Differential Behavior of Salt and Organic Matter Passage in 2-Pass RO Systems for Ultrapure Water Production
Abstract
1. Introduction
2. Methods
2.1. 4-Inch RO Membrane Module Test
2.2. Characterization of 4-Inch Membrane Module Performance
2.3. Water Quality Analysis
2.3.1. Offline TOC
2.3.2. Online TOC
3. Results
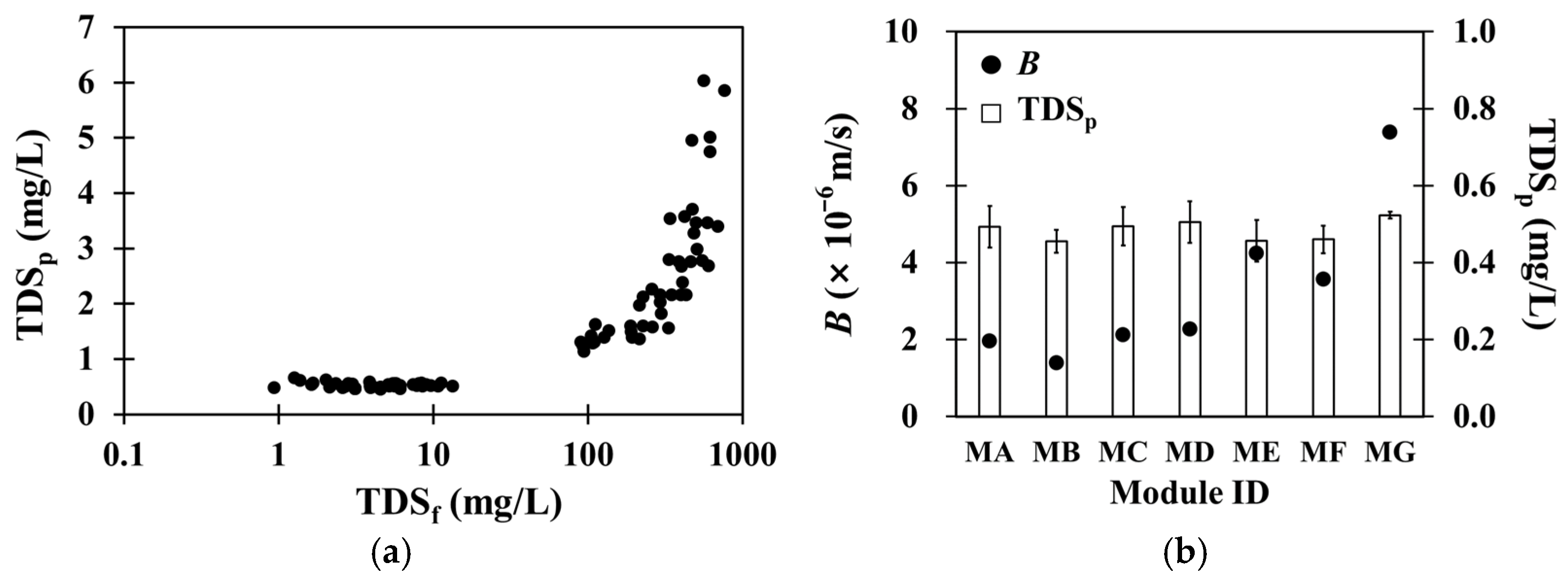
3.1. Salt Passage Through the Second-Pass RO Membranes in a Wide Range of Salt Permeabilities
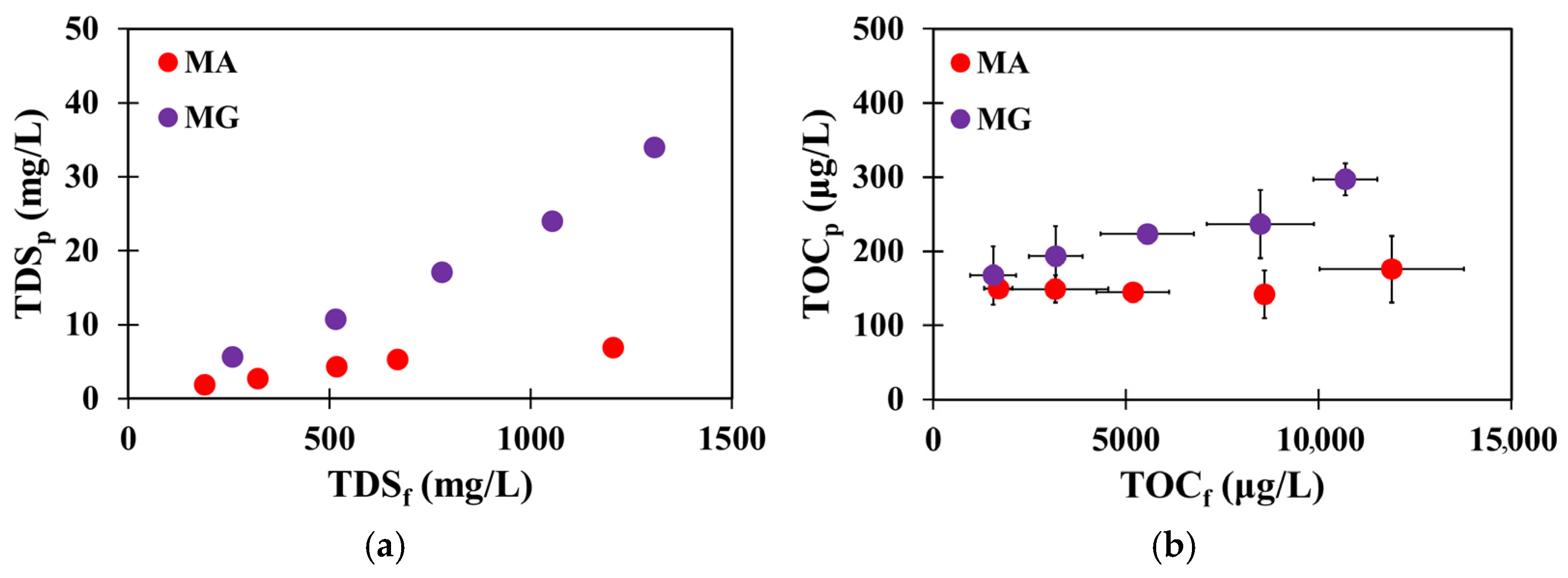
3.2. Organic Matter Passage Through the Second-Pass RO Membranes in a Wide Range of Water and Salt Permeabilities
3.3. Evaluating 2-Pass Membrane Configurations for Balancing RO Feed Pressure and TOC Control Under High-Recovery Rate Conditions
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| UPW | Ultrapure water |
| RO | Reverse osmosis |
| CEDI | Continuous electrodeionization |
| TOC | Total organic carbon |
| TDS | Total dissolved solids |
| GAC | Granular activated carbon |
| VFD | Variable frequency drive |
| C-PVC | Chlorinated polyvinyl chloride |
| BWRO | Brackish water reverse osmosis |
| CF | Concentration factor |
| NDIR | Non-Dispersive Infrared |
| ASTM | American society for testing and materials International |
| A value | Water permeability coefficient |
| B value | Salt permeability coefficient |
| CP | Concentration polarization |
| Qf | Feed flow rate |
| Qp | Permeate flow rate |
| Qc | Concentration flow rate |
| Cf | Solute concentration of feed |
| Cp | Solute concentration of permeate |
| Cc | Solute concentration of concentration |
| S | Membrane area |
| Jw | Permeate flux |
| Rs | Salt rejection |
| Cfb | Average bulk concentration |
| Y | Recovery |
| I | Reference electrolyte |
| Δπ | Osmosis pressure |
| K | Absolute temperature |
| ΔP | membrane pressure |
| Js | Solute flux |
| Cm | membrane surface concentration |
| β | CP factor |
| TDSf | Feed total dissolved solids |
| TDSp | Permeate total dissolved solids |
| TOCf | Feed total organic carbon |
| TOCp | Permeate total organic carbon |
References
- Shim, J.; Lee, S.; Park, S.; Moon, J.; Lee, C.; Cho, K.H. Optimizing membrane cleaning strategy of industrial reverse osmosis process using long sequence time-series forecasting. Desalination 2025, 614, 119193. [Google Scholar] [CrossRef]
- Wang, Q.; Luo, L.; Huang, N.; Wang, W.; Rong, Y.; Wang, Z.; Yuan, Y.; Xu, A.; Xiong, J.; Wu, Q.; et al. Evolution of low molecular weight organic compounds during ultrapure water production process: A pilot-scale study. Sci. Total Environ. 2022, 830, 154713. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Yang, Y.; Ngo, H.H.; Guo, W.; Wen, H.; Wang, X.; Zhang, J.; Long, T. A critical review on challenges and trend of ultrapure water production process. Sci. Total Environ. 2021, 785, 147254. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Kim, D.; Kim, S.; Im, S.; Lee, C.-K.; Park, K.-D.; Kim, M.; Chon, K.; Woo, Y.C.; Rho, H. Comprehensive evaluation of a pilot-scale semiconductor wastewater reuse process using ultrafiltration and two-stage reverse osmosis for securing intake water resource in ultrapure water production. Desalination 2025, 613, 119115. [Google Scholar] [CrossRef]
- Lee, H.; Jin, Y.; Hong, S. Recent transitions in ultrapure water (UPW) technology: Rising role of reverse osmosis (RO). Desalination 2016, 399, 185–197. [Google Scholar] [CrossRef]
- Jin, Y.; Lee, H.; Zhan, M.; Hong, S. UV radiation pretreatment for reverse osmosis (RO) process in ultrapure water (UPW) production. Desalination 2018, 439, 138–146. [Google Scholar] [CrossRef]
- Rho, H.; Chon, K.; Cho, J. An autopsy study of a fouled reverse osmosis membrane used for ultrapure water production. Water 2019, 11, 1116. [Google Scholar] [CrossRef]
- Wu, Y.-P.; Wu, Q.-Y.; Zhou, M.-Q.; Chen, Y.-L.; Lee, M.-Y.; Wang, W.-L. Insight into the transport mechanism of low-molecular-weight charge-neutral organics in reverse osmosis: The critical role of molecular polarity. Environ. Sci. Technol. 2026, 60, 1517–1526. [Google Scholar] [CrossRef]
- Kim, D.; Lee, H.; Jeon, J.; Kim, S. High recovery design of reverse osmosis process with high permeate water quality and low wastewater discharge for ultra-pure water production. Desalination 2024, 592, 118149. [Google Scholar] [CrossRef]
- Zhao, P.; Bai, Y.; Liu, B.; Chang, H.; Cao, Y.; Fang, J. Process optimization for producing ultrapure water with high resistivity and low total organic carbon. Process Saf. Environ. Prot. 2019, 126, 232–241. [Google Scholar] [CrossRef]
- Park, D.; Yeo, I.-H.; Lee, J.; Kim, H.; Choi, S.; Kang, S. Combined impacts of aluminum and silica ions on RO membrane fouling in full-scale ultrapure water production facilities. Desalination 2024, 577, 117380. [Google Scholar] [CrossRef]
- Muhyuddin, M.; Santoro, C.; Osmieri, L.; Ficca, V.C.A.; Friedman, A.; Yassin, K.; Pagot, G.; Negro, E.; Konovalova, A.; Lindquist, G.; et al. Anion-exchange-membrane electrolysis with alkali-free water feed. Chem. Rev. 2025, 125, 6906–6976. [Google Scholar] [CrossRef] [PubMed]
- Chekli, L.; Phuntsho, S.; Kim, J.E.; Kim, J.; Choi, J.Y.; Choi, J.-S.; Kim, S.; Kim, J.H.; Hong, S.; Sohn, J.; et al. A comprehensive review of hybrid forward osmosis systems: Performance, applications and future prospects. J. Membr. Sci. 2016, 497, 430–449. [Google Scholar] [CrossRef]
- Phuntsho, S.; Shon, H.K.; Hong, S.; Lee, S.; Vigneswaran, S. A novel low energy fertilizer driven forward osmosis desalination for direct fertigation: Evaluating the performance of fertilizer draw solutions. J. Membr. Sci. 2011, 375, 172–181. [Google Scholar] [CrossRef]
- Zhao, S.; Liao, Z.; Fane, A.; Li, J.; Tang, C.; Zheng, C.; Lin, J.; Kong, L. Engineering antifouling reverse osmosis membranes: A review. Desalination 2021, 499, 114857. [Google Scholar] [CrossRef]
- Kang, Y.; Kwon, J.; Kim, J.; Hong, S. Fate of low molecular weight organic matters in reverse osmosis and vacuum ultraviolet process for high-quality ultrapure water production in the semiconductor industry. J. Clean. Prod. 2023, 423, 138714. [Google Scholar] [CrossRef]
- Shrestha, J.; Li, J. Influence of permeate from domestic reverse osmosis filters on lead pipes corrosion and plastic pipes leaching. J. Water Process Eng. 2017, 18, 126–133. [Google Scholar] [CrossRef]
- Kim, J.; Hong, S. A novel single-pass reverse osmosis configuration for high-purity water production and low energy consumption in seawater desalination. Desalination 2018, 429, 142–154. [Google Scholar] [CrossRef]
- Busan Metropolitan City Waterworks. Water Quality Report Archive. Available online: https://www.busan.go.kr/water/qreport (accessed on 14 February 2026).
- Armendáriz-Ontiveros, M.M.; Dévora-Isiordia, G.E.; Rodríguez-López, J.; Sánchez-Duarte, R.G.; Álvarez-Sánchez, J.; Villegas-Peralta, Y.; Martínez-Macias, M.d.R. Effect of Temperature on Energy Consumption and Polarization in Reverse Osmosis Desalination Using a Spray-Cooled Photovoltaic System. Energies 2022, 15, 7787. [Google Scholar] [CrossRef]
- ASTM D4516-00(2010); Standard Practice for Standardizing Reverse Osmosis Performance Data. ASTM International: West Conshohocken, PA, USA, 2010. [CrossRef]
- Jeon, J.; Kim, D.; Kim, N.; Kim, S. Applicability and limitation of the industrial reverse osmosis system simulators. Desalination 2023, 549, 116358. [Google Scholar] [CrossRef]
- Wang, L.; Cao, T.; Dykstra, J.E.; Porada, S.; Biesheuvel, P.M.; Elimelech, M. Salt and Water Transport in Reverse Osmosis Membranes: Beyond the Solution-Diffusion Model. Environ. Sci. Technol. 2021, 55, 16665–16675. [Google Scholar] [CrossRef] [PubMed]





| Parameter | Concentration (mg/L) |
|---|---|
| Na+ | 18.0 ± 9.3 |
| Ca2+ | 23.1 ± 6.0 |
| Mg2+ | 4.6 ± 1.2 |
| K+ | 4.3 ± 1.0 |
| Cl− | 28.8 ± 8.0 |
| SO42− | 35.0 ± 7.1 |
| TDS | 172.0 ± 52.0 |
| TOC | 1.293 ± 0.246 |
| Permeate Flux (Lm−2h−1 (LMH)) | Temperature (°C) | Module Recovery (%) | |
|---|---|---|---|
| First-pass RO | 19 | 25 ± 2 | 25 |
| Second-pass RO | 23 | 25 ± 1 | 15 |
| Manufacturer | Module | Area (m2) | Salt Rejection (%) | Permeate Flow Rate (m3d−1) | Module ID |
|---|---|---|---|---|---|
| Toray Advanced Materials Korea Inc. (Seoul, Republic of Korea) | RE4040-BE | 7.9 | 99.7 a | 9.1 a | MA |
| Toray Membrane (Poway, CA, USA) | TM710D | 8.0 | 99.8 b | 9.8 b | MB |
| DuPontTM (Wilmington, DE, USA) | BW30 PRO4040 | 7.9 | 99.7 c | 9.8 c | MC |
| Toray Advanced Materials Korea Inc. (Seoul, Republic of Korea) | RE4040-BLR | 7.9 | 99.6 d | 7.9 d | MD |
| Toray Advanced Materials Korea Inc. (Seoul, Republic of Korea) | RE4040-BLN | 7.9 | 99.4 e | 7.9 e | ME |
| Toray Membrane (Poway, CA, USA) | TMG10D | 8.0 | 99.7 f | 10.0 f | MF |
| LG Chem (Seoul, Republic of Korea) | BW4040-UES | 7.9 | 99.0 g | 10.2 g | MG |
| Module ID | A (×10−11 m/s/Pa) | B (×10−6 m/s) |
|---|---|---|
| MA | 0.98 | 1.97 |
| MB | 1.04 | 1.39 |
| MC | 1.05 | 2.12 |
| MD | 1.34 | 2.28 |
| ME | 1.61 | 4.25 |
| MF | 1.71 | 3.56 |
| MG | 2.32 | 7.39 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Oh, C.; Kim, D.; Kim, S. Differential Behavior of Salt and Organic Matter Passage in 2-Pass RO Systems for Ultrapure Water Production. Separations 2026, 13, 93. https://doi.org/10.3390/separations13030093
Oh C, Kim D, Kim S. Differential Behavior of Salt and Organic Matter Passage in 2-Pass RO Systems for Ultrapure Water Production. Separations. 2026; 13(3):93. https://doi.org/10.3390/separations13030093
Chicago/Turabian StyleOh, Changryeol, Dongkeon Kim, and Suhan Kim. 2026. "Differential Behavior of Salt and Organic Matter Passage in 2-Pass RO Systems for Ultrapure Water Production" Separations 13, no. 3: 93. https://doi.org/10.3390/separations13030093
APA StyleOh, C., Kim, D., & Kim, S. (2026). Differential Behavior of Salt and Organic Matter Passage in 2-Pass RO Systems for Ultrapure Water Production. Separations, 13(3), 93. https://doi.org/10.3390/separations13030093






