Recent Advances in Hydrophobic Deep Eutectic Solvents for Extraction
Abstract
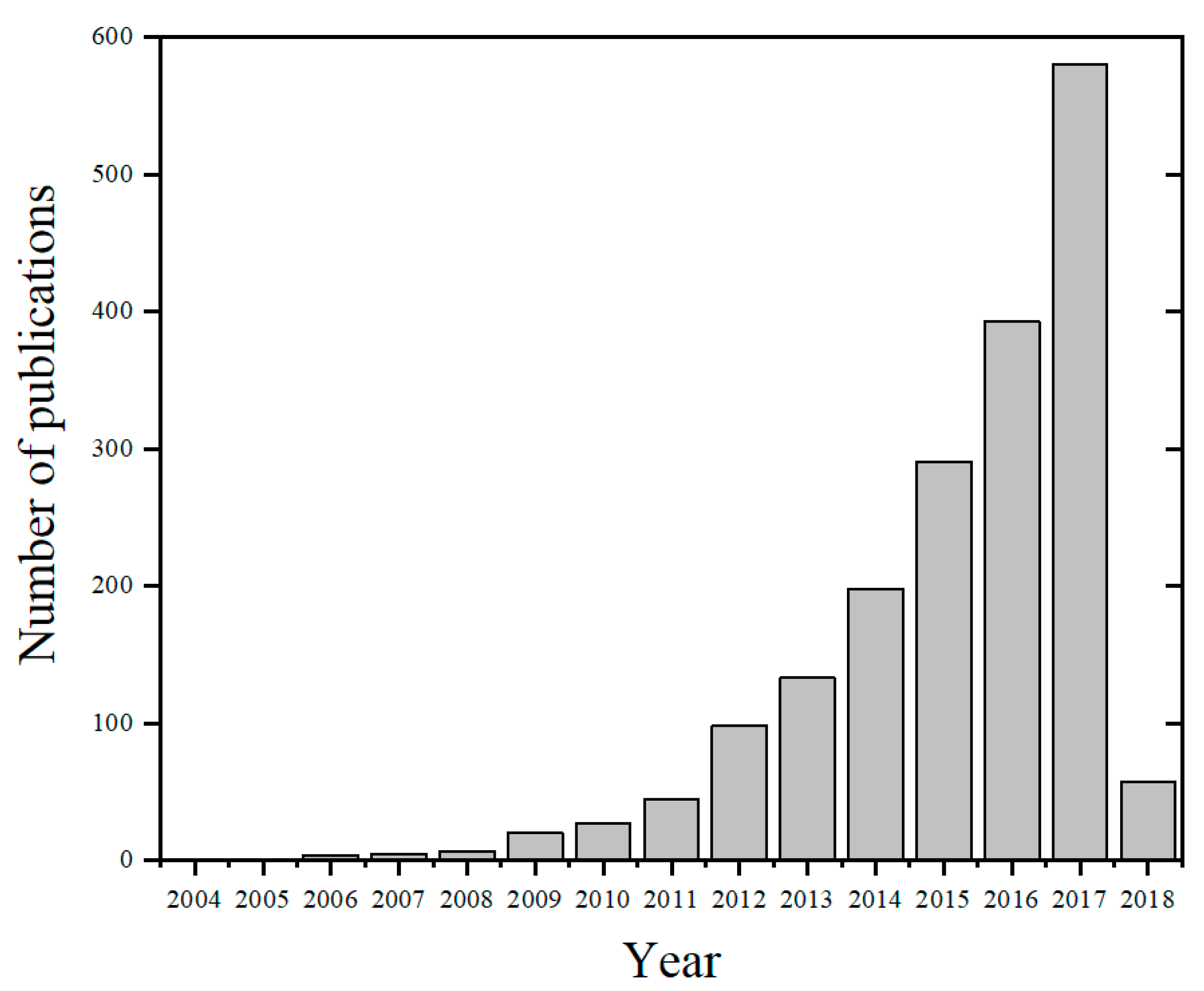
:1. Introduction
2. Formation, Classification, and Types of DES
3. Hydrophilic DES and Applications
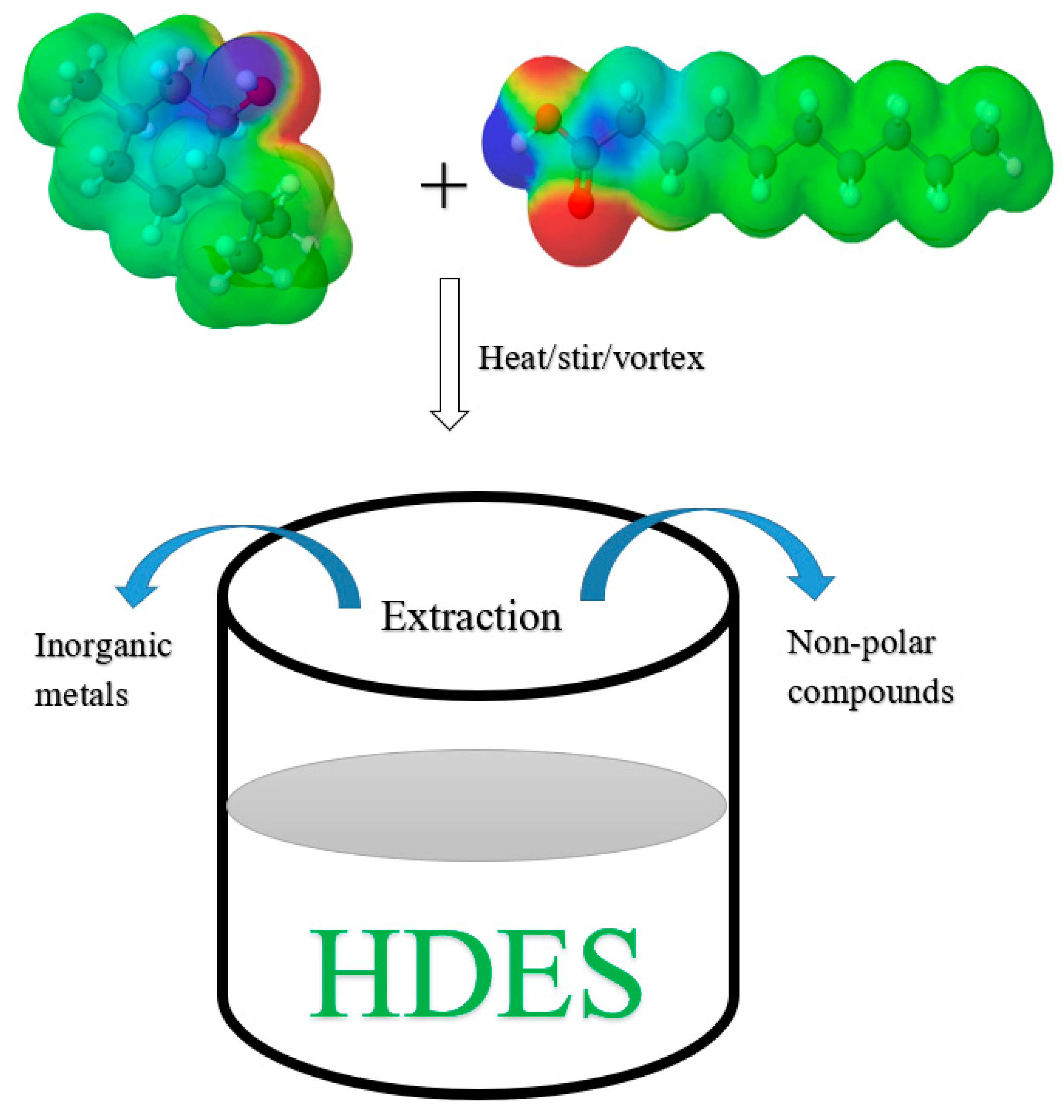
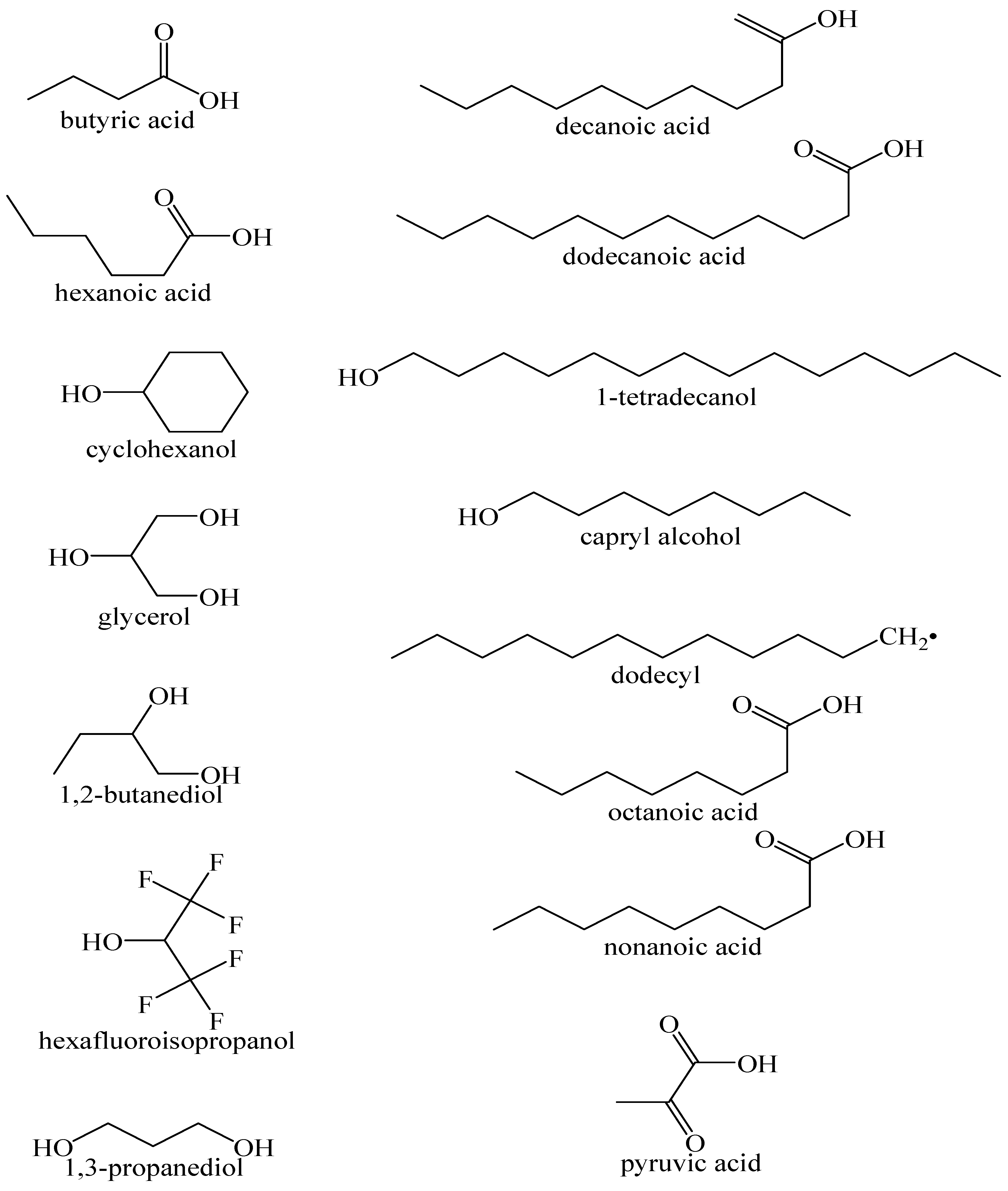
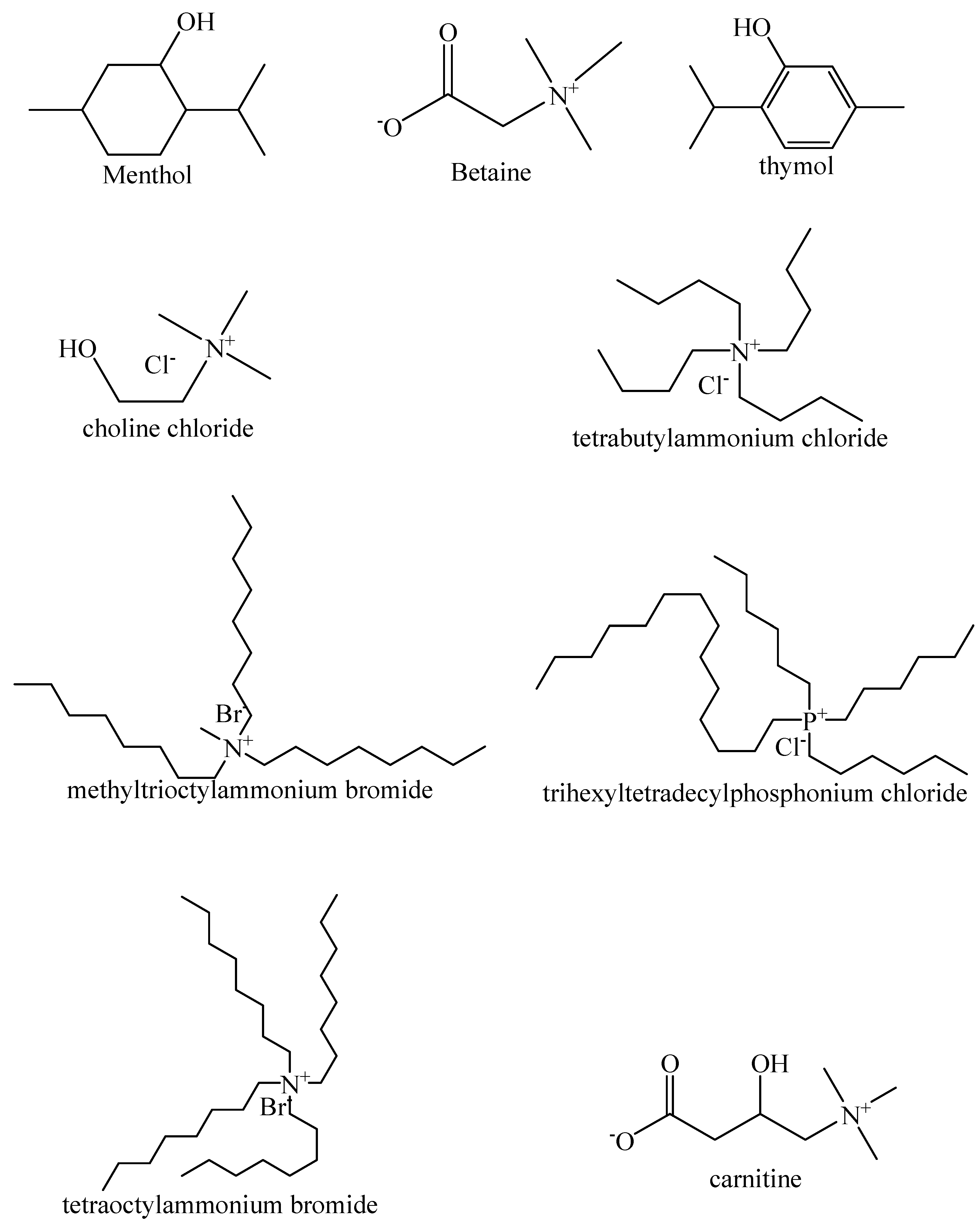
4. HDES Synthesis (Common HBA and HBD)
5. Applications of HDES in Extraction
6. Challenges, Opportunities, and Perspectives
7. Concluding Remarks
Funding
Acknowledgments
Conflicts of Interest
References
- Rogers, R.D.; Seddon, K.R. Ionic liquids--solvents of the future? Science 2003, 302, 792–793. [Google Scholar] [CrossRef] [PubMed]
- DeSimone, J.M. Practical approaches to green solvents. Science 2002, 297, 799–803. [Google Scholar] [CrossRef] [PubMed]
- Reichardt, C.; Welton, T. Solvents and Solvent Effects in Organic Chemistry; John Wiley & Sons, 2011. [Google Scholar]
- Sheldon, R.A. Green solvents for sustainable organic synthesis: State of the art. Green Chem. 2005, 7, 267–278. [Google Scholar] [CrossRef]
- Abbott, A.P.; Boothby, D.; Capper, G.; Davies, D.L.; Rasheed, R.K. Deep eutectic solvents formed between choline chloride and carboxylic acids: Versatile alternatives to ionic liquids. J. Am. Chem. Soc. 2004, 126, 9142–9147. [Google Scholar] [CrossRef] [PubMed]
- Abbott, A.P.; Capper, G.; Davies, D.L.; Rasheed, R.K.; Tambyrajah, V. Novel solvent properties of choline chloride/urea mixtures. Chem. Commun. 2003, 70–71. [Google Scholar] [CrossRef]
- Pena-Pereira, F.; Namieśnik, J. Ionic liquids and deep eutectic mixtures: Sustainable solvents for extraction processes. ChemSusChem 2014, 7, 1784–1800. [Google Scholar] [CrossRef] [PubMed]
- Imperato, G.; Höger, S.; Lenoir, D.; Koenig, B. Low melting sugar–urea–salt mixtures as solvents for organic reactions—estimation of polarity and use in catalysis. Green Chem. 2006, 8, 1051–1055. [Google Scholar] [CrossRef]
- Imperato, G.; Eibler, E.; Niedermaier, J.; König, B. Low-melting sugar–urea–salt mixtures as solvents for Diels–Alder reactions. Chem. Commun. 2005, 1170–1172. [Google Scholar] [CrossRef]
- Ruß, C.; König, B. Low melting mixtures in organic synthesis–an alternative to ionic liquids? Green Chem. 2012, 14, 2969–2982. [Google Scholar]
- Abbott, A.P.; Capper, G.; Gray, S. Design of improved deep eutectic solvents using hole theory. Chemphyschem 2006, 7, 803–806. [Google Scholar] [CrossRef]
- Smith, E.L.; Abbott, A.P.; Ryder, K.S. Deep eutectic solvents (DESs) and their applications. Chem. Rev. 2014, 114, 11060–11082. [Google Scholar] [CrossRef] [PubMed]
- Florindo, C.; Oliveira, F.; Rebelo, L.; Fernandes, A.M.; Marrucho, I. Insights into the synthesis and properties of deep eutectic solvents based on cholinium chloride and carboxylic acids. ACS Sustain. Chem. Eng. 2014, 2, 2416–2425. [Google Scholar] [CrossRef]
- Zhao, B.-Y.; Xu, P.; Yang, F.-X.; Wu, H.; Zong, M.-H.; Lou, W.-Y. Biocompatible deep eutectic solvents based on choline chloride: Characterization and application to the extraction of rutin from Sophora japonica. ACS Sustain. Chem. Eng. 2015, 3, 2746–2755. [Google Scholar] [CrossRef]
- Abbott, A.P.; Capper, G.; Davies, D.L.; Munro, H.L.; Rasheed, R.K.; Tambyrajah, V. Preparation of novel, moisture-stable, Lewis-acidic ionic liquids containing quaternary ammonium salts with functional side chains. Chem. Commun. 2001, 0, 2010–2011. [Google Scholar] [CrossRef]
- Zhang, Q.; Vigier, K.D.O.; Royer, S.; Jérôme, F. Deep eutectic solvents: Syntheses, properties and applications. Chem. Soc. Rev. 2012, 41, 7108–7146. [Google Scholar] [CrossRef] [PubMed]
- Jessop, P.G. Searching for green solvents. Green Chem. 2011, 13, 1391–1398. [Google Scholar] [CrossRef]
- Lozano, P.; Bernal, J.M.; Vaultier, M. Towards continuous sustainable processes for enzymatic synthesis of biodiesel in hydrophobic ionic liquids/supercritical carbon dioxide biphasic systems. Fuel 2011, 90, 3461–3467. [Google Scholar] [CrossRef]
- Zhao, H.; Baker, G.A. Ionic liquids and deep eutectic solvents for biodiesel synthesis: A review. J. Chem. Technol. Biotechnol. 2013, 88, 3–12. [Google Scholar] [CrossRef]
- Shishov, A.; Bulatov, A.; Locatelli, M.; Carradori, S.; Andruch, V. Application of deep eutectic solvents in analytical chemistry. A review. Microchem. J. 2017, 135, 33–38. [Google Scholar] [CrossRef]
- van Osch, D.J.; Zubeir, L.F.; van den Bruinhorst, A.; Rocha, M.A.; Kroon, M.C. Hydrophobic deep eutectic solvents as water-immiscible extractants. Green Chem. 2015, 17, 4518–4521. [Google Scholar] [CrossRef]
- Francisco, M.; van den Bruinhorst, A.; Kroon, M.C. Low-transition-temperature mixtures (LTTMs): A new generation of designer solvents. Angew. Chem. Int. Ed. 2013, 52, 3074–3085. [Google Scholar] [CrossRef] [PubMed]
- Li, J.-J.; Xiao, H.; Tang, X.-D.; Zhou, M. Green carboxylic acid-based deep eutectic solvents as solvents for extractive desulfurization. Energy Fuels 2016, 30, 5411–5418. [Google Scholar] [CrossRef]
- Aroso, I.M.; Craveiro, R.; Rocha, Â.; Dionísio, M.; Barreiros, S.; Reis, R.L.; Paiva, A.; Duarte, A.R.C. Design of controlled release systems for THEDES—therapeutic deep eutectic solvents, using supercritical fluid technology. Int. J. Pharm. 2015, 492, 73–79. [Google Scholar] [CrossRef]
- Aroso, I.M.; Silva, J.C.; Mano, F.; Ferreira, A.S.; Dionísio, M.; Sá-Nogueira, I.; Barreiros, S.; Reis, R.L.; Paiva, A.; Duarte, A.R.C. Dissolution enhancement of active pharmaceutical ingredients by therapeutic deep eutectic systems. Eur. J. Pharm. Biopharm. 2016, 98, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Patil, U.B.; Shendage, S.S.; Nagarkar, J.M. One-pot synthesis of nitriles from aldehydes catalyzed by deep eutectic solvent. Synthesis 2013, 45, 3295–3299. [Google Scholar]
- Wang, L.; Dai, D.Y.; Chen, Q.; He, M.Y. Rapid, Sustainable, and Gram-Scale Synthesis of Phenols Catalyzed by a Biodegradable Deep Eutectic Mixture in Water. Asian J. Org. Chem. 2013, 2, 1040–1043. [Google Scholar] [CrossRef]
- Avalos, M.; Babiano, R.; Cintas, P.; Jimenez, J.L.; Palacios, J.C. Greener media in chemical synthesis and processing. Angew. Chem. Int. Ed. 2006, 45, 3904–3908. [Google Scholar] [CrossRef]
- Hayyan, M.; Mjalli, F.S.; Hashim, M.A.; AlNashef, I.M. A novel technique for separating glycerine from palm oil-based biodiesel using ionic liquids. Fuel Process. Technol. 2010, 91, 116–120. [Google Scholar] [CrossRef]
- Shahbaz, K.; Mjalli, F.; Hashim, M.; AlNashef, I. Using deep eutectic solvents based on methyl triphenyl phosphunium bromide for the removal of glycerol from palm-oil-based biodiesel. Energy Fuels 2011, 25, 2671–2678. [Google Scholar] [CrossRef]
- Francisco, M.; van den Bruinhorst, A.; Kroon, M.C. New natural and renewable low transition temperature mixtures (LTTMs): Screening as solvents for lignocellulosic biomass processing. Green Chem. 2012, 14, 2153–2157. [Google Scholar] [CrossRef]
- Dai, Y.; Witkamp, G.-J.; Verpoorte, R.; Choi, Y.H. Natural deep eutectic solvents as a new extraction media for phenolic metabolites in Carthamus tinctorius L. Anal. Chem. 2013, 85, 6272–6278. [Google Scholar] [CrossRef] [PubMed]
- Nam, M.W.; Zhao, J.; Lee, M.S.; Jeong, J.H.; Lee, J. Enhanced extraction of bioactive natural products using tailor-made deep eutectic solvents: Application to flavonoid extraction from Flos sophorae. Green Chem. 2015, 17, 1718–1727. [Google Scholar] [CrossRef]
- Tang, B.; Zhang, H.; Row, K.H. Application of deep eutectic solvents in the extraction and separation of target compounds from various samples. J. Sep. Sci. 2015, 38, 1053–1064. [Google Scholar] [CrossRef]
- Karimi, M.; Dadfarnia, S.; Shabani, A.M.H.; Tamaddon, F.; Azadi, D. Deep eutectic liquid organic salt as a new solvent for liquid-phase microextraction and its application in ligandless extraction and preconcentraion of lead and cadmium in edible oils. Talanta 2015, 144, 648–654. [Google Scholar] [CrossRef] [PubMed]
- Radošević, K.; Ćurko, N.; Srček, V.G.; Bubalo, M.C.; Tomašević, M.; Ganić, K.K.; Redovniković, I.R. Natural deep eutectic solvents as beneficial extractants for enhancement of plant extracts bioactivity. LWT-Food Sci. Technol. 2016, 73, 45–51. [Google Scholar]
- Hizaddin, H.F.; Ramalingam, A.; Hashim, M.A.; Hadj-Kali, M.K. Evaluating the performance of deep eutectic solvents for use in extractive denitrification of liquid fuels by the conductor-like screening model for real solvents. J. Chem. Eng. Data 2014, 59, 3470–3487. [Google Scholar] [CrossRef]
- Brett, C.M. Deep eutectic solvents and applications in electrochemical sensing. Curr. Opin. Electrochem. 2018, 10, 143–148. [Google Scholar] [CrossRef]
- Nkuku, C.A.; LeSuer, R.J. Electrochemistry in deep eutectic solvents. J. Phys. Chem. B 2007, 111, 13271–13277. [Google Scholar] [CrossRef] [PubMed]
- Durand, E.; Lecomte, J.; Baréa, B.; Piombo, G.; Dubreucq, E.; Villeneuve, P. Evaluation of deep eutectic solvents as new media for Candida antarctica B lipase catalyzed reactions. Process Biochem. 2012, 47, 2081–2089. [Google Scholar] [CrossRef]
- Choi, Y.H.; van Spronsen, J.; Dai, Y.; Verberne, M.; Hollmann, F.; Arends, I.W.; Witkamp, G.-J.; Verpoorte, R. Are natural deep eutectic solvents the missing link in understanding cellular metabolism and physiology? Plant Physiol. 2011, 156, 1701–1705. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.; van Spronsen, J.; Witkamp, G.-J.; Verpoorte, R.; Choi, Y.H. Natural deep eutectic solvents as new potential media for green technology. Anal. Chim. Acta 2013, 766, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Paiva, A.; Craveiro, R.; Aroso, I.; Martins, M.; Reis, R.L.; Duarte, A.R.C. Natural deep eutectic solvents–solvents for the 21st century. ACS Sustain. Chem. Eng. 2014, 2, 1063–1071. [Google Scholar] [CrossRef]
- Liu, Y.; Friesen, J.B.; McAlpine, J.B.; Lankin, D.C.; Chen, S.-N.; Pauli, G.F. Natural deep eutectic solvents: Properties, applications, and perspectives. J. Nat. Prod. 2018, 81, 679–690. [Google Scholar] [CrossRef] [PubMed]
- Satlewal, A.; Agrawal, R.; Bhagia, S.; Sangoro, J.; Ragauskas, A.J. Natural deep eutectic solvents for lignocellulosic biomass pretreatment: Recent developments, challenges and novel opportunities. Biotechnol. Adv. 2018. [Google Scholar] [CrossRef]
- de los Ángeles Fernández, M.; Boiteux, J.; Espino, M.; Gomez, F.V.; Silva, M.F. Natural Deep Eutectic Solvents-Mediated Extractions: The way forward for sustainable analytical developments. Anal. Chim. Acta 2018. [Google Scholar]
- Lynam, J.G.; Kumar, N.; Wong, M.J. Deep eutectic solvents’ ability to solubilize lignin, cellulose, and hemicellulose; thermal stability; and density. Bioresour. Technol. 2017, 238, 684–689. [Google Scholar] [CrossRef] [PubMed]
- Vanda, H.; Dai, Y.; Wilson, E.G.; Verpoorte, R.; Choi, Y.H. Green solvents from ionic liquids and deep eutectic solvents to natural deep eutectic solvents. Comptes Rendus Chim. 2018, 21, 628–638. [Google Scholar] [CrossRef]
- Morrison, H.G.; Sun, C.C.; Neervannan, S. Characterization of thermal behavior of deep eutectic solvents and their potential as drug solubilization vehicles. Int. J. Pharm. 2009, 378, 136–139. [Google Scholar] [CrossRef] [PubMed]
- Rajabi, M.; Ghassab, N.; Hemmati, M.; Asghari, A. Emulsification microextraction of amphetamine and methamphetamine in complex matrices using an up-to-date generation of eco-friendly and relatively hydrophobic deep eutectic solvent. J. Chromatogr. A 2018. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Yang, M.; Cao, F.; Wang, J.; Su, E. Well-designed hydrophobic deep eutectic solvents as green and efficient media for the extraction of artemisinin from Artemisia annua leaves. ACS Sustain. Chem. Eng. 2017, 5, 3270–3278. [Google Scholar] [CrossRef]
- Phelps, T.E.; Bhawawet, N.; Jurisson, S.S.; Baker, G.A. Efficient and Selective Extraction of 99mTcO4–from Aqueous Media using Hydrophobic Deep Eutectic Solvents. ACS Sustain. Chem. Eng. 2018. [Google Scholar] [CrossRef]
- Deng, W.; Yu, L.; Li, X.; Chen, J.; Wang, X.; Deng, Z.; Xiao, Y. Hexafluoroisopropanol-based hydrophobic deep eutectic solvents for dispersive liquid-liquid microextraction of pyrethroids in tea beverages and fruit juices. Food Chem. 2019, 274, 891–899. [Google Scholar] [CrossRef] [PubMed]
- Makoś, P.; Przyjazny, A.; Boczkaj, G. Hydrophobic deep eutectic solvents as “green” extraction media for polycyclic aromatic hydrocarbons in aqueous samples. J. Chromatogr. A 2018. [Google Scholar]
- Florindo, C.; Romero, L.; Rintoul, I.; Branco, L.C.; Marrucho, I.M. From phase change materials to green solvents: Hydrophobic low viscous fatty acid–based deep eutectic solvents. ACS Sustain. Chem. Eng. 2018, 6, 3888–3895. [Google Scholar] [CrossRef]
- Florindo, C.; Branco, L.; Marrucho, I. Development of hydrophobic deep eutectic solvents for extraction of pesticides from aqueous environments. Fluid Phase Equilibria 2017, 448, 135–142. [Google Scholar] [CrossRef]
- Martins, M.A.R.; Crespo, E.A.; Pontes, P.V.; Silva, L.P.; Bülow, M.; Maximo, G.J.; Batista, E.A.C.; Held, C.; Pinho, S.P.; Coutinho, J.A. Tunable hydrophobic eutectic solvents based on terpenes and monocarboxylic acids. ACS Sustain. Chem. Eng. 2018. [Google Scholar] [CrossRef]
- Ribeiro, B.D.; Florindo, C.; Iff, L.C.; Coelho, M.A.; Marrucho, I.M. Menthol-based eutectic mixtures: Hydrophobic low viscosity solvents. ACS Sustain. Chem. Eng. 2015, 3, 2469–2477. [Google Scholar] [CrossRef]
- Safavi, A.; Ahmadi, R.; Ramezani, A.M. Vortex-assisted liquid-liquid microextraction based on hydrophobic deep eutectic solvent for determination of malondialdehyde and formaldehyde by HPLC-UV approach. Microchem. J. 2018, 143, 166–174. [Google Scholar] [CrossRef]
- Zubeir, L.F.; Van Osch, D.J.; Rocha, M.A.; Banat, F.; Kroon, M.C. Carbon dioxide solubilities in decanoic acid-based hydrophobic deep eutectic solvents. J. Chem. Eng. Data 2018, 63, 913–919. [Google Scholar] [CrossRef]
- Zubeir, L.F.; Romanos, G.E.; Weggemans, W.M.; Iliev, B.; Schubert, T.J.; Kroon, M.C. Solubility and diffusivity of CO2 in the ionic liquid 1-butyl-3-methylimidazolium tricyanomethanide within a large pressure range (0.01 MPa to 10 MPa). J. Chem. Eng. Data 2015, 60, 1544–1562. [Google Scholar] [CrossRef]
- Verma, R.; Banerjee, T. Liquid–Liquid Extraction of Lower Alcohols Using Menthol-Based Hydrophobic Deep Eutectic Solvent: Experiments and COSMO-SAC Predictions. Ind. Eng. Chem. Res. 2018, 57, 3371–3381. [Google Scholar] [CrossRef]
- Kislik, V.S. Solvent Extraction: Classical and Novel Approaches; Elsevier: Amsterdam, The Netherlands, 2011. [Google Scholar]
- Zhu, S.; Zhou, J.; Jia, H.; Zhang, H. Liquid–liquid microextraction of synthetic pigments in beverages using a hydrophobic deep eutectic solvent. Food Chem. 2018, 243, 351–356. [Google Scholar] [CrossRef] [PubMed]
- Gilmore, M.; McCourt, E.; Connolly, F.; Nockemann, P.; Swadzba-Kwasny, M.; Holbrey, J.D. Hydrophobic deep eutectic solvents incorporating trioctylphosphine oxide: Advanced liquid extractants. ACS Sustain. Chem. Eng. 2018. [Google Scholar] [CrossRef]
- Laitinen, O.; Suopajärvi, T.; Österberg, M.; Liimatainen, H. Hydrophobic, superabsorbing aerogels from choline chloride-based deep eutectic solvent pretreated and silylated cellulose nanofibrils for selective oil removal. ACS Appl. Mater. Interfaces 2017, 9, 25029–25037. [Google Scholar] [CrossRef] [PubMed]
- Křížek, T.; Bursová, M.; Horsley, R.; Kuchař, M.; Tůma, P.; Čabala, R.; Hložek, T. Menthol-based hydrophobic deep eutectic solvents: Towards greener and efficient extraction of phytocannabinoids. J. Clean. Prod. 2018, 193, 391–396. [Google Scholar]
- Cao, J.; Yang, M.; Cao, F.; Wang, J.; Su, E. Tailor-made hydrophobic deep eutectic solvents for cleaner extraction of polyprenyl acetates from Ginkgo biloba leaves. J. Clean. Prod. 2017, 152, 399–405. [Google Scholar] [CrossRef]
- Tereshatov, E.; Boltoeva, M.Y.; Folden, C. First evidence of metal transfer into hydrophobic deep eutectic and low-transition-temperature mixtures: Indium extraction from hydrochloric and oxalic acids. Green Chem. 2016, 18, 4616–4622. [Google Scholar] [CrossRef]
- van Osch, D.J.; Parmentier, D.; Dietz, C.H.; van den Bruinhorst, A.; Tuinier, R.; Kroon, M.C. Removal of alkali and transition metal ions from water with hydrophobic deep eutectic solvents. Chem. Commun. 2016, 52, 11987–11990. [Google Scholar] [CrossRef]
- Dietz, C.H.; van Osch, D.J.; Kroon, M.C.; Sadowski, G.; van Sint Annaland, M.; Gallucci, F.; Zubeir, L.F.; Held, C. PC-SAFT modeling of CO2 solubilities in hydrophobic deep eutectic solvents. Fluid Phase Equilibria 2017, 448, 94–98. [Google Scholar] [CrossRef]
- Dietz, C.H.; Kroon, M.C.; Di Stefano, M.; van Sint Annaland, M.; Gallucci, F. Selective separation of furfural and hydroxymethylfurfural from an aqueous solution using a supported hydrophobic deep eutectic solvent liquid membrane. Faraday Discuss. 2017, 206, 77–92. [Google Scholar] [CrossRef]
- Ruggeri, S.; Poletti, F.; Zanardi, C.; Pigani, L.; Zanfrognini, B.; Corsi, E.; Dossi, N.; Salomäki, M.; Kivelä, H.; Lukkari, J. Chemical and electrochemical properties of a hydrophobic deep eutectic solvent. Electrochim. Acta 2018. [Google Scholar] [CrossRef]
| Type | General Formula | Terms |
|---|---|---|
| type I | Cat+X−zMClx | M = Zn, Sn, Al, Ga, In |
| type II | Cat+X−zMClx·YH2O | M = Cr, Co, Cu, Ni, Fe |
| type III | Cat+X−zRZ | Z = CONH2, COOH, OH |
| type IV | MClx + RZ = ·RZ + | M = Al, Zn and Z = CONH2, OH |
| HBA | HBD | Molar Ratio | Synthesis | Ref. |
|---|---|---|---|---|
| DL-menthol | acetic acid | 1:1 | heating at 50 °C for 15 min | [58] |
| lactic acid | 1:2 | |||
| lauric acid | 2:1 | |||
| pyruvic acid | 1:2 | |||
| choline chloride | phenethyl alcohol | 1:4 | stirring at ambient temperature | [50] |
| DoDecA | octanoic acid | 1:3 | heating at 40 °C | [55] |
| nonanoic acid | 1:3 | |||
| DecA | 1:2 | |||
| betaine | HFI | 1:2, 1:2.5, 1:3 | heating at 80 °C in screw-cap pressure tube | [53] |
| L-carnitine | ||||
| N8881-Br | DecA | 1:2 | stirring and heating at 35 °C | [59] |
| N4444-Cl | DecA | 1:2 | vortex mixing at room temperature and heating at 308 K in oil bath | [60,61] |
| N8881-Cl | ||||
| N8881-Br | ||||
| N8888-Cl | ||||
| N8888-Br | ||||
| DL-menthol | lauric acid | 0.5:1, 1:1, 1.5:1, 2:1, 2.5:1 | reflux condensing for 1 h at 50 °C with stirring | [62] |
| DL-menthol | acetic acid, levulinic acid, butyric acid, hexanoic acid, octanoic acid, DecA (1:1), pyruvic acid (1:2), DoDecA (2:1) | 1:1, 1:2, 2:1 | mixing with mechanical stirring at 350 rpm at 353.15 K | [56] |
| N4444-Cl | acetic acid (1:1), levulinic acid, hexanoic acid, octanoic acid, DecA (1:2) | |||
| C32H68PCl | DecA | 1:2 | heating at 80 °C for 30 min | [52] |
| N8888-Br | hexanoic acid | |||
| DecA | ||||
| N81-Cl | ethylene glycol, 1-propanol, 1,3-propanediol, glycerol, 1-butanol, 1,2-butanediol, hexyl alcohol, capryl alcohol, decyl alcohol, dodecyl alcohol, 1-tetradecanol, cyclohexanol, DL-menthol | 1:2 | heating at 80 °C | [51] |
| thymol | (±)-camphor | 7:3, 3:2, 1:1 | heat and stirred magnetically at 60 °C | [54] |
| 10-undecylenic acid | 7:3, 3:2, 1:1, 1:2, 1:3, and 1:4 | |||
| DecA | 3:2, 1:1, 1:2, 1:3 |
| Analyte | Separation/Quantification | Ref. |
|---|---|---|
| caffeine, isophthalic acid, tryptophan, and vanillic acid | partitioning/UV-Vis | [58] |
| Amphetamine, Meth | HPLC/UV-Vis | [50] |
| bisphenol A | phase separation by equilibration/UV-Vis | [55] |
| pyrethroids | HPLC/DAD | [53] |
| derivatized MA and FA | HPLC/UV-Vis | [59] |
| CO2 solubility | evacuation steps/magnetic suspension balance | [60,61] |
| ethanol, 1-propanol, 1-butanol | shaking and 24 h equilibration/1H NMR | [62] |
| (neonicotinoids) imidacloprid, acetamiprid, nitenpyram and thiamethoxam | partitioning/gravimetric | [56] |
| pertechnetate (99mTcO4−) | equilibration and TLC/NaI (Tl) well counter detector | [52] |
| artemisinin | HPLC/UV-Vis | [51] |
| polycyclic aromatic hydrocarbons | GC-MS | [54] |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dwamena, A.K. Recent Advances in Hydrophobic Deep Eutectic Solvents for Extraction. Separations 2019, 6, 9. https://doi.org/10.3390/separations6010009
Dwamena AK. Recent Advances in Hydrophobic Deep Eutectic Solvents for Extraction. Separations. 2019; 6(1):9. https://doi.org/10.3390/separations6010009
Chicago/Turabian StyleDwamena, Amos K. 2019. "Recent Advances in Hydrophobic Deep Eutectic Solvents for Extraction" Separations 6, no. 1: 9. https://doi.org/10.3390/separations6010009
APA StyleDwamena, A. K. (2019). Recent Advances in Hydrophobic Deep Eutectic Solvents for Extraction. Separations, 6(1), 9. https://doi.org/10.3390/separations6010009





