Photothermal Performance Testing of Lithium Niobate After Ion Beam Radiation
Abstract
1. Introduction
2. Theoretical Analysis
3. Experiment
3.1. Parameter Design and Irradiation Protocol
- 400–800 V (100 V increments)—chosen to cover the range where ion energy is sufficient to induce surface defects without excessive bulk damage [27].
- 20–40 mA (5 mA steps)—selected to study the effect of ion flux on defect formation and annihilation [26].
- 30–150 s (30 s increments)—chosen to observe both defect accumulation and thermal annealing effects [25].
3.2. Photothermal Detection Methodology
3.3. Baseline Characterization
4. Results and Discussion
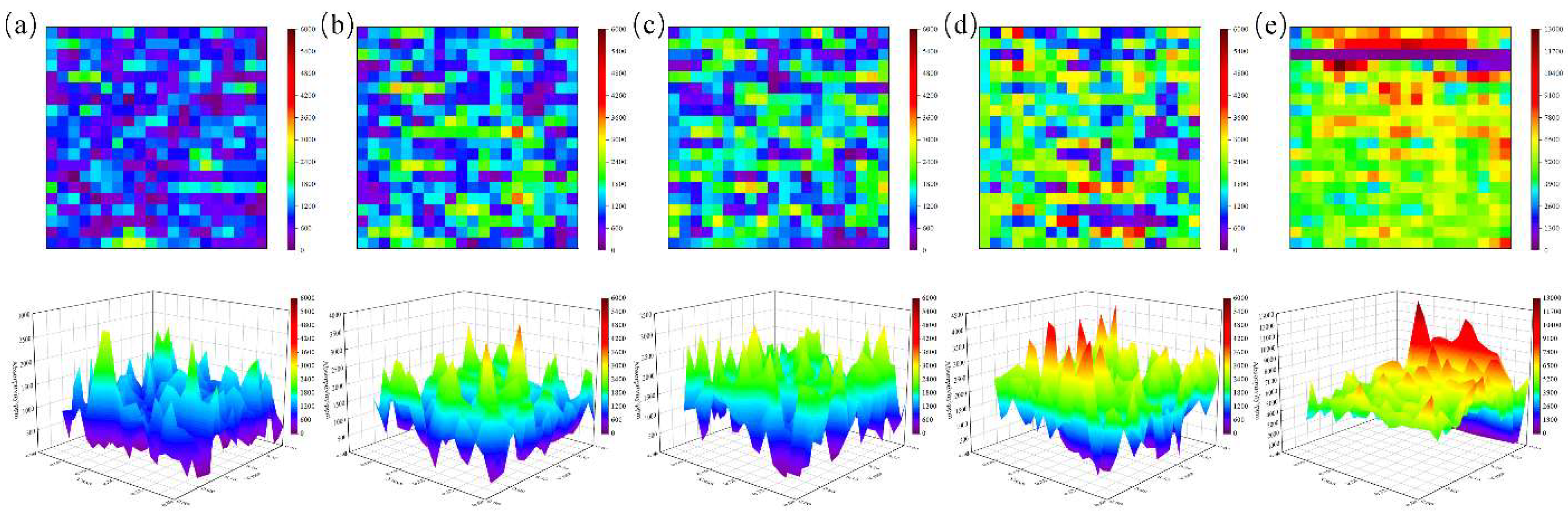
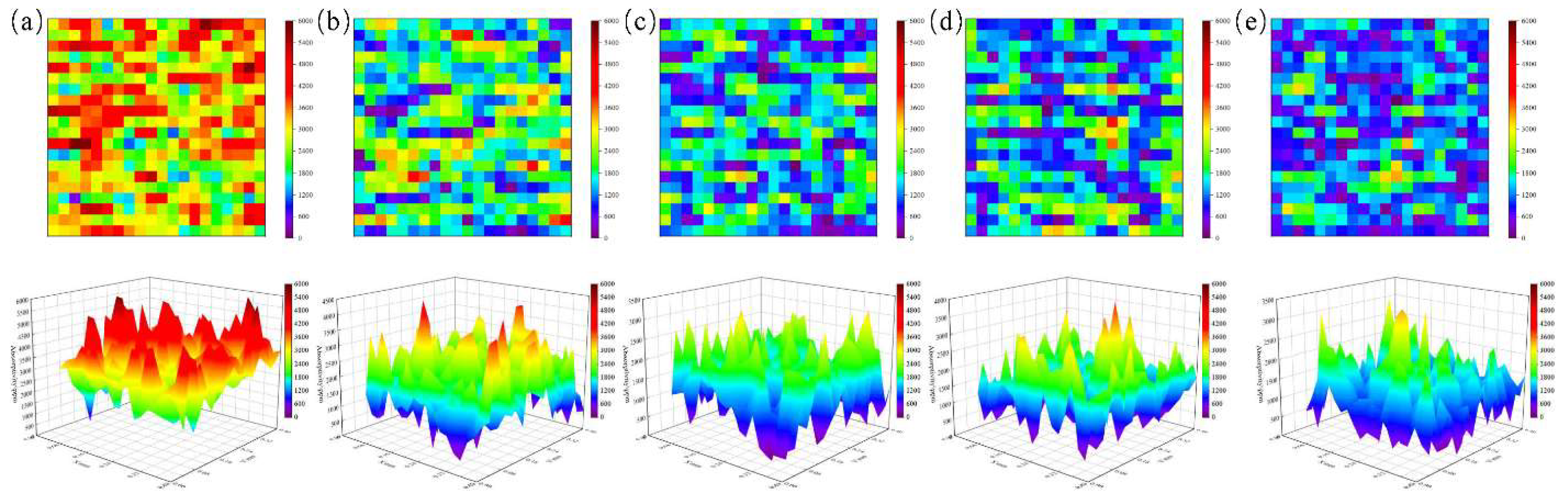
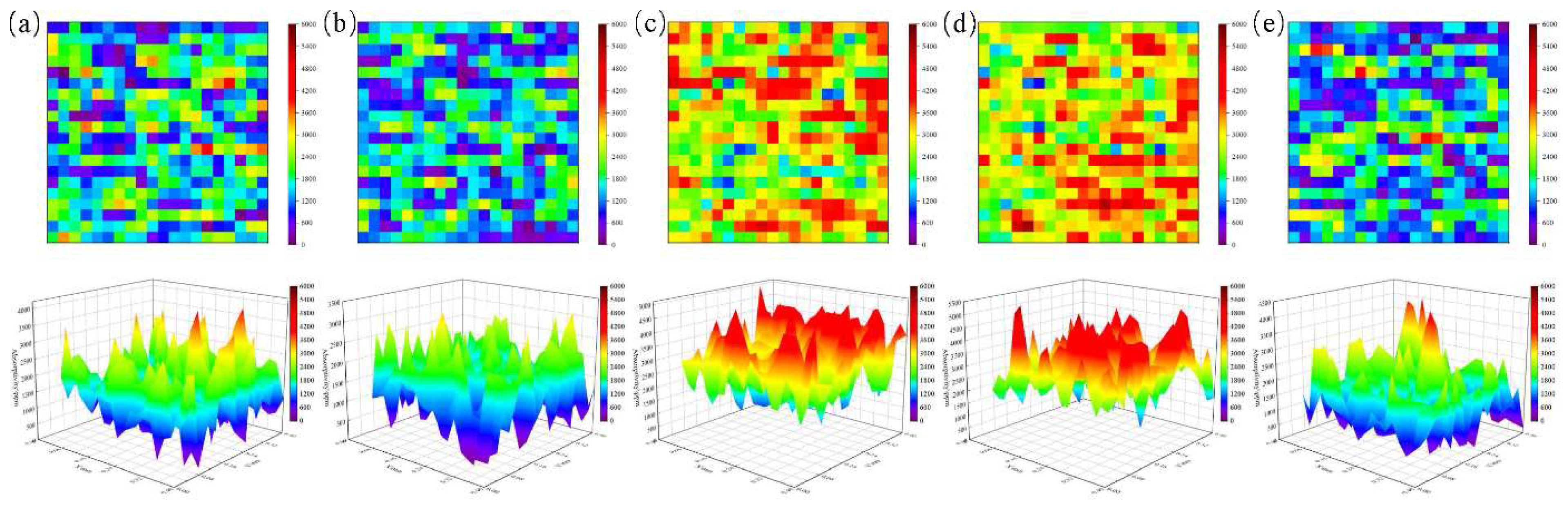
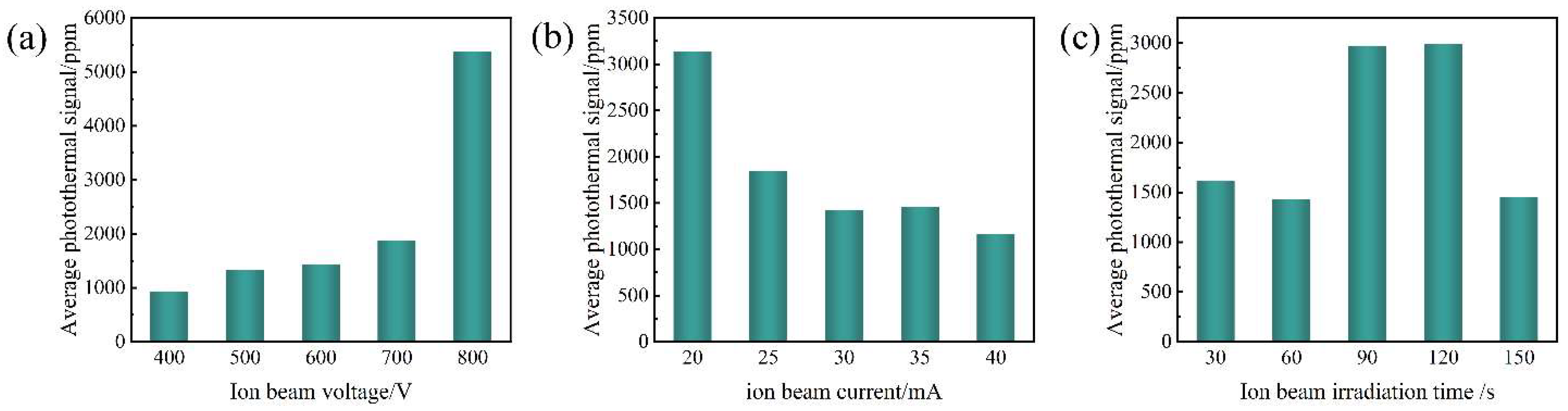
4.1. Influence of Ion Beam Voltage on Photothermal Response
4.2. Effect of Ion Beam Current on Photothermal Signal Suppression
4.3. Non-Monotonic Effect of Irradiation Time on Photothermal Response
4.4. Statistical Analysis and Mechanistic Insights
4.5. Comparison with Literature and Application Prospects
- (1)
- Exploring other ion species (e.g., Kr+, O+) to modulate defect types and densities: Preliminary simulations suggest O+ irradiation may reduce Li vacancies, potentially tuning photothermal response to shorter wavelengths.
- (2)
- Investigating temperature-dependent photothermal responses (20–300 °C): This will simulate practical device operating conditions, with a focus on maintaining signal stability above 100 °C.
- (3)
- Integrating optimized LiNbO3 with waveguides: Targeting 100 GHz modulation speed, we propose waveguides with dimensions 500 nm (width) × 200 nm (thickness), fabricated via focused ion beam milling, to enable on-chip all-optical switches with insertion loss < 3 dB and modulation depth > 20 dB [6].
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hua, Z.; Li, B.; Li, L.; Yin, X.; Chen, K.; Wang, W. Designing a Novel Photothermal Material of Hierarchical Microstructured Copper Phosphate for Solar Evaporation Enhancement. J. Phys. Chem. C 2016, 121, 60–69. [Google Scholar] [CrossRef]
- Li, J.; Zhang, W.; Ji, W.; Wang, J.; Wang, N.; Wu, W.; Wu, Q.; Hou, X.; Hu, W.; Li, L. Near infrared photothermal conversion materials: Mechanism, preparation, and photothermal cancer therapy applications. J. Mater. Chem. B 2021, 9, 7909–7926. [Google Scholar] [CrossRef]
- Liu, F.; Lai, Y.; Zhao, B.; Bradley, R.; Wu, W. Photothermal materials for efficient solar powered steam generation. Front. Chem. Sci. Eng. 2019, 13, 636–653. [Google Scholar] [CrossRef]
- Xie, Z.; Duo, Y.; Lin, Z.; Fan, T.; Xing, C.; Yu, L.; Wang, R.; Qiu, M.; Zhang, Y.; Zhao, Y.; et al. The Rise of 2D Photothermal Materials beyond Graphene for Clean Water Production. Adv. Sci. 2020, 7, 1902236. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.; Li, D.; Salazar, S.L.; Rao, Z.; Arıcı, M.; Wei, W. Photothermal properties and photothermal conversion performance of nano-enhanced paraffin as a phase change thermal energy storage material. Sol. Energy Mater. Sol. Cells 2021, 219, 110792. [Google Scholar] [CrossRef]
- Rashed, A.N.Z.; Zaky, W.F.; El-Hageen, H.M.; Alatwi, A.M. Technical specifications for an all-optical switch for information storage and processing systems. Eur. Phys. J. Plus 2021, 136, 100. [Google Scholar]
- Stabile, R.; Albores-Mejia, A.; Rohit, A.; Williams, K.A. Integrated optical switch matrices for packet data networks. Microsyst. Nanoeng. 2016, 2, 15042. [Google Scholar] [CrossRef] [PubMed]
- Arizmendi, L. Photonic applications of lithium niobate crystals. Phys. Status Solidi (A) 2004, 201, 253–283. [Google Scholar] [CrossRef]
- Li, M.; Ling, J.; He, Y.; Javid, U.A.; Xue, S.; Lin, Q. Lithium niobate photonic-crystal electro-optic modulator. Nat. Commun. 2020, 11, 4123. [Google Scholar] [CrossRef]
- Qi, Y.; Li, Y. Integrated lithium niobate photonics. Nanophotonics 2020, 9, 1287–1320. [Google Scholar] [CrossRef]
- Shur, V.Y.; Akhmatkhanov, A.R.; Baturin, I.S. Micro- and nano-domain engineering in lithium niobate. Appl. Phys. Rev. 2015, 2, 040604. [Google Scholar] [CrossRef]
- Smith, R.T.; Welsh, F.S. Temperature Dependence of the Elastic, Piezoelectric, and Dielectric Constants of Lithium Tantalate and Lithium Niobate. J. Appl. Phys. 1971, 42, 2219–2230. [Google Scholar] [CrossRef]
- Wang, C.; Zhang, M.; Stern, B.; Lipson, M.; Lončar, M. Nanophotonic lithium niobate electro-optic modulators. Opt. Express 2018, 26, 1547–1555. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Feng, H.; Wang, C.; Ren, W. On-chip photothermal gas sensor based on a lithium niobate rib waveguide. Sens. Actuators B Chem. 2024, 405, 135392. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, H.; Li, S. Comparative Study on Photothermal Stability of Lithium Niobate and Silicon under High-Power Laser Irradiation. Opt. Commun. 2024, 550, 129876. [Google Scholar]
- Cao, Y.; Zhang, D.; Yang, Y.; Lin, B.; Lv, J.; Wang, F.; Yang, X.; Yi, Y. Au Nanoparticles-Doped Polymer All-Optical Switches Based on Photothermal Effects. Polymers 2020, 12, 1960. [Google Scholar] [CrossRef]
- Che, Y.; Zhang, T.; Shi, T.; Deng, Z.-L.; Cao, Y.; Guan, B.-O.; Li, X. Ultrasensitive Photothermal Switching with Resonant Silicon Metasurfaces at Visible Bands. Nano Lett. 2023, 24, 576–583. [Google Scholar] [CrossRef]
- Atta, A.; Abdel Reheem, A.M.; Abdeltwab, E. Ion beam irradiation effects on surface morphology and optical properties of ZnO/PVA composites. Surf. Rev. Lett. 2020, 27, 1950214. [Google Scholar] [CrossRef]
- Cuerno, R.; Kim, J.S. A perspective on nanoscale pattern formation at surfaces by ion-beam irradiation. J. Appl. Phys. 2020, 128, 180902. [Google Scholar] [CrossRef]
- McCulloch, D.G.; Prawer, S.; Hoffman, A. Structural investigation of xenon-ion-beam-irradiated glassy carbon. Phys. Rev. B 1994, 50, 5905–5917. [Google Scholar] [CrossRef]
- Taller, S.; Woodley, D.; Getto, E.; Monterrosa, A.M.; Jiao, Z.; Toader, O.; Naab, F.; Kubley, T.; Dwaraknath, S.; Was, G.S. Multiple ion beam irradiation for the study of radiation damage in materials. Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interact. Mater. At. 2017, 412, 1–10. [Google Scholar] [CrossRef]
- Tyagi, C.; Khan, S.A.; Ojha, S.; Avasthi, D.K.; Tripathi, A. Effect of carbon ion-beam irradiation on graphene oxide film. Vacuum 2018, 154, 259–263. [Google Scholar] [CrossRef]
- Xiang, X.; He, Z.; Rao, J.; Fan, Z.; Wang, X.; Chen, Y. Applications of Ion Beam Irradiation in Multifunctional Oxide Thin Films: A Review. ACS Appl. Electron. Mater. 2021, 3, 1031–1042. [Google Scholar] [CrossRef]
- Hazem, R.; Izerrouken, M.; Cheraitia, A.; Djehlane, A. Raman study of ion beam irradiation damage on nanostructured TiO2 thin film. Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interact. Mater. At. 2019, 444, 62–67. [Google Scholar] [CrossRef]
- Borschel, C.; Ronning, C. Ion beam irradiation of nanostructures—A 3D Monte Carlo simulation code. Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interact. Mater. At. 2011, 269, 2133–2138. [Google Scholar] [CrossRef]
- Castro, M.; Gago, R.; Vázquez, L.; Muñoz-García, J.; Cuerno, R. Stress-induced solid flow drives surface nanopatterning of silicon by ion-beam irradiation. Phys. Rev. B 2012, 86, 214107. [Google Scholar] [CrossRef]
- Chen, Z.L.; Liu, W.G.; Yang, L.H. Self-organizing nano-structureand optical properties of sapphire induced by low energy ion beam. Chin. J. Lasers 2015, 42, 0306003. [Google Scholar] [CrossRef]
- Wang, M.J.; Chen, Z.L. Sapphire nanostructure and optical propertyeroded by different ion beam parameters. J. Appl. Opt. 2014, 35, 691–695. [Google Scholar]
- Yang, G.Y.; Cai, M.Q.; Li, J.Y. Preparation of subwavelength nanostructures based on low-energy ion bombardment. Acta Opt. Sin. 2020, 40, 1736001. [Google Scholar] [CrossRef]
- Zhao, Y.; Chen, L. Progress in Ion Beam Modification of Ferroelectric Materials for Photonic Applications. J. Phys. D Appl. Phys. 2023, 56, 243001. [Google Scholar]
- Stuart, B.C.; Feit, M.D.; Rubenchik, A.M.; Shore, B.W.; Perry, M.D. Laser-induced damage in dielectrics with nanosecond to subpicosecond pulses. Phys. Rev. Lett. 1995, 74, 2248. [Google Scholar] [CrossRef]
- Bass, M.; Barrett, H. Avalanche breakdown and the probabilistic nature of laser-induced damage. IEEE J. Quantum Electron. 1972, 8, 338–343. [Google Scholar] [CrossRef]
- Bloembergen, N. Laser-induced electric breakdown in solids. IEEE J. Quantum Electron. 1974, 10, 375–386. [Google Scholar] [CrossRef]
- Thornber, K.K. Applications of scaling to problems in high-field electronic transport. J. Appl. Phys. 1981, 52, 279–290. [Google Scholar] [CrossRef]
- Kennedy, P.K. A first-order model for computation of laser-induced breakdown thresholds in ocular and aqueous media. I. Theory. IEEE J. Quantum Electron. 1995, 31, 2241–2249. [Google Scholar] [CrossRef]
- Kennedy, P.K.; Boppart, S.A.; Hammer, D.X.; Rockwell, B.A.; Noojin, G.D.; Roach, W.P. A first-order model for computation of laser-induced breakdown thresholds in ocular and aqueous media. II. Comparison to experiment. IEEE J. Quantum Electron. 1995, 31, 2250–2257. [Google Scholar] [CrossRef]
- El-Sapa, S.; Ghazouani, N.; Becheikh, N. Effects of changing thermal conductivity on photothermal excitation in non-local semiconductor material subjected to moisture diffusion and laser pulses. J. Appl. Phys. 2023, 133, 245108. [Google Scholar] [CrossRef]
- Schelling, P.K.; Phillpot, S.R. Mechanism of thermal transport in zirconia and yttria-stabilized zirconia by molecular-dynamics simulation. J. Am. Ceram. Soc. 2001, 84, 2997–3007. [Google Scholar] [CrossRef]
- Wang, Y.; Qiu, B.; Ruan, X. Edge effect on thermal transport in graphene nanoribbons: A phonon localization mechanism beyond edge roughness scattering. Appl. Phys. Lett. 2012, 101, 013101. [Google Scholar] [CrossRef]
- Klemens, P.G. The scattering of low-frequency lattice waves by static imperfections. Proc. Phys. Soc. Sect. A 1955, 68, 1113. [Google Scholar] [CrossRef]
- Pauling, L. Atomic radii and interatomic distances in metals. J. Am. Chem. Soc. 1947, 69, 542–553. [Google Scholar] [CrossRef]
- Sun, C.Q. Size dependence of nanostructures: Impact of bond order deficiency. Prog. Solid State Chem. 2007, 35, 1–159. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, X.; Bo, M.; Zhang, X.; Liu, X.; Sun, C.Q.; Huang, Y. Number-of-layer, pressure, and temperature resolved bond–phonon–photon cooperative relaxation of layered black phosphorus. J. Raman Spectrosc. 2016, 47, 1304–1309. [Google Scholar] [CrossRef]
- Bai, X.; Shuai, Y.; Gong, C.G.; Wu, C.G. Surface Modifications of Crystal-Ion-Sliced LiNbO3 Thin Films by Low Energy Ion Irradiations. Appl. Surf. Sci. 2017, 434, 87–94. [Google Scholar] [CrossRef]

| Source | df | Sum of Squares | Mean Square | F | p |
|---|---|---|---|---|---|
| Voltage | 4 | 1.23 × 108 | 3.08 × 107 | 28.7 | <0.001 |
| Current | 4 | 8.75 × 107 | 2.19 × 107 | 20.3 | <0.01 |
| Irradiation Time | 4 | 5.42 × 107 | 1.36 × 107 | 12.6 | <0.05 |
| Error | 36 | 3.92 × 107 | 1.09 × 106 | – | – |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, J.; Lin, D.; Peng, X.; Wu, Y.; Li, J.; Hu, Z.; He, Z.; Wang, J.; Tan, Y.; Xu, X.; et al. Photothermal Performance Testing of Lithium Niobate After Ion Beam Radiation. Photonics 2025, 12, 793. https://doi.org/10.3390/photonics12080793
Liu J, Lin D, Peng X, Wu Y, Li J, Hu Z, He Z, Wang J, Tan Y, Xu X, et al. Photothermal Performance Testing of Lithium Niobate After Ion Beam Radiation. Photonics. 2025; 12(8):793. https://doi.org/10.3390/photonics12080793
Chicago/Turabian StyleLiu, Junyi, Daiyong Lin, Xing Peng, Yao Wu, Jian Li, Ziqiang Hu, Zhixuan He, Jiaqi Wang, Yuxia Tan, Xiaoshu Xu, and et al. 2025. "Photothermal Performance Testing of Lithium Niobate After Ion Beam Radiation" Photonics 12, no. 8: 793. https://doi.org/10.3390/photonics12080793
APA StyleLiu, J., Lin, D., Peng, X., Wu, Y., Li, J., Hu, Z., He, Z., Wang, J., Tan, Y., Xu, X., & Qiao, S. (2025). Photothermal Performance Testing of Lithium Niobate After Ion Beam Radiation. Photonics, 12(8), 793. https://doi.org/10.3390/photonics12080793







