A Study of Copper (II) Ions Removal by Reverse Osmosis under Various Operating Conditions
Abstract
:1. Introduction
2. Materials and Methods
2.1. Artificial Wastewater Preparation
2.2. RO Membrane Setup
2.3. Test Method
2.4. Theoretical Calculations
3. Results and Discussion
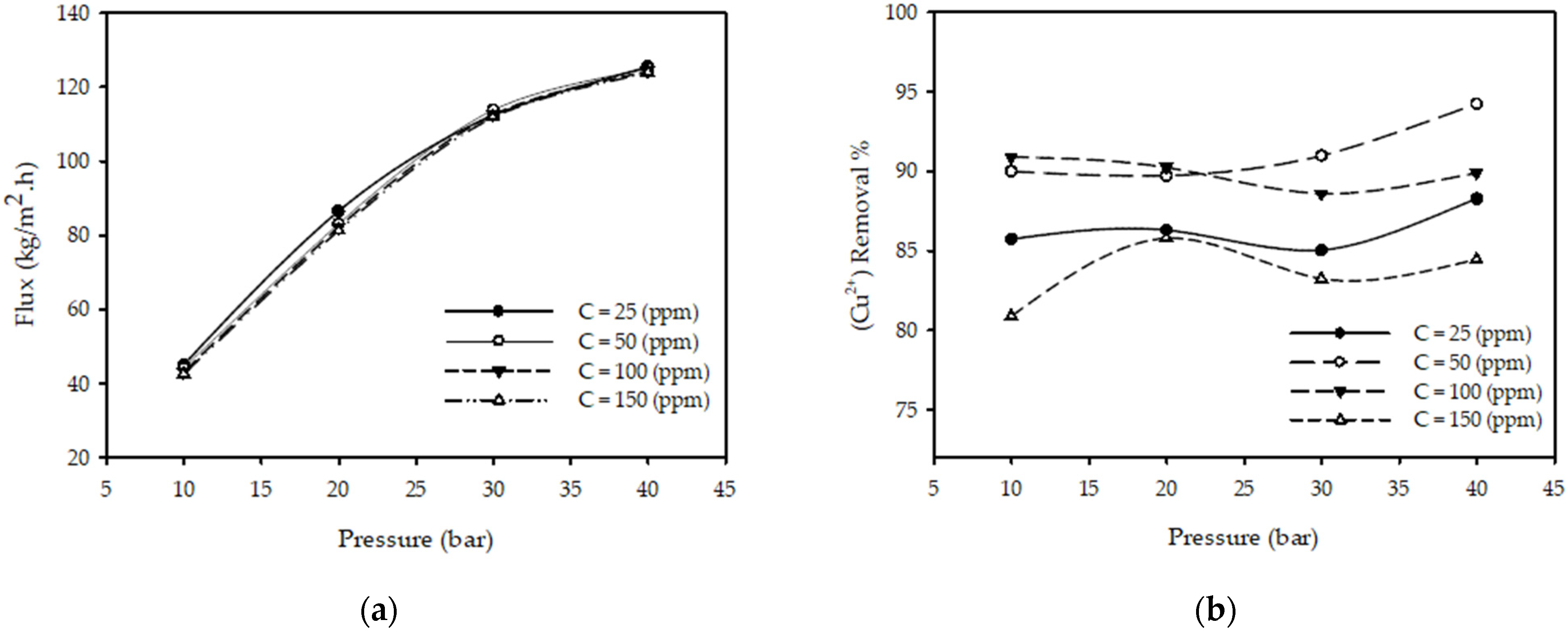
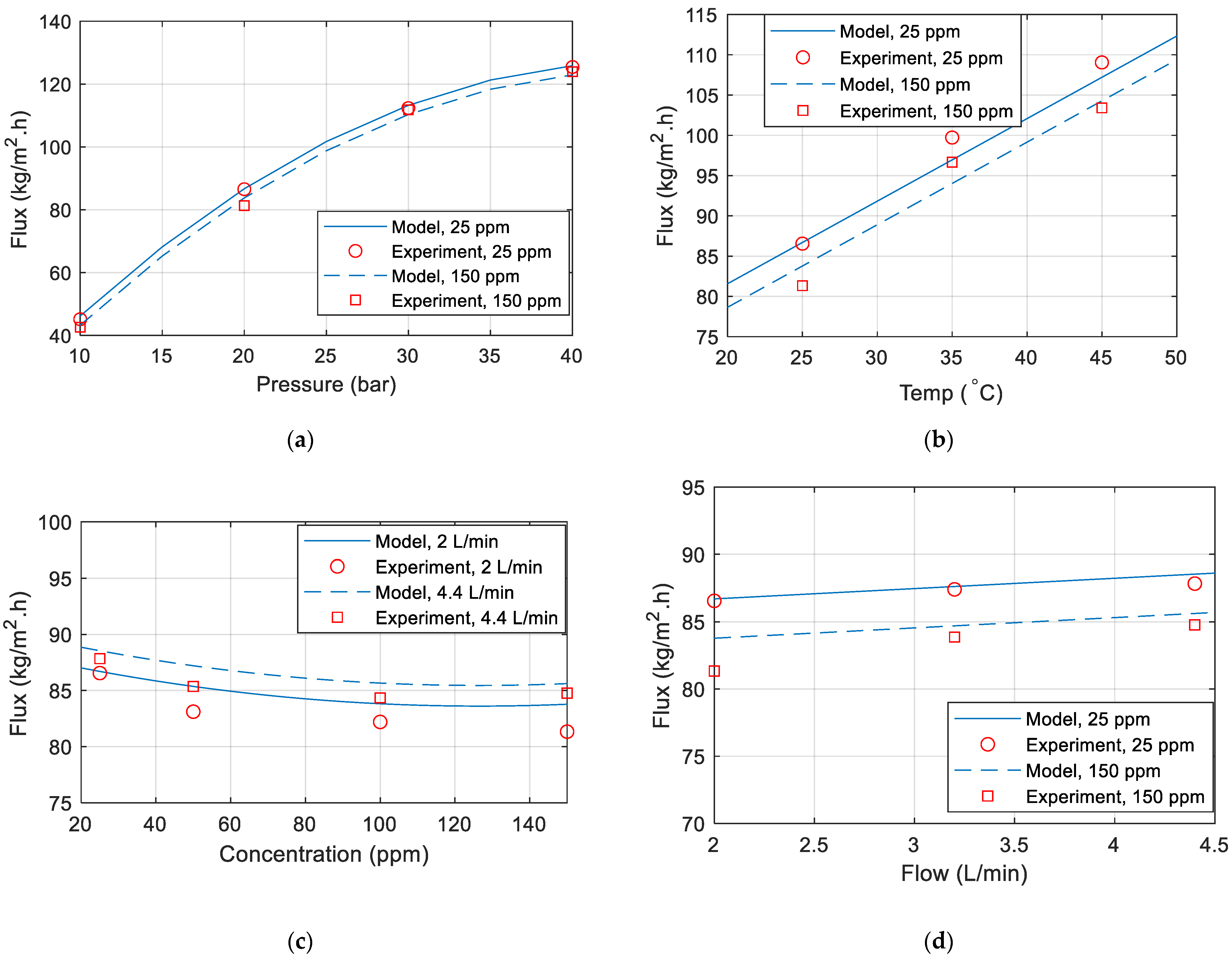
3.1. Effect of Operating Pressure
3.2. Effect of Feed Temperature
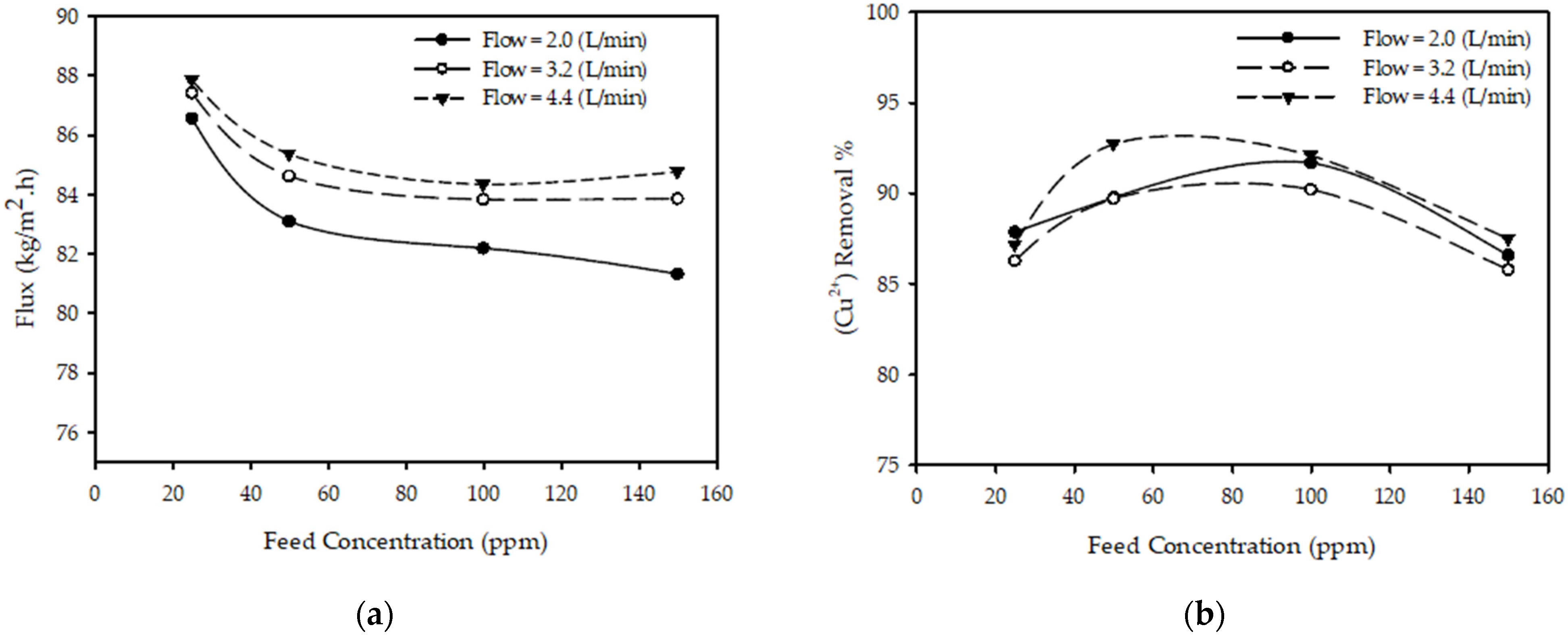
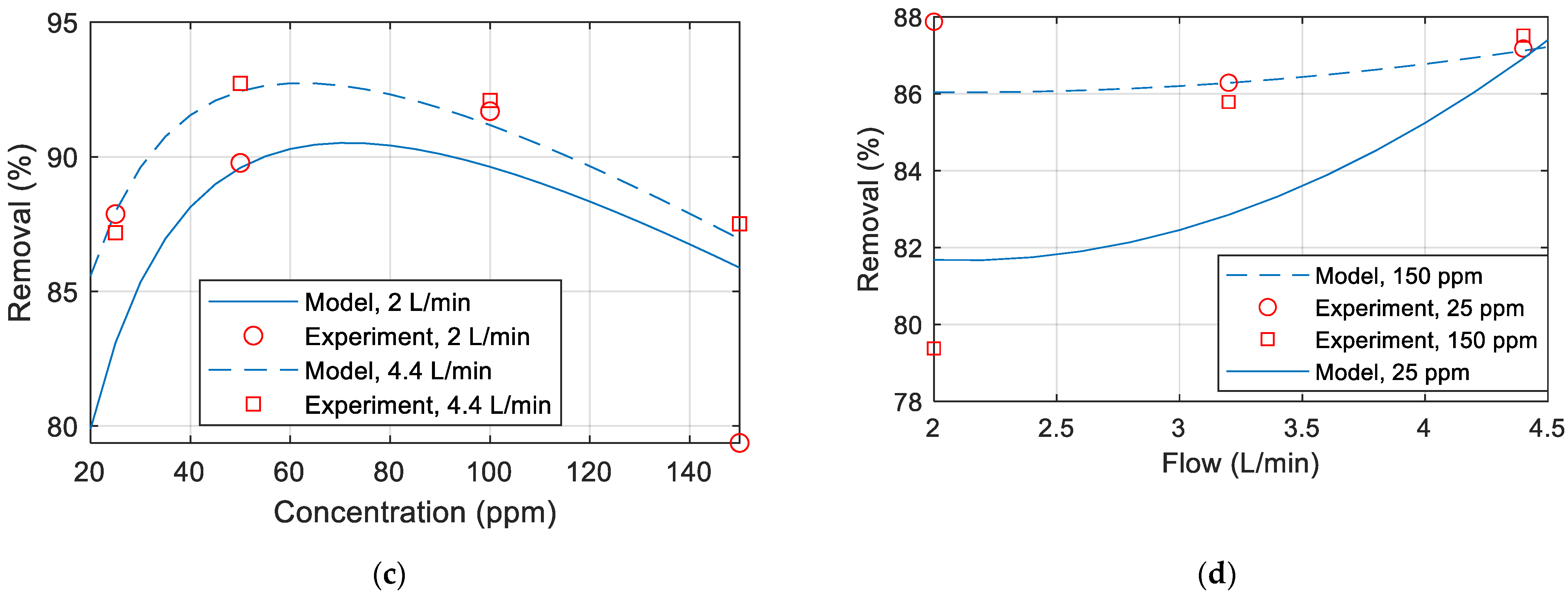
3.3. Effect of Feed Concentration
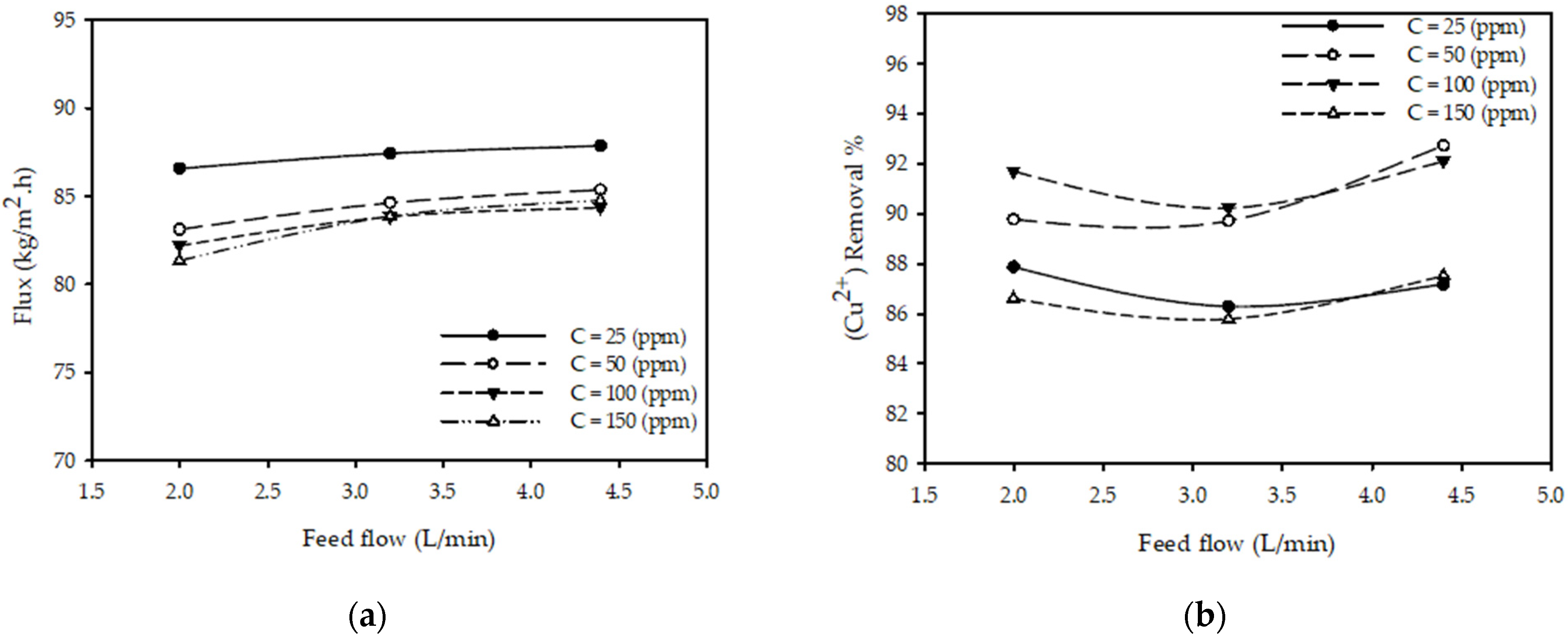
3.4. Effect of Feed Flow Rate
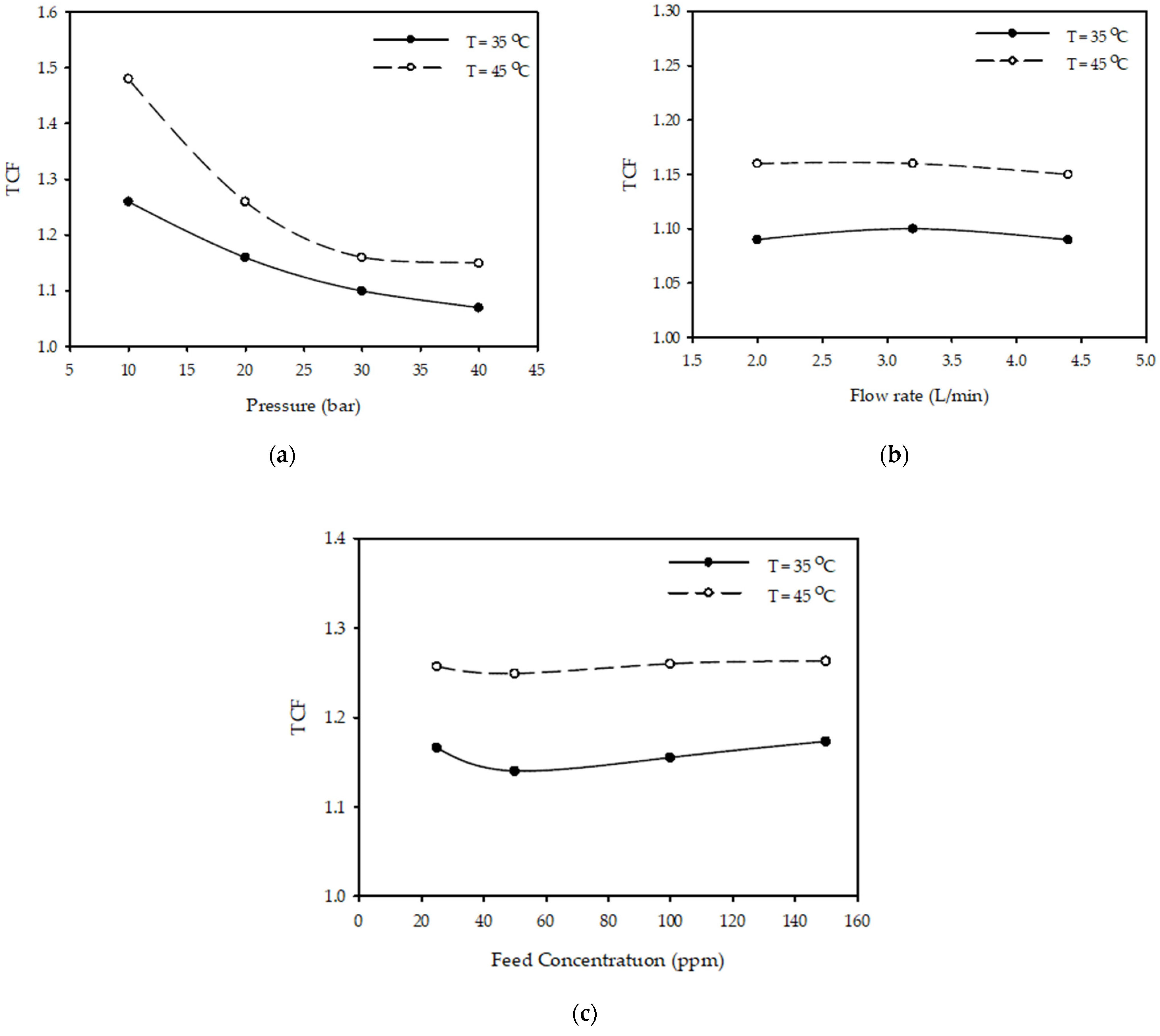
3.5. Temperature Correction Factor (TCF)
3.6. Mathematical Model
3.6.1. Permeate Flux Model
3.6.2. Copper Removal Model
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gautam, P.K.; Gautam, R.K.; Banerjee, S.; Chattopadhyaya, M.C.; Pandey, J.D. Heavy metals in the environment: Fate, transport, toxicity and remediation technologies. Heavy Met. Sources Toxic. Remediat. Tech. 2016, 60, 101–130. [Google Scholar]
- Xu, Z. Removal of Heavy Metal Ions from Aqueous Solution by Alkaline Filtration; University of Ottawa: Ottawa, ON, Canada, 2020. [Google Scholar]
- Vidu, R.; Matei, E.; Predescu, A.M.; Alhalaili, B.; Pantilimon, C.; Tarcea, C.; Predescu, C. Removal of heavy metals from wastewaters: A challenge from current treatment methods to nanotechnology applications. Toxics 2020, 8, 101. [Google Scholar] [CrossRef] [PubMed]
- Fu, F.; Xie, L.; Tang, B.; Wang, Q.; Jiang, S. Application of a novel strategy—Advanced Fenton-chemical precipitation to the treatment of strong stability chelated heavy metal containing wastewater. Chem. Eng. J. 2012, 189–190, 283–287. [Google Scholar] [CrossRef]
- Polat, H.; Erdogan, D. Heavy metal removal from waste waters by ion flotation. J. Hazard. Mater. 2007, 148, 267–273. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yuan, X.Z.; Meng, Y.T.; Zeng, G.M.; Fang, Y.Y.; Shi, J.G. Evaluation of tea-derived biosurfactant on removing heavy metal ions from dilute wastewater by ion flotation. Colloids Surf. A Physicochem. Eng. Asp. 2008, 317, 256–261. [Google Scholar] [CrossRef]
- Thakare, Y.N.; Jana, A.K. Performance of high density ion exchange resin (INDION225H) for removal of Cu(II) from waste water. J. Environ. Chem. Eng. 2015, 2, 1393–1398. Available online: https://www.infona.pl//resource/bwmeta1.element.elsevier-30052313-c7a7-35c1-a784-6e6a3cfc4070 (accessed on 1 May 2022). [CrossRef]
- Shaidan, N.H.; Eldemerdash, U.; Awad, S. Removal of Ni(II) ions from aqueous solutions using fixed-bed ion exchange column technique. J. Taiwan Inst. Chem. Eng. 2012, 1, 40–45. Available online: https://www.infona.pl//resource/bwmeta1.element.elsevier-4cae4d9c-909f-3b39-a0e9-c4185990983f (accessed on 15 May 2022). [CrossRef]
- Oussedik, S.M.; Khelifa, A. Reduction of copper ions concentration in wastewaters of galvanoplastic industry by electroflotation. Desalination 2001, 139, 383. [Google Scholar] [CrossRef]
- Konstantinos, D.; Christoforidis, A.; Valsamidou, E. Removal of Nickel, Copper, Zinc and Chromium from Synthetic and Industrial Wastewater by Electrocoagulation. Available online: https://www.researchgate.net/publication/284650056_Removal_of_nickel_copper_zinc_and_chromium_from_synthetic_and_industrial_wastewater_by_electrocoagulation (accessed on 20 January 2022).
- Zhu, S.; Chen, Y.; Khan, M.A.; Xu, H.; Wang, F.; Xia, M. In-Depth Study of Heavy Metal Removal by an Etidronic Acid-Functionalized Layered Double Hydroxide. ACS Appl. Mater. Interfaces 2022, 14, 7450–7463. [Google Scholar] [CrossRef]
- Zhu, S.; Khan, M.A.; Kameda, T.; Xu, H.; Wang, F.; Xia, M.; Yoshioka, T. New insights into the capture performance and mechanism of hazardous metals Cr3+ and Cd2+ onto an effective layered double hydroxide-based material. J. Hazard. Mater. 2022, 426, 128062. [Google Scholar] [CrossRef]
- Kosa, S.A.; Al-Zhrani, G.; Abdel Salam, M. Removal of heavy metals from aqueous solutions by multi-walled carbon nanotubes modified with 8-hydroxyquinoline. Chem. Eng. J. 2012, 181–182, 159–168. [Google Scholar] [CrossRef]
- Lo, S.F.; Wang, S.Y.; Tsai, M.J.; Lin, L.D. Adsorption capacity and removal efficiency of heavy metal ions by Moso and Ma bamboo activated carbons. Chem. Eng. Res. Des. 2012, 90, 1397–1406. [Google Scholar] [CrossRef]
- Zhu, S.; Xia, M.; Chu, Y.; Khan, M.A.; Lei, W.; Wang, F.; Muhmood, T.; Wang, A. Adsorption and Desorption of Pb(II) on l-Lysine Modified Montmorillonite and the simulation of Interlayer Structure. Appl. Clay Sci. 2019, 169, 40–47. [Google Scholar] [CrossRef]
- Barakat, M.A.; Schmidt, E. Polymer-enhanced ultrafiltration process for heavy metals removal from industrial wastewater. Desalination 2010, 256, 90–93. [Google Scholar] [CrossRef]
- Uzal, N.; Jaworska, A.; Miśkiewicz, A.; Zakrzewska-Trznadel, G.; Cojocaru, C. Optimization of Co2+ ions removal from water solutions via polymer enhanced ultrafiltration with application of PVA and sulfonated PVA as complexing agents. J. Colloid Interface Sci. 2011, 362, 615–624. [Google Scholar] [CrossRef]
- Gunatilake, S.K. Methods of Removing Heavy Metals from Industrial Wastewater. Methods 2015, 1, 12–18. [Google Scholar]
- Qdais, H.A.; Moussa, H. Removal of heavy metals from wastewater by membrane processes: A comparative study. Desalination 2004, 164, 105–110. [Google Scholar] [CrossRef]
- Mehdipour, S.; Vatanpour, V.; Kariminia, H.R. Influence of ion interaction on lead removal by a polyamide nanofiltration membrane. Desalination 2015, 362, 84–92. [Google Scholar] [CrossRef]
- Aljendeel, H.A. Removal of Heavy Metals Using Reverse Osmosis. J. Eng. 2011, 17, 647–658. [Google Scholar]
- Al-Saydeh, S.A.; El-Naas, M.H.; Zaidi, S.J. Copper removal from industrial wastewater: A comprehensive review. J. Ind. Eng. Chem. 2017, 56, 35–44. [Google Scholar] [CrossRef]
- Algureiri, A.H.; Abdulmajeed, Y.R. Removal of Heavy Metals from Industrial Wastewater by Using RO Membrane. Iraqi J. Chem. Pet. Eng. 2016, 17, 125–136. Available online: www.iasj.net (accessed on 5 January 2022).
- Myzairah, B. Hamdzah Low Pressure Reverse Osmosis Membrane for Rejection of Heavy Metals; Universiti Teknologi Malaysia: Johor Bahru, Malaysia, 2007. [Google Scholar]
- Applied Membranes Comparison of Membrane Processes. 2003. Available online: http://www.appliedmembranes.com/pdf/membrane_specs_ami/Comparison (accessed on 7 April 2022).
- Dimitriou, E.; Boutikos, P.; Mohamed, E.S.; Koziel, S.; Papadakis, G. Theoretical performance prediction of a reverse osmosis desalination membrane element under variable operating conditions. Desalination 2017, 419, 70–78. [Google Scholar] [CrossRef]
- Ruiz-García, A.; Nuez, I.; Carrascosa-Chisvert, M.D.; Santana, J.J. Simulations of BWRO systems under different feedwater characteristics. Analysis of operation windows and optimal operating points. Desalination 2020, 491, 114582. [Google Scholar] [CrossRef]
- Ansari, M.; Al-Obaidi, M.A.; Hadadian, Z.; Moradi, M.; Haghighi, A.; Mujtaba, I.M. Performance evaluation of a brackish water reverse osmosis pilot-plant desalination process under different operating conditions: Experimental study. Clean. Eng. Technol. 2021, 4, 100134. [Google Scholar] [CrossRef]
- Arvind Patil; Gary Hatch Factors that Influence the Performance of RO Systems. Available online: https://www.lenntech.com/performance-influence-factors.htm (accessed on 10 March 2022).
- Al-Mutaz, I.S.; Al-Ghunaimi, M.A. Performance of Reverse Osmosis Units at High Temperatures. The Ida World Congress on Desalination and Water Reuse. Available online: https://www.researchgate.net/publication/264420571 (accessed on 23 January 2022).
- Shigidi, I.; Anqi, A.E.; Elkhaleefa, A.; Mohamed, A.; Ali, I.H.; Brima, E.I. Temperature impact on reverse osmosis permeate flux in the remediation of hexavalent chromium. Water 2022, 14, 44. [Google Scholar] [CrossRef]
- Çimen, A.; Kılıçel, F.; Arslan, G. Removal of chromium ions from waste waters using reverse osmosis AG and SWHR membranes. Russ. J. Phys. Chem. A 2014, 88, 845–850. [Google Scholar] [CrossRef]
- Mousavi Rad, S.A.; Mirbagheri, S.A.; Mohammadi, T. Using reverse osmosis membrane for chromium removal from aqueous solution. World Acad. Sci. Eng. Technol. 2009, 57, 348–352. [Google Scholar] [CrossRef]
- Goosen, M.F.A.; Sablani, S.S.; Al-Maskari, S.S.; Al-Belushi, R.H.; Wilf, M. Effect of feed temperature on permeate flux and mass transfer coefficient in spiral-wound reverse osmosis systems. Desalination 2002, 144, 367–372. [Google Scholar] [CrossRef]
- Kocurek, P.; Kolomazník, K.; Bařinová, M. Chromium removal from wastewater by reverse osmosis. WSEAS Trans. Environ. Dev. 2014, 10, 358–365. [Google Scholar]



| Membrane Type | Initial Cocn. (ppm) | Operating Pressure | pH | Removal | References |
|---|---|---|---|---|---|
| Efficiency % | |||||
| UF | 1000 | 1 | 7 | 97.6 | [16] |
| PEUF | 5–10 | - | 6.5 | 99.98 | [17] |
| UF | 10–160 | 2–5 | 5.5–6 | 90 | [18] |
| NF | 25–200 | 7–13 | 4–11 | 90 | [19] |
| NF | 20–400 | 10–40 | 5.6 | 97.5 | [20] |
| RO | 25–200 | 7–13 | 4–11 | 98 | [19] |
| RO | 50–150 | 4–10 | - | 96.6 | [21] |
| RO | 50–200 | 1–4 | 2–5.5 | 96 | [23] |
| RO | 100–500 | 3–5 | 4–11 | 99.5 | [22] |
| Flow Rate (L/min) | Pressure (Bar) | TCF | |
|---|---|---|---|
| 35 °C | 45 °C | ||
| 2.0 | 10 | 1.26 | 1.48 |
| 20 | 1.15 | 1.24 | |
| 30 | 1.09 | 1.16 | |
| 40 | 1.07 | 1.16 | |
| 3.2 | 10 | 1.26 | 1.48 |
| 20 | 1.16 | 1.26 | |
| 30 | 1.09 | 1.15 | |
| 40 | 1.07 | 1.15 | |
| 4.4 | 10 | 1.26 | 1.48 |
| 20 | 1.16 | 1.26 | |
| 30 | 1.09 | 1.15 | |
| 40 | 1.07 | 1.16 | |
| Permeate Flux vs. | Relationship Function | Justification |
|---|---|---|
| Temperature | f(T) = aT + b | Linear curves observed in Figure 3a. |
| Pressure | g(P) = aP2 + bP + c | Curves obtained in Figure 2a are linear, but nonlinear behavior is observed at high pressure. The term aP2 will compensate for this nonlinearity. |
| Concentration | h(C) = aC2 + bC + c | Curves in Figure 4a are nonlinear, especially at low concentrations. A parabolic fitting can be used to match the permeate flux vs. concentration curve. |
| Flow | m(F) = aF + b | Small variation in the permeate flux can be observed in Figure 5a, hence a linear model should be sufficient to model these variations. |
| Constant | Optimum Value |
|---|---|
| a0 | −33.6615 |
| a1 | 1.0250 |
| a2 | 6.1397 |
| a3 | −0.0697 |
| a4 | −0.0759 |
| a5 | 0.0003 |
| a6 | 0.7670 |
| Permeate Flux vs. | Relationship Function | Justification |
|---|---|---|
| Temperature | f(T) = aT2 + bT + c | Nonlinear curves observed in Figure 3b are similar to parabolic curves. |
| Pressure | g(P) = aP3 + bP2 + cP + d | Curves in Figure 2b have maxima and minima. A quadratic equation will be insufficient to model this curve because it has only one minima/ maxima; hence, a higher-order (cubic) function is required. |
| Concentration | h(C) = aC2 + bC + c | Curves in Figure 4b are nonlinear, especially with only one maximum; hence, a parabolic fitting can be used to match the data. |
| Flow | m(F) = aF2 + bF + c | Nonlinear variation with the flow is observed in Figure 5b, with one maximum value; hence, a quadradic model is used. |
| Constant | Optimum Value |
|---|---|
| b0 | 8.9944 |
| b1 | 0.0899 |
| b2 | −0.0030 |
| b3 | −0.3250 |
| b4 | 0.0254 |
| b5 | −0.0004 |
| b6 | −0.1013 |
| b7 | 0.0008 |
| b8 | 2.0821 |
| b9 | −0.4925 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Harharah, R.H.; Abdalla, G.M.T.; Elkhaleefa, A.; Shigidi, I.; Harharah, H.N. A Study of Copper (II) Ions Removal by Reverse Osmosis under Various Operating Conditions. Separations 2022, 9, 155. https://doi.org/10.3390/separations9060155
Harharah RH, Abdalla GMT, Elkhaleefa A, Shigidi I, Harharah HN. A Study of Copper (II) Ions Removal by Reverse Osmosis under Various Operating Conditions. Separations. 2022; 9(6):155. https://doi.org/10.3390/separations9060155
Chicago/Turabian StyleHarharah, Ramzi H., Ghassan M. T. Abdalla, Abubakr Elkhaleefa, Ihab Shigidi, and Hamed N. Harharah. 2022. "A Study of Copper (II) Ions Removal by Reverse Osmosis under Various Operating Conditions" Separations 9, no. 6: 155. https://doi.org/10.3390/separations9060155
APA StyleHarharah, R. H., Abdalla, G. M. T., Elkhaleefa, A., Shigidi, I., & Harharah, H. N. (2022). A Study of Copper (II) Ions Removal by Reverse Osmosis under Various Operating Conditions. Separations, 9(6), 155. https://doi.org/10.3390/separations9060155








