 2014. [Google Scholar]
- Ke, Z.; Xin, T.; Chaohui, Y.; Meng, L.; Tanran, C.; Qian, J. Use of the Tooth Whitening CBD. China Patent CN109939012, 12 December 2017. [Google Scholar]
- Ke, Z.; Xin, T.; Chaohui, Y.; Meng, L.; Tanran, C.; Qian, J. Composition Comprising a Cannabinoid Toothpaste and its Preparation Method. China Patent CN109939011, 20 December 2017. [Google Scholar]
- Curatola, G.P. Oral Care Formulations and Methods for Use. U.S. Patent WO2019/055420, US20190076343, 14 September 2017. [Google Scholar]
- Stahl, V. Cannabis and Derivatives Thereof for the Treatment of Pain and Inflammation Related with Dental Pulp and Bone Regeneration Related to Dental Jaw Bone Defects. U.S. Patent WO2019030762A2, EP3664795A2, US20200222361A1, 8 September 2018. [Google Scholar]
- Ke, Z.; Xin, T.; Zhaohui, Y.; Meng, L.; Calm, C.; Qian, J. A kind of Toothpaste and Preparation Method Thereof Containing Cannabinoid. China Patent CN109939011A, 28 June 2019. [Google Scholar]
- Ke, Z.; Xin, T.; Zhaohui, Y.; Meng, L.; Calm, C.; Qian, J. Application of the Cannabidiol in Tooth Whitening. China Patent CN109939012A, 28 June 2019. [Google Scholar]
- Yunpeng, L.; Shuangqing, Z.; Zhipeng, L. A Kind of Toothpaste and Preparation Method Thereof with Sterilization, Anti-inflammatory and Analgesic Efficacy. China Patent CN109925246A, 25 June 2019. [Google Scholar]
- Tumey, D.M.; Panahi, H. Cannabinoid-Infused Post-Operative Dental Dressing. U.S. Patent US20200163746A1, 6 November 2019. [Google Scholar]
- Zeitouni, D.B.; Eyal, A.M. Dental Care Cannabis Device and Use Thereof. U.S. Patent WO2019239357A1, 12 June 2019. [Google Scholar]
- Haiyan, X.; Wei, H.; Meilian, X.; Zhijiang, H.; Xiao, K.; Xiao, H.; Lingyu, X. Cannabinoid-Containing Functional Toothpaste. ChinaPatent. CN111671698A, 5 August 2019. [Google Scholar]
- Qi, C.; Fengsha, X.; Lianmengm, G.; Tanran, C.; Ruyan, L.; Qingzhong, L. Oral Care Composition and Application Thereof. China Patent CN111973480A, 30 December 2019. [Google Scholar]
- Crisler, M.; Diponio, E.; Kennedy, P.J. Compositions for Preventing and Treating Viral Infections. U.S. Patent WO2020257588A1, 19 June 2020. [Google Scholar]
- Koturbash, I.; MacKay, D. Cannabidiol and other Cannabinoids: From Toxicology and Pharmacology to the Development of a Regulatory Pathway; Taylor & Francis: Abingdon, UK, 2020. [Google Scholar]
- Dick, T.N.; Marques, L.C.; Lopes, A.L.; Candreva, M.S.; Santos, L.R.; Picciani, B.L. Phytotherapy in dentistry: A literature review based on clinical data. Eur. J. Med. Plants. 2020, 1–13. [Google Scholar] [CrossRef]
- Park, H.; Kim, K.; Choi, S.; Yoon, J. A patent intelligence system for strategic technology planning. Expert Syst. Appl. 2013, 40, 2373–2390. [Google Scholar] [CrossRef]
- Nelson, K.M.; Bisson, J.; Singh, G.; Graham, J.G.; Chen, S.N.; Friesen, J.B.; Dahlin, J.L.; Niemitz, M.; Walters, M.A.; Pauli, G.F. The essential medicinal chemistry of cannabidiol (CBD). J. Med. Chem. 2020, 63, 12137–12155. [Google Scholar] [CrossRef]
- Bernardini, S.; Tiezzi, A.; Laghezza Masci, V.; Ovidi, E. Natural products for human health: An historical overview of the drug discovery approaches. Nat. Prod. Res. 2018, 32, 1926–1950. [Google Scholar] [CrossRef]
- Yau, W.P.; Goh, C.H.; Koh, H.L. Quality control and quality assurance of phytomedicines: Key considerations, methods, and analytical challenges. In Phytotherapies: Efficacy, Safety, and Regulation; Wiley: Hoboken, NJ, USA, 2015. [Google Scholar]
- Das, K.; Tiwari, R.; Shrivastava, D. Techniques for evaluation of medicinal plant products as antimicrobial agent: Current methods and future trends. J. Med. Plants Res. 2010, 4, 104–111. [Google Scholar]
| PubMed/MEDLINE | (THC) OR (Tetrahydrocannab) OR (Dronabin) OR (Cannab) OR (Phytocannab) OR (Marijuana) OR (Synthetic cannab) OR (Cannabidiol) AND (Bone and Bones [Mesh] OR (Bone and Bones) OR (Bones and Bone Tissue) OR (Bones and Bone) OR (Bone Tissue) OR (Bone Tissues) OR (Tissue, Bone) OR (Tissues, Bone) OR (Bony Apophyses) OR (Apophyses, Bony) OR (Bony Apophysis) AND (Dentistry) OR (Implants, Dental) OR (Dental Implant) OR (Implant, Dental) OR (dental Pain) OR (anti-inflammatory) OR (Pulp, Dental) OR (Pulps, Dental) OR (Dental Pulps) OR (Regeneration, Periodontal Guided Tissue) OR (Guided Periodontal Tissue Regeneration) OR (Periodontal Guided Tissue Regeneration) OR (Bone reparation) |
| Scopus | ALL (“THC” OR “Tetrahydrocannab” OR “Dronabin” OR “Cannab” OR “Phytocannab” OR “Synthetic cannab” OR “Cannabidiol” AND (“Bone and Bones” OR “Bone and Bones” OR “Bones and Bone Tissue” OR “Bones and Bone” OR “Bone Tissue” OR “Bone Tissues” OR “Tissue, Bone” OR “Tissues, Bone” OR “Bony Apophyses” OR “Apophyses, Bony” OR “Bony Apophysis”) AND (“Dentistry” OR “Implants, Dental” OR “Dental Implant” OR “Implant, Dental” OR “dental Pain” OR “anti-inflammatory” OR “Pulp, Dental” OR “Pulps, Dental” OR “Dental Pulps” OR “Regeneration, Periodontal Guided Tissue” OR “Guided Periodontal Tissue Regeneration” OR “Periodontal Guided Tissue Regeneration” OR “Bone reparation”) |
| Web of Science | TS = (THC) OR (Tetrahydrocannab *) OR (Dronabin *) OR (Cannab *) OR (Phytocannab *) OR (Marijuana *) OR (Synthetic cannab *) AND (Dentistry) OR (Implants, Dental) OR (Dental Implant) OR (Implant, Dental) OR (dental Pain) OR (anti inflamatory) OR (Pulp, Dental) OR (Pulps, Dental) OR (Dental Pulps) OR (Regeneration, Periodontal Guided Tissue) OR (Guided Periodontal Tissue Regeneration) OR (Periodontal Guided Tissue Regeneration) OR (Bone reparation) |
| Cochrane Library | (THC) OR (Tetrahydrocannab) OR (Dronabin) OR (Cannab) OR (Phytocannab) OR (Marijuana) OR (Synthetic cannab) OR (Cannabidiol) AND (Bone and Bones) [Mesh] OR (Bone and Bones) OR (Bones and Bone Tissue) OR (Bones and Bone) OR (Bone Tissue) OR (Bone Tissues) OR (Tissue, Bone) OR (Tissues, Bone) OR (Bony Apophyses) OR (Apophyses, Bony) OR (Bony Apophysis) AND (Dentistry) OR (Implants, Dental) OR (Dental Implant) OR (Implant, Dental) OR (dental Pain) OR (anti-inflammatory) OR (Pulp, Dental) OR (Pulps, Dental) OR (Dental Pulps) OR (Regeneration, Periodontal Guided Tissue) OR (Guided Periodontal Tissue Regeneration) OR (Periodontal Guided Tissue Regeneration) OR (Bone reparation) |
| Embase | (THC) OR (Tetrahydrocannab) OR (Dronabin) OR (Cannab) OR (Phytocannab) OR (Marijuana) OR (Synthetic cannab) OR (Cannabidiol) AND (Bone and Bones) [Mesh] OR (Bone and Bones) OR (Bones and Bone Tissue) OR (Bones and Bone) OR (Bone Tissue) OR (Bone Tissues) OR (Tissue, Bone) OR (Tissues, Bone) OR (Bony Apophyses) OR (Apophyses, Bony) OR (Bony Apophysis) AND (Dentistry) OR (Implants, Dental) OR (Dental Implant) OR (Implant, Dental) OR (dental Pain) OR (anti inflamatory) OR (Pulp, Dental) OR (Pulps, Dental) OR (Dental Pulps) OR (Regeneration, Periodontal Guided Tissue) OR (Guided Periodontal Tissue Regeneration) OR (Periodontal Guided Tissue Regeneration) OR (Bone reparation) |
| Orbit | THC OR Tetrahydrocannab OR Dronabin OR Cannab OR Endocannab OR Phytocannab OR Marijuana OR Synthetic cannab OR Cannabidiol OR Endocannabinoide AND “Bone and Bones OR Bones and Bone Tissue OR Bones and Bone OR Bone Tissue OR Bone Tissues OR Tissue, Bone OR Tissues, Bone OR “Bony Apophyses OR Apophyses, Bony OR Bony Apophysis OR Dentistry OR Implants, Dental OR Dental Implant OR Implant, Dental OR dental Pain OR anti-inflammatory OR Pulp, Dental OR Pulps, Dental OR Dental Pulps OR Regeneration, Periodontal Guided Tissue OR Guided Periodontal Tissue Regeneration OR Periodontal Guided Tissue Regeneration or Bone reparation |
| Google patents | THC OR Tetrahydrocannab OR Dronabin OR Cannab OR Endocannab OR Phytocannab OR Marijuana OR Synthetic cannab OR Cannabidiol OR Endocannabinoide AND “Bone and Bones OR Bones and Bone Tissue OR Bones and Bone OR Bone Tissue OR Bone Tissues OR Tissue, Bone OR Tissues, Bone OR “Bony Apophyses OR Apophyses, Bony OR Bony Apophysis OR Dentistry OR Implants, Dental OR Dental Implant OR Implant, Dental OR dental Pain OR anti-inflammatory OR Pulp, Dental OR Pulps, Dental OR Dental Pulps OR Regeneration, Periodontal Guided Tissue OR Guided Periodontal Tissue Regeneration OR Periodontal Guided Tissue Regeneration or Bone reparation |
| USPTO | THC OR Tetrahydrocannab OR Dronabin OR Cannab OR Endocannab OR Phytocannab OR Marijuana OR Synthetic cannab OR Cannabidiol OR Endocannabinoide AND “Bone and Bones OR Bones and Bone Tissue OR Bones and Bone OR Bone Tissue OR Bone Tissues OR Tissue, Bone OR Tissues, Bone OR “Bony Apophyses OR Apophyses, Bony OR Bony Apophysis OR Dentistry OR Implants, Dental OR Dental Implant OR Implant, Dental OR dental Pain OR anti-inflammatory OR Pulp, Dental OR Pulps, Dental OR Dental Pulps OR Regeneration, Periodontal Guided Tissue OR Guided Periodontal Tissue Regeneration OR Periodontal Guided Tissue Regeneration or Bone reparation |
| WIPO | THC OR Tetrahydrocannab OR Dronabin OR Cannab OR Endocannab OR Phytocannab OR Marijuana OR Synthetic cannab OR Cannabidiol OR Endocannabinoide AND “Bone and Bones OR Bones and Bone Tissue OR Bones and Bone OR Bone Tissue OR Bone Tissues OR Tissue, Bone OR Tissues, Bone OR “Bony Apophyses OR Apophyses, Bony OR Bony Apophysis OR Dentistry OR Implants, Dental OR Dental Implant OR Implant, Dental OR dental Pain OR anti-inflammatory OR Pulp, Dental OR Pulps, Dental OR Dental Pulps OR Regeneration, Periodontal Guided Tissue OR Guided Periodontal Tissue Regeneration OR Periodontal Guided Tissue Regeneration or Bone reparation |
| Espacenet | THC OR Tetrahydrocannab OR Dronabin OR Cannab OR Endocannab OR Phytocannab OR Marijuana OR Synthetic cannab OR Cannabidiol OR Endocannabinoide AND “Bone and Bones OR Bones and Bone Tissue OR Bones and Bone OR Bone Tissue OR Bone Tissues OR Tissue, Bone OR Tissues, Bone OR “Bony Apophyses OR Apophyses, Bony OR Bony Apophysis OR Dentistry OR Implants, Dental OR Dental Implant OR Implant, Dental OR dental Pain OR anti-inflammatory OR Pulp, Dental OR Pulps, Dental OR Dental Pulps OR Regeneration, Periodontal Guided Tissue OR Guided Periodontal Tissue Regeneration OR Periodontal Guided Tissue Regeneration or Bone reparation |
| First Author, Year | Journal | Country | Study Type | CBD or Analog | Dosage | Administration | Presentation | Manufacturer | Main Application | Sponsor |
|---|---|---|---|---|---|---|---|---|---|---|
| Napimoga, 2009 [18] | Int. Immunopharmacol. | Brazil | Animal | CBD | 1 mg/kg | Injected i.p | CBD powder dissolved in 2% polysorbate 20/Tween 80- in saline | THC Pharm, Germany | Periodontal therapy | CAPES and UNIUBE, Brazil |
| Klein, 2018 [19] | Phytother. Res. | Brazil | Animal | CBD | 5–10 mg/kg | Injected i.p | CBD powder dissolved in 2% polysorbate 20/Tween 80- in saline | THC Pharm, Germany | Oral therapy | CAPES and PUCRS, Brazil |
| Kogan, 2015 [8] | J. Bone Miner. Res. | Israel | Animal | CBD | 5 mg/kg | Injected i.p | CBD powder dissolved in ethanol/emulphor/saline | MAX-IV laboratory, Sweden | Aid for bone regeneration | NIH, USA and Israel Anti-Drug Authority |
| Kamali, 2019 [7] | Mater. Sci. Eng. C Mater. Biol. Appl. | Iran | Animal | CBD | 30 mg/kg | Direct contact | CBD powder dissolved in 2% polysorbate 20/Tween 80- in saline | Tocris company, USA | Aid for bone regeneration | Shiraz University, Royan Institute, and the Iran National Science Foundation, Iran |
| Cuba, 2017 [20] | J. Clin. Pharm. Ther. | Brazil | Animal | CBD | 3, 10, 30 mg/kg | Injected i.p | CBD powder dissolved in Tween 80- in saline | THC Pharm, Germany | Oral therapy | CAPES and PUCRS, Brazil |
| Scionti, 2016 [21] | Front. Physiol. | Italy | Animal | CBD | 10 mg/kg | NS | Pure CBD (>99%) | Greenhouse cultivation at CREA-CIN, Italy | Periodontal therapy | Health Ministry, Italy |
| Gu, 2019 [22] | Front. Immunol. | USA | Animal | CBD | 10 mg/kg | NS | NS | Cayman Chemical Co., USA | Periodontal therapy | NIDCR, USA |
| Cells | 0.1–1.0 µg/mL | Direct contact | NS | |||||||
| Ossola, 2016 [23] | J. Periodontol. | Argentina | Animal | HU-308 | 200 µL per tooth | Topical | HU-308 powder dissolved 100% ethanol/saline | Tocris, USA | Periodontal therapy | University of Buenos Aires and CONICET, Argentina |
| Rawal, 2012 [24] | J. Periodontal. Res. | USA | Cells | CBD | 0.01–30 µM | Direct contact | CBD powder dissolved in methanol | Sigma-Aldrich, USA | Periodontal therapy | University of Tennesse, USA |
| Stahl, 2020 [25] | Cureus | Belgium | Cells/bacteria | CBD | NS | Direct contact | NS | NS | Oral therapy | NS |
| Petrescu, 2020 [26] | Medicina | Romania | Cells | CBD | 0.75 µM | Direct contact | NS | NS | Aid for bone regeneration | CNCS, Romania |
| Patent Number(s) | Country | Year | Title | Main Claims | Inventors | Company |
|---|---|---|---|---|---|---|
| US20190076349 US10172786 EP3233021 EP3233021 WO2016/100516 US20160166498 | USA | 2014 | Oral care composition comprising cannabinoids | Toothpaste, tooth powder, or mouthwash solution | George Anastassov, Lekhram Changoer | Axim Biotechnologies |
| CN109939012 | China | 2017 | Use of the tooth whitening CBD | Toothpaste, dentifrice, mouthwash, gel, gum or film-forming agent for tooth whitening | Zhang Ke, Tan Xin, Yu Zhaohui, Lian Meng, Chang Tanran, Jin Qian | Han Yi Biotechnology |
| CN109939011 | China | 2017 | Composition comprising a cannabinoid toothpaste and its preparation method | Toothpaste with oral injury repair potential | Zhang Ke, Tan Xin, Yu Zhaohui, Lian Meng, Chang Tanran, Jin Qian | Han Yi Biotechnology |
| WO2019/055420 US20190076343 | USA | 2017 | Oral care formulations and methods for use | Toothpaste, mouthwash, chewing gum, lozenge, coated interdental device, and coated dental floss as oral care products | Gerald Curatola | NS |
| WO2019030762A2 EP3664795A2 US20200222361A1 | USA | 2018 | Cannabis and derivatives thereof for the treatment of pain and inflammation related with dental pulp and bone regeneration related to dental jaw bone defects | Composition for treating dental pulp inflammation/infection and bone defects | Veronica Stahl | CannIBite |
| CN109939011A | China | 2019 | A kind of toothpaste and preparation method thereof containing cannabinoid | Toothpaste with oral injury repair potential | Zhang Ke, Tan Xin, Yu Zhaohui, Lian Meng, Chang Tanran, Jin Qian | Han Yi Biotechnology |
| CN109939012A | China | 2019 | Application of the cannabidiol in tooth whitening | Tooth whitening product | Zhang Ke, Tan Xin, Yu Zhaohui, Lian Meng, Chang Tanran, Jin Qian | Han Yi Biotechnology |
| CN109925246A | China | 2019 | A kind of toothpaste and preparation method thereof with sterilization, anti-inflammatory, and analgesic efficacy | Toothpaste | Luan Yunpeng, Zheng Shuangqing, Li Zhipeng | NS |
| US20200163746A1 | USA | 2019 | Cannabinoid-infused post-operative dental dressing | Dental dressing pouch for an oral surgical site | David Tumey, Harry Panahi | NS |
| WO2019239357A1 | USA | 2019 | Dental care cannabis device and use thereof | Device for topical administration | Dana Zeitouni, Aharon M. Eyal | Buzzelet Development and Technologies |
| CN111671698A | China | 2019 | Cannabinoid-containing functional toothpaste | Toothpaste with antifungal, anti-inflammatory, analgesic, and neuroprotector effects | Xu Haiyan, He Wei, Xia Meilian, He Zhijiang, Kite Xiao, Xiao Hong, Xie Lingyu | Jiangxi Caoshanhu Oral Care Products |
| CN111973480A | China | 2019 | Oral care composition and application thereof | Toothpaste, dentifrice, mouthwash, oral spray, oral patch, chewing gum or buccal tablet for oral hygiene | Chen Qi, Xiao Fengsha, Gull Lianmeng, Chang Tanran, Li Ruyan, Li Qingzhong | Yunnan Hanmeng Pharmaceutical |
| WO2020257588A1 | USA | 2020 | Compositions for preventing and treating viral infections | Composition for treating viral infections | Maria Crisler, Emma Diponio, Philip Kennedy | Shaman Naturals |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
David, C.; Elizalde-Hernández, A.; Barboza, A.S.; Cardoso, G.C.; Santos, M.B.F.; Moraes, R.R. Cannabidiol in Dentistry: A Scoping Review. Dent. J. 2022, 10, 193. https://doi.org/10.3390/dj10100193
David C, Elizalde-Hernández A, Barboza AS, Cardoso GC, Santos MBF, Moraes RR. Cannabidiol in Dentistry: A Scoping Review. Dentistry Journal. 2022; 10(10):193. https://doi.org/10.3390/dj10100193
Chicago/Turabian StyleDavid, Carla, Alejandro Elizalde-Hernández, Andressa S. Barboza, Gabriela C. Cardoso, Mateus B. F. Santos, and Rafael R. Moraes. 2022. "Cannabidiol in Dentistry: A Scoping Review" Dentistry Journal 10, no. 10: 193. https://doi.org/10.3390/dj10100193
APA StyleDavid, C., Elizalde-Hernández, A., Barboza, A. S., Cardoso, G. C., Santos, M. B. F., & Moraes, R. R. (2022). Cannabidiol in Dentistry: A Scoping Review. Dentistry Journal, 10(10), 193. https://doi.org/10.3390/dj10100193







