Effects of Metal Ions on the Flotation of Fluorite and Barite: An Experimental and Mechanistic Investigation
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. Experimental and Simulation Methods
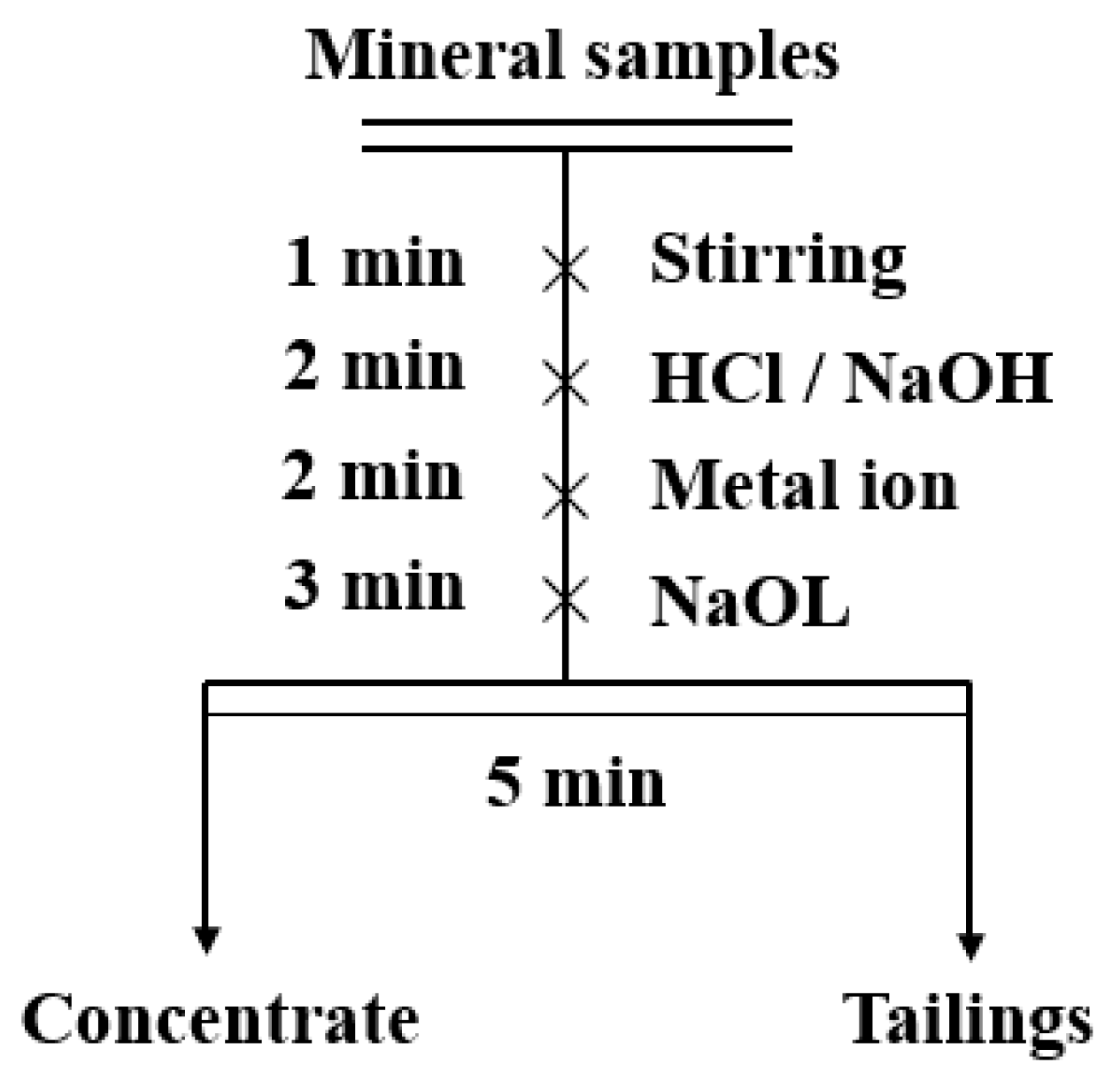
2.2.1. Micro-Flotation Test
2.2.2. Contact Angle Measurement
2.2.3. NaOL Adsorption Measurements
2.2.4. ICP-OES Measurements of Metal-Ion Adsorption
2.2.5. DFT Simulations
3. Results
3.1. Effect of Metal Ions on the Flotation of Fluorite and Barite
3.2. Effect of Metal Ions on the Surface Contact Angle of Fluorite and Barite
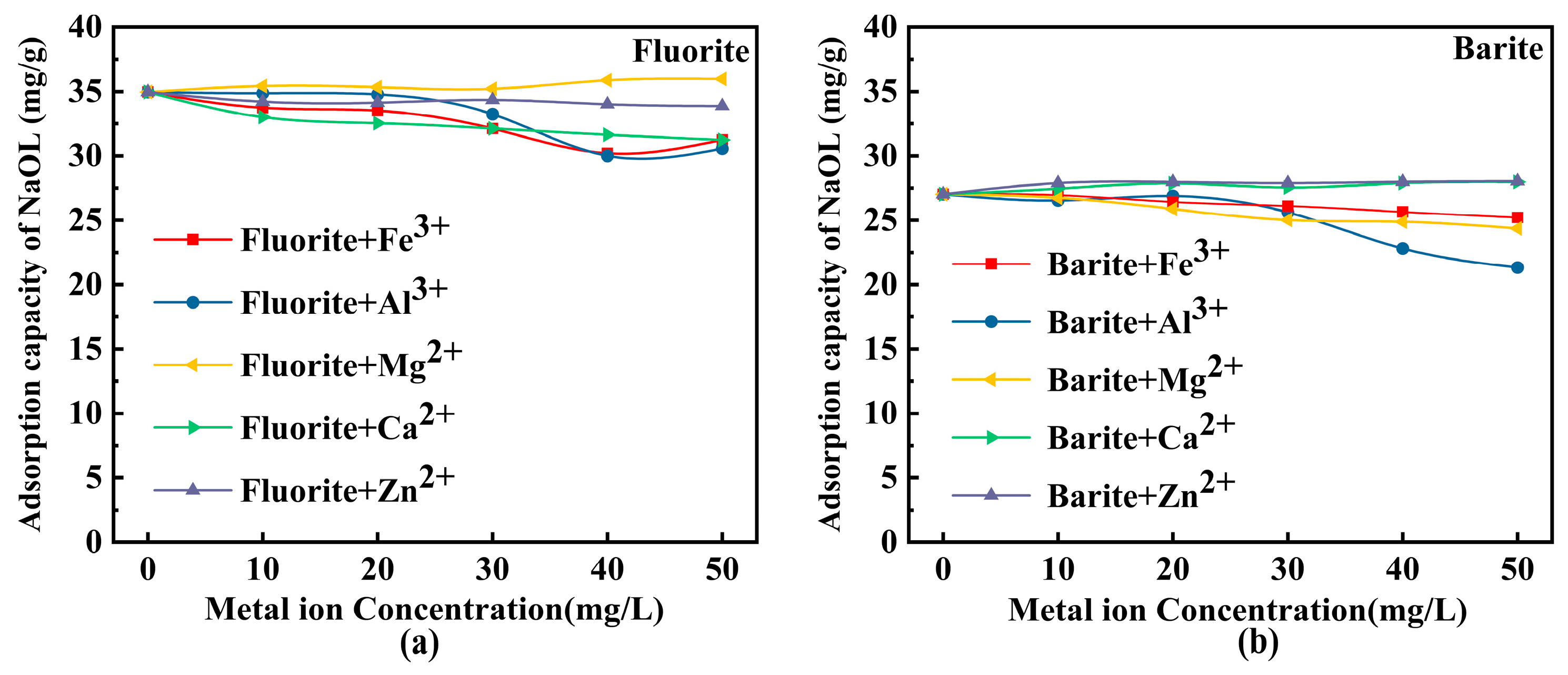
3.3. Effect of Metal Ions on Collector Adsorption Capacity
3.4. ICP Analysis
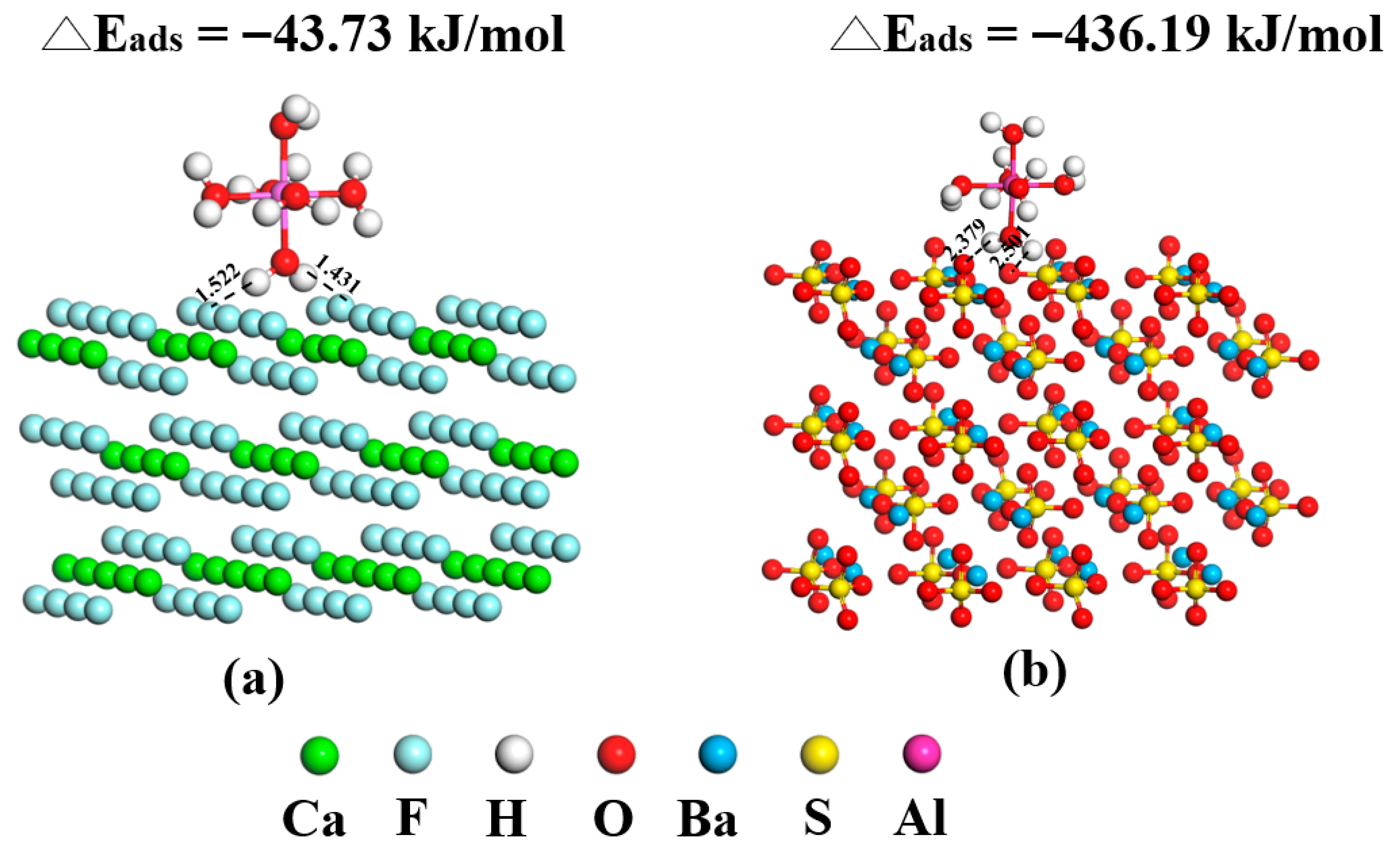
3.5. DFT Calculations
4. Discussion
5. Conclusions
- 1.
- Single-mineral flotation tests showed that, after optimizing the separation conditions, the largest floatability contrast between fluorite and barite was achieved at pH 10 with a NaOL concentration of mol/L (fluorite recovery close to 100% and barite around 80%), providing an optimal operating window for the separation process.
- 2.
- Metal ions exhibited distinct effects on fluorite and barite flotation. For barite, Al3+ strongly depressed floatability via pronounced adsorption (recovery decreased to below 20%), Fe3+ and Mg2+ caused weak depression, whereas Ca2+ and high concentrations of Zn2+ (>20 mg/L) acted as activators. For fluorite, the recovery changes induced by all five ions were below 5%, indicating strong resistance of fluorite to metal-ion interference, likely due to the stability of Ca-related surface sites.
- 3.
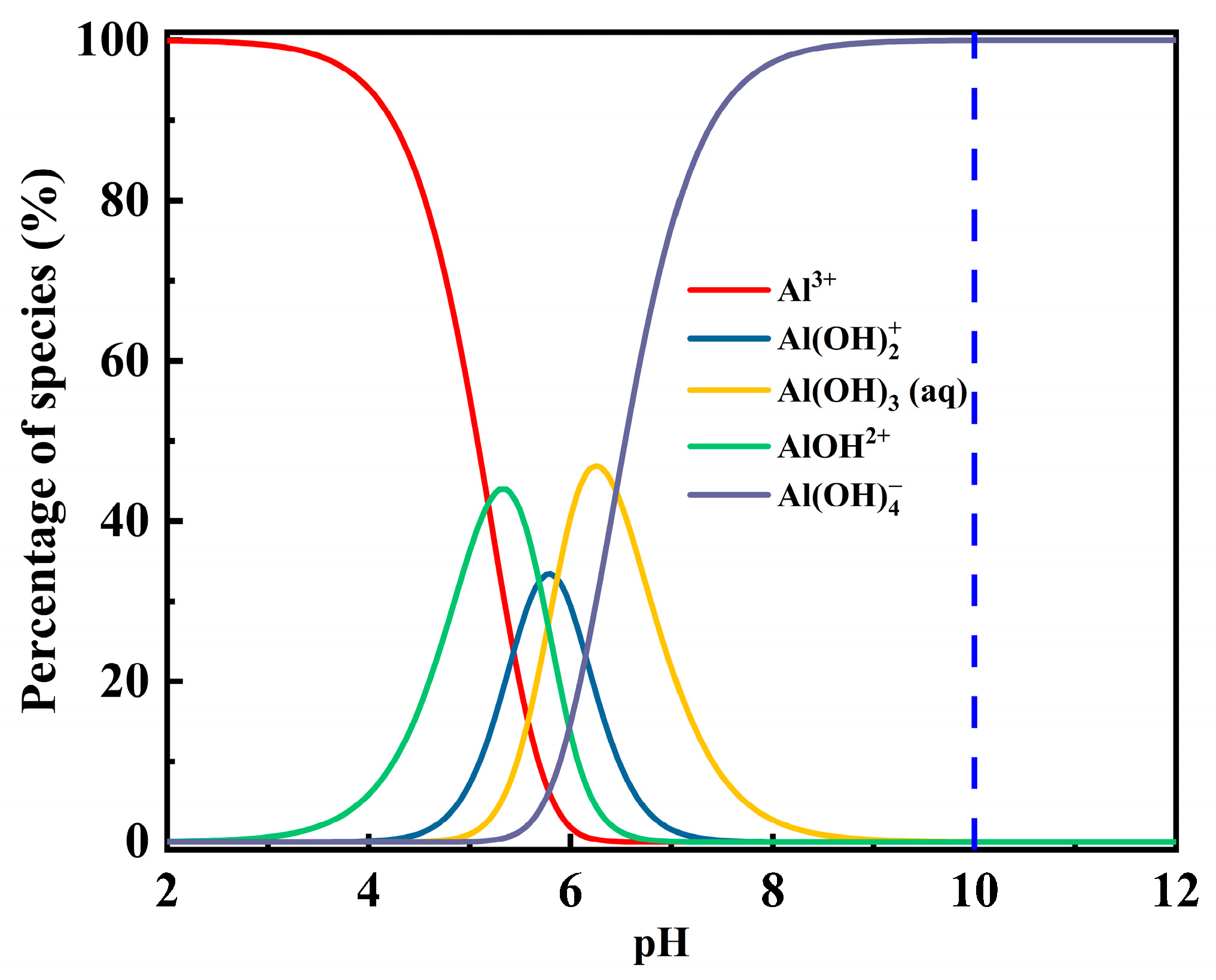
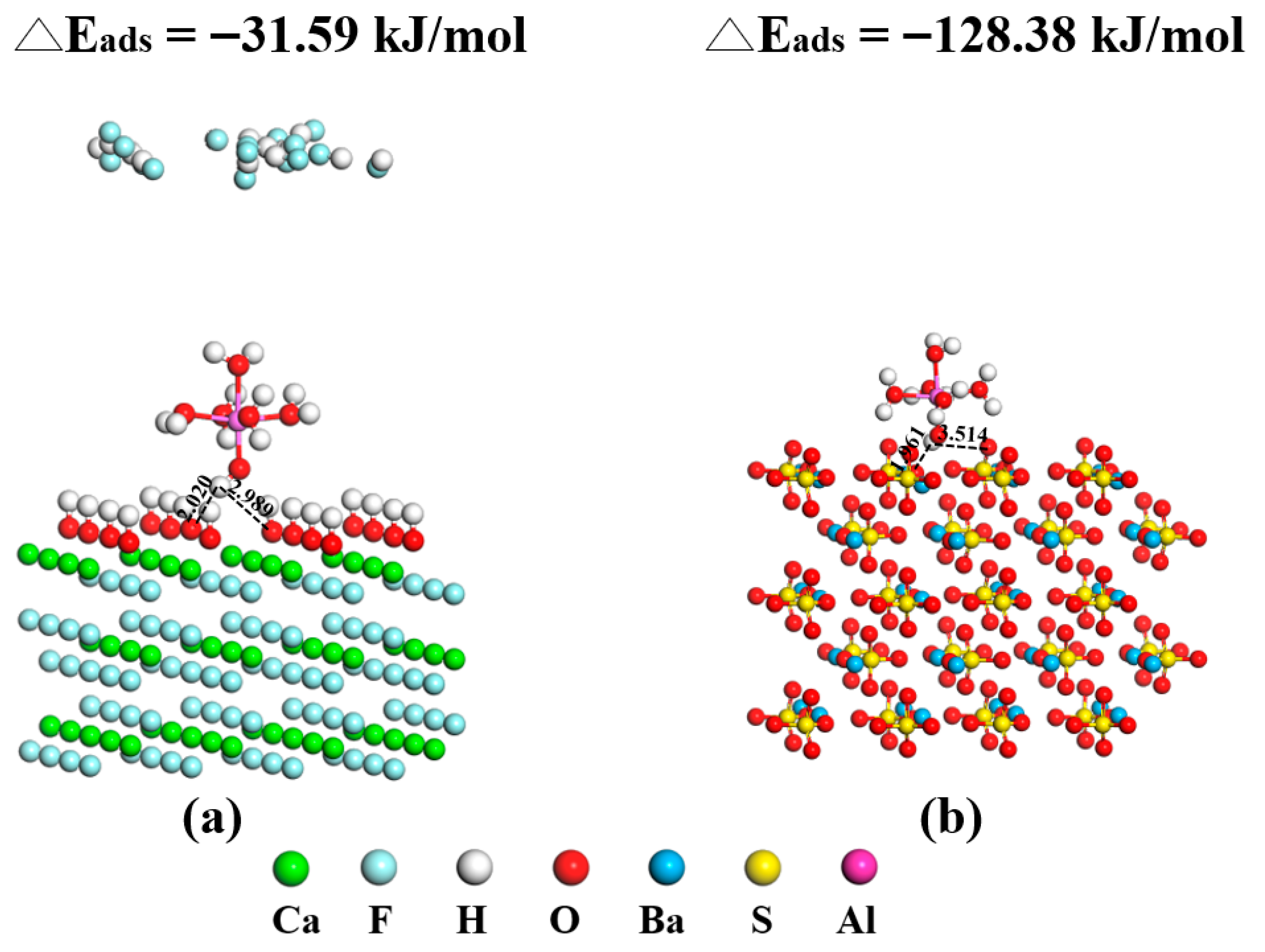
- Contact-angle measurements, ICP-OES analysis, UV–Vis adsorption tests, and DFT calculations collectively indicate that competitive adsorption and surface modification dominate the interfacial mechanism governing fluorite and barite flotation. Al3+ occupies active sites on barite, reducing NaOL adsorption by more than 40% and decreasing the contact angle from 35.6° to 23.1°, thereby significantly weakening surface hydrophobicity. ICP-OES confirmed that Al3+ adsorption on barite is far higher than that on fluorite, explaining its selective depression at the ion-adsorption level. DFT calculations further revealed at the molecular scale that barite surface SO42− groups form strong chemisorption with hydrolyzed Al species (adsorption energy: −436.19 kJ/mol), whereas only weak physisorption occurs on hydroxylated fluorite surfaces due to the presence of a “passivation layer” (adsorption energy: −43.73 kJ/mol). It should be noted that Al(III) speciation at pH 10 is dominated by Al(OH)4− according to Figure 10, whereas the DFT calculations herein used simplified hydrated/hydrolyzed Al(III) species to capture comparative adsorption trends. Future work will include explicit DFT modeling of Al(OH)4− adsorption (and hydration-layer effects) to further strengthen the computational interpretation.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| NaOL | Sodium oleate |
| DFT | Density functional theory |
| ICP | Inductively coupled plasma |
| ICP-OES | Inductively coupled plasma optical emission spectrometry |
| XRD | X-ray diffraction |
| XRF | X-ray fluorescence |
| FP | fundamental parameter |
| CASTEP | Cambridge Sequential Total Energy Package (CASTEP module) |
| GGA | Generalized gradient approximation |
| PW91 | Perdew–Wang 1991 exchange–correlation functional |
| pH | Potential of hydrogen |
| UV | Ultraviolet |
| UV-2700 | UV–Vis spectrophotometer model (Shimadzu UV-2700) |
| XFG | XFG-type self-aerated flotation machine |
| DSA | Drop shape analyzer |
| DSA100E | Drop shape analyzer model (DSA100E) |
| ICP-5000 | ICP-OES instrument model (ICP-5000) |
| IDS | Tetrasodium iminodisuccinate |
References
- Lu, Y.; Liu, W.; Wang, X.; Cheng, H.; Cheng, F.; Miller, J.D. Lauryl Phosphate Flotation Chemistry in Barite Flotation. Minerals 2020, 10, 280. [Google Scholar] [CrossRef]
- Penaloza, I.; Tita, A.; McNew, E.; Chu, P. Barite resources, production and recovery using froth flotation: A review. Miner. Eng. 2023, 203, 108327. [Google Scholar] [CrossRef]
- Jiang, L.; Feng, B.; Wang, T.; Wang, Z. Selective flotation separation of fluorite and barite with carboxylated chitosan as depressant. Miner. Eng. 2024, 219, 109089. [Google Scholar] [CrossRef]
- Gao, Z.; Wang, C.; Sun, W.; Gao, Y.; Kowalczuk, P.B. Froth flotation of fluorite: A review. Adv. Colloid Interface Sci. 2021, 290, 102382. [Google Scholar] [CrossRef]
- Li, X.; Xie, S.; Xu, Y.; Xia, Y.; Cheng, Y.; Tang, S.; Wang, D.; Ren, W.; Zhang, M.; Meng, W. Flotation recovery of barite from high-density waste drilling fluid using β-cyclodextrin as a novel depressant and its mechanism. PLoS ONE 2024, 19, e0298626. [Google Scholar] [CrossRef]
- Zhang, X.F.; Jiao, F.; Qin, W.; Liu, C. Evaluation of 2-phosphatebutane-1, 2, 4-tricarboxylic acid as a depressant in the flotation separation of fluorite from barite. Colloids Surf. A Physicochem. Eng. Asp. 2023, 656, 130453. [Google Scholar] [CrossRef]
- Wang, L.; Lyu, W.; Huang, L.; Li, F.; Zhang, H. Utilization of gellan gum as a novel eco-friendly depressant in the flotation separation of fluorite from barite. Miner. Eng. 2022, 184, 107640. [Google Scholar] [CrossRef]
- Chen, Z.; Ren, Z.; Gao, H.; Zheng, R.; Jin, Y.; Niu, C. Flotation studies of fluorite and barite with sodium petroleum sulfonate and sodium hexametaphosphate. J. Mater. Res. Technol. 2019, 8, 1267–1273. [Google Scholar] [CrossRef]
- Liu, C.; Song, S.; Li, H. Selective flotation of fluorite from barite using trisodium phosphate as a depressant. Miner. Eng. 2019, 134, 390–393. [Google Scholar] [CrossRef]
- Huang, Z.; Cheng, C.; Zhong, H.; Li, L.; Guo, Z.; Yu, X.; He, G.; Han, H.; Deng, L.; Fu, W. Flotation of sylvite from potash ore by using the Gemini surfactant as a novel flotation collector. Miner. Eng. 2019, 132, 22–26. [Google Scholar] [CrossRef]
- Chang, T.; Lv, L.; Liu, J.; Tao, Y.; Ge, W.; Jiang, K. Influence of starch on floatability in fluorite and barite in α-BLA system. J. Mol. Liq. 2024, 402, 124526. [Google Scholar] [CrossRef]
- Liu, C.; Xu, L.; Deng, J.; Han, Z.; Tian, J.; Xue, K.; Wang, D. Enhanced flotation separation of fluorite and calcite using novel collector: Experimental and theoretical Insights. J. Mol. Liq. 2024, 408, 125304. [Google Scholar] [CrossRef]
- Liu, Q.; Han, R.; Jie, L.; Gao, P.; Wang, X.; Tang, Z. Environmentally friendly separation of monazite and fluorite using carboxymethyl cellulose and octyl hydroxamic acid: Experimental and DFT calculations. Appl. Surf. Sci. 2025, 706, 163606. [Google Scholar] [CrossRef]
- Xu, W.; Chen, F.; Zhang, M.; Jiang, F.; Wang, J.; Zhang, W.; Peng, G.; Li, J.; Hu, K.; Yu, H. Flotation separation of fluorite from calcite and quartz using 1-hydroxyoctane-1,1-bisphosphonic acid. J. Mol. Liq. 2025, 424, 127169. [Google Scholar] [CrossRef]
- Somasundaran, P.; Xiao, L.; Wang, D. Solution chemistry of flotation of sparingly soluble minerals. Min. Metall. Explor. 1991, 8, 115–121. [Google Scholar] [CrossRef]
- Wei, B.; Mao, Y.; Wang, L.; Shen, P.; Wang, H.; Liu, D. Sources, Performance and Mechanisms of Metal Ions in the Flotation Process of Copper, Lead, and Zinc Ores: A Review. Minerals 2024, 14, 1105. [Google Scholar] [CrossRef]
- Zheng, Q.; Qiao, L.; Shen, P.; Zhang, T.; Dong, L.; Liu, D. Effect of metal ions on the organic small molecule depressant in the flotation separation of scheelite from calcite. Appl. Surf. Sci. 2026, 717, 164763. [Google Scholar] [CrossRef]
- Tian, J.; Xu, L.; Sun, W.; Zeng, X.; Fang, S.; Han, H.; Hong, K.; Hu, Y. Use of Al2(SO4)3 and acidified water glass as mixture depressants in flotation separation of fluorite from calcite and celestite. Miner. Eng. 2019, 137, 160–170. [Google Scholar] [CrossRef]
- Zhang, H.-L.; Rao, X.; Zhang, C.-Y.; Chen, J.-H.; Zhu, Y.-G.; Wang, R.; Sun, W. Ion coordination affinity and its application in mineral flotation. Trans. Nonferrous Met. Soc. China 2025, 35, 3468–3479. [Google Scholar] [CrossRef]
- Zhu, L.; Zhang, W.; Tian, H.; Dai, H.; Sun, W.; Xie, L.; Tian, M. Study on the mechanism of the inhibition effect of Pb2+ ion activator concealed beneath its activation action in flotation. Surf. Interfaces 2025, 72, 107332. [Google Scholar] [CrossRef]
- Xu, R.; Shen, L.; Huang, J.; Tang, H.; Han, H.; Sun, W.; Wang, L. Enhancement mechanism of metal ions on lepidolite flotation in mixed NaOL/DDA system. Miner. Eng. 2025, 234, 109670. [Google Scholar] [CrossRef]
- Huang, J.; Zhang, Q.; Jiang, T.; Chen, M.; Wang, C.; Wang, S.; Li, H.; Chen, F. Enhancing the flotation separation of fluorite and calcite: Insights into the mechanism of differential surface regulation by various metallic ions. Appl. Surf. Sci. 2025, 696, 162964. [Google Scholar] [CrossRef]
- Zhang, B.; Sun, N.; Sun, W.; Jiang, F.; Wang, L. Enhanced spodumene flotation through Zn(II) ions activation using sodium oleate as collector. Miner. Eng. 2025, 230, 109414. [Google Scholar] [CrossRef]
- Zhu, L.; Zhu, Y.; Xiong, W.; Zhou, Z.; Gao, P.; Han, Y. Aluminium-Modified sodium silicate as a selective depressant in Fluorite-Dolomite Flotation: Experimental and DFT analysis. Miner. Eng. 2025, 228, 109318. [Google Scholar] [CrossRef]
- Douglas, I.; Mohanty, M. Role of dissolved metal ions in rare earth mineral flotation: A review. J. Rare Earths 2025, in press. [Google Scholar] [CrossRef]
- Zhang, X.; Luo, B.; Lu, M.; Feng, J. Effect of Fe3+ on barite flotation and investigation of its adsorption mechanism. Adv. Powder Technol. 2025, 36, 105039. [Google Scholar] [CrossRef]
- Perdew, J.P. Generalized Gradiant Approximation for Exchange and Correlation. A Look Forward and Back-ward. Phys. B 1991, 172, 18828–18832. [Google Scholar] [CrossRef]
- Ren, Z.; Yu, F.; Gao, H.; Chen, Z.; Peng, Y.; Liu, L. Selective Separation of Fluorite, Barite and Calcite with Valonea Extract and Sodium Fluosilicate as Depressants. Minerals 2017, 7, 24. [Google Scholar] [CrossRef]
- De Leeuw, N.H.; Parker, S.C.; Rao, K.H. Modeling the Competitive Adsorption of Water and Methanoic Acid on Calcite and Fluorite Surfaces. Langmuir 1998, 14, 5900–5906. [Google Scholar] [CrossRef]
- Duan, H.; Liu, W.; Zhao, L.; Wang, X. Flotation performance and selective adsorption mechanism of novel hydroxamic acid on the separation of fluorite from barite. Miner. Eng. 2021, 171, 107101. [Google Scholar] [CrossRef]
- Fang, F.; Min, F.; Liu, L.; Chen, J.; Ren, B.; Liu, C. Adsorption of Al(OH)3(3-n)+ (n = 2–4) on Kaolinite (001) Surfaces: A DFT study. Appl. Clay Sci. 2020, 187, 105455. [Google Scholar] [CrossRef]
- Blount, C.W.; Dickson, F.W. The gypsum-anhydrite equilibrium by solubility measurements. Geochim. Cosmochim. Acta 1980, 44, 1931–1936. [Google Scholar] [CrossRef]
- Guan, Z.; Jiao, F.; Zhang, L.; Wei, T. Enhanced flotation separation of barite and fluorite using tetrasodium iminodisuccinate (IDS) as a biodegradable fluorite depressant: Experiments and DFT calculations. Sep. Purif. Technol. 2025, 354, 128847. [Google Scholar] [CrossRef]
- Wang, Z.; Song, Y.; Ren, Z.; Jiang, T. Mechanistic insights into the benzene ring effects on the adsorption activity of sulfonate collector on fluorite: A combined DFT and experimental study. Colloids Surf. A Physicochem. Eng. Asp. 2026, 728, 138481. [Google Scholar] [CrossRef]
- Xiao, F.; Zhang, B.; Lee, C. Effects of low temperature on aluminum(III) hydrolysis: Theoretical and experimental studies. J. Environ. Sci. 2008, 20, 907–914. [Google Scholar] [CrossRef]
- Bi, S.; Wang, C.; Cao, Q.; Zhang, C. Studies on the Mechanism of Hydrolysis and Polymerization of Aluminum Salts in Aqueous Solution: Correlations between the ‘Core-Links’ Model and ‘Cage-Like’ Keggin-Al13 Model. Coord. Chem. Rev. 2004, 248, 441–455. [Google Scholar] [CrossRef]








| Samples | CaF2 | BaSO4 | SiO2 | Al2O3 | Fe2O3 | SrSO4 | Purity |
|---|---|---|---|---|---|---|---|
| Fluorite | 99.90 | - | 0.04 | 0.02 | 0.01 | - | 99.90 |
| Barite | - | 99.10 | 0.18 | 0.03 | 0.05 | 0.45 | 99.10 |
| Samples | Contact Angle 1/° | Difference 2/° |
|---|---|---|
| Barite + NaOL | 35.6 ± 0.8 | - |
| Barite + Fe3+ + NaOL | 32.8 ± 1.9 | −2.8 |
| Barite + Al3+ + NaOL | 23.1 ± 1.5 | −12.5 |
| Barite + Mg2+ + NaOL | 30.7 ± 1.7 | −4.9 |
| Barite + Ca2+ + NaOL | 32.9 ± 1.3 | −2.7 |
| Barite + Zn2+ + NaOL | 34.2 ± 1.6 | −1.4 |
| Fluorite + NaOL | 73.5 ± 1.7 | - |
| Fluorite + Fe3+ + NaOL | 56.9 ± 2.1 | −16.6 |
| Fluorite + Al3+ + NaOL | 68.5 ± 2.3 | −5.0 |
| Fluorite + Mg2+ + NaOL | 69.1 ± 1.9 | −4.4 |
| Fluorite + Ca2+ + NaOL | 60.3 ± 1.8 | −13.2 |
| Fluorite + Zn2+ + NaOL | 66.6 ± 1.9 | −6.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Wei, Y.; Li, Y.; Liu, Y.; Guo, Y.; Li, C.; Yang, W. Effects of Metal Ions on the Flotation of Fluorite and Barite: An Experimental and Mechanistic Investigation. Separations 2026, 13, 85. https://doi.org/10.3390/separations13030085
Wei Y, Li Y, Liu Y, Guo Y, Li C, Yang W. Effects of Metal Ions on the Flotation of Fluorite and Barite: An Experimental and Mechanistic Investigation. Separations. 2026; 13(3):85. https://doi.org/10.3390/separations13030085
Chicago/Turabian StyleWei, Ying, Yuqiong Li, Yingchao Liu, Yuxin Guo, Caiyun Li, and Wanglin Yang. 2026. "Effects of Metal Ions on the Flotation of Fluorite and Barite: An Experimental and Mechanistic Investigation" Separations 13, no. 3: 85. https://doi.org/10.3390/separations13030085
APA StyleWei, Y., Li, Y., Liu, Y., Guo, Y., Li, C., & Yang, W. (2026). Effects of Metal Ions on the Flotation of Fluorite and Barite: An Experimental and Mechanistic Investigation. Separations, 13(3), 85. https://doi.org/10.3390/separations13030085





