Response Surface Methodology-Optimized QuEChERS Combined with Liquid Chromatography–Quadrupole-Time-of-Flight Mass Spectrometry for Simultaneous Screening of Pesticides and Mycotoxins in Astragalus
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Apparatus
2.3. Preparation of Standard Solutions
2.4. Real Samples
2.5. Sample Preparation
2.6. Instrumentation
2.7. Design and Optimization of Pretreatment
2.8. Methodological Validation
3. Results and Discussion
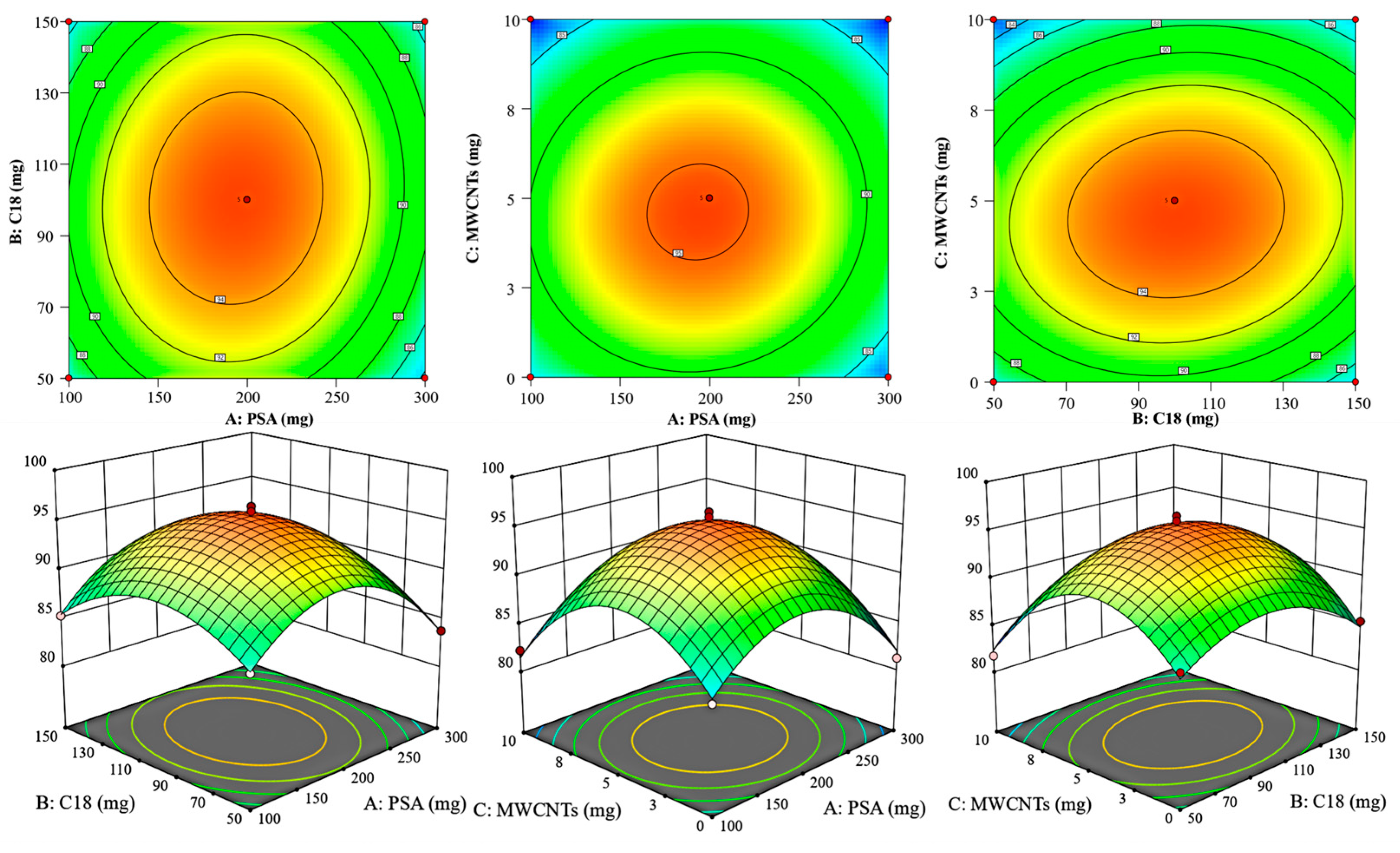
3.1. Optimization of Pretreatment Conditions
3.1.1. Optimization of Extraction Solvent
3.1.2. Single-Factor Test of Purification Conditions
3.1.3. Response Surface Optimization of Purification Conditions
3.2. Validation of the Approach
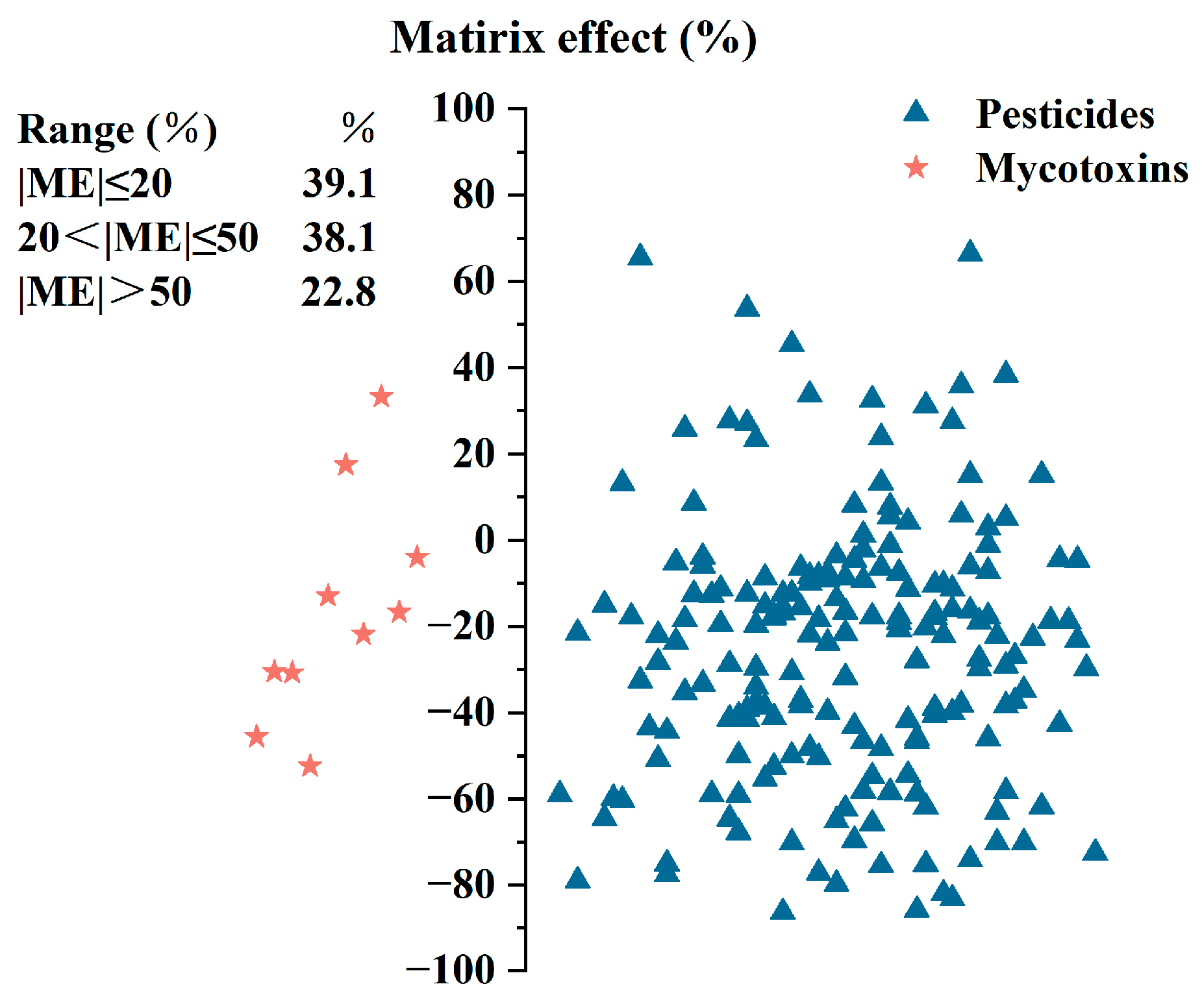
3.2.1. Matrix Effect
3.2.2. Screening Detection Limits (SDLs), Limits of Quantification (LOQs), and Linearity
3.2.3. Recovery and Precision
3.2.4. Comparison with Other Methods
3.3. Real Sample Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Guo, Z.; Lou, Y.; Kong, M.; Luo, Q.; Liu, Z.; Wu, J. A Systematic Review of Phytochemistry, Pharmacology and Pharmacokinetics on Astragali Radix: Implications for Astragali Radix as a Personalized Medicine. Int. J. Mol. Sci. 2019, 20, 1463. [Google Scholar] [CrossRef]
- Shi, Y.; Shi, X.; Zhao, M.; Ma, S.; Zhang, Y. Pharmacological Potential of Astragali Radix for the Treatment of Kidney Diseases. Phytomedicine 2024, 123, 155196. [Google Scholar] [CrossRef] [PubMed]
- Qin, H.; Xie, L.; Zang, Y.; Han, J.; Yu, J.; Luo, Z.; Ma, X. Residue of Chlormequat and Regulatory Effects on the Specialized Metabolites of Astragali Radix. Molecules 2023, 28, 6754. [Google Scholar] [CrossRef]
- Butkovskyi, A.; Jing, Y.; Bergheim, H.; Lazar, D.; Gulyaeva, K.; Odenmarck, S.; Norli, H.; Nowak, K.; Miltner, A.; Kästner, M.; et al. Retention and Distribution of Pesticides in Planted Filter Microcosms Designed for Treatment of Agricultural Surface Runoff. Sci. Total Environ. 2021, 778, 146114. [Google Scholar] [CrossRef]
- Yang, Y.; Zheng, K.; Guo, L.P.; Wang, C.X.; Zhong, D.B.; Shang, L.; Hong, J.N.; Cui, X.M.; Huang, S.J. Rapid Determination and Dietary Intake Risk Assessment of 249 Pesticide Residues in Panax Notoginseng. Ecotoxicol. Environ. Saf. 2022, 233, 113348. [Google Scholar] [CrossRef]
- Chinese Pharmacopoeia Commission. Pharmacopoeia of the People’s Republic of China; China Press of Traditional Chinese Medicine: Beijing, China, 2025. [Google Scholar]
- GB 2763-2021; National Food Safety Standard-Maximum Residue Limits for Pesticides in Food. Ministry of Agriculture of the People’s Republic of China: Beijing, China, 2021.
- Wang, Z.; Weng, Y.; Xiao, J.; Zhang, P.; Wang, X.; Chen, L. Determination Research of Prohibited Pesticide Residues in Astragali Radix. Asia-Pac. Tradit. Med. 2024, 20, 61–68. [Google Scholar]
- Wang, Z.; Ren, X.; Zhang, A.; Sun, M.; Ding, Z.; Fan, J. A Fungal Hyphae-Derived Biomass Carbon for Magnetic Solid-Phase Extraction of the Organochlorine Pesticides in Water Samples, Tea Beverages, and Chinese Traditional Medicines before Gas Chromatography-Tandem Mass Spectrometry Determination. Food Chem. 2024, 457, 140123. [Google Scholar] [CrossRef]
- Wang, Q.; Zhu, Y.; Li, T.; Fan, L.; Li, Z.; Wang, X. Determination of 69 Kinds of Pesticide Residues in Paeonia tactilora, Astragalus membranaceus, Ranunculus ternatus and Cornus officinalis by GC-MS/MS. China Pharm. 2019, 30, 2829–2834. [Google Scholar]
- Yang, W.; Wang, S.; Li, Y.; Liu, Y.; Liu, S.; Huang, Y.; Ran, F.; Ren, H. A Comprehensive Review of Detection and Removal Strategy for Organic Pesticide Residues in Chinese Herbal Medicines. Talanta 2026, 299, 129098. [Google Scholar] [CrossRef]
- Wang, G.; Jiao, M.; Hu, J.; Yi, R.X.; Chen, L.; Qiu, J.; Ji, F.; Lee, Y.W.; Shi, J.; Xu, J. Quantitative Analysis of Fungal Contamination of Different Herbal Medicines in China. Toxins 2024, 16, 229. [Google Scholar] [CrossRef]
- Hu, L.; Li, M.; Sun, Y.; Zhao, H.; Chen, J.; Dai, X.; Kong, Z. Pesticide and Mycotoxin Residues in Astragalus: Transfer Patterns, Processing Factors and Risk Assessment during Astragalus Processing. Food Chem. 2025, 481, 144167. [Google Scholar] [CrossRef]
- Zhu, Y.; Wang, S.; Xu, X.; Wang, L.; Zhou, H.; Ying, X.; Hu, Q.; Wang, X.; Ji, S.; Cai, Q. Exposure Assessment and Risk-Based Limit Levels Evaluation of Ochratoxin a in Astragali Radix in China. Ecotoxicol. Environ. Saf. 2022, 237, 113517. [Google Scholar] [CrossRef]
- Hua, Z.; Liu, R.; Chen, Y.; Liu, G.; Li, C.; Song, Y.; Cao, Z.; Li, W.; Li, W.; Lu, C. Contamination of Aflatoxins Induces Severe Hepatotoxicity through Multiple Mechanisms. Front. Pharmacol. 2021, 11, 605823. [Google Scholar] [CrossRef]
- Perestrelo, R.; Silva, P.; Porto-Figueira, P.; Pereira, J.A.M.; Silva, C.; Medina, S.; Câmara, J.S. QuEChERS—Fundamentals, Relevant Improvements, Applications and Future Trends. Anal. Chim. Acta 2019, 1070, 1–28. [Google Scholar] [CrossRef]
- Cheng, R.; Xie, Y.; Chang, Q.; Bai, Y.; Tong, K.; Wu, X.; Chen, H. Simultaneous Determination of 202 Pesticide Residues and 19 Mycotoxins in Coix Seed by QuEChERS Coupled with LC-Q-TOF/MS and Subsequent Assessment of Dietary Exposure Risk. Food Control 2025, 175, 111301. [Google Scholar] [CrossRef]
- Selim, M.T.; Almutari, M.M.; Shehab, H.I.; EL-Saeid, M.H. Risk Assessment of Pesticide Residues by GC-MSMS and UPLC-MSMS in Edible Vegetables. Molecules 2023, 28, 1343. [Google Scholar] [CrossRef]
- Jedziniak, P.; Panasiuk, Ł.; Pietruszka, K.; Posyniak, A. Multiple Mycotoxins Analysis in Animal Feed with LC-MS/MS: Comparison of Extract Dilution and Immunoaffinity Clean-Up. J. Sep. Sci. 2019, 42, 1240–1247. [Google Scholar] [CrossRef]
- Eddahby, A.; Chahine, A.; Hssaini, L. Assessing Matrix Effects in LC-MS/MS Analysis of Pesticides in Food Using Diluted QuEChERS Extracts. Food Anal. Methods 2025, 18, 1203–1212. [Google Scholar] [CrossRef]
- Tölgyesi, Á.; Cseh, A.; Simon, A.; Sharma, V.K. Development of a Novel LC-MS/MS Multi-Method for the Determination of Regulated and Emerging Food Contaminants Including Tenuazonic Acid, a Chromatographically Challenging Alternaria Toxin. Molecules 2023, 28, 1468. [Google Scholar] [CrossRef]
- Chen, Y.; Cai, W.; Lin, Z.; Ma, Y.; Wu, Y.; Li, D.; Zhang, Z.; Wang, Y.; Chen, Y.; Wang, C. A Modified QuEChERS Method Using Single-Sorbent Combined with LC-MS/MS for Simultaneous Determination of Four Phenolic Pesticide Residues in Fruits and Vegetables. J. Food Compos. Anal. 2026, 149, 108820. [Google Scholar] [CrossRef]
- Lin, R.; Feng, H.; Zhang, Z.; Liu, Y.; Wen, H.; Chen, H.; Fan, Y.; Tan, Y.; Tang, Z. Novel Magnetic Nanoparticle-Assisted QuEChERS-UHPLC-MS/MS for the Determination of Multi-Mycotoxins in Complex Sample Matrices. Microchem. J. 2026, 221, 116963. [Google Scholar] [CrossRef]
- Huang, Q.; Guo, W.; Zhao, X.; Cao, H.; Fan, K.; Meng, J.; Nie, D.; Wu, Y.; Han, Z. Universal Screening of 200 Mycotoxins and Their Variations in Stored Cereals in Shanghai, China by UHPLC-Q-TOF MS. Food Chem. 2022, 387, 132869. [Google Scholar] [CrossRef]
- Pang, G.; Fan, C.; Chang, Q.; Li, J.; Kang, J.; Lv, M. Screening of 485 Pesticide Residues in Fruits and Vegetables by Liquid Chromatography-Quadrupole-Time-of-Flight Mass Spectrometry Based on TOF Accurate Mass Database and QTOF Spectrum Library. J. AOAC Int. 2018, 101, 1156–1182. [Google Scholar] [CrossRef] [PubMed]
- Pang, G.; Chang, Q.; Bai, R.; Fan, C.; Zhang, Z.; Yan, H.; Wu, X. Simultaneous Screening of 733 Pesticide Residues in Fruits and Vegetables by a GC/LC-Q-TOFMS Combination Technique. Engineering 2020, 6, 432–441. [Google Scholar] [CrossRef]
- SANTE/11312/2021; Analytical Quality Control and Method Validation Procedures for Pesticide Residues Analysis in Food and Feed. European Commission: Brussels, Belgium, 2021.
- Kresse, M.; Drinda, H.; Romanotto, A.; Speer, K. Simultaneous Determination of Pesticides, Mycotoxins, and Metabolites as Well as Other Contaminants in Cereals by LC-LC-MS/MS. J. Chromatogr. B 2019, 1117, 86–102. [Google Scholar] [CrossRef]
- Reichert, B.; Pizzutti, I.R.; Jänisch, B.D.; Zorzella Fontana, M.E. Improving Analytical Performance for Pesticides and Mycotoxins Determination in Brazilian Table Olives: One Extraction and One Analysis. Anal. Methods 2024, 16, 4124–4135. [Google Scholar] [CrossRef]
- Rasche, C.; Fournes, B.; Dirks, U.; Speer, K. Multi-Residue Pesticide Analysis (Gas Chromatography–Tandem Mass Spectrometry Detection)—Improvement of the Quick, Easy, Cheap, Effective, Rugged, and Safe Method for Dried Fruits and Fat-Rich Cereals—Benefit and Limit of a Standardized Apple Purée Calibration (Screening). J. Chromatogr. A 2015, 1403, 21–31. [Google Scholar]
- Wu, X.; Li, J.; Wei, J.; Tong, K.; Xie, Y.; Chang, Q.; Yu, X.; Li, B.; Lu, M.; Fan, C. Multi-Residue Analytical Method Development and Dietary Exposure Risk Assessment of 345 Pesticides in Mango by LC-Q-TOF/MS. Food Control 2024, 170, 111016. [Google Scholar] [CrossRef]
- Yan, X.; Zhang, H.; Zhu, Z.; Xie, Y.; Wu, X.; Shi, Z.; Fan, C.; Chen, H. Simultaneous Determination of 78 Pesticide Residues and 16 Mycotoxins in Tsampa by an Improved QuEChERS Method Coupled with Ultra Performance Liquid Chromatography-Tandem Mass Spectrometry. Anal. Methods 2024, 16, 5178–5190. [Google Scholar] [CrossRef]
- Azimeh, K.; Ansari, M.; Amirahmadi, M.; Shahidi, M.; Mohamadi, N.; Kazemipour, M. Pesticide Residues in Dates Using a Modified QuEChERS Method and GC-MS/MS. Food Addit. Contam. Part B 2022, 15, 168–176. [Google Scholar]
- Zhang, Z.; Dong, M.; Hao, X.; Han, L.; Song, S.; Yao, W. Evaluation of Cleanup Procedures in Pesticide Multi-Residue Analysis with QuEChERS in Cinnamon Bark. Food Chem. 2019, 276, 140–146. [Google Scholar] [CrossRef]
- Zheng, K.; Zheng, H.; Yu, Y.; Su, J.; Chen, L.; Zheng, M.; Liu, L.; Wu, X.; Chen, D.; Meng, X. Simultaneous Determination of Four Pesticides Residues in Rice by Modified QuEChERS Coupled with GC-MS/MS. J. Food Compos. Anal. 2024, 133, 106396. [Google Scholar] [CrossRef]
- Xu, R.; Xie, Q.; Li, X.; Zhao, H.; Liu, X.; Wei, Y.; Qiu, A. Modified QuEChERS Method Based on Multi-Walled Carbon Nanotubes Coupled with Gas Chromatography-Tandem Mass Spectrometry for the Detection of 10 Pyrethroid Pesticide Residues in Tea. Chin. J. Chromatogr. 2022, 40, 469–476. [Google Scholar] [CrossRef]
- Yu, C.; Hao, D.; Chu, Q.; Wang, T.; Liu, S.; Lan, T.; Wang, F.; Pan, C. A One Adsorbent QuEChERS Method Coupled with LC-MS/MS for Simultaneous Determination of 10 Organophosphorus Pesticide Residues in Tea. Food Chem. 2020, 321, 126657. [Google Scholar] [CrossRef]
- Liu, S.; Xiao, P.; Xu, J.; Yang, D.; Chen, M.; Zhou, C. Deep eutectic solvent-modified multi-walled carbon nanotubes as QuEChERS adsorbents coupled with gas chromatography-mass spectrometry for the determination of pesticide residues in Lycium barbarum. J. Food Compos. Anal. 2025, 141, 107350. [Google Scholar] [CrossRef]
- Lu, Q.; Ruan, H.; Sun, X.; Luo, J.; Yang, M. Contamination status and health risk assessment of 31 mycotoxins in six edible and medicinal plants using a novel green defatting and depigmenting pretreatment coupled with LC-MS/MS. J. LWT Food Sci. Technol. 2022, 161, 113401. [Google Scholar] [CrossRef]
- Zheng, W.; Li, X.; Wang, X.; Hua, M.Z.; Wu, Y.; Wang, Y.; Wu, Z.; He, X.; Xu, D.; Zheng, W.; et al. Simultaneous Determination of 147 Pesticide Residues in Traditional Chinese Medicines by GC-MS/MS. ACS Omega 2023, 8, 28663. [Google Scholar]
- Li, R.; Li, M.; Wang, T.; Wang, T.; Chen, J.; Francis, F.; Fan, B.; Kong, Z.; Dai, X. Screening of Pesticide Residues in Traditional Chinese Medicines Using Modified QuEChERS Sample Preparation Procedure and LC-MS/MS Analysis. J. Chromatogr. B 2020, 1152, 122224. [Google Scholar] [CrossRef]
- Xie, S.; Gu, S.; Gu, L.; Li, J. Determination of 16 Mycotoxins in Three Radix and Rhizome Chinese Herbal Medicines by mPFC-QuEChERS with UPLC-MS/MS. J. Instrum. Anal. 2024, 43, 1003–1010. [Google Scholar]
- Reinholds, I.; Pugajeva, I.; Bavrins, K.; Kuckovska, G.; Bartkevics, V. Mycotoxins, Pesticides and Toxic Metals in Commercial Spices and Herbs. Food Addit. Contam. Part B 2016, 10, 5–14. [Google Scholar] [CrossRef] [PubMed]
- Tóth, E.; Bálint, M.; Tölgyesi, Á. False Positive Identification of Pesticides in Food Using the European Standard Method and LC-MS/MS Determination: Examples and Solutions from Routine Applications. Appl. Sci. 2022, 12, 12005. [Google Scholar] [CrossRef]
- Council of Europe, European Directorate for the Quality of Medicines & HealthCare (EDQM). European Pharmacopoeia, 10th ed.; Council of Europe: Strasbourg, France, 2019. [Google Scholar]
- Li, L.; Qiu, M.; He, S.; Luo, Y.; Huang, J.; Lu, M. HPLC-MS/MS determination of 119 pesticides in Astragali Radix. Chin. J. Pharm. Anal. 2012, 32, 424–433. [Google Scholar]




| No. | Compound | Retention Time (min) | Precursorion (m/z) | Production 1 (m/z) | Production 2 (m/z) | No. | Compound | Retention Time (min) | Precursorion (m/z) | Production 1 (m/z) | Production 2 (m/z) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Pesticides (187) | 96 | Metconazole | 18.2 | 320.1524 | 70.0400 | 125.0153 | |||||
| 1 | Acetamiprid | 5.4 | 223.0745 | 126.0105 | 90.0338 | 97 | Methamidophos | 1.9 | 142.0086 | 94.0052 | 124.9821 |
| 2 | Acetochlor | 17.2 | 270.1255 | 224.0837 | 148.1121 | 98 | Methidathion | 13.7 | 302.9691 | 145.0066 | 85.0396 |
| 3 | Alachlor | 17.2 | 270.1255 | 238.0993 | 162.1277 | 99 | Methiocarb | 15.2 | 226.0900 | 169.0678 | 121.0650 |
| 4 | Aldicarb-sulfone | 3.2 | 223.0747 | 86.0600 | 148.0427 | 100 | Methoxyfenozide | 16.7 | 369.2173 | 149.0597 | 313.1547 |
| 5 | Ametryn | 13.0 | 228.1277 | 186.0808 | 68.0243 | 101 | Metribuzin | 8.2 | 215.0961 | 187.1012 | 84.0808 |
| 6 | Atrazine | 12.2 | 216.1010 | 174.0541 | 104.0010 | 102 | Metsulfuron-methyl | 9.9 | 382.0816 | 167.0564 | 135.0441 |
| 7 | Avermectin | 20.5 | 895.4818 | 789.9903 | 896.4840 | 103 | Monocrotophos | 4.1 | 224.0682 | 127.0155 | 193.0260 |
| 8 | Azaconazole | 12.3 | 300.0301 | 230.9974 | 158.9763 | 104 | Myclobutanil | 16.6 | 289.1215 | 125.0153 | 70.0400 |
| 9 | Azinphos-ethyl | 17.0 | 346.0443 | 132.0444 | 232.9491 | 105 | Napropamide | 17.2 | 272.1645 | 171.0804 | 199.0754 |
| 10 | Azoxystrobin | 15.9 | 404.1241 | 372.0979 | 329.0795 | 106 | Nicosulfuron | 9.3 | 411.1081 | 182.0560 | 213.0322 |
| 11 | Benalaxyl | 18.1 | 326.1751 | 294.1489 | 208.1332 | 107 | Omethoate | 2.8 | 214.0297 | 182.9875 | 154.9926 |
| 12 | Bitertanol | 18.4 | 338.1863 | 269.1536 | 251.1417 | 108 | Oxadiazon | 19.2 | 345.0767 | 303.0298 | 219.9563 |
| 13 | Boscalid | 16.0 | 343.0399 | 307.0633 | 271.0866 | 109 | Oxadixyl | 7.8 | 279.1339 | 219.1128 | 133.0886 |
| 14 | Bromuconazole | 16.8 | 375.9614 | 158.9763 | 306.9273 | 110 | Paclobutrazol | 16.2 | 294.1368 | 70.0400 | 125.0153 |
| 15 | Buprofezin | 19.1 | 306.1635 | 201.1056 | 106.0651 | 111 | Paraoxon-ethyl | 12.8 | 276.0632 | 220.0002 | 174.0076 |
| 16 | Cadusafos | 18.6 | 271.0950 | 158.9698 | 130.9385 | 112 | Paraoxon-methyl | 7.3 | 248.0319 | 202.0389 | 109.0049 |
| 17 | Carbofuran | 9.1 | 222.1125 | 165.0910 | 123.0441 | 113 | Penconazole | 12.6 | 284.0716 | 158.9763 | 70.0400 |
| 18 | Carbofuran-3-Hydroxy | 5.2 | 238.1074 | 163.0754 | 181.0859 | 114 | Phenthoate | 17.7 | 321.0379 | 247.0011 | 163.0754 |
| 19 | Carboxin | 9.9 | 236.0740 | 143.0161 | 93.0573 | 115 | Phorate-Sulfone | 12.5 | 293.0097 | 247.0215 | 171.0230 |
| 20 | Chlordimeform | 4.0 | 197.0840 | 117.0573 | 152.0262 | 116 | Phorate-sulfoxide | 12.2 | 277.0150 | 199.0011 | 170.9698 |
| 21 | Chlorfenvinphos | 18.1 | 358.9768 | 155.0468 | 204.9371 | 117 | Phosalone | 18.3 | 367.9941 | 181.9998 | 110.9996 |
| 22 | Chlorpyrifos | 19.4 | 349.9336 | 321.9023 | 197.9268 | 118 | Phosfolan | 6.8 | 256.0217 | 227.9907 | 139.9563 |
| 23 | Chlorsulfuron | 11.3 | 358.0371 | 167.0564 | 141.0771 | 119 | Phosfolan-Methyl | 4.1 | 227.9911 | 167.9873 | 109.0044 |
| 24 | Clothianidin | 4.6 | 250.0160 | 169.0542 | 131.9669 | 120 | Phosmet | 14.5 | 318.0018 | 160.0393 | 133.0284 |
| 25 | Coumaphos | 18.1 | 363.0217 | 226.9926 | 306.9591 | 121 | Phosphamidon | 8.1 | 300.0762 | 174.0678 | 127.0155 |
| 26 | Cyproconazole | 17.4 | 292.1211 | 125.0153 | 70.0400 | 122 | Phoxim | 17.7 | 299.0614 | 129.0447 | 124.9821 |
| 27 | Cyprodinil | 17.2 | 226.1339 | 93.0573 | 210.1026 | 123 | Picoxystrobin | 17.7 | 368.1104 | 205.0859 | 145.0648 |
| 28 | Cyromazine | 1.9 | 167.1036 | 85.0511 | 125.0821 | 124 | Piperonyl Butoxide | 19.3 | 356.2423 | 177.0898 | 178.0940 |
| 29 | Dicrotophos | 4.5 | 238.0839 | 112.0757 | 127.0155 | 125 | Pirimicarb | 7.8 | 239.1503 | 182.1288 | 72.0444 |
| 30 | Diethofencarb | 15.3 | 268.1543 | 226.1074 | 152.0706 | 126 | Pirimiphos-ethyl | 19.2 | 334.1349 | 198.1059 | 170.0746 |
| 31 | Difenoconazole | 18.6 | 406.0721 | 408.0690 | 251.0021 | 127 | Pirimiphos-methyl | 18.2 | 306.1036 | 164.1182 | 108.0556 |
| 32 | Diflubenzuron | 13.9 | 311.0393 | 158.0412 | 141.0146 | 128 | Pretilachlor | 18.9 | 312.1725 | 252.1150 | 176.1434 |
| 33 | Diflufenican | 18.7 | 395.0813 | 266.0412 | 246.0350 | 129 | Prochloraz | 18.3 | 376.0381 | 307.9988 | 309.9951 |
| 34 | Dimethoate | 5.0 | 230.0069 | 198.9647 | 170.9698 | 130 | Profenofos | 19.0 | 372.9424 | 302.8642 | 344.9111 |
| 35 | Dimethomorph | 16.5 | 388.1310 | 301.0626 | 165.0546 | 131 | Promecarb | 15.9 | 208.1332 | 151.1117 | 109.0648 |
| 36 | Diniconazole | 18.4 | 326.0821 | 328.0799 | 327.0855 | 132 | Prometryn | 15.6 | 242.1434 | 158.0495 | 200.0964 |
| 37 | Dioxacarb | 8.2 | 224.0917 | 167.0703 | 123.0441 | 133 | Propachlor | 13.0 | 212.0837 | 170.0367 | 94.0651 |
| 38 | Edifenphos | 18.0 | 311.0324 | 283.0011 | 172.9821 | 134 | Propamocarb | 2.8 | 189.1598 | 102.0550 | 144.1019 |
| 39 | Epoxiconazole | 15.9 | 330.0806 | 332.0784 | 331.0837 | 135 | Propiconazole | 18.1 | 342.0771 | 158.9763 | 69.0699 |
| 40 | Ethametsulfuron | 6.9 | 397.0924 | 196.0823 | 168.0510 | 136 | Pymetrozine | 12.2 | 218.1036 | 105.0447 | 78.0338 |
| 41 | Ethion | 19.3 | 384.9949 | 199.0011 | 170.9698 | 137 | Pyraclostrobin | 18.3 | 388.1059 | 194.0812 | 163.0628 |
| 42 | Ethoprophos | 17.2 | 243.0637 | 172.9854 | 215.0324 | 138 | Pyridaben | 20.2 | 365.1449 | 309.0823 | 147.1168 |
| 43 | Etofenprox | 20.8 | 394.2378 | 399.2619 | 393.2476 | 139 | Pyrimethanil | 13.5 | 200.1182 | 168.0682 | 182.0838 |
| 44 | Etrimfos | 17.8 | 293.0719 | 265.0406 | 124.9821 | 140 | Pyriproxyfen | 19.3 | 322.1438 | 227.1067 | 96.0444 |
| 45 | Fenamiphos | 17.6 | 304.1131 | 217.0083 | 201.9848 | 141 | Quinalphos | 17.7 | 299.0614 | 271.0301 | 242.9985 |
| 46 | Fenamiphos-sulfone | 11.5 | 336.1029 | 308.0716 | 266.0247 | 142 | Quinoxyfen | 19.4 | 308.0040 | 196.9789 | 213.9821 |
| 47 | Fenamiphos-sulfoxide | 11.0 | 320.1080 | 233.0032 | 292.0767 | 143 | Quizalofop-ethyl | 19.0 | 373.0950 | 299.0577 | 255.0315 |
| 48 | Fenarimol | 17.2 | 331.0399 | 268.0524 | 259.0057 | 144 | Simazine | 8.5 | 202.0854 | 132.0323 | 124.0869 |
| 49 | Fenazaquin | 20.4 | 307.1805 | 161.1325 | 57.0699 | 145 | Simeconazole | 17.1 | 294.1432 | 70.0400 | 135.0605 |
| 50 | Fenbuconazole | 17.4 | 337.1219 | 339.1194 | 338.1248 | 146 | Spirodiclofen | 19.9 | 411.1124 | 313.0393 | 212.9505 |
| 51 | Fenhexamid | 17.0 | 302.0709 | 97.1012 | 55.0542 | 147 | Spirotetramat | 17.3 | 374.1962 | 330.2064 | 302.1751 |
| 52 | Fenpropathrin | 19.7 | 350.1751 | 125.0961 | 97.1012 | 148 | Spirotetramat-enol | 4.8 | 302.1758 | 216.1017 | 270.1487 |
| 53 | Fenpropimorph | 15.5 | 304.2625 | 305.2672 | 147.1160 | 149 | Spirotetramat-enol-glucoside | 4.8 | 464.2283 | 302.1750 | 324.1573 |
| 54 | Fensulfothion | 13.9 | 309.0379 | 252.9753 | 234.9647 | 150 | Spirotetramat-keto-hydroxy | 14.2 | 318.1702 | 300.1597 | 268.1333 |
| 55 | Fensulfothion-oxon | 7.3 | 293.0607 | 236.9980 | 218.9873 | 151 | Spirotetramat-mono-hydroxy | 9.2 | 304.1915 | 254.1537 | 211.1475 |
| 56 | Fensulfothion-Oxon-Sulfone | 7.5 | 309.0565 | 252.9932 | 175.0150 | 152 | Spiroxamine | 16.1 | 298.2741 | 144.1383 | 100.1121 |
| 57 | Fensulfothion-sulfone | 14.1 | 325.0328 | 268.9687 | 190.9914 | 153 | Sulfentrazone | 10.5 | 386.9891 | 306.9944 | 308.0038 |
| 58 | Fenthion | 17.8 | 279.0273 | 247.0011 | 169.0140 | 154 | Sulfotep | 17.8 | 323.0300 | 171.0239 | 294.9987 |
| 59 | Fenthion-oxon | 14.6 | 263.0501 | 231.0239 | 216.0005 | 155 | Tebuconazole | 17.9 | 308.1527 | 310.1498 | 309.1552 |
| 60 | Fenthion-oxon-sulfone | 5.9 | 295.0403 | 217.0624 | 296.0430 | 156 | Tebufenpyrad | 19.2 | 334.1681 | 117.0209 | 145.0522 |
| 61 | Fenthion-oxon-sulfoxide | 5.7 | 279.0451 | 264.0216 | 262.0423 | 157 | Terbufos | 16.0 | 289.0514 | 103.0576 | 57.0699 |
| 62 | Fenthion-sulfone | 11.6 | 311.0171 | 278.9909 | 124.9821 | 158 | Terbufos-Sulfone | 15.1 | 321.0412 | 171.0240 | 275.0536 |
| 63 | Fenthion-sulfoxide | 11.1 | 295.0222 | 279.9987 | 278.0195 | 159 | Terbufos-Sulfoxide | 15.3 | 305.0465 | 187.0010 | 158.9694 |
| 64 | Flonicamid | 3.6 | 230.0536 | 203.0427 | 148.0369 | 160 | Tetrachlorvinphos | 17.7 | 364.9065 | 203.9293 | 238.8982 |
| 65 | Fluacrypyrim | 18.6 | 427.1475 | 205.0859 | 145.0648 | 161 | Tetraconazole | 17.2 | 372.0290 | 374.0250 | 158.9752 |
| 66 | Flucythrinate | 19.6 | 469.1931 | 470.3460 | 468.3107 | 162 | Thiabendazole | 4.6 | 202.0433 | 175.0324 | 131.0604 |
| 67 | Flufenacet | 17.1 | 364.0737 | 194.0976 | 152.0506 | 163 | Thiacloprid | 6.3 | 253.0309 | 126.0087 | 90.0338 |
| 68 | Flufenoxuron | 19.5 | 489.0435 | 158.0401 | 141.0146 | 164 | Thiamethoxam | 3.8 | 292.0266 | 211.0648 | 181.0542 |
| 69 | Flumioxazin | 14.8 | 355.1088 | 327.1085 | 210.1506 | 165 | Thidiazuron | 8.7 | 221.0492 | 102.0120 | 77.0386 |
| 70 | Flumorph | 14.6 | 372.1606 | 285.0909 | 165.0530 | 166 | Thifluzamide | 17.4 | 526.8485 | 528.8456 | 488.8332 |
| 71 | Fluroxypyr | 7.3 | 254.9734 | 208.9679 | 180.9730 | 167 | Thiobencarb | 18.4 | 258.0714 | 125.0153 | 100.0757 |
| 72 | Flusilazole | 17.6 | 316.1076 | 247.0749 | 165.0697 | 168 | Thiocyclam | 2.0 | 182.0126 | 136.9523 | 73.0092 |
| 73 | Flutriafol | 12.9 | 302.1099 | 233.0773 | 123.0241 | 169 | Tralkoxydim | 19.6 | 330.2064 | 284.1645 | 285.1723 |
| 74 | Fonofos | 17.9 | 247.0375 | 137.0184 | 108.9871 | 170 | Triadimefon | 16.6 | 294.1004 | 197.0714 | 225.0659 |
| 75 | Fosthiazate | 11.9 | 284.0538 | 227.9909 | 104.0165 | 171 | Triadimenol | 16.8 | 296.1158 | 70.0405 | 298.1133 |
| 76 | Heptenophos | 13.7 | 251.0234 | 127.0155 | 109.0049 | 172 | Triazophos | 16.8 | 314.0723 | 162.0662 | 119.0604 |
| 77 | Hexaconazole | 18.1 | 314.0825 | 316.0790 | 315.0848 | 173 | Trichlorfon | 5.0 | 256.9299 | 220.9532 | 109.0051 |
| 78 | Hexythiazox | 19.4 | 353.1085 | 228.0240 | 168.0570 | 174 | Tricyclazole | 7.1 | 190.0433 | 136.0215 | 163.0324 |
| 79 | Imazalil | 12.7 | 297.0550 | 299.0526 | 158.9761 | 175 | Trifloxystrobin | 18.7 | 409.1370 | 186.0525 | 145.0260 |
| 80 | Imidacloprid | 4.6 | 256.0596 | 209.0589 | 175.0978 | 176 | Triflumizole | 18.8 | 346.0920 | 278.0547 | 280.0524 |
| 81 | Imidaclothiz | 4.9 | 262.0158 | 181.0537 | 265.1525 | 177 | Trifluralin | 17.8 | 336.1170 | 342.2657 | 345.2389 |
| 82 | Indanofan | 17.2 | 341.0941 | 348.2548 | 348.7550 | 178 | Triflusulfuron-methyl | 16.5 | 493.1112 | 264.0698 | 461.0850 |
| 83 | Indoxacarb | 18.7 | 528.0780 | 293.0319 | 218.0417 | 179 | Uniconazole | 17.4 | 292.1213 | 294.1191 | 293.1246 |
| 84 | Ipconazole | 19.2 | 334.1681 | 70.0400 | 125.0153 | 180 | Zoxamide | 18.0 | 336.0319 | 186.9712 | 158.9763 |
| 85 | Isazofos | 16.7 | 314.0490 | 162.0424 | 272.0020 | Mycotoxins (10) | |||||
| 86 | Isocarbophos | 13.6 | 312.0433 | 269.9950 | 236.0077 | 1 | Aflatoxin B1 | 9.6 | 313.0700 | 285.0741 | 269.0432 |
| 87 | Isoprothiolane | 16.3 | 291.0719 | 231.0144 | 188.9675 | 2 | Aflatoxin B2 | 8.5 | 315.0862 | 259.0583 | 287.0891 |
| 88 | Linuron | 14.6 | 249.0192 | 159.9710 | 182.0241 | 3 | Aflatoxin G1 | 7.5 | 329.0638 | 243.0629 | 215.0682 |
| 89 | Malaoxon | 10.3 | 315.0662 | 127.0390 | 99.0077 | 4 | Aflatoxin G2 | 6.9 | 331.0807 | 313.0709 | 285.0760 |
| 90 | Malathion | 16.3 | 331.0433 | 127.0390 | 285.0015 | 5 | Deoxynivalenol | 3.4 | 297.1328 | 298.3453 | 249.1101 |
| 91 | Mandipropamid | 16.5 | 412.1310 | 328.1094 | 356.1043 | 6 | Ochratoxin A | 16.8 | 404.0895 | 239.0098 | 220.9990 |
| 92 | Matrine | 2.3 | 249.1967 | 148.1117 | 250.1991 | 7 | Stachybotrylactam | 18.2 | 386.2326 | 387.2331 | 178.0488 |
| 93 | Mecarbam | 17.2 | 330.0593 | 226.9960 | 142.9385 | 8 | T-2 toxin | 15.6 | 484.2541 | 215.1063 | 185.0960 |
| 94 | Mepronil | 16.3 | 270.1489 | 228.1019 | 119.0491 | 9 | Tentoxin | 14.3 | 415.2340 | 358.2125 | 302.1499 |
| 95 | Metalaxyl | 13.7 | 280.1543 | 220.1332 | 192.1383 | 10 | Zearalenone | 16.6 | 319.1540 | 283.1309 | 301.1413 |
| Number | A: PSA (mg) | B: C18 (mg) | C: MWCNTs (mg) | Y (%) |
|---|---|---|---|---|
| 1 | 300 | 50 | 5 | 83.76 |
| 2 | 200 | 150 | 10 | 83.76 |
| 3 | 100 | 100 | 10 | 82.23 |
| 4 | 100 | 50 | 5 | 86.30 |
| 5 | 300 | 100 | 10 | 81.73 |
| 6 | 300 | 100 | 0 | 81.22 |
| 7 | 100 | 150 | 5 | 85.28 |
| 8 | 200 | 50 | 10 | 81.73 |
| 9 | 200 | 100 | 5 | 94.92 |
| 10 | 200 | 100 | 5 | 95.94 |
| 11 | 200 | 150 | 0 | 85.28 |
| 12 | 300 | 150 | 5 | 85.28 |
| 13 | 200 | 50 | 0 | 86.8 |
| 14 | 200 | 100 | 5 | 94.42 |
| 15 | 200 | 100 | 5 | 96.45 |
| 16 | 100 | 100 | 0 | 84.26 |
| 17 | 200 | 100 | 5 | 95.43 |
| Source | Sum of Squares | df | Mean Square | F-Value | p-Value | Significance |
|---|---|---|---|---|---|---|
| Model | 499.8 | 9 | 55.53 | 65.68 | <0.0001 | ** |
| A-PSA | 4.62 | 1 | 4.62 | 5.46 | 0.052 | |
| B-C18 | 0.1275 | 1 | 0.1275 | 0.1508 | 0.7093 | |
| C-MWCNTs | 8.22 | 1 | 8.22 | 9.72 | 0.0169 | * |
| AB | 1.61 | 1 | 1.61 | 1.91 | 0.2097 | |
| AC | 1.61 | 1 | 1.61 | 1.91 | 0.2097 | |
| BC | 3.15 | 1 | 3.15 | 3.73 | 0.0949 | |
| A2 | 159.5 | 1 | 159.5 | 188.63 | <0.0001 | ** |
| B2 | 71.55 | 1 | 71.55 | 84.62 | <0.0001 | ** |
| C2 | 201.47 | 1 | 201.47 | 238.27 | <0.0001 | ** |
| Residual | 5.92 | 7 | 0.8455 | |||
| Lack of fit | 3.34 | 3 | 1.11 | 1.72 | 0.2994 | |
| Pure error | 2.58 | 4 | 0.6452 | |||
| Cor total | 505.72 | 16 |
| Matrix | Number of Analytes | Extraction Method | Instrumentation | LOQ Range (μg/kg) | Total Analysis Time | References |
|---|---|---|---|---|---|---|
| Astragalus | 33 pesticides | QuEChERS | LC-MS/MS, GC-MS/MS | 10–100 | 50 min | [8] |
| Six edible and medicinal plants | 31 mycotoxins | QuEChERS | LC-MS/MS | 0.03–50 | 15 min | [39] |
| Two TCM | 147 pesticides | QuEChERS | GC-MS/MS | 10–50 | 30 min | [40] |
| Three TCM | 108 pesticides | QuEChERS | UHPLC-MS/MS | 0.01–50 | 21 min | [41] |
| Three rhizome TCM | 16 mycotoxins | mPFC-QuEChERS | UPLC-MS/MS | 0.5–50 | 17 min | [42] |
| Spices and herbs | 134 pesticides and 11 mycotoxins | QuEChERS | HPLC-QqQ-MS/MS | 2.4–12 | 15 min | [43] |
| Astragalus | 187 pesticides and 10 mycotoxins | QuEChERS | LC-Q-TOF/MS | 2–20 | 23 min | This work |
| No. | Compound | Detected Quantity | Detection Rate (%) | Concentration Range (µg/kg) |
|---|---|---|---|---|
| 1 | Azoxystrobin | 2 | 10.0 | 5.08–10.88 |
| 2 | Difenoconazole | 1 | 5.0 | 5.66 |
| 3 | Tebuconazole | 3 | 15.0 | 11.15–34.28 |
| 4 | Ochratoxin A | 1 | 5.0 | 45.10 |
| 5 | Aflatoxin B2 | 2 | 10.0 | 16.40–21.43 |
| 6 | Aflatoxin G2 | 2 | 10.0 | 6.22–27.24 |
| Sum of aflatoxins | 2 | 10.0 | 22.62–48.67 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Yin, H.; Chen, Y.; Wang, Y.; Shi, Z.; Hu, X.; Zhang, H. Response Surface Methodology-Optimized QuEChERS Combined with Liquid Chromatography–Quadrupole-Time-of-Flight Mass Spectrometry for Simultaneous Screening of Pesticides and Mycotoxins in Astragalus. Separations 2026, 13, 76. https://doi.org/10.3390/separations13030076
Yin H, Chen Y, Wang Y, Shi Z, Hu X, Zhang H. Response Surface Methodology-Optimized QuEChERS Combined with Liquid Chromatography–Quadrupole-Time-of-Flight Mass Spectrometry for Simultaneous Screening of Pesticides and Mycotoxins in Astragalus. Separations. 2026; 13(3):76. https://doi.org/10.3390/separations13030076
Chicago/Turabian StyleYin, Hang, Yanlong Chen, Yingchun Wang, Zhihong Shi, Xueyan Hu, and Hongyi Zhang. 2026. "Response Surface Methodology-Optimized QuEChERS Combined with Liquid Chromatography–Quadrupole-Time-of-Flight Mass Spectrometry for Simultaneous Screening of Pesticides and Mycotoxins in Astragalus" Separations 13, no. 3: 76. https://doi.org/10.3390/separations13030076
APA StyleYin, H., Chen, Y., Wang, Y., Shi, Z., Hu, X., & Zhang, H. (2026). Response Surface Methodology-Optimized QuEChERS Combined with Liquid Chromatography–Quadrupole-Time-of-Flight Mass Spectrometry for Simultaneous Screening of Pesticides and Mycotoxins in Astragalus. Separations, 13(3), 76. https://doi.org/10.3390/separations13030076





