A Proof-of-Concept Study for the Strong Electrolyte (SE) Switching and the Combined CO2-SE Switching of the Polarity of Tertiary Amine for Lipid Separation Application
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents and Instruments
2.2. Samples
2.3. Procedure for Switching and Back-Switching
2.4. Characterization
2.5. Lipids Extraction from Algae
3. Results and Discussion
3.1. Background of Solvent Switching Techniques
3.2. Oil and Amine Recovery with CO2, SE and CO2-SE Switching
4. Further Discussion
4.1. Pros and Cons of Examined Switching Methods
4.2. Preliminary Results on Lipids Extraction from Microalgae
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Mohsenpour, S.F.; Hennige, S.; Willoughby, N.; Adeloye, A.; Gutierrez, T. Integrating micro-algae into wastewater treatment: A review. Sci. Total Environ. 2021, 752, 142168. [Google Scholar] [CrossRef] [PubMed]
- Lage, S.; Gentili, F.G. Chemical composition and species identification of microalgal biomass grown at pilot-scale with municipal wastewater and CO2 from flue gases. Chemosphere 2023, 313, 137344. [Google Scholar] [CrossRef] [PubMed]
- Pandey, S.; Narayanan, I.; Selvaraj, R.; Varadavenkatesan, T.; Vinayagam, R. Biodiesel production from microalgae: A comprehensive review on influential factors, transesterification processes, and challenges. Fuel 2024, 367, 131547. [Google Scholar] [CrossRef]
- Zhou, J.; Wang, M.; Saraiva, J.A.; Martins, A.P.; Pinto, C.A.; Prieto, M.A.; Simal-Gandara, J.; Cao, H.; Xiao, J.; Barba, F.J. Extraction of lipids from microalgae using classical and innovative approaches. Food Chem. 2022, 384, 132236. [Google Scholar] [CrossRef]
- Wetterwald, L.; Leybros, A.; Fleury, G.; Delrue, F.; Dimitriades-Lemaire, A.; Chambonniere, P.; Hertz, A. Supercritical CO2 extraction of neutral lipids from dry and wet Chlorella vulgaris NIES 227 microalgae for biodiesel production. J. Environ. Chem. Eng. 2023, 11, 110628. [Google Scholar] [CrossRef]
- Spain, O.; Plöhn, M.; Funk, C. The cell wall of green microalgae and its role in heavy metal removal. Physiol. Plant. 2021, 173, 526–535. [Google Scholar] [CrossRef]
- Yong, T.C.; Chiu, P.-H.; Chen, C.-H.; Hung, C.-H.; Chen, C.-N.N. Disruption of thin- and thick-wall microalgae using high pressure gases: Effects of gas species, pressure and treatment duration on the extraction of proteins and carotenoids. J. Biosci. Bioeng. 2020, 129, 502–507. [Google Scholar] [CrossRef]
- Araujo, G.S.; Matos, L.J.B.L.; Fernandes, J.O.; Cartaxo, S.J.M.; Gonçalves, L.R.B.; Fernandes, F.A.N.; Farias, W.R.L. Extraction of lipids from microalgae by ultrasound application: Prospection of the optimal extraction method. Ultrason. Sonochemistry 2013, 20, 95–98. [Google Scholar] [CrossRef]
- Motlagh, S.R.; Khezri, R.; Etesami, M.; Chee, C.Y.; Kheawhom, S.; Nootong, K.; Harun, R. Microwave-assisted extraction of lipid and eicosapentaenoic acid from the microalga Nanochloropsis sp. using imidazolium-based ionic liquids as an additive in water. J. Appl. Phycol. 2024, 36, 1709–1724. [Google Scholar] [CrossRef]
- Jessop, P.G.; Heldebrant, D.J.; Li, X.; Eckert, C.A.; Liotta, C.L. Reversible nonpolar-to-polar solvent. Nature 2005, 436, 1102. [Google Scholar] [CrossRef] [PubMed]
- Al-Ameri, M.; Al-Zuhair, S. Using switchable solvents for enhanced, simultaneous microalgae oil extraction-reaction for biodiesel production. Biochem. Eng. J. 2019, 141, 217–224. [Google Scholar] [CrossRef]
- Du, Y.; Schuur, B.; Samorì, C.; Tagliavini, E.; Brilman, D.W.F. Secondary amines as switchable solvents for lipid extraction from non-broken microalgae. Bioresour. Technol. 2013, 149, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Anto, S.; Premalatha, M.; Mathimani, T. Tertiary amine as an efficient CO2 switchable solvent for extracting lipids from hypersaline microalgae. Chemosphere 2022, 288, 132442. [Google Scholar] [CrossRef]
- Russell, C.; Rodriguez, C. Lipid extraction from Chlorella vulgaris & Haematococcus pluvialis using the switchable solvent DMCHA for biofuel production. Energy 2023, 278, 127983. [Google Scholar] [CrossRef]
- Samorì, C.; López Barreiro, D.; Vet, R.; Pezzolesi, L.; Brilman, D.W.F.; Galletti, P.; Tagliavini, E. Effective lipid extraction from algae cultures using switchable solvents. Green Chem. 2013, 15, 353–356. [Google Scholar] [CrossRef]
- Tang, W.; Ho Row, K. Evaluation of CO2-induced azole-based switchable ionic liquid with hydrophobic/hydrophilic reversible transition as single solvent system for coupling lipid extraction and separation from wet microalgae. Bioresour. Technol. 2020, 296, 122309. [Google Scholar] [CrossRef]
- Lestari, G.; Salari, A.; Abolhasani, M.; Kumacheva, E. A microfluidic study of liquid–liquid extraction mediated by carbon dioxide. Lab A Chip 2016, 16, 2710–2718. [Google Scholar] [CrossRef]
- Han, S.; Ramezani, M.; TomHon, P.; Abdel-Latif, K.; Epps, R.W.; Theis, T.; Abolhasani, M. Intensified continuous extraction of switchable hydrophilicity solvents triggered by carbon dioxide. Green Chem. 2021, 23, 2900–2906. [Google Scholar] [CrossRef]
- Patel, S.D.; Hill, S.J.; Weber, C.C. Enhancing the Efficiency of Switching Switchable Hydrophilicity Solvents Using Continuous Flow Approaches. ChemSusChem 2025, 18, e202501531. [Google Scholar] [CrossRef]
- Perdomo, F.A.; Khalit, S.H.; Graham, E.J.; Tzirakis, F.; Papadopoulos, A.I.; Tsivintzelis, I.; Seferlis, P.; Adjiman, C.S.; Jackson, G.; Galindo, A. A predictive group-contribution framework for the thermodynamic modelling of CO2 absorption in cyclic amines, alkyl polyamines, alkanolamines and phase-change amines: New data and SAFT-γ Mie parameters. Fluid Phase Equilibria 2023, 566, 113635. [Google Scholar] [CrossRef]
- Tzirakis, F.; Papadopoulos, A.I.; Seferlis, P.; Tsivintzelis, I. CO solubility in aqueous N-methylcyclohexylamine (MCA) and N-cyclohexyl-1,3-propanediamine (CHAP) solutions. AIChE J. 2023, 69, e17982. [Google Scholar] [CrossRef]
- Shahvandi, S.K.; Banitaba, M.H.; Ahmar, H. Development of a new pH assisted homogeneous liquid-liquid microextraction by a solvent with switchable hydrophilicity: Application for GC-MS determination of methamphetamine. Talanta 2018, 184, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Billet, R.; Zeng, B.; Wu, H.; Lockhart, J.; Gattrell, M.; Zhao, H.; Zhang, X. Hydrophilic solvent recovery from switched-on microdroplet dissolution. Droplet 2023, 2, e82. [Google Scholar] [CrossRef]
- Jing, J.; Li, X.; Zhang, Y.; Liu, Y.; Lu, H.; Wang, J.; Wu, Y. pH-Responsive Regulation of a Surfactant-Free Microemulsion Based on Hydrophobic Deep Eutectic Solvents. Langmuir 2022, 38, 7898–7905. [Google Scholar] [CrossRef]
- Guan, X.; Huang, Z.; Lu, H.; Sun, D. Microheterogeneity and CO2 Switchability of N,N-Dimethylcyclohexylamine–Water Binary Mixtures. J. Phys. Chem. B 2019, 123, 3096–3102. [Google Scholar] [CrossRef] [PubMed]
- European Union. Commission Regulation (EEC) No 2568/91 of 11 July 1991 on the Characteristics of Olive Oil and Olive-Residue Oil and on the Relevant Methods of Analysis. Available online: https://eur-lex.europa.eu/eli/reg/1991/2568/oj/eng (accessed on 26 November 2025).
- Palikrousis, T.L.; Manolis, C.; Kalamaras, S.D.; Samaras, P. Effect of Light Intensity on the Growth and Nutrient Uptake of the Microalga Chlorella sorokiniana Cultivated in Biogas Plant Digestate. Water 2024, 16, 2782. [Google Scholar] [CrossRef]
- Lo, C.; Wijffels, R.H.; Eppink, M.H.M. Lipid recovery from deep eutectic solvents by polar antisolvents. Food Bioprod. Process. 2024, 143, 21–27. [Google Scholar] [CrossRef]
- Kazarian, S.G.; Vincent, M.F.; Bright, F.V.; Liotta, C.L.; Eckert, C.A. Specific Intermolecular Interaction of Carbon Dioxide with Polymers. J. Am. Chem. Soc. 1996, 118, 1729–1736. [Google Scholar] [CrossRef]
- Cundari, T.R.; Wilson, A.K.; Drummond, M.L.; Gonzalez, H.E.; Jorgensen, K.R.; Payne, S.; Braunfeld, J.; De Jesus, M.; Johnson, V.M. CO2-Formatics: How Do Proteins Bind Carbon Dioxide? J. Chem. Inf. Model. 2009, 49, 2111–2115. [Google Scholar] [CrossRef]
- Romanić, R. Chapter 17—Cold pressed sunflower (Helianthus annuus L.) oil. In Cold Pressed Oils; Ramadan, M.F., Ed.; Academic Press: Cambridge, MA, USA, 2020; pp. 197–218. [Google Scholar]
- Zambiazi, R.C.; Przybylski, R.; Zambiazi, M.W.; Mendonça, C.B. Fatty Acid Composition of Vegetable Oils and Fats. Bol. Cent. Pesqui. Process. Aliment. 2007, 25, 111–120. [Google Scholar] [CrossRef]
- Available online: https://srdata.nist.gov/solubility/sol_detail.aspx?sysID=62_1 (accessed on 24 January 2025).
- Yoo, Y.; Kim, I.; Lee, D.; Choi, W.; Choi, J.; Jang, K.; Park, J.; Kang, D. Review of Contemporary Research on Inorganic CO2 Utilization via CO2 Conversion into Metal Carbonate-Based Materials. J. Ind. Eng. Chem. 2022, 116, 60–74. [Google Scholar] [CrossRef]
- Tsagdi, A.; Drossos, I.; Georgiou, D.; Exarhopoulos, S.; Karasiotas, G.; Kallitsis, J.K.; Kalogianni, E.P. Injection Molded PP Foams Using Food Ingredients for Food Packaging Applications. Polymers 2021, 13, 288. [Google Scholar] [CrossRef] [PubMed]
- Tsioptsias, C.; Mitis, S.; Rentzela, A.; Alvanou, K.; Kelesi, D.; Manolis, C.; Stergiou, A.; Kalamaras, S.D.; Samaras, P. A Simplified Methodology for Solvent Screening in Selective Extraction of Lipids from Microalgae Based on Hansen Solubility Parameters. Molecules 2025, 30, 4428. [Google Scholar] [CrossRef] [PubMed]



| Experiment | Conditions | Target |
|---|---|---|
| 1 (CO2 switching) | Forward switching with CO2 bubbling at room temperature for 80 min | Recovery of oil |
| 2 (CO2 switching) | Forward switching with CO2 bubbling at room temperature for 30 min and back-switching by heating at 80–105 °C for 2.5 h | Recovery of amine |
| 3 (SE switching) | Forward switching with HCl solution at HCl:amine ratio 1:1 | Recovery of oil |
| 4 (SE switching) | Forward switching with HCl solution at HCl:amine ratio 1:1 and back-switching with 10 M NaOH solution at NaOH:amine ratio 1:1 | Recovery of amine |
| 5 (combined CO2-SEswitching) | Forward switching with CO2 bubbling at room temperature for 30 min and back-switching with 10 M NaOH solution at NaOH:amine ratio 1:1 | Recovery of amine |
| CO2 Switching | SE (HCl) Switching | Combined CO2-SE Switching | |
|---|---|---|---|
| Required time, min | 80 | ~1 | 80 |
| Purity of recovered oil, %v/v | 80 | 95 | 80 |
| % recovery of oil | 93.3 | 95 | 93.3 |
| Oxidation Index | Untreated Sunflower Oil | Sunflower Oil Treated with HCl |
|---|---|---|
| K232 | 2.27 ± 0.01 | 2.29 ± 0.01 |
| K268 | 1.93 ± 0.02 | 2.29 ± 0.03 |
| CO2: Switching with CO2 and Back Switching with Heat | SE: Switching with HCl and Back-Switching with NaOH | Combined CO2-SE: Switching with CO2 and Back-Switching with NaOH | |
|---|---|---|---|
| Required time only for the back-switching, min | 150 | ~1 min | ~1 min |
| Temperature of back-switching, °C | 80–105 | Room temperature (~18) | Room temperature (~18) |
| Purity of recovered amine, %v/v | 96.5 | 90.5 | 94.0 |
| % recovery of amine | 42.9 | 85.5 | 68 |
| Approach for Polarity Switching | Issues | |||
|---|---|---|---|---|
| Technical | Economic | Environmental | Safety | |
| CO2 | High oil purity and oil recovery. High amine purity. Low amine recovery (44%). High processing time (4 h). | No useful byproducts. Cost for energy consumption for back-switching. Cost for additional amine due to low % recovery of the amine. | No CO2 is captured. Amine toxic residues in water and air (unless additional capital and operational cost is invested) | Risk of fire since back-switching is performed at temperatures well above the flash point of amine. |
| SE | High oil purity and oil recovery. High amine purity. High amine recovery (85%). Very short processing time (2 min). | Cost for HCl and NaOH. Decrease in cost of demand of amine due to high % recovery of the amine. | No CO2 is captured. Production of highly saline effluent. Minimization of amine residues in water and air. | Corrosion and safety issues due to concentrated HCl and NaOH. Production of HCl vapors. Minimization of risk of fire accident since the process is carried out in air atmosphere (typically below the flash point of the amine) |
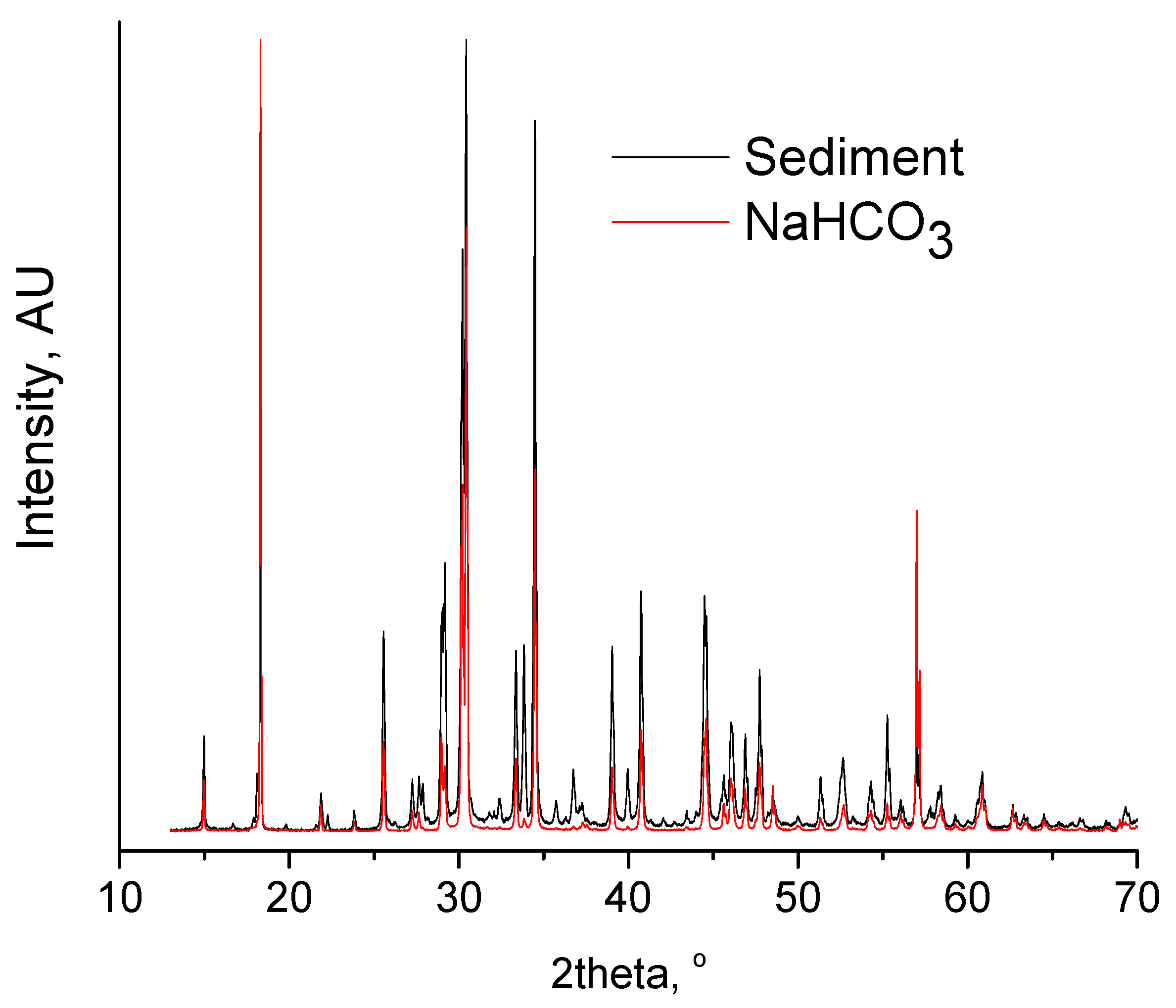
| CO2-SE | High oil purity and oil recovery. High amine purity. High amine recovery (68%). Short processing time (80 min). Difficulties related to the handling the NaHCO3 slurry. | Cost for NaOH. This cost can be balanced by the useful byproduct (NaHCO3). Decrease in cost of demand of amine due to high % recovery of amine. | Reduced carbon footprint process since CO2 is captured and converted to NaHCO3 (in the case that flue gas is used). Minimization of amine residues in water and air. | Corrosion and safety issues due to concentrated NaOH. Minimization of risk of fire accident since the process is carried out at temperatures typically below the flash point of amine. No strong acid is used. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Tsioptsias, C.; Maletskos, I.; Tachias, G.; Palikrousis, T.; Ntampou, X.; Kalogianni, E.P.; Samaras, P. A Proof-of-Concept Study for the Strong Electrolyte (SE) Switching and the Combined CO2-SE Switching of the Polarity of Tertiary Amine for Lipid Separation Application. Separations 2026, 13, 81. https://doi.org/10.3390/separations13030081
Tsioptsias C, Maletskos I, Tachias G, Palikrousis T, Ntampou X, Kalogianni EP, Samaras P. A Proof-of-Concept Study for the Strong Electrolyte (SE) Switching and the Combined CO2-SE Switching of the Polarity of Tertiary Amine for Lipid Separation Application. Separations. 2026; 13(3):81. https://doi.org/10.3390/separations13030081
Chicago/Turabian StyleTsioptsias, Costas, Ioannis Maletskos, George Tachias, Thomas Palikrousis, Xanthi Ntampou, Eleni P. Kalogianni, and Petros Samaras. 2026. "A Proof-of-Concept Study for the Strong Electrolyte (SE) Switching and the Combined CO2-SE Switching of the Polarity of Tertiary Amine for Lipid Separation Application" Separations 13, no. 3: 81. https://doi.org/10.3390/separations13030081
APA StyleTsioptsias, C., Maletskos, I., Tachias, G., Palikrousis, T., Ntampou, X., Kalogianni, E. P., & Samaras, P. (2026). A Proof-of-Concept Study for the Strong Electrolyte (SE) Switching and the Combined CO2-SE Switching of the Polarity of Tertiary Amine for Lipid Separation Application. Separations, 13(3), 81. https://doi.org/10.3390/separations13030081










