Characterization of the Natural Colloidal TiO2 Background in Soil
Abstract
1. Introduction
2. Materials and Methods
2.1. Soil Sampling and Characterization
2.2. X-ray Fluorescence Analysis (XRFA)
2.3. Inductively Coupled Plasma Optical Emission Spectroscopy (ICP-OES)
2.4. Extraction of Colloids
2.5. Hydrodynamic Chromatography Coupled to Inductively Coupled Mass Spectrometry (HDC-ICP-MS)
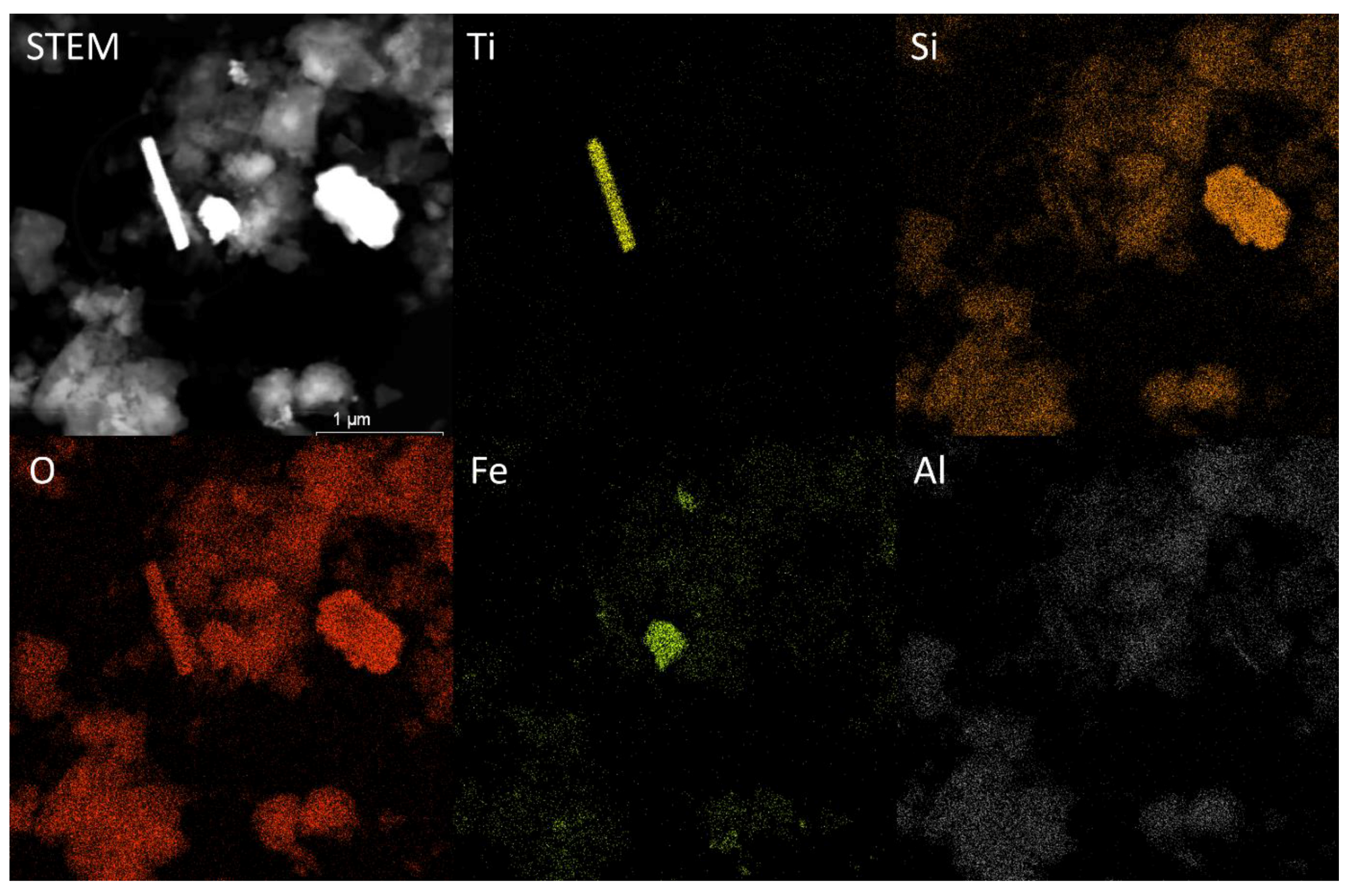
2.6. Scanning Electron Microscopy (SEM)
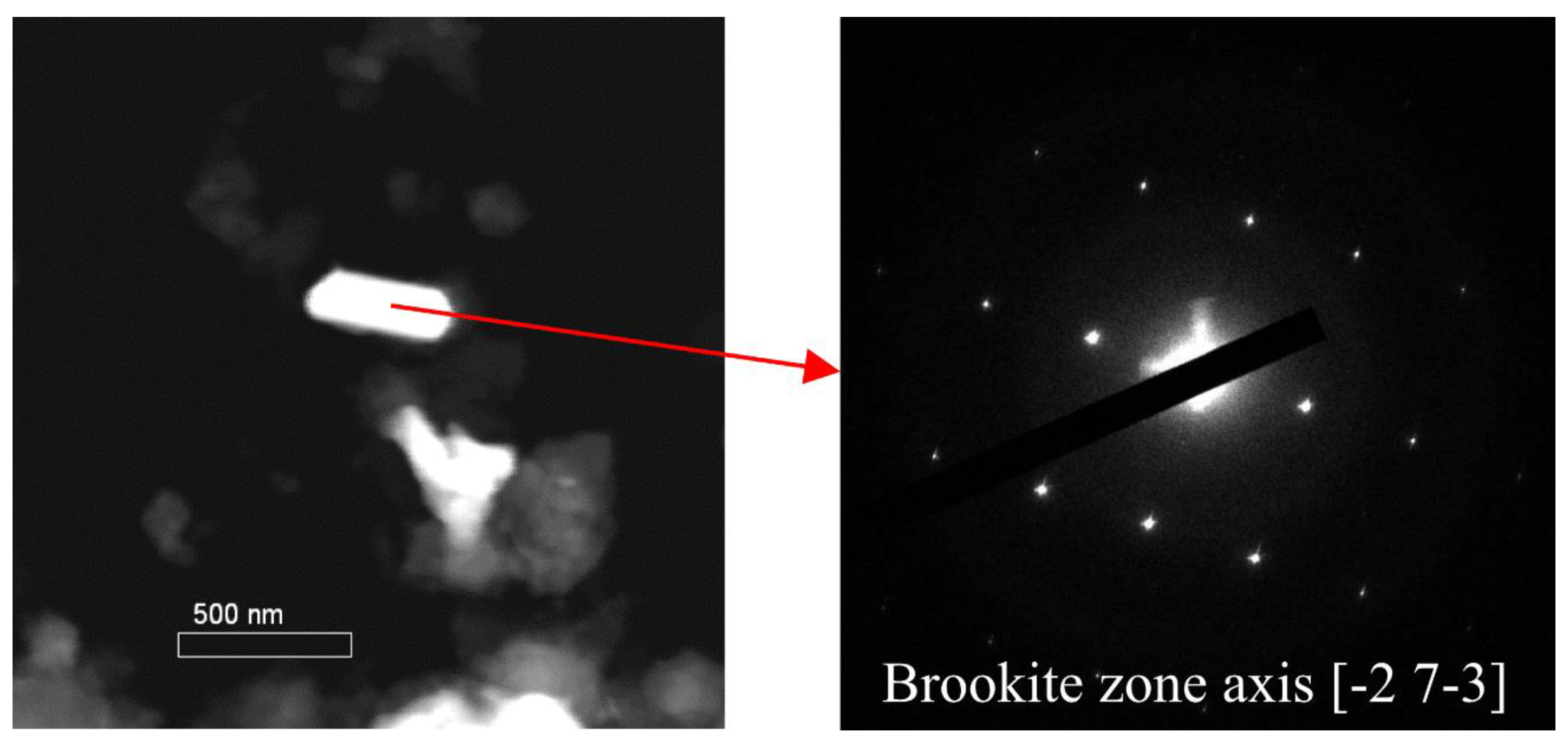
2.7. High Resolution Transmission Electron Microscopy (HR-TEM)
3. Results and Discussion
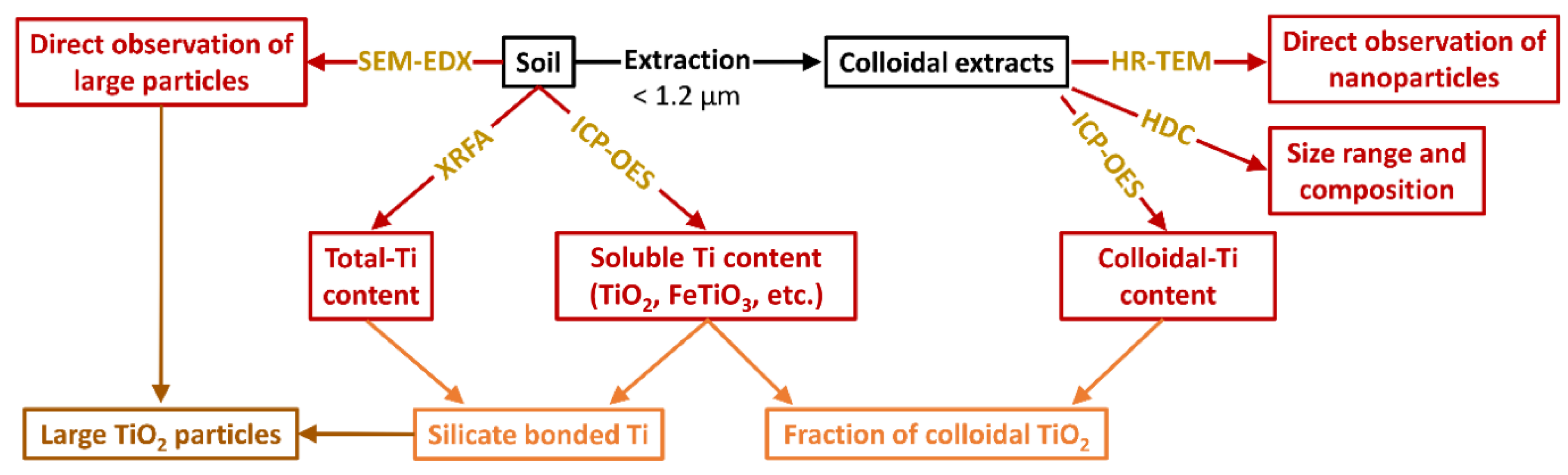
3.1. Analytical Workflow
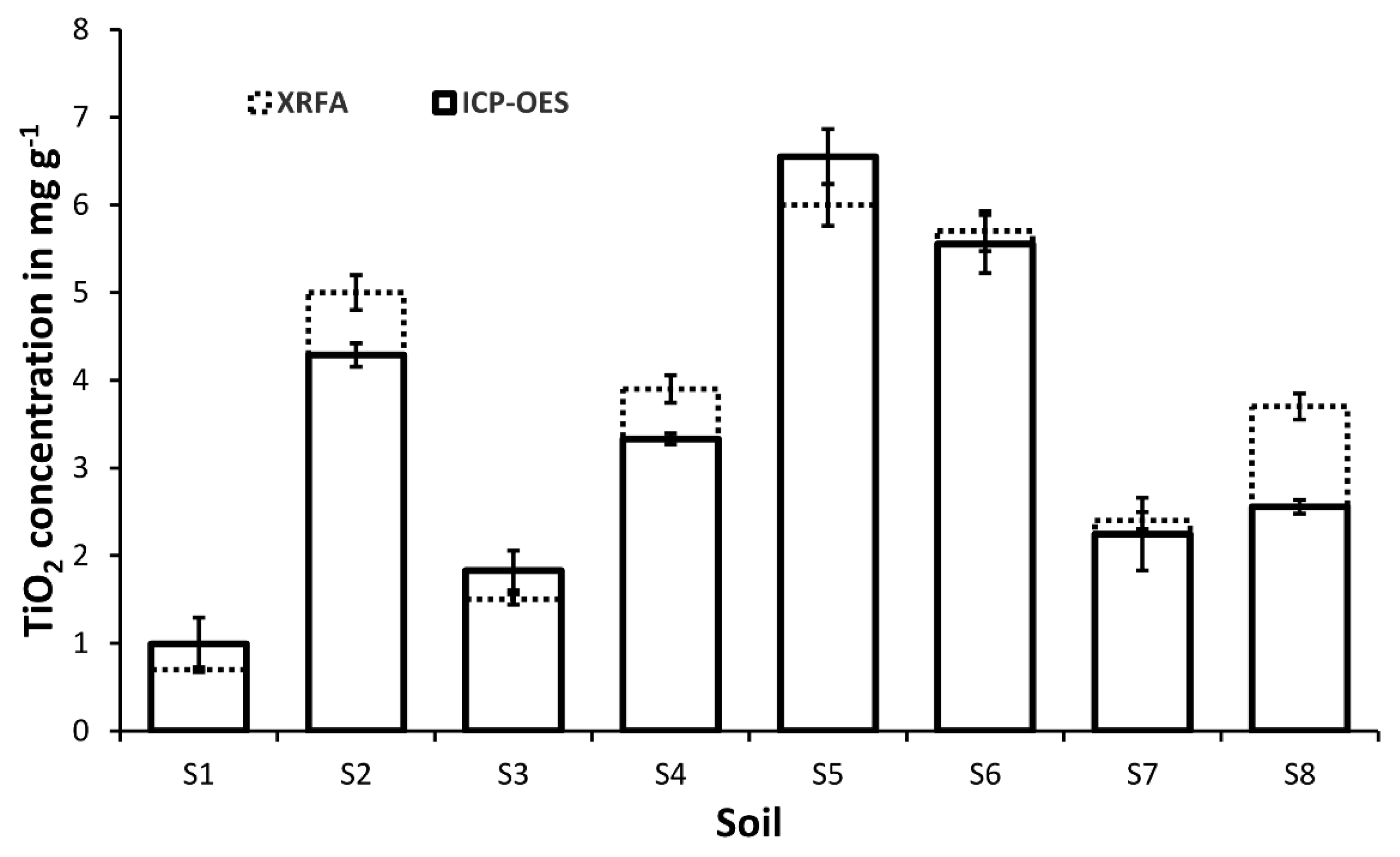
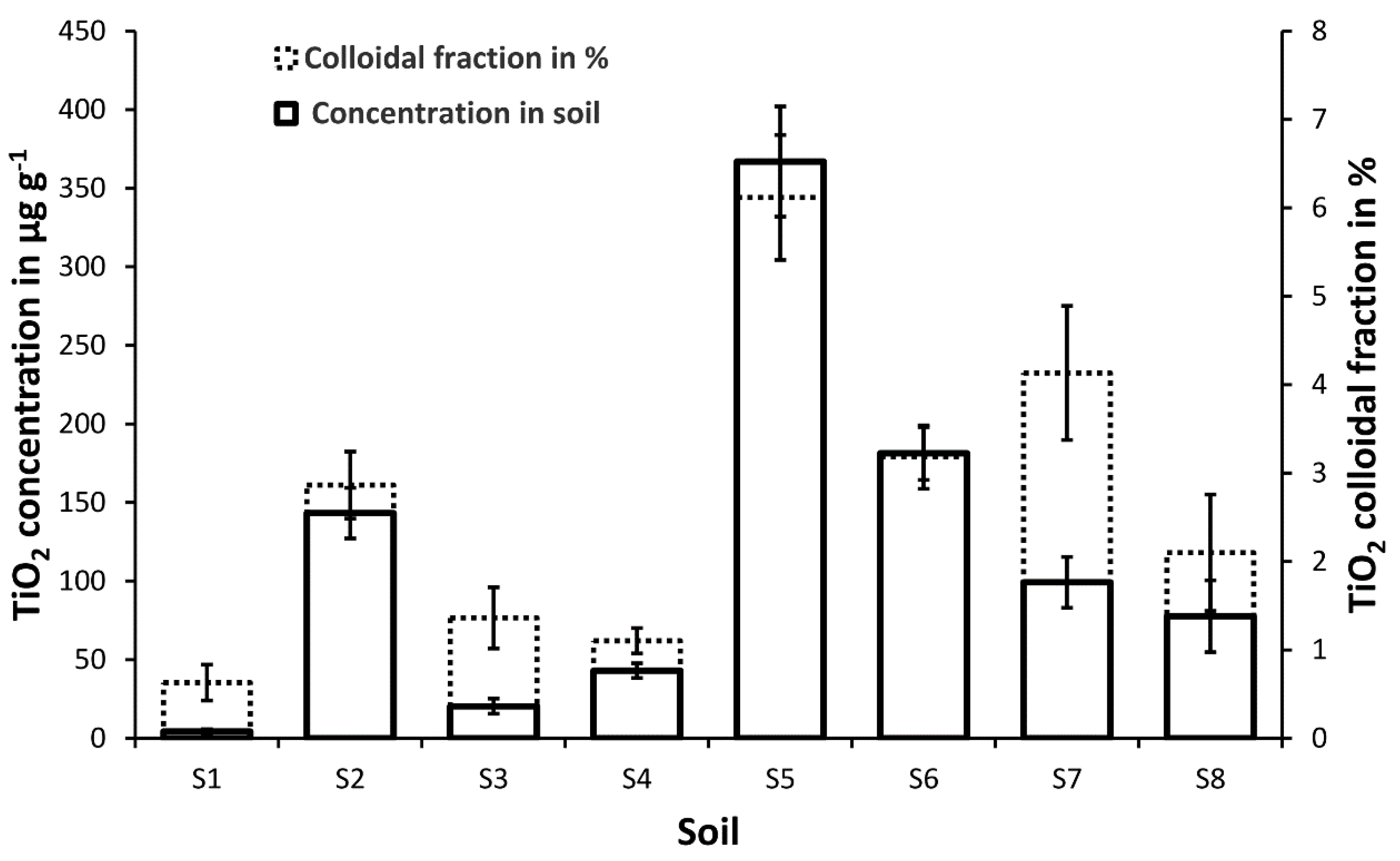
3.2 Ti-Concentration in Soils
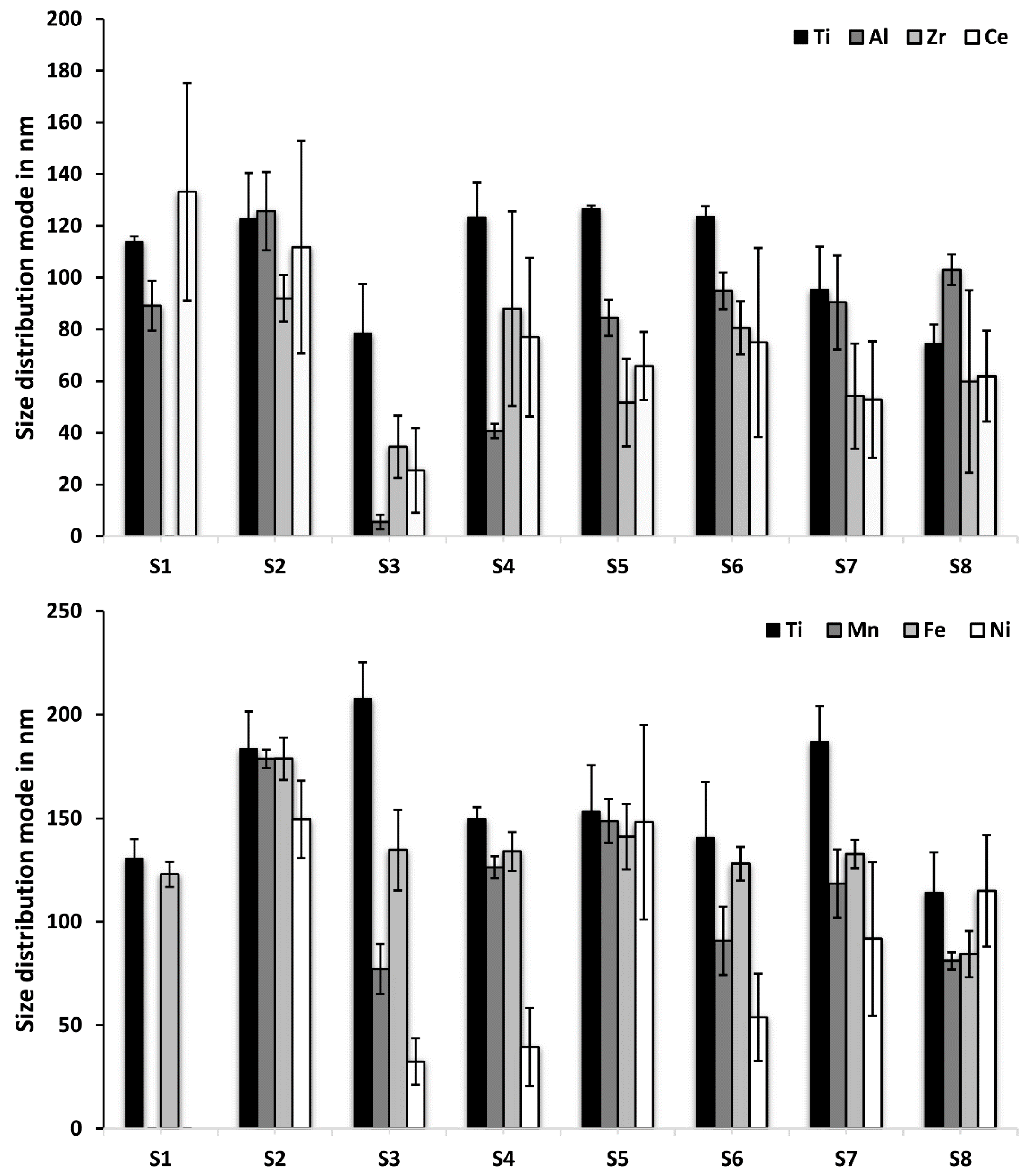
3.3 Size and Composition of Ti-Containing Particles
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bairi, V.G.; Lim, J.-H.; Fong, A.; Linder, S.W. Size characterization of metal oxide nanoparticles in commercial sunscreen products. J. Nanopart. Res. 2017, 19, 256. [Google Scholar] [CrossRef]
- Philippe, A.; Košík, J.; Welle, A.; Guigner, J.-M.; Clemens, O.; Schaumann, G.E. Extraction and characterization methods for titanium dioxide nanoparticles from commercialized sunscreens. Environ. Sci. Nano 2018, 5, 191–202. [Google Scholar] [CrossRef]
- Weir, A.; Westerhoff, P.; Fabricius, L.; Hristovski, K.; von Goetz, N. Titanium dioxide nanoparticles in food and personal care products. Environ. Sci. Technol. 2012, 46, 2242–2250. [Google Scholar] [CrossRef] [PubMed]
- Baalousha, M.; Yang, Y.; Vance, M.E.; Colman, B.P.; McNeal, S.; Xu, J.; Blaszczak, J.; Steele, M.; Bernhardt, E.; Hochella, M.F. Outdoor urban nanomaterials: The emergence of a new, integrated, and critical field of study. Sci. Total Environ. 2016, 557, 740–753. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Mao, S.S. Titanium dioxide nanomaterials: Synthesis, properties, modifications, and applications. Chem. Rev. 2007, 107, 2891–2959. [Google Scholar] [CrossRef] [PubMed]
- Kiser, M.; Westerhoff, P.; Benn, T.; Wang, Y.; Perez-Rivera, J.; Hristovski, K. Titanium nanomaterial removal and release from wastewater treatment plants. Environ. Sci. Technol. 2009, 43, 6757–6763. [Google Scholar] [CrossRef] [PubMed]
- Gondikas, A.P.; von der Kammer, F.; Reed, R.B.; Wagner, S.; Ranville, J.F.; Hofmann, T. Release of TiO2 nanoparticles from sunscreens into surface waters: A one-year survey at the old Danube recreational Lake. Environ. Sci. Technol. 2014, 48, 5415–5422. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.H.; Cohen, Y. Multimedia environmental distribution of engineered nanomaterials. Environ. Sci. Technol. 2014, 48, 3281–3292. [Google Scholar] [CrossRef] [PubMed]
- Gottschalk, F.; Lassen, C.; Kjoelholt, J.; Christensen, F.; Nowack, B. Modeling flows and concentrations of nine engineered nanomaterials in the Danish environment. Int. J. Environ. Res. Public Health 2015, 12, 5581–5602. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.K. Aggregation and toxicity of titanium dioxide nanoparticles in aquatic environment—A Review. J. Environ. Sci. Health Part A 2009, 44, 1485–1495. [Google Scholar] [CrossRef] [PubMed]
- Kulacki, K.J.; Cardinale, B.J.; Keller, A.A.; Bier, R.; Dickson, H. How do stream organisms respond to, and influence, the concentration of titanium dioxide nanoparticles? A mesocosm study with algae and herbivores. Environ. Toxicol. Chem. 2012, 31, 2414–2422. [Google Scholar] [CrossRef] [PubMed]
- Cornelis, G.; Hund-Rinke, K.; Kuhlbusch, T.; Van den Brink, N.; Nickel, C. Fate and bioavailability of engineered nanoparticles in soils: A review. Crit. Rev. Environ. Sci. Technol. 2014, 44, 2720–2764. [Google Scholar] [CrossRef]
- Gondikas, A.; von der Kammer, F.; Kaegi, R.; Borovinskaya, O.; Neubauer, E.; Navratilova, J.; Praetorius, A.; Cornelis, G.; Hofmann, T. Where is the nano? Analytical approaches for the detection and quantification of TiO2 engineered nanoparticles in surface waters. Environ. Sci. Nano 2018, 5, 313–326. [Google Scholar] [CrossRef]
- Schwertfeger, D.M.; Velicogna, J.R.; Jesmer, A.H.; Saatcioglu, S.; McShane, H.A.; Scroggins, R.P.; Princz, J.I. Extracting metallic nanoparticles from soils for quantitative analysis: Method development using engineered silver nanoparticles and SP-ICP-MS. Anal. Chem. 2017, 89, 2505–2513. [Google Scholar] [CrossRef] [PubMed]
- Mahdi, K.N.; Peters, R.J.; Klumpp, E.; Bohme, S.; Van der Ploeg, M.; Ritsema, C.; Geissen, V. Silver nanoparticles in soil: Aqueous extraction combined with single-particle ICP-MS for detection and characterization. Environ. Nanotechnol. Monit. Manag. 2017, 7, 24–33. [Google Scholar] [CrossRef]
- Geochemical Atlas of Europe. Available online: http://weppi.gtk.fi/publ/foregsatlas/ (accessed on 19 October 2018).
- Fitzpatrick, R.W.; Chittleborough, D. Titanium and zirconium minerals. In Soil Mineralogy with Environmental Applications; Soil Science Society of America: Madison, WI, USA, 2002; pp. 667–690. [Google Scholar]
- Theng, B.K.; Yuan, G. Nanoparticles in the soil environment. Elements 2008, 4, 395–399. [Google Scholar] [CrossRef]
- Li, W.; He, Y.; Wu, J.; Xu, J. Extraction and characterization of natural soil nanoparticles from Chinese soils. Eur. J. Soil Sci. 2012, 63, 754–761. [Google Scholar] [CrossRef]
- Waychunas, G.A.; Kim, C.S.; Banfield, J.F. Nanoparticulate iron oxide minerals in soils and sediments: Unique properties and contaminant scavenging mechanisms. J. Nanopart. Res. 2005, 7, 409–433. [Google Scholar] [CrossRef]
- Tang, Z.; Wu, L.; Luo, Y.; Christie, P. Size fractionation and characterization of nanocolloidal particles in soils. Environ. Geochem. Health 2009, 31, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Beckett, R. Development of SdFFF-ETAAS for characterising soil and sediment colloids. Analyst 2001, 126, 1588–1593. [Google Scholar] [CrossRef]
- Ryzak, M.; Bieganowski, A. Methodological aspects of determining soil particle-size distribution using the laser diffraction method. J. Plant Nutr. Soil Sci. 2011, 174, 624–633. [Google Scholar] [CrossRef]
- Navratilova, J.; Praetorius, A.; Gondikas, A.; Fabienke, W.; von der Kammer, F.; Hofmann, T. Detection of engineered copper nanoparticles in soil using single particle ICP-MS. Int. J. Environ. Res. Public Health 2015, 12, 15756–15768. [Google Scholar] [CrossRef] [PubMed]
- El Hadri, H.; Louie, S.M.; Hackley, V.A. Assessing the interactions of metal nanoparticles in soil and sediment matrices-a quantitative analytical multi-technique approach. Environ. Sci. Nano 2018, 5, 203–214. [Google Scholar] [CrossRef]
- Loomis, G.A. Grain size of whiteware clays as determined by the Andreasen pipette. J. Am. Ceram. Soc. 1938, 21, 393–399. [Google Scholar] [CrossRef]
- Vinther, E.; Lasson, M. Grain-size measurements of kaolins and clays. Berl. Deut. Keram. Ges. 1933, 14, 259–279. [Google Scholar]
- Nickel, C.; Hellack, B.; Gartiser, S.; Schiwy, A.; Schäffer, A.; Gabsch, S.; Stintz, M.; Erdinger, L.; Kuhlbusch, T.A.J. Fate and Behaviour of TiO2 Nanomaterials in the Environment, Influenced by Their Shape, Size and Surface Area; UBA Final Report 25/2012; Environmental Protection Agency: Berlin, Germany, 2012. [Google Scholar]
- Rakcheev, D.; Philippe, A.; Schaumann, G.E. Hydrodynamic chromatography coupled with single particle-inductively coupled plasma mass spectrometry for investigating nanoparticles agglomerates. Anal. Chem. 2013, 85, 10643–10647. [Google Scholar] [CrossRef] [PubMed]
- Wintermyer, A.; Kinter, E.B. Dispersing Agents for Particle-Size Analysis of Soils; Highway Research Board Bulletin; Highway Research Board: Washington, DC, USA, 1955. [Google Scholar]
- Stegeman, G.; Kraak, J.C.; Poppe, H. Dispersion in packed-column hydrodynamic chromatography. J. Chromatogr. A 1993, 634, 149–159. [Google Scholar] [CrossRef]
- Striegel, A.M.; Brewer, A.K. Hydrodynamic Chromatography. Annu. Rev. Anal. Chem. 2012, 5, 15–34. [Google Scholar] [CrossRef] [PubMed]
- Tiede, K.; Boxall, A.B.; Tiede, D.; Tear, S.P.; David, H.; Lewis, J. A robust size-characterisation methodology for studying nanoparticle behaviour in “real”environmental samples, using hydrodynamic chromatography coupled to ICP-MS. J. Anal. Atomic Spectrom. 2009, 24, 964–972. [Google Scholar] [CrossRef]
- Tiede, K.; Boxall, A.B.; Wang, X.; Gore, D.; Tiede, D.; Baxter, M.; David, H.; Tear, S.P.; Lewis, J. Application of hydrodynamic chromatography-ICP-MS to investigate the fate of silver nanoparticles in activated sludge. J. Anal. Atomic Spectrom. 2010, 25, 1149–1154. [Google Scholar] [CrossRef]
- Philippe, A.; Schaumann, G.E. Evaluation of Hydrodynamic Chromatography Coupled with UV-Visible, Fluorescence and Inductively Coupled Plasma Mass Spectrometry Detectors for Sizing and Quantifying Colloids in Environmental Media. PLoS ONE 2014, 9, e90559. [Google Scholar] [CrossRef] [PubMed]
- Philippe, A.; Gangloff, M.; Rakcheev, D.; Schaumann, G. Evaluation of hydrodynamic chromatography coupled with inductively coupled plasma mass spectrometry detector for analysis of colloids in environmental media-effects of colloid composition, coating and shape. Anal. Methods 2014, 6, 8722–8728. [Google Scholar] [CrossRef]
- Roman, M.; Rigo, C.; Castillo-Michel, H.; Munivrana, I.; Vindigni, V.; Mičetić, I.; Benetti, F.; Manodori, L.; Cairns, W.R. Hydrodynamic chromatography coupled to single-particle ICP-MS for the simultaneous characterization of AgNPs and determination of dissolved Ag in plasma and blood of burn patients. Anal. Bioanal. Chem. 2016, 408, 5109–5124. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Wang, X.; Chen, C.; Sun, A. Effect of soil humic and fulvic acids, pH and ionic strength on Th(IV) sorption to TiO2 nanoparticles. Appl. Radiat. Isot. 2007, 65, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Yin, D.; Zhu, S.; Hu, X. Adsorption of cadmium (II) on humic acid coated titanium dioxide. J. Colloid Interface Sci. 2012, 367, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Burleson, D.J.; Driessen, M.D.; Penn, R.L. On the characterization of environmental nanoparticles. J. Environ. Sci. Health Part A 2005, 39, 2707–2753. [Google Scholar] [CrossRef]
- Bourgeault, A.; Cousin, C.; Geertsen, V.; Cassier-Chauvat, C.; Chauvat, F.; Durupthy, O.; Chaneac, C.; Spalla, O. The challenge of studying TiO2 nanoparticle bioaccumulation at environmental concentrations: Crucial use of a stable isotope tracer. Environ. Sci. Technol. 2015, 49, 2451–2459. [Google Scholar] [CrossRef] [PubMed]


| Soil | Soil Description | pH | EC (µS m−1) | N (%) | C (%) | H (%) | Clay (%) | Silt (%) | Sand (%) |
|---|---|---|---|---|---|---|---|---|---|
| S1 | Commercial sand | 6.78 | 12 | <LOD | <LOD | 0.11 | 0.0 | 0.0 | 100.0 |
| S2 | Calcareous soil | 6.70 | 464 | 0.26 | 6.03 | 0.85 | 8.12 | 58.58 | 33.30 |
| S3 | Agricultural soil | 5.00 | 166 | 0.06 | 0.67 | 0.20 | 2.6 | 12.7 | 84.7 |
| S4 | Agricultural soil | 5.80 | 203 | 0.08 | 0.67 | 0.26 | 6.4 | 36.0 | 57.6 |
| S5 | Agricultural soil | 7.10 | 176 | 0.18 | 1.73 | 0.86 | 41.7 | 36.0 | 22.3 |
| S6 | Flooded meadow | 5.94 | 154 | 0.60 | 6.28 | 1.15 | 41.9 | 34.2 | 23.9 |
| S7 | Peat soil | 6.70 | 1687 | 1.22 | 9.16 | 1.51 | 29.0 | 62.5 | 34.6 |
| S8 | Tropical native soil | 3.91 | 10 | 0.03 | 0.02 | 0.14 | 5.1 | 26.6 | 68.4 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Philippe, A.; Campos, D.A.; Guigner, J.-M.; Buchmann, C.; Diehl, D.; Schaumann, G.E. Characterization of the Natural Colloidal TiO2 Background in Soil. Separations 2018, 5, 50. https://doi.org/10.3390/separations5040050
Philippe A, Campos DA, Guigner J-M, Buchmann C, Diehl D, Schaumann GE. Characterization of the Natural Colloidal TiO2 Background in Soil. Separations. 2018; 5(4):50. https://doi.org/10.3390/separations5040050
Chicago/Turabian StylePhilippe, Allan, Daniel Armando Campos, Jean-Michel Guigner, Christian Buchmann, Dörte Diehl, and Gabriele E. Schaumann. 2018. "Characterization of the Natural Colloidal TiO2 Background in Soil" Separations 5, no. 4: 50. https://doi.org/10.3390/separations5040050
APA StylePhilippe, A., Campos, D. A., Guigner, J.-M., Buchmann, C., Diehl, D., & Schaumann, G. E. (2018). Characterization of the Natural Colloidal TiO2 Background in Soil. Separations, 5(4), 50. https://doi.org/10.3390/separations5040050