Abstract
Biocatalytic oxidation of a fatty acid preparation by enzymes released from a plant extract provided (2E,4E)-deca-2,4-dienal (2,4-DDAL), a valuable natural aroma. The post-reaction mixture contained several other compounds, from which 2,4-DDAL must be recovered. These included the main by-product trans-2-hexenal (2-HAL) and the unreacted substrate composed mainly of linoleic, palmitic, and oleic acids. The adsorption capacity and selectivity of 17 hydrophobic adsorbents were examined in batch equilibrium experiments. The highest binding capacity for 2,4-DDAL was around 30 mg/g. Due to a large excess of fatty acids in the mixture, the total adsorbed amounts of other components analyzed reached the values up to 150 mg/g. In addition, 2-HAL was always adsorbed more strongly than 2,4-DDAL, whereas fatty acids were mostly not adsorbed as effectively. The best-performing adsorbent, AmberLite FPX66, had a selectivity ratio of 6 for 2,4-DDAL with respect to the key impurity and linoleic acid. Ethanol, isopropanol, and acetone were examined as potential desorbents using batch adsorption experiments on AmberLite FPX66 from 2,4-DDAL solutions in organic solvents. Ethanol was found to be the best choice. Based on the batch equilibrium data, a column elution experiment was performed to purify 2,4-DDAL from the biocatalytic reaction mixture using elution with 96% ethanol.
1. Introduction
Research on the development of new, eco-friendly methods for utilizing natural plant-based substrates to produce bioproducts with high added value has gained significant attention in recent years. These components should be isolated from natural sources and applied in biocatalytic processes. Plant enzymes have an important role in the evolution of green chemistry due to their ability to produce natural regiospecific and stereospecific substances. Such commercially interesting natural substances include doubly or triply unsaturated aldehydes.
Moreover, (2E,4E)-deca-2,4-dienal 2,4-DDAL is an aromatic substance naturally present in butter, cooked beef, fish, potato chips, roasted peanuts, buckwheat, and wheat breadcrumbs [1]. In an isolated state, it smells of a deep-fat flavor, characteristic of chicken and meat aroma [2]. At lower concentrations, it has the odor of citrus, orange, or grapefruit. It is frequently used in the food industry as a flavor [3] but needs to be used in an appropriate amount due to its negative association with human health. Wu et al. indicated 2,4-DDAL as a strong carcinogenic and cytotoxic mutation biomarker [4]. Several authors identified 2,4-DDAL as a toxic aldehyde responsible for stomach or lung carcinoma [5,6,7]. In addition, 2,4-DDAL is also a by-product of the peroxidation of polyunsaturated lipids during storage [8] or heated oils during cooking [9].
It was shown that 2,4-DDAL was efficiently produced by the catalytic action of combined plant lipoxygenase and hydroperoxide lyase or lipoxygenase alone on arachidonic acid [10] or linoleic acid [11] with the supply of air oxygen [12]. These conversions of polyunsaturated fatty acids can become a basis for the production of 2,4-DDAL as a natural aroma from mixtures of fatty acids obtained by the hydrolysis of different plant oils. This makes the enzymatic process more complicated when several substances may participate in the reaction. Therefore, they are also present in the post-production mixture. Since 2,4-DDAL is used in the food industry and, therefore, requires high purity, there is a need to develop an effective method for 2,4-DDAL isolation. It can be found together with other tastes and odors present in drinking water, rendering the drinking water fishy or oily [13]. As a result, the purification of drinking water is another motivation for developing this separation process.
Sun et al. studied methods to improve the extraction efficiency and evaluate optimal conditions of derivatization for 2,4-DDAL in oils to quantitatively determine 2,4-DDAL levels in edible oils [2]. Satchwill et al. [14] demonstrated the use of aluminum sulphate coagulation combined with conventional gravity sedimentation or dissolved air flotation as an effective method to remove 2,4-DDAL and other odors (2,4-heptadienal, 2,4,7-octatriene, and 2,4,7-decatrienal) from natural waters.
Adsorption has been proven to be an effective method for removing 2,4-DDAL and 2,4-heptadienal from drinking water. Li et al. investigated powdered-activated carbon adsorption for this purpose [15]. Obendorf et al. [16] studied the adsorption of 2,4-DDAL and other aromas in cotton fabric in different aqueous environments. A novel hyper-cross-linked sorbent material composed of silica gel, activated carbon, octadecylsilane, and other high-purity materials was used for the adsorption of aroma active compounds: 2,4-DDAL and other aldehydes, as well as alcohols and acids in walnut oil [17]. Adsorption on yeast β-glucan for off-odor compounds in silver carp mince was analyzed by Zhang et al. [18].
The objective of this study was to examine the suitability of industrial hydrophobic adsorbents for the separation of 2,4-DDAL prepared from a fatty acid substrate by the catalytic action of lipoxygenase/hydroperoxide lyase released from a plant extract. A biocatalytic reaction mixture, which contained fatty acids and 2-HAL as main impurities, was used for batch adsorption experiments. The adsorbed amounts of 2,4-DDAL and the key impurities and selectivities of 2,4-DDAL relative to these components were calculated for 17 hydrophobic adsorbents. Model solutions of each component using different solvents were prepared, and batch adsorption experiments with these model mixtures were carried out to study desorption conditions for a selected adsorbent. This adsorbent, which has the most promising results, was selected for the separation of the biocatalytic mixture in a chromatographic column.
2. Materials and Methods
2.1. Chemicals
Palmitic acid, (2E,4E)-deca-2,4-dienal, oleic acid, linoleic acid, and trans-2-hexenal were purchased from Sigma–Aldrich (St. Louis, MO, USA), while ethanol, isopropanol, and acetone of analytical grade were purchased from Centralchem (Bratislava, Slovakia). The fatty acid preparation Nouracid HE 30 was from Oleon (Ertvelde, Belgium). The adsorbents tested were various types of AmberLite resins from Dow Chemical Company (Midland, MI, USA) and Macronet or PuroSorb resins from PuroLite (Philadelphia, PA, USA). All adsorbents except AmberLite XAD 7HP and AmberLite FPX62 are nonfunctionalized poly(styrene-divinylbenzene) resins. Their list with essential particle textural properties is provided in Table 1. The adsorbents were conditioned for 24 h in 96% ethanol and subsequently washed several times with redistilled water.

Table 1.
Adsorbents used and their properties provided by the producers.
2.2. Biocatalytic Reaction Mixture Preparation
A 2,4-DDAL reaction mixture was prepared from the fatty acid substrate by catalyzed process on fatty acids at the presence of oxygen. Representative average composition of the reaction mixture is provided in Table 2.

Table 2.
Representative composition of the reaction mixture.
2.3. Batch Adsorption Experiments
Approximately 0.1 g of wet adsorbent was weighed in a glass tube, and 15 mL of adsorption solution (reaction mixture) was added. The glass tube was closed and sealed to prevent stripping losses. The suspension was stirred at the ambient temperature for 24 h using a reciprocal shaker to achieve adsorption equilibrium. The equilibrium concentrations of all components, were then measured using gas chromatography analysis. Each adsorption experiment was performed in duplicate. Batch experiments for single-component adsorption of 2,4-DDAL in ethanol (EtOH), isopropanol (IPA), and acetone solutions were performed using 25 mg of adsorbent particles and 1 mL of liquid phase. The same conditions were used in batch experiments of single-component adsorption from ethanol/water solutions of 2-HAL and fatty acids.
The adsorbed amount of each substance qi was then determined from the material balance as follows:
where VL is the volume of the liquid phase and mads the wet adsorbent weight. The selectivity S was defined by Equation (2)
where is the adsorbed amount of 2,4-DDAL at equilibrium concentration
2.4. Column Experiments
Column elution separation experiments were performed using ÄKTA FPLC system (Cytiva, Marlborough, MA, USA). The column Tricorn 10/100 (bed volume 7.85 mL) was filled with AmberLite FPX66 adsorbent. Experiments were carried out at volumetric flow of rate 1 mL/min during both the adsorption and desorption phase. The feed was formed by 1 mL of the biocatalytic reaction mixture containing 0.40 g/L of 2,4-DDAL, 0.54 g/L of 2-HAL, 0.51 g/L of palmitic acid, 2.35 g/L of oleic acid, and 2.20 g/L of linoleic acid. A feed pulse was injected into a water-equilibrated adsorbent bed. After this injection, the mobile phase was switched to 96% ethanol that was used as eluent.
2.5. Analytical Methods
Fractions eluted from the separation column were analyzed by Agilent 7890A gas chromatograph with FID detector. Polar capillary column DB-WAX (Agilent Technologies, Santa Clara, CA, USA) with dimensions 30 m × 0.25 mm i.d. and 0.15 μm film thickness was used for the analysis. Hydrogen was used as a carrier gas at a flow rate of 2 mL/min. One microliter of sample was injected using a split mode in the ratio of 10:1; the inlet temperature was 250 °C. The initial column temperature was 95 °C. The temperature was held for 6 min, then increased at a rate of 10 °C/min to 120 °C, subsequently ramped again at 20 °C/min to 220 °C, and finally held for 31.5 min. Standard of 2,4-DDAL (Sigma-Aldrich, St. Louis, MO, USA) was used to obtain the retention time, and standard of ethyl decadienoate (Sigma-Aldrich, St. Louis, MO, USA) was used as an internal standard for quantification of 2,4-DDAL.
2.6. Evaluation of Separation Efficiency
Yield is an essential parameter for evaluation of process efficiency. The yield of 2,4-DDAL was calculated from Equation (3)
where is amount of purified 2,4-DDAL and is total amount of 2,4-DDAL. The amount of purified 2,4-DDAL was calculated from the experimental data using numerical integration. The total amount of 2,4-DDAL was calculated from the total volume of the feed and the concentration of 2,4-DDAL.
3. Results
3.1. Adsorbent Screening
Seventeen hydrophobic adsorbents were examined as potential materials for the separation of 2,4-DDAL present in the biocatalytic reaction mixture. The composition of the reaction mixture is variable and depends on the type and seasonal conditions of the plant material used as a source of enzymes. In general, matrices that do not contain starch and other macromolecules are more suitable for this biocatalytic process. The reaction mixture, in addition to the 2,4-DDAL, contained significant amounts of three organic fatty acid substrates, palmitic acid, linoleic acid, and oleic acid, as well as by-product trans-2-hexenal (Table 2). Therefore, our research focused not only on the adsorption capacity of 2,4-DDAL but also selectivity of each adsorbent considering other components represented in the reaction mixture.
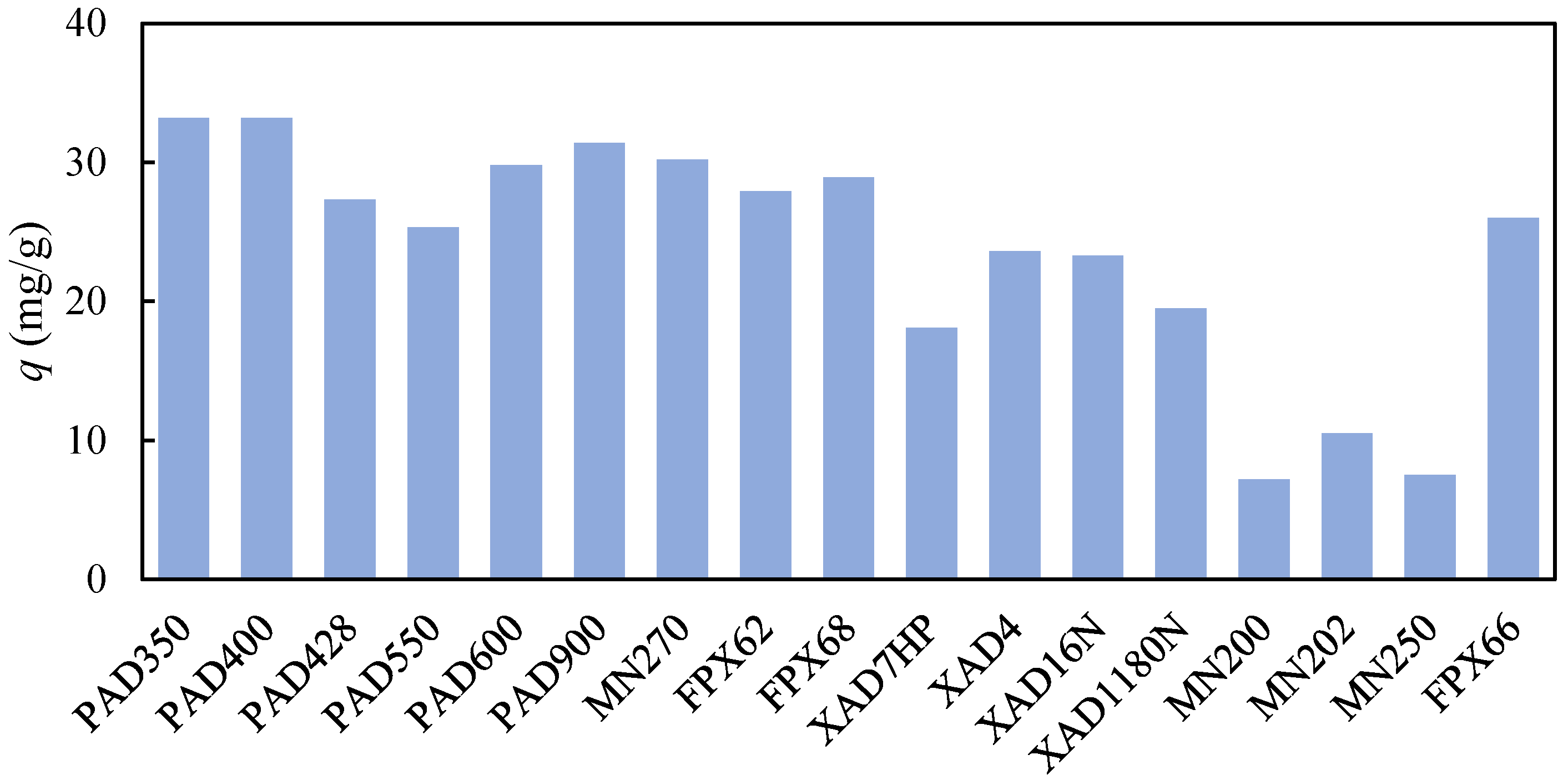
For this purpose, batch adsorption experiments with the reaction mixture were performed for each examined adsorbent. The adsorbed amount of 2,4-DDAL was calculated from Equation (1), and its value for each adsorbent is provided in Figure 1. The highest adsorbed amount of 2,4-DDAL reached in our screening was approximately 33 mg/g, when the q-values of 2,4-DDAL ranged from 8 mg/g to 33 mg/g. Li et al. [15] reached the values of about 240–260 mg/g for 2,4-DDAL adsorption on activated carbon from pure 2,4-DDAL solution. On the other hand, Zhang et al. [18] observed the competitive adsorption of 2,4-DDAL and other odor compounds (hexanal, nonanal,1-octen-3-ol) on β-glucan from silver carp mince. The 2,4-DDAL adsorption capacities were only 1–2 mg/g.

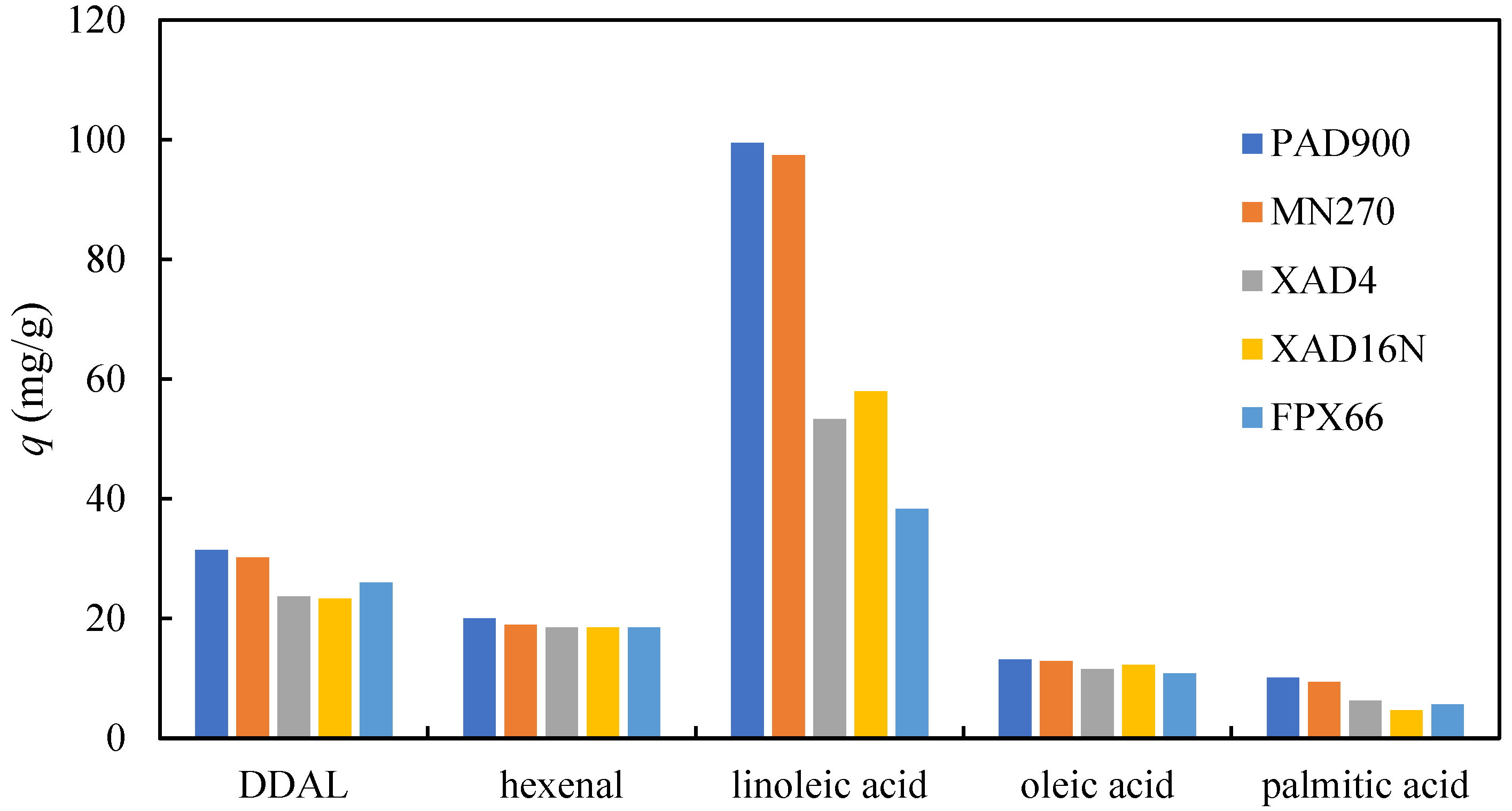
For five selected adsorbents with a good adsorption capacity for 2,4-DDAL, the adsorption capacities of other components present in the reaction mixture were calculated and are provided in Figure 2. The adsorption capacities of 2-HAL, palmitic, and oleic acid are lower compared to 2,4-DDAL, but the concentration of these components in the reaction mixture was also lower. In the case of palmitic and oleic acid, their adsorption capacity was approximately proportional to their content in the reaction mixture. However, the adsorption capacity for hexanal was relatively higher than that for 2,4-DDAL, considering that the hexanal concentration in the reaction mixture was only about 30% of the 2,4-DDAL concentration. The reaction mixture contained, on average, approximately 4-times higher amounts of linoleic acid compared to 2,4-DDAL. The adsorbed amounts of linoleic acid were from roughly 40 mg/g to 100 mg/g, which are the values about 2–3 times higher than the values reached for 2,4-DDAL (Figure 2).

Figure 2.
Adsorbed amounts of key components on selected adsorbents determined in batch experiments using the biocatalytic reaction mixture with the composition specified in Table 2.
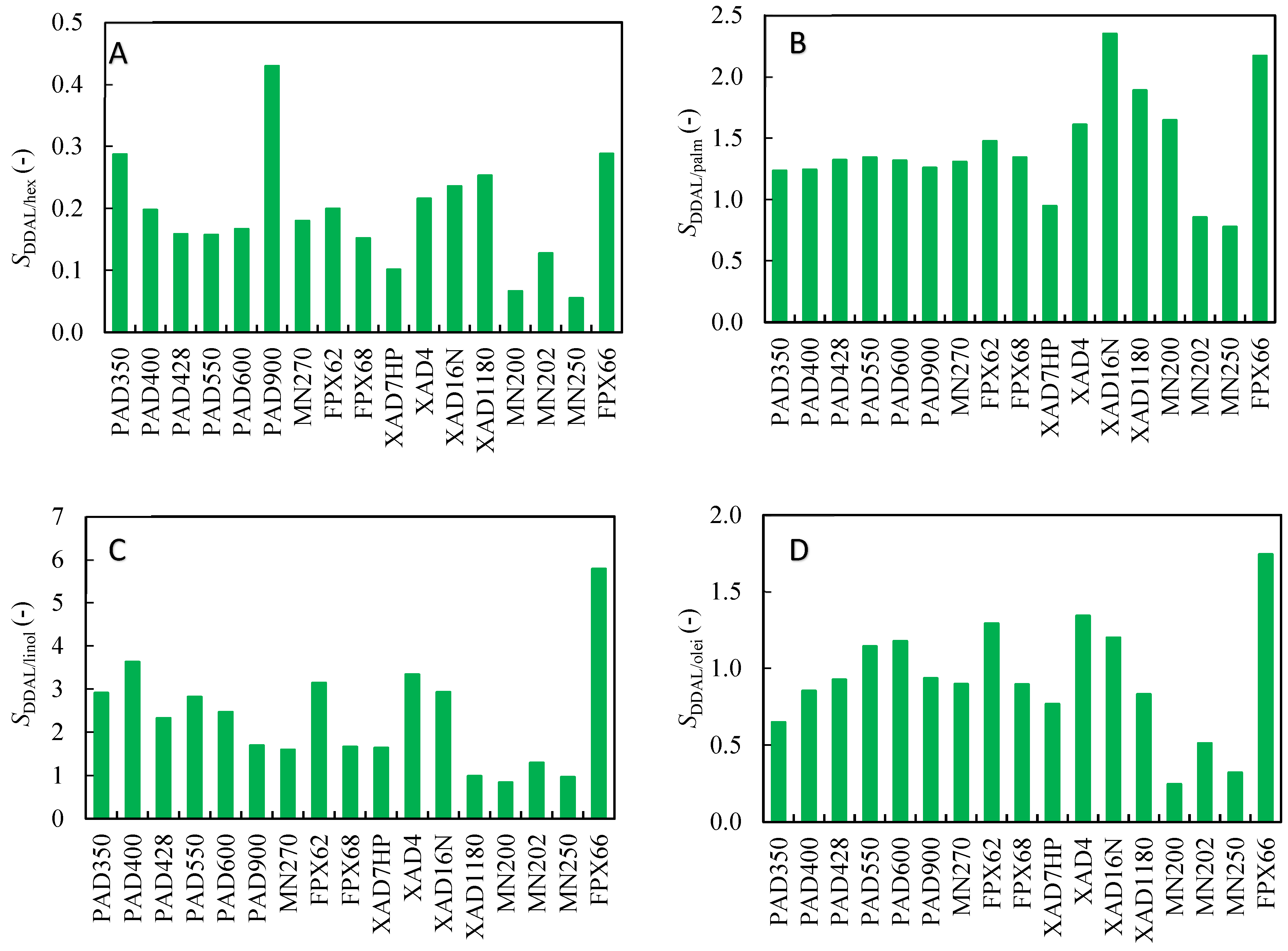
As mentioned above, selectivity is an essential parameter for evaluating the efficiency of the adsorption process. The selectivity of competitive binding of 2,4-DDAL and other substances presented in the mixture was calculated from Equation (2). The results are presented in Figure 3. Almost all adsorbents in the screening had selectivity values of 2,4-DDAL related to linoleic acid higher than 1. Moreover, the FPX66 adsorbent reached the selectivity value as high as 6. The selectivity values related to oleic acid and palmitic acid were, on average, close to 1. The selectivity values related to 2-HAL were for all adsorbents less than 1 when the highest value reached was about 0.4 for the adsorbent PuroSorb PAD900. It can be assumed from the results presented in Figure 3 that 2,4-DDAL binds preferentially compared to linoleic acid, approximately equally compared to oleic and palmitic acid, and worse compared to 2-HAL.

Figure 3.
Selectivities of adsorbents determined in batch experiments using the biocatalytic reaction mixture. The selectivity 2,4-DDAL related to (A) 2-HAL; (B) Palmitic acid; (C) Linoleic acid; (D) Oleic acid.
Almost all adsorbents applied for batch adsorption experiments are hydrophobic nonfunctionalized poly(styrene-divinylbenzene) resins. As shown in Table 1, they differ in texture properties, such as specific surface area or pore volume. Manufacturers provide specific surface area values in a broad range of 250 m2/g to 1100 m2/g, with the pore volume varying from 0.2 mL/g to 1.9 mL/g. In our previous work, a clear correlation between the specific pore surface area and adsorption capacity of acetophenone and 1-phenylethanol was observed for the identical group of 17 hydrophobic adsorbents [19]. A positive impact of high pore surface area on the selectivity of acetophenone binding was also observed, albeit this relationship did not have a monotonous trend.
No such correlation for the adsorption capacity was achieved in this study primarily due to the significantly lower concentrations of reaction mixture components. It means that the degree of surface coverage of the adsorbents was well below the saturation limit. It can be assumed that the correlation for the group of adsorbents of the same chemical structure will exist between the specific pore surface area and the saturation capacity achieved at sufficiently high concentrations of adsorbing compounds. At lower concentrations, pore structure and size will also play a role.
This hypothesis can be supported by the observation of some correlation for a series of adsorbent types. Figure 1 shows that the highest adsorption capacities of 2,4-DDAL were achieved for the Purosorb PAD and AmberLite FPX series. They were somewhat lower for the Amberlite XAD series and several times lower for the Macronet MN series (except for MN 270). A different performance of the Macronet MN series is clearly visible at the selectivity values in Figure 3. These adsorbents bound much more selectively 2-HAL and oleic acid compared to other adsorbent series and were not selective toward the binding of linoleic acid.
Following the results obtained, Amberlite FPX66 was chosen for further experiments in this work. The decisive factor was the highest selectivity of 2,4-DDAL with respect to the main impurity, linoleic acid. The selectivity value of roughly 6 implies that about 80% of linoleic acid can be theoretically removed in the breakthrough of the adsorption phase [19]. However, only a limited separation effect can be achieved during the adsorption phase due to the low selectivity values for other mixture components. The separation of all other components of the reaction mixture can be achieved via selective desorption.
3.2. Desorbent Selection
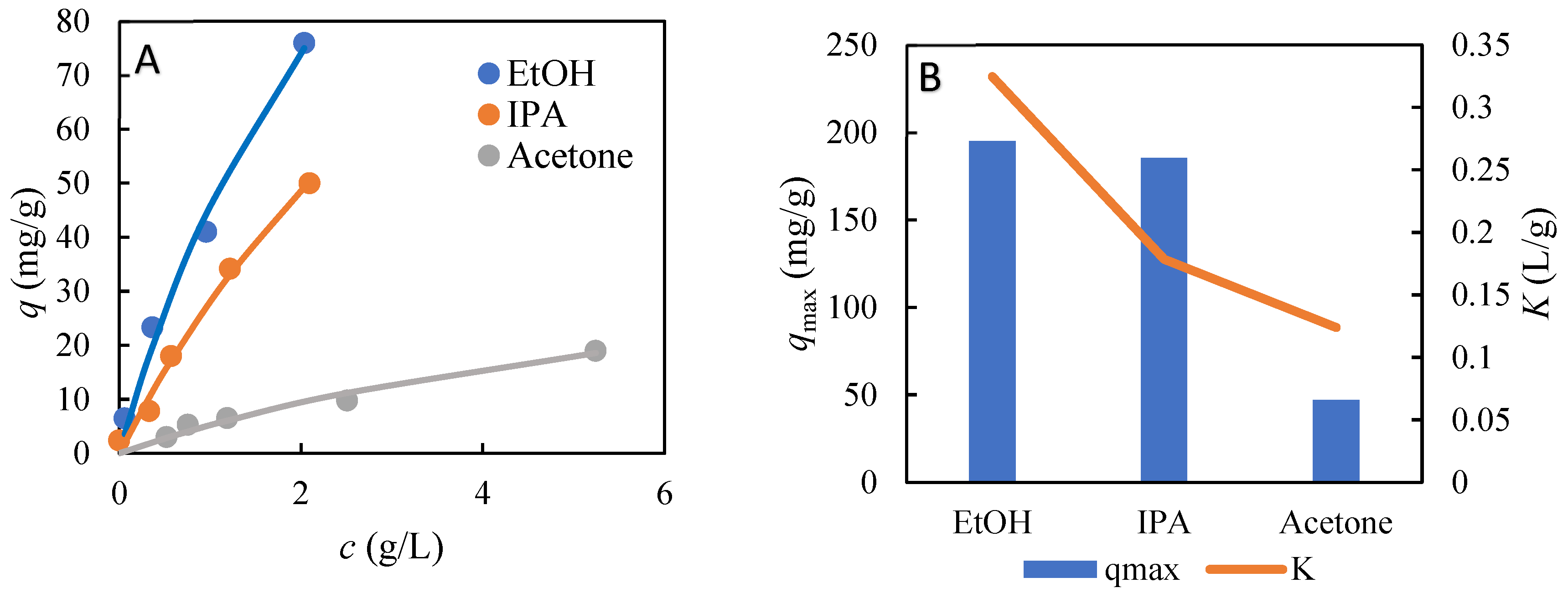
As mentioned above, the optimization of the desorption conditions could lead to selective desorption of each substance bound to the stationary phase. The selection of a suitable desorption agent is, therefore, very important. For this purpose, batch adsorption experiments from the single-component solutions of different solvents were performed using AmberLite FPX66 as an adsorbent. For 2,4-DDAL, its solutions in ethanol (96% aqueous solution), isopropanol, and acetone were examined. The equilibrium data obtained (Figure 4A) were described by a single-component Langmuir isotherm (Figure 4B). Acetone was found to be the strongest desorbent, with the adsorption capacities of 2,4-DDAL in the tested concentration range less than 10 mg/g (Figure 4A). This observation can also be supported by the lowest affinity constant value of 2,4-DDAL, K, for the adsorption from acetone solutions (Figure 4B).

Figure 4.
Single-component adsorption isotherms of 2,4-DDAL from organic solvent solutions for AmberLite FPX66. (A) Equilibrium data fitted with the Langmuir isotherm and (B) Langmuir isotherm parameters.
Acetone, as the strongest desorption agent, appeared not to be very suitable for selective desorption since it could cause an elution of all components at once. The concentration of 2,4-DDAL in the reaction mixture was approximately 0.3 g/L. Figure 4A shows that, for this concentration, the adsorption capacity reached in ethanol was the highest, specifically 20 mg/g. It was much lower (7 mg/g) for isopropanol and approximately only 2 mg/g for acetone. On the other hand, the adsorption capacity from water solutions of about 100 mg/g can be inferred from the total adsorbed amount of all components (Figure 1). The difference between 100 mg/g in water solutions and 20 mg/g for ethanol solutions creates a good basis to achieve selective desorption. Therefore, ethanol was determined to be the most suitable desorption agent. For further optimization, batch adsorption experiments were designed for all components contained in the biocatalytic reaction mixture dissolved in 96% EtOH.
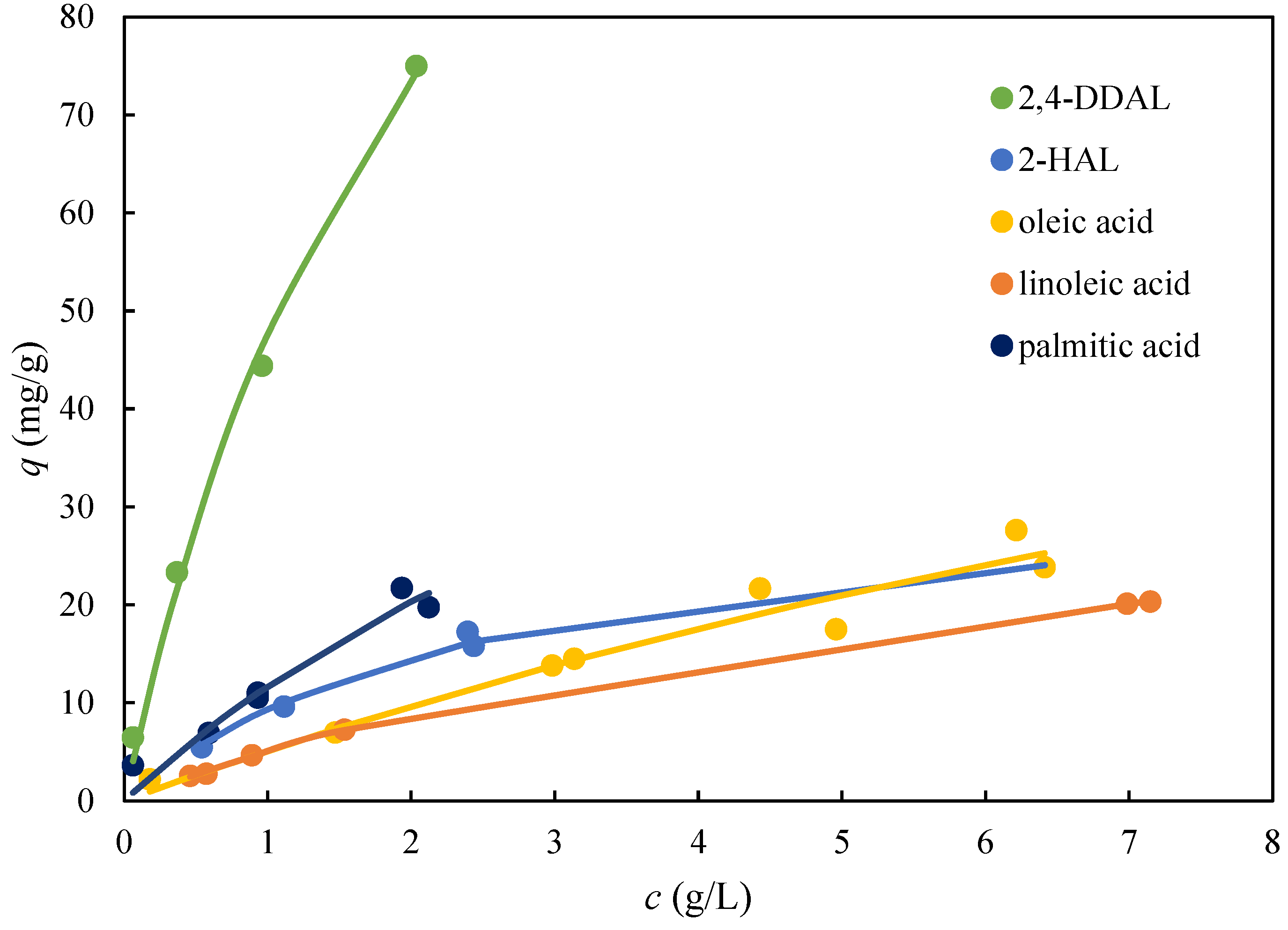
Single component adsorption equilibria were measured for 2,4-DDAL, 2-HAL, and three fatty acids on AmberLite FPX66 (Figure 5). The equilibrium data obtained were well-fitted with the single-component Langmuir adsorption isotherm. Figure 5 shows that the adsorbent capacity and affinity for 2,4-DDAL was several times higher than for all other compounds. The maximum adsorbed amounts of 2-HAL and fatty acids did not exceed the value of 20 mg/g. On the contrary, the adsorbed amount of 75 mg/g was observed for 2,4-DDAL, well below the saturation threshold when the extrapolated value of qmax was close to 200 mg/g (Figure 4B). These results implied that a highly selective separation of 2,4-DDAL from the remaining components of the biocatalytic mixture could be achieved in the desorption phase.
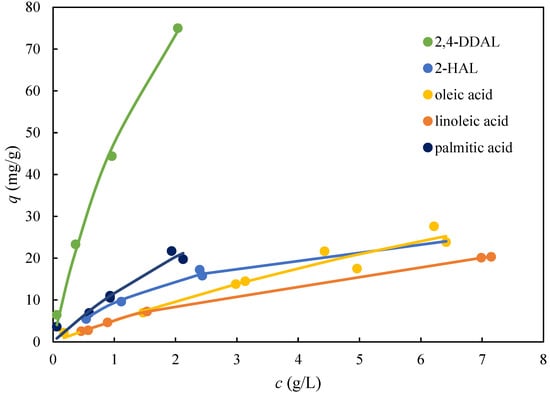
Figure 5.
Single-component adsorption isotherms of the biocatalytic reaction mixture compounds for AmberLite FPX66 from 96% aqueous solutions of EtOH. The points are experimental values, and the lines represent the fits with the Langmuir isotherm.
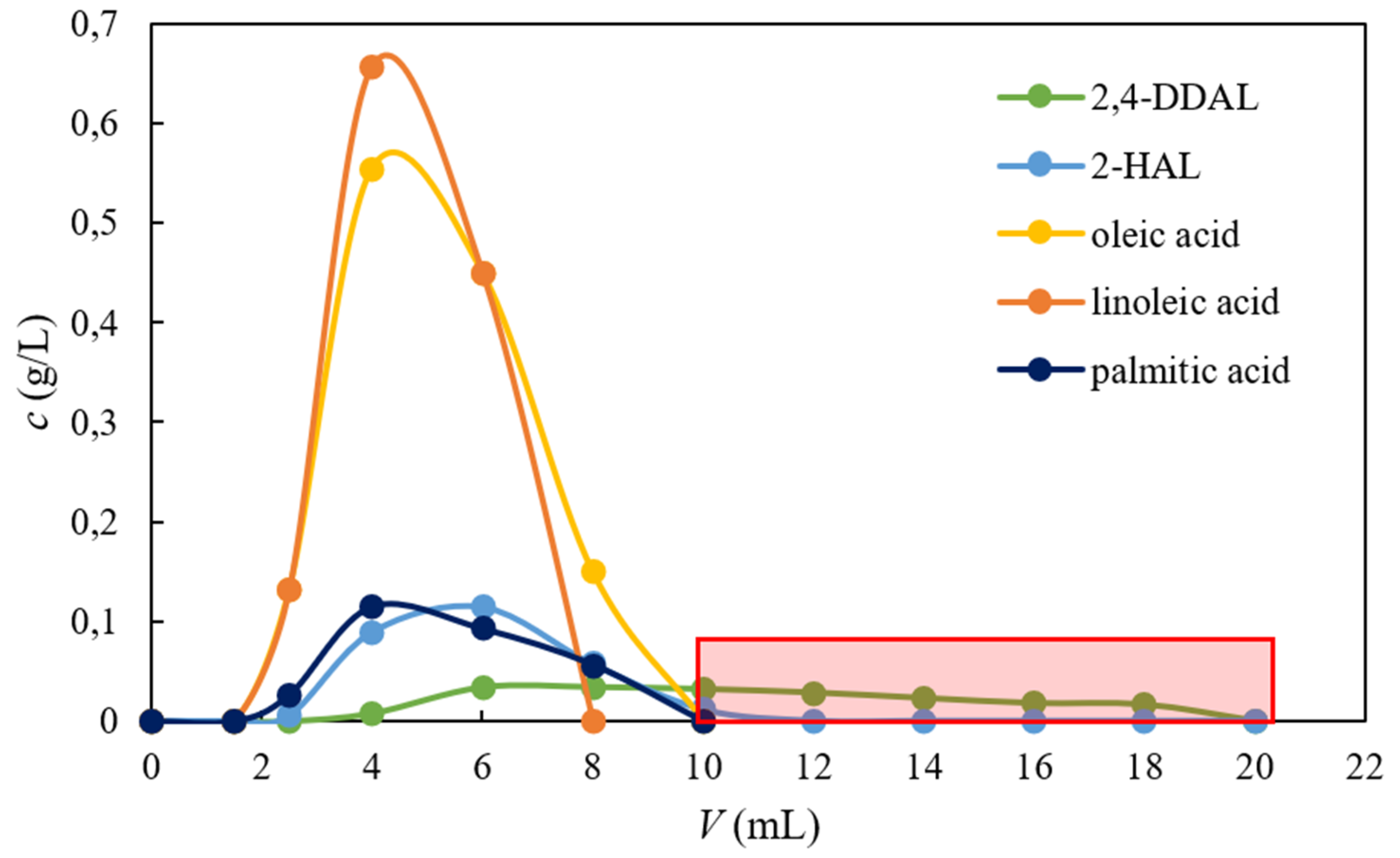
For this purpose, column chromatographic experiments were carried out to verify this hypothesis. The AmberLite FPX66 particles were packed into a small laboratory column. The biocatalytic reaction mixture was fed using a low loading of the adsorbent bed. After the pulse-injection of the 1-mL feed, an elution step using 96% EtOH was applied. The course of an illustrative experiment is presented in Figure 6 where the outlet concentrations of the mixture components are plotted as a function of the retention volume. Figure 6 shows that oleic and linoleic acid eluted out of the column practically in dead volume (approximately 2 mL). The elution of palmitic acid and 2-HAL was slightly delayed compared to the other two fatty acids but also occurred early. These results are in agreement with the adsorption equilibrium data presented in Figure 5. The elution of 2,4-DDAL started last and the peak of 2,4-DDAL was the most broadened, as expected considering the results of batch adsorption experiments in 96% EtOH solution. In the retention volume of 10 mL, all components except for 2,4-DDAL were completely washed out. From this point on, the 2,4-DDAL product was collected. The yield of 2,4-DDAL calculated from Equation (3) reached a value of 53%. The product purity was 97%. It was shown in our recent paper that by the scaling up a bed length formed by AmberLite FPX68 particles from 10 cm to 1 m, the yield of 1-phenylethanol increased from roughly 50% to over 90% [20]. Therefore, these results are promising for further process development.

Figure 6.
Illustrative chromatogram of separation of 2,4-DDAL from the biocatalytic reaction mixture. Experimental details are given in Part 2.4. The red rectangle delimits the period of product collection.
4. Conclusions
The potential of hydrophobic adsorbents for the purification of 2,4-DDAL from a biocatalytic reaction mixture was investigated. The adsorbed amount of all components significantly represented in the mixture was evaluated from batch adsorption experiments. The highest adsorbed amount of 2,4-DDAL was roughly 30 mg/g. For each adsorbent, the selectivity toward 2,4-DDAL was calculated with respect to each component of the mixture. Selectivity values were higher than 1 for all components except 2-HAL; selectivities for linolenic acid were the highest, specifically 4 to 6. The adsorbent AmberLite FPX66 was selected as the most promising for experiments in the chromatographic column.
To set the desorption conditions, batch adsorption experiments were performed with pure 2,4-DDAL solutions using three different solvents: ethanol, isopropanol, and acetone. All three desorbents were suitable for an elution of 2,4-DDAL, but 96% ethanol was chosen as the most suitable for further experiments. Single-component batch adsorption experiments were then performed for all components of the biocatalytic mixture. Obtained adsorption isotherms revealed a very good selectivity of 2,4-DDAL with respect to other components.
To verify the expected separation performance based on the batch adsorption experiments, pulse-injection chromatographic experiments were performed. After the injection of the biocatalytic mixture feed, elution was performed with 96% EtOH. The results obtained from the column experiment were in good agreement with the expected outcome, as 2,4-DDAL was eluted out of the column last and, in the final elution stage, pure 2,4-DDAL was collected. The potential of hydrophobic adsorbents for 2,4-DDAL was confirmed. The yield of 2,4-DDAL in this experiment was 53% with 97% purity, which is promising. The optimization of the elution step and a focus for further process development could increase this yield.
Author Contributions
Conceptualization, K.V. and M.P.; methodology, H.D., J.Č., M.A. and M.O.; validation, M.O. and M.P.; formal analysis, H.D., J.Č., M.A. and M.O.; investigation, M.O.; resources, M.P.; data curation, M.O.; writing—original draft, M.O.; writing—review & editing, M.O. and M.P.; visualization, M.O.; supervision, K.V., M.A. and M.P.; project administration, M.P., funding acquisition, K.V. and M.P. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by grants from the Slovak Research and Development Agency (Grant number: APVV-20-0312) and the Slovak Grant Agency for Science (Grant number: VEGA 1/0515/22).
Data Availability Statement
Data are contained within the article.
Conflicts of Interest
The authors declare no conflict of interest..
References
- Vermeulen, N.; Czerny, M.; Gänzle, M.G.; Schieberle, P.; Vogel, R.F. Reduction, of (E)-2-nonenal and (E,E)-2,4-decadienal during sourdough fermentation. J. Cereal Sci. 2007, 45, 78–87. [Google Scholar] [CrossRef]
- Sun, H.; Peng, X.; Li, C.; Zhang, W.M.; Cao, J. Determination of 2,4-decadienal in edible oils using reversed-phase liquid chromatography and its application as an alternative indicator of lipid oxidation. J. Food Sci. 2020, 85, 1418–1426. [Google Scholar] [CrossRef] [PubMed]
- Opdyke, D.L.J. Monographs on Fragrance Raw Materials. Food Cosmet. Toxicol. 1979, 17, 357–390. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.C.; Yen, G.C. Preventive effect of adding antioxidants on mutagenic compound formation in fumes of cooking oil. J. Sci. Food Agric. 2004, 84, 459–464. [Google Scholar] [CrossRef]
- Chang, Y.C.; Lin, P. Trans, trans-2,4-decadienal induced cell proliferation via p27 pathway in human bronchial epithelial cells. Toxicol. Appl. Pharmacol. 2008, 228, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Boskou, G.; Salta, F.N.; Chiou, A.; Troullidou, E.; Andrikopoulos, N.K. Content of trans,trans-2,4-decadienal in deep-fried and pan-fried potatoes. Eur. J. Lipid Sci. Technol. 2006, 108, 109–115. [Google Scholar] [CrossRef]
- Carvalho, V.M.; Asahara, F.; Di Masciol, P.; De Arruda, I.P.; Jean, C.; Medeiros, M.H.G. Trans, trans-2, 4-decadienal and trans-2-octenal characterization of etheno-2-deoxyguanosine adducts from 2, 4-decadienal and 2-octenal. Biol. React. Intermed. 2001, 6, 229–232. [Google Scholar]
- Chang, L.W.; Lo, W.S.; Lin, P. Trans, Trans-2, 4-decadienal, a product found in cooking oil fumes, induces cell proliferation and cytokine production due to reactive oxygen species in human bronchial epithelial cells. Toxicol. Sci. 2005, 87, 337–343. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Yen, G.; Sheu, F. Mutagenicity and Identication of Mutagenic Compounds of fumes obtained from heating peanut oil. J. Food Protect. 2001, 64, 240–245. [Google Scholar] [CrossRef] [PubMed]
- Boonprab, K.; Matsui, K.; Akakabe, Y.; Yotsokura, N.; Kajiwara, T. 11-Hydroperoxide eicosanoid-mediated 2(E),4(E)-decadienal production from arachidonic acid in the brown algae, Saccharina angustata. J. Appl. Phycol. 2019, 31, 2719–2727. [Google Scholar] [CrossRef]
- Nielsen, G.S.; Larsen, L.M.; Poll, L. Formation of volatile compounds in model experiments with crude leek (Allium ampeloprasum var. Lancelot) enzyme extract and linoleic acid or linolenic acid. J. Agric Food Chem. 2004, 52, 2315–2321. [Google Scholar] [CrossRef] [PubMed]
- Almosnino, A.M.; Bensoussan, M.; Belin, J.M. Unsaturated fatty acid bioconversion by apple pomace enzyme system. Factors influencing the production of aroma compounds. Food Chem. 1996, 55, 327–332. [Google Scholar] [CrossRef]
- Watson, S.B. Aquatic taste and odor: A primary signal of drinking-water integrity. J. Toxicol. Environ. Health Part A 2004, 67, 1779–1795. [Google Scholar] [CrossRef] [PubMed]
- Satchwill, T.; Watson, S.B.; Dixon, E. Odourous algal-derived alkenes: Differences in stability and treatment responses in drinking water. Water Sci. Technol. 2007, 55, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, J.; Zhang, X.; Chen, C. Powdered activated carbon adsorption of two fishy odorants in water: Trans,trans-2,4-heptadienal and trans,trans-2,4-decadienal. J. Environ. Sci. 2015, 32, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Obendorf, S.K.; Liu, H.; Tan, K.; Leonard, M.J.; Young, T.J.; Incorvia, M.J. Adsorption of aroma chemicals on cotton fabric in different aqueous environments. J. Surfactants Deterg. 2009, 12, 43–58. [Google Scholar] [CrossRef]
- Xu, Y.; Bi, S.; Xiong, C.; Dai, Y.; Zhou, Q.; Liu, Y. Identification of aroma active compounds in walnut oil by monolithic material adsorption extraction of RSC18 combined with gas chromatography-olfactory-mass spectrometry. Food Chem. 2023, 402, 134303. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H. Adsorption kinetics and thermodynamics of yeast β-glucan for off-odor compounds in silver carp mince. Food Chem. 2020, 319, 126232. [Google Scholar] [CrossRef] [PubMed]
- Ostrihoňová, M.; Gramblička, M.; Polakovič, M. Industrial hydrophobic adsorbent screening for the separation of 1-phenylethanol and acetophenone, Food Bioproduct. Proc. 2023, 137, 124–134. [Google Scholar] [CrossRef]
- Ostrihoňová, M.; . Cabadaj, P.; Polakovič, M. Design of frontal chromatography separation of 1-phenylethanol and acetophenone using a hydrophobic resin. Separ. Purif. Technol. 2023, 314, 123578. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).