A Preliminary Life Cycle Analysis of Bioethanol Production Using Seawater in a Coastal Biorefinery Setting
Abstract
:1. Introduction
2. Materials and Methods
2.1. Goal and Scope
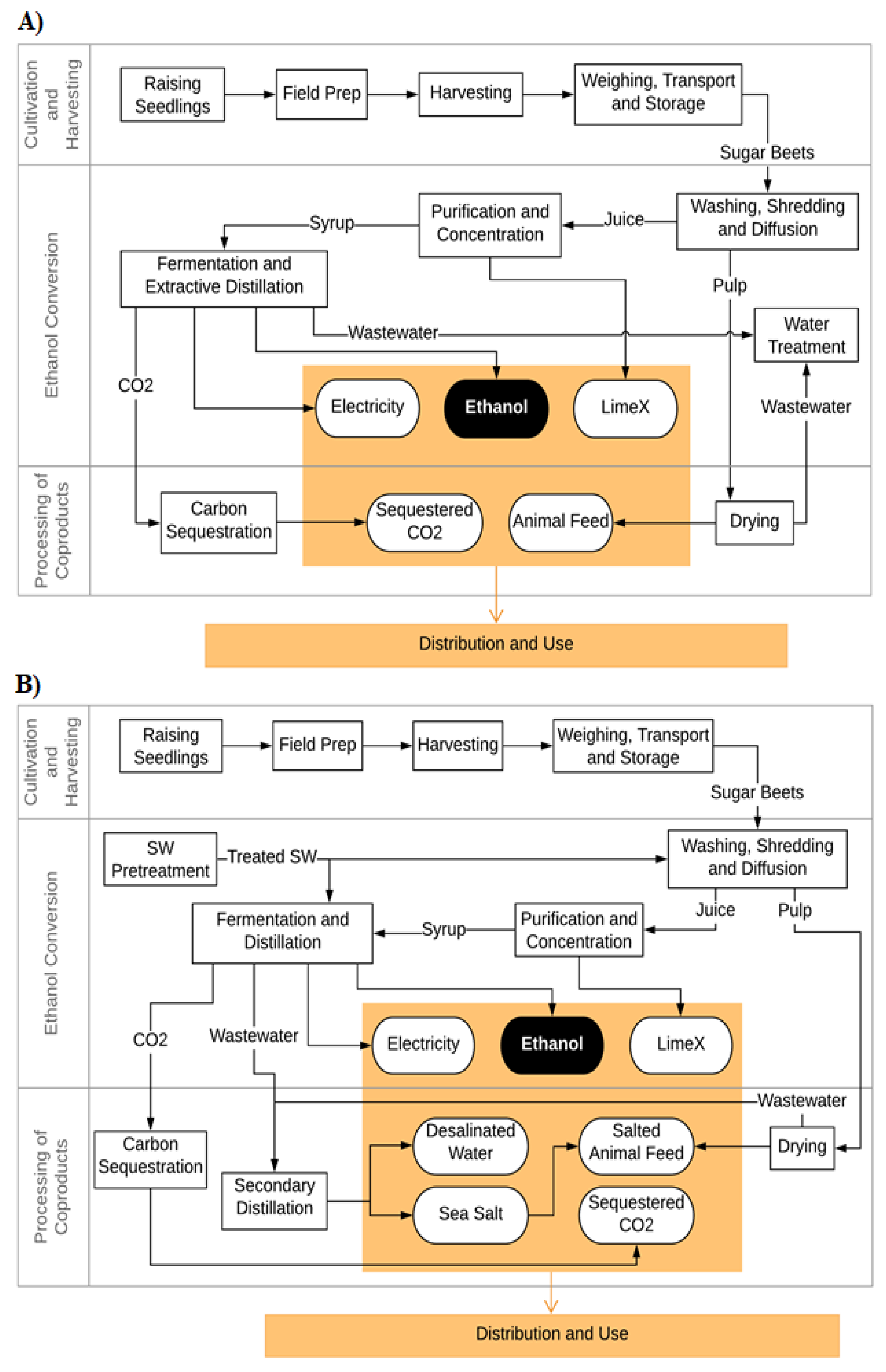
2.2. Modeling and Input Data
2.2.1. Sugar Beet Cultivation and Harvesting
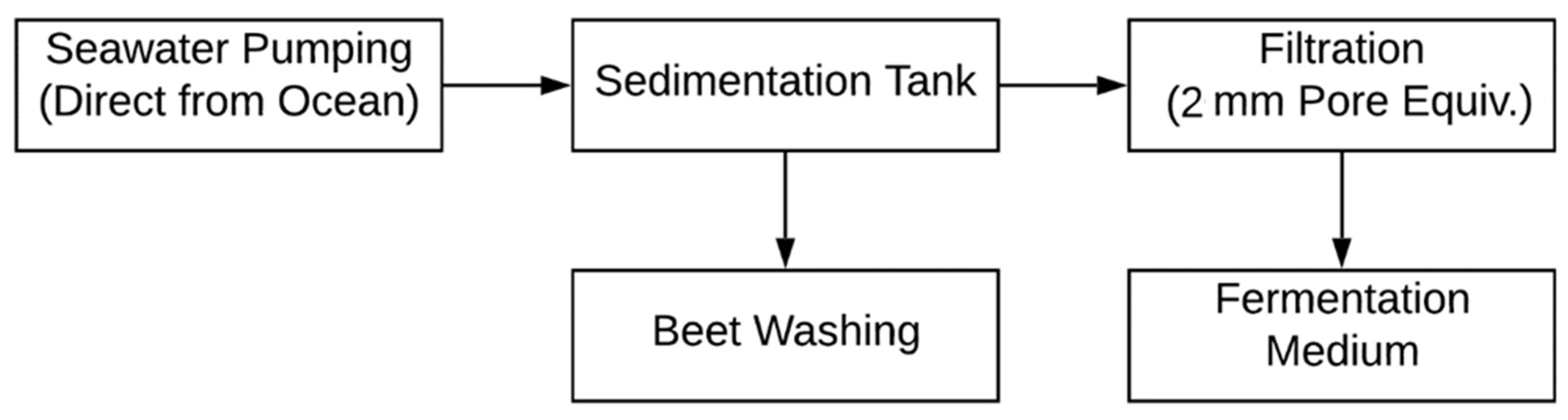
2.2.2. Seawater Pretreatment
2.2.3. Sugar Beet Washing
2.2.4. Syrup Production
2.2.5. Fermentation and Ethanol Recovery
2.2.6. Processing of Coproducts
2.3. Allocation
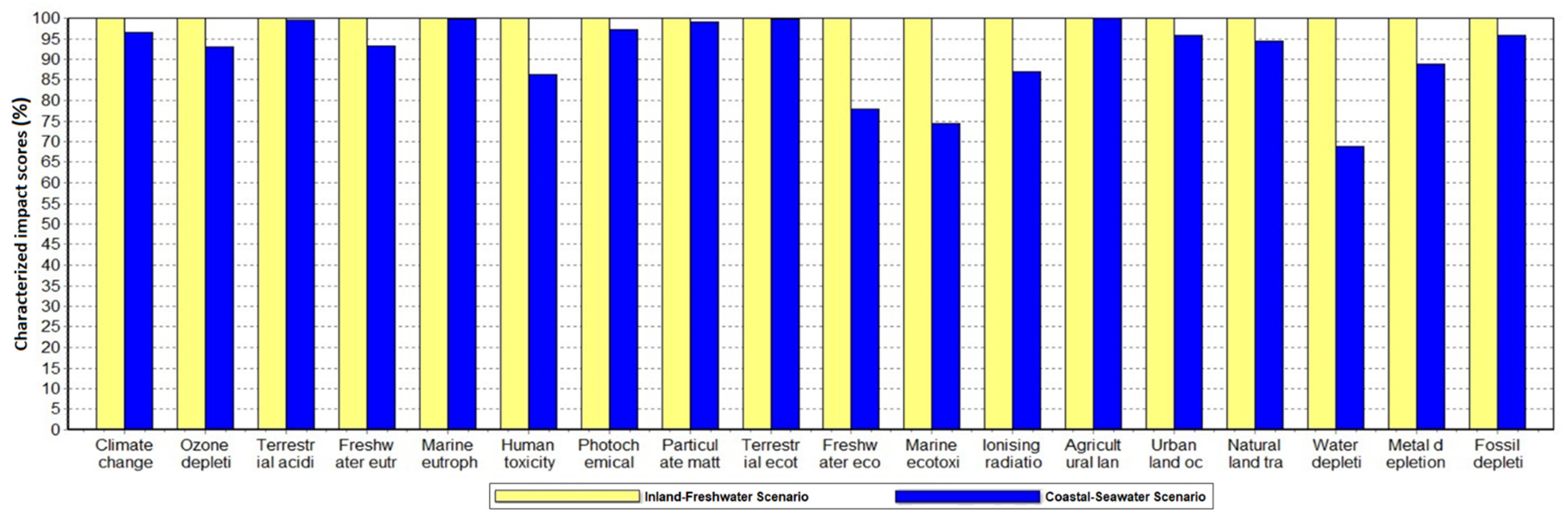
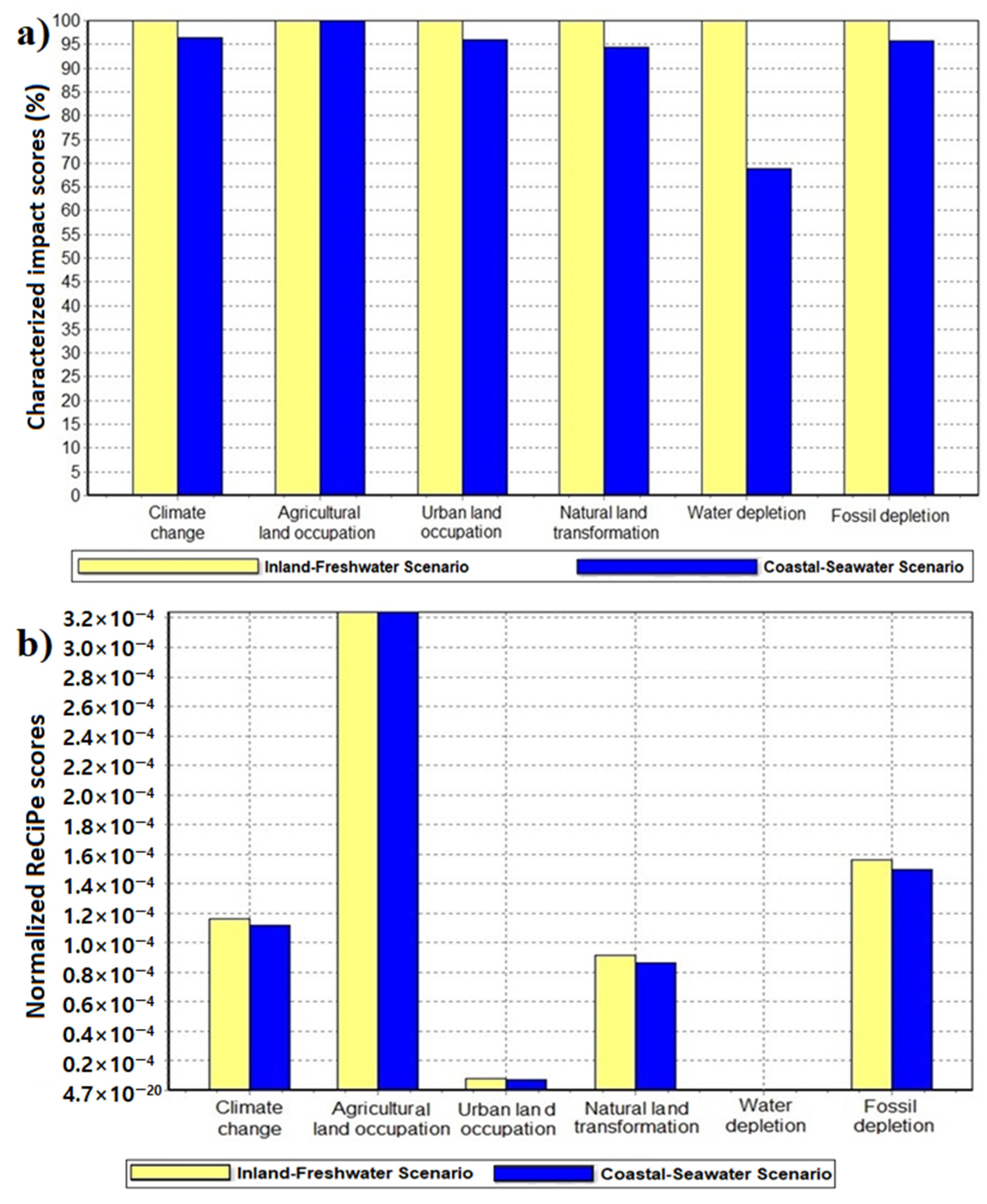
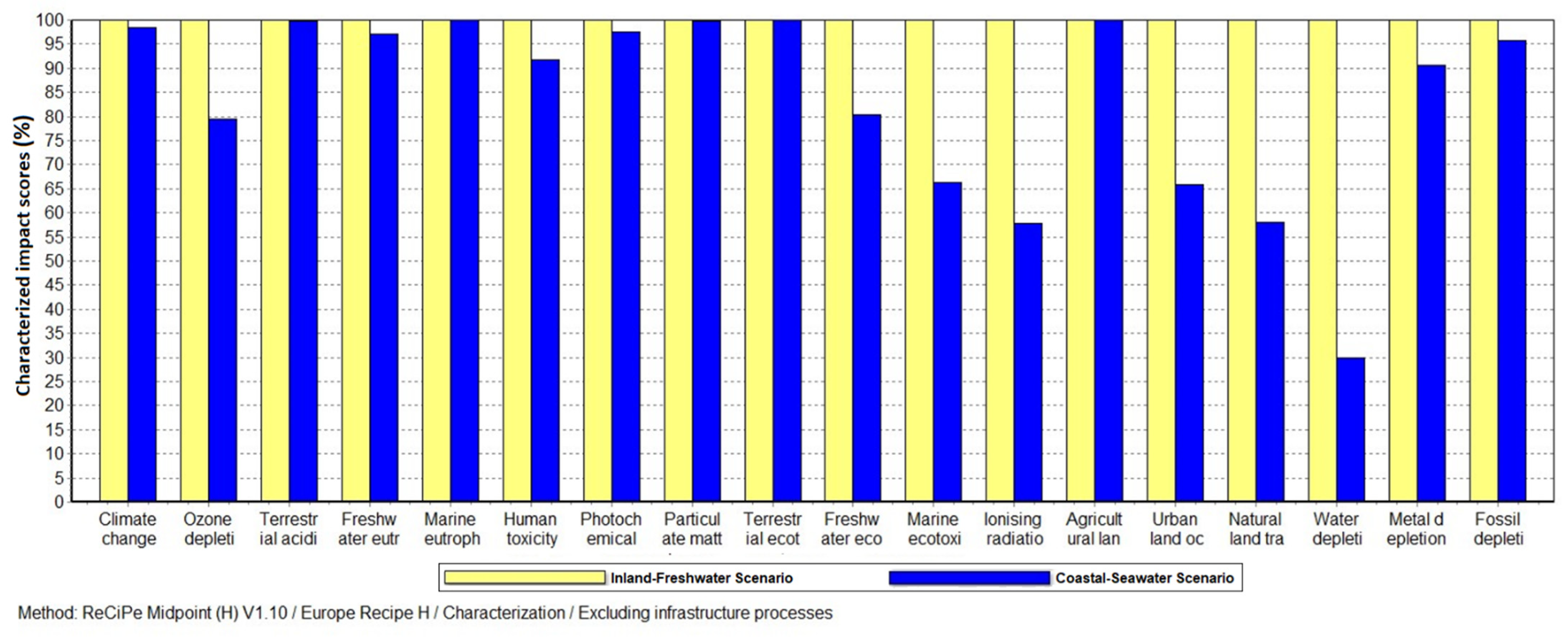
3. Results
4. Discussion
4.1. Water Depletion Impact and Other Benefits of Seawater Fermentation
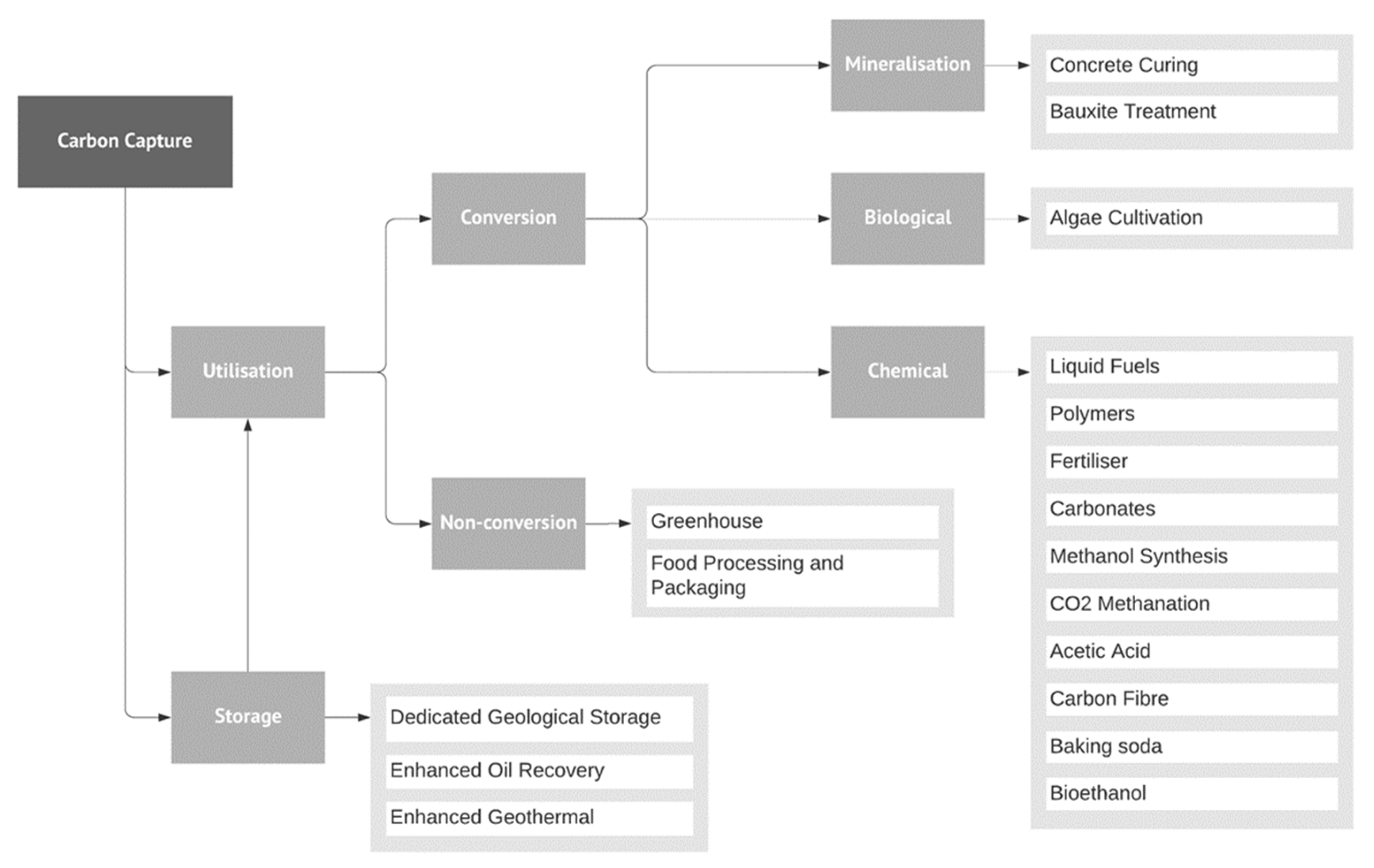
4.2. Climate Change and CCS Technology
4.3. Coproduct Profiles and Economic Discussion
4.4. Assumptions and Limitations
4.5. Future Perspectives: Toward a Coastal Integrated Marine Biorefinery (CIMB) System
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- United Nations. Paris Agreement; United Nations: Paris, France, 2016; pp. 1–27. [Google Scholar]
- Ritchie, H.; Rosner, M. CO2 and Greenhouse Gas Emissions. 2020. Available online: https://ourworldindata.org/co2-and-other-greenhouse-gas-emissions (accessed on 20 April 2020).
- Duque, A.; Álvarez, C.; Doménech, P.; Manzanares, P.; Moreno, A.D. Advanced Bioethanol Production: From Novel Raw Materials to Integrated Biorefineries. Processes 2021, 9, 206. [Google Scholar] [CrossRef]
- Fulton, L.M.; Lynd, L.R.; Körner, A.; Greene, N.; Tonachel, L.R. The need for biofuels as part of a low carbon energy future. Biofuels Bioprod. Biorefining 2015, 9, 476–483. [Google Scholar] [CrossRef]
- Zhang, J.; Fisher, T.S.; Ramachandran, P.V.; Gore, J.P.; Mudawar, I. A review of heat transfer issues in hydrogen storage technologies. J. Heat Transf. 2005, 127, 1391–1399. [Google Scholar] [CrossRef]
- Felderhoff, M.; Weidenthaler, C.; von Helmolt, R.; Eberle, U. Hydrogen storage: The remaining scientific and technological challenges. Phys. Chem. Chem. Phys. 2007, 9, 2643–2653. [Google Scholar] [CrossRef]
- Den Boer, E.; Aarnink, S.; Kleiner, F.; Pagenkopf, J. Zero Emissions Trucks: An Overview of State-of-the-Art Technologies and Their Potential. 2013. Available online: https://theicct.org/sites/default/files/publications/CE_Delft_4841_Zero_emissions_trucks_Def.pdf (accessed on 11 August 2021).
- Metz, B.; Davidson, O.; De Coninck, H.; Loos, M.; Meyer, L. IPCC Special Report on Carbon Dioxide Capture and Storage; Cambridge University Press: Cambridge, UK, 2005; Available online: https://www.ipcc.ch/site/assets/uploads/2018/03/srccs_wholereport-1.pdf (accessed on 11 August 2021).
- GlobalCCSInstitute. Global Status of CCS 2019: Targeting Climate Change; The Global CCS Institute: Melbourne, Australia, 2019. [Google Scholar]
- Pal, A. Blending of ethanol in gasoline: Impact on SI engine performance and emissions. Int. J. Therm. Technol. 2014, 4, 1–5. [Google Scholar]
- Al-Hasan, M. Effect of ethanol–unleaded gasoline blends on engine performance and exhaust emission. Energy Convers. Manag. 2003, 44, 1547–1561. [Google Scholar] [CrossRef]
- Muñoz, M.; Heeb, N.V.; Haag, R.; Honegger, P.; Zeyer, K.; Mohn, J.; Comte, P.; Czerwinski, J. Bioethanol blending reduces nanoparticle, PAH, and alkyl-and nitro-PAH emissions and the genotoxic potential of exhaust from a gasoline direct injection flex-fuel vehicle. Environ. Sci. Technol. 2016, 50, 11853–11861. [Google Scholar] [CrossRef]
- Department for Transport. Fuelling a Greener Future: E10 Petrol Set for September 2021 Launch. 2021. Available online: https://www.gov.uk/government/news/fuelling-a-greener-future-e10-petrol-set-for-september-2021-launch (accessed on 4 March 2021).
- Bailey, B.K. Performance of ethanol as a transportation fuel. In Handbook on Bioethanol; Routledge: Abingdon, UK, 2018; pp. 37–60. [Google Scholar]
- Speidel, H.K.; Ahmed, I. Biodegradability Characteristics of Current and Newly-Developed Alternative Fuels; SAE Technical Paper 1999-01-351; SAE International: Warrendale, PA, USA, 1999. [Google Scholar] [CrossRef]
- Dias De Oliveira, M.E.; Vaughan, B.E.; Rykiel, E.J. Ethanol as fuel: Energy, carbon dioxide balances, and ecological footprint. BioScience 2005, 55, 593–602. [Google Scholar] [CrossRef]
- European Union. Directive (EU) 2018/2001 of the European Parliament and of the Council of 11 December 2018 on the promotion of the use of energy from renewable sources (Text with EEA relevance.). Off. J. Eur. Union 2018, 328, 82–209. [Google Scholar]
- Liu, X.V.; Hoekman, S.K.; Broch, A. Potential water requirements of increased ethanol fuel in the USA. Energy Sustain. Soc. 2017, 7, 18. [Google Scholar] [CrossRef] [Green Version]
- Zaky, A.; Greetham, D.; Tucker, G.; Du, C. The establishment of a marine focused biorefinery for bioethanol production using seawater and a novel marine yeast strain. Sci. Rep. 2018, 8, 12127. [Google Scholar] [CrossRef] [Green Version]
- Gerbens-Leenes, W.; Hoekstra, A.Y.; van der Meer, T.H. The water footprint of bioenergy. Proc. Natl. Acad. Sci. USA 2009, 106, 10219–10223. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gerbens-Leenes, P. Green, blue and grey bioenergy water footprints, a comparison of feedstocks for bioenergy supply in 2040. Environ. Process. 2018, 5, 167–180. [Google Scholar] [CrossRef] [Green Version]
- de María, P.D. On the use of seawater as reaction media for large-scale applications in biorefineries. ChemCatChem 2013, 5, 1643–1648. [Google Scholar] [CrossRef]
- Zaky, A.; Greetham, D.; Louis, E.; Tucker, G.; Du, C. A New Isolation and Evaluation Method for Marine-Derived Yeast spp. with Potential Applications in Industrial Biotechnology. J. Microbiol. Biotechnol. 2016, 26, 1891–1907. [Google Scholar] [CrossRef] [Green Version]
- Zaky, A.S.; French, C.E.; Tucker, G.A.; Du, C. Improving the productivity of bioethanol production using marine yeast and seawater-based media. Biomass Bioenergy 2020, 139, 105615. [Google Scholar] [CrossRef]
- International Organization for Standardization. Environmental Management: Life Cycle Assessment; Requirements and Guidelines; ISO: Geneva, Switzerland, 2006. [Google Scholar]
- Goedkoop, M.; Oele, M.; de Schryver, A.; Vieira, M.; Hegger, S. SimaPro Database Manual Methods Library; PRé Consultants: Amersfoort, The Netherlands, 2008; pp. 22–25. [Google Scholar]
- Wernet, G.; Bauer, C.; Steubing, B.; Reinhard, J.; Moreno-Ruiz, E.; Weidema, B. The ecoinvent database version 3 (part I): Overview and methodology. Int. J. Life Cycle Assess. 2016, 21, 1218–1230. [Google Scholar] [CrossRef]
- Durlinger, B.; Tyszler, M.; Scholten, J.; Broekema, R.; Blonk, H.; Beatrixstraat, G. Agri-Footprint; a Life Cycle Inventory database covering food and feed production and processing. In Proceedings of the 9th International Conference on Life Cycle Assessment in the Agri-Food Sector, San Francisco, CA, USA, 8–10 October 2014; pp. 310–317. [Google Scholar]
- Vierhout, R. Renewable Ethanol: Driving Jobs, Growth and Innovation throughout Europe; State of the Industry Report; ePure European Renewable Ethanol Association, 2014; Available online: https://www.liquid-energy.ch/wp/wp-content/uploads/2014-06-14-Ethanol-State-of-the-Industry-Report-2014.pdf (accessed on 11 August 2021).
- Šantek, B.; Gwehenberger, G.; Šantek, M.I.; Narodoslawsky, M.; Horvat, P. Evaluation of energy demand and the sustainability of different bioethanol production processes from sugar beet. Resour. Conserv. Recycl. 2010, 54, 872–877. [Google Scholar] [CrossRef]
- Mundi, G.S.; Zytner, R.G.; Warriner, K. Fruit and vegetable wash-water characterization, treatment feasibility study and decision matrices. Can. J. Civ. Eng. 2017, 44, 971–983. [Google Scholar] [CrossRef] [Green Version]
- Campiotti, C.A.; Latini, A.; Scoccianti, M.; Biagiotti, D.; Giagnacovo, G.; Viola, C. Energy efficiency in Italian fruit and vegetables processing industries in the EU agro-food sector context. Riv. Studi Sulla Sostenibilita 2014, 2, 159–174. [Google Scholar]
- Mortimer, N.; Elsayed, M.; Horne, R. Energy and Greenhouse Gas Emissions for Bioethanol Production from Wheat Grain and Sugar Beet; Resources Research Unit School of Environment and Development, Sheffield Hallam University: Sheffield, UK, 2004. [Google Scholar]
- Rao, P.; Morrow, W.R., III; Aghajanzadeh, A.; Sheaffer, P.; Dollinger, C.; Brueske, S.; Cresko, J. Energy considerations associated with increased adoption of seawater desalination in the United States. Desalination 2018, 445, 213–224. [Google Scholar] [CrossRef] [Green Version]
- Pfromm, P.H. The minimum water consumption of ethanol production via biomass fermentation. Open Chem. Eng. J. 2008, 2, 1–5. [Google Scholar] [CrossRef] [Green Version]
- Aspelund, A.; Jordal, K. Gas conditioning—The interface between CO2 capture and transport. Int. J. Greenh. Gas Control 2007, 1, 343–354. [Google Scholar] [CrossRef]
- Zaky, A.S. Marine Fermentation, the Sustainable Approach for Bioethanol Production. EC Microbiol. 2017, 25–27. Available online: https://www.ecronicon.com/eco16/pdf/ECMI-01-ECO-11.pdf (accessed on 11 August 2021).
- European Commission. Directive of the European Parliament and of the Council on the Promotion of the Use of Energy from Renewable Sources; European Commission: Brussels, Belgium, 2008. [Google Scholar]
- Stocker, T.; Qin, D.; Plattner, G.; Tignor, M.; Allen, S.; Boschung, J.; Nauels, A.; Xia, Y.; Bex, B.; Midgley, B. IPCC, 2013: Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; IPCC: Cambridge, UK; New York, NY, USA, 2013; Available online: https://www.ipcc.ch/site/assets/uploads/2017/09/WG1AR5_Frontmatter_FINAL.pdf (accessed on 11 August 2021).
- Lee, F.M.; Pahl, R.H. Solvent screening study and conceptual extractive distillation process to produce anhydrous ethanol from fermentation broth. Ind. Eng. Chem. Process Des. Dev. 1985, 24, 168–172. [Google Scholar] [CrossRef]
- Pinto, R.; Wolf-Maciel, M.; Lintomen, L. Saline extractive distillation process for ethanol purification. Comput. Chem. Eng. 2000, 24, 1689–1694. [Google Scholar] [CrossRef]
- Budinis, S.; Krevor, S.; Mac Dowell, N.; Brandon, N.; Hawkes, A. An assessment of CCS costs, barriers and potential. Energy Strategy Rev. 2018, 22, 61–81. [Google Scholar] [CrossRef]
- Laude, A.; Ricci, O.; Bureau, G.; Royer-Adnot, J.; Fabbri, A. CO2 capture and storage from a bioethanol plant: Carbon and energy footprint and economic assessment. Int. J. Greenh. Gas Control 2011, 5, 1220–1231. [Google Scholar] [CrossRef]
- Schrag, D.P. Storage of carbon dioxide in offshore sediments. Science 2009, 325, 1658–1659. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rackley, S.A. (Ed.) Chapter 12—Ocean Storage. In Carbon Capture and Storage; Butterworth-Heinemann: Boston, MA, USA, 2010; pp. 267–286. [Google Scholar] [CrossRef]
- Morcos, S.A. Chemical Composition of Seawater and the Variation of Calcium and Alkalinity. ICES J. Mar. Sci. 1970, 33, 126–133. [Google Scholar] [CrossRef]
- Behera, M.R.; Varade, S.R.; Ghosh, P.; Paul, P.; Negi, A.S. Foaming in Micellar Solutions: Effects of Surfactant, Salt, and Oil Concentrations. Ind. Eng. Chem. Res. 2014, 53, 18497–18507. [Google Scholar] [CrossRef]
- Zaky, A.S.; Pensupa, N.; Andrade-Eiroa, Á.; Tucker, G.A.; Du, C. A new HPLC method for simultaneously measuring chloride, sugars, organic acids and alcohols in food samples. J. Food Compos. Anal. 2017, 56, 25–33. [Google Scholar] [CrossRef]
- Papong, S.; Rewlay-ngoen, C.; Itsubo, N.; Malakul, P. Environmental life cycle assessment and social impacts of bioethanol production in Thailand. J. Clean. Prod. 2017, 157, 254–266. [Google Scholar] [CrossRef]
- Milazzo, M.F.; Spina, F. The use of the risk assessment in the life cycle assessment framework. Manag. Environ. Qual. Int. J. 2015, 26, 389–406. [Google Scholar] [CrossRef]
- Zaky, A.S.; Tucker, G.A.; Daw, Z.Y.; Du, C. Marine yeast isolation and industrial application. FEMS Yeast Res. 2014, 14, 813–825. [Google Scholar] [CrossRef]
- Greetham, D.; Zaky, A.; Makanjuola, O.; Du, C. A brief review on bioethanol production using marine biomass, marine microorganism and seawater. Curr. Opin. Green Sustain. Chem. 2018, 14, 53–59. [Google Scholar] [CrossRef] [Green Version]
| Input Data | Unit | Inland Freshwater or Coastal Seawater (a) |
|---|---|---|
| Occupation, arable | m2a | 10,000 |
| Water, unspecified natural origin, DE | m3/ha | 186.1 |
| Energy from diesel burned in machinery, RER Energy | MJ/ha | 7367.25 |
| Manure from pigs, at pig farm, RER Energy | kg/ha | 8732.39 |
| Potassium chloride (NPK 0-0-60) at regional storehouse, RER Energy | kg/ha | 162.38 |
| NPK compound (NPK 15-15-15) at regional storehouse, RER Energy | kg/ha | 213.41 |
| PK compound (NPK 0-22-22) at regional storehouse, RER Energy | kg/ha | 91.55 |
| Potassium sulfate (NPK 0-0-50) at regional storehouse, RER Energy | kg/ha | 20.84 |
| Triple superphosphate as 80% Ca(H2PO4)2 (NPK 0-48-0) at regional storehouse, RER Energy | kg/ha | 16.35 |
| Ammonium sulfate as 100% (NH4)2SO4 (NPK 21-0-0) at regional storehouse, RER Energy | kg/ha | 28.55 |
| Calcium ammonium nitrate (CAN) (NPK 26.5-0-0) at regional storehouse, RER Energy | kg/ha | 242.87 |
| Liquid urea-ammonium nitrate solution (NPK 30-0-0) at regional storehouse, RER Energy | kg/ha | 56.34 |
| Urea as 100% CO(NH2)2 (NPK 46.6-0-0) at regional storehouse, RER Energy | kg/ha | 65.94 |
| Lime fertilizer at regional storehouse, RER Energy | kg/ha | 290.74 |
| Input Data | Unit | Inland Freshwater | Seawater Coastal | Source (a) |
|---|---|---|---|---|
| Washing and Syrup Production | ||||
| Tap Water | L/kg SB | 5.000 | 0 | [31] |
| Seawater | L/kg SB | 0 | 5.000 | |
| Electricity (Washing) | kWh/kg SB | 4.000 | 4.000 | [32] |
| Hydrochloric Acid | g/kg SB | 0.025 | 0.025 | [33] |
| Formaldehyde | g/kg SB | 0.150 | 0.150 | |
| LimeX Recovered | g/kg SB | 45.000 | 45.000 | |
| Water Inlet or Treatment | ||||
| Wastewater Treatment (for Disposal) | m3 H2O/kg Et | 3.140 | 0 | [30] |
| Electricity (Seawater Pumping) | kWh/m3 H2O | 0 | 0.004 | [34] |
| Fermentation | ||||
| Clean Sugar Beet | kg/kg Et | 8.535 | 8.535 | [27] |
| Sulfuric Acid | kg/kg Et | 0.026 | 0.026 | |
| Sodium Sulfate | kg/kg Et | 0.003 | 0.003 | |
| Fresh Water for Medium | L/kg Et | 0.814 | 0 | |
| Seawater for Medium | L/kg Et | 0 | 0.814 | |
| Water for Cooling | kg/kg Et | 0.3 | 0.3 | [35] |
| Antiscalant | g/kg SB | 0.040 | 0.040 | [33] |
| Coke | g/kg SB | 1.800 | 1.800 | |
| Anti-Foam | g/kg SB | 0.200 | 0.200 | |
| Electricity (Surplus) | kWh/kg SB | 0.694 | 0.694 | |
| Input Data | Unit | Inland Freshwater | Coastal Seawater | Source (a) |
|---|---|---|---|---|
| Carbon Sequestration | ||||
| CO2 from Fermentation | kg/kg Et | 0.713 | 0.713 | [27] |
| Electricity | kWh/kg CO2 | 0.105 (b) | 0.105 (b) | [36] |
| Animal Feed Production | ||||
| Wet Sugar Beet Pulp | kg/kg Et | 1.392 | 1.392 | [27] |
| Process Steam | MJ/kg wet pulp | 1.413 | 1.413 | [28] |
| Electricity for Drying | MJ/kg wet pulp | 0.043 | 0.043 | |
| Dried Sugar Beet Pulp | kg/kg wet pulp | 0.243 | 0.243 | |
| Distillate Water Treatment | ||||
| Water from Distillation | kg/L Et | 0 | 7.000 | [37] |
| Sea Salt from Distillation | kg/L Et | 0 | 0.208 |
| Impact Category | Unit | Inland Freshwater | Coastal Seawater |
|---|---|---|---|
| Climate change | kg CO2 eq | 1.30 | 1.26 |
| Ozone depletion | kg CFC-11 eq | 4.37 × 10−8 | 4.06 × 10−8 |
| Terrestrial acidification | kg SO2 eq | 4.13 × 10−2 | 4.11 × 10−2 |
| Freshwater eutrophication | kg P eq | 3.96 × 10−4 | 3.69 × 10−4 |
| Marine eutrophication | kg N eq | 2.44 × 10−2 | 2.44 × 10−2 |
| Human toxicity | kg 1,4-DB eq | 1.33 × 10−1 | 1.15 × 10−1 |
| Photochemical oxidant formation | kg NMVOC | 3.30 × 10−3 | 3.21 × 10−3 |
| Particulate matter formation | kg PM10 eq | 6.05 × 10−3 | 5.99 × 10−3 |
| Terrestrial ecotoxicity | kg 1,4-DB eq | 1.82 × 10−3 | 1.81 × 10−3 |
| Freshwater ecotoxicity | kg 1,4-DB eq | 2.30 × 10−3 | 1.79 × 10−3 |
| Marine ecotoxicity | kg 1,4-DB eq | 1.66 × 10−3 | 1.23 × 10−3 |
| Ionizing radiation | kBq U235 eq | 1.38 × 10−1 | 1.20 × 10−1 |
| Agricultural land occupation | m2a | 1.47 | 1.47 |
| Urban land occupation | m2a | 3.14 × 10−3 | 3.01 × 10−3 |
| Natural land transformation | m2 | 1.48 × 10−5 | 1.40 × 10−5 |
| Water depletion | m3 | 9.13 × 10−1 | 6.28 × 10−1 |
| Metal depletion | kg Fe eq | 3.84 × 10−3 | 3.41 × 10−3 |
| Fossil depletion | kg oil eq | 2.43 × 10−1 | 2.32 × 10−1 |
| Impact Category | Unit | Inland Freshwater | Coastal Seawater |
|---|---|---|---|
| Climate change | kg CO2 eq | 8.90 × 10−2 | 8.76 × 10−2 |
| Ozone depletion | kg CFC-11 eq | 3.50 × 10−10 | 2.78 × 10−10 |
| Terrestrial acidification | kg SO2 eq | 4.62 × 10−3 | 4.61 × 10−3 |
| Freshwater eutrophication | kg P eq | 4.10 × 10−5 | 3.97 × 10−5 |
| Marine eutrophication | kg N eq | 2.85 × 10−3 | 2.85 × 10−3 |
| Human toxicity | kg 1,4-DB eq | 1.08 × 10−2 | 9.92 × 10−3 |
| Photochemical oxidant formation | kg NMVOC | 1.21 × 10−4 | 1.18 × 10−4 |
| Particulate matter formation | kg PM10 eq | 6.28 × 10−4 | 6.26 × 10−4 |
| Terrestrial ecotoxicity | kg 1,4-DB eq | 2.07 × 10−4 | 2.07 × 10−4 |
| Freshwater ecotoxicity | kg 1,4-DB eq | 1.51 × 10−4 | 1.21 × 10−4 |
| Marine ecotoxicity | kg 1,4-DB eq | 6.18 × 10−5 | 4.09 × 10−5 |
| Ionizing radiation | kBq U235 eq | 1.63 × 10−3 | 9.43 × 10−4 |
| Agricultural land occupation | m2a | 1.70 × 10−1 | 1.70 × 10−1 |
| Urban land occupation | m2a | 1.97 × 10−5 | 1.30 × 10−5 |
| Natural land transformation | m2 | 9.13 × 10−8 | 5.31 × 10−8 |
| Water depletion | m3 | 2.35 × 10−2 | 7.06 × 10−3 |
| Metal depletion | kg Fe eq | 3.19 × 10−5 | 2.90 × 10−5 |
| Fossil depletion | kg oil eq | 7.72 × 10−3 | 7.40 × 10−3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zaky, A.S.; Carter, C.E.; Meng, F.; French, C.E. A Preliminary Life Cycle Analysis of Bioethanol Production Using Seawater in a Coastal Biorefinery Setting. Processes 2021, 9, 1399. https://doi.org/10.3390/pr9081399
Zaky AS, Carter CE, Meng F, French CE. A Preliminary Life Cycle Analysis of Bioethanol Production Using Seawater in a Coastal Biorefinery Setting. Processes. 2021; 9(8):1399. https://doi.org/10.3390/pr9081399
Chicago/Turabian StyleZaky, Abdelrahman S., Claudia E. Carter, Fanran Meng, and Christopher E. French. 2021. "A Preliminary Life Cycle Analysis of Bioethanol Production Using Seawater in a Coastal Biorefinery Setting" Processes 9, no. 8: 1399. https://doi.org/10.3390/pr9081399
APA StyleZaky, A. S., Carter, C. E., Meng, F., & French, C. E. (2021). A Preliminary Life Cycle Analysis of Bioethanol Production Using Seawater in a Coastal Biorefinery Setting. Processes, 9(8), 1399. https://doi.org/10.3390/pr9081399







