Geopolymers Based on Fly Ash for Organic Dye Removal from Water
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Synthesis of Materials
2.3. Material Characterization
2.4. Adsorption Experiments
2.5. Box–Behnken Design for Adsorption Parameter Optimization
2.6. Computational Modelling by Artificial Neural Networks
3. Results and Discussion
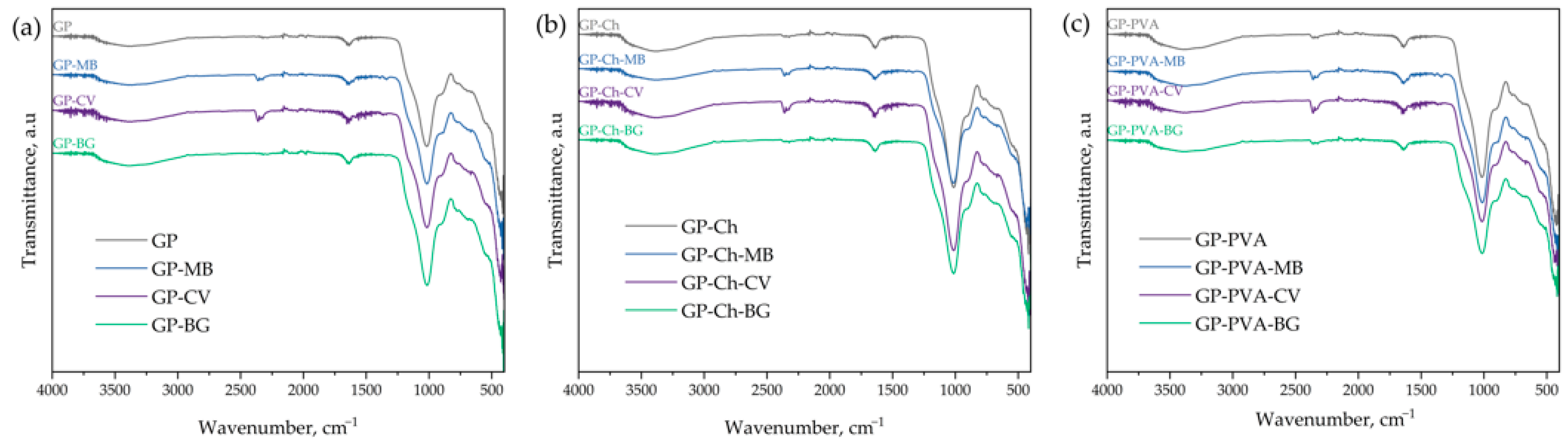
3.1. Material Characterization
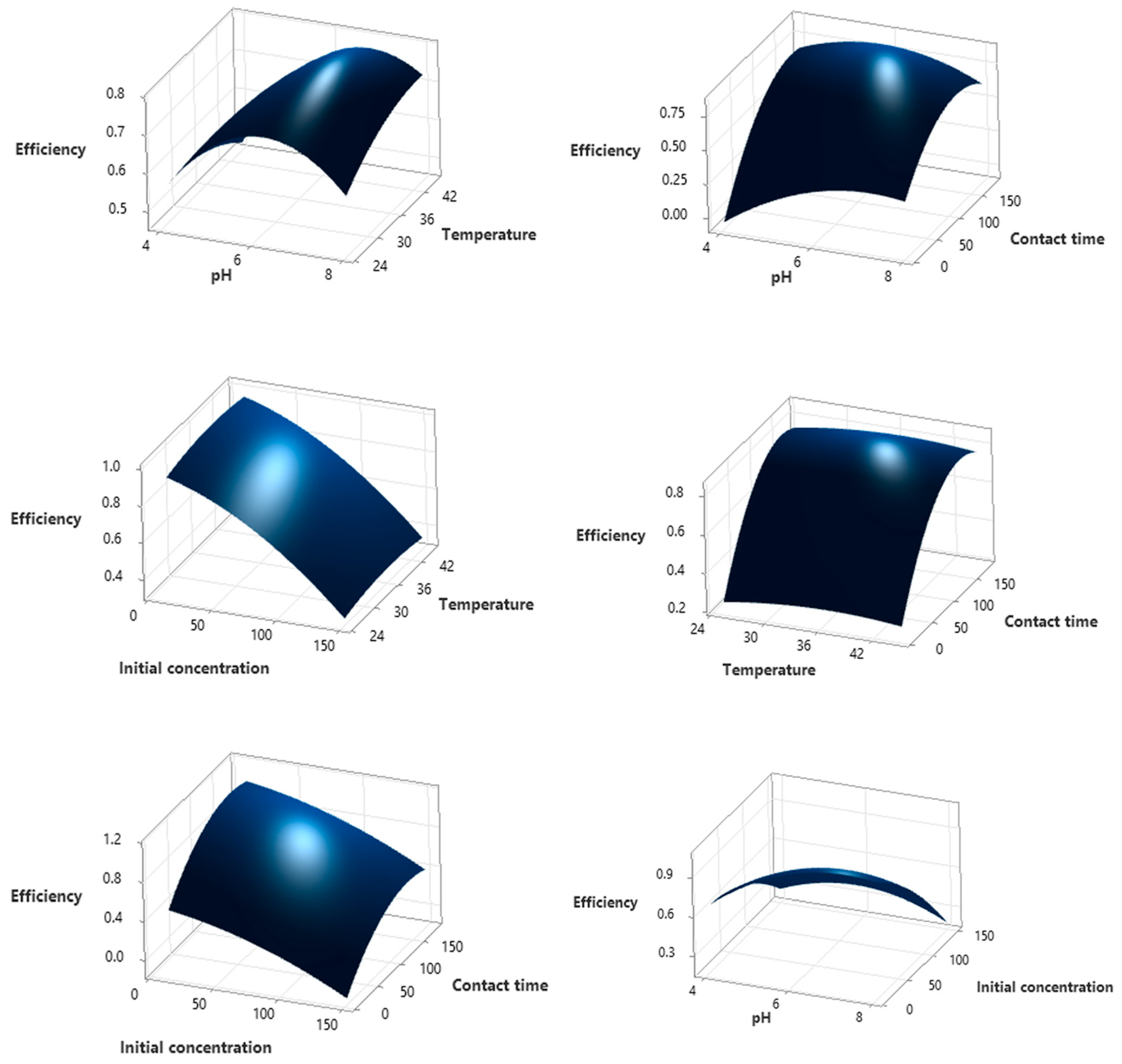
3.2. Optimization of Adsorption Parameters Using Box–Behnken Design
3.3. Kinetic Models of the Adsorption Process
3.4. Isotherm Models of the Adsorption Process
3.5. Thermodynamics of the Adsorption Process
3.6. Application of Geopolymeric Adsorbents for Dye Adsorption from Real Wastewater Samples
3.7. Comparison of Adsorption Process with Other Studies
3.8. Prediction of the Dye Adsorption Efficiency on Geopolymers Using Artificial Neural Network
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hmoudah, M.; Paparo, R.; De Luca, M.; Fortunato, M.E.; Tesser, R.; Di Serio, M.; Ferone, C.; Roviello, G.; Tarallo, O.; Russo, V. Adsorption of Methylene Blue on Metakaolin-Based Geopolymers: A Kinetic and Thermodynamic Investigation. ChemEngineering 2025, 9, 79. [Google Scholar] [CrossRef]
- Chafi, S.; Cuevas-Aranda, M.; Martínez-Cartas, M.L.; Sánchez, S. Production of Bioadsorbents via Low-Temperature Pyrolysis of Exhausted Olive Pomace for the Removal of Methylene Blue from Aqueous Media. Molecules 2025, 30, 3254. [Google Scholar] [CrossRef]
- Azlan Zahari, K.F.; Sahu, U.K.; Khadiran, T.; Surip, S.N.; Alothman, Z.A.; Jawad, A.H. Mesoporous Activated Carbon from Bamboo Waste via Microwave-Assisted K2CO3 Activation: Adsorption Optimization and Mechanism for Methylene Blue Dye. Separations 2022, 9, 390. [Google Scholar] [CrossRef]
- Milojković, N.; Simović, B.; Žunić, M.; Radovanović, L.; Prekajski-Đorđević, M.; Dapčević, A. Modified Z-scheme Heterojunction of TiO2 /Polypyrrole Recyclable Photocatalyst. J. Am. Ceram. Soc. 2025, 108, e20431. [Google Scholar] [CrossRef]
- Bounaas, M.; Bouguettoucha, A.; Chebli, D.; Derbal, K.; Benalia, A.; Pizzi, A. Effect of Washing Temperature on Adsorption of Cationic Dyes by Raw Lignocellulosic Biomass. Appl. Sci. 2024, 14, 10365. [Google Scholar] [CrossRef]
- Boumezough, Y.; Viscusi, G.; Arris, S.; Gorrasi, G.; Carabineiro, S.A.C. Synthesis and Characterization of a Nanocomposite Based on Opuntia Ficus Indica for Efficient Removal of Methylene Blue Dye: Adsorption Kinetics and Optimization by Response Surface Methodology. IJMS 2025, 26, 6717. [Google Scholar] [CrossRef]
- Gul, S.; Gul, A.; Gul, H.; Khattak, R.; Ismail, M.; Khan, S.U.; Khan, M.S.; Aouissi, H.A.; Krauklis, A. Removal of Brilliant Green Dye from Water Using Ficus Benghalensis Tree Leaves as an Efficient Biosorbent. Materials 2023, 16, 521. [Google Scholar] [CrossRef] [PubMed]
- Šekuljica, N.Ž.; Prlainović, N.Ž.; Stefanović, A.B.; Žuža, M.G.; Čičkarić, D.Z.; Mijin, D.Ž.; Knežević-Jugović, Z.D. Decolorization of Anthraquinonic Dyes from Textile Effluent Using Horseradish Peroxidase: Optimization and Kinetic Study. Sci. World J. 2015, 2015, 371625. [Google Scholar] [CrossRef] [PubMed]
- Ong, M.D.; Vasquez, I.; Alvarez, B.; Cho, D.R.; Williams, M.B.; Vincent, D.; Ali, M.A.; Aich, N.; Pinto, A.H.; Choudhury, M.R. Modification of Cellulose Acetate Microfiltration Membranes Using Graphene Oxide–Polyethyleneimine for Enhanced Dye Rejection. Membranes 2023, 13, 143. [Google Scholar] [CrossRef]
- Kuzminova, A.; Dmitrenko, M.; Salomatin, K.; Vezo, O.; Kirichenko, S.; Egorov, S.; Bezrukova, M.; Karyakina, A.; Eremin, A.; Popova, E.; et al. Holmium-Containing Metal-Organic Frameworks as Modifiers for PEBA-Based Membranes. Polymers 2023, 15, 3834. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.-S.; Liu, C.-H.; Chu, K.H.; Suen, S.-Y. Removal of Cationic Dye Methyl Violet 2B from Water by Cation Exchange Membranes. J. Membr. Sci. 2008, 309, 239–245. [Google Scholar] [CrossRef]
- El-Moselhy, M.M.; Kamal, S.M. Selective Removal and Preconcentration of Methylene Blue from Polluted Water Using Cation Exchange Polymeric Material. Groundw. Sustain. Dev. 2018, 6, 6–13. [Google Scholar] [CrossRef]
- Li, H.; Lin, Y.; Luo, Y.; Yu, P.; Hou, L. Relating Organic Fouling of Reverse Osmosis Membranes to Adsorption during the Reclamation of Secondary Effluents Containing Methylene Blue and Rhodamine B. J. Hazard. Mater. 2011, 192, 490–499. [Google Scholar] [CrossRef]
- Antanasković, A.; Lopičić, Z.; Dimitrijević-Branković, S.; Ilić, N.; Adamović, V.; Šoštarić, T.; Milivojević, M. Biochar as an Enzyme Immobilization Support and Its Application for Dye Degradation. Processes 2024, 12, 2418. [Google Scholar] [CrossRef]
- Shah, L.A.; Malik, T.; Siddiq, M.; Haleem, A.; Sayed, M.; Naeem, A. TiO2 Nanotubes Doped Poly(Vinylidene Fluoride) Polymer Membranes (PVDF/TNT) for Efficient Photocatalytic Degradation of Brilliant Green Dye. J. Environ. Chem. Eng. 2019, 7, 103291. [Google Scholar] [CrossRef]
- Hoang, N.T.; Manh, T.D.; Nguyen, V.T.; Thy Nga, N.T.; Mwazighe, F.M.; Nhi, B.D.; Hoang, H.Y.; Chang, S.W.; Chung, W.J.; Nguyen, D.D. Kinetic Study on Methylene Blue Removal from Aqueous Solution Using UV/Chlorine Process and Its Combination with Other Advanced Oxidation Processes. Chemosphere 2022, 308, 136457. [Google Scholar] [CrossRef] [PubMed]
- Bhatnagar, A.; Hogland, W.; Marques, M.; Sillanpää, M. An Overview of the Modification Methods of Activated Carbon for Its Water Treatment Applications. Chem. Eng. J. 2013, 219, 499–511. [Google Scholar] [CrossRef]
- Gebre Meskel, A.; Kwikima, M.M.; Meshesha, B.T.; Habtu, N.G.; Naik, S.V.C.S.; Vellanki, B.P. Malachite Green and Methylene Blue Dye Removal Using Modified Bagasse Fly Ash: Adsorption Optimization Studies. Environ. Chall. 2024, 14, 100829. [Google Scholar] [CrossRef]
- Radoor, S.; Karayil, J.; Devrim, Y.; Kim, H. Polyethyleneimine Functionalized Waste Tissue Paper@waste PET Composite for the Effective Adsorption and Filtration of Organic Dyes from Wastewater. Sustain. Mater. Technol. 2025, 45, e01494. [Google Scholar] [CrossRef]
- El-Eswed, B.I.; Yousef, R.I.; Alshaaer, M.; Hamadneh, I.; Al-Gharabli, S.I.; Khalili, F. Stabilization/Solidification of Heavy Metals in Kaolin/Zeolite Based Geopolymers. Int. J. Miner. Process. 2015, 137, 34–42. [Google Scholar] [CrossRef]
- Kaya, M.; Koksal, F.; Gencel, O.; Munir, M.J.; Kazmi, S.M.S. Influence of Micro Fe2O3 and MgO on the Physical and Mechanical Properties of the Zeolite and Kaolin Based Geopolymer Mortar. J. Build. Eng. 2022, 52, 104443. [Google Scholar] [CrossRef]
- Ettahiri, Y.; Bouna, L.; Brahim, A.; Benlhachemi, A.; Bakiz, B.; Sánchez-Soto, P.J.; Eliche-Quesada, D.; Pérez-Villarejo, L. Synthesis and Characterization of Porous and Photocatalytic Geopolymers Based on Natural Clay: Enhanced Properties and Efficient Rhodamine B Decomposition. Appl. Mater. Today 2024, 36, 102048. [Google Scholar] [CrossRef]
- Zhong, W.L.; Fan, L.F.; Zhang, B. Experimental Research on the Flexural Properties and Pore Structure Characteristics of Engineered Geopolymer Composites Prepared by Calcined Natural Clay. J. Build. Eng. 2024, 98, 111244. [Google Scholar] [CrossRef]
- Wang, Y.; Iqtidar, A.; Amin, M.N.; Nazar, S.; Hassan, A.M.; Ali, M. Predictive Modelling of Compressive Strength of Fly Ash and Ground Granulated Blast Furnace Slag Based Geopolymer Concrete Using Machine Learning Techniques. Case Stud. Constr. Mater. 2024, 20, e03130. [Google Scholar] [CrossRef]
- Faraji, M.F.; Ahıskalı, A.; Bayraktar, O.Y.; Ahıskalı, M.; Kaplan, G.; Aydın, A.C.; Ozbakkaloglu, T. A Green Approach to Construction: Fly Ash-Based One-Part Geopolymer Foam Concrete Reinforced with Waste Concrete Powder and Polypropylene Fibers. Constr. Build. Mater. 2025, 494, 143429. [Google Scholar] [CrossRef]
- Mladenović, N.; Kljajević, L.; Nenadović, S.; Ivanović, M.; Čalija, B.; Gulicovski, J.; Trivunac, K. The Applications of New Inorganic Polymer for Adsorption Cadmium from Waste Water. J. Inorg. Organomet. Polym. 2020, 30, 554–563. [Google Scholar] [CrossRef]
- Wang, S.; Zhang, Z.; Mao, J.; Lin, J.; Xiao, F.; Chen, S.; Liu, Y.; Qian, H.; Yan, D. Modeling the Thermal Conductivity of the Ground Granulated Blast Furnace Slag-Based Foam Geopolymer Based on Its Multi-Scale Pore Structure. J. Build. Eng. 2025, 105, 112487. [Google Scholar] [CrossRef]
- Chen, J. Thermally Activated Red Mud-Based Geopolymer Concrete: Synergistic Design for High Strength and Multidimensional Sustainability. Sustain. Chem. Pharm. 2025, 48, 102236. [Google Scholar] [CrossRef]
- Guo, Z.; Liu, T.; Zhang, Y.; Wan, Q.; Zhang, R. Efficient and Green Utilization of Bayer Red Mud in Solid Waste Based Geopolymer. J. Environ. Chem. Eng. 2025, 13, 119717. [Google Scholar] [CrossRef]
- Buha Marković, J.Z.; Marinković, A.D.; Savić, J.Z.; Mladenović, M.R.; Erić, M.D.; Marković, Z.J.; Ristić, M.Đ. Risk Evaluation of Pollutants Emission from Coal and Coal Waste Combustion Plants and Environmental Impact of Fly Ash Landfilling. Toxics 2023, 11, 396. [Google Scholar] [CrossRef] [PubMed]
- Djukic, D.; Suljagic, M.; Andjelkovic, L.; Pavlovic, V.; Bucevac, D.; Vrbica, B.; Mirkovic, M. Effect of Sintering Temperature and Calcium Amount on Compressive Strength of Brushite-Metakaolin Polymer Materials. Sci. Sinter. 2022, 54, 287–294. [Google Scholar] [CrossRef]
- Štulović, M.; Radovanović, D.; Dikić, J.; Gajić, N.; Djokić, J.; Kamberović, Ž.; Jevtić, S. Utilization of Copper Flotation Tailings in Geopolymer Materials Based on Zeolite and Fly Ash. Materials 2024, 17, 6115. [Google Scholar] [CrossRef]
- Genua, F.; Lancellotti, I.; Leonelli, C. Geopolymer-Based Stabilization of Heavy Metals, the Role of Chemical Agents in Encapsulation and Adsorption: Review. Polymers 2025, 17, 670. [Google Scholar] [CrossRef] [PubMed]
- Mladenović Nikolić, N.; Kandić, A.; Potočnik, J.; Latas, N.; Ivanović, M.; Nenadović, S.; Kljajević, L. Microstructural Analysis and Radiological Characterization of Alkali-Activated Materials Based on Aluminosilicate Waste and Metakaolin. Gels 2025, 11, 57. [Google Scholar] [CrossRef]
- Al Obeidy, N.F.; Khalil, W.I.; Ahmed, H.K. Optimization of Local Modified Metakaolin-Based Geopolymer Concrete by Taguchi Method. Open Eng. 2024, 14, 20220561. [Google Scholar] [CrossRef]
- Buzukashvili, S.; Sommerville, R.; Kökkılıç, O.; Ouzilleau, P.; Rowson, N.A.; Waters, K.E. Exploring Efficiency and Regeneration of Magnetic Zeolite Synthesized from Coal Fly Ash for Water Treatment Applications. JCIS Open 2025, 17, 100127. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, S.; Zhang, G.; Wang, X.; Zang, S.; Jia, Y. Removal of As(V) and As(III) Species from Wastewater by Adsorption on Coal Fly Ash. Desalination Water Treat. 2019, 151, 242–250. [Google Scholar] [CrossRef]
- Huang, Y.; Wang, Z.; Zhou, Z.; Zhou, Q.; Chen, J. Enhanced Adsorption of Pb(II), Cd(II), and Zn(II) by Tannic Acid-Modified Magnetic Fly Ash-Based Tobermorite. Environ. Res. 2025, 283, 122206. [Google Scholar] [CrossRef] [PubMed]
- Joseph, I.V.; Tosheva, L.; Doyle, A.M. Simultaneous Removal of Cd(II), Co(II), Cu(II), Pb(II), and Zn(II) Ions from Aqueous Solutions via Adsorption on FAU-Type Zeolites Prepared from Coal Fly Ash. J. Environ. Chem. Eng. 2020, 8, 103895. [Google Scholar] [CrossRef]
- Cui, J.; Wang, Q.; Gao, J.; Guo, Y.; Cheng, F. The Selective Adsorption of Rare Earth Elements by Modified Coal Fly Ash Based SBA-15. Chin. J. Chem. Eng. 2022, 47, 155–164. [Google Scholar] [CrossRef]
- Radojičić, T.; Trivunac, K.; Vukčević, M.; Maletić, M.; Palić, N.; Janković-Častvan, I.; Perić Grujić, A. Rare Earth Element Adsorption from Water Using Alkali-Activated Waste Fly Ash. Materials 2025, 18, 699. [Google Scholar] [CrossRef]
- Vukčević, M.; Trajković, D.; Maletić, M.; Mirković, M.; Perić Grujić, A.; Živojinović, D. Modified Fly Ash as an Adsorbent for the Removal of Pharmaceutical Residues from Water. Separations 2024, 11, 337. [Google Scholar] [CrossRef]
- Hussain, Z.; Chang, N.; Sun, J.; Xiang, S.; Ayaz, T.; Zhang, H.; Wang, H. Modification of Coal Fly Ash and Its Use as Low-Cost Adsorbent for the Removal of Directive, Acid and Reactive Dyes. J. Hazard. Mater. 2022, 422, 126778. [Google Scholar] [CrossRef]
- Huang, J.; Ma, H.; Li, Y.; Zhang, X.; Liu, Y.; Li, Z. Efficient Adsorption and Performance Optimization of Coal Fly Ash/Desulphurized Manganese Residue Composite Microspheres on Dyes. Sep. Purif. Technol. 2025, 357, 130174. [Google Scholar] [CrossRef]
- Wu, R.; Hapiz, A.; Musa, S.A.; ALOthman, Z.A.; Sillanpää, M.; Jawad, A.H. Hydrothermal Fabrication of Composite Chitosan Grafted Salicylaldehyde/Coal Fly Ash/Algae for Malachite Green Dye Removal: A Statistical Optimization. Int. J. Biol. Macromol. 2024, 280, 135897. [Google Scholar] [CrossRef]
- Rodwihok, C.; Suwannakaew, M.; Han, S.W.; Lim, Y.J.; Park, S.Y.; Woo, S.W.; Choe, J.W.; Wongratanaphisan, D.; Kim, H.S. Effective Removal of Hazardous Organic Contaminant Using Integrated Photocatalytic Adsorbents: Ternary Zinc Oxide/Zeolite-Coal Fly Ash/Reduced Graphene Oxide Nanocomposites. Colloids Surf. A Physicochem. Eng. Asp. 2023, 662, 131044. [Google Scholar] [CrossRef]
- Malek, N.N.A.; Jawad, A.H.; Ismail, K.; Razuan, R.; ALOthman, Z.A. Fly Ash Modified Magnetic Chitosan-Polyvinyl Alcohol Blend for Reactive Orange 16 Dye Removal: Adsorption Parametric Optimization. Int. J. Biol. Macromol. 2021, 189, 464–476. [Google Scholar] [CrossRef]
- Andrunik, M.; Skalny, M.; Bajda, T. Functionalized Adsorbents Resulting from the Transformation of Fly Ash: Characterization, Modification, and Adsorption of Pesticides. Sep. Purif. Technol. 2023, 309, 123106. [Google Scholar] [CrossRef]
- Zhou, L.; Chen, Y.-L.; Zhang, X.-H.; Tian, F.-M.; Zu, Z.-N. Zeolites Developed from Mixed Alkali Modified Coal Fly Ash for Adsorption of Volatile Organic Compounds. Mater. Lett. 2014, 119, 140–142. [Google Scholar] [CrossRef]
- Zhu, T.; Zhang, X.; Han, Y.; Liu, T.; Wang, B.; Zhang, Z. Preparation of Zeolite X by the Aluminum Residue from Coal Fly Ash for the Adsorption of Volatile Organic Compounds. Front. Chem. 2019, 7, 341. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; Jiang, J.; Gao, X.; Ding, M. Improving the Mechanical Properties of Fly Ash-Based Geopolymer Composites with PVA Fiber and Powder. Materials 2022, 15, 2363. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Malik, M.A.; Qi, Z.; Huang, B.; Li, Q.; Sarkar, M. Influence of the PVA Fibers and SiO2 NPs on the Structural Properties of Fly Ash Based Sustainable Geopolymer. Constr. Build. Mater. 2018, 164, 238–245. [Google Scholar] [CrossRef]
- Li, Z.; Chen, R.; Zhang, L. Utilization of Chitosan Biopolymer to Enhance Fly Ash-Based Geopolymer. J. Mater. Sci. 2013, 48, 7986–7993. [Google Scholar] [CrossRef]
- Chen, L.; Yuan, N.; Tan, Z.; Zhang, J.; Zhang, L.; Tang, W.; Chen, C.; Lin, D. Transparent Elastic Wound Dressing Gel Supporting Drug Release: Synergistic Effects of Poly(Vinyl Alcohol)/Chitosan Hybrid Matrix. Gels 2025, 11, 771. [Google Scholar] [CrossRef] [PubMed]
- Jang, T.; Yoon, S.; Choi, J.-H.; Kim, N.; Park, J.-A. Simultaneous Removal of Heavy Metals and Dyes on Sodium Alginate/Polyvinyl Alcohol/κ-Carrageenan Aerogel Beads. Gels 2025, 11, 211. [Google Scholar] [CrossRef]
- Lazar, M.M.; Ghiorghita, C.-A.; Rusu, D.; Dinu, M.V. Nanocomposite Cryogels Based on Chitosan for Efficient Removal of a Triphenylmethane Dye from Aqueous Systems. Gels 2025, 11, 729. [Google Scholar] [CrossRef]
- Dalvand, A.; Nabizadeh, R.; Reza Ganjali, M.; Khoobi, M.; Nazmara, S.; Hossein Mahvi, A. Modeling of Reactive Blue 19 Azo Dye Removal from Colored Textile Wastewater Using L-Arginine-Functionalized Fe3O4 Nanoparticles: Optimization, Reusability, Kinetic and Equilibrium Studies. J. Magn. Magn. Mater. 2016, 404, 179–189. [Google Scholar] [CrossRef]
- Lagergren, S. Zur Theorie Der Sogenannten Adsorption Gelöster Stoffe. K. Sven. Vetenskapsakademiens Handl. 1898, 24, 1–39. [Google Scholar]
- Ho, Y.S.; McKay, G. Pseudo-Second Order Model for Sorption Processes. Process Biochem. 1999, 34, 451–465. [Google Scholar] [CrossRef]
- Weber, W.J.; Morris, J.C. Removal of Biological Resistant Pollutions from Wastewater by Adsorption. In Advances in Water Pollution Research: Proceedings of the International Conference on Water Pollution Symposium; Pergamon Press: Oxford, UK, 1962. [Google Scholar]
- Aharoni, C.; Tompkins, F.C. Kinetics of Adsorption and Desorption and the Elovich Equation. In Advances in Catalysis; Eley, D.D., Pines, H., Weisz, P.B., Eds.; Academic Press: New York, NY, USA, 1970; Volume 21, pp. 1–49. ISBN 978-0-12-007821-9. [Google Scholar]
- Langmuir, I. The Adsorption of Gases on Plane Surfaces of Glass, Mica and Platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef]
- Freundlich, H. Adsorption in Solutions. J. Phys. Chem. 1906, 57, 384–410. [Google Scholar]
- Temkin, M.I. Kinetics of Ammonia Synthesis on Promoted Iron Catalysts. Acta Physiochimica URSS 1940, 12, 327–356. [Google Scholar]
- Dubinin, M.M.; Radushkevich, L.W. Equation of the Characteristic Curve of Activated Charcoal. Proc. Acad. Sci. USSR Phys. Chem. Sect. 1947, 55, 331–333. [Google Scholar]
- Grahovac, N.; Aleksić, M.; Pezo, L.; Đurović, A.; Stojanović, Z.; Jocković, J.; Cvejić, S. Box–Behnken-Assisted Optimization of High-Performance Liquid Chromatography Method for Enhanced Sugar Determination in Wild Sunflower Nectar. Separations 2025, 12, 244. [Google Scholar] [CrossRef]
- Tsitsirigka, T.; Kalompatsios, D.; Athanasiadis, V.; Bozinou, E.; Sfougaris, A.I.; Lalas, S.I. Valorization of the Bioactive Potential of Juniperus communis L. Berry Extracts Using a Box–Behnken Design and Characterization of Kernel Oil Compounds. Separations 2025, 12, 209. [Google Scholar] [CrossRef]
- Mecheri, R.; Zobeidi, A.; Atia, S.; Neghmouche Nacer, S.; Salih, A.A.M.; Benaissa, M.; Ghernaout, D.; Arni, S.A.; Ghareba, S.; Elboughdiri, N. Modeling and Optimizing the Crystal Violet Dye Adsorption on Kaolinite Mixed with Cellulose Waste Red Bean Peels: Insights into the Kinetic, Isothermal, Thermodynamic, and Mechanistic Study. Materials 2023, 16, 4082. [Google Scholar] [CrossRef]
- Leardi, R. Nature-Inspired Methods in Chemometrics: Genetic Algorithms and Artificial Neural Networks; Elsevier: Amsterdam, The Netherlands, 2003; ISBN 978-0-08-052262-3. [Google Scholar]
- Lek, S.; Delacoste, M.; Baran, P.; Dimopoulos, I.; Lauga, J.; Aulagnier, S. Application of Neural Networks to Modelling Nonlinear Relationships in Ecology. Ecol. Model. 1996, 90, 39–52. [Google Scholar] [CrossRef]
- Basheer, I.A.; Hajmeer, M. Artificial Neural Networks: Fundamentals, Computing, Design, and Application. J. Microbiol. Methods 2000, 43, 3–31. [Google Scholar] [CrossRef]
- Islam, M.H.; Law, D.; Patrisia, Y.; Gunasekara, C. Blended Brown Coal and Class F Fly Ash Based Geopolymer. Case Stud. Constr. Mater. 2025, 23, e05036. [Google Scholar] [CrossRef]
- Li, Y.; Wang, H. Alkali Cation Effects on Compressive Strength of Metakaolin–Low-Calcium Fly Ash-Based Geopolymers. Materials 2025, 18, 4080. [Google Scholar] [CrossRef]
- Zhang, G.; Li, K.; Shi, H.; Chen, C.; Yuan, C. Study on the Effects and Mechanisms of Fly Ash, Silica Fume, and Metakaolin on the Properties of Slag–Yellow River Sediment-Based Geopolymers. Materials 2025, 18, 1845. [Google Scholar] [CrossRef]
- Liu, Y.-L.; Wu, K.; Qian, L.-P.; Wang, Y.-S.; Guo, D. Effect of Polyethylene Glycol (PEG) Modification on Strength and Microstructure of Geopolymer Paste. Constr. Build. Mater. 2025, 489, 142378. [Google Scholar] [CrossRef]
- Mladenović Nikolić, N.; Kljajević, L.; Nenadović, S.S.; Potočnik, J.; Knežević, S.; Dolenec, S.; Trivunac, K. Adsorption Efficiency of Cadmium (II) by Different Alkali-Activated Materials. Gels 2024, 10, 317. [Google Scholar] [CrossRef]
- Kmiotek, A.; Figiela, B.; Łach, M.; Aruova, L.; Korniejenko, K. An Investigation of Key Mechanical and Physical Characteristics of Geopolymer Composites for Sustainable Road Infrastructure Applications. Buildings 2025, 15, 1262. [Google Scholar] [CrossRef]
- Nath, S.K.; Maitra, S.; Mukherjee, S.; Kumar, S. Microstructural and Morphological Evolution of Fly Ash Based Geopolymers. Constr. Build. Mater. 2016, 111, 758–765. [Google Scholar] [CrossRef]
- Van Riessen, A.; Jamieson, E.; Gildenhuys, H.; Skane, R.; Allery, J. Using XRD to Assess the Strength of Fly-Ash- and Metakaolin-Based Geopolymers. Materials 2025, 18, 2093. [Google Scholar] [CrossRef]
- Jankovic, A.; Cujic, M.; Stojkovic, M.; Djolic, M.; Zivojinovic, D.; Onjia, A.; Ristic, M.; Peric-Grujic, A. Impact of Leaching Procedure on Heavy Metals Removal from Coal Fly Ash. Hem. Ind. 2024, 78, 51–62. [Google Scholar] [CrossRef]
- Yavuz, E.; Kul Gul, N.I.; Kockal, N.U. Characterization of Class C and F Fly Ashes Based Geopolymers Incorporating Silica Fume. Ceram. Int. 2022, 48, 32213–32225. [Google Scholar] [CrossRef]
- Li, J.; Ma, Z.; Gao, J.; Guo, Y.; Cheng, F. Synthesis and Characterization of Geopolymer Prepared from Circulating Fluidized Bed-Derived Fly Ash. Ceram. Int. 2022, 48, 11820–11829. [Google Scholar] [CrossRef]
- Wang, T.; Fan, X.; Gao, C. Strength, Pore Characteristics, and Characterization of Fly Ash-Slag-Based Geopolymer Mortar Modified with Silica Fume. Structures 2024, 69, 107525. [Google Scholar] [CrossRef]
- Qiao, J.; Lv, Y.; Peng, H.; Han, W.; Pan, B.; Zhang, B. Performance and Characterization of Fly Ash-Metakaolin-Based Geopolymer Pastes. Mater. Today Commun. 2024, 41, 110293. [Google Scholar] [CrossRef]
- Bhavyasree, P.G.; Xavier, T.S. Adsorption Studies of Methylene Blue, Coomassie Brilliant Blue, and Congo Red Dyes onto CuO/C Nanocomposites Synthesized via Vitex Negundo Linn Leaf Extract. Curr. Res. Green. Sustain. Chem. 2021, 4, 100161. [Google Scholar] [CrossRef]
- Sokić, K.; Dikić, J.; Veljović, Đ.; Jelić, I.; Radovanović, D.; Štulović, M.; Jevtić, S. Preparation and Characterization of Hydroxyapatite-Modified Natural Zeolite: Application as Adsorbent for Ni2+ and Cr3+ Ion Removal from Aqueous Solutions. Processes 2025, 13, 818. [Google Scholar] [CrossRef]
- Prokić, D.; Vukčević, M.; Mitrović, A.; Maletić, M.; Kalijadis, A.; Janković-Častvan, I.; Đurkić, T. Adsorption of Estrone, 17β-Estradiol, and 17α-Ethinylestradiol from Water onto Modified Multi-Walled Carbon Nanotubes, Carbon Cryogel, and Carbonized Hydrothermal Carbon. Environ. Sci. Pollut. Res. 2022, 29, 4431–4445. [Google Scholar] [CrossRef]
- Andrew Ofudje, E.; Sodiya, E.F.; Olanrele, O.S.; Akinwunmi, F. Adsorption of Cd2+ onto Apatite Surface: Equilibrium, Kinetics and Thermodynamic Studies. Heliyon 2023, 9, e12971. [Google Scholar] [CrossRef]
- Ofudje, E.A.; Adeogun, I.A.; Idowu, M.A.; Kareem, S.O.; Ndukwe, N.A. Simultaneous Removals of Cadmium(II) Ions and Reactive Yellow 4 Dye from Aqueous Solution by Bone Meal-Derived Apatite: Kinetics, Equilibrium and Thermodynamic Evaluations. J. Anal. Sci. Technol. 2020, 11, 7. [Google Scholar] [CrossRef]
- Vukcevic, M.; Pejic, B.; Pajic-Lijakovic, I.; Kalijadis, A.; Kostic, M.; Lausevic, Z.; Lausevic, M. Influence of the Precursor Chemical Composition on Heavy Metal Adsorption Properties of Hemp (Cannabis Sativa) Fibers Based Biocarbon. J. Serb. Chem. Soc. 2017, 82, 1417–1431. [Google Scholar] [CrossRef]
- Işıtan, A. Sustainable Adsorption of Amoxicillin and Sulfamethoxazole onto Activated Carbon Derived from Food and Agricultural Waste: Isotherm Modeling and Characterization. Processes 2025, 13, 2528. [Google Scholar] [CrossRef]
- Siddiqui, S.I.; Alsebaii, N.M.; Al-Ghamdi, A.A.; Aldahiri, R.H.; Alzahrani, E.A.; Hafeez, S.; Oh, S.; Chaudhry, S.A. Fe3O4/BC for Methylene Blue Removal from Water: Optimization, Thermodynamic, Isotherm, and Kinetic Studies. Materials 2025, 18, 2049. [Google Scholar] [CrossRef]
- Mphuthi, B.R.; Thabede, P.M.; Modise, J.S.; Xaba, T.; Shooto, N.D. Adsorption of Cadmium and Methylene Blue Using Highly Porous Carbon from Hemp Seeds. Appl. Sci. 2023, 13, 9659. [Google Scholar] [CrossRef]
- Ali, R.; Elsagan, Z.; AbdElhafez, S. Lignin from Agro-Industrial Waste to an Efficient Magnetic Adsorbent for Hazardous Crystal Violet Removal. Molecules 2022, 27, 1831. [Google Scholar] [CrossRef]
- Gul, S.; Gul, S.; Gul, H.; Khitab, F.; Khattak, R.; Khan, M.; Ullah, R.; Ullah, R.; Wasil, Z.; Krauklis, A.; et al. Dried Leaves Powder of Adiantum Capillus-Veneris as an Efficient Biosorbent for Hazardous Crystal Violet Dye from Water Resources. Separations 2023, 10, 165. [Google Scholar] [CrossRef]
- Huang, Y.-T.; Shih, M.-C. Kinetic, Isotherm, and Thermodynamic Modeling of Methylene Blue Adsorption Using Natural Rice Husk: A Sustainable Approach. Separations 2025, 12, 189. [Google Scholar] [CrossRef]
- Amin, M.T.; Alazba, A.A.; Shafiq, M. Successful Application of Eucalyptus Camdulensis Biochar in the Batch Adsorption of Crystal Violet and Methylene Blue Dyes from Aqueous Solution. Sustainability 2021, 13, 3600. [Google Scholar] [CrossRef]
- Supelano, G.I.; Gómez Cuaspud, J.A.; Moreno-Aldana, L.C.; Ortiz, C.; Trujillo, C.A.; Palacio, C.A.; Parra Vargas, C.A.; Mejía Gómez, J.A. Synthesis of Magnetic Zeolites from Recycled Fly Ash for Adsorption of Methylene Blue. Fuel 2020, 263, 116800. [Google Scholar] [CrossRef]
- Kemdjien, L.; Tchakounte, A.; Lenou, I.; Tome, S.; Kede, C. Optimization of Synthesis of a Geopolymer Based on Laterite/Beer Bottles Composites for Adsorption of Methylene Blue in Aqueous Solution. Desalination Water Treat. 2025, 322, 101189. [Google Scholar] [CrossRef]
- Kurniasih, M.; Aprilita, N.H.; Roto, R.; Mudasir, M. Modification of Coal Fly Ash for High Capacity Adsorption of Methylene Blue. Case Stud. Chem. Environ. Eng. 2025, 11, 101101. [Google Scholar] [CrossRef]
- Singh, K.P.; Basant, A.; Malik, A.; Jain, G. Artificial Neural Network Modeling of the River Water Quality—A Case Study. Ecol. Model. 2009, 220, 888–895. [Google Scholar] [CrossRef]



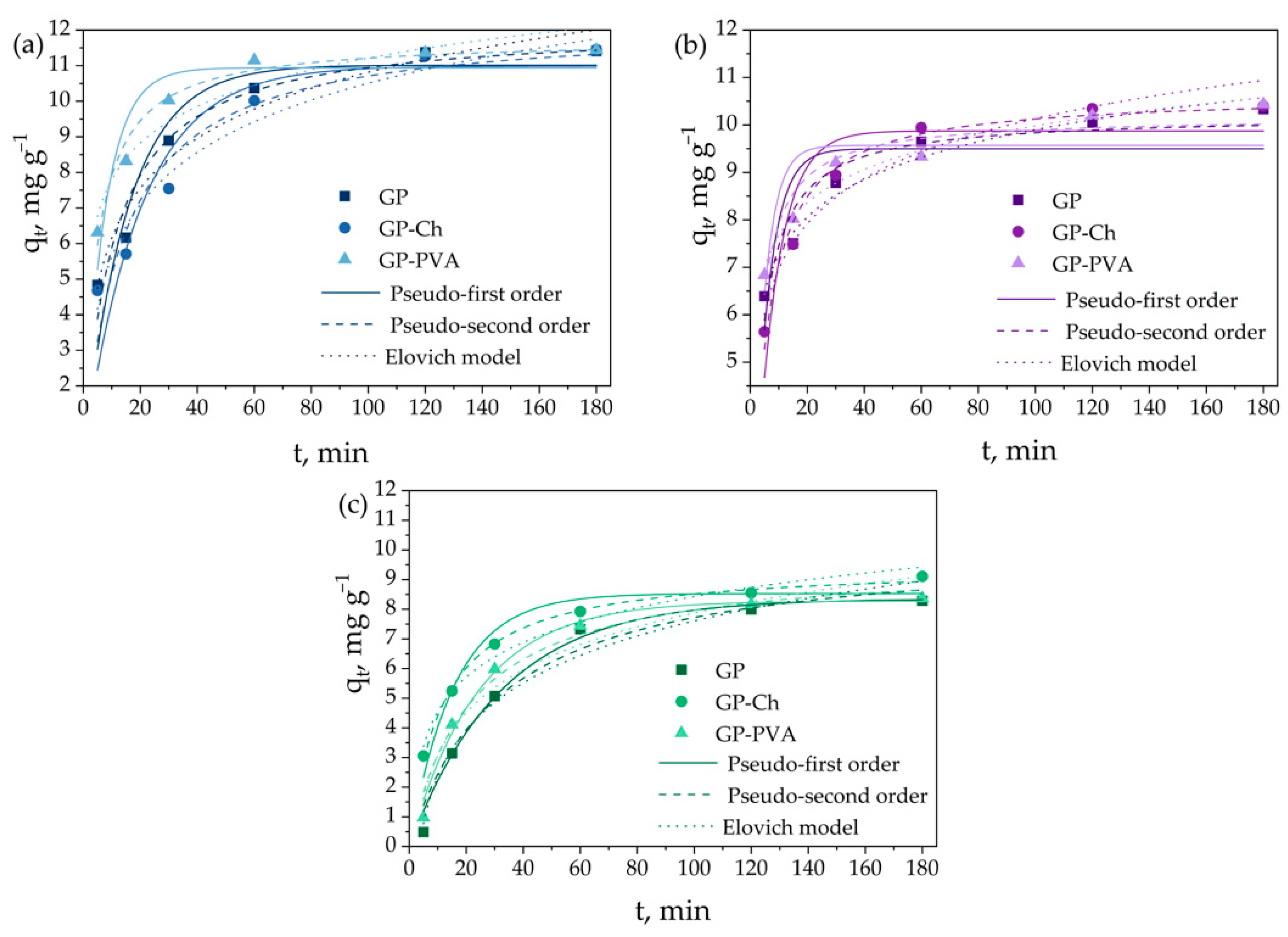







| Geopolymer | Dye | Pseudo-First Order | Pseudo-Second Order | Elovich Kinetic | qe,exp, mg g−1 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| qe,cal, mg g−1 | k1, min−1 | R2 | qe,cal, mg g−1 | k2 × 105, g mg−1 min−1 | R2 | α mg g−1 min−1 | β g mg−1 | R2 | |||
| GP | MB | 11.01 | 0.0644 | 0.85651 | 12.11 | 0.0078 | 0.94014 | 4.2106 | 0.493 | 0.94620 | 11.41 |
| CV | 9.498 | 0.1823 | 0.59997 | 10.19 | 0.0268 | 0.90054 | 64.242 | 0.871 | 0.96996 | 10.34 | |
| BG | 9.569 | 0.2232 | 0.98411 | 10.18 | 0.0344 | 0.96290 | 0.639 | 0.438 | 0.96445 | 8.289 | |
| GP-Ch | MB | 10.99 | 0.0503 | 0.80493 | 12.18 | 0.0060 | 0.90256 | 3.020 | 0.472 | 0.95138 | 11.45 |
| CV | 9.875 | 0.1284 | 0.80170 | 10.64 | 0.0185 | 0.97144 | 21.581 | 0.726 | 0.93593 | 10.39 | |
| BG | 8.529 | 0.0639 | 0.94085 | 9.503 | 0.0091 | 0.99536 | 2.488 | 0.593 | 0.97401 | 9.107 | |
| GP-PVA | MB | 10.94 | 0.1317 | 0.80942 | 11.76 | 0.0174 | 0.96945 | 28.157 | 0.673 | 0.91285 | 11.44 |
| CV | 9.569 | 0.2232 | 0.60297 | 10.18 | 0.0344 | 0.89237 | 208.63 | 0.996 | 0.96691 | 10.44 | |
| BG | 8.279 | 0.0414 | 0.98625 | 9.684 | 0.0048 | 0.96832 | 0.903 | 0.478 | 0.94559 | 8.370 | |
| Geopolymer | Dye | Langmuir Isotherm | Freundlich Isotherm | Temkin Isotherm | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| q0, mg g−1 | b | R2 | Kf, mg g−1 (mg dm−3) −1/n | 1/n | R2 | A, dm3 g−1 | B, J mol−1 | R2 | ||
| GP | MB | 27.99 | 0.5285 | 0.99004 | 11.58 | 0.170 | 0.94674 | 25.56 | 3.176 | 0.98058 |
| CV | 29.79 | 0.0653 | 0.96539 | 8.679 | 0.289 | 0.80086 | 1.372 | 6.536 | 0.89201 | |
| BG | 26.16 | 0.0104 | 0.98209 | 3.283 | 0.456 | 0.8996 | 0.261 | 7.954 | 0.96490 | |
| GP-Ch | MB | 29.03 | 0.5444 | 0.99374 | 11.95 | 0.169 | 0.95116 | 29.06 | 3.196 | 0.98408 |
| CV | 28.57 | 0.0847 | 0.94911 | 10.18 | 0.247 | 0.74965 | 2.517 | 5.679 | 0.84108 | |
| BG | 25.16 | 0.0218 | 0.96786 | 6.611 | 0.311 | 0.80327 | 0.837 | 6.179 | 0.88640 | |
| GP-PVA | MB | 24.19 | 0.8079 | 0.99237 | 12.89 | 0.139 | 0.90938 | 101.5 | 2.620 | 0.95155 |
| CV | 36.07 | 0.0646 | 0.92654 | 8.762 | 0.334 | 0.80366 | 1.053 | 8.308 | 0.88869 | |
| BG | 27.35 | 0.0122 | 0.98843 | 3.738 | 0.444 | 0.89619 | 0.299 | 8.178 | 0.96726 | |
| Geopolymer | Dye | R2 | qm mg g−1 | Β mol2 J−2 | E kJ mol−1 |
|---|---|---|---|---|---|
| GP | MB | 0.72284 | 20.44 | 0.44 | 1.069 |
| CV | 0.76026 | 24.09 | 3.07 | 0.403 | |
| BG | 0.92298 | 21.56 | 23.97 | 0.144 | |
| GP-Ch | MB | 0.65172 | 20.57 | 0.32 | 1.243 |
| CV | 0.80453 | 24.47 | 2.26 | 0.470 | |
| BG | 0.92382 | 22.77 | 8.35 | 0.245 | |
| GP-PVA | MB | 0.77171 | 20.42 | 0.26 | 1.380 |
| CV | 0.60338 | 25.45 | 2.22 | 0.474 | |
| BG | 0.91663 | 22.36 | 18.51 | 0.164 |
| Geopolymer | Dye | ΔH°, kJ mol−1 | ΔS°, J mol−1 K−1 | ΔG°, kJ mol−1 | ||
|---|---|---|---|---|---|---|
| 303.15 K | 308.15 K | 318.15 K | ||||
| GP | MB | 11.22 | 53.1 | −4.90 | −5.16 | −5.70 |
| CV | 11.20 | 49.6 | −3.84 | −4.08 | −4.58 | |
| BG | 8.268 | 29.1 | −0.55 | −0.70 | −0.99 | |
| GP-Ch | MB | 46.41 | 167.3 | −4.32 | −5.15 | −6.83 |
| CV | 42.28 | 149.7 | −3.12 | −3.86 | −5.36 | |
| BG | 31.26 | 105.3 | −0.67 | −1.20 | −2.25 | |
| GP-PVA | MB | −34.95 | −97.4 | −5.42 | −4.93 | −3.96 |
| CV | 9.612 | 42.8 | −3.38 | −3.59 | −4.02 | |
| BG | −6.987 | 20.3 | −0.82 | −0.71 | −0.51 | |
| Adsorbent | Dye | C0, mg dm−3 | Langmuir maximum Adsorption Capacity, qmax (mg g−1) | Removal Efficiency | Ref. |
|---|---|---|---|---|---|
| Fe3O4/Black cumin (Nigella sativa) carbon framework | MB | 10–60 | 40.0–48.5 | 99% | [92] |
| Pristine hemp seeds Carbonized hemp seeds | MB | 20–100 | 19.43–52.61 49.12–53.98 * | / | [93] |
| Lignin copper ferrite (from agro-industrial waste corn stover lignin) | CV | 5–100 | 30.5 34.125 * | 97% | [94] |
| Biochar from exhausted olive pomace | MB | 10–200 | 6.414 | / | [2] |
| Dried leaves powder of Adiantum capillus-veneris | CV | 10–100 | 18.51 9.12 * | 91.26% | [95] |
| Natural rice husk | MB | 10–100 | 5.72 4.30 * | / | [96] |
| Eucalyptus camaldulensis biochar | MB CV | 20–100 | 114.6 123.3 * 54.7 56.0 * | / | [97] |
| Magnetic zeolites synthesized from coal fly ash | MB | 10–100 | 16.53 27.05 | / | [98] |
| Geopolymer based on laterite and recycled beer bottle glass | MB | 10–100 | 10.54 12.63 | / | [99] |
| Modified Bagasse Fly Ash | MB | 50–100 | 5.19 15.5 * | 67.2% | [18] |
| Polyethyleneimine functionalized waste tissue paper/waste PET composite | CV | 10–50 | 4.44 3.94 * | 92% | [19] |
| Modified coal fly ash | MB | 50–300 | 95.81–151.33 | 74.34–94.62% | [100] |
| Geopolymer Based on Fly Ash | MB | 20–75 | 24.19–29.03 11.36–11.45 * | 95.4–95.9% | This study |
| Geopolymer Based on Fly Ash | CV | 20–75 | 28.57–36.07 10.36–10.44 * | 86.0–87.3% | This study |
| Geopolymer Based on Fly Ash | BG | 20–75 | 25.16–27.35 8.29–9.11 * | 69.1–77.3% | This study |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Trajković, D.V.; Maletić, M.M.; Vukčević, M.M.; Veljović, Đ.N.; Perić Grujić, A.A.; Živojinović, D.Z. Geopolymers Based on Fly Ash for Organic Dye Removal from Water. Separations 2025, 12, 299. https://doi.org/10.3390/separations12110299
Trajković DV, Maletić MM, Vukčević MM, Veljović ĐN, Perić Grujić AA, Živojinović DZ. Geopolymers Based on Fly Ash for Organic Dye Removal from Water. Separations. 2025; 12(11):299. https://doi.org/10.3390/separations12110299
Chicago/Turabian StyleTrajković, Dušan V., Marina M. Maletić, Marija M. Vukčević, Đorđe N. Veljović, Aleksandra A. Perić Grujić, and Dragana Z. Živojinović. 2025. "Geopolymers Based on Fly Ash for Organic Dye Removal from Water" Separations 12, no. 11: 299. https://doi.org/10.3390/separations12110299
APA StyleTrajković, D. V., Maletić, M. M., Vukčević, M. M., Veljović, Đ. N., Perić Grujić, A. A., & Živojinović, D. Z. (2025). Geopolymers Based on Fly Ash for Organic Dye Removal from Water. Separations, 12(11), 299. https://doi.org/10.3390/separations12110299








