Nanoadsorbants for the Removal of Heavy Metals from Contaminated Water: Current Scenario and Future Directions
Abstract
:1. Introduction
2. Nanomaterials Applied for the Removal of Metallic Ions from Water
2.1. Carbon Based Nanomaterials
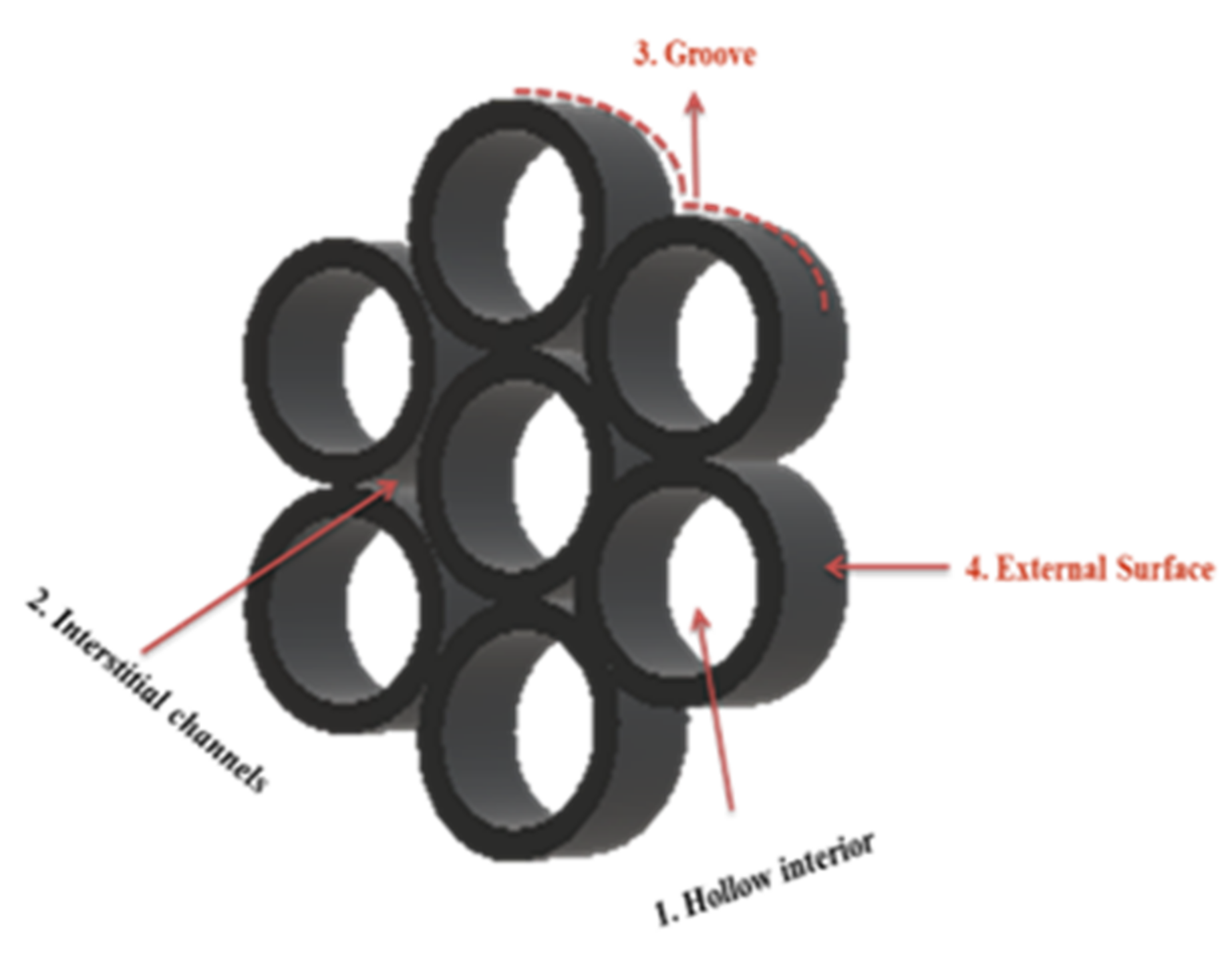
2.1.1. Carbon Nanotubes
2.1.2. Graphene Based Adsorbents
2.2. Silica Based Nanomaterials
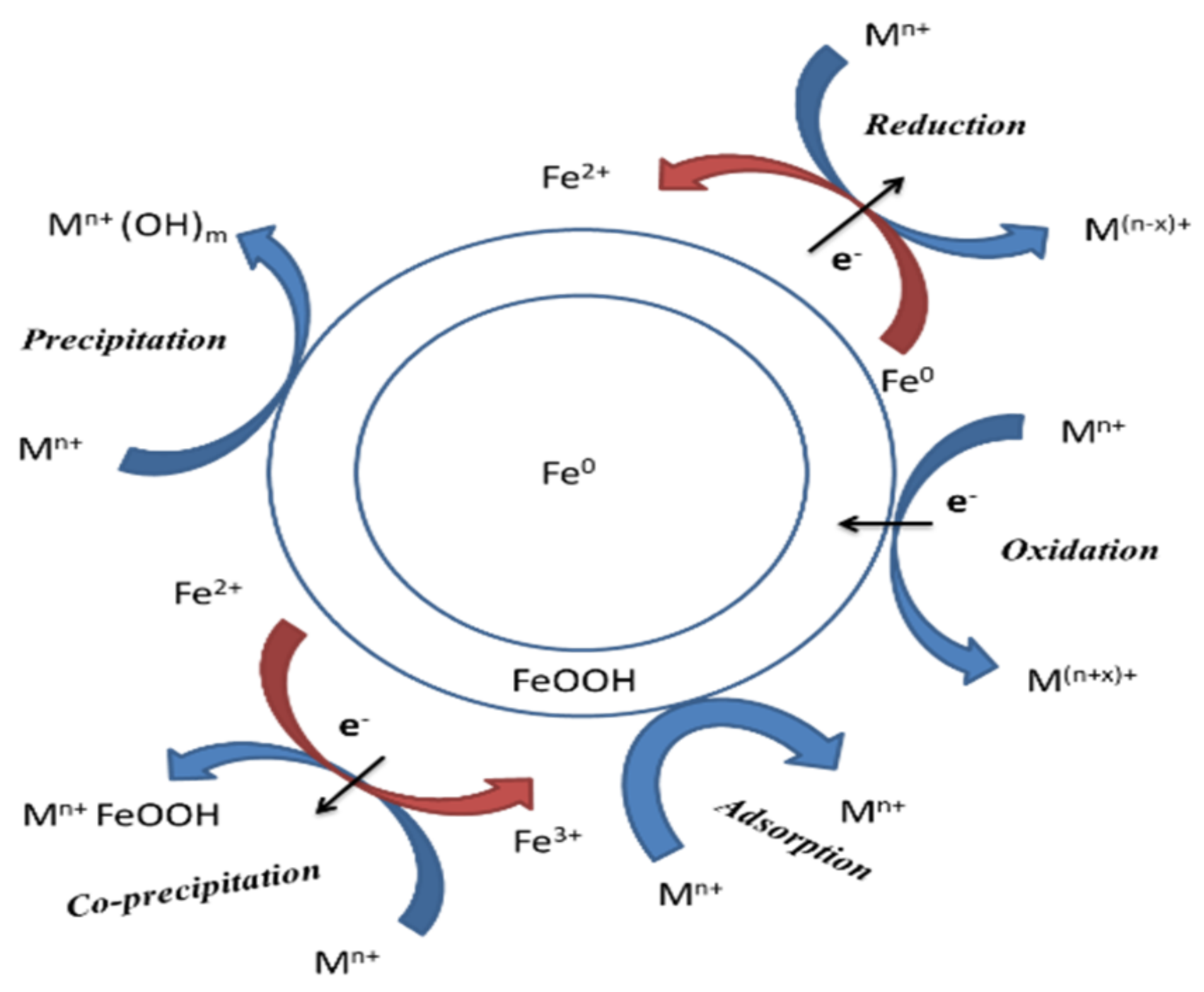
2.3. Zero-Valent Iron Nanoparticles
2.4. Magnetic Nanoparticles
2.4.1. Magnetic Iron-Oxide Nanoparticles
2.4.2. Magnetic Nanocomposites
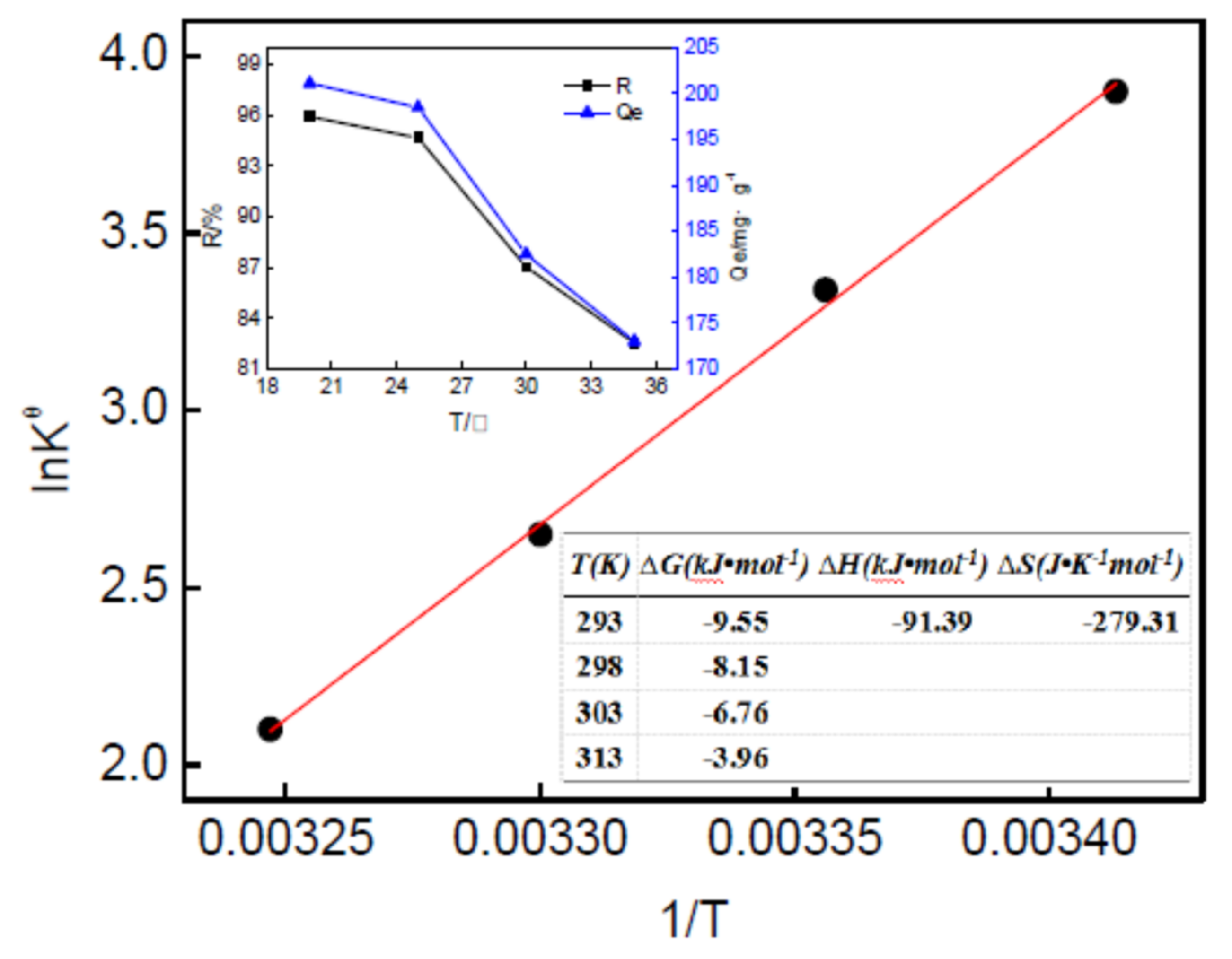
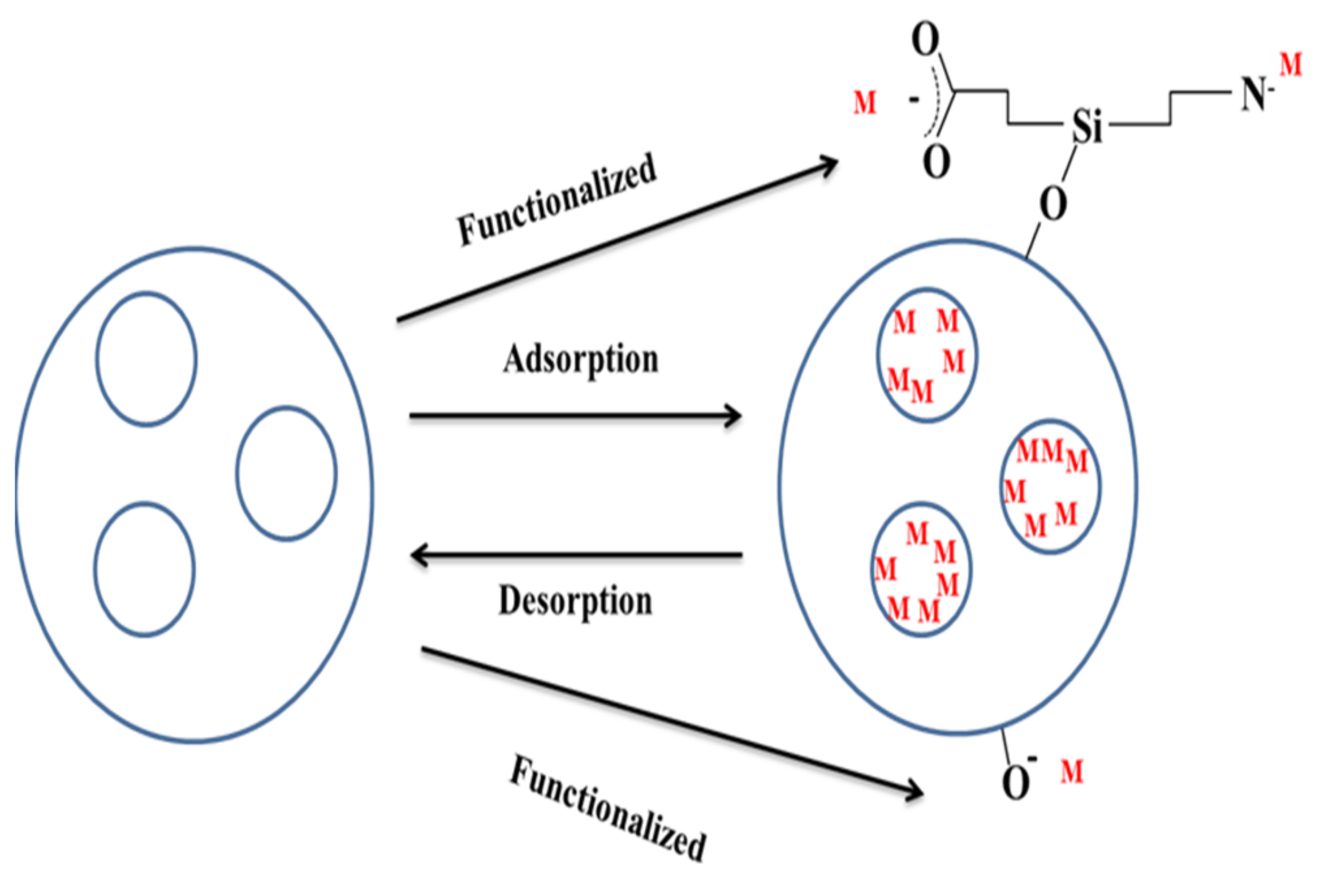
2.4.3. Reusability of Magnetic Nanoparticles
3. Nanomaterial Modified Bioelectrochemical Systems for Enhanced Power Production and the Remediation of Heavy Metals
3.1. Electrode Materials for Anode in BESs
3.1.1. Conventional Anode Materials
3.1.2. Nanomaterial Modified Anode Materials
3.2. Electrode Materials for Cathode in BESs
3.2.1. Conventional Cathode Materials
3.2.2. Nanomaterials Modified Cathode Materials
4. Conclusions and Future Perspectives
4.1. Roadmap of the Nanomaterial Based Adsorbents for the Extraction of Heavy Metals
4.2. Challenges and Opportunities for Large Scale Implementation of BESs for Heavy Metals Removal
4.3. Future Outlook for Nanomaterial Assisted BESs
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rogowska, J.; Cieszynska-Semenowicz, M.; Ratajczyk, W.; Wolska, L. Micropollutants in treated wastewater. Ambio 2020, 49, 487–503. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ali, H.; Khan, E.; Ilahi, I. Environmental Chemistry and Ecotoxicology of Hazardous Heavy Metals: Environmental Persistence, Toxicity, and Bioaccumulation. J. Chem. 2019, 2019, 6730305. [Google Scholar] [CrossRef] [Green Version]
- Gautam, P.K.; Gautam, R.; Banerjee, S.; Chattopadhyaya, M.; Pandey, J. Heavy metals in the environment: Fate, transport, toxicity and remediation technologies. In Heavy Metals; Nava Science Publishers: Plzen, Czech Republic, 2016; pp. 101–130. [Google Scholar]
- Vardhan, K.H.; Kumar, P.S.; Panda, R.C. A review on heavy metal pollution, toxicity and remedial measures: Current trends and future perspectives. J. Mol. Liq. 2019, 290, 111197. [Google Scholar] [CrossRef]
- Sharma, R.; Agrawal, M. Biological effects of heavy metals: An overview. J. Environ. Biol. Acad. Environ. Biol. India 2005, 26, 301–313. [Google Scholar]
- Tchounwou, P.B.; Yedjou, C.G.; Patlolla, A.K.; Sutton, D.J. Heavy metal toxicity and the environment. Exp. Suppl. 2012, 101, 133–164. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dominguez-Benetton, X.; Varia, J.C.; Pozo, G.; Modin, O.; Ter Heijne, A.; Fransaer, J.; Rabaey, K. Metal recovery by microbial electro-metallurgy. Prog. Mater. Sci. 2018, 94, 435–461. [Google Scholar] [CrossRef]
- Lesmana, S.O.; Febriana, N.; Soetaredjo, F.E.; Sunarso, J.; Ismadji, S. Studies on potential applications of biomass for the separation of heavy metals from water and wastewater. Biochem. Eng. J. 2009, 44, 19–41. [Google Scholar] [CrossRef]
- Morais, S.; Costa, F.; Pereira, M. Heavy Metals and Human Health. Environ. Health Emerg. Issues Pract. 2012, 10, 227–245. [Google Scholar]
- Tokar, E.J.; Benbrahim-Tallaa, L.; Waalkes, M.P. Metal ions in human cancer development. Met. Ions Life Sci 2011, 8, 375–401. [Google Scholar] [PubMed]
- Jomova, K.; Valko, M. Advances in metal-induced oxidative stress and human disease. Toxicology 2011, 283, 65–87. [Google Scholar] [CrossRef]
- Cruz-Olivares, J.; Martínez-Barrera, G.; Pérez-Alonso, C.; Barrera-Díaz, C.E.; Chaparro-Mercado, M.d.C.; Ureña-Núñez, F. Adsorption of Lead Ions from Aqueous Solutions Using Gamma Irradiated Minerals. J. Chem. 2016, 2016, 8782469. [Google Scholar] [CrossRef] [Green Version]
- Khulbe, K.C.; Matsuura, T. Removal of heavy metals and pollutants by membrane adsorption techniques. Appl. Water Sci. 2018, 8, 19. [Google Scholar] [CrossRef] [Green Version]
- Lee, I.H.; Kuan, Y.-C.; Chern, J.-M. Equilibrium and kinetics of heavy metal ion exchange. J. Chin. Inst. Chem. Eng. 2007, 38, 71–84. [Google Scholar] [CrossRef]
- Burakov, A.; Galunin, E.; Burakova, I.; Memetova, A.; Agarwal, S.; Tkachev, A.; Gupta, V. Adsorption of heavy metals on conventional and nanostructured materials for wastewater treatment purposes: A review. Ecotoxicol. Environ. Saf. 2017, 148, 702–712. [Google Scholar] [CrossRef] [PubMed]
- Abdullah, N.; Yusof, N.; Lau, W.J.; Jaafar, J.; Ismail, A. Recent trends of heavy metal removal from water/wastewater by membrane technologies. J. Ind. Eng. Chem. 2019, 76. [Google Scholar] [CrossRef]
- Kumar, V.; Shahi, S.; Singh, S. Bioremediation: An eco-sustainable approach for restoration of contaminated sites. In Microbial Bioprospecting for Sustainable Development; Springer: Berlin/Heidleberg, Germany, 2018; pp. 115–136. [Google Scholar]
- Donati, E.R.; Sani, R.K.; Goh, K.M.; Chan, K.-G. Editorial: Recent Advances in Bioremediation/Biodegradation by Extreme Microorganisms. Front. Microbiol. 2019, 10. [Google Scholar] [CrossRef] [Green Version]
- Hlihor, R.M.; Gavrilescu, M.; Tavares, T.; Favier, L.; Olivieri, G. Bioremediation: An Overview on Current Practices, Advances, and New Perspectives in Environmental Pollution Treatment. Biomed. Res. Int. 2017, 2017, 6327610. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jin, W.; Zhang, Y. Sustainable Electrochemical Extraction of Metal Resources from Waste Streams: From Removal to Recovery. ACS Sustain. Chem. Eng. 2020, 8, 4693–4707. [Google Scholar] [CrossRef]
- Jin, W.; Hu, M.; Hu, J. Selective and Efficient Electrochemical Recovery of Dilute Copper and Tellurium from Acidic Chloride Solutions. ACS Sustain. Chem. Eng. 2018, 6, 13378–13384. [Google Scholar] [CrossRef]
- Karbasi, M.; Alamdari, E.K.; Dehkordi, E.A. Electrochemical performance of PbCo composite anode during Zinc electrowinning. Hydrometallurgy 2019, 183, 51–59. [Google Scholar] [CrossRef]
- Tahoon, M.A.; Siddeeg, S.M.; Salem Alsaiari, N.; Mnif, W.; Ben Rebah, F. Effective Heavy Metals Removal from Water Using Nanomaterials: A Review. Processes 2020, 8, 645. [Google Scholar] [CrossRef]
- Babel, S.; Kurniawan, T.A. Low-cost adsorbents for heavy metals uptake from contaminated water: A review. J. Hazard. Mater. 2003, 97, 219–243. [Google Scholar] [CrossRef]
- Yan, H.; Li, H.; Tao, X.; Li, K.; Yang, H.; Li, A.; Xiao, S.; Cheng, R. Rapid Removal and Separation of Iron(II) and Manganese(II) from Micropolluted Water Using Magnetic Graphene Oxide. ACS Appl. Mater. Interfaces 2014, 6, 9871–9880. [Google Scholar] [CrossRef] [PubMed]
- Marsh, H.; Rodríguez-Reinoso, F. Chapter 8—Applicability of Activated Carbon. In Activated Carbon; Marsh, H., Rodríguez-Reinoso, F., Eds.; Elsevier Science Ltd.: Oxford, UK, 2006; pp. 383–453. [Google Scholar]
- Saleem, J.; Shahid, U.B.; Hijab, M.; Mackey, H.; McKay, G. Production and applications of activated carbons as adsorbents from olive stones. Biomass Convers. Biorefin. 2019, 9, 775–802. [Google Scholar] [CrossRef] [Green Version]
- Sarma, G.K.; Sen Gupta, S.; Bhattacharyya, K.G. Nanomaterials as versatile adsorbents for heavy metal ions in water: A review. Environ. Sci. Pollut. Res. 2019, 26, 6245–6278. [Google Scholar] [CrossRef] [PubMed]
- Theron, J.; Walker, J.A.; Cloete, T.E. Nanotechnology and Water Treatment: Applications and Emerging Opportunities. Crit. Rev. Microbiol. 2008, 34, 43–69. [Google Scholar] [CrossRef] [PubMed]
- Yaqoob, A.A.; Parveen, T.; Umar, K.; Mohamad Ibrahim, M.N. Role of Nanomaterials in the Treatment of Wastewater: A Review. Water 2020, 12, 495. [Google Scholar] [CrossRef] [Green Version]
- Parvin, F.; Rikta, S.Y.; Tareq, S.M. 8—Application of nanomaterials for the removal of heavy metal from wastewater. In Nanotechnology in Water and Wastewater Treatment; Ahsan, A., Ismail, A.F., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 137–157. [Google Scholar]
- Yang, J.; Hou, B.; Wang, J.; Tian, B.; Bi, J.; Wang, N.; Li, X.; Huang, X. Nanomaterials for the Removal of Heavy Metals from Wastewater. Nanomaterials 2019, 9, 424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lei, T.; Li, S.-J.; Jiang, F.; Ren, Z.-X.; Wang, L.-L.; Yang, X.-J.; Tang, L.-H.; Wang, S.-X. Adsorption of Cadmium Ions from an Aqueous Solution on a Highly Stable Dopamine-Modified Magnetic Nano-Adsorbent. Nanoscale Res. Lett 2019, 14, 352. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, P.; Kumar, P. Removal of cadmium (Cd-II) from aqueous solution using gas industry-based adsorbent. SN Appl. Sci. 2019, 1, 365. [Google Scholar] [CrossRef] [Green Version]
- Pyrzyńska, K.; Bystrzejewski, M. Comparative study of heavy metal ions sorption onto activated carbon, carbon nanotubes, and carbon-encapsulated magnetic nanoparticles. Colloids Surf. A Physicochem. Eng. Asp. 2010, 362, 102–109. [Google Scholar] [CrossRef]
- El-sayed, M.E.A. Nanoadsorbents for water and wastewater remediation. Sci. Total Environ. 2020, 739, 139903. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.C.; Rodrigues, D.F. Carbon-based nanomaterials for removal of chemical and biological contaminants from water: A review of mechanisms and applications. Carbon 2015, 91, 122–143. [Google Scholar] [CrossRef]
- Menezes, B.R.C.d.; Rodrigues, K.F.; Fonseca, B.C.d.S.; Ribas, R.G.; Montanheiro, T.L.d.A.; Thim, G.P. Recent advances in the use of carbon nanotubes as smart biomaterials. J. Mater. Chem. B 2019, 7, 1343–1360. [Google Scholar] [CrossRef] [PubMed]
- Mujawar, M.; Sahu, J.; Abdullah, E.; Natesan, J. Removal of Heavy Metals from Wastewater Using Carbon Nanotubes. Sep. Purif. Rev. 2014, 43, 311–338. [Google Scholar] [CrossRef]
- Bassyouni, M.; Mansi, A.E.; Elgabry, A.; Ibrahim, B.A.; Kassem, O.A.; Alhebeshy, R. Utilization of carbon nanotubes in removal of heavy metals from wastewater: A review of the CNTs’ potential and current challenges. Appl. Phys. A 2019, 126, 38. [Google Scholar] [CrossRef]
- Yu, G.; Lu, Y.; Guo, J.; Patel, M.; Bafana, A.; Wang, X.; Qiu, B.; Jeffryes, C.; Wei, S.; Guo, Z.; et al. Carbon nanotubes, graphene, and their derivatives for heavy metal removal. Adv. Compos. Hybrid. Mater. 2018, 1, 56–78. [Google Scholar] [CrossRef]
- Arora, B.; Attri, P. Carbon Nanotubes (CNTs): A Potential Nanomaterial for Water Purification. J. Compos. Sci. 2020, 4, 135. [Google Scholar] [CrossRef]
- Zhang, X.; Cui, H.; Gui, Y.; Tang, J. Mechanism and Application of Carbon Nanotube Sensors in SF(6) Decomposed Production Detection: A Review. Nanoscale Res. Lett 2017, 12, 177. [Google Scholar] [CrossRef] [Green Version]
- Alijani, H.; Shariatinia, Z. Synthesis of high growth rate SWCNTs and their magnetite cobalt sulfide nanohybrid as super-adsorbent for mercury removal. Chem. Eng. Res. Des. 2018, 129, 132–149. [Google Scholar] [CrossRef]
- Gupta, A.; Vidyarthi, S.R.; Sankararamakrishnan, N. Enhanced sorption of mercury from compact fluorescent bulbs and contaminated water streams using functionalized multiwalled carbon nanotubes. J. Hazard. Mater. 2014, 274, 132–144. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Bhatiya, D.; Murthy, C.N. Metal Removal Studies by Composite Membrane of Polysulfone and Functionalized Single-Walled Carbon Nanotubes. Sep. Sci. Technol. 2015, 50, 421–429. [Google Scholar] [CrossRef]
- Robati, D. Pseudo-second-order kinetic equations for modeling adsorption systems for removal of lead ions using multi-walled carbon nanotube. J. Nanostruct. Chem. 2013, 3, 55. [Google Scholar] [CrossRef] [Green Version]
- Farghali, A.A.; Abdel Tawab, H.A.; Abdel Moaty, S.A.; Khaled, R. Functionalization of acidified multi-walled carbon nanotubes for removal of heavy metals in aqueous solutions. J. Nanostruct. Chem. 2017, 7, 101–111. [Google Scholar] [CrossRef] [Green Version]
- Elsehly, E.M.I.; Chechenin, N.G.; Bukunov, K.A.; Makunin, A.V.; Priselkova, A.B.; Vorobyeva, E.A.; Motaweh, H.A. Removal of iron and manganese from aqueous solutions using carbon nanotube filters. Water Supply 2015, 16, 347–353. [Google Scholar] [CrossRef]
- Mallakpour, S.; Khadem, E. 8—Carbon nanotubes for heavy metals removal. In Composite Nanoadsorbents; Kyzas, G.Z., Mitropoulos, A.C., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 181–210. [Google Scholar]
- Faur-Brasquet, C.; Kadirvelu, K.; Le Cloirec, P. Removal of metal ions from aqueous solution by adsorption onto activated carbon cloths: Adsorption competition with organic matter. Carbon 2002, 40, 2387–2392. [Google Scholar] [CrossRef]
- Hur, J.; Shin, J.; Yoo, J.; Seo, Y.-S. Competitive Adsorption of Metals onto Magnetic Graphene Oxide: Comparison with Other Carbonaceous Adsorbents. Sci. World J. 2015, 2015, 836287. [Google Scholar] [CrossRef] [PubMed]
- Borji, H.; Ayoub, G.M.; Bilbeisi, R.; Nassar, N.; Malaeb, L. How Effective Are Nanomaterials for the Removal of Heavy Metals from Water and Wastewater? Water Air Soil Pollut. 2020, 231, 330. [Google Scholar] [CrossRef]
- Rodríguez, C.; Briano, S.; Leiva, E. Increased Adsorption of Heavy Metal Ions in Multi-Walled Carbon Nanotubes with Improved Dispersion Stability. Molecules 2020, 25, 3106. [Google Scholar] [CrossRef]
- Amin, M.T.; Alazba, A.A.; Manzoor, U. A Review of Removal of Pollutants from Water/Wastewater Using Different Types of Nanomaterials. Adv. Mater. Sci. Eng. 2014, 2014, 825910. [Google Scholar] [CrossRef] [Green Version]
- Chowdhury, S.; Balasubramanian, R. Recent advances in the use of graphene-family nanoadsorbents for removal of toxic pollutants from wastewater. Adv. Colloid Interface Sci. 2014, 204, 35–56. [Google Scholar] [CrossRef] [PubMed]
- Ali, I.; Basheer, A.A.; Mbianda, X.Y.; Burakov, A.; Galunin, E.; Burakova, I.; Mkrtchyan, E.; Tkachev, A.; Grachev, V. Graphene based adsorbents for remediation of noxious pollutants from wastewater. Environ. Int. 2019, 127, 160–180. [Google Scholar] [CrossRef]
- Woo, Y.C.; Kim, S.-H.; Shon, H.K.; Tijing, L.D. Introduction: Membrane Desalination Today, Past, and Future. In Current Trends and Future Developments on (Bio-) Membranes; Basile, A., Curcio, E., Inamuddin, Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. xxv–xlvi. [Google Scholar]
- Smith, A.T.; LaChance, A.M.; Zeng, S.; Liu, B.; Sun, L. Synthesis, properties, and applications of graphene oxide/reduced graphene oxide and their nanocomposites. Nano Mater. Sci. 2019, 1, 31–47. [Google Scholar] [CrossRef]
- Azizighannad, S.; Mitra, S. Stepwise Reduction of Graphene Oxide (GO) and Its Effects on Chemical and Colloidal Properties. Sci. Rep. 2018, 8, 10083. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Wang, J. The application of graphene-based materials for the removal of heavy metals and radionuclides from water and wastewater. Crit. Rev. Environ. Sci. Technol. 2017, 47, 1042–1105. [Google Scholar] [CrossRef]
- Zhao, G.; Li, J.; Ren, X.; Chen, C.; Wang, X. Few-Layered Graphene Oxide Nanosheets As Superior Sorbents for Heavy Metal Ion Pollution Management. Environ. Sci. Technol. 2011, 45, 10454–10462. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Yuan, X.; Wu, Y.; Huang, H.; Zeng, G.; Liu, Y.; Wang, X.; Lin, N.; Qi, Y. Adsorption characteristics and behaviors of graphene oxide for Zn(II) removal from aqueous solution. Appl. Surf. Sci. 2013, 279, 432–440. [Google Scholar] [CrossRef]
- Guo, T.; Bulin, C.; Li, B.; Zhao, Z.; Yu, H.; Sun, H.; Ge, X.; Xing, R.; Zhang, B. Efficient removal of aqueous Pb(II) using partially reduced graphene oxide-Fe3O4. Adsorpt. Sci. Technol. 2017, 36, 1031–1048. [Google Scholar] [CrossRef] [Green Version]
- Tabish, T.A.; Memon, F.A.; Gomez, D.E.; Horsell, D.W.; Zhang, S. A facile synthesis of porous graphene for efficient water and wastewater treatment. Sci. Rep. 2018, 8, 1817. [Google Scholar] [CrossRef]
- Zhang, C.-Z.; Chen, B.; Bai, Y.; Xie, J. A new functionalized reduced graphene oxide adsorbent for removing heavy metal ions in water via coordination and ion exchange. Sep. Sci. Technol. 2018, 53, 2896–2905. [Google Scholar] [CrossRef]
- Awad, F.S.; AbouZied, K.M.; Abou El-Maaty, W.M.; El-Wakil, A.M.; Samy El-Shall, M. Effective removal of mercury(II) from aqueous solutions by chemically modified graphene oxide nanosheets. Arab. J. Chem. 2020, 13, 2659–2670. [Google Scholar] [CrossRef]
- Zheng, S.; Hao, L.; Zhang, L.; Wang, K.; Zheng, W.; Wang, X.; Zhou, X.; Li, W.; Zhang, L. Tea Polyphenols Functionalized and Reduced Graphene Oxide-ZnO Composites for Selective Pb(2+) Removal and Enhanced Antibacterial Activity. J. Biomed. Nanotechnol. 2018, 14, 1263–1276. [Google Scholar] [CrossRef]
- Arshad, F.; Selvaraj, M.; Zain, J.; Banat, F.; Haija, M.A. Polyethylenimine modified graphene oxide hydrogel composite as an efficient adsorbent for heavy metal ions. Sep. Purif. Technol. 2019, 209, 870–880. [Google Scholar] [CrossRef]
- Chauke, V.P.; Maity, A.; Chetty, A. High-performance towards removal of toxic hexavalent chromium from aqueous solution using graphene oxide-alpha cyclodextrin-polypyrrole nanocomposites. J. Mol. Liq. 2015, 211, 71–77. [Google Scholar] [CrossRef]
- Hadi Najafabadi, H.; Irani, M.; Roshanfekr Rad, L.; Heydari Haratameh, A.; Haririan, I. Removal of Cu2+, Pb2+ and Cr6+ from aqueous solutions using a chitosan/graphene oxide composite nanofibrous adsorbent. RSC Adv. 2015, 5, 16532–16539. [Google Scholar] [CrossRef]
- Mahmoud, M.E.; Fekry, N.A.; El-Latif, M.M.A. Nanocomposites of nanosilica-immobilized-nanopolyaniline and crosslinked nanopolyaniline for removal of heavy metals. Chem. Eng. J. 2016, 304, 679–691. [Google Scholar] [CrossRef]
- Nguyen, T.T.; Ma, H.T.; Avti, P.; Bashir, M.J.K.; Ng, C.A.; Wong, L.Y.; Jun, H.K.; Ngo, Q.M.; Tran, N.Q. Adsorptive Removal of Iron Using SiO2 Nanoparticles Extracted from Rice Husk Ash. J. Anal. Methods Chem. 2019, 2019, 6210240. [Google Scholar] [CrossRef] [Green Version]
- Najafi, M.; Yousefi, Y.; Rafati, A.A. Synthesis, characterization and adsorption studies of several heavy metal ions on amino-functionalized silica nano hollow sphere and silica gel. Sep. Purif. Technol. 2012, 85, 193–205. [Google Scholar] [CrossRef]
- Kotsyuda, S.S.; Tomina, V.V.; Zub, Y.L.; Furtat, I.M.; Lebed, A.P.; Vaclavikova, M.; Melnyk, I.V. Bifunctional silica nanospheres with 3-aminopropyl and phenyl groups. Synthesis approach and prospects of their applications. Appl. Surf. Sci. 2017, 420, 782–791. [Google Scholar] [CrossRef]
- Liu, A.M.; Hidajat, K.; Kawi, S.; Zhao, D.Y. A new class of hybrid mesoporous materials with functionalized organic monolayers for selective adsorption of heavy metal ions. Chem. Commun. 2000, 1145–1146. [Google Scholar] [CrossRef]
- Li, X.; Han, C.; Zhu, W.; Ma, W.; Luo, Y.; Zhou, Y.; Yu, J.; Wei, K. Cr(VI) Removal from Aqueous by Adsorption on Amine-Functionalized Mesoporous Silica Prepared from Silica Fume. J. Chem. 2014, 2014, 765856. [Google Scholar] [CrossRef]
- Li, R.; Zhang, L.; Wang, P. Rational design of nanomaterials for water treatment. Nanoscale 2015, 7, 17167–17194. [Google Scholar] [CrossRef] [Green Version]
- Huang, X.; Wang, L.; Chen, J.; Jiang, C.; Wu, S.; Wang, H. Effective removal of heavy metals with amino-functionalized silica gel in tea polyphenol extracts. J. Food Meas. Charact. 2020, 14, 2134–2144. [Google Scholar] [CrossRef]
- Wang, H.; Kang, J.; Liu, H.; Qu, J. Preparation of organically functionalized silica gel as adsorbent for copper ion adsorption. J. Environ. Sci. 2009, 21, 1473–1479. [Google Scholar] [CrossRef]
- Wieszczycka, K.; Filipowiak, K.; Wojciechowska, I.; Buchwald, T.; Siwińska-Ciesielczyk, K.; Strzemiecka, B.; Jesionowski, T.; Voelkel, A. Novel highly efficient ionic liquid-functionalized silica for toxic metals removal. Sep. Purif. Technol. 2021, 265, 118483. [Google Scholar] [CrossRef]
- Crane, R.A.; Scott, T.B. Nanoscale zero-valent iron: Future prospects for an emerging water treatment technology. J. Hazard. Mater. 2012, 211–212, 112–125. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, N.R.; Shankar, P.A.; Bandyopadhyaya, R. Plasma treated activated carbon impregnated with silver nanoparticles for improved antibacterial effect in water disinfection. Carbon 2013, 57, 1–10. [Google Scholar] [CrossRef]
- O’Carroll, D.; Sleep, B.; Krol, M.; Boparai, H.; Kocur, C. Nanoscale zero valent iron and bimetallic particles for contaminated site remediation. Adv. Water Resour. 2013, 51, 104–122. [Google Scholar] [CrossRef]
- Phenrat, T.; Lowry, G.V.; Babakhani, P. Nanoscale zerovalent iron (NZVI) for environmental decontamination: A brief history of 20 years of research and field-scale application. In Nanoscale Zerovalent Iron Particles for Environmental Restoration: From Fundamental Science to Field Scale Engineering Applications; Phenrat, T., Lowry, G.V., Eds.; Springer International Publishing: Cham, Switzerland, 2019; pp. 1–43. [Google Scholar]
- Liu, T.; Wang, Z.-L.; Sun, Y. Manipulating the morphology of nanoscale zero-valent iron on pumice for removal of heavy metals from wastewater. Chem. Eng. J. 2015, 263, 55–61. [Google Scholar] [CrossRef]
- Zhang, Z.; Hao, Z.W.; Liu, W.L.; Xu, X.H. Synchronous treatment of heavy metal ions and nitrate by zero-valent iron. Huan Jing Ke Xue 2009, 30, 775–779. [Google Scholar]
- Seyedi, S.M.; Rabiee, H.; Shahabadi, S.M.S.; Borghei, S.M. Synthesis of Zero-Valent Iron Nanoparticles Via Electrical Wire Explosion for Efficient Removal of Heavy Metals. CLEAN Soil Air Water 2017, 45, 1600139. [Google Scholar] [CrossRef]
- Guo, J.J.; Fan, M.D. Progress of Removing Heavy Metals by Zero Valent Iron Nanoparticles from Wastewater. Adv. Mater. Res. 2013, 726–731, 2563–2566. [Google Scholar] [CrossRef]
- Sarathy, V.; Tratnyek, P.G.; Nurmi, J.T.; Baer, D.R.; Amonette, J.E.; Chun, C.L.; Penn, R.L.; Reardon, E.J. Aging of Iron Nanoparticles in Aqueous Solution: Effects on Structure and Reactivity. J. Phys. Chem. C 2008, 112, 2286–2293. [Google Scholar] [CrossRef]
- Fu, F.; Dionysiou, D.D.; Liu, H. The use of zero-valent iron for groundwater remediation and wastewater treatment: A review. J. Hazard. Mater. 2014, 267, 194–205. [Google Scholar] [CrossRef]
- Huang, D.-L.; Chen, G.-M.; Zeng, G.-M.; Xu, P.; Yan, M.; Lai, C.; Zhang, C.; Li, N.-J.; Cheng, M.; He, X.-X.; et al. Synthesis and Application of Modified Zero-Valent Iron Nanoparticles for Removal of Hexavalent Chromium from Wastewater. Water Air Soil Pollut. 2015, 226, 375. [Google Scholar] [CrossRef]
- Su, Y.; Adeleye, A.S.; Huang, Y.; Sun, X.; Dai, C.; Zhou, X.; Zhang, Y.; Keller, A.A. Simultaneous removal of cadmium and nitrate in aqueous media by nanoscale zerovalent iron (nZVI) and Au doped nZVI particles. Water Res. 2014, 63, 102–111. [Google Scholar] [CrossRef] [Green Version]
- Tamjidi, S.; Esmaeili, H.; Kamyab Moghadas, B. Application of magnetic adsorbents for removal of heavy metals from wastewater: A review study. Mater. Res. Express 2019, 6, 102004. [Google Scholar] [CrossRef]
- Vojoudi, H.; Badiei, A.; Bahar, S.; Mohammadi Ziarani, G.; Faridbod, F.; Ganjali, M.R. A new nano-sorbent for fast and efficient removal of heavy metals from aqueous solutions based on modification of magnetic mesoporous silica nanospheres. J. Magn. Magn. Mater. 2017, 441, 193–203. [Google Scholar] [CrossRef]
- Etale, A.; Tutu, H.; Drake, D.C. The effect of silica and maghemite nanoparticles on remediation of Cu(II)-, Mn(II)- and U(VI)-contaminated water by Acutodesmus sp. J. Appl. Phycol. 2016, 28, 251–260. [Google Scholar] [CrossRef]
- Tuutijärvi, T.; Lu, J.; Sillanpää, M.; Chen, G. As(V) adsorption on maghemite nanoparticles. J. Hazard. Mater. 2009, 166, 1415–1420. [Google Scholar] [CrossRef] [PubMed]
- Akhbarizadeh, R.; Shayestefar, M.R.; Darezereshki, E. Competitive Removal of Metals from Wastewater by Maghemite Nanoparticles: A Comparison Between Simulated Wastewater and AMD. Mine Water Environ. 2014, 33, 89–96. [Google Scholar] [CrossRef]
- Madrakian, T.; Afkhami, A.; Zadpour, B.; Ahmadi, M. New synthetic mercaptoethylamino homopolymer-modified maghemite nanoparticles for effective removal of some heavy metal ions from aqueous solution. J. Ind. Eng. Chem. 2015, 21, 1160–1166. [Google Scholar] [CrossRef]
- Tzabar, N.; ter Brake, H.J.M. Adsorption isotherms and Sips models of nitrogen, methane, ethane, and propane on commercial activated carbons and polyvinylidene chloride. Adsorption 2016, 22, 901–914. [Google Scholar] [CrossRef] [Green Version]
- Fato, F.P.; Li, D.-W.; Zhao, L.-J.; Qiu, K.; Long, Y.-T. Simultaneous Removal of Multiple Heavy Metal Ions from River Water Using Ultrafine Mesoporous Magnetite Nanoparticles. ACS Omega 2019, 4, 7543–7549. [Google Scholar] [CrossRef] [Green Version]
- Shipley, H.J.; Engates, K.E.; Grover, V.A. Removal of Pb(II), Cd(II), Cu(II), and Zn(II) by hematite nanoparticles: Effect of sorbent concentration, pH, temperature, and exhaustion. Environ. Sci. Pollut. Res. 2013, 20, 1727–1736. [Google Scholar] [CrossRef]
- Giraldo, L.; Erto, A.; Moreno-Piraján, J. Magnetite nanoparticles for removal of heavy metals from aqueous solutions: Synthesis and characterization. Adsorption 2013, 19. [Google Scholar] [CrossRef]
- Feng, L.; Cao, M.; Ma, X.; Zhu, Y.; Hu, C. Superparamagnetic high-surface-area Fe3O4 nanoparticles as adsorbents for arsenic removal. J. Hazard. Mater. 2012, 217–218, 439–446. [Google Scholar] [CrossRef]
- Yu, X.; Tong, S.; Ge, M.; Zuo, J.; Cao, C.; Song, W. One-step synthesis of magnetic composites of cellulose@iron oxide nanoparticles for arsenic removal. J. Mater. Chem. A 2013, 1, 959–965. [Google Scholar] [CrossRef]
- Ahmed, M.A.; Ali, S.M.; El-Dek, S.I.; Galal, A. Magnetite–hematite nanoparticles prepared by green methods for heavy metal ions removal from water. Mater. Sci. Eng. B 2013, 178, 744–751. [Google Scholar] [CrossRef]
- Hu, J.; Chen, G.; Lo, I.M. Removal and recovery of Cr(VI) from wastewater by maghemite nanoparticles. Water Res. 2005, 39, 4528–4536. [Google Scholar] [CrossRef] [PubMed]
- Watts, M.P.; Coker, V.S.; Parry, S.A.; Pattrick, R.A.; Thomas, R.A.; Kalin, R.; Lloyd, J.R. Biogenic nano-magnetite and nano-zero valent iron treatment of alkaline Cr(VI) leachate and chromite ore processing residue. Appl. Geochem. 2015, 54, 27–42. [Google Scholar] [CrossRef]
- Shi, J.; Li, H.; Lu, H.; Zhao, X. Use of Carboxyl Functional Magnetite Nanoparticles as Potential Sorbents for the Removal of Heavy Metal Ions from Aqueous Solution. J. Chem. Eng. Data 2015, 60, 2035–2041. [Google Scholar] [CrossRef]
- Tu, Y.-J.; You, C.-F.; Chang, C.-K. Kinetics and thermodynamics of adsorption for Cd on green manufactured nano-particles. J. Hazard. Mater. 2012, 235–236, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Panneerselvam, P.; Morad, N.; Tan, K.A. Magnetic nanoparticle (Fe3O4) impregnated onto tea waste for the removal of nickel(II) from aqueous solution. J. Hazard. Mater. 2011, 186, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Karvelas, E.; Liosis, C.; Benos, L.; Karakasidis, T.; Sarris, I. Micromixing Efficiency of Particles in Heavy Metal Removal Processes under Various Inlet Conditions. Water 2019, 11, 1135. [Google Scholar] [CrossRef] [Green Version]
- Mourdikoudis, S.; Kostopoulou, A.; LaGrow, A.P. Magnetic Nanoparticle Composites: Synergistic Effects and Applications. Adv. Sci. 2021, 8, 2004951. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Shi, X.; Zhang, Y.; Gu, W.; Li, B.; Xian, Y. Synthesis of water-soluble magnetic graphene nanocomposites for recyclable removal of heavy metal ions. J. Mater. Chem. A 2013, 1, 1745–1753. [Google Scholar] [CrossRef]
- Elmi, F.; Hosseini, T.; Taleshi, M.S.; Taleshi, F. Kinetic and thermodynamic investigation into the lead adsorption process from wastewater through magnetic nanocomposite Fe3O4/CNT. Nanotechnol. Environ. Eng. 2017, 2, 13. [Google Scholar] [CrossRef] [Green Version]
- Takafuji, M.; Ide, S.; Ihara, H.; Xu, Z. Preparation of Poly(1-vinylimidazole)-Grafted Magnetic Nanoparticles and Their Application for Removal of Metal Ions. Chem. Mater. 2004, 16, 1977–1983. [Google Scholar] [CrossRef]
- Faghihian, H.; Moayed, M.; Firooz, A.; Iravani, M. Evaluation of a new magnetic zeolite composite for removal of Cs+ and Sr2+ from aqueous solutions: Kinetic, equilibrium and thermodynamic studies. C. R. Chim. 2014, 17, 108–117. [Google Scholar] [CrossRef]
- Ge, L.; Wang, W.; Peng, Z.; Tan, F.; Wang, X.; Chen, J.; Qiao, X. Facile fabrication of Fe@MgO magnetic nanocomposites for efficient removal of heavy metal ion and dye from water. Powder Technol. 2018, 326, 393–401. [Google Scholar] [CrossRef]
- Samrot, A.V.; Sahithya, C.S.; Selvarani, A.J.; Pachiyappan, S.; Kumar, S.S. Surface-Engineered Super-Paramagnetic Iron Oxide Nanoparticles For Chromium Removal. Int. J. Nanomed. 2019, 14, 8105–8119. [Google Scholar] [CrossRef] [Green Version]
- Rauwel, P.; Uhl, W.; Rauwel, E. Editorial for the Special Issue on ‘Application and Behavior of Nanomaterials in Water Treatment’. Nanomaterials 2019, 9, 880. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, H.; Hodges, C.S.; Mishra, P.K.; Yoon, J.Y.; Hunter, T.N.; Lee, J.W.; Harbottle, D. Bio-Inspired Preparation of Clay–Hexacyanoferrate Composite Hydrogels as Super Adsorbents for Cs+. ACS Appl. Mater. Interfaces 2020, 12, 33173–33185. [Google Scholar] [CrossRef]
- Rauwel, P.; Rauwel, E. Towards the Extraction of Radioactive Cesium-137 from Water via Graphene/CNT and Nanostructured Prussian Blue Hybrid Nanocomposites: A Review. Nanomaterials 2019, 9, 682. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Perumal, S.; Atchudan, R.; Edison, T.N.J.I.; Babu, R.S.; Karpagavinayagam, P.; Vedhi, C. A Short Review on Recent Advances of Hydrogel-Based Adsorbents for Heavy Metal Ions. Metals 2021, 11, 864. [Google Scholar] [CrossRef]
- Nicola, R.; Costişor, O.; Ciopec, M.; Negrea, A.; Lazău, R.; Ianăşi, C.; Picioruş, E.-M.; Len, A.; Almásy, L.; Szerb, E.I.; et al. Silica-Coated Magnetic Nanocomposites for Pb2+ Removal from Aqueous Solution. Appl. Sci. 2020, 10, 2726. [Google Scholar] [CrossRef] [Green Version]
- Ahmad, N.; Sereshti, H.; Mousazadeh, M.; Rashidi Nodeh, H.; Kamboh, M.A.; Mohamad, S. New magnetic silica-based hybrid organic-inorganic nanocomposite for the removal of lead(II) and nickel(II) ions from aqueous solutions. Mater. Chem. Phys. 2019, 226, 73–81. [Google Scholar] [CrossRef]
- Culita, D.C.; Simonescu, C.M.; Patescu, R.-E.; Dragne, M.; Stanica, N.; Oprea, O. o-Vanillin functionalized mesoporous silica—Coated magnetite nanoparticles for efficient removal of Pb(II) from water. J. Solid State Chem. 2016, 238, 311–320. [Google Scholar] [CrossRef]
- Hu, H.; Wang, Z.; Pan, L. Synthesis of monodisperse Fe3O4@silica core–shell microspheres and their application for removal of heavy metal ions from water. J. Alloy. Compd. 2010, 492, 656–661. [Google Scholar] [CrossRef]
- Morsi, R.E.; Al-Sabagh, A.M.; Moustafa, Y.M.; ElKholy, S.G.; Sayed, M.S. Polythiophene modified chitosan/magnetite nanocomposites for heavy metals and selective mercury removal. Egypt. J. Pet. 2018, 27, 1077–1085. [Google Scholar] [CrossRef]
- Suleiman, J.S.; Hu, B.; Peng, H.; Huang, C. Separation/preconcentration of trace amounts of Cr, Cu and Pb in environmental samples by magnetic solid-phase extraction with Bismuthiol-II-immobilized magnetic nanoparticles and their determination by ICP-OES. Talanta 2009, 77, 1579–1583. [Google Scholar] [CrossRef]
- Zhang, W.; An, Y.; Li, S.; Liu, Z.; Chen, Z.; Ren, Y.; Wang, S.; Zhang, X.; Wang, X. Enhanced heavy metal removal from an aqueous environment using an eco-friendly and sustainable adsorbent. Sci. Rep. 2020, 10, 16453. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Hu, Q.; Fang, Z.; Zhang, X.; Zhang, B. Magnetic Chitosan Nanocomposites: A Useful Recyclable Tool for Heavy Metal Ion Removal. Langmuir 2009, 25, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Awut, T.; Liu, B.; Ma, Y.; Wang, T.; Nurulla, I. Functionalized magnetic Fe3O4 nanoparticles for removal of heavy metal ions from aqueous solutions. e-Polymers 2016, 16, 313–322. [Google Scholar] [CrossRef]
- Huang, Z.-n.; Wang, X.-l.; Yang, D.-s. Adsorption of Cr(VI) in wastewater using magnetic multi-wall carbon nanotubes. Water Sci. Eng. 2015, 8, 226–232. [Google Scholar] [CrossRef] [Green Version]
- Rivera, F.L.; Palomares, F.J.; Herrasti, P.; Mazario, E. Improvement in Heavy Metal Removal from Wastewater Using an External Magnetic Inductor. Nanomaterials 2019, 9, 1508. [Google Scholar] [CrossRef] [Green Version]
- Tao, S.; Wang, C.; Ma, W.; Wu, S.; Meng, C. Designed multifunctionalized magnetic mesoporous microsphere for sequential sorption of organic and inorganic pollutants. Microporous Mesoporous Mater. 2012, 147, 295–301. [Google Scholar] [CrossRef]
- Zheng, T.; Li, J.; Ji, Y.; Zhang, W.; Fang, Y.; Xin, F.; Dong, W.; Wei, P.; Ma, J.; Jiang, M. Progress and Prospects of Bioelectrochemical Systems: Electron Transfer and Its Applications in the Microbial Metabolism. Front. Bioeng. Biotechnol. 2020, 8, 10. [Google Scholar] [CrossRef] [Green Version]
- Bajracharya, S.; Sharma, M.; Mohanakrishna, G.; Dominguez Benneton, X.; Strik, D.P.B.T.B.; Sarma, P.M.; Pant, D. An overview on emerging bioelectrochemical systems (BESs): Technology for sustainable electricity, waste remediation, resource recovery, chemical production and beyond. Renew. Energy 2016, 98, 153–170. [Google Scholar] [CrossRef]
- Sukrampal; Kumar, R.; Patil, S.A. 3—Removal of heavy metals using bioelectrochemical systems. In Integrated Microbial Fuel Cells for Wastewater Treatment; Abbassi, R., Yadav, A.K., Khan, F., Garaniya, V., Eds.; Butterworth-Heinemann: Oxford, UK, 2020; pp. 49–71. [Google Scholar]
- Rabaey, K.; Lissens, G.; Siciliano, S.D.; Verstraete, W. A microbial fuel cell capable of converting glucose to electricity at high rate and efficiency. Biotechnol. Lett. 2003, 25, 1531–1535. [Google Scholar] [CrossRef]
- Kumar, R.; Yadav, S.; Patil, S.A. Bioanode-Assisted Removal of Hg2+ at the Cathode of Microbial Fuel Cells. J. Hazard. Toxic Radioact. Waste 2020, 24, 04020034. [Google Scholar] [CrossRef]
- Zhang, Y.; Sun, J.; Hu, Y.; Li, S.; Xu, Q. Bio-cathode materials evaluation in microbial fuel cells: A comparison of graphite felt, carbon paper and stainless steel mesh materials. Int. J. Hydrogen Energy 2012, 37, 16935–16942. [Google Scholar] [CrossRef]
- Yang, Q.; Liang, S.; Liu, J.; Lv, J.; Feng, Y. Analysis of Anodes of Microbial Fuel Cells When Carbon Brushes Are Preheated at Different Temperatures. Catalysts 2017, 7, 312. [Google Scholar] [CrossRef] [Green Version]
- He, Y.-R.; Xiao, X.; Li, W.-W.; Sheng, G.-P.; Yan, F.-F.; Yu, H.-Q.; Yuan, H.; Wu, L.-J. Enhanced electricity production from microbial fuel cells with plasma-modified carbon paper anode. Phys. Chem. Chem. Phys. 2012, 14, 9966–9971. [Google Scholar] [CrossRef] [PubMed]
- Ezziat, L.; Elabed, A.; Ibnsouda, S.; El Abed, S. Challenges of Microbial Fuel Cell Architecture on Heavy Metal Recovery and Removal From Wastewater. Front. Energy Res. 2019, 7. [Google Scholar] [CrossRef]
- Logan, B.; Cheng, S.; Watson, V.; Estadt, G. Graphite Fiber Brush Anodes for Increased Power Production in Air-Cathode Microbial Fuel Cells. Environ. Sci. Technol. 2007, 41, 3341–3346. [Google Scholar] [CrossRef]
- Fan, Y.; Liu, H. Materials for Low-Temperature Fuel Cells; Wiley: Hoboken, NJ, USA, 2014; pp. 145–166. [Google Scholar]
- Savla, N.; Anand, R.; Pandit, S.; Prasad, R. Utilization of Nanomaterials as Anode Modifiers for Improving Microbial Fuel Cells Performance. J. Renew. Mater. 2020, 8, 1581–1605. [Google Scholar] [CrossRef]
- Nakamura, R.; Kai, F.; Okamoto, A.; Newton, G.J.; Hashimoto, K. Self-Constructed Electrically Conductive Bacterial Networks. Angew. Chem. Int. Ed. 2009, 48, 508–511. [Google Scholar] [CrossRef]
- Zhao, C.; Wang, Y.; Shi, F.; Zhang, J.; Zhu, J.-J. High biocurrent generation in Shewanella-inoculated microbial fuel cells using ionic liquid functionalized graphene nanosheets as an anode. Chem. Commun. 2013, 49, 6668–6670. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Gai, P.; Liu, C.; Wang, X.; Xu, H.; Zhang, J.; Zhu, J.-J. Polyaniline networks grown on graphene nanoribbonscoated carbon paper with a synergistic effect for highperformance microbial fuel cells. J. Mater. Chem. A. 2013, 1, 12587. [Google Scholar] [CrossRef]
- Hou, J.; Liu, Z.; Zhang, P. A new method for fabrication of graphene/polyaniline nanocomplex modified microbial fuel cell anodes. J. Power Sources 2013, 224, 139–144. [Google Scholar] [CrossRef]
- Dessie, Y.; Tadesse, S.; Eswaramoorthy, R.; Adimasu, Y. Biosynthesized α-MnO2-based polyaniline binary composite as efficient bioanode catalyst for high-performance microbial fuel cell. All Life 2021, 14, 541–568. [Google Scholar] [CrossRef]
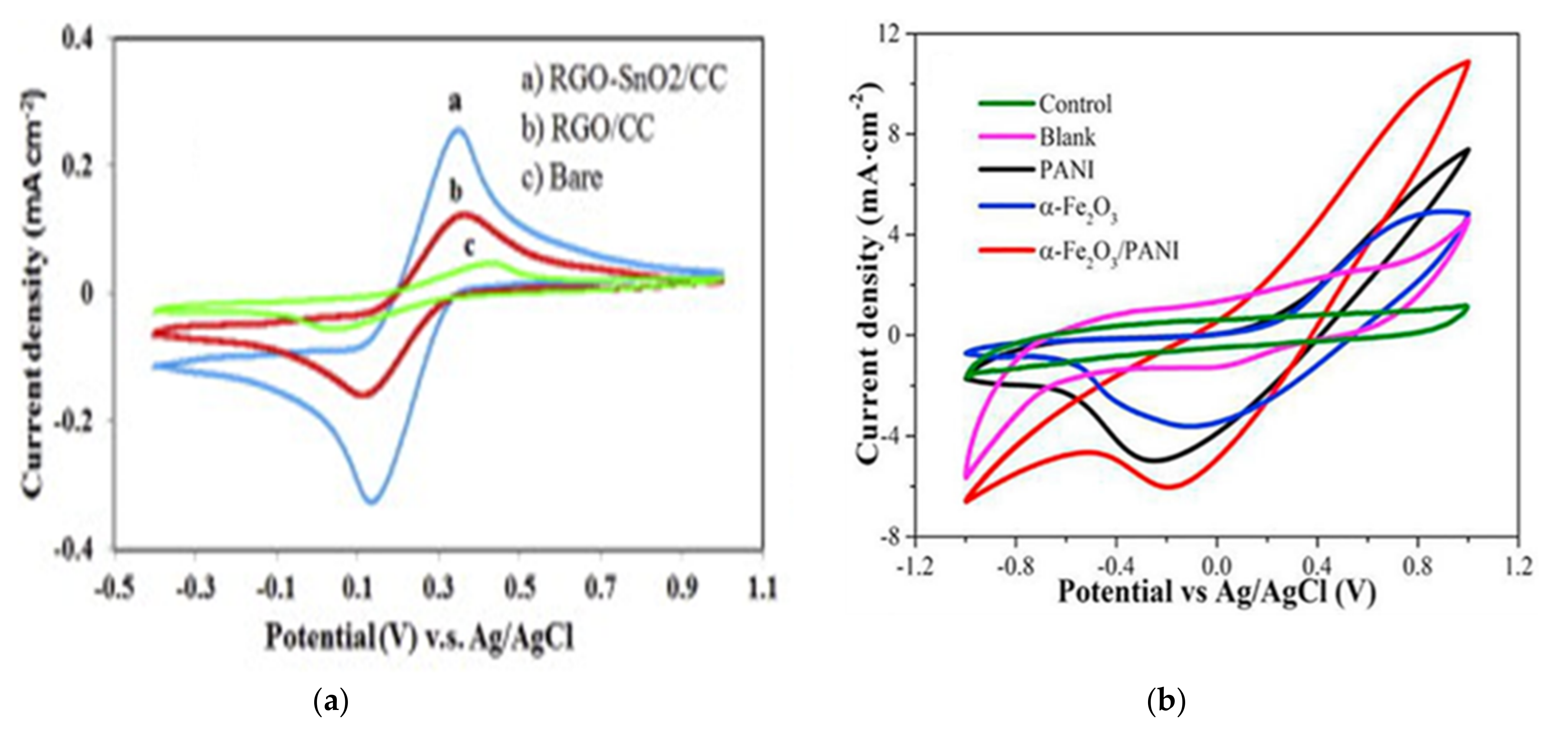
- Mehdinia, A.; Ziaei, E.; Jabbari, A. Facile microwave-assisted synthesized reduced graphene oxide/tin oxide nanocomposite and using as anode material of microbial fuel cell to improve power generation. Int. J. Hydrogen Energy 2014, 39, 10724–10730. [Google Scholar] [CrossRef]
- Velvizhi, G.; Venkata Mohan, S. Biocatalyst behavior under self-induced electrogenic microenvironment in comparison with anaerobic treatment: Evaluation with pharmaceutical wastewater for multi-pollutant removal. Bioresour. Technol. 2011, 102, 10784–10793. [Google Scholar] [CrossRef] [PubMed]
- Tao, C.-H.; Liang, M.; Li, W.; Zhang, J.-L.; Ni, R.-J.; Wu, M.-W. Removal of copper from aqueous solution by electrodeposition in cathode chamber of microbial fuel cell. J. Hazard. Mater. 2011, 189, 186–192. [Google Scholar] [CrossRef]
- Huang, L.; Chen, J.; Quan, X.; Yang, F. Enhancement of hexavalent chromium reduction and electricity production from a biocathode microbial fuel cell. Bioprocess. Biosyst. Eng. 2010, 33, 937–945. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Feng, C.; Ni, J.; Zhang, J.; Huang, W. Simultaneous reduction of vanadium (V) and chromium (VI) with enhanced energy recovery based on microbial fuel cell technology. J. Power Sources 2012, 204, 34–39. [Google Scholar] [CrossRef]
- Wang, G.; Huang, L.; Zhang, Y. Cathodic reduction of hexavalent chromium [Cr(VI)] coupled with electricity generation in microbial fuel cells. Biotechnol. Lett. 2008, 30, 1959. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Lim, B.; Choi, C. Removal of Hg2+ as an electron acceptor coupled with power generation using a microbial fuel cell. Bioresour. Technol. 2011, 102, 6304–6307. [Google Scholar] [CrossRef]
- Rodenas Motos, P.; ter Heijne, A.; van der Weijden, R.; Saakes, M.; Buisman, C.J.N.; Sleutels, T.H.J.A. High rate copper and energy recovery in microbial fuel cells. Front. Microbiol. 2015, 6, 527. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choi, C.; Cui, Y. Recovery of silver from wastewater coupled with power generation using a microbial fuel cell. Bioresour. Technol. 2012, 107, 522–525. [Google Scholar] [CrossRef] [PubMed]
- Catal, T.; Bermek, H.; Liu, H. Removal of selenite from wastewater using microbial fuel cells. Biotechnol. Lett. 2009, 31, 1211–1216. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Li, T.; Liu, C.; Quan, X.; Chen, L.; Wang, A.; Chen, G. Synergetic interactions improve cobalt leaching from lithium cobalt oxide in microbial fuel cells. Bioresour. Technol. 2013, 128, 539–546. [Google Scholar] [CrossRef] [PubMed]
- Lim, B.S.; Lu, H.; Choi, C.; Liu, Z.X. Recovery of silver metal and electric power generation using a microbial fuel cell. Desalination Water Treat. 2015, 54, 3675–3681. [Google Scholar] [CrossRef]
- Gupta, S.; Yadav, A.; Verma, N. Simultaneous Cr(VI) reduction and bioelectricity generation using microbial fuel cell based on alumina-nickel nanoparticles-dispersed carbon nanofiber electrode. Chem. Eng. J. 2017, 307, 729–738. [Google Scholar] [CrossRef]
- Shi, J.; Zhao, W.; Liu, C.; Jiang, T.; Ding, H. Enhanced Performance for Treatment of Cr (VI)-Containing Wastewater by Microbial Fuel Cells with Natural Pyrrhotite-Coated Cathode. Water 2017, 9, 979. [Google Scholar] [CrossRef] [Green Version]
- Wu, Y.; Wang, L.; Jin, M.; Kong, F.; Qi, H.; Nan, J. Reduced graphene oxide and biofilms as cathode catalysts to enhance energy and metal recovery in microbial fuel cell. Bioresour. Technol. 2019, 283, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Zhou, S. α-Fe2O3/polyaniline nanocomposites as an effective catalyst for improving the electrochemical performance of microbial fuel cell. Chem. Eng. J. 2018, 339, 539–546. [Google Scholar] [CrossRef]
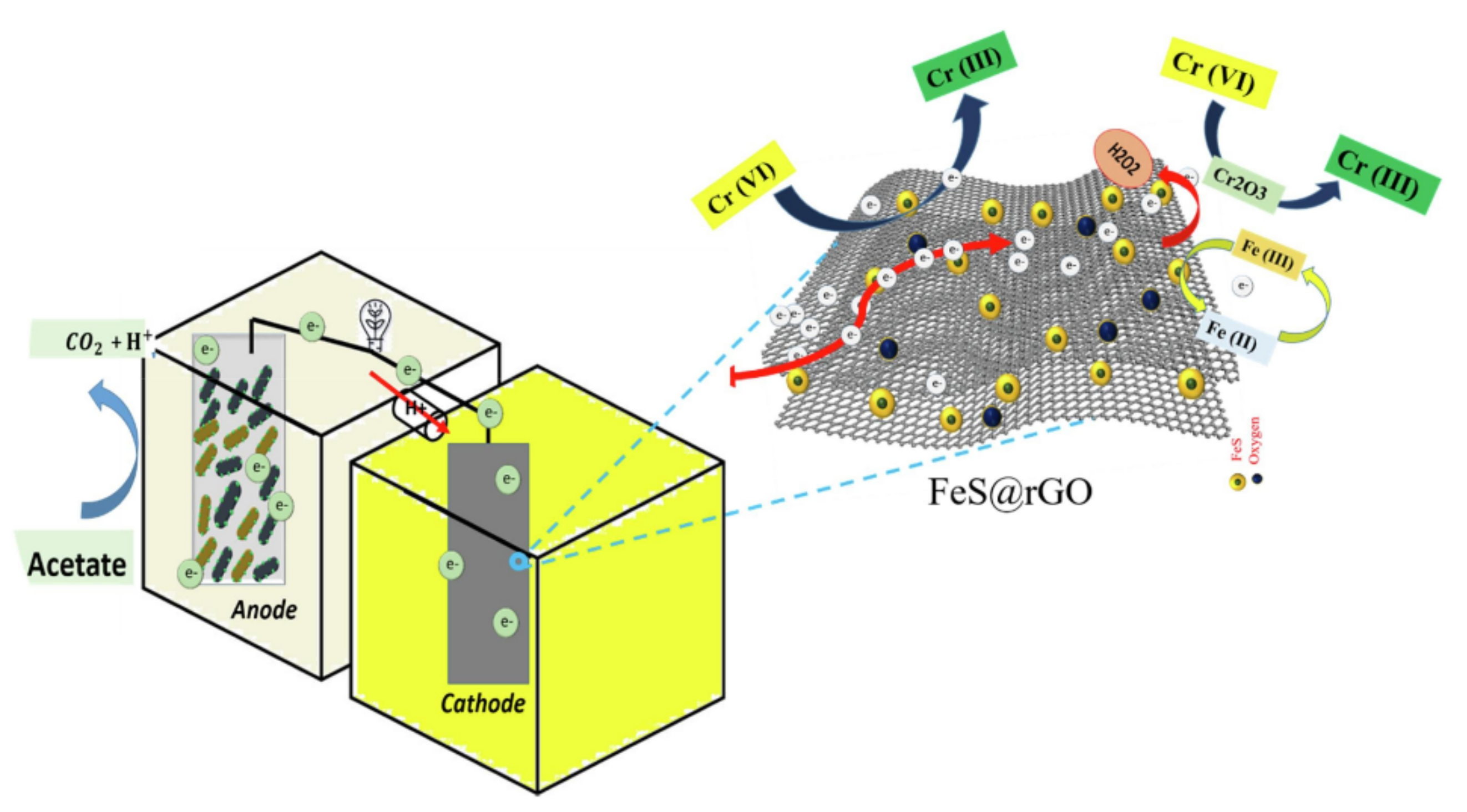
- Ali, J.; Wang, L.; Waseem, H.; Djellabi, R.; Oladoja, N.A.; Pan, G. FeS@rGO nanocomposites as electrocatalysts for enhanced chromium removal and clean energy generation by microbial fuel cell. Chem. Eng. J. 2020, 384, 123335. [Google Scholar] [CrossRef]
- Xue, A.; Shen, Z.-Z.; Zhao, B.; Zhao, H.-Z. Arsenite removal from aqueous solution by a microbial fuel cell–zerovalent iron hybrid process. J. Hazard. Mater. 2013, 261, 621–627. [Google Scholar] [CrossRef] [PubMed]
- Qin, B.; Luo, H.; Liu, G.; Zhang, R.; Chen, S.; Hou, Y.; Luo, Y. Nickel ion removal from wastewater using the microbial electrolysis cell. Bioresour. Technol. 2012, 121, 458–461. [Google Scholar] [CrossRef] [PubMed]
- Choi, C.; Hu, N.; Lim, B. Cadmium recovery by coupling double microbial fuel cells. Bioresour. Technol. 2014, 170, 361–369. [Google Scholar] [CrossRef] [PubMed]
- Ge, Z.; Li, J.; Xiao, L.; Tong, Y.; He, Z. Recovery of Electrical Energy in Microbial Fuel Cells. Environ. Sci. Technol. Lett. 2014, 1, 137–141. [Google Scholar] [CrossRef]
- Fan, Y.; Hu, H.; Liu, H. Enhanced Coulombic efficiency and power density of air-cathode microbial fuel cells with an improved cell configuration. J. Power Sources 2007, 171, 348–354. [Google Scholar] [CrossRef]
- Liu, H.; Cheng, S.; Logan, B.E. Production of Electricity from Acetate or Butyrate Using a Single-Chamber Microbial Fuel Cell. Environ. Sci. Technol. 2005, 39, 658–662. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Guo, K.; Li, H.; Du, Z.; Tian, J. Microfiltration membrane performance in two-chamber microbial fuel cells. Biochem. Eng. J. 2010, 52, 194–198. [Google Scholar] [CrossRef]

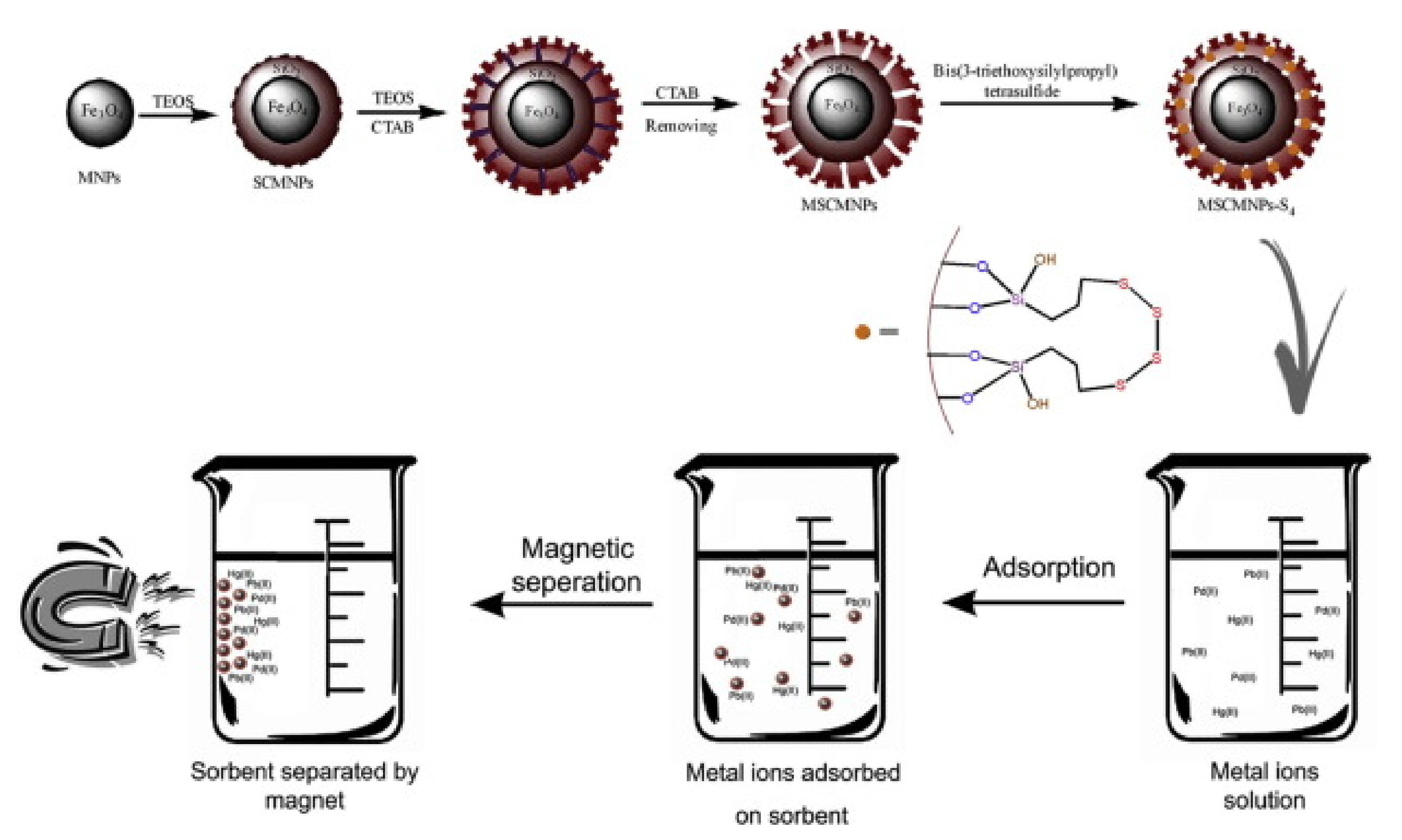
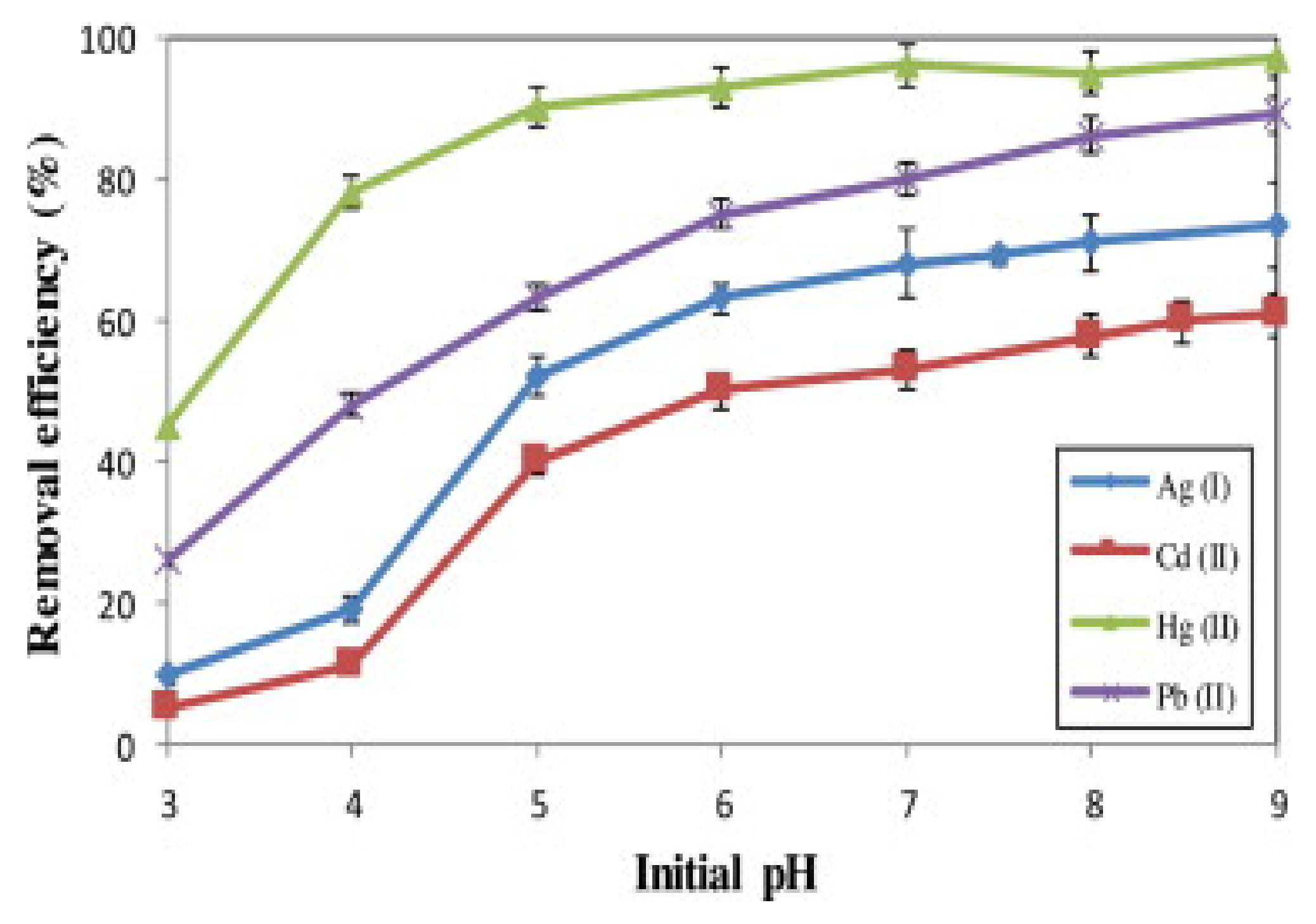
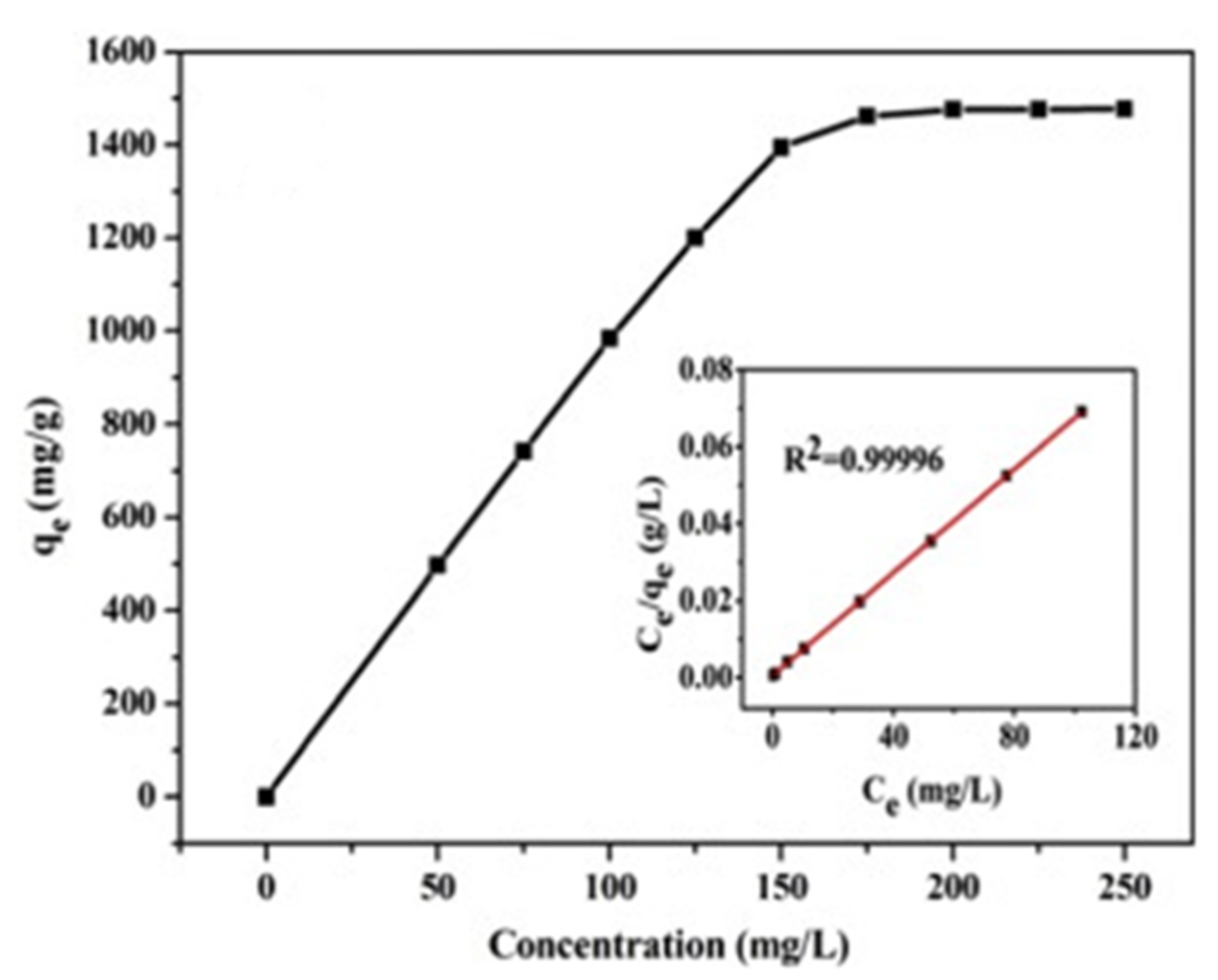

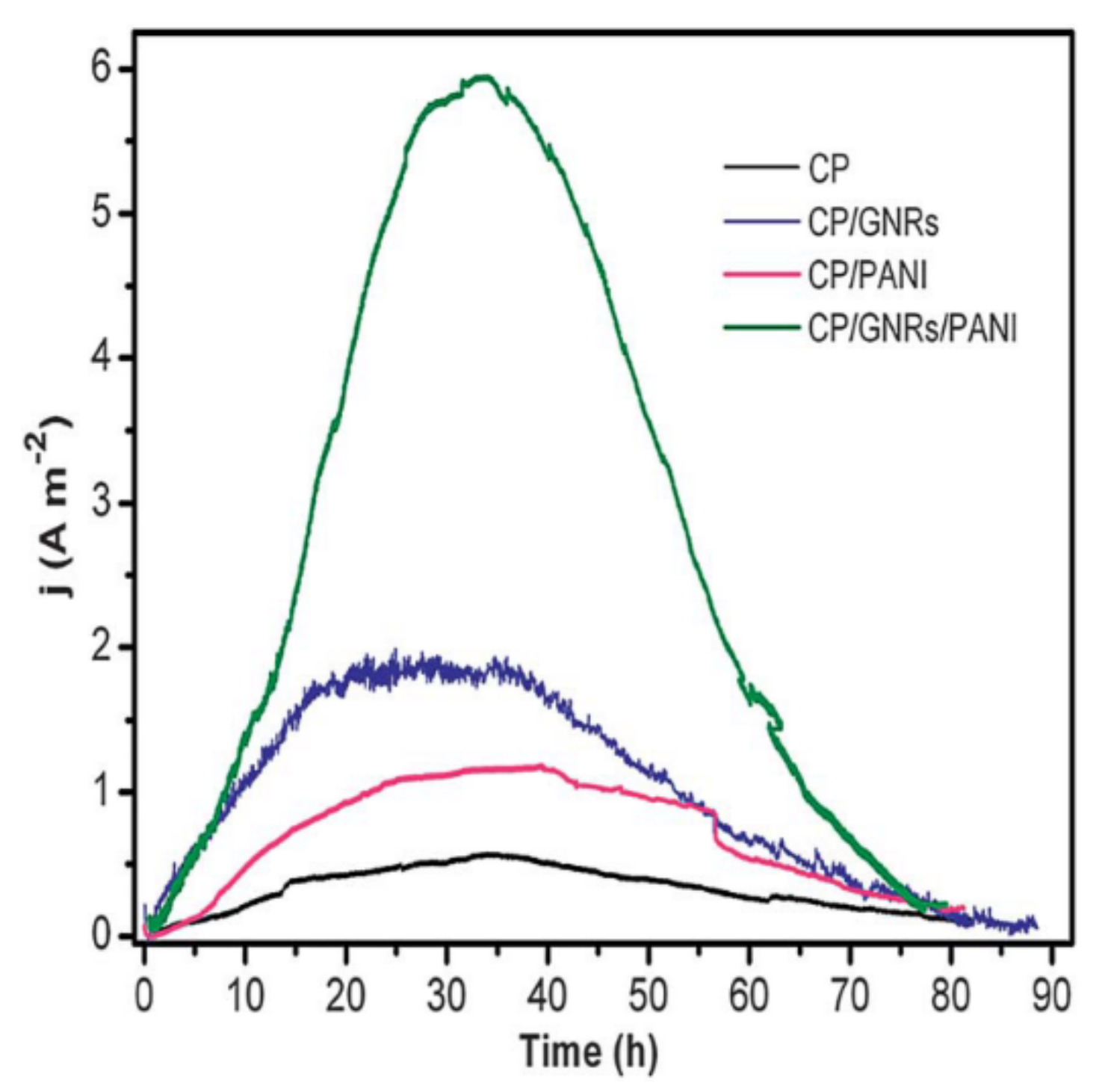
| Technology | Advantages | Disadvantages | References |
|---|---|---|---|
Physical
|
|
| [12,13] |
Chemical
|
|
| [14,15,16] |
| Biological (Microbes assisted remediation) |
|
| [17,18,19] |
| Electrochemical |
|
| [20,21,22] |
| Type of CNT | Target Metal/s | Initial Concentration (mg/L) | %Removal Efficiency | References |
|---|---|---|---|---|
| SWCNT | Hg2+ | (1–2000) mg/L | 4.16% | [44] |
| MSWCNT-CoS | Hg2+ | (1–2000) mg/L | 166.6% | [39] |
| MWCNT-SH | Hg2+ | 10 mg/L | 15.15% | [45] |
| SWCNTs-polysulfone nanocomposite-based membrane | Pb2+, As3+ | 1 mg/L | 94.2%, 87.6% | [46] |
| MWCNT-COOH functionalized nanotube | Pb2+ | (10–100) mg/L | 99.1% | [47] |
| Acidified MWCNT | Pb2+, Cu2+, Ni2+ | 100 mg/L | 93%, 78%, 83% | [48] |
| MWCNT | Mn7+ | (50–800) mg/L | 71.5% | [49] |
| MWCNTs | Cd2+ | 100 mg/L | 10.7% (pH = 2) 94.2% (pH = 7) 100% (pH = 10) | [48] |
| Oxidized MWCNT | Cu2+ | 100 mg/L | 78% | [48] |
| MWCNT | Fe2+ | 200 mg/L | 52% | [49] |
| Oxidized MWCNT | Ni2+ | 100 mg/L | 83% | [48] |
| Adsorbate | Target Metal | Initial Concentration | Removal % | References |
|---|---|---|---|---|
| Tea polyphenols—rGO-ZnO | Pb2+ | 20 mg/L | 98.9% | [68] |
| Porous Graphene | As3+ | 130 mg/L | >90% | [65] |
| rGO-Fe3O4 | Pb2+ | 20 mg/L | 37.314% | [64] |
| rGO-Sulfophenylazo | Cu2+, Ni2+ | 40 mg/L | 5.9 %, 6.6 % | [66] |
| GO embedded calcium alginate (GOCA) beads | Pb2+, Cd2+, Hg2+ | 50 mg/L | 60.2%, 18.1%, 37.4% | [69] |
| rGO-Sulfophenylazo | Cd2+ | 40 mg/L | 26.7% | [66] |
| GO-alpha cyclodextrin-polypyrrole | Cr6+ | 100–700 mg/L | 66.67% | [70] |
| rGO-Sulfophenylazo | Cr3+ | 40 mg/L | 19.1% | [66] |
| -COOH functionalized GO | Hg2+ | 400 mg/L | 12.2% | [67] |
| Chitosan/GO composite nanofibrous adsorbent | Cr6+ | 10–1000 mg/L | 31.04% | [71] |
| Adsorbent | Target Metal | Initial Concentration | Removal% | References |
|---|---|---|---|---|
| Thiol and Amino functionalized SBA-15 Silica | Hg2+ | 10.1 mg/L | 29.2% | [76] |
| Amino functionalized mesoporous silica | Cr6+ | 40 mg/L | 8.205% | [77] |
| Functionalized silica with –SH | Hg2+ | - | 50.5% | [78] |
| Amino functionalized silica gel in Tea Polyphenol extracts | Pb2+ | 5–1200 mg/L (Pb2+) 5–800 mg/L (Cu2+) | 98.1% | [79] |
| Amino-functionalized and pure silica nano hollow sphere (NH2-SNHS, SHNS) and silica gel(NH2-SG) | Ni2+ | 100 mg/L | 0.84% (SHNS), 2.59%(NH2-SG), and 3.13% (NH2-SNHS) mg/g | [74] |
| Amino-functionalized and pure silica nano hollow sphere (NH2-SNHS, SHNS) and silica gel(NH2-SG) | Pb2+ | 100 mg/L | 26.85%(SHNS), 54.35%(NH2-SG), and 96.78%(NH2-SNHS) | [74] |
| Amino functionalized silica gel in Tea Polyphenol extracts | Cd2+ | 5–800 mg/L | 99.78% | [74] |
| Organically functionalized silica gel | Cu2+ | 63 mg/L | 1.99% | [80] |
| Amino-functionalized and pure silica nano hollow sphere (NH2-SNHS, SHNS) and silica gel(NH2-SG) | Cd2+ | 100 mg/L | 2.6% (SHNS), 3.2%(NH2-SG), and 4.1%(NH2-SNHS) | [74] |
| Ionic liquid-functionalized silica | Pb2+ | 50–200 mg/L | 20.23% and | [81] |
| Amino functionalized silica gel in Tea Polyphenol extracts | Cu2+ | 5–800 mg/L | 99.59% | [79] |
| Ionic liquid-functionalized silica | Cd2+ | 50–200 mg/L | 15% | [76] |
| Adsorbent | Target Metal/s | Initial Concentration | Removal% | References |
|---|---|---|---|---|
| Hematite-Magnetite hybrid | Pb2+ | 2 mg/L | 97.67%, | [106] |
| Hematite-Magnetite hybrid | Cd2+ | 2 mg/L | 99.84% | [106] |
| Maghemite NP | As5+ | 1–11 mg/L | 50% | [97] |
| Hematite-Magnetite hybrid | Cr3+ | 2 mg/L | 99.50% | [106] |
| Maghemite NP | Cr6+ | 5–200 mg/L | 1.92% | [107] |
| Biogenic nano-magnetite | Cr6+ | 16.69 mg/L | 3.2% | [108] |
| Magnetite NP | Pb2+ | 10–600 mg/L | 3730% | [103] |
| Magnetite NP | Mn2+ | 10–600 mg/L | 7700% | [103] |
| Carboxyl functionalized magnetite NP | Cu2+ | 10 mg/L | 0.983% | [109] |
| Carboxyl functionalized magnetite NP | Cd2+ | 10 mg/L | 1.03% | [109] |
| Magnetite NP | Zn2+ | 10–600 mg/L | 1046% | [103] |
| Adsorbent | Target Metal | Initial Concentration or Concentration Range | Removal Efficiency (%) | References |
|---|---|---|---|---|
| Silica coated magnetic nanocomposites | Pb2+ | 5–120 mg/L | 1.49% | [124] |
| Silica based hybrid organic inorganic magnetic nanocomposites(MNPs@SiO2-TSD-TEOS) | Pb2+ | 100 mg/L | 41.7% | [125] |
| Silica based hybrid organic inorganic magnetic nanocomposites(MNPs@SiO2-TSD-TEOS) | Ni2+ | 100 mg/L | 35.7% | [125] |
| o-Vanillin functionalized mesoporous silica–coated magnetite nanoparticles (Fe3O4@MCM-41) | Pb2+ | 120 mg/L | 15.57% | [126] |
| Silica coated iron oxide magnetic nanocomposites(Fe3O4@SiO2) | Pb2+ | 10 mg/L | 97% | [127] |
| Silica coated iron oxide magnetic nanocomposites(Fe3O4@SiO2) | Hg2+ | 10 mg/L | 94.12% | [127] |
| Polythiophene modified chitosan/magnetite nanocomposites | Hg2+ | 0.02–100 mg/L | 5.28% | [128] |
| Bismuthiol-II-immobilized magnetic nanoparticles | Cr3+ | - | >90% | [129] |
| Bismuthiol-II-immobilized magnetic nanoparticles | Cu2+ | - | >90% | [129] |
| Thiol-lignocellulose sodium bentonite (TLSB) nanocomposites | Cd2+ | (0.20–1.70) × 103 mg/L | 45.832% | [130] |
| Thiol-lignocellulose sodium bentonite (TLSB) nanocomposites | Zn2+ | (0.20–1.70) × 103 mg/L | 35.729% | [130] |
| Magnetic Chitosan Nanocomposites | Cd2+ | 10 mg/L | 92.1% | [131] |
| Water-soluble magnetic graphene nanocomposites | Cd2+ | NA | >85% | [114] |
| Functionalized magnetic Fe3O4 | Cu2+ | 10 mg/L | 96% | [132] |
| Functionalized magnetic Fe3O4 | Hg2+ | 10 mg/L | 96% | [132] |
| Fe@MgO nanocomposite | Pb2+ | 100 mg/L | 147.64% | [118] |
| Magnetic MWCNT | Cr6+ | 1–25 mg/L | 1.14% | [133] |
| Target Metal | BES Configuration | Anode | Cathode | Power Output | Reference |
|---|---|---|---|---|---|
| Cr6+ | Double chambered MFC | Graphite plate | Graphite plate | 150 mW/m2 | [158] |
| Hg2+ | Double chambered MFC | Grpahite rod | Graphite rod | 32.6 ± 0.5 W/m2 | [140] |
| Hg2+ | Double chambered MFC | Graphite felt | Carbon paper | 433.1 mW/m2 | [159] |
| Cu2+ | Double chambered MFC | Carbon felt | Carbon plate | 5.5 W/m2 | [160] |
| Ag+ | Double chambered MFC | Carbon brush | Carbon felt | 4.25 mW/m2 | [161] |
| Se | Single chambered MFC | Carbon cloth | Carbon cloth | 13–1500 mW/m2 depending on initial concentration | [162] |
| Co | Double chambered MFC | Graphite felt | Graphite felt | 258 mW/m2 | [163] |
| Ag+ | Double chambered MFC | Carbon felt | Carbon felt | 0.109 mW/m2 | [164] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumar, R.; Rauwel, P.; Rauwel, E. Nanoadsorbants for the Removal of Heavy Metals from Contaminated Water: Current Scenario and Future Directions. Processes 2021, 9, 1379. https://doi.org/10.3390/pr9081379
Kumar R, Rauwel P, Rauwel E. Nanoadsorbants for the Removal of Heavy Metals from Contaminated Water: Current Scenario and Future Directions. Processes. 2021; 9(8):1379. https://doi.org/10.3390/pr9081379
Chicago/Turabian StyleKumar, Rohit, Protima Rauwel, and Erwan Rauwel. 2021. "Nanoadsorbants for the Removal of Heavy Metals from Contaminated Water: Current Scenario and Future Directions" Processes 9, no. 8: 1379. https://doi.org/10.3390/pr9081379
APA StyleKumar, R., Rauwel, P., & Rauwel, E. (2021). Nanoadsorbants for the Removal of Heavy Metals from Contaminated Water: Current Scenario and Future Directions. Processes, 9(8), 1379. https://doi.org/10.3390/pr9081379








