Electrocatalytic Activities towards the Electrochemical Oxidation of Formic Acid and Oxygen Reduction Reactions over Bimetallic, Trimetallic and Core–Shell-Structured Pd-Based Materials
Abstract
1. Introduction
2. Electrocatalysts for Direct Fuel Cells
2.1. Electrocatalytic Activity for the Oxidation of Small Organic Compounds
2.2. Electrooxidation of Formic Acid on Pt-Based Electrocatalysts
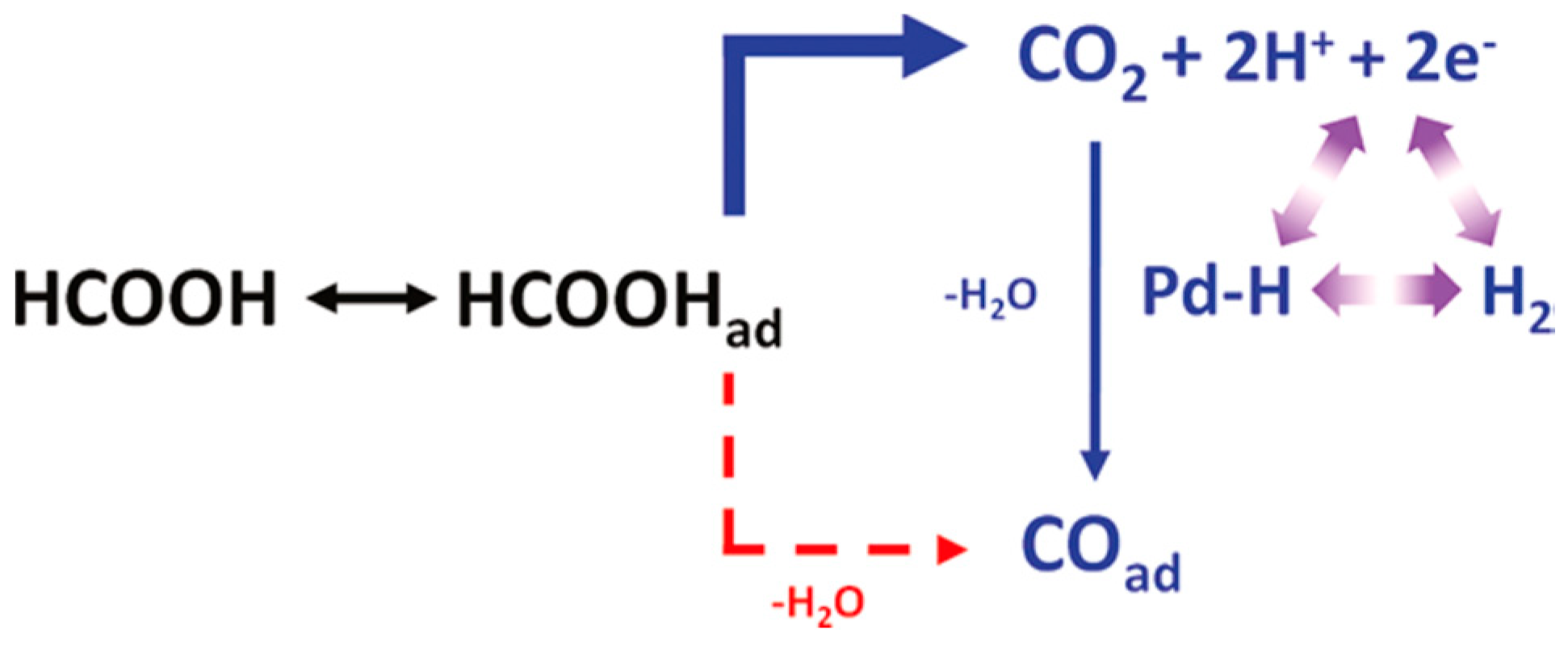
2.3. Electrooxidation of Formic Acid on Pd Electrocatalysts
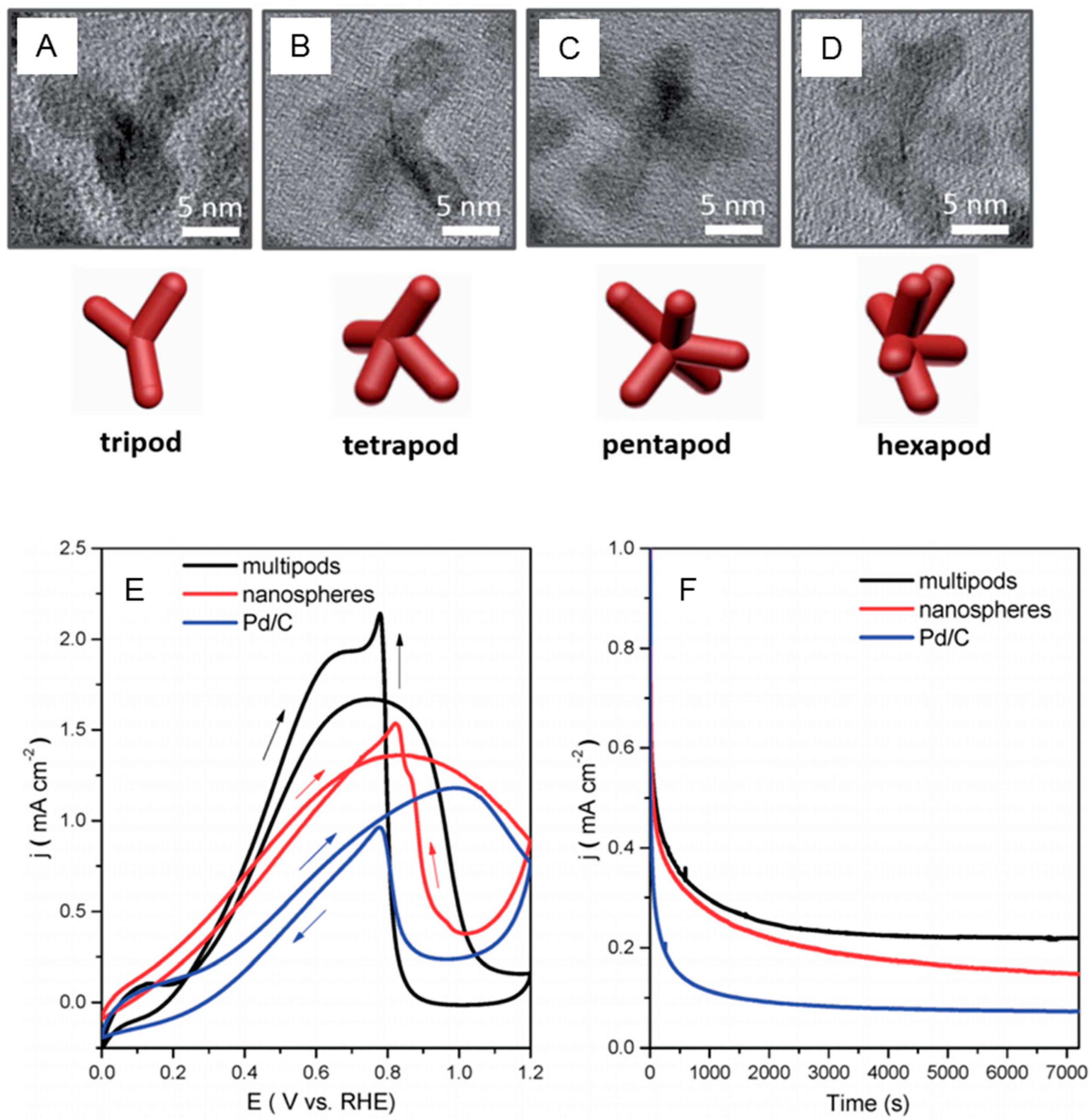
2.4. Effect of Electronic State, Morphology and Structure on the Catalytic Activity of Pd towards Formic Acid Oxidation
3. ORR
3.1. ORR on Metal Catalysts
3.2. ORR on Pd-Based Catalysts in Alkaline Media
3.3. Electrocatalytic Activity of Pd-Based Materials towards ORR in Acidic Media
3.4. The Development of Electrocatalysts for ORR Based on Theoretical Calculations
3.5. The Development of Electrocatalysts with Simultaneously Improved Activity and Durability
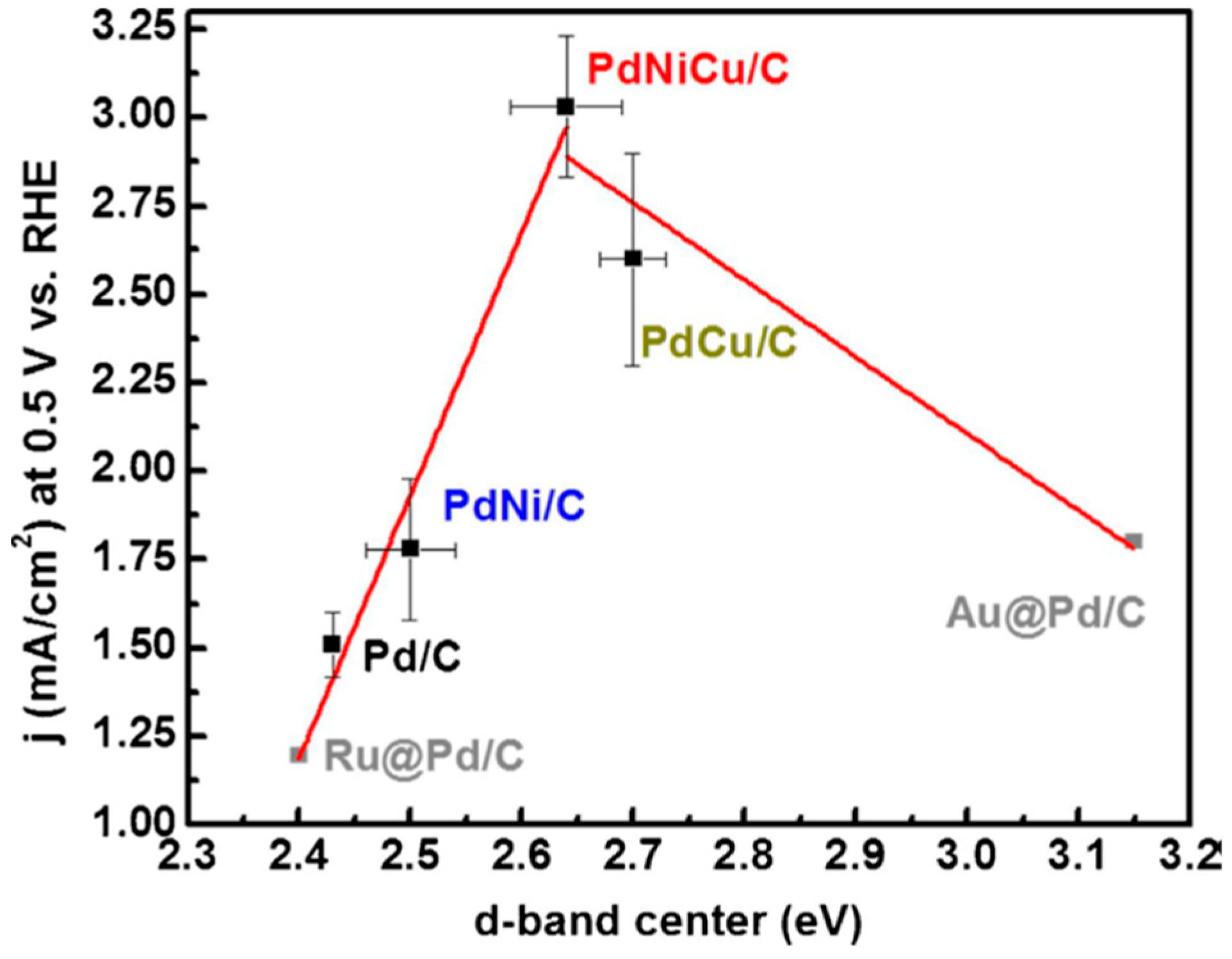
3.6. Pt-Decorated Pd and Pd Bimetallic Alloy Catalysts for ORR Electrocatalysts in Acidic Media
4. Conclusions
Funding
Conflicts of Interest
References
- Ahmadi, T.S.; Wang, Z.L.; Green, T.C.; Henglein El-Sayed, A.M. Shape-Controlled Synthesis of Colloidal Platinum Nanoparticles. Science 1996, 272, 1924–1925. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Pickup, P.G. Recent advances in direct formic acid fuel cells (DFAFC). J. Power Sources 2008, 182, 124–132. [Google Scholar] [CrossRef]
- Demirci, U.B. Direct liquid-feed fuel cells: Thermodynamic and environmental concerns. J. Power Sources 2007, 169, 239–246. [Google Scholar] [CrossRef]
- Rice, C.; Ha, S.; Masel, R.I.; Waszczuk, P.; Wieckowski, A.; Barnard, T. Direct formic acid fuel cells. J. Power Sources 2002, 111, 83–89. [Google Scholar] [CrossRef]
- Raj, C.R.; Samanta, A.; Noh, S.H.; Mondal, S.; Okajima, T.; Ohsaka, T. Emerging new generation electrocatalysts for the oxygen reduction reaction. J. Mater. Chem. A 2016, 4, 11156–11178. [Google Scholar] [CrossRef]
- Chen, A.; Ostrom, C. Palladium-Based Nanomaterials: Synthesis and Electrochemical Applications. Chem. Rev. 2015, 115, 11999–12044. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Lee, K.; Zhang, J. The effect of heat treatment on nanoparticle size and ORR activity for carbon-supported Pd–Co alloy electrocatalysts. Electrochim. Acta 2007, 52, 3088–3094. [Google Scholar] [CrossRef]
- Shi, Q.; Zhu, C.; Bi, C.; Xi, H.; Engelhard, H.M.; Du, D.; Lin, Y. Intermetallic Pd3Pb nanowire networks boost ethanol oxidation and oxygen reduction reactions with significantly improved methanol tolerance. J. Mater. Chem. A 2017, 5, 23952–23959. [Google Scholar] [CrossRef]
- Wang, D.; Xin, H.L.; Wang, H.; Yu, Y.; Rus, E.; Muller, D.A.; DiSalvo, F.J.; Abruña, H.D. Facile Synthesis of Carbon-Supported Pd–Co Core–Shell Nanoparticles as Oxygen Reduction Electrocatalysts and Their Enhanced Activity and Stability with Monolayer Pt Decoration. Chem. Mater. 2012, 24, 2274–2281. [Google Scholar] [CrossRef]
- Stamenkovic, V.R.; Mun, B.S.; Arenz, M.; Mayrhofer, K.J.J.; Lucas, C.A.; Wang, G.; Ross, P.N.; Markovic, N.M. Trends in electrocatalysis on extended and nanoscale Pt-bimetallic alloy surfaces. Nat. Mater. 2007, 6, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Greeley, J.; Stephens, I.E.L.; Bondarenko, A.S.; Johansson, T.P.; Hansen, H.A.; Jaramillo, T.F.; Rossmeisl, J.; Chorkendorff, I.; Nørskov, J.K. Alloys of platinum and early transition metals as oxygen reduction electrocatalysts. Nat. Chem. 2009, 1, 552–556. [Google Scholar] [CrossRef] [PubMed]
- Shao, M.; Chang, Q.; Dodelet, J.-P.; Chenitz, R. Recent Advances in Electrocatalysts for Oxygen Reduction Reaction. Chem. Rev. 2016, 116, 3594–3657. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zou, S.; Cai, W.-B. Recent Advances on Electro-Oxidation of Ethanol on Pt- and Pd-Based Catalysts: From Reaction Mechanisms to Catalytic Materials. Catalysts 2015, 5, 1507–1534. [Google Scholar] [CrossRef]
- Antolini, E. Alloy vs. intermetallic compounds: Effect of the ordering on the electrocatalytic activity for oxygen reduction and the stability of low temperature fuel cell catalysts. Appl. Catal. B Environ. 2017, 217, 201–213. [Google Scholar] [CrossRef]
- Erikson, H.; Sarapuu, A.; Gullón, J.S.; Tammeveski, K. Recent progress in oxygen reduction electrocatalysis on Pd-based catalysts. J. Electroanal. Chem. 2016, 780, 327–336. [Google Scholar] [CrossRef]
- Bianchini, C.; Shen, P.K. Palladium-Based Electrocatalysts for Alcohol Oxidation in Half Cells and in Direct Alcohol Fuel Cells. Chem. Rev. 2009, 109, 4183–4206. [Google Scholar] [CrossRef] [PubMed]
- Yin, Z.; Lin, L.; Ma, D. Construction of Pd-based nano-catalysts for fuel cells: Opportunities and challenges. Catal. Sci. Technol. 2014, 4, 4116–4128. [Google Scholar] [CrossRef]
- Gómez, J.C.C.; Moliner, R.; Lázaro, M.J. Palladium-Based Catalysts as Electrodes for Direct Methanol Fuel Cells: A Last Ten Years Review. Catalysts 2016, 6, 130. [Google Scholar] [CrossRef]
- Meng, H.; Zeng, D.; Xie, F. Recent Development of Pd-Based Electrocatalysts for Proton Exchange Membrane Fuel Cells. Catalysts 2015, 5, 1221–1274. [Google Scholar] [CrossRef]
- Antolini, E. Structural parameters of supported fuel cell catalysts: The effect of particle size, inter-particle distance and metal loading on catalytic activity and fuel cell performance. Appl. Catal. B Environ. 2016, 181, 298–313. [Google Scholar] [CrossRef]
- Halseid, M.C.; Jusys, Z.; Behm, R.J. Ethanol Oxidation over a Pt/C Catalyst at High emperatures and Pressure: An Online Electrochemical Mass Spectrometry Study. J. Phys. Chem. C 2010, 114, 22573–22581. [Google Scholar] [CrossRef]
- Bagotzky, V.S.; Vassiliev, Y.B.; Khazova, O.A.J. Generalized scheme of chemisorption, electrooxidation and electroreduction of simple organic compounds on platinum group metals. Electroanal. Chem. 1977, 81, 229. [Google Scholar] [CrossRef]
- Iwasita, T.; Hoster, H.; Anacker, A.J.; Lin, W.F.; Vielstich, W. Methanol Oxidation on PtRu Electrodes. Influence of Surface Structure and Pt-Ru Atom Distribution. Langmuir 2000, 16, 522–529. [Google Scholar] [CrossRef]
- Kodiyath, R.; Ramesh, G.V.; Koudelkova, E.; Tanabe, T.; Ito, M.; Manikandan, M.; Ueda, S.; Fujita, T.; Umezawa, N.; Noguchi, H.; et al. Promoted C–C bond cleavage over intermetallic TaPt3 catalyst toward low-temperature energy extraction from ethanol. Energy Environ. Sci. 2015, 8, 1685–1689. [Google Scholar] [CrossRef]
- Kunimatsu, K.; Kita, H. Infrared spectroscopic study of methanol and formic acid absorbates on a platinum electrode: Part II. Role of the linear CO(a) derived from methanol and formic acid in the electrocatalytic oxidation of CH3OH and HCOOH. Electroanal. Chem. 1987, 218, 155. [Google Scholar] [CrossRef]
- Corrigan, D.S.; Weaver, M.J. Mechanisms of formic acid, methanol, and carbon monoxide electrooxidation at platinum as examined by single potential alteration infrared spectroscopy. J. Electroanal. Chem. 1988, 241, 143. [Google Scholar] [CrossRef]
- Gao, W.; Mueller, J.E.; Jiang, Q.; Jacob, T. The Role of Co-Adsorbed CO and OH in the Electrooxidation of Formic Acid on Pt(111). Angew. Chem. Int. Ed. 2012, 51, 9448. [Google Scholar] [CrossRef]
- Chen, Y.-X.; Heinen, M.; Jusys, Z.; Behm, R.J. Bridge-Bonded Formate: Active Intermediate or Spectator Species in Formic Acid Oxidation on a Pt Film Electrode? Langmuir 2006, 22, 10399–10408. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Keith, J.A.; Anton, J.; Jacob, T. Theoretical Elucidation of the Competitive Electro-oxidation Mechanisms of Formic Acid on Pt(111). J. Am. Chem. Soc. 2010, 132, 18377–18385. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Keith, J.A.; Anton, J.; Jacob, T. Oxidation of formic acid on the Pt(111) surface in the gas phase. Dalton Trans. 2010, 39, 8450–8456. [Google Scholar] [CrossRef] [PubMed]
- Gunji, T.; Noh, S.H.; Tanabe, T.; Han, B.; Nien, C.Y.; Ohsaka, T.; Matsumoto, F. Enhanced Electrocatalytic Activity of Carbon-Supported Ordered Intermetallic Palladium–Lead (Pd3Pb) Nanoparticles toward Electrooxidation of Formic Acid. Chem. Mater. 2017, 29, 2906–2913. [Google Scholar] [CrossRef]
- Zhang, S.; Shao, Y.Y.; Liao, H.G.; Liu, J.; Aksay, I.A.; Yin, G.P.; Lin, Y.H. Graphene Decorated with PtAu Alloy Nanoparticles: Facile Synthesis and Promising Application for Formic Acid Oxidation. Chem. Mater. 2011, 23, 1079–1081. [Google Scholar] [CrossRef]
- Roychowdhury, C.; Matsumoto, F.; Mutolo, P.F.; Abruña, H.D.; DiSalvo, F.J. Synthesis and characterization and electrochemical activity of PtBi nanoparticles prepared by the polyol process. Chem. Mater. 2005, 17, 5871–5876. [Google Scholar] [CrossRef]
- Roychowdhury, C.; Matsumoto, F.; Zeldovich, V.B.; Warren, S.C.; Mutolo, P.F.; Ballesteros, M.; Wiesner, U.; Abruña, H.D.; DiSalvo, F. Synthesis, Characterization and Electrocatalytic activity of PtBi and PtPb Nanoparticles prepared by Borohydride reduction in Methanol. Chem. Mater. 2006, 18, 3365–3372. [Google Scholar] [CrossRef]
- Matsumoto, F.; Roychowdhury, C.; DiSalvo, F.J.; Abruña, H.D. Electrochemical Activity of Ordered Intermetallic PtPb Nanoparticles Prepared by Borohydride Reduction towards Formic Acid Oxidation for Fuel Cell Applications. J. Electrochem. Soc. 2008, 155, B148–B154. [Google Scholar] [CrossRef]
- Wang, H.S.; Alden, L.; DiSalvo, F.J.; Abruña, H.D. Electrocatalytic mechanism and kinetics of SOMs oxidation on ordered PtPb and PtBi intermetallic compounds: DEMS and FTIRS study. Phys. Chem. Chem. Phys. 2008, 10, 3739–3751. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, F. Ethanol and Methanol Oxidation Activity of PtPb, PtBi, and PtBi2 Intermetallic Compounds in Alkaline Media. Electrochemistry 2012, 80, 132–138. [Google Scholar] [CrossRef]
- Capon, A.; Parsons, R.J. The oxidation of formic acid on noble metal electrodes: II. A comparison of the behaviour of pure electrodes. J. Electroanal. Chem. Interfacial Electrochem. 1973, 44, 239–254. [Google Scholar] [CrossRef]
- Zhang, H.-X.; Wang, C.; Wang, J.-Y.; Zhai, J.-J.; Cai, W.-B. Carbon-Supported Pd–Pt Nanoalloy with Low Pt Content and Superior Catalysis for Formic Acid Electro-oxidation. J. Phys. Chem. C 2010, 114, 6446–6451. [Google Scholar] [CrossRef]
- Elnabawy, A.O.; Herron, J.A.; Scaranto, J.; Mavrikakis, M. Structure Sensitivity of Formic Acid Electrooxidation on Transition Metal Surfaces: A First-Principles Study. J. Electrochem. Soc. 2018, 165, J3109–J3121. [Google Scholar] [CrossRef]
- Scaranto, J.; Mavrikakis, M. Density functional theory studies of HCOOH decomposition on Pd(111). Surf. Sci. 2016, 650, 111–120. [Google Scholar] [CrossRef]
- Melchionna, M.; Bracamonte, M.V.; Giuliani, A.; Nasi, L.; Montini, T.; Tavagnacco, C.; Bonchio, M.; Fornasiero, P.; Prato, M. Pd@TiO2/carbon nanohorn electrocatalysts: Reversible CO2 hydrogenation to formic acid. Energy Environ. Sci. 2018, 11, 1571–1580. [Google Scholar] [CrossRef]
- Jiang, K.; Zhang, H.X.; Zou, S.; Cai, W.B. Electrocatalysis of formic acid on palladium and platinum surfaces: From fundamental mechanisms to fuel cell applications. Phys. Chem. Chem. Phys. 2014, 16, 20360–20376. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.-X.; Wang, S.-H.; Jiang, K.; André, T.; Cai, W.-B. In Situ Spectroscopic Investigation of CO Accumulation and Poisoning on Pd Black Surfaces in Concentrated HCOOH. J. Power Sources 2012, 199, 165–169. [Google Scholar] [CrossRef]
- Wang, J.-Y.; Zhang, H.-X.; Jiang, K.; Cai, W.-B. From HCOOH to CO at Pd Electrodes: A Surface-Enhanced Infrared Spectroscopy Study. J. Am. Chem. Soc. 2011, 133, 14876–14879. [Google Scholar] [CrossRef] [PubMed]
- Xi, Z.; Erdosy, D.P.; Mendoza-Garcia, A.; Duchesne, P.N.; Li, J.; Muzzio, M.; Li, Q.; Zhang, P.; Sun, S. Pd Nanoparticles Coupled to WO2.72 Nanorods for Enhanced Electrochemical Oxidation of Formic Acid. Nano Lett. 2017, 17, 2727–2731. [Google Scholar] [CrossRef] [PubMed]
- Mazumder, V.; Chi, M.; Mankin, M.; Liu, Y.; Metin, O.; Sun, D.; More, K.L.; Sun, S. A Facile Synthesis of MPd (M = Co, Cu) Nanoparticles and Their Catalysis for Formic Acid Oxidation. Nano Lett. 2012, 12, 1102–1106. [Google Scholar] [CrossRef] [PubMed]
- Du, C.Y.; Chen, M.; Wang, W.G.; Yin, G.P. Nanoporous PdNi Alloy Nanowires as Highly Active Catalysts for the Electro-Oxidation of Formic Acid. ACS Appl. Mater. Interfaces 2011, 3, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.Z.; Munoz, F.; Noborikawa, J.; Haan, J.; Scudiero, L.; Ha, S. Carbon Supported Pd-Based Bimetallic and Trimetallic Catalyst for Formic Acid Electrochemical Oxidation. Appl. Catal. B 2016, 180, 758–765. [Google Scholar] [CrossRef]
- Chen, D.; Sun, P.; Liu, H.; Yang, J. Bimetallic Cu–Pd alloy multipods and their highly electrocatalytic performance for formic acid oxidation and oxygen reduction. J. Mater. Chem. A 2017, 5, 4421–4429. [Google Scholar] [CrossRef]
- Feng, L.; Sun, X.; Liu, C.; Xing, W. Poisoning effect diminished on a novel PdHoOx/C catalyst for the electrooxidation of formic acid. Chem. Commun. 2012, 48, 419–421. [Google Scholar] [CrossRef] [PubMed]
- Abe, H.; Yoshikawa, H.; Umezawa, N.; Xu, Y.; Saravanan, G.; Ramesh, G.V.; Tanabe, T.; Kodiyath, R.; Ueda, S.; Sekido, N.; et al. Correlation between the surface electronic structure and CO-oxidation activity of Pt alloys. Phys. Chem. Chem. Phys. 2015, 17, 4879–4887. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.J.; Lee, J.Y. Highly active core–shell Au@Pd catalyst for formic acid electrooxidation. Electrochem. Commun. 2007, 9, 1725–1729. [Google Scholar] [CrossRef]
- Liu, P.; Norskov, J.K. Ligand and ensemble effects in adsorption on alloy surfaces. Phys. Chem. Chem. Phys. 2001, 3, 3814–3818. [Google Scholar] [CrossRef]
- Yin, X.; Chen, Q.; Tian, P.; Zhang, P.; Zhang, Z.; Voyles, P.M.; Wang, X. Ionic Layer Epitaxy of Nanometer-Thick Palladium Nanosheets with Enhanced Electrocatalytic Properties. Chem. Mater. 2018, 30, 3308. [Google Scholar] [CrossRef]
- He, N.; Gong, Y.; Yang, Y. ScienceDirect an effective Pd@Ni-B/C anode catalyst for electro- oxidation of formic acid. Int. J. Hydrogen Energy 2018, 43, 3216–3222. [Google Scholar] [CrossRef]
- Huang, L.; Yang, J.; Wu, M.; Shi, Z.Q.; Lin, Z.; Kang, X.W.; Chen, S.W. PdAg@Pd core–shell nanotubes: Superior catalytic performance towards electrochemical oxidation of formic acid and methanol. J. Power Sources 2018, 398, 201–208. [Google Scholar] [CrossRef]
- Zhang, W.; Huang, H.; Li, F.; Deng, K.; Wang, X. Palladium nanoparticles supported on graphitic carbon nitride-modified reduced graphene oxide as highly efficient catalysts for formic acid and methanol electrooxidation. J. Mater. Chem. A 2014, 2, 19084–19094. [Google Scholar] [CrossRef]
- Cui, Z.; Yang, M.; DiSalvo, F.J. Mesoporous Ti0.5Cr0.5N Supported PdAg Nanoalloy as Highly Active and Stable Catalysts for the Electro-Oxidation of Formic Acid and Methanol. ACS Nano 2014, 8, 6106–6113. [Google Scholar] [CrossRef] [PubMed]
- Hua, S.; Scudiero, L.; Ha, S. Effect of Au and Ru in Au@Pd and Ru@Pd Core@Shell Nanoparticles for Direct Formic Acid Fuel Cells. ECS Trans. 2014, 64, 1121–1127. [Google Scholar] [CrossRef]
- Stamenkovic, V.R.; Fowler, B.; Mun, B.S.; Wang, G.; Ross, P.N.; Lucas, C.A.; Markovic, N.M. Improved Oxygen Reduction Activity on Pt3Ni(111) via Increased Surface Site Availability. Science 2007, 315, 493–497. [Google Scholar] [CrossRef] [PubMed]
- Hodnik, N.; Jeyabharathi, C.; Meier, J.C.; Kostka, A.; Phani, K.L.; Recnik, A.; Bele, M.; Hocevar, S.; Gaberscek, M.; Mayrhofer, K.J.J. Effect of ordering of PtCu3 nanoparticle structure on the activity and stability for the oxygen reduction reaction. Phys. Chem. Chem. Phys. 2014, 16, 13610–13615. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Yu, Y.; Xin, H.L.; Hovden, R.; Ercius, P.; Mundy, J.A.; Chen, H.; Richard, J.H.; Muller, D.A.; DiSalvo, F.J. Tuning Oxygen Reduction Reaction Activity Via Controllable Dealloying: A Model Study of Ordered Cu3Pt/C Intermetallic Nanocatalysts. Nano Lett. 2012, 12, 5230–5238. [Google Scholar] [CrossRef] [PubMed]
- Abe, H.; Matsumoto, F.; Alden, L.R.; Warren, S.C.; Abruña, H.D.; DiSalvo, F.J. High Electrocatalytic Activity of Atomically Ordered Pt3Ti Nanoparticles. J. Am. Chem. Soc. 2008, 130, 5452–5458. [Google Scholar] [CrossRef] [PubMed]
- De-los-Santos-Alvarez, N.; Alden, L.R.; Rus, E.; Wang, H.; DiSalvo, F.J.; Abruña, H.D. CO tolerance of ordered intermetallic phases. J. Electroanal. Chem. 2009, 626, 14–22. [Google Scholar] [CrossRef]
- Shao, Y.Y.; Zhang, S.; Wang, C.M.; Nie, Z.M.; Liu, J.; Wang, Y.; Lin, Y.H. Highly durable graphene nanoplatelets supported Pt nanocatalysts for oxygen reduction. J. Power Sources 2010, 195, 4600–4605. [Google Scholar] [CrossRef]
- Fu, K.; Wang, Y.; Mao, L.; Jin, J.; Yang, S.; Li, G. Facile one-pot synthesis of graphene-porous carbon nanofibers hybrid support for Pt nanoparticles with high activity towards oxygen reduction. Electrochim. Acta 2016, 215, 427–434. [Google Scholar] [CrossRef]
- Nassr, A.B.A.A.; Quetschke, A.; Koslowski, E.; Bron, M. Electrocatalytic Oxidation of Formic Acid on Pd/MWCNTs Nanocatalysts Prepared by the Polyol Method. Electrochim. Acta 2013, 102, 202–211. [Google Scholar] [CrossRef]
- Mondal, S.L.; Ghosh, S.; Raj, C.R. Unzipping of Single-Walled Carbon Nanotube for the Development of Electrocatalytically Active Hybrid Catalyst of Graphitic Carbon and Pd Nanoparticles. ACS Omega 2018, 3, 622–630. [Google Scholar] [CrossRef]
- Hammer, B.; Nørskov, J.K. Theoretical surface science and catalysis—Calculations and concepts. Adv. Catal. 2000, 45, 71–129. [Google Scholar]
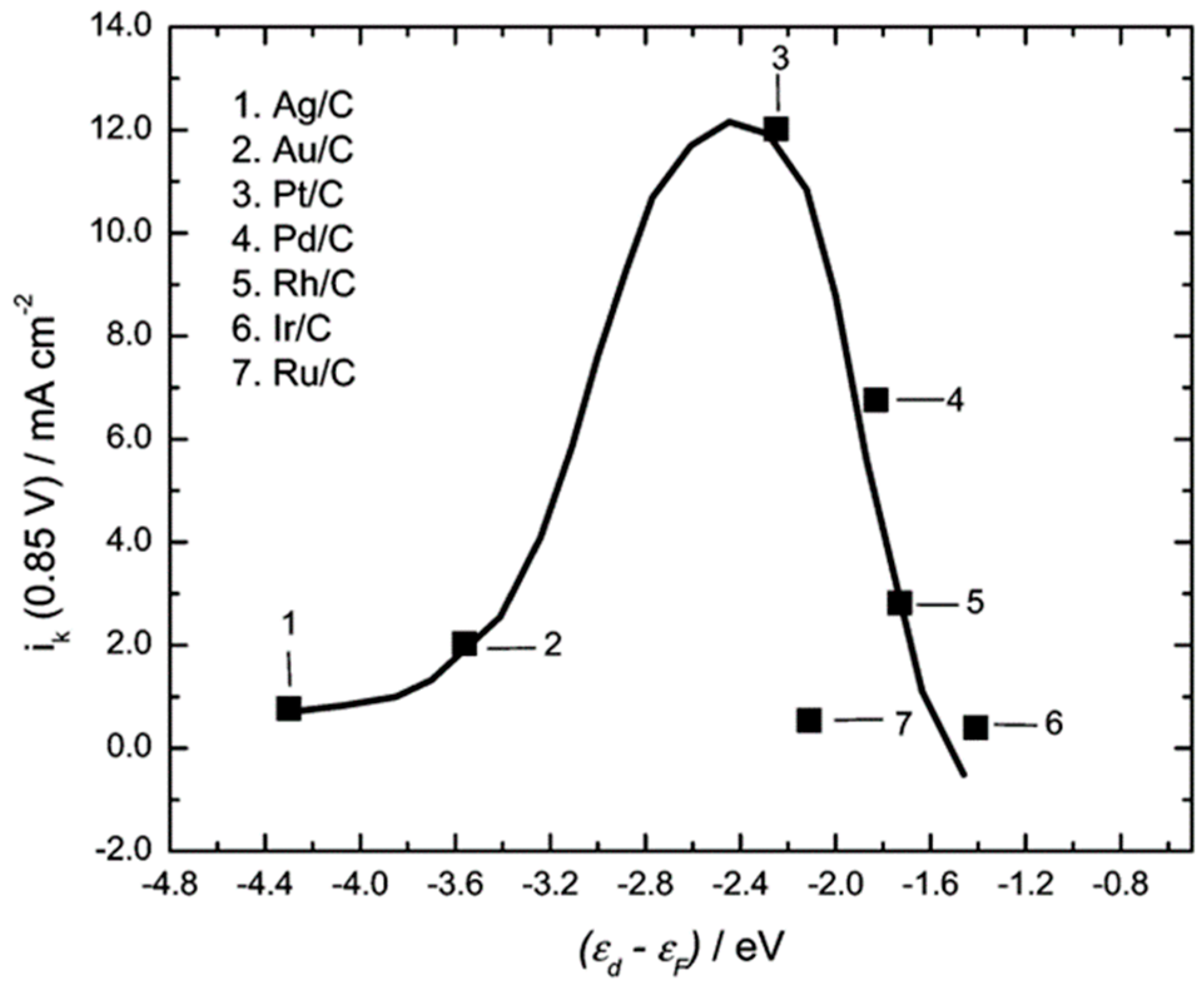
- Stamenkovic, V.; Mun, B.S.; Mayrhofer, K.J.J.; Ross, P.N.; Markovic, N.M.; Rossmeisl, J.; Greeley, J.; Norskov, J.K. Changing the Activity of Electrocatalysts for Oxygen Reduction by Tuning the Surface Electronic Structure. Angew. Chem. Int. Ed. 2006, 45, 2897–2906. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.X.; Zhang, J.L.; Adzic, R.R. Catalytic Activity–d-Band Center Correlation for the O2 Reduction Reaction on Platinum in Alkaline Solutions. J. Phys. Chem. A 2007, 111, 12702–12710. [Google Scholar] [CrossRef] [PubMed]
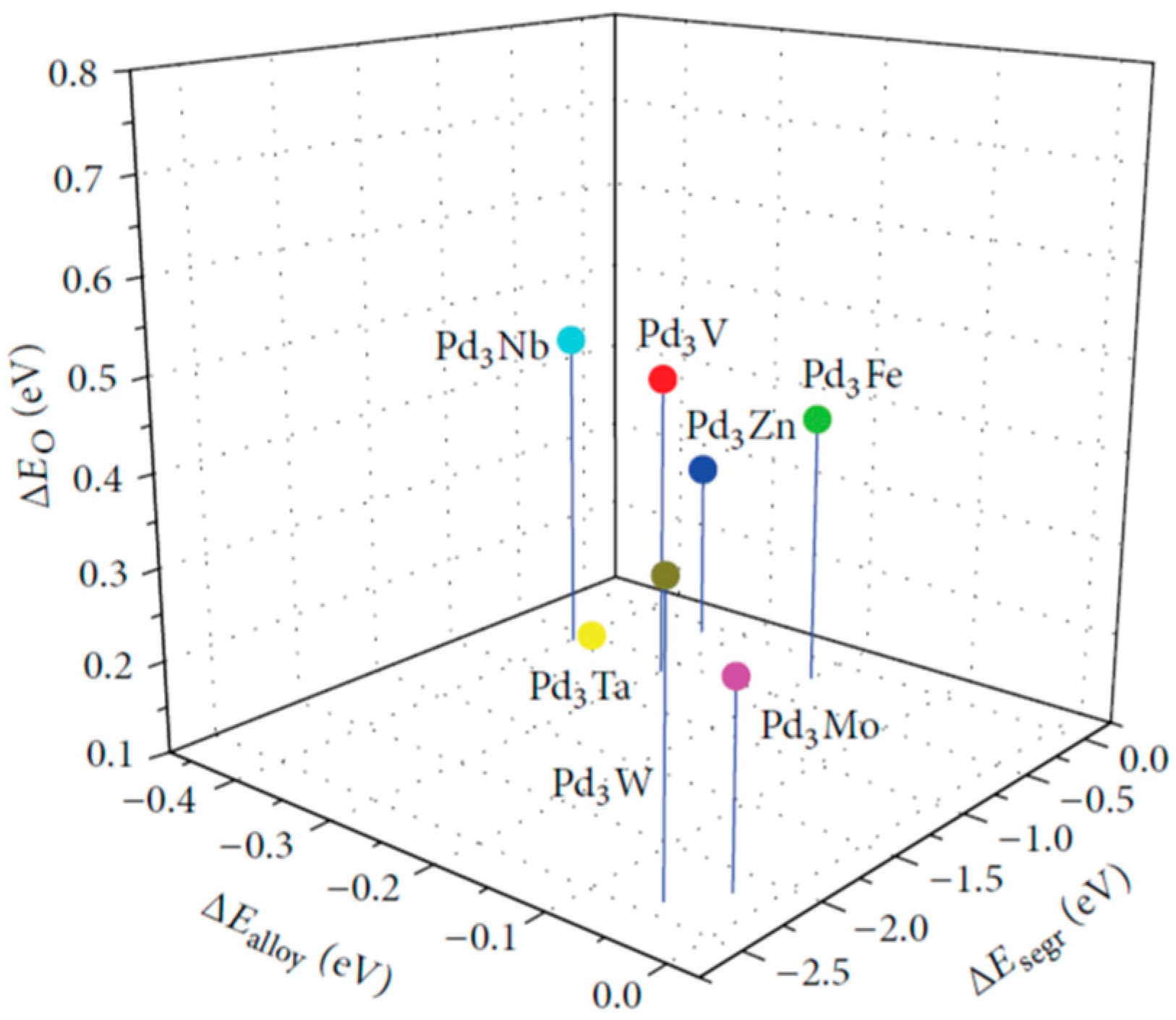
- Gunji, T.; Wakabayashi, R.H.; Noh, S.H.; Han, B.; Matsumoto, F.; DiSalvo, F.J.; Abruña, H.D. The effect of alloying of transition metals (M = Fe, Co, Ni) with palladium catalysts on the electrocatalytic activity for the oxygen reduction reaction in alkaline media. Electrochim. Acta 2018, 283, 1045–1052. [Google Scholar] [CrossRef]
- Cui, Z.M.; Chen, H.; Zhao, M.; DiSalvo, F.J. High Performance Pd3Pb Intermetallic Catalyst for Electrochemical Oxygen Reduction. Nano Lett. 2016, 16, 2560–2566. [Google Scholar] [CrossRef] [PubMed]
- Bu, L.Z.; Shao, Q.; Pi, Y.C.; Yao, J.L.; Luo, M.C.; Lang, J.P.; Hwang, S.; Xin, H.L.; Huang, B.L.; Guo, J.; et al. Coupled s-p-d Exchange in Facet-Controlled Pd3Pb Tripods Enhances Oxygen Reduction. Catal. Chem. 2018, 4, 359–371. [Google Scholar] [CrossRef]
- Luo, S.; Tang, M.; Wu, X.; Ou, Y.; Wang, Z.; Jian, N.; Li, X.; Lin, Y.; Yan, Y.; Huang, J.; et al. Intermetallic Pd3Pb square nanoplates as highly efficient electrocatalysts for oxygen reduction reaction. CrystEngComm 2019. Advance Article. [Google Scholar] [CrossRef]
- Wang, K.; Qin, Y.; Lv, F.; Li, M.; Liu, Q.; Lin, F.; Feng, J. Intermetallic Pd3Pb nanoplates enhance oxygen reduction catalysis with excellent methanol tolerance. Small 2018, 1700331, 1–8. [Google Scholar]
- Liu, S.; Wang, Y.; Liu, L.; Li, M.; Lv, W.; Zhao, X.; Qin, Z.; Zhu, P.; Wang, G.; Long, Z.; et al. One-Pot Synthesis of Pd@PtNi Core–shell Nanoflowers Supported on the Multi-Walled Carbon Nanotubes with Boosting Activity toward Oxygen Reduction in Alkaline Electrolyte. J. Power Sources 2017, 365, 26–33. [Google Scholar] [CrossRef]
- Lu, Y.; Jiang, Y.; Gao, X.; Wang, X.; Chen, W. Strongly Coupled Pd anotetrahedron/Tungsten Oxide Nanosheet Hybrids with Enhanced Catalytic Activity and Stability as Oxygen Reduction Electrocatalysts. J. Am. Chem. Soc. 2014, 136, 11687–11697. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Qin, X.; Jiang, K.; Dong, Y.; Shao, M.; Cai, W.-B. Electrocatalytic Activities of Oxygen Reduction Reaction on Pd/C and Pd–B/C Catalysts. J. Phys. Chem. C 2017, 121, 3416–3423. [Google Scholar] [CrossRef]
- Xue, Q.; Bai, J.; Han, C.; Chen, P.; Jiang, J.-X.; Chen, Y. Au Nanowires@Pd-Polyethylenimine Nanohybrids as Highly Active and Methanol-Tolerant Electrocatalysts toward Oxygen Reduction Reaction in Alkaline Media. ACS Catal. 2018, 8, 11287–11295. [Google Scholar] [CrossRef]
- Cui, Z.; Li, L.; Manthiram, A.; Goodenough, J.B. Enhanced Cycling Stability of Hybrid Li–Air Batteries Enabled by Ordered Pd3Fe Intermetallic Electrocatalyst. J. Am. Chem. Soc. 2015, 137, 7278–7281. [Google Scholar] [CrossRef] [PubMed]
- Păsti, I.; Mentus, S. Electronic properties of the PtxMe1−x/Pt(1 1 1) (Me = Au, Bi, In, Pb, Pd, Sn and Cu) surface alloys: DFT study. Mater. Chem. Phys. 2009, 116, 94–101. [Google Scholar] [CrossRef]
- Verdaguer-Casadevall, A.; Hernandez-Fernandez, P.; Stephens, I.E.L.; Chorkendorff, I.; Dahl, S. The effect of ammonia upon the electrocatalysis of hydrogen oxidation and oxygen reduction on polycrystalline platinum. J. Power Sources 2012, 220, 205–210. [Google Scholar] [CrossRef]
- Siahrostami, S.; Verdaguer-Casadevall, A.; Karamad, M.; Deiana, D.; Malacrida, P.; Wickman, B.; Escudero-Escribano, M.; Paoli, E.A.; Frydendal, R.; Hansen, T.W.; et al. Enabling direct H2O2 production through rational electrocatalyst design. Nat. Mater. 2013, 12, 1137–1143. [Google Scholar] [CrossRef] [PubMed]
- Verdaguer-Casadevall, A.; Deiana, D.; Karamad, M.; Siahrostami, S.; Malacrida, P.; Hansen, T.W.; Rossmeisl, J.; Chorkendorff, I.; Stephens, I.E.L. Trends in the Electrochemical Synthesis of H2O2: Enhancing Activity and Selectivity by Electrocatalytic Site Engineering. Nano Lett. 2014, 14, 1603–1608. [Google Scholar] [CrossRef] [PubMed]
- Jirkovsky, J.S.; Panas, I.; Ahlberg, E.; Halasa, M.; Romani, S.; Schiffrin, D.J. Single Atom Hot-Spots at Au–Pd Nanoalloys for Electrocatalytic H2O2 Production. J. Am. Chem. Soc. 2011, 133, 19432–19441. [Google Scholar] [CrossRef] [PubMed]
- Erikson, H.; Jürmann, G.; Sarapuu, A.; Potter, R.J.; Tammeveski, K. Electroreduction of oxygen on carbon-supported gold catalysts. Electrochim. Acta 2009, 54, 7483–7489. [Google Scholar] [CrossRef]
- Gasteiger, H.A.; Kocha, S.S.; Sompalli, B.; Wagner, F.T. Activity Benchmarks and Requirements for Pt, Pt-Alloy, and Non-Pt Oxygen Reduction Catalysts for PEMFCs. Appl. Catal. B 2005, 56, 9–35. [Google Scholar] [CrossRef]
- Kim, J.; Lee, S.W.; Carlton, C.; Shao-Horn, Y. Oxygen Reduction Activity of PtXNi1−X Alloy Nanoparticles on Multiwall Carbon Nanotubes. Electrochem. Solid-State Lett. 2011, 14, B110–B113. [Google Scholar] [CrossRef]
- Higuchi, E.; Taguchi, A.; Hayashi, K.; Inoue, H. Electrocatalytic Activity for Oxygen Reduction Reaction of Pt Nanoparticle Catalysts with Narrow Size Distribution Prepared from [Pt3(Co)3(M-Co)3]n2− (n = 3–8) Complexes. J. Electroanal. Chem. 2011, 663, 84–89. [Google Scholar] [CrossRef]
- Wang, D.; Liu, S.; Wang, J.; Lin, R.; Kawasaki, M.; Rus, E.; Silberstein, K.E.; Lowe, M.A.; Lin, F.; Nordlund, D.; et al. Spontaneous incorporation of gold in palladium-based ternary nanoparticles makes durable electrocatalysts for oxygen reduction reaction. Nat. Commun. 2016, 7, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Chen, D.P.; Sang, X.; Unocic, R.R.; Skrabalak, S.E. Size-Dependent Disorder–Order Transformation in the Synthesis of Monodisperse Intermetallic PdCu Nanocatalysts. ACS Nano 2016, 10, 6345–6353. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.P.; Kariuki, N.; Vaughey, J.T.; Goodpaster, J.; Kumar, R.; Myers, D.J. Bimetallic Pd–Cu Oxygen Reduction Electrocatalysts. J. Electrochem. Soc. 2008, 155, B602–B609. [Google Scholar] [CrossRef]
- Gunji, T.; Noh, S.H.; Ando, F.; Tanabe, T.; Han, B.; Ohsaka, T.; Matsumoto, F. Electrocatalytic activity of electrochemically dealloyed PdCu3 intermetallic compound towards oxygen reduction reaction in acidic media. J. Mater. Chem. A 2018, 6, 14828. [Google Scholar] [CrossRef]
- Xiao, W.; Zhu, J.; Han, L.; Liu, S.; Wang, J.; Wu, Z.; Lei, W.; Xuan, C.; Xin, H.L.; Wang, D. Pt skin on Pd-Co-Zn/C ternary nanoparticles with enhanced Pt efficiency toward ORR. Nanoscale 2016, 8, 14793–14802. [Google Scholar] [CrossRef] [PubMed]
- Duan, H.; Xu, C. Nanoporous PdCr alloys as highly active electrocatalysts for oxygen reduction reaction. Phys. Chem. Chem. Phys. 2016, 18, 4166–4173. [Google Scholar] [CrossRef] [PubMed]
- Jiang, G.; Zhu, H.; Zhang, X.; Shen, B.; Wu, L.; Zhang, S.; Lu, G.; Wu, Z.; Sun, S. Core/Shell Face-Centered Tetragonal FePd/Pd Nanoparticles as an Efficient Non-Pt Catalyst for the Oxygen Reduction Reaction. ACS Nano 2015, 9, 11014–11022. [Google Scholar] [CrossRef] [PubMed]
- Zuo, Y.; Rao, D.; Li, S.; Li, T.; Zhu, G.; Chen, S.; Song, L.; Chai, Y.; Han, H. Atomic Vacancies Control of Pd-Based Catalysts for Enhanced Electrochemical Performance. Adv. Mater. 2018, 30, 1704171–1704177. [Google Scholar] [CrossRef] [PubMed]
- Neergat, M.; Gunasekar, V.; Rahul, R. Carbon-supported Pd–Fe electrocatalysts for oxygen reduction reaction (ORR) and their methanol tolerance. J. Electroanal. Chem. 2011, 658, 25–32. [Google Scholar] [CrossRef]
- Shao, M.-H.; Sasaki, K.; Adzic, R.R. Pd–Fe Nanoparticles as Electrocatalysts for Oxygen Reduction. J. Am. Chem. Soc. 2006, 128, 3526–3527. [Google Scholar] [CrossRef] [PubMed]
- Ou, L. Design of Pd-Based Bimetallic Catalysts for ORR: A DFT Calculation Study. J. Chem. 2015, 2015, 932616. [Google Scholar] [CrossRef] [PubMed]
- Sha, Y.; Yu, T.H.; Merinov, B.V.; Goddard, W.A. DFT Prediction of Oxygen Reduction Reaction on Palladium–Copper Alloy Surfaces. ACS Catal. 2014, 4, 1189–1197. [Google Scholar] [CrossRef]
- Noh, S.H.; Kwak, D.H.; Seo, M.H.; Ohsaka, T.; Han, B. First principles study of oxygen reduction reaction mechanisms on N-doped graphene with a transition metal support. Electrochim. Acta 2014, 140, 225–231. [Google Scholar] [CrossRef]
- Kondo, S.; Nakamura, M.; Maki, N.; Hoshi, N. Active Sites for the Oxygen Reduction Reaction on the Low and High Index Planes of Palladium. J. Phys. Chem. C 2009, 113, 12625–12628. [Google Scholar] [CrossRef]
- Meku, E.; Du, C.; Sun, Y.; Du, L.; Wang, Y.; Yin, G. Electrocatalytic Activity and Stability of Ordered Intermetallic Palladium-Iron Nanoparticles toward Oxygen Reduction Reaction. J. Electrochem. Soc. 2016, 163, F132–F138. [Google Scholar] [CrossRef]
- Zhao, X.; Takao, S.; Higashi, K.; Kaneko, T.; Samjeskè, G.; Sekizawa, O.; Sakata, T.; Yoshida, Y.; Uruga, T.; Iwasawa, Y. Simultaneous Improvements in Performance and Durability of an Octahedral PtNix/C Electrocatalyst for Next-Generation Fuel Cells by Continuous, Compressive, and Concave Pt Skin Layers. ACS Catal. 2017, 7, 4642–4654. [Google Scholar] [CrossRef]
- Wang, H.; Yin, S.; Li, Y.; Yu, H.; Li, C.; Deng, K.; Xu, Y.; Li, X.; Xue, H.; Wang, L. One-step fabrication of tri-metallic PdCuAu nanothorn assemblies as an efficient catalyst for oxygen reduction reaction. J. Mater. Chem. A 2018, 6, 3642–3648. [Google Scholar] [CrossRef]
- Rivera Gavidia, L.M.; García, G.; Anaya, D.; Querejeta, A.; Alcaide, F.; Pastor, E. Carbon-Supported Pt-Free Catalysts with High Specificity and Activity toward the Oxygen Reduction Reaction in Acidic Medium. Appl. Catal. B 2016, 184, 12–19. [Google Scholar] [CrossRef]
- Wang, X.; Orikasa, Y.; Takesue, Y.; Inoue, H.; Nakamura, M.; Minato, T.; Hoshi, N.; Uchimoto, Y. Quantitating the Lattice Strain Dependence of Monolayer Pt Shell Activity toward Oxygen Reduction. J. Am. Chem. Soc. 2013, 135, 5938–5941. [Google Scholar] [CrossRef] [PubMed]
- Xie, S.; Choi, S.-I.; Lu, N.; Roling, L.T.; Herron, J.A.; Zhang, L.; Park, J.; Wang, J.; Kim, M.J.; Xie, Z.; et al. Atomic Layer-by-Layer Deposition of Pt on Pd Nanocubes for Catalysts with Enhanced Activity and Durability toward Oxygen Reduction. Nano Lett. 2014, 14, 3570–3576. [Google Scholar] [CrossRef] [PubMed]
- US DOE, Fuel Cell Technologies Program: Multi-Year Research, Development, and Demonstration Plan—Planned Program Activities for 2011–2020. Available online: https://www.energy.gov/sites/prod/files/2017/05/f34/fcto_myrdd_fuel_cells.pdf (accessed on 1 November 2018).
- Chen, G.; Kuttiyiel, K.A.; Li, M.; Su, D.; Du, L.; Du, C.; Gao, Y.; Fei, W.; Yin, G.; Sasaki, K.; et al. Correlating the electrocatalytic stability of platinum monolayer catalysts with their structural evolution in the oxygen reduction reaction. J. Mater. Chem. A 2018, 6, 20725–20736. [Google Scholar] [CrossRef]
- Humbert, M.P.; Smith, B.H.; Wang, Q.; Ehrlich, S.N.; Shao, M. Synthesis and Characterization of Palladium-Platinum Core–Shell Electrocatalysts for Oxygen Reduction. Electrocatalysis 2012, 3, 298–303. [Google Scholar] [CrossRef]
- Bliznakov, S.T.; Vukmirovic, M.B.; Yang, L.; Sutter, E.A.; Adzic, R.R. Pt Monolayer on Electrodeposited Pd Nanostructures: Advanced Cathode Catalysts for PEM Fuel Cells. J. Electrochem. Soc. 2012, 159, F501–F506. [Google Scholar] [CrossRef]
- Xu, Q.; Chen, W.; Yan, Y.; Wu, Z.; Jiang, Y.; Li, J.; Bian, T.; Zhang, H.; Wu, J.; Yang, D. Multimetallic AuPd@Pd@Pt core–interlayer-shell icosahedral electrocatalysts for highly efficient oxygen reduction reaction. Sci. Bull. 2018, 63, 494–501. [Google Scholar] [CrossRef]
- He, D.S.; He, D.P.; Wang, J.; Lin, X.; Yin, P.Q.; Hong, X.; Wu, Y.E.; Li, Y.D. Ultrathin Icosahedral Pt-Enriched Nanocage with Excellent Oxygen Reduction Reaction Activity. J. Am. Chem. Soc. 2016, 138, 1494–1497. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Vara, M.; Luo, M.; Huang, H.; Ruditskiy, A.; Park, J.; Bao, S.; Liu, J.; Howe, J.; Chi, M.; et al. Pd@Pt Core–Shell Concave Decahedra: A Class of Catalysts for the Oxygen Reduction Reaction with Enhanced Activity and Durability. J. Am. Chem. Soc. 2015, 137, 15036–15042. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; Yang, Y.; DiSalvo, F.J.; Abruña, H.D. Pt-Decorated Composition-Tunable Pd–Fe@Pd/C Core–Shell Nanoparticles with Enhanced Electrocatalytic Activity toward the Oxygen Reduction Reaction. J. Am. Chem. Soc. 2018, 140, 7248–7255. [Google Scholar] [CrossRef] [PubMed]
- Kuttiyiel, K.A.; Sasaki, K.; Su, D.; Vukmirovic, M.B.; Marinkovic, N.S.; Adzic, R.R. Pt Monolayer on Au-Stabilized PdNi Core–Shell Nanoparticles for Oxygen Reduction Reaction. Electrochim. Acta 2013, 110, 267–272. [Google Scholar] [CrossRef]
- Huang, J.-F.; Tseng, P.-K. High performance layer-by-layer Pt3Ni(Pt-skin)-modified Pd/C for the oxygen reduction reaction. Chem. Sci. 2018, 9, 6134–6142. [Google Scholar] [CrossRef] [PubMed]
- Marković, N.M.; Ross, P.N., Jr. Surface science studies of model fuel cell electrocatalysts. Surf. Sci. Rep. 2002, 45, 117–229. [Google Scholar] [CrossRef]
- Shao, M.H.; Huang, T.; Liu, P.; Zhang, J.; Sasaki, K.; Vukmirovic, M.B.; Adzic, R.R. Palladium Monolayer and Palladium Alloy Electrocatalysts for Oxygen Reduction. Langmuir 2006, 22, 10409–10415. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Kang, Y.J.; Huo, Z.Y.; Zhu, Z.W.; Huang, W.Y.; Xin, H.L.L.; Snyder, J.D.; Li, D.G.; Herron, J.A.; Mavrikakis, M.; et al. Highly Crystalline Multimetallic Nanoframes with Three-Dimensional Electrocatalytic Surfaces. Science 2014, 343, 1339–1343. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.Q.; Zhao, Z.P.; Cao, L.; Chen, Y.; Zhu, E.B.; Lin, Z.Y.; Li, M.F.; Yan, A.M.; Zettl, A.; Wang, Y.M.; et al. High-performance transition metal-doped Pt3Ni octahedra for oxygen reduction reaction. Science 2015, 348, 1230–1234. [Google Scholar] [CrossRef] [PubMed]
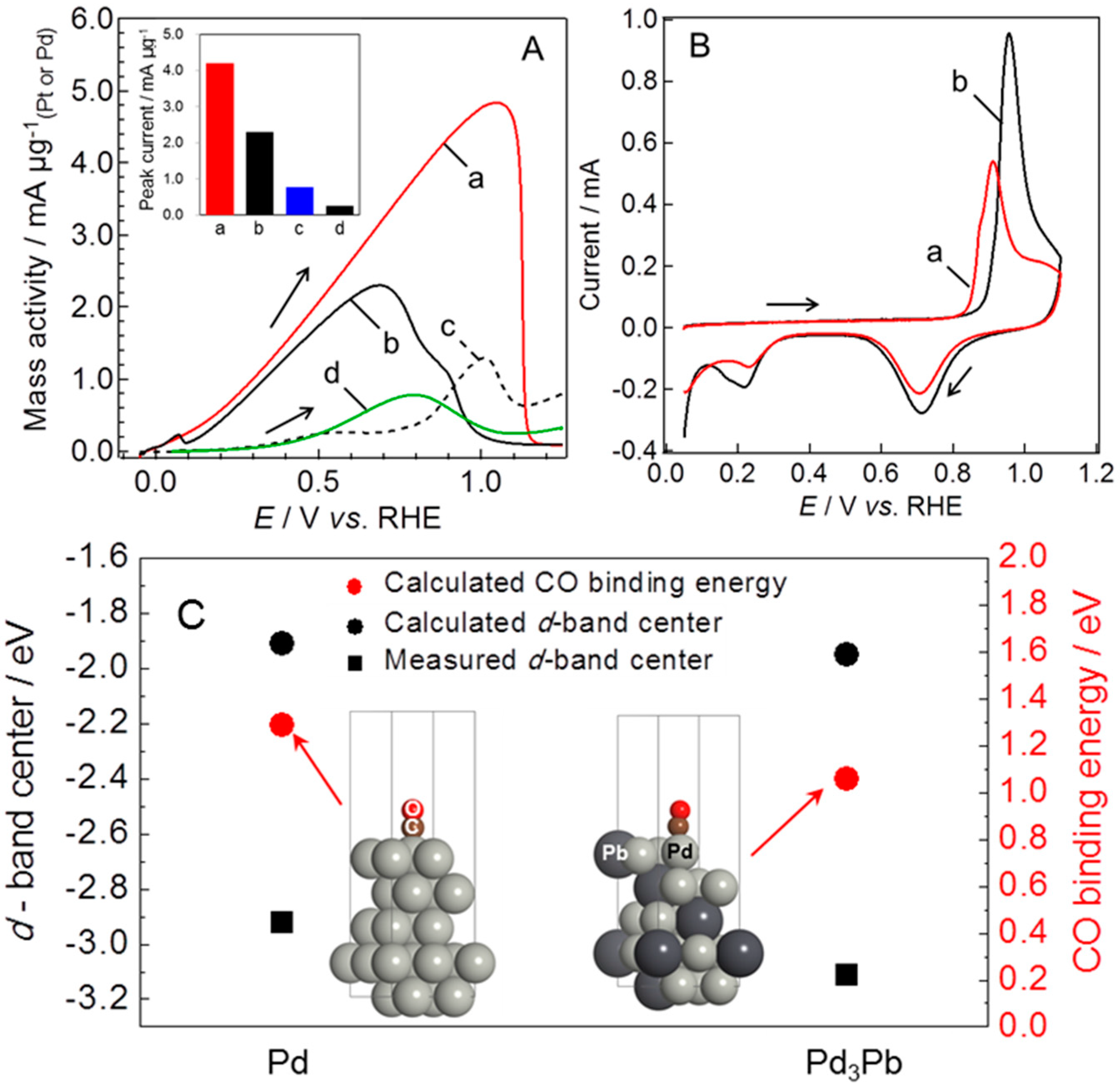
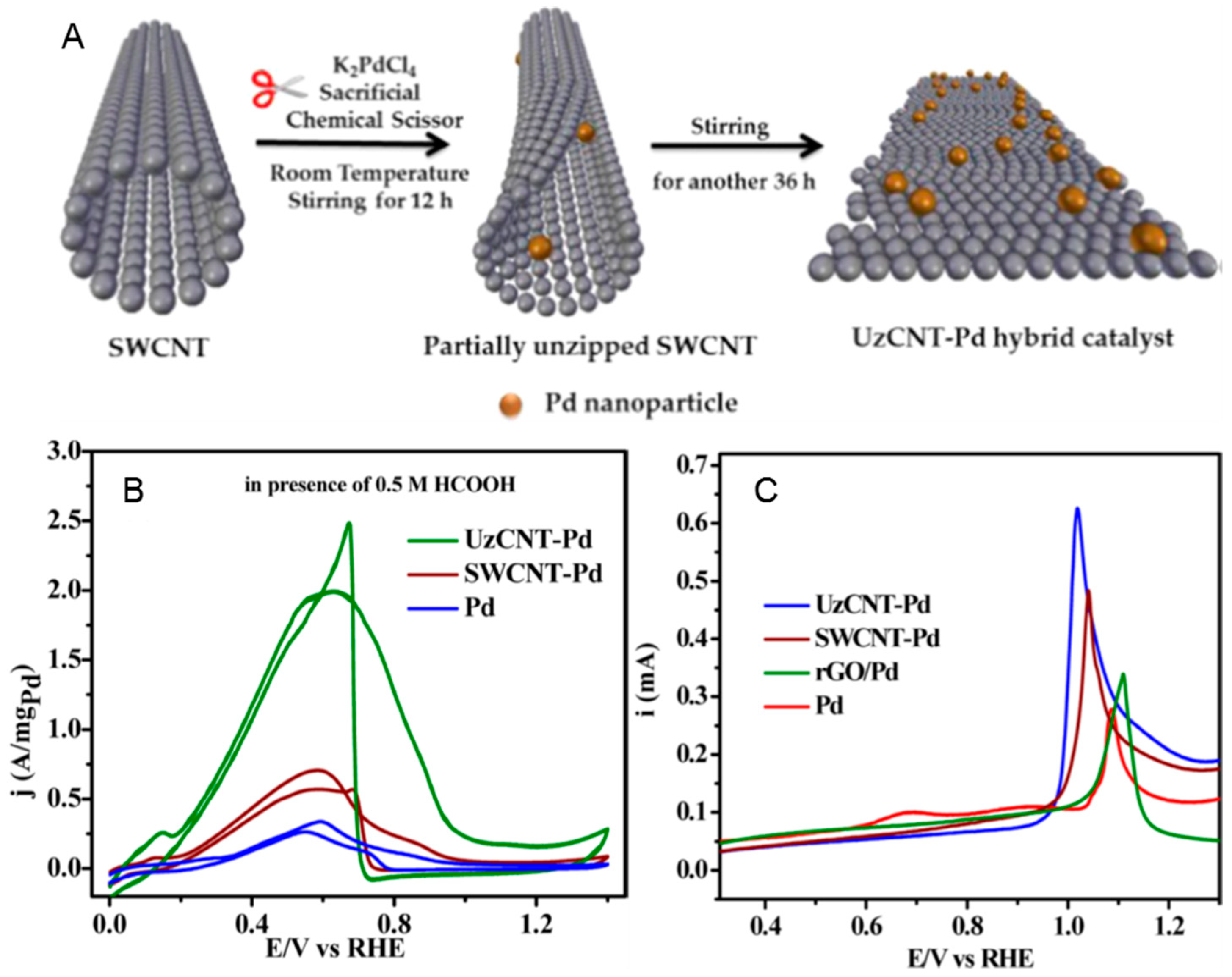
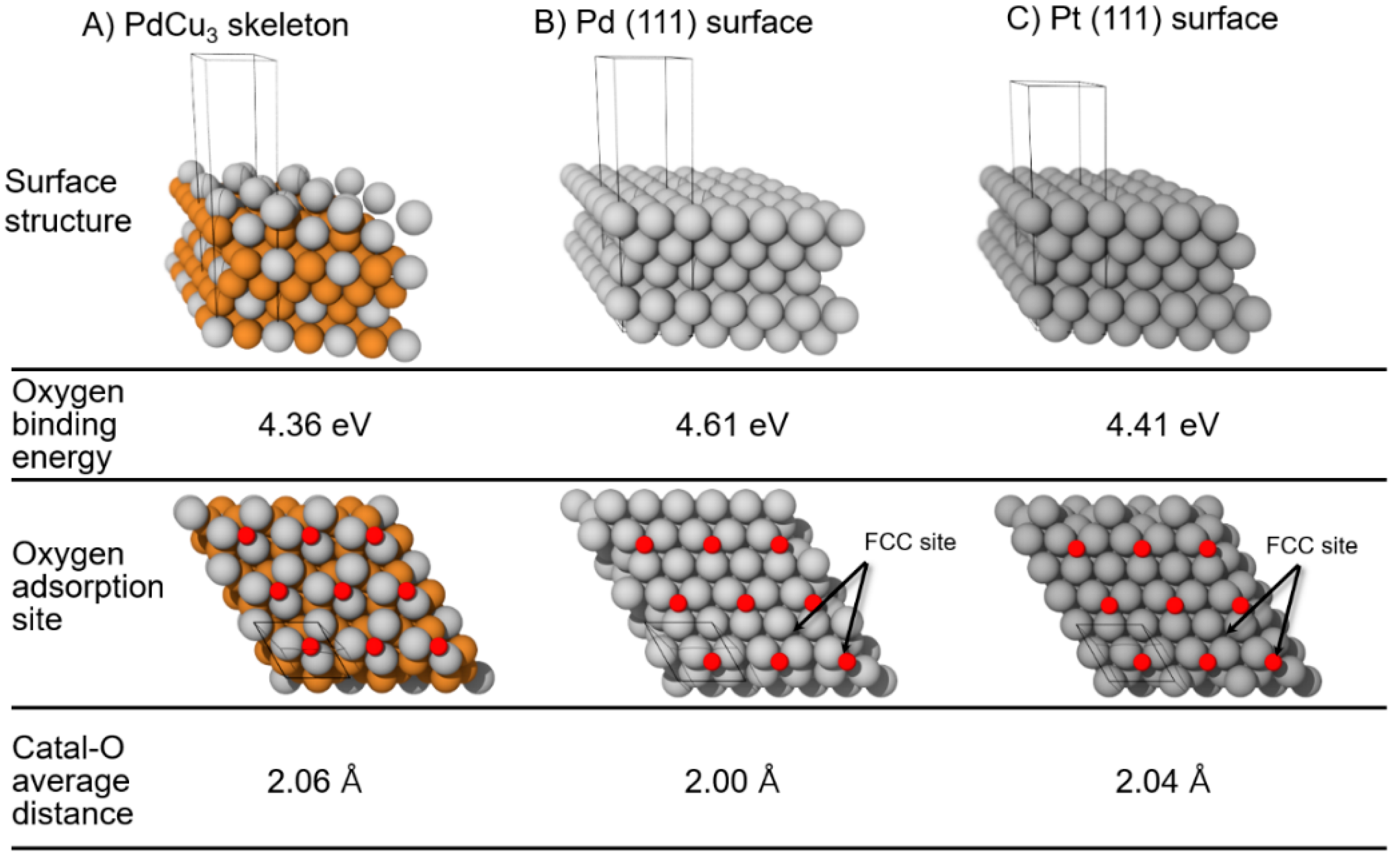
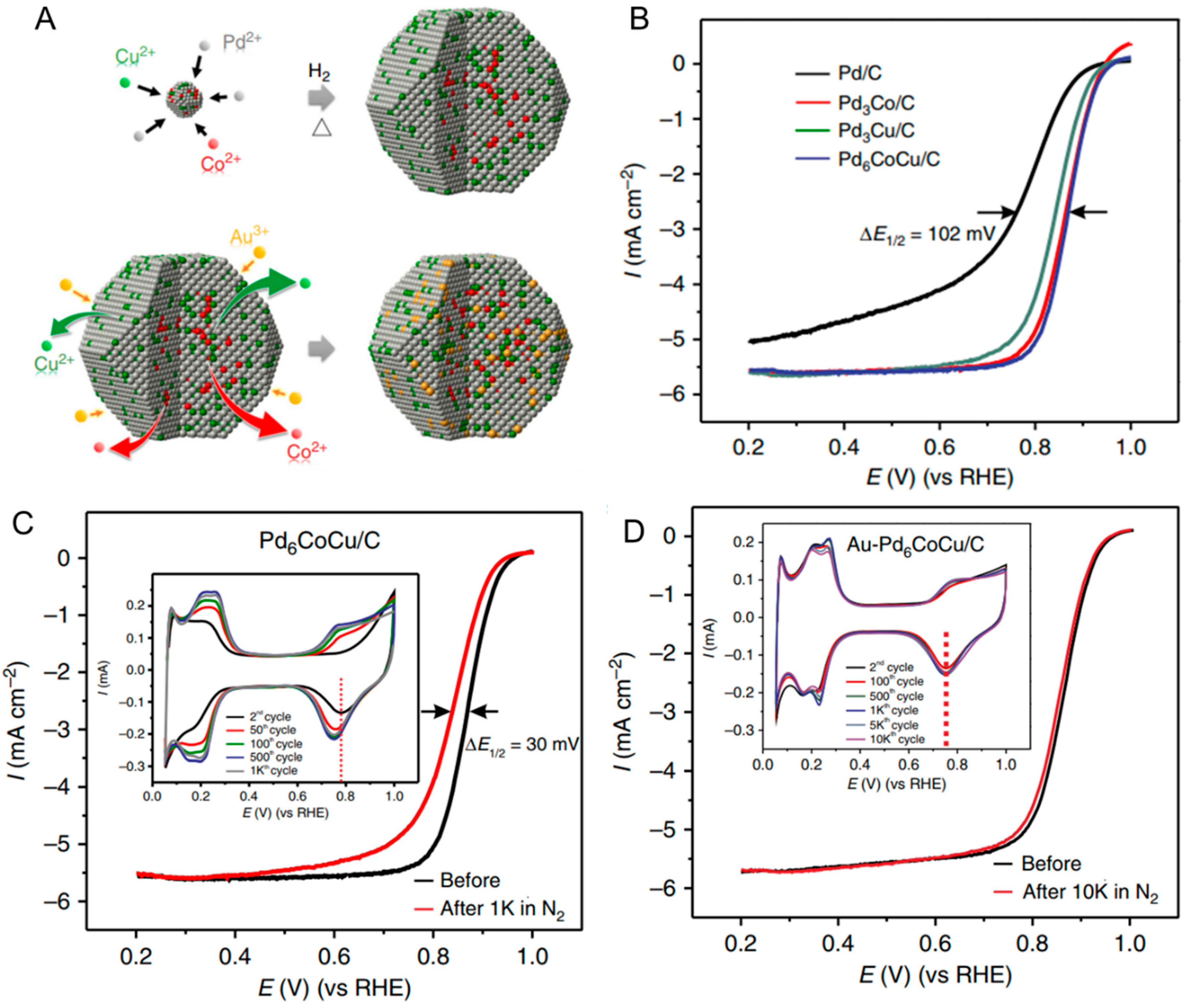
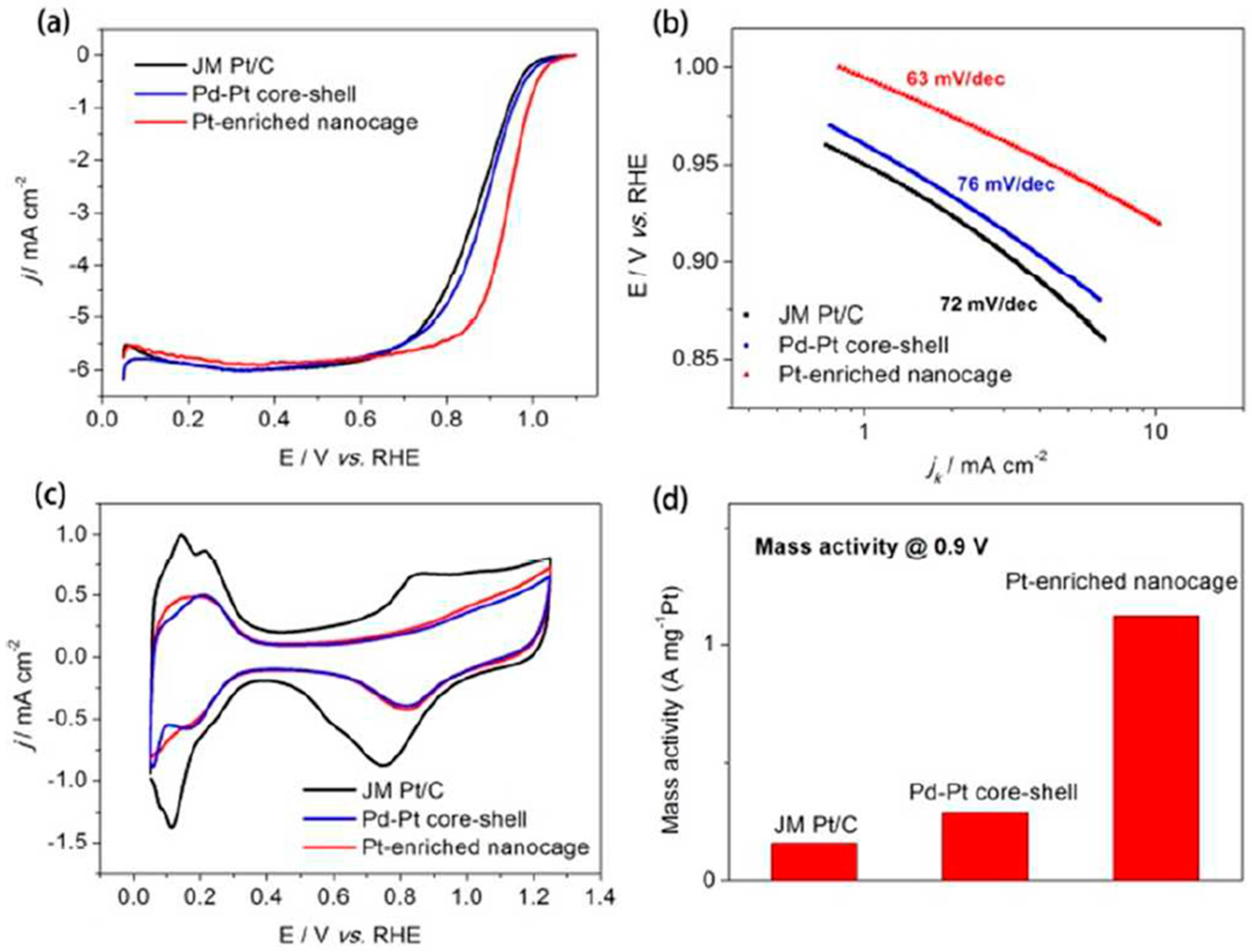
| Catalyst | MA (A mg−1Pd) | Concentration of Formic Acid (mol L−1) | E for Peak Top of CO Stripping (V versus RHE) | References |
|---|---|---|---|---|
| Pd | 0.35 | 0.1 | 1.00 | [46] |
| Pd1.1/WO2.72 | 1.15 | 0.1 | 0.93 | [46] |
| Pd@Au | 0.52 | 3 | 0.872 | [53] |
| 8 nm Pd0.5Co0.5 | 0.18 a | 2 | - | [47] |
| Pd nanosheets | 0.3 a | 0.25 | - | [55] |
| Pd@Ni1.6B0.02 | 0.21 a | 0.5 | 0.939 | [56] |
| Pd0.41Ag | 1.7 a | 0.5 | - | [57] |
| Pd–g-C3N4–rGO-2 | 1.02 | 0.5 | 0.96 | [58] |
| Pd57Ni43 nanowires | 0.62 | 0.5 | - | [48] |
| PdCu alloy multipods | 0.15 | 1 | 0.96 | [50] |
| PdAg/Ti0.5Cr0.5N | 0.31 | 1 | - | [59] |
| Pd0.57Ni0.13Cu0.30 | 0.48 | 0.5 | 0.92 | [49] |
| PdCu | 0.38 | 0.5 | 0.909 | [49] |
| PdNi | 0.22 | 0.5 | - | [49] |
| UzCNT-Pd | 0.71 | - | 1.01 | [49] |
| Pd3Pb (dealloy) | 1.00 | 0.5 | 0.906 | [31] |
| Catalyst | MA at 0.90 V/A mg−1Pd | E1/2/V versus RHE | References |
|---|---|---|---|
| Pd3Pb nanoparticles | 0.17 | 0.92 | [74] |
| Pd3Pb nanowires (NWs) | 0.61 | 0.92 | [8] |
| Pd3Pb tripods | 0.56 | 0.91 | [75] |
| Pd3Pb square nanoplates | 0.62 | 0.91 | [76] |
| Pd3Pb square nanoplates | 0.78 | 0.88 | [77] |
| Pd/W18O49 | 0.22 | 0.88 | [79] |
| Pd-B | 0.15 | 0.88 | [80] |
| Pd@Pd-Ni MWCNTs | 0.071 | 0.87 | [78] |
| Au NWs@Pd@PEI | 0.30 | 0.90 | [81] |
| Pd3Fe | 0.097 | 0.89 | [82] |
| Catalyst | MA at 0.90 (0.85) V/A mg−1Pt or Pd | E1/2/V versus RHE | References |
|---|---|---|---|
| Pt | 0.21–0.23 | about 0.826 | [89,90,91] |
| Pd | (0.03–0.025) | about 0.78 | [92] |
| PdNiCo NPs | 0.047 (0.137) | 0.86 | [92] |
| PdCu | 0.041 (0.156) | 0.85 | [93] |
| PdCu3 at 600 °C | (0.10) a | 0.82 | [94] |
| PdCu3 dealloyed | 0.047 | 0.88 | [95] |
| Pd8CoZn | 0.019 (0.141) | 0.84 | [96] |
| NP-Pd0.75Cr0.25 | 0.158 | 0.89 | [97] |
| PdCu alloy multipods | - | 0.819 | [50] |
| C-fct-Fe46Pd54 | 0.1 | 0.87 | [98] |
| v-PdCuCo | 0.175 | 0.91 | [99] |
| Pd3Fe | (0.12) | 0.80 | [100] |
| Pd3Fe | (0.6) | 0.85 | [101] |
| Catalyst | MA at 0.90 V/A mg−1Pt | SA at 0.90 V/A m−2 | Ref |
|---|---|---|---|
| Pt | 0.20–0.23 | 0.23–0.31 | [89,90,91] |
| Pd@Pt | 0.34, 0.8 | - | [111,114] |
| PdNR@PtML | 1.81 | 0.93 | [115] |
| Au60Pd40@Pt | 1.82 | 2.2 | [116] |
| Pt-enriched nanocage | 1.12 | 2.48 | [117] |
| Pd@Pt concave decahedra | 1.60 | 1.66 | [118] |
| Pd8CoZn@Pt | 2.62 | 4.76 | [96] |
| Pd42Fe58@Pt | 2.8 | - | [119] |
| Pd@PtNi | 2.5 | 2.7 | [120] |
| Pd@Pt3Ni(Pt-skin) | 16.7 | 14.2 | [121] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gunji, T.; Matsumoto, F. Electrocatalytic Activities towards the Electrochemical Oxidation of Formic Acid and Oxygen Reduction Reactions over Bimetallic, Trimetallic and Core–Shell-Structured Pd-Based Materials. Inorganics 2019, 7, 36. https://doi.org/10.3390/inorganics7030036
Gunji T, Matsumoto F. Electrocatalytic Activities towards the Electrochemical Oxidation of Formic Acid and Oxygen Reduction Reactions over Bimetallic, Trimetallic and Core–Shell-Structured Pd-Based Materials. Inorganics. 2019; 7(3):36. https://doi.org/10.3390/inorganics7030036
Chicago/Turabian StyleGunji, Takao, and Futoshi Matsumoto. 2019. "Electrocatalytic Activities towards the Electrochemical Oxidation of Formic Acid and Oxygen Reduction Reactions over Bimetallic, Trimetallic and Core–Shell-Structured Pd-Based Materials" Inorganics 7, no. 3: 36. https://doi.org/10.3390/inorganics7030036
APA StyleGunji, T., & Matsumoto, F. (2019). Electrocatalytic Activities towards the Electrochemical Oxidation of Formic Acid and Oxygen Reduction Reactions over Bimetallic, Trimetallic and Core–Shell-Structured Pd-Based Materials. Inorganics, 7(3), 36. https://doi.org/10.3390/inorganics7030036