Advanced Microsamples: Current Applications and Considerations for Mass Spectrometry-Based Metabolic Phenotyping Pipelines
Abstract
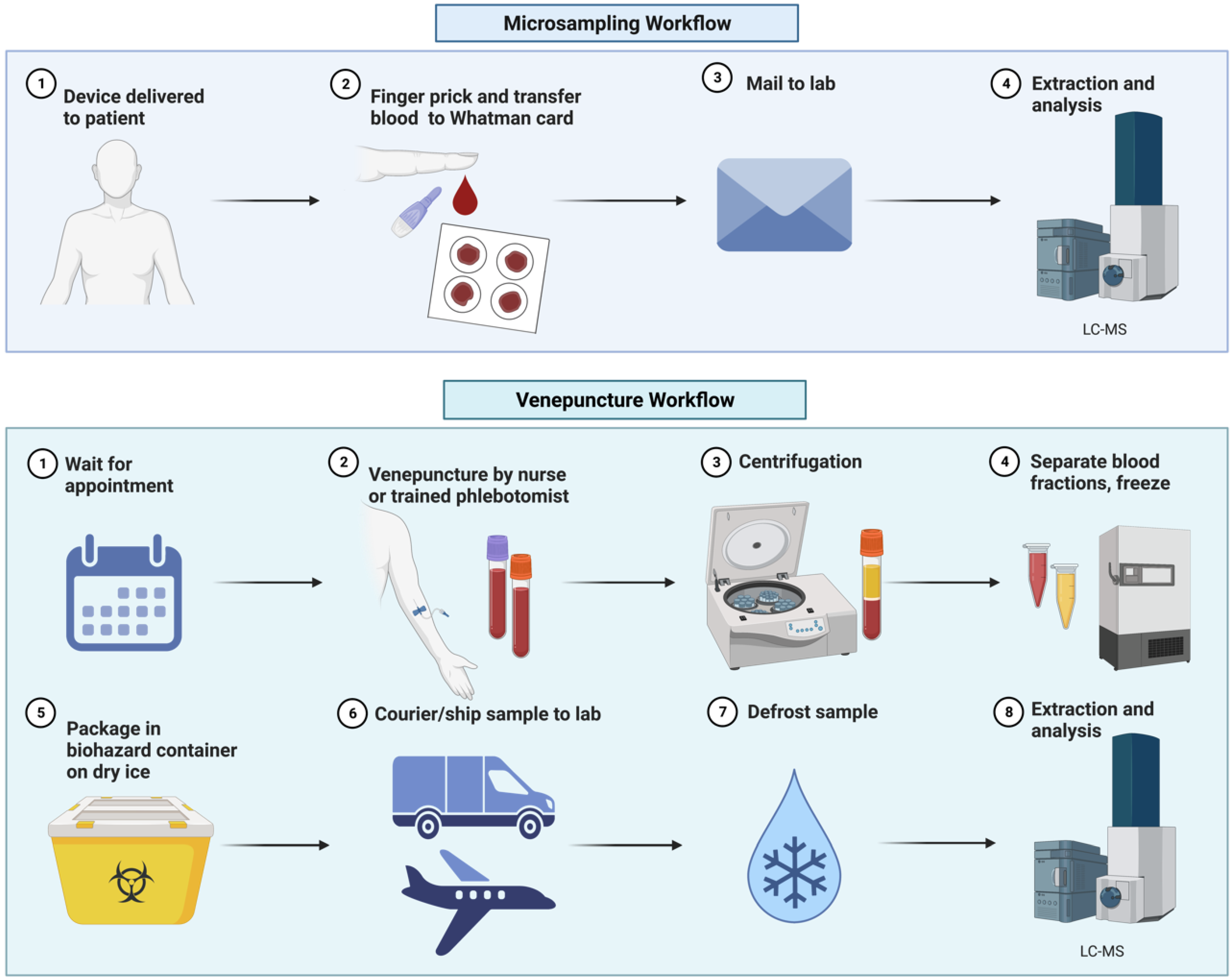
:1. Introduction
2. Approaches to Microsample Collections
2.1. Traditional Dried Samples: Dry Blood Spots (DBSs)
2.2. Improving Microsample Collection: Are Advanced Devices the Future for Metabolic Phenotyping?
3. Advanced Microsamples: Current Applications in Metabolic Phenotyping
3.1. Advanced Dried Microsamples
3.1.1. Metabolite Coverage and Stability of Different Advanced Dried Microsample Devices
3.1.2. Analysis of Haematocrit
3.1.3. Summary of Advanced Dried Microsample Findings
3.2. Passive Separation Devices
3.3. Whole Biofluid Collectors
4. Considerations for Future Application of Advanced Microsampling in Metabolic Phenotyping Workflows
4.1. Microsample Collection and Stability
4.2. Microsample Preparation
4.3. Determining an Equivalent Concentration Factor
4.4. Self-Sampling
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 25OHD | 25-hydroxyvitamin D |
| ACN | Acetonitrile |
| CV | Coefficient of variation |
| DBS | Dried blood spot |
| DPS | Dried plasma spot |
| DSS | Dried serum spot |
| DUS | Dried urine spot |
| EDTA | Ethylenediaminetetraacetic |
| GC–MS | Gas chromatography–mass spectrometry |
| h | Hours |
| hct | Haematocrit |
| LC–MS | Liquid chromatography–mass spectrometry |
| MeOH | Methanol |
| mL | Millilitre |
| MS | Mass spectrometry |
| NIR | Near-infrared |
| P4 medicine | Predictive, preventive, personalised, and participatory medicine |
| QC | Quality control |
| qDBS | Quantitative dried blood spot |
| TAP | Touch-activated phlebotomy |
| UHPLC–MS | Ultra-high-performance liquid chromatography–mass spectrometry |
| VAMS | Volumetric absorptive microsampling |
| WADA | World Anti-Doping Agency |
| WB | Whole blood |
| µL | Microlitre |
References
- Flores, M.; Glusman, G.; Brogaard, K.; Price, N.D.; Hood, L. P4 medicine: How systems medicine will transform the healthcare sector and society. Pers. Med. 2013, 10, 565–576. [Google Scholar] [CrossRef] [Green Version]
- Gray, N.; Lawler, N.G.; Yang, R.; Morillon, A.-C.; Gay, M.C.L.; Bong, S.-H.; Holmes, E.; Nicholson, J.K.; Whiley, L. A simultaneous exploratory and quantitative amino acid and biogenic amine metabolic profiling platform for rapid disease phenotyping via UPLC-QToF-MS. Talanta 2021, 223, 121872. [Google Scholar] [CrossRef]
- Nicholson, J.K.; Holmes, E.; Kinross, J.M.; Darzi, A.W.; Takats, Z.; Lindon, J.C. Metabolic phenotyping in clinical and surgical environments. Nature 2012, 491, 384–392. [Google Scholar] [CrossRef]
- Rainville, P.D.; Theodoridis, G.; Plumb, R.S.; Wilson, I.D. Advances in liquid chromatography coupled to mass spectrometry for metabolic phenotyping. TrAC Trends Anal. Chem. 2014, 61, 181–191. [Google Scholar] [CrossRef]
- Holen, T.; Norheim, F.; Gundersen, T.E.; Mitry, P.; Linseisen, J.; Iversen, P.O.; Drevon, C.A. Biomarkers for nutrient intake with focus on alternative sampling techniques. Genes Nutr. 2016, 11, 12. [Google Scholar] [CrossRef] [Green Version]
- Evans, C.A.; Bowen, C.L.; Filali-Ansary, A. Dried blood spots: Challenges. Future Sci. Group (FSG) 2013, 1, 30–46. [Google Scholar] [CrossRef]
- Bowen, C.L.; Licea-Perez, H.; Karlinsey, M.Z.; Jurusik, K.; Pierre, E.; Siple, J.; Kenney, J.; Stokes, A.; Spooner, N.; Evans, C.A. A novel approach to capillary plasma microsampling for quantitative bioanalysis. Bioanalysis 2013, 5, 1131–1135. [Google Scholar] [CrossRef]
- Bowen, C.L.; Dopson, W.; Kemp, D.C.; Lewis, M.; Lad, R.; Overvold, C. Investigations into the environmental conditions experienced during ambient sample transport: Impact to dried blood spot sample shipments. Bioanalysis 2011, 3, 1625–1633. [Google Scholar] [CrossRef]
- Thomas, S.L.; Wakerman, J.; Humphreys, J.S. Ensuring equity of access to primary health care in rural and remote Australia-what core services should be locally available? Int. J. Equity Health 2015, 14, 111. [Google Scholar] [CrossRef] [Green Version]
- Xing, J.; Loureiro, J.; Patel, M.T.; Mikhailov, D.; Gusev, A.I. Evaluation of a novel blood microsampling device for clinical trial sample collection and protein biomarker analysis. Bioanalysis 2020, 12, 919–935. [Google Scholar] [CrossRef]
- Australian Institute of Health and Welfare (AIHW). Rural & Remote Health; AIHW: Canberra, Australia, 2019.
- Ahmetaj-Shala, B.; Olanipekun, M.; Tesfai, A.; Maccallum, N.; Kirkby, N.; Quinlan, G.J.; Shih, C.-C.; Kawai, R.; Mumby, S.; Paul-Clark, M.; et al. Development of a novel UHPLC–MS/MS-based platform to quantify amines, amino acids and methylarginines for applications in human disease phenotyping. Sci. Rep. 2018, 8, 13987. [Google Scholar] [CrossRef]
- Want, E.J.; Wilson, I.; Gika, H.G.; Theodoridis, G.; Plumb, R.S.; Shockcor, J.P.; Holmes, E.; Nicholson, J. Global metabolic profiling procedures for urine using UPLC–MS. Nat. Protoc. 2010, 5, 1005–1018. [Google Scholar] [CrossRef]
- Want, E.J. LC–MS Untargeted Analysis. In Metabolic Profiling: Methods and Protocols; Theodoridis, G.A., Gika, H.G., Wilson, I.D., Eds.; Springer: New York, NY, USA, 2018; pp. 9–116. [Google Scholar]
- Contrepois, K.; Mahmoudi, S.; Ubhi, B.K.; Papsdorf, K.; Hornburg, D.; Brunet, A.; Snyder, M. Cross-Platform Comparison of Untargeted and Targeted Lipidomics Approaches on Aging Mouse Plasma. Sci. Rep. 2018, 8, 1–9. [Google Scholar] [CrossRef]
- Climaco Pinto, R.; Karaman, I.; Lewis, M.R.; Hällqvist, J.; Kaluarachchi, M.; Graça, G.; Chekmeneva, E.; Durainayagam, B.; Ghanbari, M.; Ikram, M.A.; et al. Finding Correspondence between Metabolomic Features in Untargeted Liquid Chromatography–Mass Spectrometry Metabolomics Datasets. Anal. Chem. 2022, 94, 5493–5503. [Google Scholar] [CrossRef]
- Whiley, L.; Chappell, K.E.; D’Hondt, E.; Lewis, M.R.; Jiménez, B.; Snowden, S.G.; Soininen, H.; Kłoszewska, I.; Mecocci, P.; Tsolaki, M.; et al. Metabolic phenotyping reveals a reduction in the bioavailability of serotonin and kynurenine pathway metabolites in both the urine and serum of individuals living with Alzheimer’s disease. Alzheimer’s Res. Ther. 2021, 13, 20. [Google Scholar] [CrossRef]
- Sands, C.J.; Gómez-Romero, M.; Correia, G.; Chekmeneva, E.; Camuzeaux, S.; Izzi-Engbeaya, C.; Dhillo, W.S.; Takats, Z.; Lewis, M.R. Representing the Metabolome with High Fidelity: Range and Response as Quality Control Factors in LC–MS-Based Global Profiling. Anal. Chem. 2021, 93, 1924–1933. [Google Scholar] [CrossRef]
- Letertre, M.P.; Myridakis, A.; Whiley, L.; Camuzeaux, S.; Lewis, M.R.; Chappell, K.E.; Thaikkatil, A.; Dumas, M.-E.; Nicholson, J.K.; Swann, J.R.; et al. A targeted ultra performance liquid chromatography–Tandem mass spectrometric assay for tyrosine and metabolites in urine and plasma: Application to the effects of antibiotics on mice. J. Chromatogr. B 2020, 1164, 122511. [Google Scholar] [CrossRef]
- Whiley, L.; Nye, L.C.; Grant, I.; Andreas, N.J.; Chappell, K.E.; Sarafian, M.H.; Misra, R.; Plumb, R.S.; Lewis, M.R.; Nicholson, J.K.; et al. Ultrahigh-Performance Liquid Chromatography Tandem Mass Spectrometry with Electrospray Ionization Quantification of Tryptophan Metabolites and Markers of Gut Health in Serum and Plasma—Application to Clinical and Epidemiology Cohorts. Anal. Chem. 2019, 91, 5207–5216. [Google Scholar] [CrossRef] [Green Version]
- Liu, L.; Jin, X.; Wu, Y.; Yang, M.; Xu, T.; Li, X.; Ren, J.; Yan, L.L. A Novel Dried Blood Spot Detection Strategy for Characterizing Cardiovascular Diseases. Front. Cardiovasc. Med. 2020, 7, 542519. [Google Scholar] [CrossRef]
- Brignardello, J.; Holmes, E.; Garcia-Perez, I. Chapter Seven-Metabolic Phenotyping of Diet and Dietary Intake. In Advances in Food and Nutrition Research; Toldrá, F., Ed.; Academic Press: Cambridge, MA, USA, 2017; pp. 231–270. [Google Scholar]
- Morgan, P.E. Microsampling Devices for Routine Therapeutic Drug Monitoring-Are We There Yet? Ther. Drug Monit. 2021, 43, 322–334. [Google Scholar] [CrossRef]
- Constantinou, M.; Papakonstantinou, E.; Benaki, D.; Spraul, M.; Shulpis, K.; Koupparis, M.; Mikros, E. Application of nuclear magnetic resonance spectroscopy combined with principal component analysis in detecting inborn errors of metabolism using blood spots: A metabonomic approach. Anal. Chim. Acta 2004, 511, 303–312. [Google Scholar] [CrossRef]
- Mingas, P.-D.; Zdovc, J.; Grabnar, I.; Vovk, T. The Evolving Role of Microsampling in Therapeutic Drug Monitoring of Monoclonal Antibodies in Inflammatory Diseases. Molecules 2021, 26, 1787. [Google Scholar] [CrossRef]
- Beck, O.; Mellring, M.; Löwbeer, C.; Seferaj, S.; Helander, A. Measurement of the alcohol biomarker phosphatidylethanol (PEth) in dried blood spots and venous blood—Importance of inhibition of post-sampling formation from ethanol. Anal. Bioanal. Chem. 2021, 413, 5601–5606. [Google Scholar] [CrossRef]
- Deprez, S.; Paniagua-González, L.; Velghe, S.; Stove, C.P. Evaluation of the Performance and Hematocrit Independence of the HemaPEN as a Volumetric Dried Blood Spot Collection Device. Anal. Chem. 2019, 91, 14467–14475. [Google Scholar] [CrossRef] [Green Version]
- Kocher, S.; Tshiananga, J.K.T.; Koubek, R. Comparison of Lancing Devices for Self-Monitoring of Blood Glucose regarding Lancing Pain. J. Diabetes Sci. Technol. 2009, 3, 1136–1143. [Google Scholar] [CrossRef]
- Jacob, M.; Malkawi, A.; Albast, N.; Al Bougha, S.; Lopata, A.; Dasouki, M.; Rahman, A.M.A. A targeted metabolomics approach for clinical diagnosis of inborn errors of metabolism. Anal. Chim. Acta 2018, 1025, 141–153. [Google Scholar] [CrossRef]
- Delahaye, L.; Veenhof, H.; Koch, B.; Alffenaar, J.; Linden, R.; Stove, C. Alternative Sampling Devices to Collect Dried Blood Microsamples: State-of-the-Art. Ther. Drug Monit. 2021, 43, 310–321. [Google Scholar] [CrossRef]
- Tasso, Inc. Tasso+. 2022. Available online: https://www.tassoinc.com/tasso-plus (accessed on 10 March 2022).
- Shimadzu. MSW² Type Udck. 2022. Available online: https://www.shimadzu.eu/msw2 (accessed on 10 March 2022).
- Telimmune. Telimmune Plasma Separation Cards (Formerly Noviplex). 2022. Available online: https://www.telimmune.com/plasma-separation-cards (accessed on 10 March 2022).
- Spot On Sciences. HEMASPOT SE. 2022. Available online: https://www.spotonsciences.com/hemaspot-se/ (accessed on 10 March 2022).
- Protti, M.; Marasca, C.; Cirrincione, M.; Sberna, A.E.; Mandrioli, R.; Mercolini, L. Dried Urine Microsampling Coupled to Liquid Chromatography—Tandem Mass Spectrometry (LC–MS/MS) for the Analysis of Unconjugated Anabolic Androgenic Steroids. Molecules 2020, 25, 3210. [Google Scholar] [CrossRef]
- Salamin, O.; Nicoli, R.; Xu, C.; Boccard, J.; Rudaz, S.; Pitteloud, N.; Saugy, M.; Kuuranne, T. Steroid profiling by UHPLC–MS/MS in dried blood spots collected from healthy women with and without testosterone gel administration. J. Pharm. Biomed. Anal. 2021, 204, 114280. [Google Scholar] [CrossRef]
- Hemmati, M.; Nix, C.; Crommen, J.; Servais, A.-C.; Fillet, M. Benefits of microsampling and microextraction for metabolomics studies. TrAC Trends Anal. Chem. 2020, 127, 115899. [Google Scholar] [CrossRef]
- Bictash, M.; Ebbels, T.M.; Chan, Q.; Loo, R.L.; Yap, I.K.; Brown, I.J.; de Iorio, M.; Daviglus, M.L.; Holmes, E.; Stamler, J.; et al. Opening up the "Black Box": Metabolic phenotyping and metabolome-wide association studies in epidemiology. J. Clin. Epidemiol. 2010, 63, 970–979. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carpentieri, D.; Colvard, A.; Petersen, J.; Marsh, W.; David-Dirgo, V.; Huentelman, M.; Pirrotte, P.; Sivakumaran, T. Mind the Quality Gap When Banking on Dry Blood Spots. Biopreservation Biobanking 2021, 19, 136–142. [Google Scholar] [CrossRef] [PubMed]
- Grüner, N.; Stambouli, O.; Ross, R.S. Dried Blood Spots-Preparing and Processing for Use in Immunoassays and in Molecular Techniques. J. Vis. Exp. 2015, 97, e52619. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lehmann, S.; Delaby, C.; Vialaret, J.; Ducos, J.; Hirtz, C. Current And future use of “dried blood spot” analyses in clinical chemistry. Clin. Chem. Lab. Med. CCLM 2013, 51, 1897–1909. [Google Scholar] [CrossRef]
- Cao, Y.; Wang, Q.; Gao, P.; Dong, J.; Zhu, Z.; Fang, Y.; Fang, Z.; Sun, X.; Sun, T. A dried blood spot mass spectrometry metabolomic approach for rapid breast cancer detection. Onco Targets Ther. 2016, 9, 1389–1398. [Google Scholar] [CrossRef] [Green Version]
- Hasanah, Y.I.F.; Harahap, Y.; Purwanto, D.J. Phenotyping Study of Cyclophosphamide 4-Hydroxylation in Malay Cancer Patients. Drug Des. Dev. Ther. 2021, 15, 305–313. [Google Scholar] [CrossRef]
- Loo, R.L.; Lu, Q.; Carter, E.M.; Liu, S.; Clark, S.; Wang, Y.; Baumgartner, J.; Tang, H.; Chan, Q. A feasibility study of metabolic phenotyping of dried blood spot specimens in rural Chinese women exposed to household air pollution. J. Expo. Sci. Environ. Epidemiol. 2020, 31, 328–344. [Google Scholar] [CrossRef]
- Lang, W.; Qi, J.; Caldwell, G.W. Drug, Lipid, and Acylcarnitine Profiling Using Dried Blood Spot (DBS) Technology in Drug Discovery. In Optimization in Drug Discovery; Humana Press: Totowa, NJ, USA, 2013; pp. 461–475. [Google Scholar] [CrossRef]
- Haijes, H.A.; Jans, J.; Van Der Ham, M.; Van Hasselt, P.M.; Verhoeven-Duif, N.M. Understanding acute metabolic decompensation in propionic and methylmalonic acidemias: A deep metabolic phenotyping approach. Orphanet J. Rare Dis. 2020, 15, 68. [Google Scholar] [CrossRef]
- Van Dooijeweert, B.; Broeks, M.H.; Verhoeven-Duif, N.M.; Van Beers, E.J.; Nieuwenhuis, E.E.S.; Van Solinge, W.W.; Bartels, M.; Jans, J.J.M.; van Wijk, R. Untargeted metabolic profiling in dried blood spots identifies disease fingerprint for pyruvate kinase deficiency. Haematologica 2020, 106, 2720. [Google Scholar] [CrossRef]
- O’Mara, M.; Hudson-Curtis, B.; Olson, K.; Yueh, Y.; Dunn, J.; Spooner, N. The effect of hematocrit and punch location on assay bias during quantitative bioanalysis of dried blood spot samples. Bioanalysis 2011, 3, 2335–2347. [Google Scholar] [CrossRef]
- Watson, P.; Maughan, R.J. Artifacts in Plasma Volume Changes due to Hematology Analyzer-Derived Hematocrit. Med. Sci. Sports Exerc. 2014, 46, 52–59. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kundrapu, S.; Noguez, J. Chapter Six-Laboratory Assessment of Anemia. In Advances in Clinical Chemistry; Makowski, G.S., Ed.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 197–225. [Google Scholar]
- Fischbach, F.T.; Fischbach, M.A. Fischbach’s Manual of Laboratory and Diagnostic Tests, 10th ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2018. [Google Scholar]
- Hematocrit. Nursing Critical Care. 2020, 15, 38. [CrossRef]
- Cvetko, A.; Tijardović, M.; Bilandžija-Kuš, I.; Gornik, O. Comparison of self-sampling blood collection for N-glycosylation analysis. BMC Res. Notes 2022, 15, 61. [Google Scholar] [CrossRef] [PubMed]
- Petrick, L.; Edmands, W.; Schiffman, C.; Grigoryan, H.; Perttula, K.; Yano, Y.; Dudoit, S.; Whitehead, T.; Metayer, C.; Rappaport, S. An untargeted metabolomics method for archived newborn dried blood spots in epidemiologic studies. Metabolomics 2017, 13, 27. [Google Scholar] [CrossRef] [Green Version]
- Palmer, E.A.; Cooper, H.J.; Dunn, W.B. Investigation of the 12-Month Stability of Dried Blood and Urine Spots Applying Untargeted UHPLC–MS Metabolomic Assays. Anal. Chem. 2019, 91, 14306–14313. [Google Scholar] [CrossRef]
- Yu, M.; Dolios, G.; Yong-Gonzalez, V.; Björkqvist, O.; Colicino, E.; Halfvarson, J.; Petrick, L. Untargeted metabolomics profiling and hemoglobin normalization for archived newborn dried blood spots from a refrigerated biorepository. J. Pharm. Biomed. Anal. 2020, 191, 113574. [Google Scholar] [CrossRef]
- Baillargeon, K.R.; Brooks, J.C.; Miljanic, P.R.; Mace, C.R. Patterned Dried Blood Spot Cards for the Improved Sampling of Whole Blood. ACS Meas. Sci. Au 2022, 2, 31–38. [Google Scholar] [CrossRef]
- Kong, S.T.; Lin, H.-S.; Ching, J.; Ho, P.C. Evaluation of Dried Blood Spots as Sample Matrix for Gas Chromatography/Mass Spectrometry Based Metabolomic Profiling. Anal. Chem. 2011, 83, 4314–4318. [Google Scholar] [CrossRef]
- Michopoulos, F.; Theodoridis, G.; Smith, C.J.; Wilson, I.D. Metabolite profiles from dried blood spots for metabonomic studies using UPLC combined with orthogonal acceleration ToF-MS: Effects of different papers and sample storage stability. Bioanalysis 2011, 3, 2757–2767. [Google Scholar] [CrossRef]
- Ward, C.; Nallamshetty, S.; Watrous, J.D.; Acres, E.; Long, T.; Mathews, I.T.; Sharma, S.; Cheng, S.; Imam, F.; Jain, M. Nontargeted mass spectrometry of dried blood spots for interrogation of the human circulating metabolome. Biol. Mass Spectrom. 2021, 56, e4772. [Google Scholar] [CrossRef]
- Koulman, A.; Prentice, P.; Wong, M.C.; Matthews, L.; Bond, N.J.; Eiden, M.; Griffin, J.; Dunger, D. The development and validation of a fast and robust dried blood spot based lipid profiling method to study infant metabolism. Metabolomics 2020, 10, 1018–1025. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, K.; Naviaux, J.C.; Monk, J.M.; Wang, L.; Naviaux, R.K. Improved Dried Blood Spot-Based Metabolomics: A Targeted, Broad-Spectrum, Single-Injection Method. Metabolites 2020, 10, 82. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matsuda, K.C.; Hall, C.; Baker-Lee, C.; Soto, M.; Scott, G.; Prince, P.J.; Retter, M.W. Measurement of in vivo therapeutic mAb concentrations: Comparison of conventional serum/plasma collection and analysis to dried blood spot sampling. Bioanalysis 2013, 5, 1979–1990. [Google Scholar] [CrossRef] [PubMed]
- Stevens, V.L.; Hoover, E.; Wang, Y.; Zanetti, K.A. Pre-Analytical Factors that Affect Metabolite Stability in Human Urine, Plasma, and Serum: A Review. Metabolites 2019, 9, 156. [Google Scholar] [CrossRef] [Green Version]
- Yin, P.; Lehmann, R.; Xu, G. Effects of pre-analytical processes on blood samples used in metabolomics studies. Anal. Bioanal. Chem. 2015, 407, 4879–4892. [Google Scholar] [CrossRef] [Green Version]
- Mandrioli, R.; Mercolini, L.; Protti, M. Blood and Plasma Volumetric Absorptive Microsampling (VAMS) Coupled to LC–MS/MS for the Forensic Assessment of Cocaine Consumption. Molecules 2020, 25, 1046. [Google Scholar] [CrossRef] [Green Version]
- Jacobs, C.M.; Wagmann, L.; Meyer, M.R. Development, validation, and application of a quantitative volumetric absorptive microsampling–based method in finger prick blood by means of LC-HRMS/MS applicable for adherence monitoring of antipsychotics. Anal. Bioanal. Chem. 2021, 413, 1729–1737. [Google Scholar] [CrossRef]
- Protti, M.; Mandrioli, R.; Mercolini, L. Tutorial: Volumetric absorptive microsampling (VAMS). Anal. Chim. Acta 2018, 1046, 32–47. [Google Scholar] [CrossRef]
- Gao, X.; Chen, C.; Geng, D.; Bateman, K.P.; Shi, S.; Woolf, E.J.; Xu, Y. Volumetric absorptive microsampling (VAMS®) in therapeutic protein quantification by LC–MS/MS: Investigation of anticoagulant impact on assay performance and recommendations for best practices in method development. J. Pharm. Biomed. Anal. 2021, 196, 113895. [Google Scholar] [CrossRef]
- Denniff, P.; Spooner, N. Volumetric Absorptive Microsampling: A Dried Sample Collection Technique for Quantitative Bioanalysis. Anal. Chem. 2014, 86, 8489–8495. [Google Scholar] [CrossRef]
- Kok, M.G.; Fillet, M. Volumetric absorptive microsampling: Current advances and applications. J. Pharm. Biomed. Anal. 2018, 147, 288–296. [Google Scholar] [CrossRef] [PubMed]
- Nix, C.; Hemmati, M.; Cobraiville, G.; Servais, A.-C.; Fillet, M. Blood Microsampling to Monitor Metabolic Profiles During Physical Exercise. Front. Mol. Biosci. 2021, 8, 681400. [Google Scholar] [CrossRef] [PubMed]
- Velghe, S.; Stove, C.P. Evaluation of the Capitainer-B Microfluidic Device as a New Hematocrit-Independent Alternative for Dried Blood Spot Collection. Anal. Chem. 2018, 90, 12893–12899. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hauser, J.; Lenk, G.; Ullah, S.; Beck, O.; Stemme, G.; Roxhed, N. An Autonomous Microfluidic Device for Generating Volume-Defined Dried Plasma Spots. Anal. Chem. 2019, 91, 7125–7130. [Google Scholar] [CrossRef] [PubMed]
- Ryona, I.; Henion, J. A Book-Type Dried Plasma Spot Card for Automated Flow-Through Elution Coupled with Online SPE-LC–MS/MS Bioanalysis of Opioids and Stimulants in blood. Anal. Chem. 2016, 88, 11229–11237. [Google Scholar] [CrossRef]
- Forchelet, D.; Béguin, S.; Sajic, T.; Bararpour, N.; Pataky, Z.; Frias, M.; Grabherr, S.; Augsburger, M.; Liu, Y.; Charnley, M.; et al. Separation of blood microsamples by exploiting sedimentation at the microscale. Sci. Rep. 2018, 8, 14101. [Google Scholar] [CrossRef]
- Roadcap, B.; Hussain, A.; Dreyer, D.; Carter, K.; Dube, N.; Xu, Y.; Anderson, M.; Berthier, E.; Vazvaei, F.; Bateman, K.; et al. Clinical application of volumetric absorptive microsampling to the gefapixant development program. Bioanalysis 2020, 12, 893–904. [Google Scholar] [CrossRef]
- Hotta, K.; Ishida, T.; Noritake, K.-I.; Kita, K.; Mano, Y. Quantitative and qualitative application of a novel capillary microsampling device, Microsampling Wing™ (MSW), using antiepileptic drugs in rats. J. Pharm. Biomed. Anal. 2020, 194, 113788. [Google Scholar] [CrossRef]
- Protti, M.; Marasca, C.; Cirrincione, M.; Cavalli, A.; Mandrioli, R.; Mercolini, L. Assessment of capillary volumetric blood microsampling for the analysis of central nervous system drugs and metabolites. Analyst 2020, 145, 5744–5753. [Google Scholar] [CrossRef]
- Wilson, I. Global metabolic profiling (metabonomics/metabolomics) using dried blood spots: Advantages and pitfalls. Bioanalysis 2011, 3, 2255–2257. [Google Scholar] [CrossRef]
- Amsterdam, P.V.; Waldrop, C. The application of dried blood spot sampling in global clinical trials. Bioanalysis 2010, 2, 1783–1786. [Google Scholar] [CrossRef] [Green Version]
- Drolet, J.; Tolstikov, V.; Williams, B.A.; Greenwood, B.P.; Hill, C.; Vishnudas, V.K.; Sarangarajan, R.; Narain, N.R.; Kiebish, M.A. Integrated Metabolomics Assessment of Human Dried Blood Spots and Urine Strips. Metabolites 2017, 7, 35. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cytiva. Whatman 903 Proteinsaver Card. 2022. Available online: https://www.cytivalifesciences.com/en/us/shop/whatman-laboratory-filtration/whatman-dx-components/blood-collection-cards-and-accessories/903-proteinsaver-card-p-01011 (accessed on 10 March 2022).
- PerkinElmer Inc. PerkinElmer 226 Spot Saver RUO Card. 2022. Available online: https://www.perkinelmer.com/uk/product/pki-ruo-spot-saver-card-pack-of-100-gr2261005 (accessed on 10 March 2022).
- Ahlstrom-Munksjö. Biosamples Collection Cards. 2022. Available online: https://www.ahlstrom-munksjo.com/products/medical-life-sciences-and-laboratory/specimen-collection-cards/Biosamples-collection-cards/ (accessed on 10 March 2022).
- Tasso, Inc. Tasso-M20. 2022. Available online: https://www.tassoinc.com/tasso-m20 (accessed on 10 March 2022).
- Capitainer. Capitainer®B Vanadate. 2022. Available online: https://capitainer.se/capitainer-b-vanadate/ (accessed on 10 March 2022).
- Capitainer. Capitainer®qDBS. 2022. Available online: https://capitainer.se/capitainer-qdbs/ (accessed on 10 March 2022).
- Neoteryx. The Mitra 96-Autorack. 2022. Available online: https://www.neoteryx.com/mitra-high-throughput-rack-systems?hsLang=en (accessed on 10 March 2022).
- Neoteryx. The Mitra Cartridge. 2022. Available online: https://www.neoteryx.com/mitra-cartridge-blood-sampling-device-dbs?hsLang=en (accessed on 10 March 2022).
- Neoteryx. The Mitra Clamshell. 2022. Available online: https://www.neoteryx.com/mitra-clamshell-blood-collection-device?hsLang=en (accessed on 10 March 2022).
- Medical TSa. Hemapen. 2022. Available online: https://www.trajanscimed.com/pages/hemapen (accessed on 10 March 2022).
- HemaXis. HemaXis DB10 Whole Blood Collection Device. 2022. Available online: https://hemaxis.com/products/hemaxis-db10/ (accessed on 10 March 2022).
- Spot On Sciences. HEMASPOT HD. Available online: https://www.spotonsciences.com/hemaspot-hd/ (accessed on 10 March 2022).
- Spot On Sciences. HEMASPOT HF. Available online: https://www.spotonsciences.com/hemaspot-hf/ (accessed on 10 March 2022).
- YourBioHealth. Touch Activated Phlebotomy (TAP). 2022. Available online: https://company.yourbiohealth.com/products/tap/ (accessed on 10 March 2022).
- YourBioHealth. Touch Activated Phlebotomy II (TAP II). 2022. Available online: https://company.yourbiohealth.com/products/tap-ii/ (accessed on 10 March 2022).
- Tasso, Inc. Tasso-SST. 2022. Available online: https://www.tassoinc.com/tasso-sst (accessed on 10 March 2022).
- Kok, M.G.; Nix, C.; Nys, G.; Fillet, M. Targeted metabolomics of whole blood using volumetric absorptive microsampling. Talanta 2019, 197, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Volani, C.; Caprioli, G.; Calderisi, G.; Sigurdsson, B.B.; Rainer, J.; Gentilini, I.; Hicks, A.A.; Pramstaller, P.P.; Weiss, G.; Smarason, S.V.; et al. Pre-analytic evaluation of volumetric absorptive microsampling and integration in a mass spectrometry-based metabolomics workflow. Anal. Bioanal. Chem. 2017, 409, 6263–6276. [Google Scholar] [CrossRef] [Green Version]
- Crimmins, E.M.; Zhang, Y.S.; Kim, J.K.; Frochen, S.; Kang, H.; Shim, H.; Ailshire, J.; Potter, A.; Cofferen, J.; Faul, J. Dried blood spots: Effects of less than optimal collection, shipping time, heat, and humidity. Am. J. Hum. Biol. 2020, 32, e23390. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Whiley, L.; Godzien, J.; Ruperez, F.J.; Legido-Quigley, C.; Barbas, C. In-Vial Dual Extraction for Direct LC–MS Analysis of Plasma for Comprehensive and Highly Reproducible Metabolic Fingerprinting. Anal. Chem. 2012, 84, 5992–5999. [Google Scholar] [CrossRef] [PubMed]
- WHO. Guidelines on Drawing Blood: Best Practices in Phlebotomy, Capillary Sampling; World Health Organization: Geneva, Switzerland, 2010. [Google Scholar]
- Hannon, W. Blood Collection on Filter Paper for Newborn Screening Programs; Clinical and Laboratory Standards Institute: Wayne, IL, USA, 2013. [Google Scholar]
- CDC. Laboratory Quality Assurance and Standardization Programs. In Guidelines for the Shipment of Dried Blood Spot Specimens; Centers for Disease Control and Prevention: Atlanta, GA, USA, 1993. [Google Scholar]
- Kirwan, J.A.; Brennan, L.; Broadhurst, D.; Fiehn, O.; Cascante, M.; Dunn, W.B.; Schmidt, M.A.; Velagapudi, V. Preanalytical Processing and Biobanking Procedures of Biological Samples for Metabolomics Research: A White Paper, Community Perspective (for “Precision Medicine and Pharmacometabolomics Task Group”—The Metabolomics Society Initiative). Clin. Chem. 2018, 64, 1158–1182. [Google Scholar] [CrossRef] [Green Version]
- Li, W.; Lee, M.S. Dried Blood Spots: Applications and Technique, 1st ed.; John Wiley & Sons: Hoboken, NJ, USA, 2014. [Google Scholar]
- Strnadová, K.A.; Holub, M.; Mühl, A.; Heinze, G.; Ratschmann, R.; Mascher, H.; Stöckler-Ipsiroglu, S.; Waldhauser, F.; Votava, F.; Lebl, J.; et al. Long-Term Stability of Amino Acids and Acylcarnitines in Dried Blood Spots. Clin. Chem. 2007, 53, 717–722. [Google Scholar] [CrossRef] [Green Version]
- Gao, F.; McDaniel, J.; Chen, E.Y.; Rockwell, H.E.; Drolet, J.; Vishnudas, V.K.; Tolstikov, V.; Sarangarajan, R.; Narain, N.R.; Kiebish, M.A. Dynamic and temporal assessment of human dried blood spot MS/MSALL shotgun lipidomics analysis. Nutr. Metab. 2017, 14, 28. [Google Scholar] [CrossRef] [Green Version]
- Grecsó, N.; Zádori, A.; Baráth, Á.; Galla, Z.; Rácz, G.; Bereczki, C.; Monostori, P. Comparison of different preparation techniques of dried blood spot quality controls in newborn screening for congenital adrenal hyperplasia. PLoS ONE 2021, 16, e0252091. [Google Scholar] [CrossRef]
- González-Domínguez, R.; González-Domínguez, Á.; Sayago, A.; Fernández-Recamales, Á. Recommendations and Best Practices for Standardizing the Pre-Analytical Processing of Blood and Urine Samples in Metabolomics. Metabolites 2020, 10, 229. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.Y.; Summerhill, K.; Rodriguez-Canas, C.; Mather, I.; Patel, P.; Eiden, M.; Young, S.; Forouhi, N.G.; Koulman, A. Development and validation of a robust automated analysis of plasma phospholipid fatty acids for metabolic phenotyping of large epidemiological studies. Genome Med. 2013, 5, 39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Börsch-Supan, A.; Weiss, L.M.; Börsch-Supan, M.; Potter, A.J.; Cofferen, J.; Kerschner, E. Dried blood spot collection, sample quality, and fieldwork conditions: Structural validations for conversion into standard values. Am. J. Hum. Biol. 2020, 33, e23517. [Google Scholar] [CrossRef] [PubMed]
- Karvaly, G.; Molnár-Világos, G.; Kovács, K.; Mészáros, K.; Patócs, A.; Vásárhelyi, B. Evaluation of the Analytical and Clinical Concordance of 25-Hydroxyvitamin D Levels in Dried Blood Spots, Dried Serum Spots, and Serum as Potential Biorepository Specimens. Biopreservation Biobanking 2017, 15, 285–292. [Google Scholar] [CrossRef]
- Mastronardi, C.A.; Whittle, B.; Tunningley, R.; Neeman, T.; Paz-Filho, G. The use of dried blood spot sampling for the measurement of HbA1c: A cross-sectional study. BMC Clin. Pathol. 2015, 15, 13. [Google Scholar] [CrossRef] [Green Version]

| Sampling Technology | Device (Manufacturer) | Reps (Model); Volume | Biofluids | Description | Refs. |
|---|---|---|---|---|---|
| Traditional dried samples | Whatman (Cytiva; Marlborough, MA, USA) | 5 (903 Protein Saver Card); 75–80 µL | Urine, WB | Traditional paper-based carrier for collection of dried samples. | [82,83] |
| PerkinElmer (PerkinElmer; Waltham, MA, USA) | 5 (226 Spot Saver Card); Up to 70 µL | WB | Traditional paper-based carrier for collection of dried samples. | [84] | |
| Ahlstrom-Munksjö (Ahlstrom-Munksjö; Helsinki, Finland) | 5 (BioSample Card); Up to 70 µL | WB | Traditional paper-based carrier for collection of dried samples. | [85] | |
| Advanced dried samples | TASSO (HemoLink; Seattle, WA, USA) | 4 (M20); 20 µL | WB | M20 device collects four dried whole blood samples. The sample pod can be removed from the button and sent to the lab. | [36,77,86] |
| Capitainer (Capitainer AB; Solna, Sweden) | 2 (qDBS); 10 µL 2 (B Vanadate); 10 µL | WB | The qDBS (quantitative dried blood spot) collects two fixed-volume DBSs. A drop of blood is applied to the two microchannels within the device. After filling, the film at the capillary outlet dissolves, emptying the blood on the pre-punched Ahlstrom filter paper discs via capillary action. Successful sampling is indicated by the exit area of the device turning dark. Discs are then dried at room temperature for a minimum of 2 h. Then, the DBSs can be removed with tweezers for analysis. B Vanadate operates under the same mechanism with an additional phospholipase D inhibitor, sodium metavanadate (NaVO3), in the DBS disc, to allow for accurate phosphatidylethanol testing. | [87,88] | |
| Volumetric Absorptive Microsampling (VAMS) Mitra (Neoteryx; Torrance, CA, USA) | 2 (Cartridge); 10, 20, or 30 µL 4 (Clamshell) 96 (Autorack) | Urine, WB (incl. serum and plasma) | A porous, absorbent white tip attached to a plastic handler. Tip consists of hydrophilic polymer and permits haematocrit-independent collection of desired fluid by wicking up an accurate volume via capillary action. Different sample volumes can be collected depending on tip size. Sample preparation can be performed manually by detaching the tip to transfer for extraction. | [89,90,91] | |
| hemaPEN (Trajan; Melbourne, VIC, Australia) | 4; 2.74 µL | WB | Haematocrit-independent sampling system for volumetric collection of capillary blood on pre-punched filter paper discs. Contains four EDTA-coated capillaries, which fill by touching surface of blood drop of minimally 20 µL. Subsequently, clicking device into plastic base and inverting it allows capillaries to transfer blood onto four integrated pre-punched filter paper discs. Device is available with both Whatman and Perkin Elmer discs. TGA approved. | [92] | |
| HemaXis (DBS System SA; Gland, Switzerland) | 4 (DB10); 10 µL | WB | DB10 utilises a conventional Whatman or Perkin Elmer card combined with 4 volumetric capillary channels. Following fingerstick lancing, a blood drop is applied to the inlet of the capillary, collecting 10 µL of blood per channel. When filling of a channel is completed, blood can be observed at the outlet of the channel. This indicates sufficient application. The device is then manually closed to allow contact between the capillary outlet and the card, taking approximately 5 s for transfer of blood. | [93] | |
| HemaSpot (Spot On Sciences; San Francisco, CA, USA) | Multiple (HD); 160 µL 8 (HF); 9.2 µL | WB | The HD is a large DBS that allows for multiple technical replicates upon performing a sub-punch; HF fan-shaped device has eight identical blades in protective plastic cassette. Two–three drops of blood must be applied to the centre of the device to ensure equal distribution of blood (taking approximately one minute). Each blade holds 1/8th of the total sample volume (~9.2 µL). The device can be closed after filling and is then left to dry, facilitated by the integrated desiccant, before it is ready for extraction. | [94,95] | |
| Passive separation devices | Capitainer (Capitainer AB; Solna, Sweden) | 1 (DPS *); 11.6 µL | WB (separated into plasma) | The autonomous microfluidic DPS device collects plasma samples in under 6 min. It consists of a filtration membrane, a capillary metering channel, absorbent paper, and a drainage valve to remove excess plasma. A blood drop is applied to the filtration membrane, and the filtered plasma fills the capillary metering channel before it is absorbed by the DPS paper. Excess plasma is removed by the drainage valve. | [74] |
| Book-Type Dried Plasma Spot Cards (Q2 Solutions; Morrisville, NC, USA) | 4 (DPS *); variable (8–14 mm spots) | WB (separated into plasma) | Device consists of two layers that filter erythrocytes from WB to produce plasma. Blood is applied to the card in the closed-book configuration. After approximately 3 min, the sample book can be opened to collect the DPSs by removing the paper substrate. Volume of the DPS obtained depends on applied WB volume (approximately 0.303 µL plasma per 1 µL of WB). | [75] | |
| HemaXis (DBS System SA; Gland, Switzerland) | 1 (DX *); 2µL | WB (separated into plasma) | The DX operates in the same way; however, it creates DPSs under the process of sedimentation once the device is closed. This is achieved through capillary pressure in a microfluidic system created with two polydimethylsiloxane sub-units. | [76] | |
| Telimmune Plasma Separation Cards (Novilytic; West Lafayette, IN, USA) | 1 (Uno); 2.5 µL (plasma) 2 (Duo); 3.8 µL (plasma) | WB (separated into plasma) | Telimmune (formerly Noviplex) Uno and Duo collect dried plasma through membrane filtration. Blood is applied to the top layer of the device. Respectively, 25 µL of blood and 60 µL of blood are required for application on the devices; an indicator control spot on the top layer changes colour once this amount is reached. A separation membrane beneath the top layer retains erythrocytes through size filtration. This allows plasma to flow through. The process takes approximately 3 min, after which the top layer is peeled and discarded before drying the DPS for an additional 15 min. The DPS can then be removed using tweezers. | [33] | |
| HemaSpot (Spot On Sciences; San Francisco, CA, USA) | Multiple (SE); ~4 µL | WB (separated into serum) | SE device has a spiral-shaped design. It separates large whole blood components such as RBCs, platelets, and leukocytes from serum. Three–four blood drops applied to the centre allow the spiral-shaped membrane to separate different blood components through lateral flow. The serum components flow freely over the membrane, whereas the larger components are retained near the centre of the spiral. After sample is dried for two minutes, the cartridge is closed and is ready for analysis. Sub-punches can be made in different parts of spiral. | [34] | |
| Whole biofluid collectors | MSW2 (Shimadzu; Kyoto, Japan) | 1 (Wing); 23 µL (WB) 5.6 µL (plasma) 14 (Windmill) | WB (separated into plasma) | Blood is collected at the tip of the Wing after performing a fingerstick (or similar) with a lancing device. The device is filled once blood reaches the reservoir (23 µL). A specialised centrifuge rotor is required (the Windmill), which can hold 14 Wings for centrifugation. Typically, the sample is then spun down as 2000 × G for 10 min. Following centrifugation, two plasma zones are created and can be snapped off with hands (i.e., does not require a specialised cutter) into a microtube for extraction. | [32] |
| Touch Activated Phlebotomy (TAP) (Yourbio Health; Medford, MA, USA) | 1 (TAP); 100 µL 1 (TAP II *); 250 µL | WB | Collects blood under vacuum pressure through microneedles, painlessly. Device sticks onto the skin (upper arm). Once button pushed, a ring of 30 small microneedles projects out of the device to micropuncture the skin. The needles immediately withdraw, creating a vacuum to induce collection of capillary blood. Blood flows through microfluidic channels into storage space, prefilled with lithium heparin anticoagulant. Subsequently, indicator window turns red when the reservoir is full (up to three minutes). Entire device can be sent to lab for collection and analysis of blood from device. | [96,97] | |
| TASSO (HemoLink; Seattle, WA, USA) | 1 (TASSO+); 200–600 µL 1 (SST); 200–300 µL | WB (incl. serum and plasma) | Placed on upper arm. Collects WB by pressing the button, which results in a lancet puncturing the skin. Blood collected from capillaries under vacuum pressure into the sample pod (different formats). TASSO+ device collects whole blood for a variety of standard collection tubes (i.e., EDTA and lithium-heparin) and passive separation tubes (serum and plasma); SST device pod collects liquid WB, which can be spun into serum at lab. | [31,98] |
| Microsampling Technology | Extraction Solvent | Metabolic Phenotyping Method | Title | Ref. |
|---|---|---|---|---|
| Traditional dried samples (DBS) | MeOH | Targeted DI-HRMS for amino acids, ketones, lipids Untargeted DI-HRMS | Understanding acute metabolic decompensation in propionic and methylmalonic acidemias: A deep metabolic phenotyping approach | [46] |
| Traditional dried samples (DBS) | MeOH | UPLC–MS/MS TQ, BEH C18 column Targeted analysis of anticancer drug, cyclophosphamide, metabolites | Phenotyping study of cyclophosphamide 4-hydroxylation in Malay cancer patients | [43] |
| Traditional dried samples (DBS) | MeOH:H2O (4:1, v/v) 2% formic acid | Untargeted UHPLC–qTOF–MS RP and HILIC separation | A feasibility study of metabolic phenotyping of dried blood spot specimens in rural Chinese women exposed to household air pollution | [44] |
| Traditional dried samples (DBS) | 100 µL of MilliQ H2O 250 µL of MeOH 500 µL of MTBE | Orbitrap—DI-HRMS Lipidomics | The development and validation of a fast and robust dried blood spot based lipid profiling method to study infant metabolism | [61] |
| Traditional dried samples (DBS, DPS) | MeOH | 7890A GC System coupled to 5975 inert MSD with Triple-Axis Detector | Evaluation of dried blood spots as sample matrix for gas chromatography/mass spectrometry based metabolomic profiling | [58] |
| Traditional dried samples (DBS, DUS) | DBS: MeOH:H2O (4:1, v/v) DUS: MeOH:H2O (8:1, v/v) | UHPLC–MS RP, HILIC, lipid methods | Investigation of the 12-month stability of dried blood and urine spots applying untargeted UHPLC–MS metabolomic assays | [55] |
| Traditional dried samples (DBS) | ACN/MeOH with H2O at a ratio of 2:1–4:1 | LC–MS/MS Drug, lipid, acylcarnitine analyses | Drug, lipid, and acylcarnitine profiling using dried blood spot (DBS) technology in drug discovery | [45] |
| Traditional dried samples (DBS) | Not specified: “relatively uncomplicated sample extraction steps” | DI-HRMS | Untargeted metabolic profiling in dried blood spots identifies disease fingerprint for pyruvate kinase deficiency | [47] |
| Traditional dried samples (DBS) | MeOH:ACN (1:1) | LC–MS | Nontargeted mass spectrometry of dried blood spots for interrogation of the human circulating metabolome | [60] |
| Traditional dried samples (DBS) | 25% (v/v) aqueous MeOH | UPLC–MS | Metabolite profiles from dried blood spots for metabonomic studies using UPLC combined with orthogonal acceleration ToF-MS: effects of different papers and sample storage stability | [59] |
| Traditional dried samples (DBS) Advanced dried samples (HemaXis DB10, TASSO-M20) | MeOH:H2O (95:5, v/v) | UHPLC–MS/MS MRM of steroid hormones | Steroid profiling by UHPLC–MS/MS in dried blood spots collected from healthy women with and without testosterone gel administration | [36] |
| Advanced dried samples (VAMS) | ACN:H2O (60:40, v/v) | UHPLC–MS/MS for organic acids HILIC–MS/MS for amino acids | Targeted metabolomics of whole blood using volumetric absorptive microsampling | [99] |
| Advanced dried samples (VAMS) | ACN:H2O (70:30, v/v) | UHPLC–qToF–MS HILIC Untargeted analysis | Pre-analytical evaluation of volumetric absorptive microsampling and integration in a mass spectrometry-based metabolomics workflow | [100] |
| Advanced dried samples (hemaPEN) | ACN:H2O (60:40, v/v) | UHPLC–MS/MS Organic acid, amino acid methods | Blood microsampling to monitor metabolic profiles during physical exercise | [72] |
| Advanced dried samples (hemaPEN) | MeOH:H2O (80:20, v/v) 0.01% formic acid | LC–MS/MS Targeted analysis of caffeine and paraxanthine | Evaluation of the performance and hematocrit independence of the hemaPEN as a volumetric dried blood spot collection device | [27] |
| Advanced dried samples (Capitainer B Vanadate) | MeOH:H2O (80:20, v/v) 0.01% formic acid | UPLC–MS/MS Targeted analysis of caffeine and paraxanthine | Evaluation of the Capitainer-B microfluidic device as a new hematocrit-independent alternative for dried blood spot collection | [73] |
| Traditional dried samples (DBS) Advanced dried samples (VAMS) Passive separation devices (Telimmune, formerly Noviplex plasma prep cards) | H2O | HILIC–UPLC–FLR Glycoprofiling | Comparison of self-sampling blood collection for N-glycosylation analysis | [53] |
| Whole biofluid collectors (MSW2) | 5 µL IS: ACN/MeOH (70:30, v/v) +100 µL MeOH | LC–MS/MS Targeted analysis of carbamazepine (and 7 of its metabolites), lamotrigine, phenytoin antiepileptic drugs | Quantitative and qualitative application of a novel capillary microsampling device, Microsampling Wing™ (MSW), using antiepileptic drugs in rats | [78] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roberts, J.L.; Whiley, L.; Gray, N.; Gay, M.; Lawler, N.G. Advanced Microsamples: Current Applications and Considerations for Mass Spectrometry-Based Metabolic Phenotyping Pipelines. Separations 2022, 9, 175. https://doi.org/10.3390/separations9070175
Roberts JL, Whiley L, Gray N, Gay M, Lawler NG. Advanced Microsamples: Current Applications and Considerations for Mass Spectrometry-Based Metabolic Phenotyping Pipelines. Separations. 2022; 9(7):175. https://doi.org/10.3390/separations9070175
Chicago/Turabian StyleRoberts, Jayden Lee, Luke Whiley, Nicola Gray, Melvin Gay, and Nathan G. Lawler. 2022. "Advanced Microsamples: Current Applications and Considerations for Mass Spectrometry-Based Metabolic Phenotyping Pipelines" Separations 9, no. 7: 175. https://doi.org/10.3390/separations9070175
APA StyleRoberts, J. L., Whiley, L., Gray, N., Gay, M., & Lawler, N. G. (2022). Advanced Microsamples: Current Applications and Considerations for Mass Spectrometry-Based Metabolic Phenotyping Pipelines. Separations, 9(7), 175. https://doi.org/10.3390/separations9070175







