Continuous Emission Monitoring of Tetrafluoromethane Using Quantum Cascade Lasers
Abstract
:1. Introduction
2. In-Situ Tetrafluoromethane Sensor
3. Laboratory Evaluation
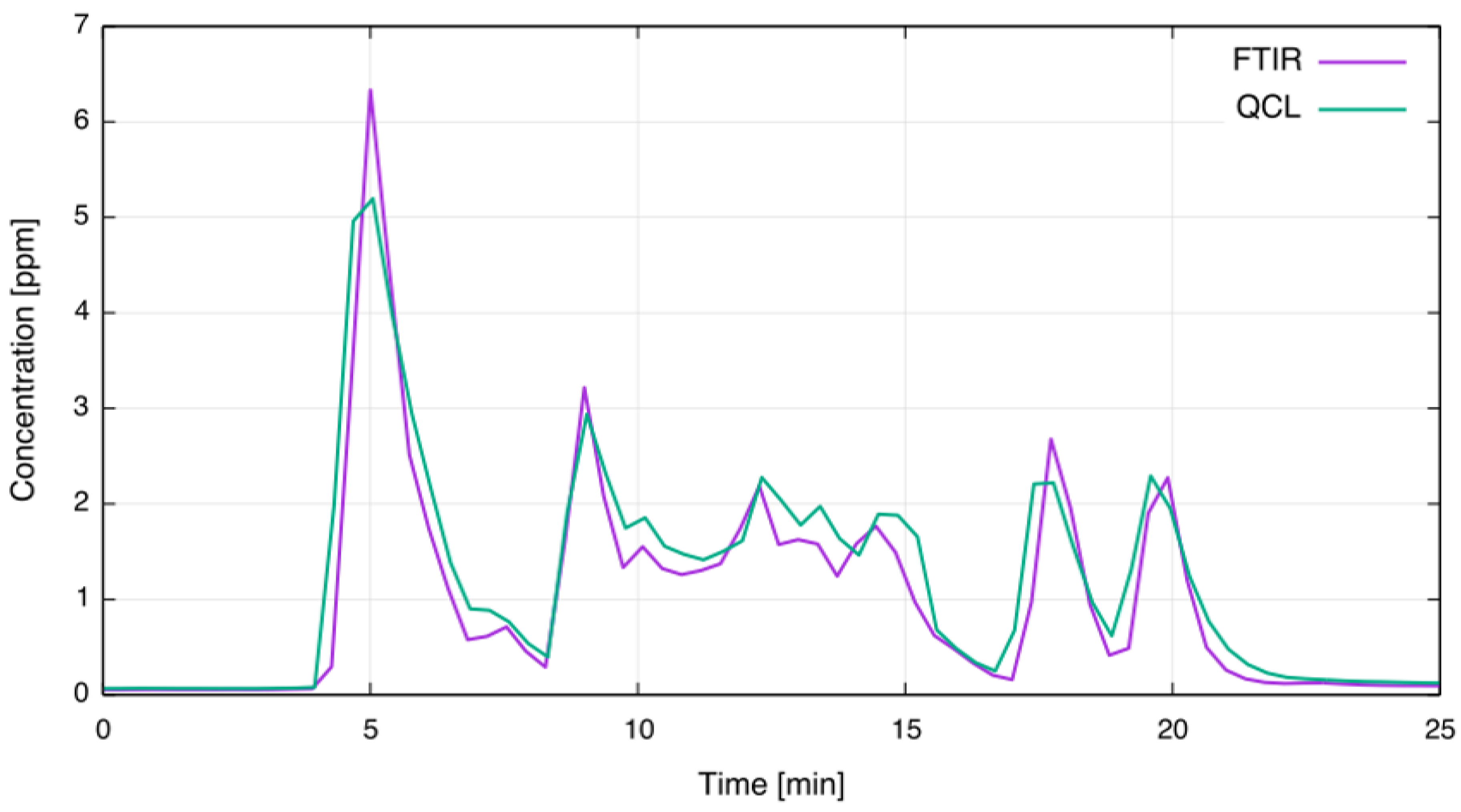
4. Field-Test at an Alcoa Aluminum Smelter
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Solomon, S.; Qin, D.; Manning, M.; Chen, Z.; Marquis, M.; Averyt, K.; Tignor, M.M.; Miller, H.L. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK, 2007. [Google Scholar]
- Kim, J.; Fraser, P.J.; Li, S.; Mühle, J.; Ganesan, A.L.; Krummel, P.B.; Steele, L.P.; Park, S.; Kim, S.K.; Park, M.K.; et al. Quantifying aluminum and semiconductor industry perfluorocarbon emissions from atmospheric measurements. Geophys. Res. Lett. 2014, 41, 4787–4794. [Google Scholar] [CrossRef]
- Grjotheim, K.; Kvande, H. Introduction to Aluminum Electrolysis; Aluminum-Verlag: Düsseldorf, Germany, 1993. [Google Scholar]
- Dando, N.R.; Xu, W.; Marks, J. Comparison of PFC Emission for Operating and Newly Started Pots in the Alcoa Fjardaal Point Fed Prebake Smelter. In Light Metals 2009; Bearne, G., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA; pp. 269–273.
- Zarouni, A.; Reverdy, M.; Zarouni, A.A.; Venkatasubramaniam, K. A Study of Low Voltage PFC Emissions at DUBAL. In Light Metals 2013; Sadler, B.A., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA; pp. 859–863.
- Dando, N.R.; Menegazzo, N.; Espinoza-Nava, L.; Westendorf, N.; Batista, E. Non Anode Effect PFCs: Measurement Considerations and Potential Impacts. In Light Metals 2015; Hyland, M., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA; pp. 551–554.
- Dando, N.R.; Xu, W.; Espinoza-Nava, L. In-Plant Performance Comparison of Fourier Transform and Photoacoustic Infra-Red PFC Monitors. In Light Metals 2004; Tabereaux, A.T., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA; pp. 381–385.
- U.S. Environmental Protection Agency; International Aluminium Institute. Available online: http://www3.epa.gov/highgwp/aluminum-pfc/documents/measureprotocol.pdf (accessed on 31 March 2016).
- Gouvernement du Québec. Available online: http://www2.publicationsduquebec.gouv.qc.ca/dynamicSearch/telecharge.php?type=3&file=/Q_2/Q2R15_A.htm (accessed on 31 March 2016).
- Linnerud, I.; Kaspersen, P.; Jaeger, T. Gas monitoring in the process industry using diode laser spectroscopy. Appl. Phys. B 1998, 67, 297–305. [Google Scholar] [CrossRef]
- Rothman, L.; Gordon, I.; Babikov, Y.; Barbe, A.; Benner, D.C.; Bernath, P.; Birk, M.; Bizzocchi, L.; Boudon, V.; Brown, L.; et al. The HITRAN2012 molecular spectroscopic database. J. Quant. Spectrosc. Radiat. Transf. 2013, 130, 4–50. [Google Scholar] [CrossRef]
- Pacific Northwest National Laboratory. Available online: https://secure2.pnl.gov/nsd/nsd.nsf/ (accessed on 31 March 2016).
- Rothman, L.; Gordon, I.; Barber, R.; Dothe, H.; Gamache, R.; Goldman, A.; Perevalov, V.; Tashkun, S.; Tennyson, J. HITEMP, the high-temperature molecular spectroscopic database. J. Quant. Spectrosc. Radiat. Transf. 2010, 111, 2139–2150. [Google Scholar] [CrossRef]
- Bouchard, G.; Kallmeyer, J.; Marks, J. PFC Emissions Measurements from Canadian Primary Aluminum Production. In Light Metals 2001; Anjier, J.L., Ed.; The Minerals, Metals & Materials Society: Warrendale, PA, USA; pp. 283–288.
- Gamble, H.A.; Mackay, G.I.; Karecki, D.R.; Pisano, J.T.; Schiff, H.I. Development of a TDLAS based Methodology for Monitoring Perfluorocarbon Production during the Aluminium Smelting Process. In Light Metals 2001; Anjier, J.L., Ed.; The Minerals, Metals & Materials Society: Warrendale, PA, USA; pp. 275–281.
- Kimmerle, F.M.; Potvin, G.; Pisano, J.T. Measured Versus Calculated Reduction of the PFC Emissions from Prebaked Hall Héroult Cells. In Light Metals 1997; Huglen, R., Ed.; The Minerals, Metals & Materials Society: Warrendale, PA, USA; pp. 165–171.
- Faist, J. Quantum Cascade Lasers; Oxford University Press: Oxford, UK, 2013. [Google Scholar]
- Geiser, P. New Opportunities in Mid-Infrared Emission Control. Sensors 2015, 15, 22724–22736. [Google Scholar] [CrossRef] [PubMed]
- Tao, L.; Sun, K.; Khan, M.A.; Miller, D.J.; Zondlo, M.A. Compact and portable open-path sensor for simultaneous measurements of atmospheric N2O and CO using a quantum cascade laser. Opt. Express 2012, 20, 28106–28118. [Google Scholar] [CrossRef] [PubMed]
- NEO Monitors AS. Available online: http://neomonitors.com/products/lasergas-q-no/ (accessed on 31 March 2016).
- Geiser, P.; Dang, D.; Bohman, A.; Kaspersen, P. Mid-infrared Sulfur Dioxide Measurements at Elevated Temperatures for Emission Control. In Lasers, Sources, and Related Photonic Devices; Optical Society of America: Washington, DC, USA, 2012. [Google Scholar]
- Kluczynski, P.; Gustafsson, J.; Lindberg, Å.M.; Axner, O. Wavelength modulation absorption spectrometry—An extensive scrutiny of the generation of signals. Spectrochim. Acta Part B At. Spectrosc. 2001, 56, 1277–1354. [Google Scholar] [CrossRef]
- Geiser, P.; Kaspersen, P. Quantum cascade laser based tetrafluoromethane and nitrogen oxide measurements for emission monitoring applications. In Imaging and Applied Optics 2014; Optical Society of America: Washington, DC, USA, 2014. [Google Scholar]
- Ren, W.; Jiang, W.; Tittel, F.K. Single-QCL-based absorption sensor for simultaneous trace-gas detection of CH4 and N2O. Appl. Phys. B 2014, 117, 245–251. [Google Scholar] [CrossRef]
- Cao, Y.; Sanchez, N.P.; Griffin, R.; Tittel, F.K.; Dong, L. Mid-infrared detection of atmospheric CH4, N2O and H2O based on a single continuous wave quantum cascade laser. In CLEO: 2015; Optical Society of America: Washington, DC, USA, 2015. [Google Scholar]
- Werle, P. Accuracy and precision of laser spectrometers for trace gas sensing in the presence of optical fringes and atmospheric turbulence. Appl. Phys. B 2010, 102, 313–329. [Google Scholar] [CrossRef]
- Klein, A.; Witzel, O.; Ebert, V. Rapid, Time-Division Multiplexed, Direct Absorption- and Wavelength Modulation-Spectroscopy. Sensors 2014, 14, 21497–21513. [Google Scholar] [CrossRef] [PubMed]
- Espinoza-Nava, L.; Menegazzo, N.; Dando, N.R.; Geiser, P. QCL-Based Perfluorocarbon Emission Monitoring. In Light Metals 2016; Williams, E., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA; pp. 541–544.
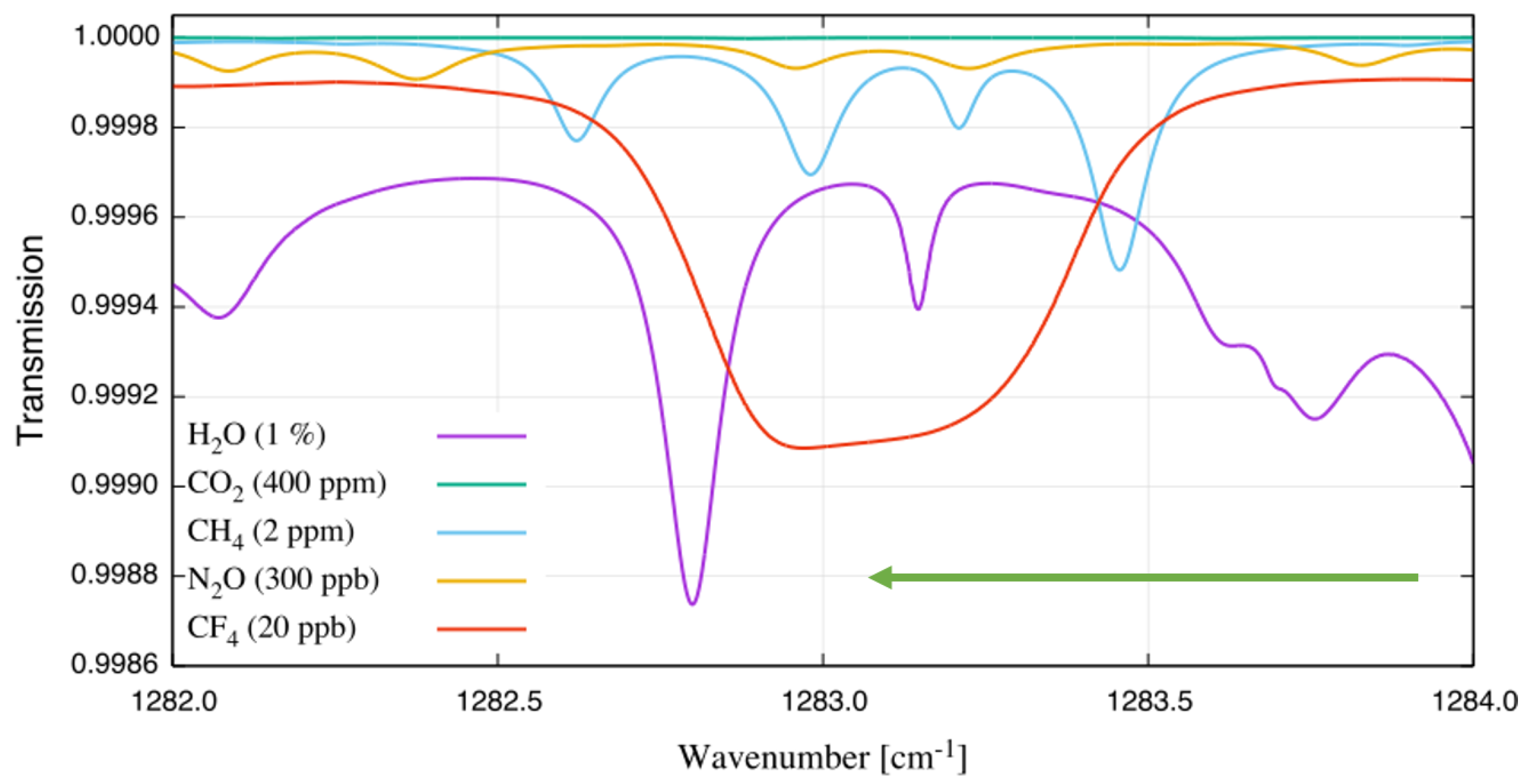
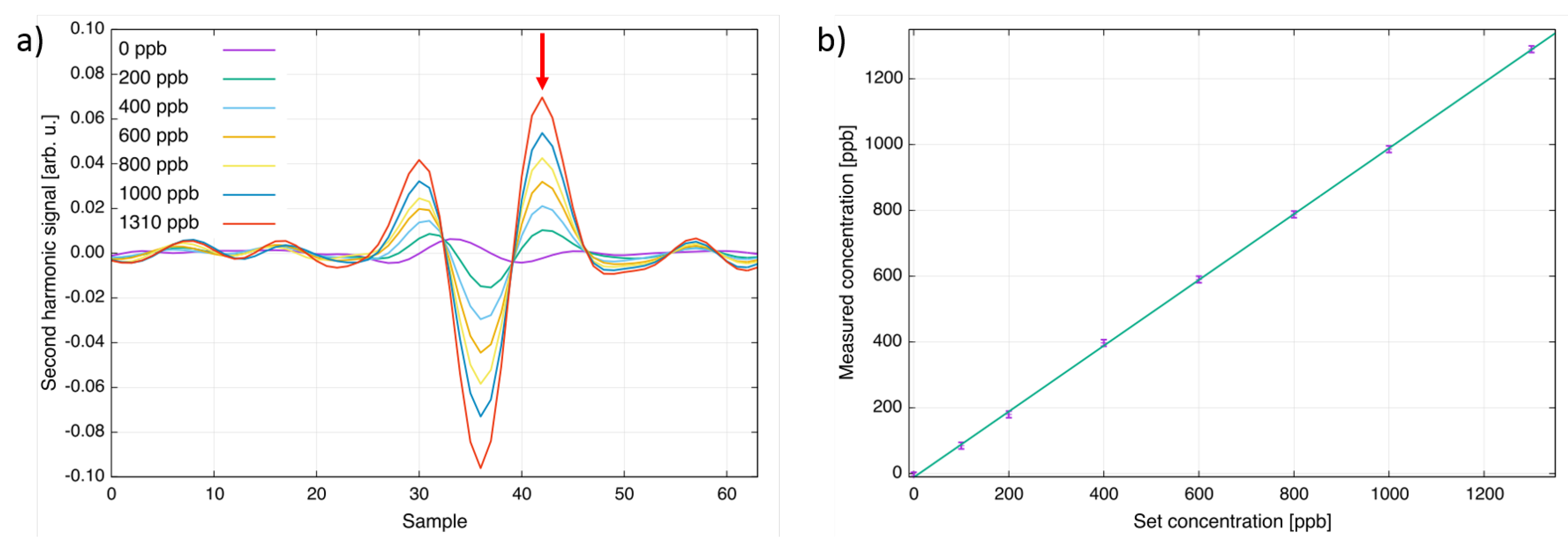
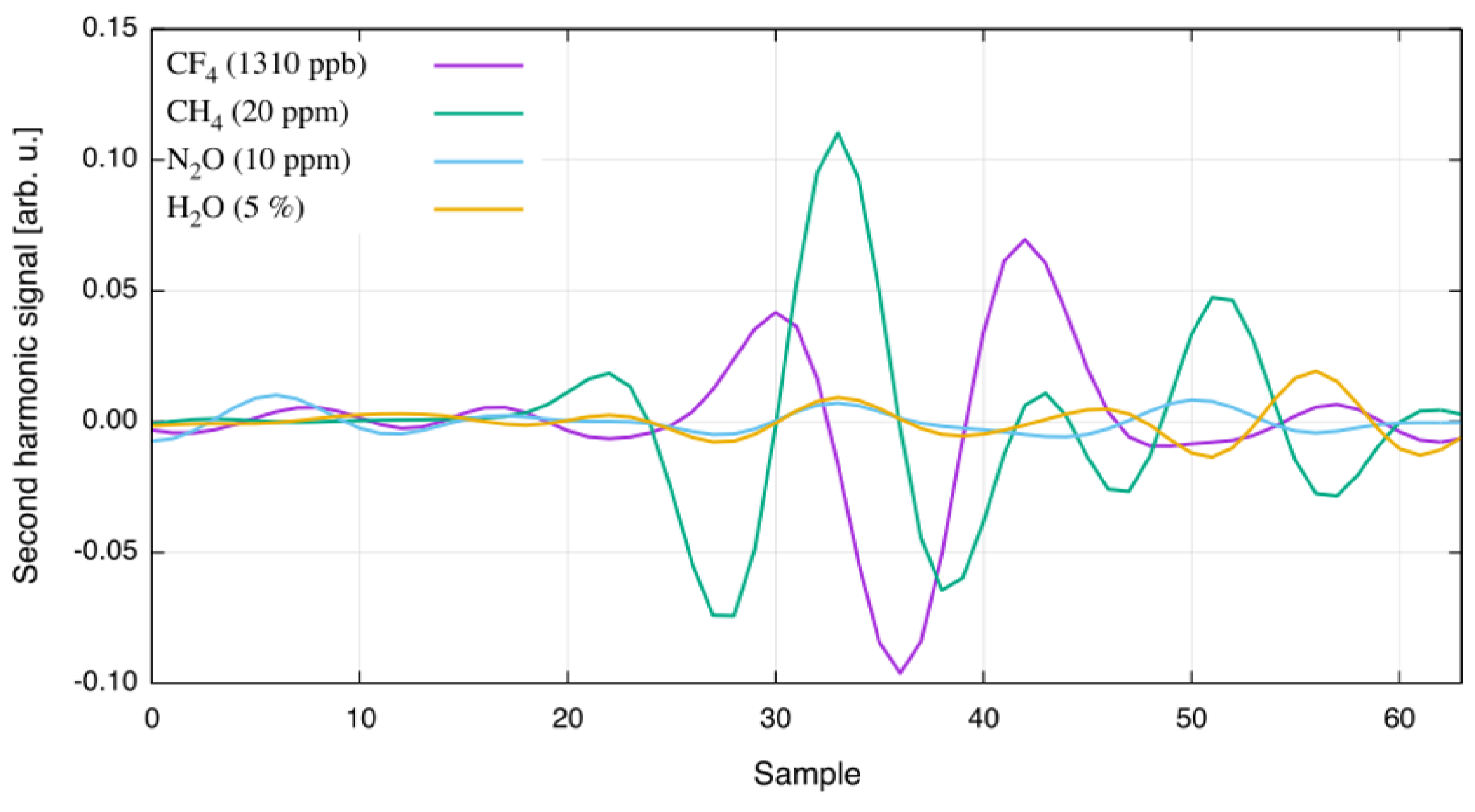


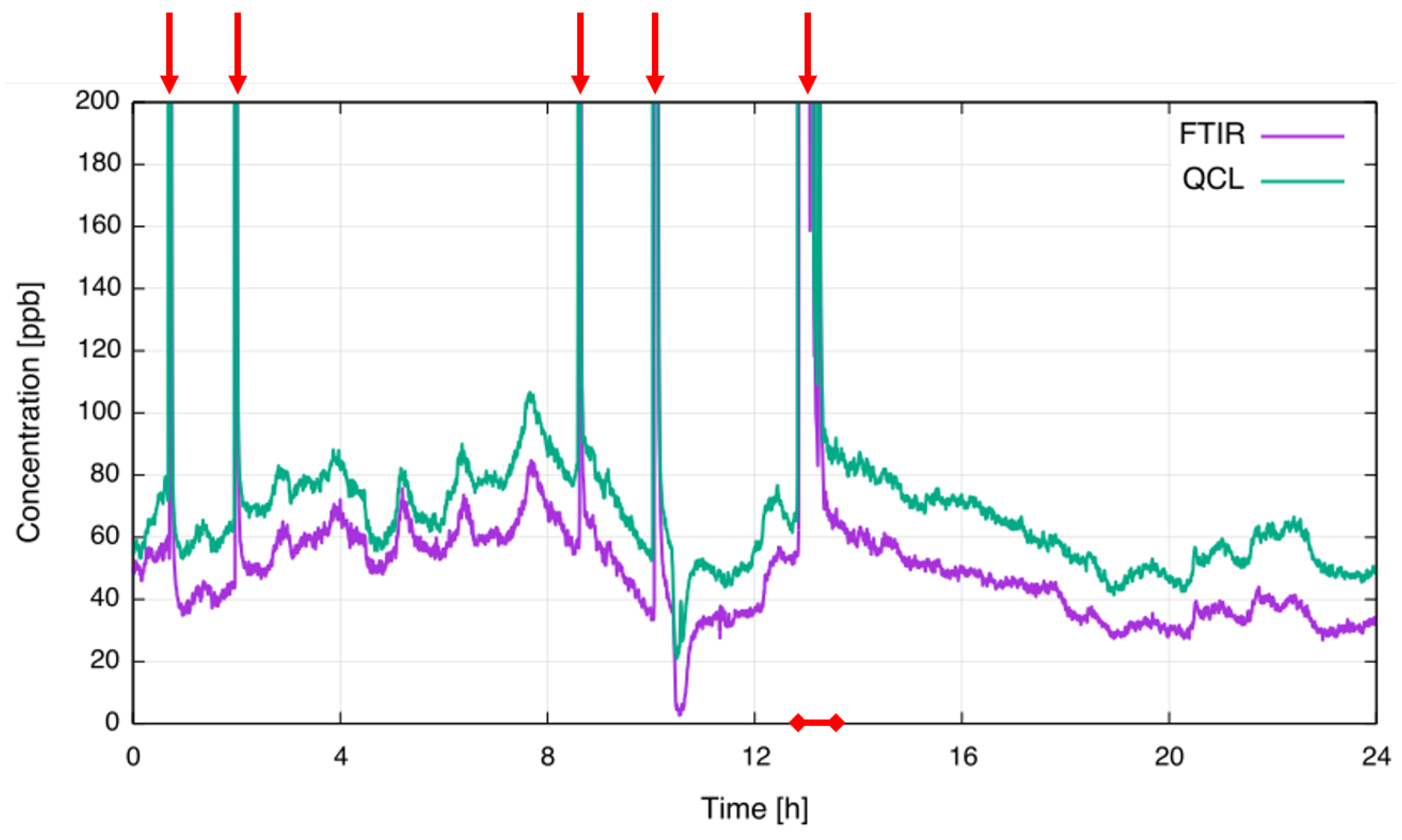
| Species | Atmospheric Concentration | Lifetime (y) | GWP 500-y |
|---|---|---|---|
| Carbon dioxide (CO) | 379 ppm ± 0.65 ppm | Variable | 1 |
| Methane (CH) | 1.774 ppm ± 1.8 ppb | 12 | 7.6 |
| Nitrous oxide (NO) | 319 ppb ± 0.12 ppb | 114 | 153 |
| Tetrafluoromethane (CF) | 74 ppt ± 1.6 ppt | 50,000 | 11,200 |
| Hexafluoroethane (CF) | 2.9 ppt ± 0.025 ppt | 10,000 | 18,200 |
| Species | Chemical Formula | Concentration | Interference |
|---|---|---|---|
| Water vapor | HO | 1% | 3 ppb |
| Methane | CH | 1 ppm | 10 ppb |
| Nitrous oxide | NO | 1 ppm | –5 ppb |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Geiser, P.; Avetisov, V.; Espinoza-Nava, L.; Menegazzo, N.; Kaspersen, P. Continuous Emission Monitoring of Tetrafluoromethane Using Quantum Cascade Lasers. Photonics 2016, 3, 16. https://doi.org/10.3390/photonics3020016
Geiser P, Avetisov V, Espinoza-Nava L, Menegazzo N, Kaspersen P. Continuous Emission Monitoring of Tetrafluoromethane Using Quantum Cascade Lasers. Photonics. 2016; 3(2):16. https://doi.org/10.3390/photonics3020016
Chicago/Turabian StyleGeiser, Peter, Viacheslav Avetisov, Luis Espinoza-Nava, Nicola Menegazzo, and Peter Kaspersen. 2016. "Continuous Emission Monitoring of Tetrafluoromethane Using Quantum Cascade Lasers" Photonics 3, no. 2: 16. https://doi.org/10.3390/photonics3020016
APA StyleGeiser, P., Avetisov, V., Espinoza-Nava, L., Menegazzo, N., & Kaspersen, P. (2016). Continuous Emission Monitoring of Tetrafluoromethane Using Quantum Cascade Lasers. Photonics, 3(2), 16. https://doi.org/10.3390/photonics3020016





