Gold Nanoparticle Uptake in Tumor Cells: Quantification and Size Distribution by sp-ICPMS
Abstract
1. Introduction
2. Materials and Methods
2.1. Instruments
2.2. Reagents
2.3. Samples and Sample Preparation
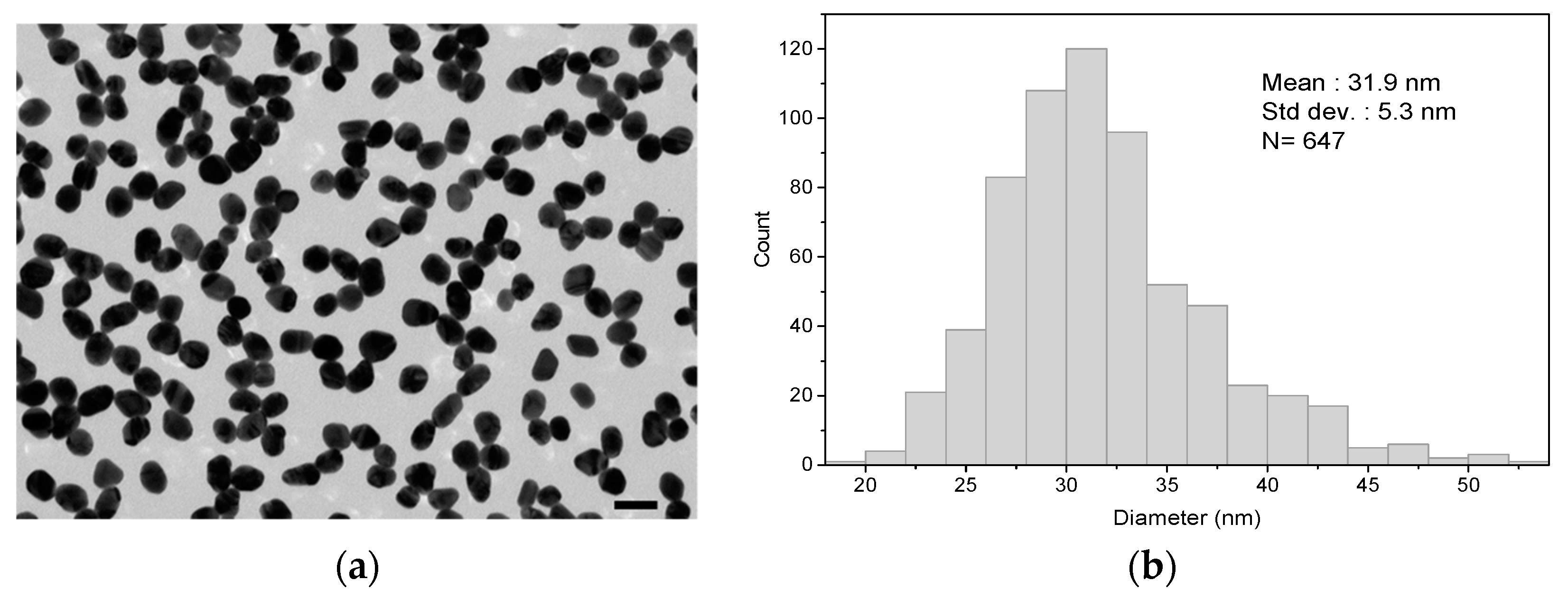
2.3.1. Gold Nanoparticle Synthesis and Functionalization
2.3.2. Cell Growth
2.3.3. Sample Preparation for Total Gold Mass Concentration Measurements
2.3.4. Sample Preparation for Sp-ICPMS Measurement and Method Validation
2.4. Calculations
2.4.1. Limits of Detection and Uncertainty
2.4.2. Particle Size
2.4.3. Particle Mass Concentration
2.4.4. Number Particle Concentration and Transport Efficiency
3. Results and Discussion
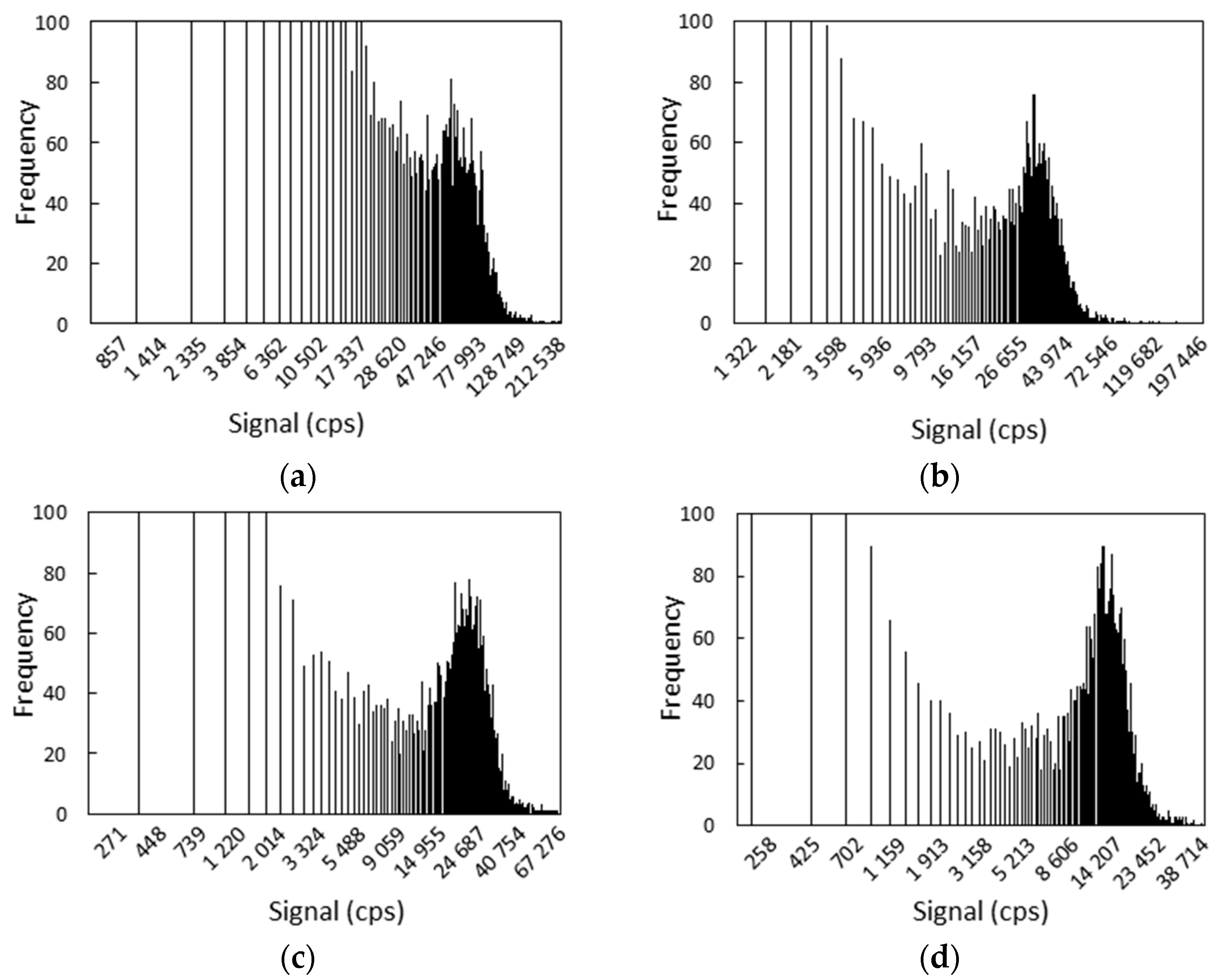
3.1. Optimization of Sp-ICPMS Measurements
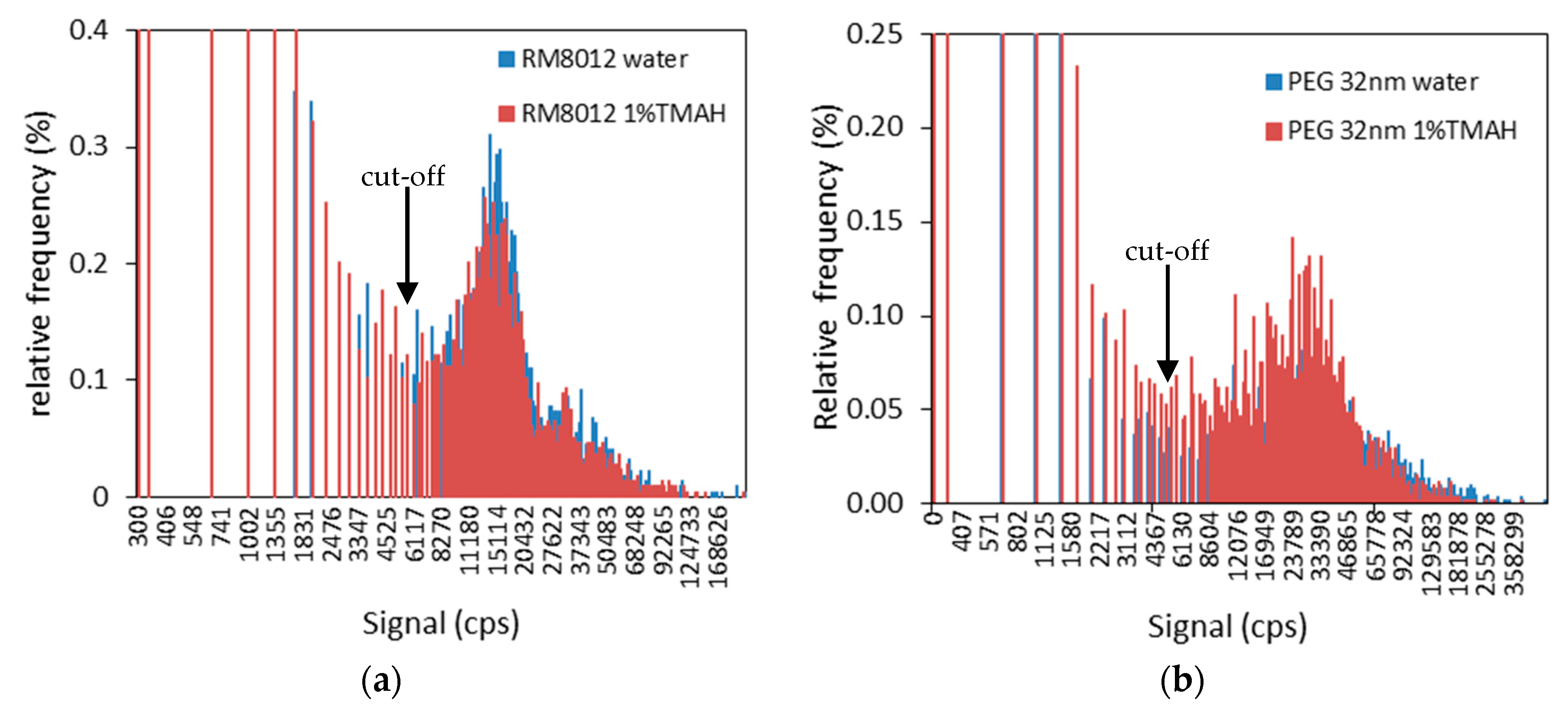
3.2. Effect of Sample Preparation
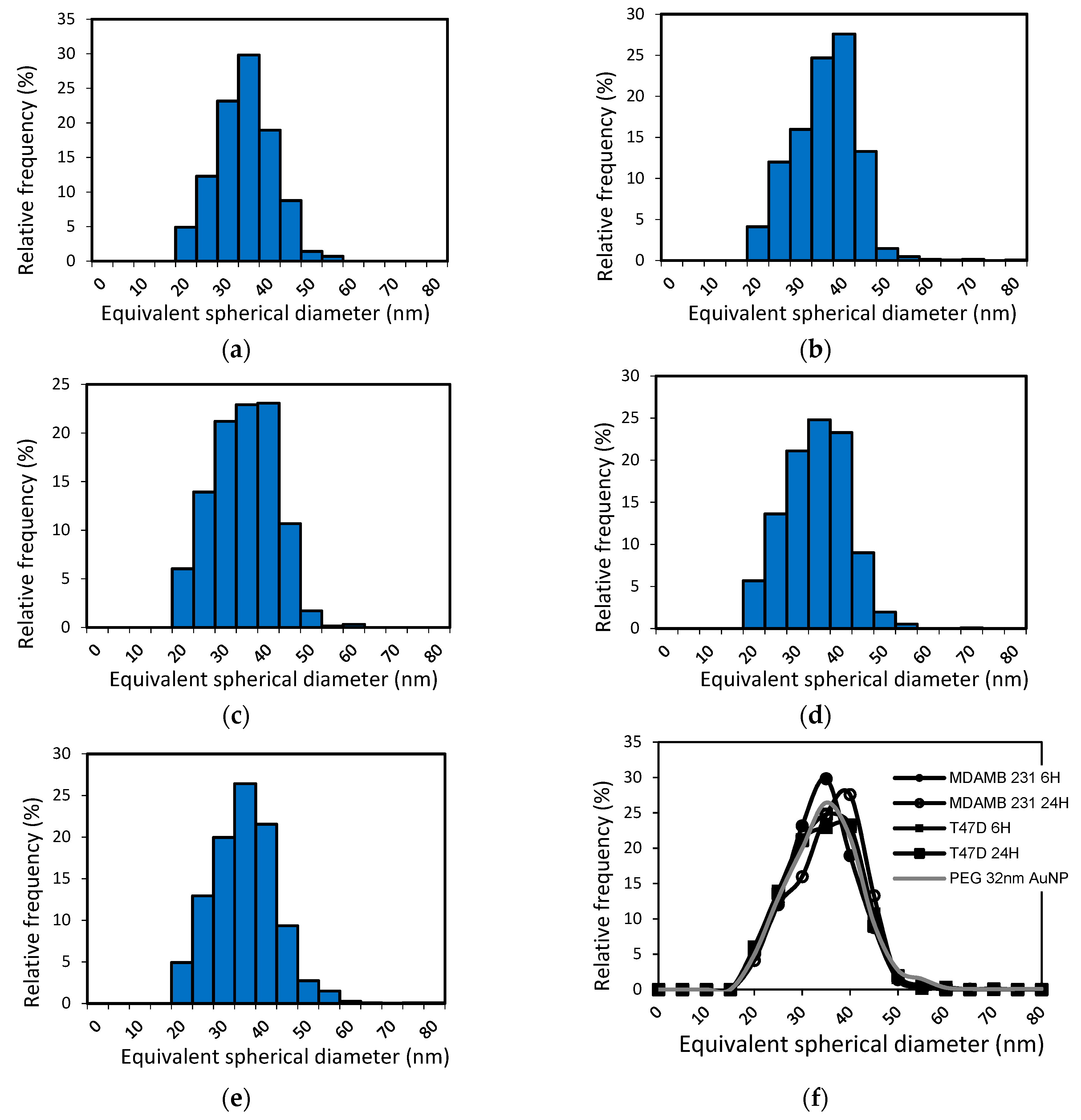
3.3. Gold Recovery and Size Measurements
3.4. Average Particle Number Per Cell
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bobo, D.; Robinson, K.J.; Islam, J.; Thurecht, K.J.; Corrie, S.R. Nanoparticle-Based Medicines: A Review of FDA-Approved Materials and Clinical Trials to Date. Pharm. Res. 2016, 33, 2373–2387. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.; Hirst, D.G.; O’Sullivan, J.M. Gold nanoparticles as novel agents for cancer therapy. Br. J. Radiol. 2012, 85, 101–113. [Google Scholar] [CrossRef] [PubMed]
- Hadioui, M.; Merdzan, V.; Wilkinson, K.J. Detection and Characterization of ZnO Nanoparticles in Surface and Waste Waters Using Single Particle ICPMS. Environ. Sci. Technol. 2015, 49, 6141–6148. [Google Scholar] [CrossRef] [PubMed]
- Adeleye, A.S.; Pokhrel, S.; Mädler, L.; Keller, A.A. Influence of nanoparticle doping on the colloidal stability and toxicity of copper oxide nanoparticles in synthetic and natural waters. Water Res. 2018, 132, 12–22. [Google Scholar] [CrossRef] [PubMed]
- Peters, R.J.B.; van Bemmel, G.; Herrera-Rivera, Z.; Helsper, H.P.F.G.; Marvin, H.J.P.; Weigel, S.; Tromp, P.C.; Oomen, A.G.; Rietveld, A.G.; Bouwmeester, H. Characterization of titanium dioxide nanoparticles in food products: Analytical methods to define nanoparticles. Agric. Food Chem. 2014, 62, 6285–6293. [Google Scholar] [CrossRef] [PubMed]
- Loeschner, K.; Correia, M.; Chaves, C.L.; Rokkjær, I.; Sloth, J.J. Detection and characterization of aluminium-containing nanoparticles in Chinese noodles by single particle ICP-MS. Food Addit. Contam. Part A 2018, 35, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Dan, Y.; Shi, H.; Stephan, C.; Liang, X. Rapid analysis of titanium dioxide nanoparticles in sunscreens using single particle inductively coupled plasma-mass spectrometry. Microchem. J. 2015, 122, 119–126. [Google Scholar] [CrossRef]
- de la Calle, I.; Menta, M.; Klein, M.; Séby, F. Screening of TiO2 and Au nanoparticles in cosmetics and determination of elemental impurities by multiple techniques (DLS, SP-ICP-MS, ICP-MS and ICP-OES). Talanta 2017, 171, 291–306. [Google Scholar] [CrossRef]
- Adeleye, A.S.; Oranu, E.A.; Tao, M.; Keller, A.A. Release and detection of nanosized copper from a commercial antifouling paint. Water Res. 2016, 102, 374–382. [Google Scholar] [CrossRef]
- Bao, D.; Oh, Z.G.; Chen, Z. Characterization of Silver Nanoparticles Internalized by Arabidopsis Plants Using Single Particle ICP-MS Analysis. Plant Sci. 2016, 7, 32. [Google Scholar] [CrossRef]
- Dan, Y.; Zhang, W.; Xue, R.; Ma, X.; Stephan, C.; Shi, H. Characterization of gold nanoparticle uptake by tomato plants using enzymatic extraction followed by single-particle Inductively Coupled plasma-mass spectrometry analysis. Environ. Sci. Technol. 2015, 49, 3007–3014. [Google Scholar] [CrossRef] [PubMed]
- Jimenez-Lamana, J.; Wojcieszek, J.; Jakubiak, M.; Asztemborska, M.; Szpunar, J. Single particle ICP-MS characterization of platinum nanoparticles uptake and bioaccumulation by Lepidium sativum and Sinapis alba plants. J. Anal. Atomic Spectrom. 2016, 31, 2321–2329. [Google Scholar] [CrossRef]
- Keller, A.A.; Huang, Y.; Nelson, J. Detection of nanoparticles in edible plant tissues exposed to nano-copper using single-particle ICP-MS. J. Nanopart. Res. 2018, 20, 101. [Google Scholar] [CrossRef]
- Klingberg, H.; Oddershede, L.B.; Loeschner, K.; Larsen, E.H.; Loft, S.; Møller, P. Uptake of gold nanoparticles in primary human endothelial cells. Toxicol. Res. 2015, 4, 655–666. [Google Scholar] [CrossRef]
- Kińska, K.; Jiménez-Lamana, J.; Kowalska, J.; Krasnodębska-Ostręga, B.; Szpunar, J. Study of the uptake and bioaccumulation of palladium nanoparticles by Sinapis alba using single particle ICP-MS. Sci. Total Environ. 2018, 615, 1078–1085. [Google Scholar] [CrossRef] [PubMed]
- Dan, Y.; Ma, X.; Zhang, W.; Liu, K.; Stephan, C.; Shi, H. Single particle ICP-MS method development for the determination of plant uptake and accumulation of CeO2 nanoparticles. Anal. Bioanal. Chem. 2016, 408, 5157–5167. [Google Scholar] [CrossRef] [PubMed]
- Loeschner, K.; Brabrand, M.S.J.; Sloth, J.J.; Larsen, E.H. Use of alkaline or enzymatic sample pretreatment prior to characterization of gold nanoparticles in animal tissue by single-particle ICPMS. Anal. Bioanal. Chem. 2014, 406, 3845–3851. [Google Scholar] [CrossRef] [PubMed]
- Vidmar, J.; Buerki-Thurnherr, T.; Loeschner, K. Comparison of the suitability of alkaline or enzymatic sample pre-treatment for characterization of silver nanoparticles in human tissue by single particle ICP-MS. J. Anal. Atomic Spectrom. 2018, 33, 752–761. [Google Scholar] [CrossRef]
- Peters, R.J.B.; Rivera, Z.H.; van Bemmel, G.; Marvin, H.J.P.; Weigel, S.; Bouwmeester, H. Development and validation of single particle ICP-MS for sizing and quantitative determination of nano-silver in chicken meat. Anal. Bioanal. Chem. 2014, 406, 3875–3885. [Google Scholar] [CrossRef]
- Gray, E.P.; Coleman, J.; Bednar, A.; Kennedy, A.J.; Ranville, J.F.; Higgins, C.P. Extraction and analysis of silver and gold nanoparticles from biological tissues using single particle inductively coupled plasma mass spectrometry. Environ. Sci. Technol. 2013, 47, 14315–14323. [Google Scholar] [CrossRef]
- Jokerst, J.V.; Lobovkina, T.; Zare, R.N.; Gambhir, S.S. Nanoparticle PEGylation for imaging and therapy. Nanomedicine 2011, 6, 715–728. [Google Scholar] [CrossRef] [PubMed]
- Peters, R.; Herrera-Rivera, Z.; Undas, A.; van der Lee, M.; Marvin, H.; Bouwmeester, H.; Weigel, S. Single particle ICP-MS combined with a data evaluation tool as a routine technique for the analysis of nanoparticles in complex matrices. J. Anal. Atomic Spectrom. 2015, 30, 1274–1285. [Google Scholar] [CrossRef]
- Pace, H.E.; Rogers, N.J.; Jarolimek, C.; Coleman, V.A.; Higgins, C.P.; Ranville, J.F. Determining Transport Efficiency for the Purpose of Counting and Sizing Nanoparticles via Single Particle Inductively Coupled Plasma Mass Spectrometry. Anal. Chem. 2011, 83, 9361–9369. [Google Scholar] [CrossRef] [PubMed]
- Turkevich, J. Colloidal gold. Part I. Gold Bull. 1985, 18, 86–91. [Google Scholar] [CrossRef]
- Gilles, M.; Brun, E.; Sicard-Roselli, C. Gold nanoparticles functionalization notably decreases radiosensitization through hydroxyl radical production under ionizing radiation. Colloids Surf. B Biointerfaces 2014, 123, 770–777. [Google Scholar] [CrossRef] [PubMed]
- Xia, X.; Yang, M.; Wang, Y.; Zheng, Y.; Li, Q.; Chen, J.; Xia, Y. Quantifying the Coverage Density of Poly(ethylene glycol) Chains on the Surface of Gold Nanostructures. ACS Nano 2012, 6, 512–522. [Google Scholar] [CrossRef] [PubMed]
- International Organization for Standardization. Nanotechnologies—Size Distribution and Concentration of Inorganic Nanoparticles in Aqueous Media via Single Particle Inductively Coupled Plasma Mass Spectrometry; XP ISO/TS 19590:20178; International Organization for Standardization: Geneva, Switzerland, 2017. [Google Scholar]
- Joint Committee for Guides in Metrology. Evaluation of Measurement Data—Guide to the Expression of Uncertainty in Measurement; JCGM 100:2008; Joint Committee for Guides in Metrology: Paris, France, 2008. [Google Scholar]
- Cohen, E.R.; Cvitas, T.; Frey, J.G.; Holmström, B.; Kuchitsu, K.; Marquardt, R.; Mills, I.; Pavese, F.; Quack, M.; Stohner, J.; et al. Quantities, Units and Symbols in Physical Chemistry, 3rd ed.; IUPAC Green Book; IUPAC & RSC Publishing: Cambridge, UK, 2008. [Google Scholar]
- Laborda, F.; Jiménez-Lamana, J.; Bolea, E.; Castillo, J.R. Critical considerations for the determination of nanoparticle number concentrations, size and number size distributions by single particle ICP-MS. J. Anal. Atomic Spectrom. 2013, 28, 1220–1232. [Google Scholar] [CrossRef]
- Hineman, A.; Stephan, C. Effect of dwell time on single particle inductively coupled plasma mass spectrometry data acquisition quality. J. Anal. Atomic Spectrom. 2014, 29, 1252–1257. [Google Scholar] [CrossRef]
- Liu, J.; Murphy, K.E.; MacCuspie, R.I.; Winchester, M.R. Capabilities of Single Particle Inductively Coupled Plasma Mass Spectrometry for the Size Measurement of Nanoparticles: A Case Study on Gold Nanoparticles. Anal. Chem. 2014, 86, 3405–3414. [Google Scholar] [CrossRef] [PubMed]
- Tuoriniemi, J.; Cornelis, G.; Hassellöv, M. Size discrimination and detection capabilities of single-particle ICP-MS for environmental analysis of silver nanoparticles. Anal. Chem. 2012, 29, 743–752. [Google Scholar] [CrossRef]
- International Organization for Standardization. Conformity Assessment—General Requirements for Proficiency Testing; NF EN ISO/CEI 17043:2010; International Organization for Standardization: Geneva, Switzerland, 2010. [Google Scholar]
- Lee, S.; Bi, X.; Reed, R.B.; Ranville, J.F.; Herckes, P.; Westerhoff, P. Nanoparticle Size Detection Limits by Single Particle ICP-MS for 40 Elements. Environ. Sci. Technol. 2014, 48, 10291–10300. [Google Scholar] [CrossRef] [PubMed]
- Espinosa, A.; Curcio, A.; Cabana, S.; Radtke, G.; Bugnet, M.; Kolosnjaj-Tabi, J.; Péchoux, C.; Alvarez-Lorenzo, C.; Botton, G.A.; Silva, A.K.A.; et al. Intracellular Biodegradation of Ag Nanoparticles, Storage in Ferritin, and Protection by a Au Shell for Enhanced Photothermal Therapy. ACS Nano 2018. [Google Scholar] [CrossRef] [PubMed]
- Cruje, C.; Chithrani, B.D. Integration of Peptides for Enhanced Uptake of PEGylated Gold Nanoparticles. J. Nanosci. Nanotechnol. 2015, 15, 2125–2131. [Google Scholar] [CrossRef] [PubMed]
- Unciti-Broceta, J.D.; Cano-Cortés, V.; Altea-Manzano, P.; Pernagallo, S.; Díaz-Mochón, J.J.; Sánchez-Martín, R.M. Number of Nanoparticles per Cell through a Spectrophotometric Method—A key parameter to Assess Nanoparticle-based Cellular Assays. Sci. Rep. 2015, 5, 10091. [Google Scholar] [CrossRef] [PubMed]
- Coulter, J.A.; Jain, S.; Butterworth, K.T.; Taggart, L.E.; Dickson, G.R.; McMahon, S.J.; Hyland, W.B.; Muir, M.F.; Trainor, C.; Hounsell, A.R.; et al. Cell type-dependent uptake, localization, and cytotoxicity of 1.9 nm gold nanoparticles. Int. J. Nanomed. 2012, 7, 2673–2685. [Google Scholar] [CrossRef] [PubMed]
| ICP-MS | sp-ICPMS | |
|---|---|---|
| Plasma power (W) | 1550 | 1550 |
| Plasma gas (mL/min) | 0.8 | 0.8 |
| Cool gas (mL/min) | 14 | 14 |
| Nebulizer gas (mL/min) | 1.0 | 1.0 |
| Spray chamber | Quartz cyclonic spray chamber | |
| Nebulizer | Quartz concentric | |
| Measurement mode | STD | STD |
| Isotope | 197 Au | 197 Au |
| Sample uptake rate (g/min) | ~0.4 | 0.259 ± 0.008 |
| Dwell time (ms) | 200 | 5 |
| Number of scans per measurement | 60 | 18000 |
| Sample | Gold Mass in Cell Medium | Number of Cells |
|---|---|---|
| MDAMB231 (control) | 0 µg | 2.21 × 106 |
| MDAMB231 (6 h) | 250 µg | 2.01 × 106 |
| MDAMB231 (24 h) | 250 µg | 2.25 × 106 |
| T47D (control) | 0 µg | 1.8 × 106 |
| T47D (6 h) | 250 µg | 2.49 × 106 |
| T47D (24 h) | 250 µg | 2.21 × 106 |
| sp-ICPMS | ICPMS | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cell Samples | d50 nm | davg nm | Cp 107 Part | SD | Cm µg | SD | U (k = 2) | N | Cm µg | SD | U (k = 2) | N | EN |
| MDAMB231 (control) | - | - | - | - | <0.009 | - | - | 1 | <0.006 | - | - | 1 | - |
| MDAMB231 (6 h) | 32 | 32 | 4.8 | - | 0.019 | - | 0.003 | 1 | 0.020 | 0.001 | 0.004 | 2 | 0.3 |
| MDAMB231 (24 h) | 33 | 33 | 41.5 | 11.8 | 0.152 | 0.009 | 0.031 | 2 | 0.123 | 0.001 | 0.007 | 2 | 0.9 |
| T47D (control) | - | - | - | - | <0.004 | - | - | 1 | <0.006 | - | - | 1 | - |
| T47D (6 h) | 32 | 32 | 3.3 | 0.4 | 0.014 | 0.001 | 0.003 | 2 | 0.015 | 0.001 | 0.003 | 2 | 0.2 |
| T47D (24 h) | 32 | 32 | 14.9 | 5 | 0.057 | 0.014 | 0.029 | 2 | 0.067 | 0.007 | 0.013 | 2 | 0.3 |
| T47D (test) | 31 | 31 | 56 | - | 0.194 | - | 0.041 | 1 | 0.223 | - | 0.037 | 1 | 0.6 |
| Cell Samples | Part/Cell (a) | Part/Cell (b) | Part/Cell (c) | Mean | RSD |
|---|---|---|---|---|---|
| MDAMB231 (6 h) | 24 | 28 | 30 | 27 | 11% |
| MDAMB231 (24 h) | 184 | 190 | 154 | 176 | 11% |
| T47D (6 h) | 13 | 17 | 18 | 16 | 15% |
| T47D (24 h) | 67 | 77 | 90 | 78 | 15% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Noireaux, J.; Grall, R.; Hullo, M.; Chevillard, S.; Oster, C.; Brun, E.; Sicard-Roselli, C.; Loeschner, K.; Fisicaro, P. Gold Nanoparticle Uptake in Tumor Cells: Quantification and Size Distribution by sp-ICPMS. Separations 2019, 6, 3. https://doi.org/10.3390/separations6010003
Noireaux J, Grall R, Hullo M, Chevillard S, Oster C, Brun E, Sicard-Roselli C, Loeschner K, Fisicaro P. Gold Nanoparticle Uptake in Tumor Cells: Quantification and Size Distribution by sp-ICPMS. Separations. 2019; 6(1):3. https://doi.org/10.3390/separations6010003
Chicago/Turabian StyleNoireaux, Johanna, Romain Grall, Marie Hullo, Sylvie Chevillard, Caroline Oster, Emilie Brun, Cécile Sicard-Roselli, Katrin Loeschner, and Paola Fisicaro. 2019. "Gold Nanoparticle Uptake in Tumor Cells: Quantification and Size Distribution by sp-ICPMS" Separations 6, no. 1: 3. https://doi.org/10.3390/separations6010003
APA StyleNoireaux, J., Grall, R., Hullo, M., Chevillard, S., Oster, C., Brun, E., Sicard-Roselli, C., Loeschner, K., & Fisicaro, P. (2019). Gold Nanoparticle Uptake in Tumor Cells: Quantification and Size Distribution by sp-ICPMS. Separations, 6(1), 3. https://doi.org/10.3390/separations6010003





