Hunting Molecules in Complex Matrices with SPME Arrows: A Review
Abstract
1. Introduction
- First, all SPME technologies comprise a minute amount of extraction phase or sorbent material. This sorbent is typically adhered to a solid substrate, and said substrate can take multiple geometries [1]. The purpose of the sorptive material or coating, is to collect/enrich analytes of interest present in a complex matrix while preventing other matrix components from adhering to said surface [2].
- Second, analyte collection is based on partitioning between the extraction phase and the matrix. Thus, controlling the extraction conditions (e.g., temperature, ionic strength, and humidity) and the extraction times is critical to assure reproducible results and use this tool for quantitative applications [3].
- Third, a SPME device can carry out multiple steps of the analytical workflow such as analyte collection (e.g., sampling), sample preparation (e.g., the clean-up of analytes of interest from other matrix components), analyte transportation (e.g., when the sampling is performed outside of a laboratory), and analyte transfer into an analytical instrument (e.g., when thermal desorption is used on gas chromatography (GC)) [4].
- Fourth, most SPME technologies reduce/eliminate the use of solvents/additives during the sample preparation step and can consequently be considered green analytical chemistry technologies [5].
- Fifth, analyte elution can be performed via thermal, liquid, or laser desorption, depending on the characteristics of the extraction phase, and it can be introduced into an analytical instrument such a mass spectrometer via a chromatographic separation technique [6]. In the case of gas chromatography, analyte introduction onto the instrument is typically performed via the direct thermal desorption of the SPME device.
2. SPME Arrow Design
2.1. Physical Attributes
2.2. Phase
3. SPME Arrow Accommodations
4. SPME Arrow Performance
4.1. Benchmarking
4.2. Method Development
4.3. Troubleshooting
5. SPME Arrow Applications
5.1. Environmental Analysis
5.2. Food Analysis
5.3. Terpenes in Cannabis
6. Future Directions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Reyes-Garcés, N.; Gionfriddo, E.; Gómez-Ríos, G.A.; Alam, M.N.; Boyaci, E.; Bojko, B.; Singh, V.; Grandy, J.J.; Pawliszyn, J. Advances in solid phase microextraction and perspective on future directions. Anal. Chem. 2018, 90, 302–360. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Garcés, N.; Gionfriddo, E. Recent developments and applications of solid phase microextraction as a sample preparation approach for mass-spectrometry-based metabolomics and lipidomics. TrAC Trends Anal. Chem. 2019, 113, 172–181. [Google Scholar] [CrossRef]
- Alam, M.N.; Nazdrajić, E.; Singh, V.; Tascon, M.; Pawliszyn, J. Effect of Transport Parameters and Device Geometry on Extraction Kinetics and Efficiency in Direct Immersion Solid-phase Microextraction. Anal. Chem. 2018. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Ríos, G.A.; Mirabelli, M.F. Solid Phase Microextraction-Mass Spectrometry: Metanoia. TrAC Trends Anal. Chem. 2019, 112, 201–211. [Google Scholar] [CrossRef]
- Armenta, S.; Garrigues, S.; de la Guardia, M. The role of green extraction techniques in Green Analytical Chemistry. TrAC Trends Anal. Chem. 2015. [Google Scholar] [CrossRef]
- Gómez-Ríos, G.A.; Garcés, N.R.; Tascon, M. Smart Materials in Solid Phase Microextraction (SPME). In Handbook of Smart Materials in Analytical Chemistry; John Wiley & Sons, Ltd.: Chichester, UK, 2019; pp. 581–620. [Google Scholar] [CrossRef]
- Belardi, R.P.; Pawliszyn, J.B. The application of chemically modified fused silica fibers in the extraction of organics from water matrix samples and their rapid transfer to capillary columns. Water Pollut. Res. J. Can. 1989, 24, 179–191. [Google Scholar] [CrossRef]
- Godage, N.H.; Gionfriddo, E. A critical outlook on recent developments and applications of matrix compatible coatings for solid phase microextraction. TrAC Trends Anal. Chem. 2019, 111, 220–228. [Google Scholar] [CrossRef]
- Olcer, Y.A.; Tascon, M.; Eroglu, A.E.; Boyacı, E. Thin film microextraction: Towards faster and more sensitive microextraction. TrAC Trends Anal. Chem. 2019, 113, 93–101. [Google Scholar] [CrossRef]
- Gutiérrez-Serpa, A.; Schorn-García, D.; Jiménez-Moreno, F.; Jiménez-Abizanda, A.I.; Pino, V. Braid solid-phase microextraction of polycyclic aromatic hydrocarbons by using fibers coated with silver-based nanomaterials in combination with HPLC with fluorometric detection. Microchim. Acta 2019, 186, 311. [Google Scholar] [CrossRef]
- Poole, J.J.; Grandy, J.J.; Yu, M.; Boyaci, E.; Gómez-Ríos, G.A.; Reyes-Garcés, N.; Bojko, B.; Heide, H.V.; Pawliszyn, J. Deposition of a Sorbent into a Recession on a Solid Support to Provide a New, Mechanically Robust Solid-Phase Microextraction Device. Anal. Chem. 2017, 89, 8021–8026. [Google Scholar] [CrossRef]
- Truong, T.V.; Lee, E.D.; Black, B.D.; Truong, T.X.; Lee, M.L. Coiled wire filament sample introduction for gas chromatography–mass spectrometry. Int. J. Mass Spectrom. 2018, 427, 123–132. [Google Scholar] [CrossRef]
- Ziegler, M.; Schmarr, H.-G. Comparison of Solid-Phase Microextraction Using Classical Fibers Versus Mini-Arrows Applying Multiple Headspace Extraction and Various Agitation Techniques. Chromatographia 2019, 82, 635–640. [Google Scholar] [CrossRef]
- Dugheri, S.; Mucci, N.; Bonari, A.; Marrubini, G.; Cappelli, G.; Ubiali, D.; Campagna, M.; Montalti, M.; Arcangeli, G. Solid phase microextraction techniques used for gas chromatography: A review. Acta Chromatogr. 2019, 1–9. [Google Scholar] [CrossRef]
- Grandy, J.J.; Boyacı, E.; Pawliszyn, J. Development of a Carbon Mesh Supported Thin Film Microextraction Membrane As a Means to Lower the Detection Limits of Benchtop and Portable GC/MS Instrumentation. Anal. Chem. 2016, 88, 1760–1767. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Jimenez, J.; Zanca, N.; Lan, H.; Jussila, M.; Hartonen, K.; Riekkola, M.-L. Aerial drone as a carrier for miniaturized air sampling systems. J. Chromatogr. A 2019, 1597, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Helin, A.; Rönkkö, T.; Parshintsev, J.; Hartonen, K.; Schilling, B.; Läubli, T.; Riekkola, M.-L. Solid phase microextraction Arrow for the sampling of volatile amines in wastewater and atmosphere. J. Chromatogr. A 2015, 1426, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Kaziur, W.; Salemi, A.; Jochmann, M.A.; Schmidt, T.C. Automated determination of picogram-per-liter level of water taste and odor compounds using solid-phase microextraction arrow coupled with gas chromatography-mass spectrometry. Anal. Bioanal. Chem. 2019, 411, 2653–2662. [Google Scholar] [CrossRef]
- Risticevic, S.; Lord, H.; Górecki, T.; Arthur, C.L.; Pawliszyn, J. Protocol for solid-phase microextraction method development. Nat. Protoc. 2010, 5, 122–139. [Google Scholar] [CrossRef]
- Setkova, L.; Risticevic, S.; Linton, C.M.; Ouyang, G.; Bragg, L.M.; Pawliszyn, J. Solid-phase microextraction-gas chromatography-time-of-flight mass spectrometry utilized for the evaluation of the new-generation super elastic fiber assemblies. Anal. Chim. Acta 2007, 581, 221–231. [Google Scholar] [CrossRef]
- Gómez-Ríos, G.A.; Reyes-Garcés, N.; Pawliszyn, J. Evaluation of a multi-fiber exchange solid-phase microextraction system and its application to on-site sampling. J. Sep. Sci. 2015, 38. [Google Scholar] [CrossRef]
- Herrington, J. SPME Arrow Blog 7 ChromaBLOGraphy: Restek’s Chromatography Blog. Available online: https://blog.restek.com/?p=41704 (accessed on 31 July 2019).
- Herrington, J. SPME Arrow Blog 2 ChromaBLOGraphy: Restek’s Chromatography Blog. Available online: https://blog.restek.com/?p=37889 (accessed on 31 July 2019).
- Herrington, J.S. Solid-Phase Microextraction Liners for Headspace Volatile Organic Compounds. Column 2018, 14, 36–40. Available online: http://files.alfresco.mjh.group/alfresco_images/pharma//2019/03/07/aae3f1b3-af2d-4eb0-9d05-fdd7ed4bb372/LCTC121718%20North%20America.pdf (accessed on 31 July 2019).
- Herrington, J.S.; Myers, C.; Stidsen, G.; Kozel, S. Determination of Volatile Organic Compounds in Water (ISO 17943) with SPME Arrow. In Proceedings of the ExTech 2017, Santiago de Compostela, Spain, 27–30 June 2017. [Google Scholar]
- Cernosek, T.; Eckert, K.E.; Carter, D.O.; Perrault, A.P. Volatile Organic Compound Profiling from Postmortem Microbes using Gas Chromatography–Mass Spectrometry. J. Forensic Sci. 2020, 65, 134–143. [Google Scholar] [CrossRef] [PubMed]
- Hartonen, K.; Helin, A.; Parshintsev, J.; Riekkola, M.-L. Problems Caused by Moisture in Gas Chromatographic Analysis of Headspace SPME Samples of Short-Chain Amines. Chromatographia 2019, 82, 307–316. [Google Scholar] [CrossRef]
- Gionfriddo, E.; Passarini, A.; Pawliszyn, J. A facile and fully automated on-fiber derivatization protocol for direct analysis of short-chain aliphatic amines using a matrix compatible solid-phase microextraction coating. J. Chromatogr. A 2016, 1457, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Kremser, A.; Jochmann, M.A.; Schmidt, T.C. Systematic comparison of static and dynamic headspace sampling techniques for gas chromatography. Anal. Bioanal. Chem. 2016, 408, 6567–6579. [Google Scholar] [CrossRef] [PubMed]
- Wardencki, W.; Curyło, J.; Namieśnik, J. Trends in Solventless Sample Preparation Techniques for Environmental Analysis. J. Biochem. Biophys. Methods 2007, 70, 275–288. [Google Scholar] [CrossRef]
- Namieśnik, J.; Zygmunt, B.; Jastrzȩbska, A. Application of Solid-Phase Microextraction for Determination of Organic Vapours in Gaseous Matrices. J. Chromatogr. A 2000, 885, 405–418. [Google Scholar] [CrossRef]
- Flórez Menéndez, J.C.; Fernández Sánchez, M.L.; Sánchez Uría, J.E.; Fernández Martínez, E.; Sanz-Medel, A. Static Headspace, Solid-Phase Microextraction and Headspace Solid-Phase Microextraction for BTEX Determination in Aqueous Samples by Gas Chromatography. Anal. Chim. Acta 2000, 415, 9–20. [Google Scholar] [CrossRef]
- Jochmann, M.A.; Yuan, X.; Schilling, B.; Schmidt, T.C. In-Tube Extraction for Enrichment of Volatile Organic Hydrocarbons from Aqueous Samples. J. Chromatogr. A 2008, 1179, 96–105. [Google Scholar] [CrossRef]
- Laaks, J.; Jochmann, M.A.; Schilling, B.; Schmidt, T.C. In-Tube Extraction of Volatile Organic Compounds from Aqueous Samples: An Economical Alternative to Purge and Trap Enrichment. Anal. Chem. 2010, 82, 7641–7648. [Google Scholar] [CrossRef]
- Jakubowska, N.; Polkowska, Ż.; Kujawski, W.; Konieczka, P.; Namieśnik, J. A Comparison of Three Solvent-Free Techniques Coupled with Gas Chromatography for Determining Trihalomethanes in Urine Samples. Anal. Bioanal. Chem. 2007, 388, 691–698. [Google Scholar] [CrossRef] [PubMed]
- Meyer-Monath, M.; Beaumont, J.; Morel, I.; Rouget, F.; Tack, K.; Lestremau, F. Analysis of BTEX and Chlorinated Solvents in Meconium by Headspace-Solid-Phase Microextraction Gas Chromatography Coupled with Mass Spectrometry. Anal. Bioanal. Chem. 2014, 406, 4481–4490. [Google Scholar] [CrossRef] [PubMed]
- Schulz, K.; Dreßler, J.; Sohnius, E.-M.; Lachenmeier, D.W. Determination of Volatile Constituents in Spirits Using Headspace Trap Technology. J. Chromatogr. A 2007, 1145, 204–209. [Google Scholar] [CrossRef] [PubMed]
- German Standard Procedure DIN 38 407-41. German Standard Procedure NA 119-01-03-02 AK. German Standard Method Procedure 32465:2008–11. Available online: http://www.wasserchemische-gesellschaft.de/dev/validierungsdokumente?download=32:f41-din-38407-41-2011-06&lang=de (accessed on 16 January 2020).
- Jeleń, H.H.; Wlazły, K.; Wąsowicz, E.; Kamiński, E. Solid-Phase Microextraction for the Analysis of Some Alcohols and Esters in Beer: Comparison with Static Headspace Method. J. Agric. Food Chem. 1998, 64, 1469–1473. [Google Scholar] [CrossRef]
- Sriseadka, T.; Wongpornchai, S.; Kitsawatpaiboon, P. Rapid Method for Quantitative Analysis of the Aroma Impact Compound, 2-Acetyl-1-Pyrroline, in Fragrant Rice Using Automated Headspace Gas Chromatography. J. Agric. Food Chem. 2006, 54, 8183–8189. [Google Scholar] [CrossRef] [PubMed]
- Keith, L.H.; Crummett, W.; Deegan, J.; Libby, R.A.; Taylor, J.K.; Wentler, G. Principles of Environmental Analysis. Anal. Chem. 1983, 55, 2210–2218. [Google Scholar] [CrossRef]
- Ma, J.; Lu, W.; Li, J.; Song, Z.; Liu, D.; Chen, L. Determination of Geosmin and 2-Methylisoborneol in Water by Headspace Liquid-Phase Microextraction Coupled with Gas Chromatography-Mass Spectrometry. Anal. Lett. 2011, 44, 1544–1557. [Google Scholar] [CrossRef]
- Yu, S.; Xiao, Q.; Zhu, B.; Zhong, X.; Xu, Y.; Su, G.; Chen, M. Gas Chromatography–Mass Spectrometry Determination of Earthy–Musty Odorous Compounds in Waters by Two Phase Hollow-Fiber Liquid-Phase Microextraction Using Polyvinylidene Fluoride Fibers. J. Chromatogr. A 2014, 1329, 45–51. [Google Scholar] [CrossRef]
- Nakamura, S.; Nakamura, N.; Ito, S. Determination of 2-Methylisoborneol and Geosmin in Water by Gas Chromatography-Mass Spectrometry Using Stir Bar Sorptive Extraction. J. Sep. Sci. 2001, 24, 674–677. [Google Scholar] [CrossRef]
- Bagheri, H.; Salemi, A. Headspace Solvent Microextraction as a Simple and Highly Sensitive Sample Pretreatment Technique for Ultra Trace Determination of Geosmin in Aquatic Media. J. Sep. Sci. 2006, 29, 57–65. [Google Scholar] [CrossRef]
- Wu, D.; Duirk, S.E. Quantitative Analysis of Earthy and Musty Odors in Drinking Water Sources Impacted by Wastewater and Algal Derived Contaminants. Chemosphere 2013, 91, 1495–1501. [Google Scholar] [CrossRef] [PubMed]
- Boyaci, E.; Rodriguez-Lafuente, A.; Gorynski, K.; Mirnaghi, F.; Souza-Silva, E.A.; Hein, D.; Pawliszyn, J. Sample Preparation with Solid Phase Microextraction and Exhaustive Extraction Approaches: Comparison for Challenging Cases. Anal Chim Acta 2015, 873, 14–30. [Google Scholar] [CrossRef] [PubMed]
- Kremser, A.; Jochmann, M.A.; Schmidt, T.C. PAL SPME Arrow—Evaluation of a Novel Solid-Phase Microextraction Device for Freely Dissolved PAHs in Water. Anal. Bioanal. Chem. 2016, 408, 943–952. [Google Scholar] [CrossRef] [PubMed]
- Barreira, L.M.F.; Parshintsev, J.; Kärkkäinen, N.; Hartonen, K.; Jussila, M.; Kajos, M.; Kulmala, M.; Riekkola, M.-L. Field Measurements of Biogenic Volatile Organic Compounds in the Atmosphere by Dynamic Solid-Phase Microextraction and Portable Gas Chromatography-Mass Spectrometry. Atmos. Environ. 2015, 115, 214–222. [Google Scholar] [CrossRef]
- Cheng, X.; Forsythe, J.; Peterkin, E. Some Factors Affecting SPME Analysis and PAHs in Philadelphia’s Urban Waterways. Water Res. 2013, 47, 2331–2340. [Google Scholar] [CrossRef]
- Pérez-Carrera, E.; León, V.M.L.; Parra, A.G.; González-Mazo, E. Simultaneous Determination of Pesticides, Polycyclic Aromatic Hydrocarbons and Polychlorinated Biphenyls in Seawater and Interstitial Marine Water Samples, Using Stir Bar Sorptive Extraction–Thermal Desorption–Gas Chromatography–Mass Spectrometry. J. Chromatogr. A 2007, 1170, 82–90. [Google Scholar] [CrossRef]
- Nam, T.G.; Lee, J.-Y.; Kim, B.-K.; Song, N.-E.; Jang, H.W. Analyzing Volatiles in Brown Rice Vinegar by Headspace Solid-Phase Microextraction (SPME)–Arrow: Optimizing the Extraction Conditions and Comparisons with Conventional SPME. Int. J. Food Prop. 2019, 22, 1195–1204. [Google Scholar] [CrossRef]
- Song, N.-E.; Lee, J.-Y.; Lee, Y.-Y.; Park, J.-D.; Jang, H.W. Comparison of Headspace–SPME and SPME-Arrow–GC–MS Methods for the Determination of Volatile Compounds in Korean Salt–Fermented Fish Sauce. Appl. Biol. Chem. 2019, 62, 16. [Google Scholar] [CrossRef]
- Lan, H.; Rönkkö, T.; Parshintsev, J.; Hartonen, K.; Gan, N.; Sakeye, M.; Sarfraz, J.; Riekkola, M.-L. Modified Zeolitic Imidazolate Framework-8 as Solid-Phase Microextraction Arrow Coating for Sampling of Amines in Wastewater and Food Samples Followed by Gas Chromatography-Mass Spectrometry. J. Chromatogr. A 2017, 1486, 76–85. [Google Scholar] [CrossRef]
- Yuan, Y.; Lin, X.; Li, T.; Pang, T.; Dong, Y.; Zhuo, R.; Wang, Q.; Cao, Y.; Gan, N. A Solid Phase Microextraction Arrow with Zirconium Metal–Organic Framework/Molybdenum Disulfide Coating Coupled with Gas Chromatography–Mass Spectrometer for the Determination of Polycyclic Aromatic Hydrocarbons in Fish Samples. J. Chromatogr. A 2019. [Google Scholar] [CrossRef]
- Lan, H.; Zhang, W.; Smått, J.-H.; Koivula, R.T.; Hartonen, K.; Riekkola, M.-L. Selective Extraction of Aliphatic Amines by Functionalized Mesoporous Silica-Coated Solid Phase Microextraction Arrow. Microchim. Acta 2019, 186, 412. [Google Scholar] [CrossRef] [PubMed]
- Russo, E.B. Taming THC: Potential Cannabis Synergy and Phytocannabinoid-Terpenoid Entourage Effects. Br. J. Pharmacol. 2011, 163, 1344–1364. [Google Scholar] [CrossRef] [PubMed]
- Halpenny, M.; Stenerson, K.K. Quantitative Determination of Terpenes in Cannabis Using Headspace Solid Phase Microextraction and GC/MS. Available online: https://www.gerstel.com/pdf/AppNote-189.pdf (accessed on 31 July 2019).
- Huba, A.K.; Mirabelli, M.F.; Zenobi, R. High-Throughput Screening of PAHs and Polar Trace Contaminants in Water Matrices by Direct Solid-Phase Microextraction Coupled to a Dielectric Barrier Discharge Ionization Source. Anal. Chim. Acta 2018, 1030, 125–132. [Google Scholar] [CrossRef]
- Gionfriddo, E.; Boyacı, E.; Pawliszyn, J. New Generation of Solid-Phase Microextraction Coatings for Complementary Separation Approaches: A Step toward Comprehensive Metabolomics and Multiresidue Analyses in Complex Matrices. Anal. Chem. 2017, 89, 4046–4054. [Google Scholar] [CrossRef]
- Gomez-Rios, G.A. High Throughput Analysis for On-Site Sampling. MSc. Thesis, University of Waterloo, Waterloo, ON, Canada, November 2012. [Google Scholar]
- Stilo, F.; Cordero, C.; Sgorbini, B.; Bicchi, C.; Liberto, E.; Stilo, F.; Cordero, C.; Sgorbini, B.; Bicchi, C.; Liberto, E. Highly Informative Fingerprinting of Extra-Virgin Olive Oil Volatiles: The Role of High Concentration-Capacity Sampling in Combination with Comprehensive Two-Dimensional Gas Chromatography. Separations 2019, 6, 34. [Google Scholar] [CrossRef]
- Bee, M.Y.; Jastrzembski, J.A.; Sacks, G.L. Parallel Headspace Extraction onto Etched Sorbent Sheets Prior to Ambient-Ionization Mass Spectrometry for Automated, Trace-Level Volatile Analyses. Anal. Chem. 2018, 90, 13806–13813. [Google Scholar] [CrossRef]
- Rafson, J.P.; Bee, M.Y.; Sacks, G.L. Spatially Resolved Headspace Extractions of Trace-Level Volatiles from Planar Surfaces for High-Throughput Quantitation and Mass Spectral Imaging. J. Agric. Food Chem. 2019, 67, 13840–13847. [Google Scholar] [CrossRef]
- Vasiljevic, T.; Gómez-Ríos, G.A.; Pawliszyn, J. Single-Use Poly(Etheretherketone) Solid-Phase Microextraction—Transmission Mode Devices for Rapid Screening and Quantitation of Drugs of Abuse in Oral Fluid and Urine via Direct Analysis in Real-Time Tandem Mass Spectrometry. Anal. Chem. 2018, 90, 952–960. [Google Scholar] [CrossRef]
- Mirabelli, M.F.; Zenobi, R. Solid-Phase Microextraction Coupled to Capillary Atmospheric Pressure Photoionization-Mass Spectrometry for Direct Analysis of Polar and Nonpolar Compounds. Anal. Chem. 2018, 90, 5015–5022. [Google Scholar] [CrossRef]
- Wang, C.H.; Su, H.; Chou, J.H.; Huang, M.Z.; Lin, H.J.; Shiea, J. Solid Phase Microextraction Combined with Thermal-Desorption Electrospray Ionization Mass Spectrometry for High-Throughput Pharmacokinetics Assays. Anal. Chim. Acta 2018, 1021, 60–68. [Google Scholar] [CrossRef]
- Hu, B.; Zheng, B.; Rickert, D.; Gómez-Ríos, G.A.; Bojko, B.; Pawliszyn, J.; Yao, Z.-P. Direct Coupling of Solid Phase Microextraction with Electrospray Ionization Mass Spectrometry: A Case Study for Detection of Ketamine in Urine. Anal. Chim. Acta 2019, 1075, 112–119. [Google Scholar] [CrossRef]
- Lendor, S.; Gómez-Ríos, G.A.; Boyacı, E.; Vander Heide, H.; Pawliszyn, J. Space-Resolved Tissue Analysis by Solid-Phase Microextraction Coupled to High-Resolution Mass Spectrometry via Desorption Electrospray Ionization. Anal. Chem. 2019, 91, 10141–10148. [Google Scholar] [CrossRef] [PubMed]
- Tascon, M.; Alam, M.N.; Gómez-Ríos, G.A.; Pawliszyn, J. Development of a Microfluidic Open Interface with Flow Isolated Desorption Volume for the Direct Coupling of SPME Devices to Mass Spectrometry. Anal. Chem. 2018, 90, 2631–2638. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Ríos, G.A.; Liu, C.; Tascon, M.; Reyes-Garcés, N.; Arnold, D.W.; Covey, T.R.; Pawliszyn, J. Open Port Probe Sampling Interface for the Direct Coupling of Biocompatible Solid-Phase Microextraction to Atmospheric Pressure Ionization Mass Spectrometry. Anal. Chem. 2017, 89. [Google Scholar] [CrossRef] [PubMed]
- Looby, N.T.; Tascon, M.; Acquaro, V.R.; Reyes-Garcés, N.; Vasiljevic, T.; Gomez-Rios, G.A.; Wąsowicz, M.; Pawliszyn, J. Solid Phase Microextraction Coupled to Mass Spectrometry via a Microfluidic Open Interface for Rapid Therapeutic Drug Monitoring. Analyst 2019, 144, 3721–3728. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Ríos, G.A.; Reyes-Garcés, N.; Bojko, B.; Pawliszyn, J. Biocompatible Solid-Phase Microextraction Nanoelectrospray Ionization: An Unexploited Tool in Bioanalysis. Anal. Chem. 2016, 88, 1259–1265. [Google Scholar] [CrossRef]
- Gómez-Ríos, G.A.; Vasiljevic, T.; Gionfriddo, E.; Yu, M.; Pawliszyn, J. Towards On-Site Analysis of Complex Matrices by Solid-Phase Microextraction-Transmission Mode Coupled to a Portable Mass Spectrometer via Direct Analysis in Real Time. Analyst 2017, 142, 2928–2935. [Google Scholar] [CrossRef]
- Feider, C.L.; Krieger, A.; DeHoog, R.J.; Eberlin, L.S. Ambient Ionization Mass Spectrometry: Recent Developments and Applications. Anal. Chem. 2019, 91, 4266–4290. [Google Scholar] [CrossRef]
- Prabhu, G.R.D.; Urban, P.L. The Dawn of Unmanned Analytical Laboratories. TrAC Trends Anal. Chem. 2017, 88, 41–52. [Google Scholar] [CrossRef]
- Trujillo-Rodriguez, M.J.; Pino, V.; Psillakis, E.; Anderson, J.L.; Ayala, J.H.; Yiantzi, E.; Afonso, A.M. Vacuum-Assisted Headspace-Solid Phase Microextraction for Determining Volatile Free Fatty Acids and Phenols. Investigations on the Effect of Pressure on Competitive Adsorption Phenomena in a Multicomponent System. Anal. Chim. Acta 2017, 962, 41–51. [Google Scholar] [CrossRef]
- Zhang, S.; Yang, Q.; Wang, W.; Wang, C.; Wang, Z. Covalent Bonding of Metal-Organic Framework-5/Graphene Oxide Hybrid Composite to Stainless Steel Fiber for Solid-Phase Microextraction of Triazole Fungicides from Fruit and Vegetable Samples. J. Agric. Food Chem. 2016, 64, 2792–2801. [Google Scholar] [CrossRef]
- Song, X.-Y.; Chen, J.; Shi, Y.-P. Different Configurations of Carbon Nanotubes Reinforced Solid-Phase Microextraction Techniques and Their Applications in the Environmental Analysis. TrAC Trends Anal. Chem. 2017, 86, 263–275. [Google Scholar] [CrossRef]
- Kalsoom, U.; Nesterenko, P.N.; Paull, B. Current and Future Impact of 3D Printing on the Separation Sciences. TrAC Trends Anal. Chem. 2018, 105, 492–502. [Google Scholar] [CrossRef]
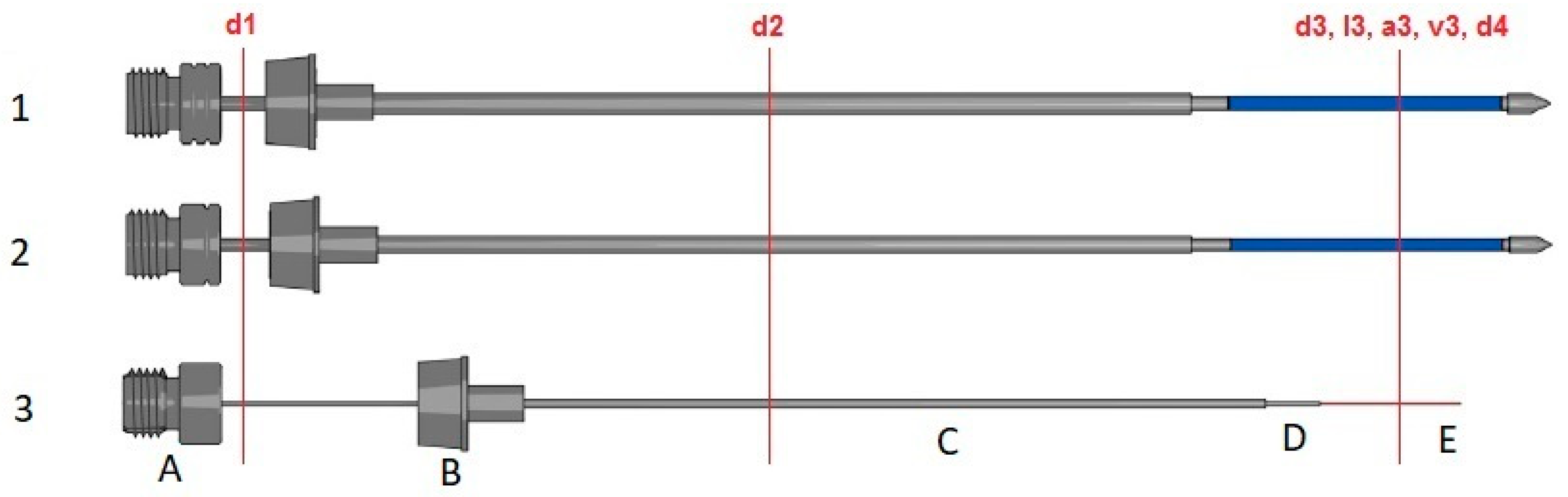



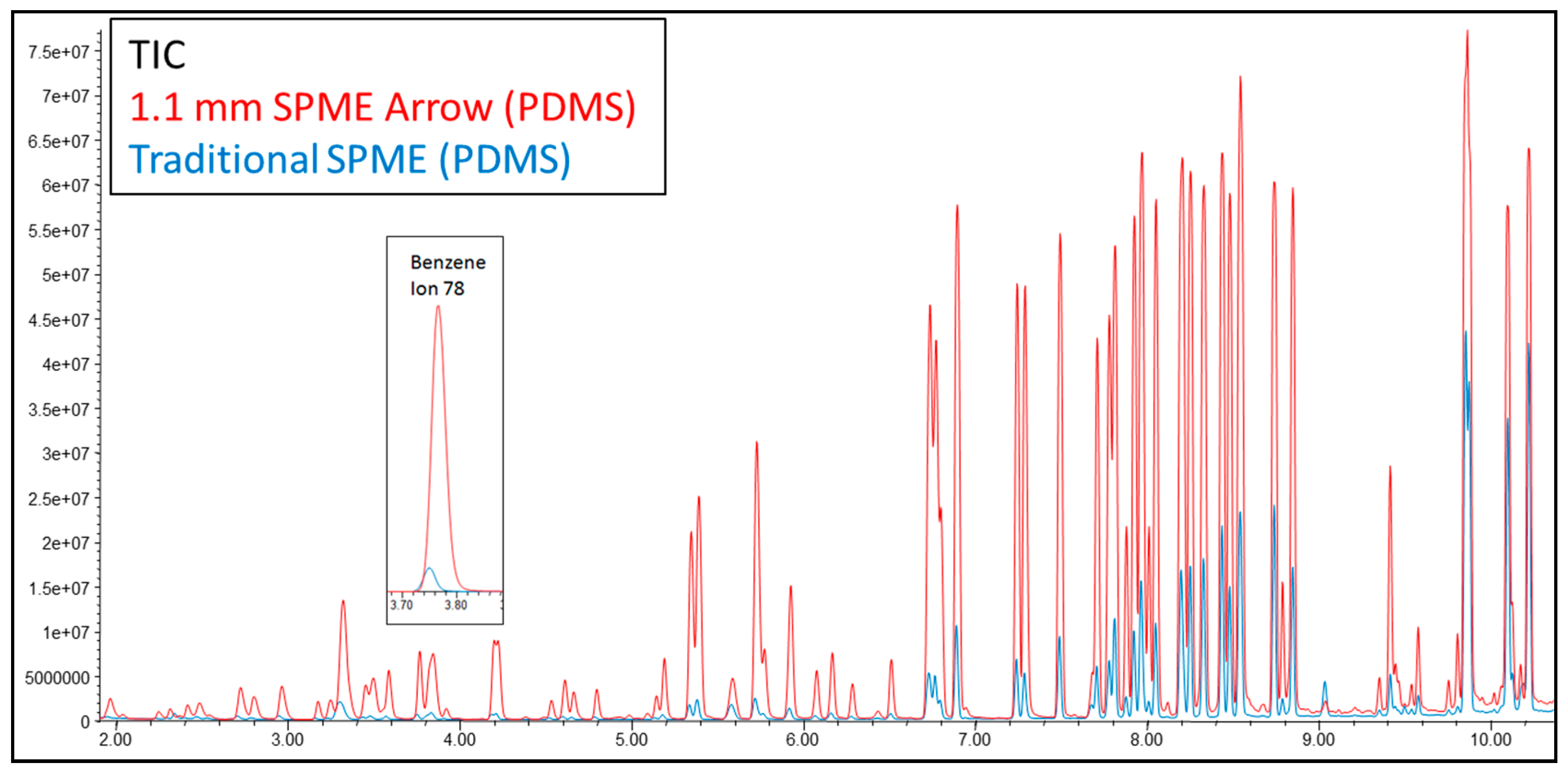
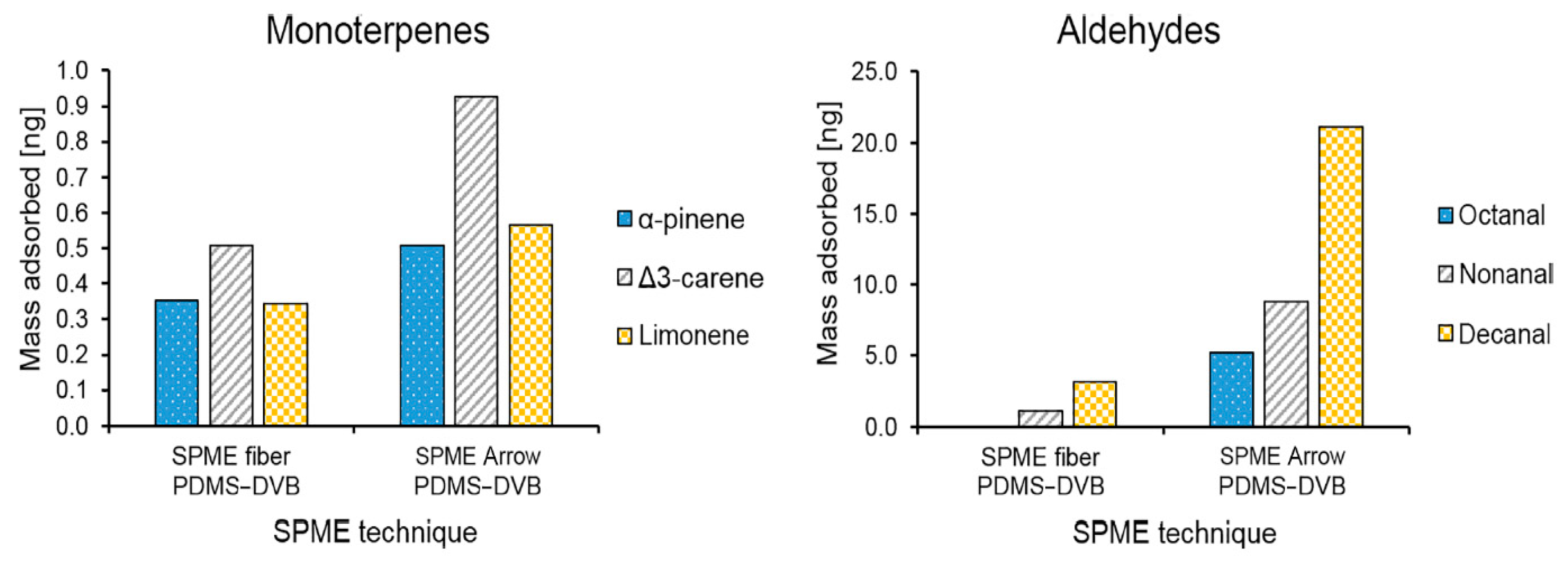
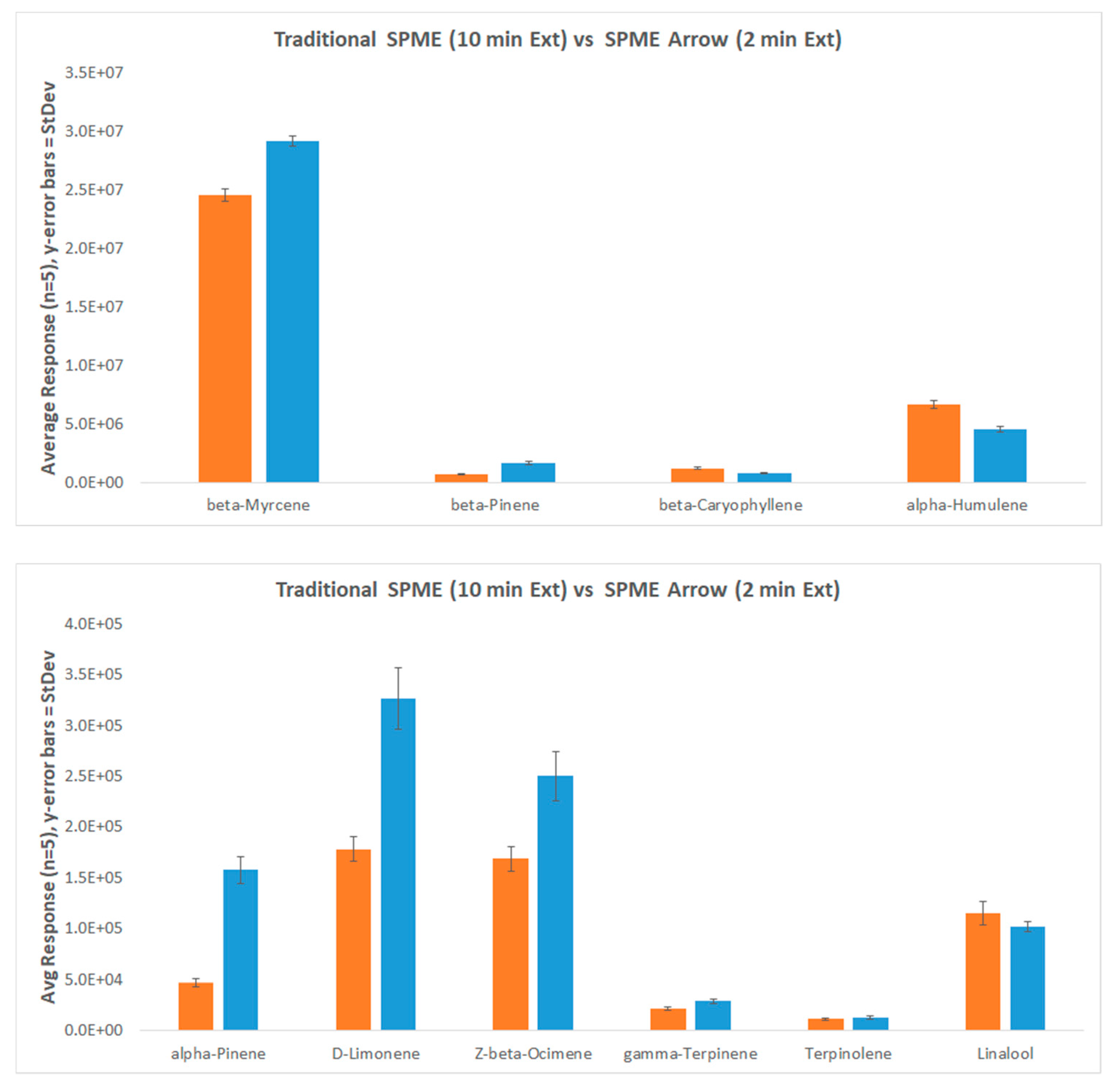
| Label | Description | Units | Traditional SPME | 1.1 mm SPME Arrow | 1.5 mm SPME Arrow | 1.1 mm% Increase | 1.5 mm% Increase |
|---|---|---|---|---|---|---|---|
| d1 | Support tubing | mm | 0.304 | 0.804 | 1.01 | 264 | 332 |
| d2 | Septum piercing needle | mm | 0.634 | 1.10 | 1.50 | 174 | 237 |
| d3 | Phase diameter | mm | 0.285 | 0.721 | 0.912 | 253 | 320 |
| d4 | Phase support tubing diameter | mm | 0.111 | 0.647 | 0.498 | 583 | 449 |
| l3 | Phase length | mm | 10.0 | 20.0 | 20.0 | 200 | 200 |
| a3 | Phase area | mm² | 9.40 | 44.0 | 62.8 | 468 | 668 |
| v3 | Phase volume | µL | 0.600 | 3.80 | 11.8 | 633 | 1967 |
| Analytes | Molecular Weight * | Stationary Phase | Thickness (µm) | Needle Diameter (mm) |
|---|---|---|---|---|
| Volatile | 60–275 | Polydimethylsiloxane (PDMS) | 100 | 1.1 and 1.5 * |
| Volatile (high capacity) | 60–275 | PDMS | 250 | 1.5 |
| Polar, semi-volatile | 80–300 | Polyacrylate | 100 | 1.1 |
| Very volatile | 30–225 | Carbon Wide Range (WR)/PDMS | 120 | 1.1 and 1.5 * |
| Aromatic, semi-volatile | 60–300 | Divinylbenzene (DVB)/PDMS | 120 | 1.1 and 1.5 * |
| Volatile and semi-volatile | 40–275 | DVB/Carbon WR/PDMS | 120 | 1.1 and 1.5 * |
| Method Name | Sources with Reported Detection Limits in ng L−1 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| This Work | [30] | [31] | [32] | [33] | [34] | [35] | [36] | [37] | [38] | [39] | [40] | |
| Syringe | 25–143 | / | / | 1–2 × 103 | / | / | 25–53 | / | 66–570 × 103 | / | / | / |
| Loop | 25–168 | / | / | / | / | / | / | / | / | / | 8–20 × 103 | 5–20 × 103 |
| SPME | 0.5–79.6 | 5.0–50 | 2.0–550 | 80–600 | / | / | / | 8.0–12 | / | / | 2–26 × 103 | / |
| PAL SPME Arrow | 0.7–4.9 | / | / | / | / | / | / | / | / | / | / | / |
| Trap | 0.6–9.6 | / | / | / | / | 1.0–10 | / | / | 7–149 × 103 | 0.5–91 | / | / |
| ITEX | 0.9–9.1 | / | / | / | 28–799 | 1.0–70 | / | / | / | / | / | / |
| Sample matrix | Lab Water | Spirit | Wastewater | Water | Lab water | Lab water | Urine | Meconium | Spirit | Water | Beer | Wet rice |
| MDL method | a | b | NS | b | a | a | NS | NS | c | d | e | b |
| Method Name | Analytes | Sample Volume (mL) | Calibration Range (ng L−1) | LOD (ng L−1) | RSD (%) | Ref. |
|---|---|---|---|---|---|---|
| Purge and trap | IPMP, IBMP, GSM, MIB, TCA | 20 | 10–200 | 0.2–2 | <8 | [42] |
| Solvent microextraction | GSM | 5 | 5–900 | 0.8 | <5 | [43] |
| SBSE | GSM, MIB, TCA | 60 | 0.1–100 | 0.02–0.16 | <3.7 | [44] |
| DLLME | GSM, MIB | 12 | 10–1000 | 2 and 9 | <11 | [45] |
| SPME | IPMP, GSM, MIB, TCA, BIN | 40 | 5–100 | 0.2–0.5 | <7 | [46] |
| PAL SPME Arrow | IPMP, IBMP, GSM, MIB, TCA, BIN, TBA | 10 | 1 (2.6)–1000 (2600) | 0.05–0.6 | <11 | - |
| Compound | PAL SPME Arrow | SPME (Cheng et al.) [50] | SBSE (Carrera et al.) [51] | |||
|---|---|---|---|---|---|---|
| MDL (ng L−1) | RSD (%) (at 10 ng L−1) | LOD (SD × 3) | RSD (conc. at S/N = 3 × 3) | LOD (conc. at S/N = 3 × 3) | RSD (%) (at 50 ng L−1) | |
| Naphthalene | 0.3 | 5.7 | 2.7 | 9 | / | / |
| Acenaphthylene | 0.2 | 6 | 1.8 | 6 | 0.1 | / |
| Acenaphthene | 0.1 | 7.1 | 0.9 | 3 | / | / |
| Fluorene | 0.2 | 5.6 | 3 | 10 | 0.1 | 8.3 |
| Phenanthrene | 0.2 | 5.5 | 2.1 | 7 | 0.1 | 1.1 |
| Anthracene | 0.3 | 7.6 | 2.1 | 7 | 0.2 | 2.1 |
| Pyrene | 0.2 | 6.4 | 3.6 | 12 | 0.2 | / |
| Fluoranthene | 0.2 | 6.2 | 2.1 | 7 | 0.2 | / |
| 1,2-Benzanthracene | 0.1 | 6.2 | 2.1 | 7 | 0.2 | 6 |
| Chrysene | 0.1 | 11 | 1.5 | 5 | 0.2 | 10.6 |
| Benzo(b)fluoranthene | 0.2 | 10.5 | 2.7 | 9 | 0.1 | / |
| Benzo(k)fluoranthene | 0.2 | 8.6 | 1.8 | 6 | 0.1 | / |
| Benzo[a]pyrene | 0.3 | 7.2 | 3.6 | 12 | 0.1 | / |
| Indeno(1,2,3 cd)pyrene | 0.8 | 9.2 | 3.6 | 12 | 0.3 | / |
| Dibenz(ah)anthracene | 0.6 | 11.3 | / | / | 0.3 | / |
| Benzo(ghi)perylene | 0.8 | 11.9 | 1.8 | 6 | 0.3 | / |
| MDL values calculated with a 99% confidence interval/not determined | ||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Herrington, J.S.; Gómez-Ríos, G.A.; Myers, C.; Stidsen, G.; Bell, D.S. Hunting Molecules in Complex Matrices with SPME Arrows: A Review. Separations 2020, 7, 12. https://doi.org/10.3390/separations7010012
Herrington JS, Gómez-Ríos GA, Myers C, Stidsen G, Bell DS. Hunting Molecules in Complex Matrices with SPME Arrows: A Review. Separations. 2020; 7(1):12. https://doi.org/10.3390/separations7010012
Chicago/Turabian StyleHerrington, Jason S., German A. Gómez-Ríos, Colton Myers, Gary Stidsen, and David S. Bell. 2020. "Hunting Molecules in Complex Matrices with SPME Arrows: A Review" Separations 7, no. 1: 12. https://doi.org/10.3390/separations7010012
APA StyleHerrington, J. S., Gómez-Ríos, G. A., Myers, C., Stidsen, G., & Bell, D. S. (2020). Hunting Molecules in Complex Matrices with SPME Arrows: A Review. Separations, 7(1), 12. https://doi.org/10.3390/separations7010012





