Recycling of 1,2-Dimethyl-3-propylimidazolium bis(trifluoromethylsulfonyl)imide Ionic Liquid by Stacked Cation and Anion Exchange Adsorption-Desorption
Abstract
1. Introduction
2. Experimental
3. Results and Discussion
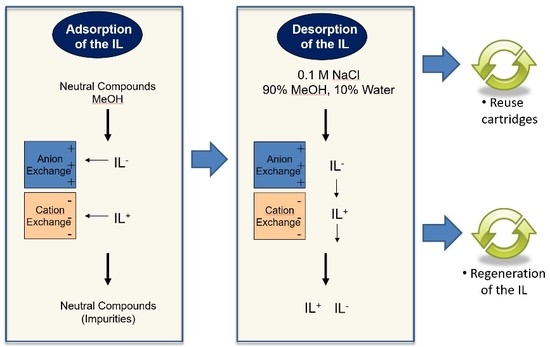
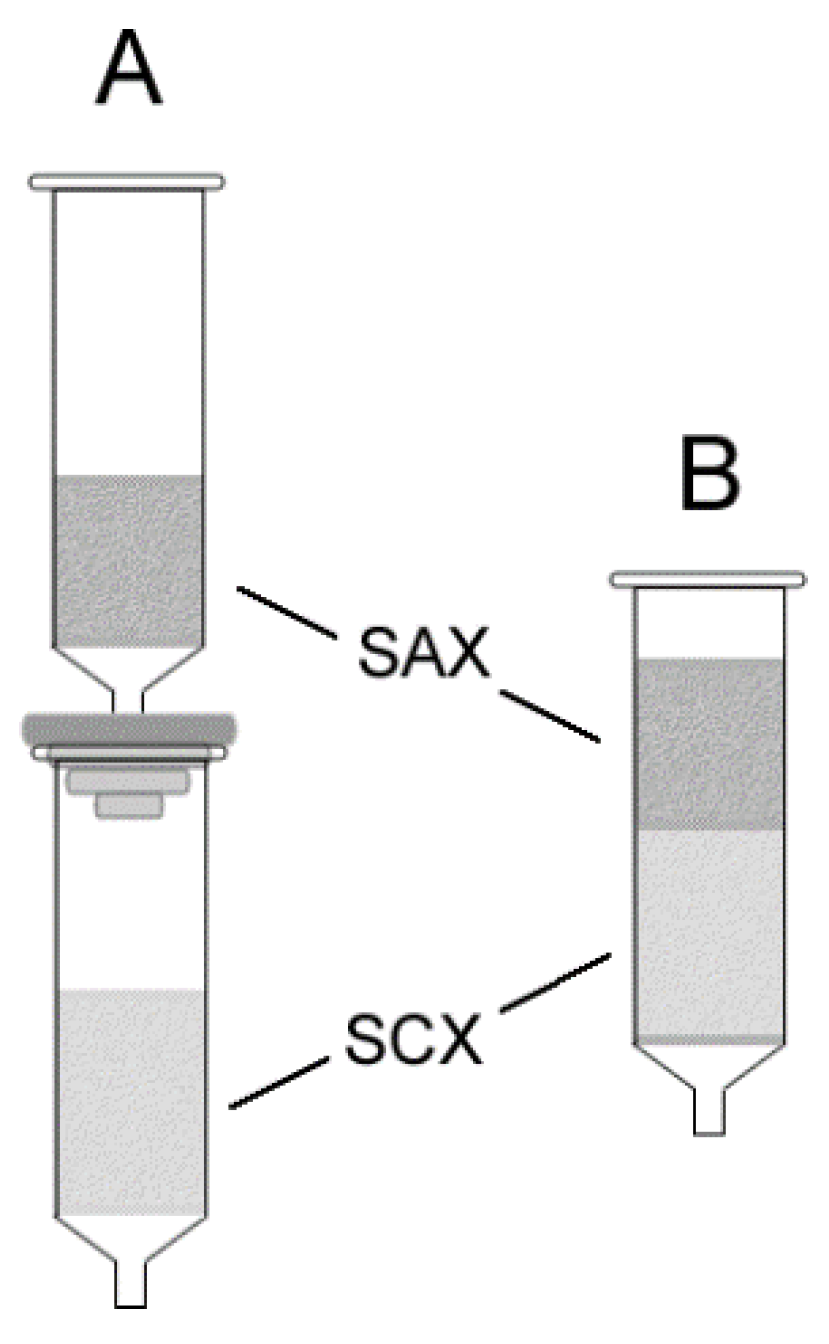
3.1. Column Configuration
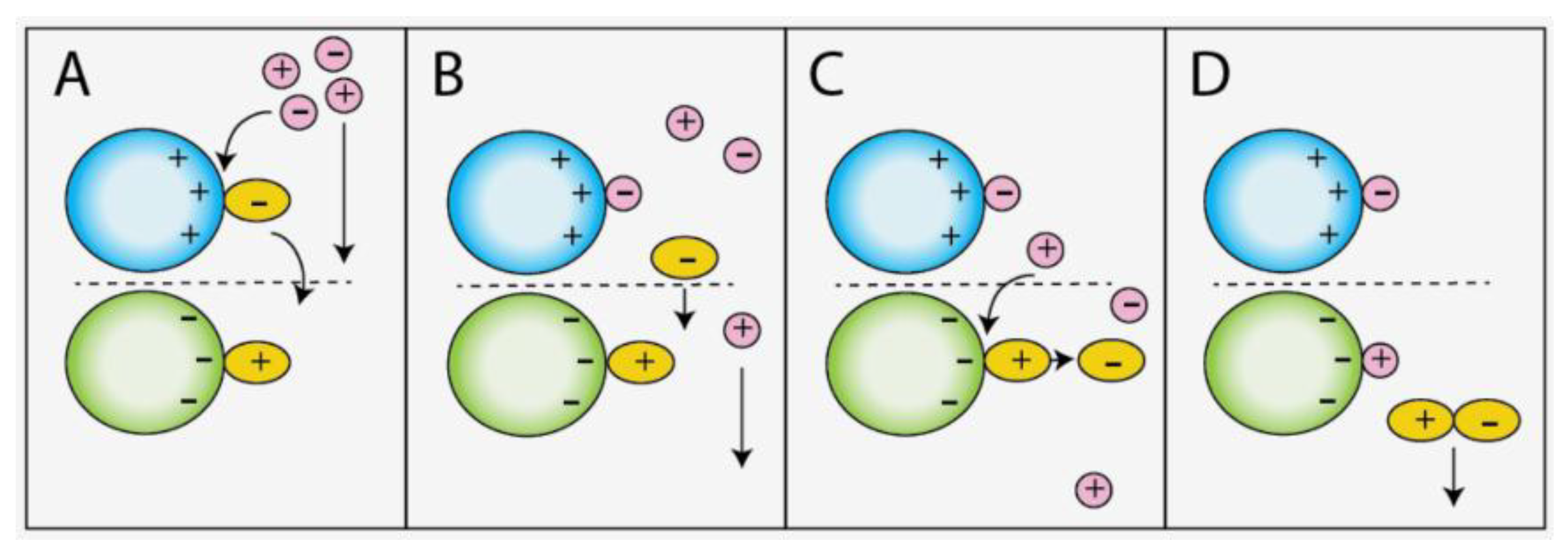
3.2. Sorbent Order
3.3. Eluent Composition
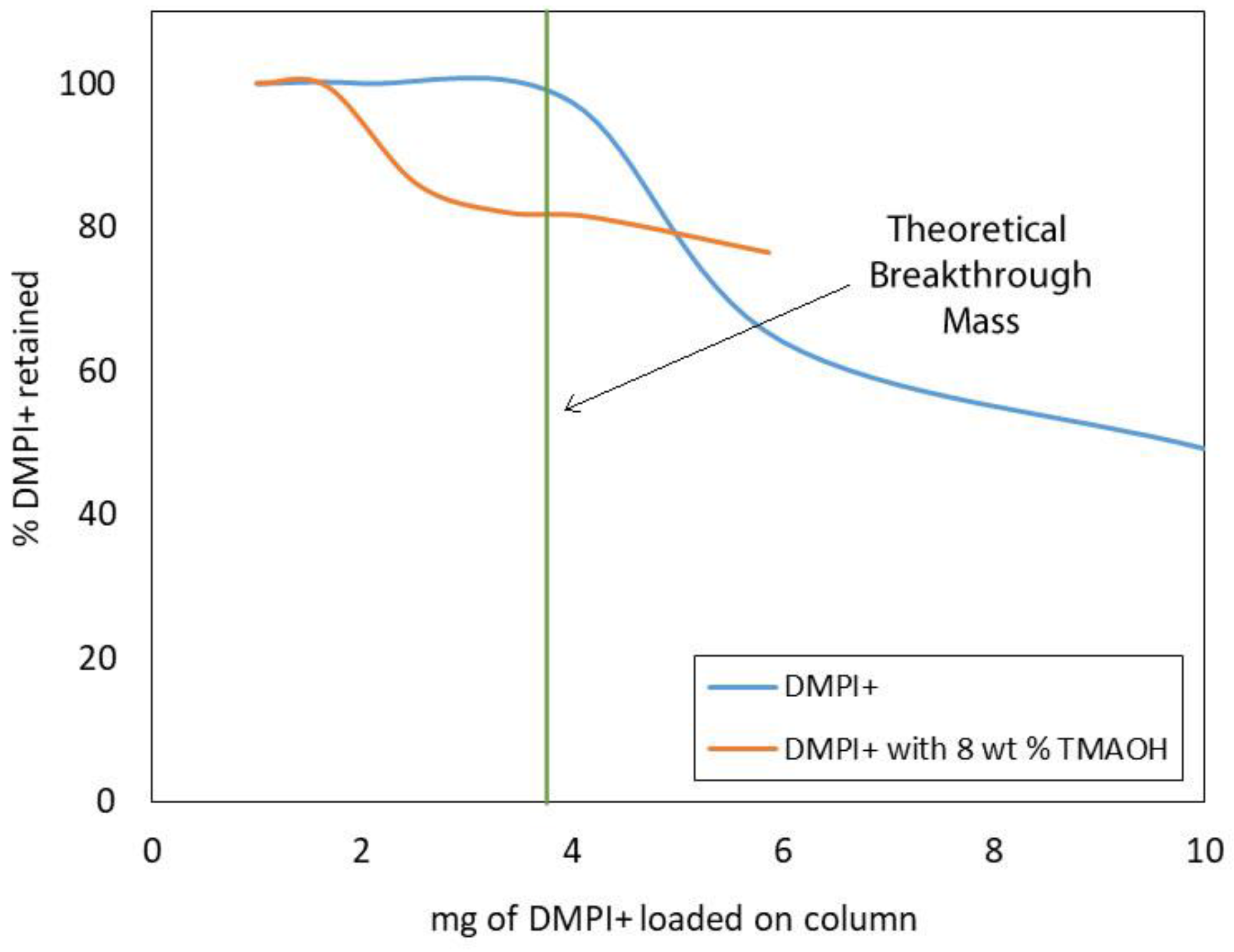
3.4. SAX/SCX Ion Exchange Capacity
3.5. Recovery and Recycling of the Ionic Liquid
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Walden, P. Ueber die Molekulargrösse und electrische Leitfähigkeit einiger geschmolzenen Salze. Bulletin de l’Academie Imperiale des Sciences de St. Petersbourg 1914, 8, 405–422. [Google Scholar]
- Wilkes, J.S.; Levisky, J.A.; Wilson, R.A.; Hussey, C.L. Dialkylimidazolium chloroaluminate melts: A new class of room-temperature ionic liquids for electrochemistry, spectroscopy and synthesis. Inorg. Chem. 1982, 21, 1263–1264. [Google Scholar] [CrossRef]
- Koel, M. Ionic Liquids in Chemical Analysis; CRC Press: Boca Raton, FL, USA, 2009. [Google Scholar]
- Del Sesto, R.E.; Corley, C.; Robertson, A.; Wilkes, J. Tetraalkylphosphonium-based ionic liquids. J. Organomet. Chem. 2005, 690, 2536–2542. [Google Scholar] [CrossRef]
- Lei, Z.; Chen, B.; Koo, Y.; MacFarlane, D.R. Introduction: Ionic Liquids. Chem. Rev. 2017, 117, 6633–6635. [Google Scholar] [CrossRef] [PubMed]
- Welton, T. Ionic liquids: A brief history. Biophys. Rev. 2018, 10, 691–706. [Google Scholar] [CrossRef]
- Fischer, T.; Sethi, A.; Welton, T.; Woolf, J. Diels-Alder reactions in room-temperature ionic liquids. Tetrahedron Lett. 1999, 40, 793–796. [Google Scholar] [CrossRef]
- Earle, M.J.; McCormac, P.B.; Seddon, K.R. Diels-Alder reactions in ionic liquids. A safe recyclable alternative to lithium perchlorate-diethyl ether mixture. Green Chem. 1999, 1, 23–25. [Google Scholar] [CrossRef]
- Earle, M.J.; McCormac, P.B.; Seddon, K.R. Regioselective alkylation in ionic liquids. Chem. Commun. 1998, 2245–2246. [Google Scholar] [CrossRef]
- Ramakoteswararao, P.; Tulasi, S.L.; Pavani, Y. Impacts of solvents on Environmental Pollution. JCHPS 2014, 3, 132–135. [Google Scholar]
- Ikeda, M. Public health problems of organic solvents. Toxicol. Lett. 1992, 64–65, 191–201. [Google Scholar] [CrossRef]
- Mallakpour, S.; Dinari, M. Green Solvents II: Properties and Applications of Ionic Liquids. In Ionic Liquids as Green Solvents: Progress and Prospects; Springer: New York, NY, USA, 2012; Chapter 1. [Google Scholar]
- Shaikh, M.A.; Huang, X. Green Solvents II: Properties and Applications of Ionic Liquids. In Organic Ionic Liquids: Ultimate Green Solvents in Organic Synthesis; Springer: New York, NY, USA, 2012; Chapter 17. [Google Scholar]
- Halett, J.P.; Welton, T. Room-Temperature Ionic Liquids: Solvents for Synthesis and Catalysis. Chem. Rev. 2011, 111, 3508–3576. [Google Scholar] [CrossRef] [PubMed]
- Przemyslaw, P. Application of Ionic Liquids as Solvents for Polymerization Processes. Prog. Polym. Sci. 2014, 29, 3–12. [Google Scholar]
- Bailey, M.M.; Jernigan, M.B.; Jennigan, P.L.; Sturdivant, J.; Hought-Toutman, W.L.; Rasco, J.F.; Swatloski, R.P.; Rogers, R.D.; Hood, R.D. Developmental toxicity assessment of the ionic liquid 1-butyl-3-methylimidazolium chloride in CD-1 mice. Green Chem. 2008, 10, 1213–1217. [Google Scholar] [CrossRef]
- Siedlecka, E.M.; Czerwicka, M.; Neumann, J.; Stepnowski, P.; Fernandez, J.F.; Thoming, J. Ionic Liquids: Theory, Properties, New Approaches. In Ionic Liquids: Methods of Degradation and Recovery; InTech: London, UK, 2011. [Google Scholar]
- Maton, C.; Vos, N.D.; Stevens, C. Ionic liquid thermal stabilities: Decomposition mechanisms and analysis tools. Chem. Soc. Rev. 2013, 42, 5963–5977. [Google Scholar] [CrossRef] [PubMed]
- Wooster, T.J.; Johanson, K.M.; Fraser, K.J.; MacFarlane, D.R.; Scott, J.L. Thermal degradation of cyano containing ionic liquids. Green Chem. 2006, 8, 691–696. [Google Scholar] [CrossRef]
- Zhou, J.; Sui, H.; Jia, Z.; Yang, Z.; He, L.; Li, X. Recovery and purification of ionic liquids from solutions: A review. RSC Adv. 2018, 8, 32832–32864. [Google Scholar] [CrossRef]
- Kim, Y.; Hendrickson, R.; Mosier, N.S.; Ladish, M.R. Liquid hot pretreatment of cel- lulosic biomass. Methods Mol. Biol. 2009, 581, 93–102. [Google Scholar] [PubMed]
- Balan, V. Current challenges in commercially producing biofuels from lignocellulosic biomass. ISRN Biotechnol. 2014, 463074. [Google Scholar] [CrossRef] [PubMed]
- Klein-Marcuschamer, D.; Simmons, B.A.; Blanch, H.W. Techno-economic analysis of a lignocellulosic ethanol biorefi nery with ionic liquid pre-treatment. Biofuel. Bioprod. Bioref. 2011, 5, 562. [Google Scholar] [CrossRef]
- Chen, L.; Sharifzadeh, M.; MacDowell, N.; Welton, T.; Shah, N.; Halett, J.P. Inexpensive ionic liquids: [HSO4]−-based solvent production at bulk scale. Green Chem. 2014, 16, 3098. [Google Scholar] [CrossRef]
- Sirwardana, A.I. Industrial Applications of Ionic Liquids. In Electrochemistry in Ionic Liquids; Torriero, A., Ed.; Springer: Cham, Switzerland, 2015. [Google Scholar]
- Plechkova, N.V.; Seddon, K.R. Applications of ionic liquids in the chemical industry. Chem. Soc. Rev. 2008, 37, 123–150. [Google Scholar] [CrossRef] [PubMed]
- Claire, B.; Sirwardana, A.; MacFarlane, D.R. Synthesis, purification and characterization of ionic liquids. Top. Curr. Chem. 2010, 290, 1–40. [Google Scholar]
- Seddon, K.R.; Stark, A.; Torres, M. Influence of chloride, water, and organic solvents on the physical properties of ionic liquids. Pure Appl. Chem. 2000, 72, 2275–2287. [Google Scholar] [CrossRef]
- Ekalavounos, E.; Helminen, J.; Kyllonen, L.; Kilpelainen, I.; King, A. Ionic Liquids: Recycling. In Encyclopedia of Inorganic and Bioinorganic Chemistry; John Wiley and Sons: Hoboken, NJ, USA, 2016. [Google Scholar]
- Abu-Eishah, S.I. Ionic Liquids Recycling for Reuse. Available online: https://www.intechopen.com/books/ionic-liquids-classes-and-properties/ionic-liquids-recycling-for-reuse (accessed on 10 October 2011).
- Thermal, E.; Mills, M.S. Solid-Phase Extraction: Principles and Practice; John Wiley and Sons: New York, NY, USA, 1998. [Google Scholar]
- Palomar, J.; Lemus, J.; Gilarranz, M.A.; Rodriguez, J.J. Adsorption of ionic liquids from aqueous effluents by activated carbon. Carbon 2009, 47, 1846–1856. [Google Scholar] [CrossRef]
- Lin, K.A.; Wu, C. Efficient and recyclable removal of imidazolium ionic liquids from water using resorcinol–formaldehyde polymer resin. RSC Adv. 2016, 6, 68111–68119. [Google Scholar]
- Lemus, J.; Palomar, J.; Heras, F.; Gilarranz, M.A.; Rodriguez, J.M. Characterization of Supported Ionic Liquid Phase (SILP) materials prepared from different supports. Adsorption 2011, 17, 561–571. [Google Scholar] [CrossRef]
- Ma, C.; Zu, Y.; Yang, L.; Li, J. Two solid-phase recycling method for basic ionic liquid [C4mim]Ac by macroporous resin and ion exchange resin from Schisandra chinensis fruits extract. J. Chromatogr. B 2015, 976–977, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Faizee, N.; Rao, K.; Dadi, A.P.; Varanasi, S.; Schall, C.A. Ion Exchange Process For the Recovery of Ionic Liquids. In Proceedings of the 2007 Annual Meeting, Salt Lake City, UT, USA, 7 November 2007. [Google Scholar]
- Sui, H.; Zhou, J.; Ma, G.; Niu, Y.; Cheng, J.; He, L.; Li, X. Removal of Ionic Liquids from Oil Sands Processing solution by Ion-Exchange Resin. Appl. Sci. 2018, 8, 1611. [Google Scholar] [CrossRef]
- Anthony, J.; Magnin, E.J.; Brennecke, J.F. Solution Thermodynamics of Imidazolium-Based Ionic Liquids and Water. J. Phys. Chem. B 2001, 105, 10942–10949. [Google Scholar] [CrossRef]
- Ventura, S.P.M.; Silva, F.A.; Quental, M.V.; Mondal, D.; Freire, M.G.; Coutinho, J.A.P. Ionic-Liquid-Mediated Extraction and Separation Processes for Bioactive Compounds: Past, Present, and Future Trends. Chem. Rev. 2017, 117, 6984–7052. [Google Scholar] [CrossRef]
- Gorman-Lewis, P.J.; Fen, J.B. Experimental Study of the Adsorption of an Ionic Liquid onto Bacterial and Mineral Surfaces. Environ. Sci. Technol. 2004, 38, 2491–2495. [Google Scholar] [CrossRef] [PubMed]
- Stepnowski, P. Preliminary Assessment of the Sorption of some Alkyl Imidazolium Cations as used in Ionic Liquids to Soils and Sediments. Aust. J. Chem. 2005, 58, 170–173. [Google Scholar] [CrossRef]
- Beaulieu, J.J.; Tank, J.L.; Kopacz, M. Sorption of imidazolium-based ionic liquids to aquatic sediments. Chemosphere 2008, 70, 1320–1328. [Google Scholar] [CrossRef] [PubMed]
- Binder, J.B.; Raines, R.T. Fermentable sugars by chemicalhydrolysis of biomass. Proc. Natl. Acad. Sci. USA 2010, 107, 4516–4521. [Google Scholar] [CrossRef]
- He, A.; Dong, B.; Feng, X.; Yao, S. Recovery of benzothiazolium ionic liquids from the coexisting glucose by ion-exchange resins. J. Mol. Liq. 2017, 227, 178–183. [Google Scholar] [CrossRef]
- Corley, C.A.; Iacono, S.T. A rapid approach to isolating nitro-explosives from imidazolium and pyrrolidinium ionic liquid solutions using solid phase extraction (SPE). Anal. Methods 2015, 7, 6911–6915. [Google Scholar] [CrossRef]
- Watanabe, M.; Thomas, M.; Zhang, S.; Uedo, K.; Tomohiro, Y.; Dokko, K. Application of Ionic Liquids to Energy Storage and Conversion Materials and Devices. Chem. Rev. 2017, 117, 7190–7239. [Google Scholar] [CrossRef]
- Fletcher, S.I.; Sillars, F.B.; Hudson, N.E.; Hall, P.J. Physical Properties of Selected Ionic Liquids for Use as Electrolytes and Other Industrial Applications. J. Chem. Eng. Data 2010, 55, 778–782. [Google Scholar] [CrossRef]
- Seki, S.; Kobayashi, Y.; Miyashiro, H.; Ohno, Y.; Usami, A.; Mita, Y.; Kihira, N.; Watanabe, M.; Terada, N. Lithium secondary batteries using modified-imidazolium room-temperature ionic liquid. J. Phys. Chem. B 2006, 110, 10228–10230. [Google Scholar] [CrossRef]
- Dilasari, B.; Jung, Y.; Kim, G.; Kwon, K. Effect of Cation Structure on Electrochemical Behavior of Lithium in [NTf2]-based Ionic Liquids. Sustain. Chem. Eng. 2015, 4, 491–496. [Google Scholar] [CrossRef]
- Rennie, A.J.R.; Martins, V.L.; Torres, R.M.; Hall, P.J. Ionic Liquids Containing Sulfonium Cations as Electrolytes for Electrochemical Double Layer Capacitors. J. Phys. Chem. C 2015, 119, 23865–23874. [Google Scholar] [CrossRef]
- Partridge, S.M.; Brimley, R.C. Displacement Chromatography on Synthetic Ion-exchange Resins. Biochem. J. 1952, 51, 628–639. [Google Scholar] [CrossRef] [PubMed]
- Thurman, E.M.; Mills, M.S. Solid Phase Extraction Principles and Practice; John Wiley & Sons: Hoboken, NJ, USA, 1998; Chapter 6. [Google Scholar]
- Thurman, E.M.; Mills, M.S. Solid Phase Extraction Principles and Practice; John Wiley & Sons: Hoboken, NJ, USA, 1998; Chapter 2. [Google Scholar]
- McMaster, M.C. LC/MS.; A Practical User’s Guide; John Wiley & Sons: Hoboken, NJ, USA, 2005. [Google Scholar]


| Extraction Step | Eluent Volume (mL) | |
|---|---|---|
| Analytical (Flowrate = 0.5 mL/min) | Semi-Preparative (Flowrate = 5 mL/min) | |
| Conditioning | 3 | 300 |
| Sample Load | n/a | n/a |
| Wash | 1–1.5 | 100–125 |
| Desorption | 1–1.5 | 100–125 |
| Entry | Eluent | DMPIm+ % Recovery * | NTf2− % Recovery * |
|---|---|---|---|
| 1 | 1.4 N NH4OH | 48 | 47 |
| 2 | 1.4 N NH4OH | 50 | 46 |
| 3 | 1.4 N NH4OH | 0 | 41 |
| 4 | 0.1 N NaOH | 87 | 45 |
| 5 | 1.0 N NaOH | 85 | 36 |
| 6 | 0.1 M NaCl | 71 | 68 |
| 7 | 0.1 M NaCl/0% MeOH | 68 | 72 |
| 8 | 0.1 M NaCl/50% MeOH | 80 | 75 |
| 9 | 0.1 M NaCl/90% MeOH | 98 | 96 |
| Entry | Column Number | Yield (%) |
|---|---|---|
| 1 | 1 | 60 |
| 2 | 1 | 60 |
| 3 | 2 | 58 |
| 4 | 2 | 65 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Corley, C.A.; Iacono, S.T. Recycling of 1,2-Dimethyl-3-propylimidazolium bis(trifluoromethylsulfonyl)imide Ionic Liquid by Stacked Cation and Anion Exchange Adsorption-Desorption. Separations 2019, 6, 29. https://doi.org/10.3390/separations6020029
Corley CA, Iacono ST. Recycling of 1,2-Dimethyl-3-propylimidazolium bis(trifluoromethylsulfonyl)imide Ionic Liquid by Stacked Cation and Anion Exchange Adsorption-Desorption. Separations. 2019; 6(2):29. https://doi.org/10.3390/separations6020029
Chicago/Turabian StyleCorley, Cynthia A., and Scott T. Iacono. 2019. "Recycling of 1,2-Dimethyl-3-propylimidazolium bis(trifluoromethylsulfonyl)imide Ionic Liquid by Stacked Cation and Anion Exchange Adsorption-Desorption" Separations 6, no. 2: 29. https://doi.org/10.3390/separations6020029
APA StyleCorley, C. A., & Iacono, S. T. (2019). Recycling of 1,2-Dimethyl-3-propylimidazolium bis(trifluoromethylsulfonyl)imide Ionic Liquid by Stacked Cation and Anion Exchange Adsorption-Desorption. Separations, 6(2), 29. https://doi.org/10.3390/separations6020029