CO Preferential Oxidation in a Microchannel Reactor Using a Ru-Cs/Al2O3 Catalyst: Experimentation and CFD Modelling
Abstract
1. Introduction
2. Materials and Methods
2.1. Microchannel Reactor Design
2.2. Catalyst Preparation and Washcoating
2.3. Experimental Apparatus
2.4. Experimental Procedure
2.5. Performance Criteria
3. CFD Modelling Procedure
3.1. Model Geometry
3.2. Model Assumptions and Boundary Conditions
3.3. Governing Equations
3.4. Equilibrium Calculations
3.5. Parameter Estimation Procedure
3.6. Solution Method
3.7. Goodness of Fit Analysis Using the Bootstrap Method
4. Results and Discussion
4.1. Experimental Results
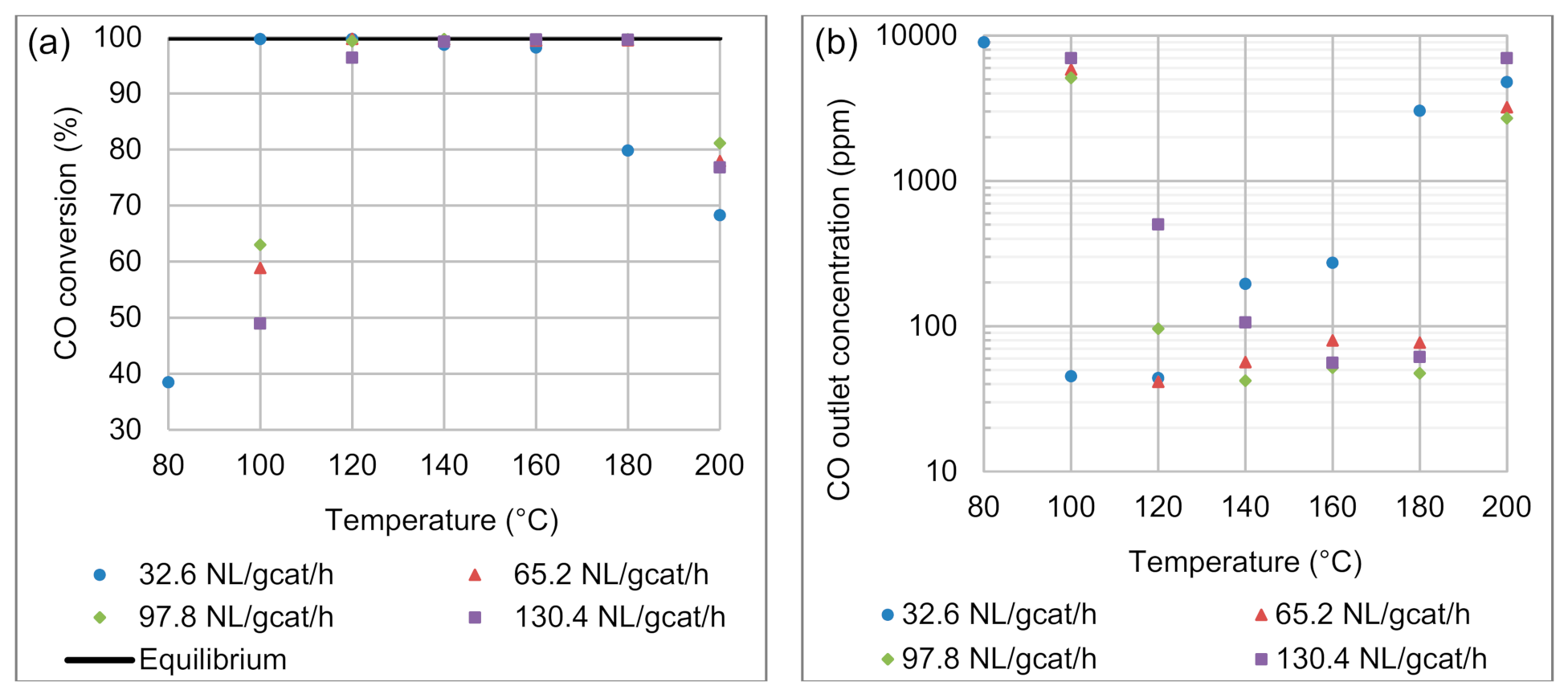
4.1.1. Effects of Reaction Temperature and Space Velocity on CO Conversion
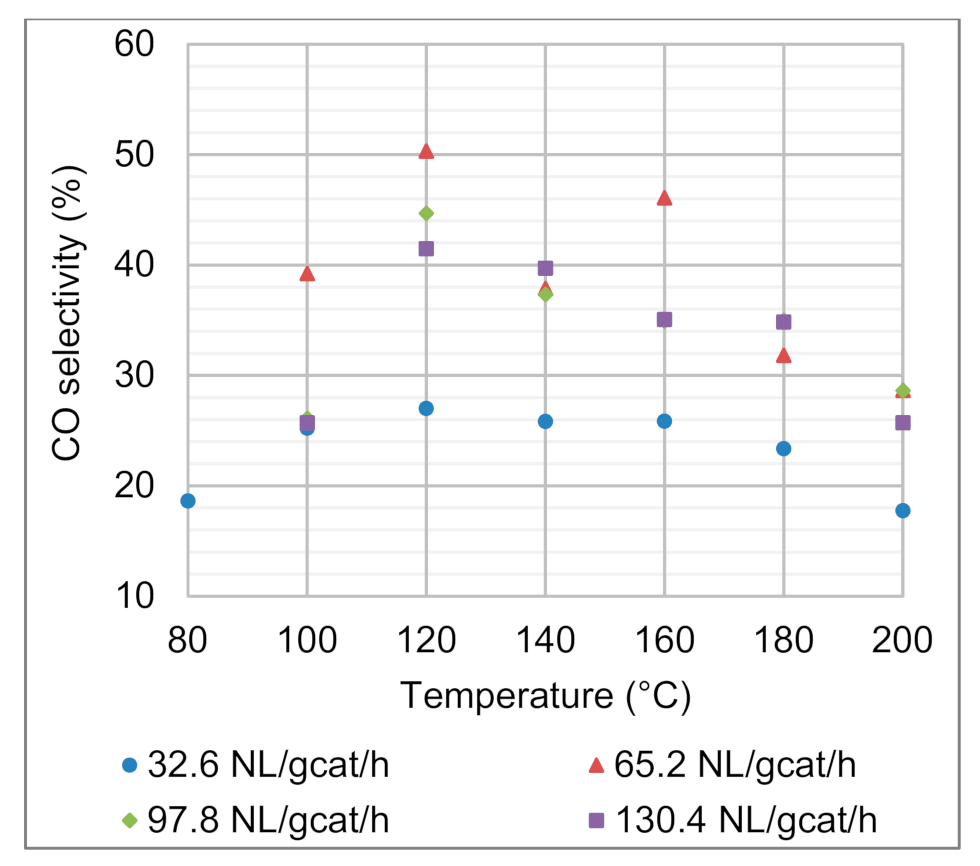
4.1.2. Effects of Reaction Temperature and Space Velocity on CO Selectivity
4.2. Modelling Results
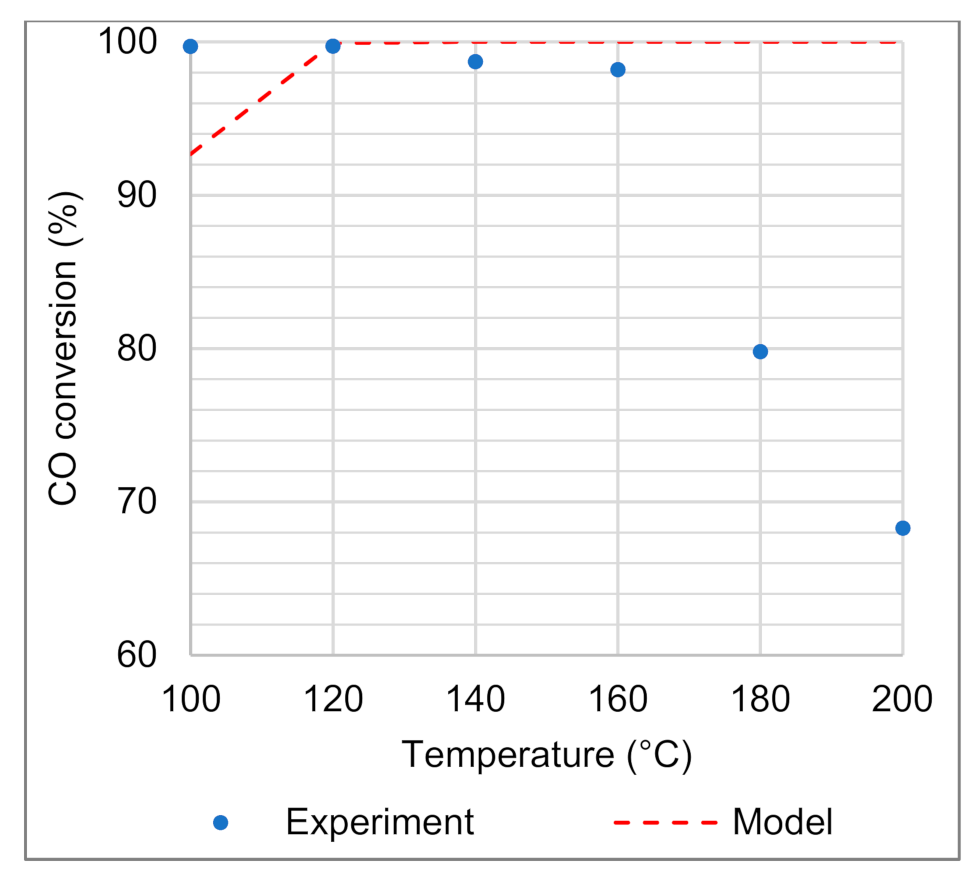
4.2.1. Estimation of Kinetic Parameters
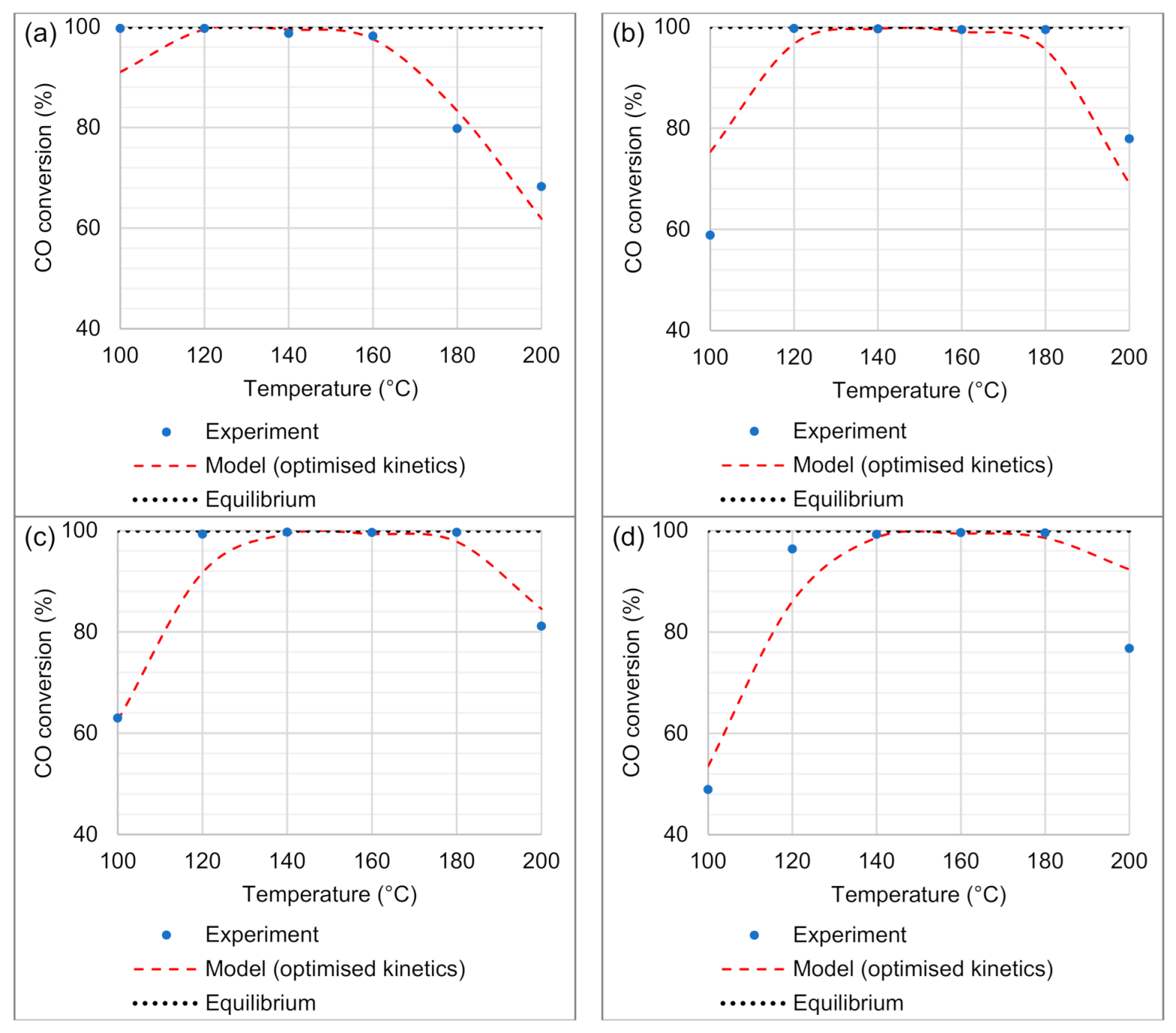
4.2.2. Model Validation
4.2.3. Goodness of Fit Results
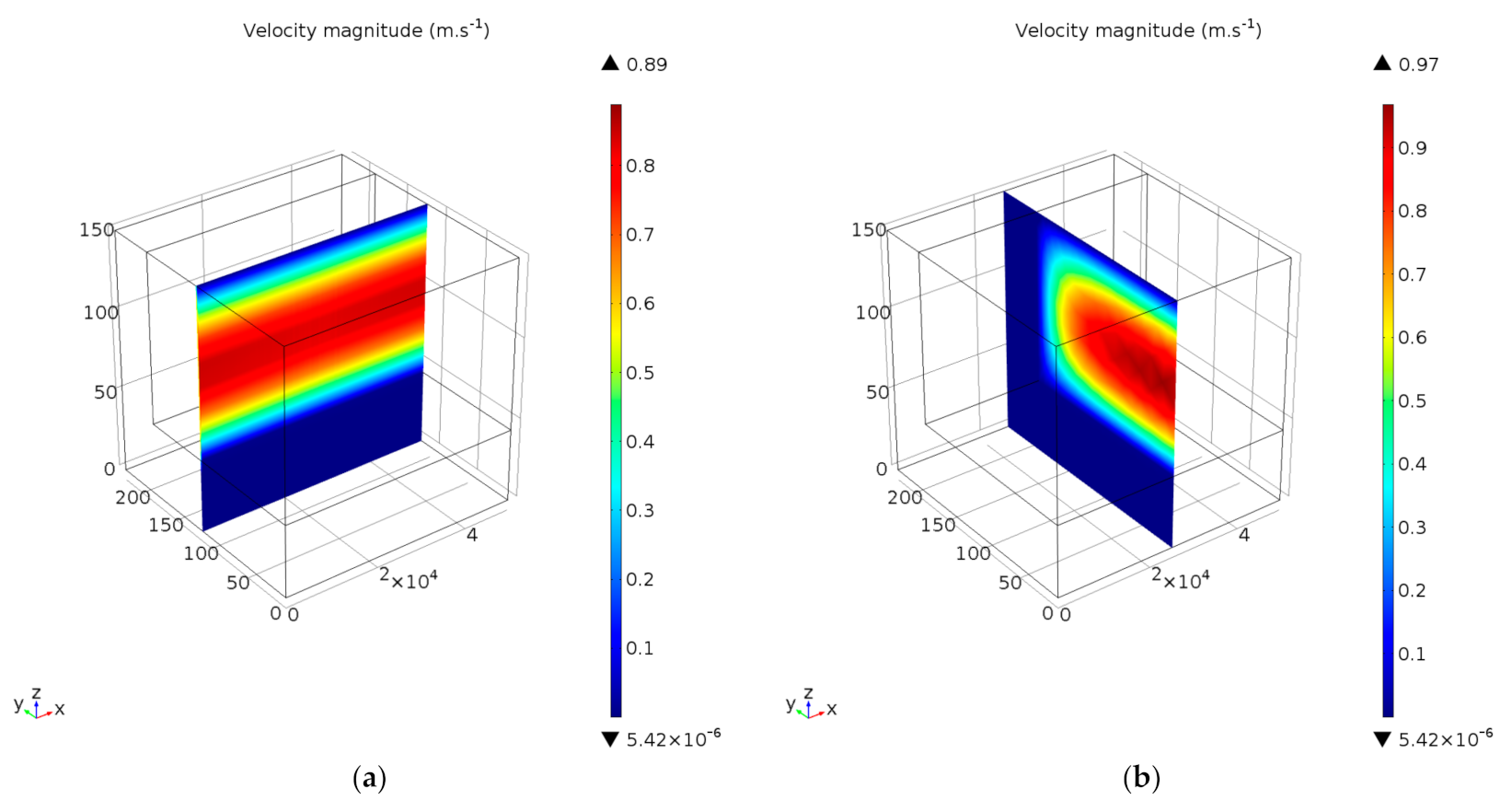
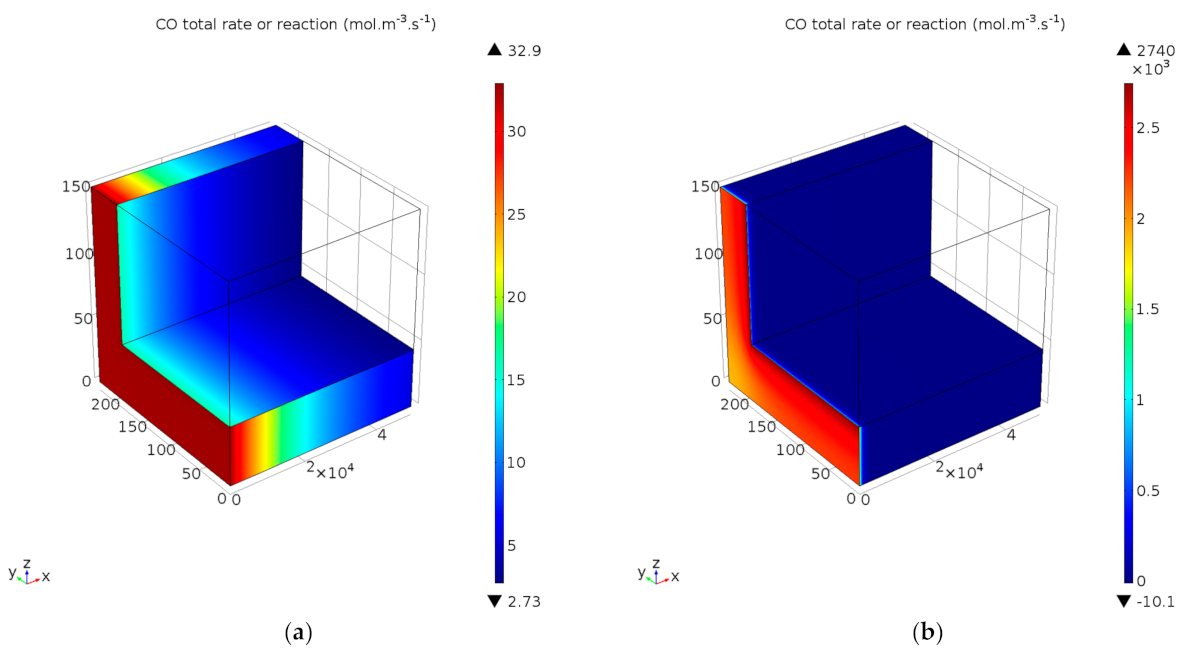
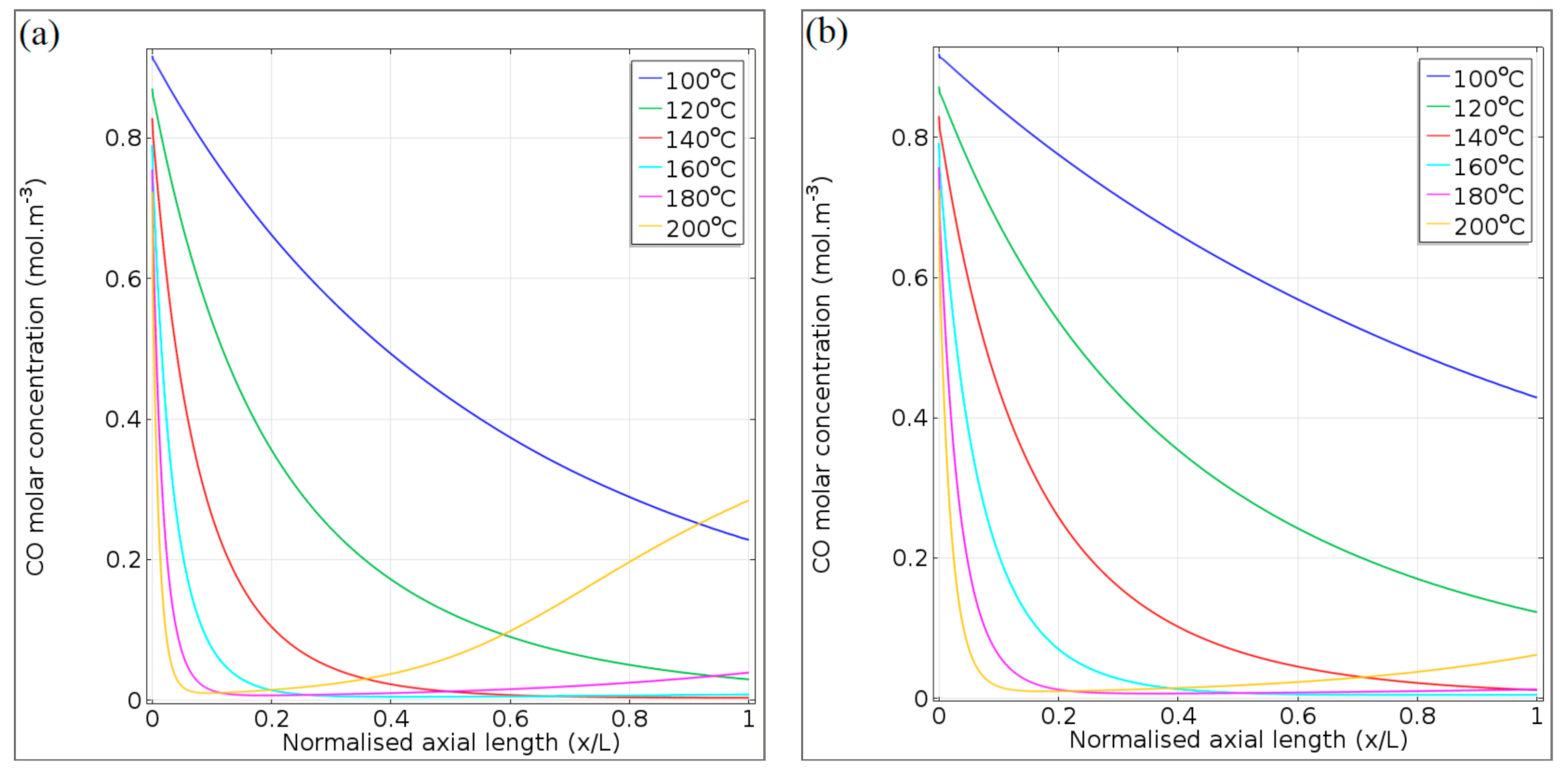
4.2.4. Simulation of Transport Phenomena
Velocity Profiles
Reaction Rate Profiles
Concentration Profiles
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Nomenclature
| CFD | Computational fluid dynamic |
| CO PROX | Preferential oxidation of CO |
| OD | Outside diameter |
| PEMFC | Proton exchange membrane fuel cell |
| RWGS | Reverse water–gas shift |
| vol.% | Volume percent |
| WGS | Water–gas shift |
| wt.% | Weight percent |
| a, b, c | Orders of reaction |
| CF | Forchheimer drag coefficient |
| Ci | Molar concentration of species i in the mixture, mol m−3 |
| Cp | Specific heat capacity, J kg−1 K−1 |
| DH | Microchannel hydraulic diameter, m |
| Deff | Effective binary diffusion coefficient, m2 s−1 |
| Dij | Binary diffusion coefficient of species i in species j, m2 s−1 |
| Dij eff | Effective binary diffusion coefficient of species i in species j, m2 s−1 |
| e | Residuals between calculated experimental CO conversions |
| E | Activation energy, J mol−1 |
| Fi | Volumetric flow rate of species i, m3 s−1 |
| gcat | Gram of catalyst |
| k | Thermal conductivity, W m−1 K−1 |
| Keq | Equilibrium constant |
| ki | Reaction rate pre-exponential constant of reaction i |
| Ki | Adsorption constant of component i, m3 mol−1 |
| M | Mean molar mass, kg mol−1 |
| Mi | Molar mass of species i, kg mol−1 |
| n | Vector normal to the symmetry plane |
| Ni | Number of moles of species i |
| P | Pressure, Pa |
| Patm | Atmospheric pressure, Pa |
| Pi | Partial pressure of species i, Pa |
| R | Universal gas constant, Pa m3 mol−1 K−1 |
| r | Rate of reaction, mol m−3 s−1 |
| Rr | Rate of reaction, mol kg−1 s−1 |
| SCO | CO selectivity, % |
| T | Temperature, K |
| T0 | Reference temperature, K |
| u | Linear velocity, m s−1 |
| XCO | CO conversion, % |
| Xg,h | CO conversions at temperature g and flow rate h |
| ΔH | Enthalpy of reaction, J mol−1 |
| ΔHi | Adsorption enthalpy of species i, J mol−1 |
| ΔHr | Heat of reaction, J mol−1 |
| α | Significance level, used in the F-test |
| β0 | Average of simulated CO conversions |
| δ | Porous washcoat thickness, m |
| ε | Porosity of the catalyst |
| κ | Permeability of the catalyst, m2 |
| ρ | Gas density, kg m−3 |
| ρs | Solid catalyst density, kg m−3 |
| θ | Parameters used during optimisation |
| μ | Viscosity, Pa.s |
| υi | Atomic volume of species i, cm3 mol−1 |
| ωi | Weight fraction of species i, cm3 mol−1 |
| eff | Effective |
| g | Number of temperatures used in the model (1, …, 6) |
| h | Number of flow rates used in the model (1, …, 4) |
| i, j | Component i or j |
| in | Reactor inlet |
| out | Reactor outlet |
| s | Solid |
| wall | Reactor wall |
Appendix A. Governing Equations Describing the Microchannel Reactor within the CFD Model
| Governing Equation | Formula |
|---|---|
| Ideal gas law | |
| Fuller–Schettler–Giddings equation | |
| Free-fluid region | |
| Continuity equation | |
| Navier–Stokes momentum equation | |
| Species continuity equation | |
| Energy equation | |
| Porous catalyst layer | |
| Continuity equation | |
| Brinkman–Forchheimer–Darcy equation | |
| Species continuity equation | |
| Bruggeman correlation | |
| Energy equation | |
| Condition | Formula |
|---|---|
| No slip condition at the wall | |
| Average inlet gas velocity | |
| Outlet pressure | |
| Symmetry plane | |
| Inlet mass fraction |
Appendix B. Goodness of Fit Using the Bootstrap Statistical Method

References
- Du, X.; Lang, Y.; Cao, K.; Yang, J.; Cai, J.; Shan, B.; Chen, R. Bifunctionally faceted Pt/Ru nanoparticles for preferential oxidation of CO in H2. J. Catal. 2021, 396, 148–156. [Google Scholar] [CrossRef]
- Qiu, Z.; Guo, X.; Mao, J.; Zhou, R. The catalytic performance of isolated-dispersed Au on nanosized CeO2 for CO preferential oxidation in H2-rich stream. Appl. Surf. Sci. 2019, 481, 1072–1079. [Google Scholar] [CrossRef]
- Fiorenza, R.; Spitaleri, L.; Gulino, A. Ru–Pd Bimetallic catalysts supported on purification through CO preferential oxidation. Catalysts 2018, 8, 203. [Google Scholar] [CrossRef]
- Galletti, C.; Specchia, S.; Saracco, G.; Specchia, V. CO preferential oxidation in H2-rich gas for fuel cell applications: Microchannel reactor performance with Rh-based catalyst. Int. J. Hydrogen Energy 2008, 33, 3045–3048. [Google Scholar] [CrossRef]
- Mohaideen, K.K.; Kim, W.; Koo, K.Y.; Yoon, W.L. Highly dispersed Ni particles on Ru/NiAl catalyst derived from layered double hydroxide for selective CO methanation. Catal. Commun. 2015, 60, 8–13. [Google Scholar] [CrossRef]
- Nunez, N.E.; Bideberripe, H.P.; Mizrahi, M.; Ramallo-Lopez, J.M.; Casella, M.L.; Siri, G.J. CO selective oxidation using Co-promoted Pt-Al2O3 catalysts. Int. J. Hydrogen Energy 2016, 41, 19005–19013. [Google Scholar] [CrossRef]
- Arzamendi, G.; Uriz, I.; Diéguez, P.M.; Laguna, O.H.; Hernández, W.Y.; Álvarez, A.; Centeno, M.A.; Odriozola, J.A.; Montes, M.; Gandía, L.M. Selective CO removal over Au/CeFe and CeCu catalysts in microreactors studied through kinetic analysis and CFD simulations. Chem. Eng. J. 2011, 167, 588–596. [Google Scholar] [CrossRef]
- Grisel, R.J.H.; Nieuwenhuys, B.E. Selective oxidation of CO, over supported Au catalysts. J. Catal. 2001, 199, 48–59. [Google Scholar] [CrossRef]
- Zou, X.; Qi, S.; Suo, Z.; An, L.; Li, F. Activity and deactivation of Au/Al2O3 catalyst for low-temperature CO oxidation. Catal. Commun. 2007, 8, 784–788. [Google Scholar] [CrossRef]
- Miao, Y.X.; Shi, L.; Sun, Q.; Li, W.C. A highly efficient potassium-treated Au-Cu/Al2O3 catalyst for the preferential oxidation of carbon monoxide. RSC Adv. 2016, 6, 24603–24609. [Google Scholar] [CrossRef]
- Beck, A.; Yang, A.; Leland, A.R.; Riscoe, A.R.; Lopez, F.A.; Goodman, E.D.; Cargnello, M. Understanding the preferential oxidation of carbon monoxide (PROX) using size-controlled Au nanocrystal catalyst. Am. Inst. Chem. Eng. 2018, 64, 3159–3167. [Google Scholar] [CrossRef]
- Bion, N.; Epron, F.; Moreno, M.; Mariño, F.; Duprez, D. Preferential oxidation of carbon monoxide in the presence of hydrogen (PROX) over noble metals and transition metal oxides: Advantages and drawbacks. Top. Catal. 2008, 51, 76–88. [Google Scholar] [CrossRef]
- Wang, C.; Cheng, Q.; Wang, X.; Ma, K.; Bai, X.; Tan, S.; Tian, Y.; Ding, T.; Zheng, L.; Zhang, J.; et al. Applied surface science enhanced catalytic performance for CO preferential oxidation over CuO catalysts supported on highly defective CeO2 nanocrystals. Appl. Surf. Sci. 2017, 422, 932–943. [Google Scholar] [CrossRef]
- Han, Y.F.; Kahlich, M.J.; Kinne, M.; Behm, R.J. Kinetic study of selective CO oxidation in H2-rich gas on a Ru/γ-Al2O3 catalyst. Phys. Chem. Chem. Phys. 2002, 4, 389–397. [Google Scholar] [CrossRef]
- Xu, G.; Zhang, Z.G. Preferential CO oxidation on Ru/Al2O3 catalyst: An investigation by considering the simultaneously involved methanation. J. Power Sources 2006, 157, 64–77. [Google Scholar] [CrossRef]
- Kim, Y.H.; Park, E.D. The effect of the crystalline phase of alumina on the selective CO oxidation in a hydrogen-rich stream over Ru/Al2O3. Appl. Catal. B Environ. 2010, 96, 41–50. [Google Scholar] [CrossRef]
- Gokhale, S.V.; Tayal, R.K.; Jayaraman, V.K.; Kulkarni, B.D. Microchannel reactors: Applications and use in process development. Int. J. Chem. React. Eng. 2005, 3, 1–53. [Google Scholar] [CrossRef]
- Snytnikov, P.V.; Potemkin, D.I.; Rebrov, E.V.; Sobyanin, V.A.; Hessel, V.; Schouten, J.C. Design, scale-out, and operation of a microchannel reactor with a Cu/CeO2-x catalytic coating for preferential CO oxidation. Chem. Eng. J. 2010, 160, 923–929. [Google Scholar] [CrossRef]
- Uriz, I.; Arzamendi, G.; Diéguez, P.M.; Laguna, O.H.; Centeno, M.A.; Odriozola, J.A.; Gandía, L.M. Preferential oxidation of CO over Au/CuOx-CeO2 catalyst in microstructured reactors studied through CFD simulations. Catal. Today 2013, 216, 283–291. [Google Scholar] [CrossRef]
- Laguna, O.H.; Ngassa, E.M.; Oraá, S.; Álvarez, A.; Domínguez, M.I.; Romero-Sarria, F.; Arzamendi, G.; Gandía, L.M.; Centeno, M.A.; Odriozola, J.A. Preferential oxidation of CO (CO-PROX) over CuOx/CeO2 coated microchannel reactor. Catal. Today 2012, 180, 105–110. [Google Scholar] [CrossRef]
- Chiuta, S.; Everson, R.C.; Neomagus, H.W.J.P.; Le Grange, L.A.; Bessarabov, D.G. A modelling evaluation of an ammonia-fuelled microchannel reformer for hydrogen generation. Int. J. Hydrogen Energy 2014, 39, 11390–11402. [Google Scholar] [CrossRef]
- O’Connell, M.; Kolb, G.; Schelhaas, K.P.; Wichert, M.; Tiemann, D.; Pennemann, H.; Zapf, R. Towards mass production of microstructured fuel processors for application in future distributed energy generation systems: A review of recent progress at IMM. Chem. Eng. Res. Des. 2012, 90, 11–18. [Google Scholar] [CrossRef]
- Engelbrecht, N.; Chiuta, S.; Everson, R.C.; Neomagus, H.W.J.P.; Bessarabov, D.G. Experimentation and CFD modelling of a microchannel reactor for carbon dioxide methanation. Chem. Eng. J. 2017, 313, 847–857. [Google Scholar] [CrossRef]
- Chiuta, S.; Everson, R.C.; Neomagus, H.W.J.P.; Bessarabov, D.G. Performance evaluation of a high-throughput microchannel reactor for ammonia decomposition over a commercial Ru-based catalyst. Int. J. Hydrogen Energy 2015, 40, 2921–2926. [Google Scholar] [CrossRef]
- COMSOL. Chemical Reaction Engineering Module User’s Guide. Available online: https://doc.comsol.com/5.4/doc/com.comsol.help.chem/ChemicalReactionEngineeringModuleUsersGuide.pdf (accessed on 5 May 2021).
- CHERIC. Korean Thermophysical Properties Database. Available online: www.cheric.org (accessed on 4 March 2020).
- Fuller, E.N.; Schettler, P.D.; Giddings, J.C. A new method for prediction of binary gas-phase diffusion coefficients. Ind. Eng. Chem. 1966, 58, 18–27. [Google Scholar] [CrossRef]
- Baughman, A.C.; Huang, X.; Martin, L.L. Evaluating kinetic models for preferential CO-oxidation catalysts using optimization-based parameter estimation. J. Power Sources 2012, 210, 402–408. [Google Scholar] [CrossRef]
- Garbis, P.; Kern, C.; Jess, A. Kinetics and reactor design aspects of selective methanation of CO over a Ru/γ-Al2O3 catalyst in CO2/H2 rich gases. Energies 2019, 12, 469. [Google Scholar] [CrossRef]
- Froment, G.; Bischoff, K.; De Wilde, J. Chemical Reactor Analysis and Design, 3rd ed.; John Wiley and Sons: Hoboken, NJ, USA, 2011. [Google Scholar]
- Ramanathan, K.; Sharma, C.S. Kinetic parameters estimation for three way catalyst modeling. Ind. Eng. Chem. Res. 2011, 50, 9960–9979. [Google Scholar] [CrossRef]
- Spiess, A.-N.; Neumeyer, N. An evaluation of R2 as an inadequate measure for nonlinear models in pharmacological and biochemical research: A Monte Carlo approach. BMC Pharmacol. 2010, 10, 6. [Google Scholar] [CrossRef]
- Wehrens, R.; van der Linden, W.E. Bootstrapping principal component regression models. J. Chemom. 1997, 11, 157–171. [Google Scholar] [CrossRef]
- Wehrens, R.; Putter, H.; Buydens, L.M.C. The bootstrap: A tutorial. Chemom. Intell. Lab. Syst. 2000, 54, 35–52. [Google Scholar] [CrossRef]
- Wu, C. Jackknife, Bootstrap and other resampling methods in regression analysis. Ann. Stat. 1986, 14, 1261–1295. [Google Scholar] [CrossRef]
- Liu, R.Y. Bootstrap procedures under some non-I.I.D. Models. Ann. Stat. 1988, 16, 1696–1708. [Google Scholar] [CrossRef]





| Kinetic Parameter | Published Values [28,29] | Manually Estimated Values | Regressed Values |
|---|---|---|---|
| a | 0.01 | 1.00 | 1.00 |
| EC [kJ mol−1] | 35.56 | 72.00 | 72.15 |
| kC [s−1 (mol m−3)−1.07] | 1.331 × 106 | 2.81 × 1011 | 3.421 × 1011 |
| ED [kJ mol−1] | 97.00 | 74.00 | 74.74 |
| kD [s−1 (mol m−3)−1] | 6.451 × 108 | 2.401 × 109 | 2.891 × 109 |
| Fitted Model Using: | p-Value | Conclusion |
|---|---|---|
| Published parameters | 27.0% (α > 0.05) | Do not reject (Equation (10)) |
| Regressed parameters | <1.0 × 10−3% | Reject (Equation (10)) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Musavuli, K.C.; Engelbrecht, N.; Everson, R.C.; Grobler, G.L.; Bessarabov, D. CO Preferential Oxidation in a Microchannel Reactor Using a Ru-Cs/Al2O3 Catalyst: Experimentation and CFD Modelling. Processes 2021, 9, 867. https://doi.org/10.3390/pr9050867
Musavuli KC, Engelbrecht N, Everson RC, Grobler GL, Bessarabov D. CO Preferential Oxidation in a Microchannel Reactor Using a Ru-Cs/Al2O3 Catalyst: Experimentation and CFD Modelling. Processes. 2021; 9(5):867. https://doi.org/10.3390/pr9050867
Chicago/Turabian StyleMusavuli, Kyatsinge Cedric, Nicolaas Engelbrecht, Raymond Cecil Everson, Gerrit Lodewicus Grobler, and Dmitri Bessarabov. 2021. "CO Preferential Oxidation in a Microchannel Reactor Using a Ru-Cs/Al2O3 Catalyst: Experimentation and CFD Modelling" Processes 9, no. 5: 867. https://doi.org/10.3390/pr9050867
APA StyleMusavuli, K. C., Engelbrecht, N., Everson, R. C., Grobler, G. L., & Bessarabov, D. (2021). CO Preferential Oxidation in a Microchannel Reactor Using a Ru-Cs/Al2O3 Catalyst: Experimentation and CFD Modelling. Processes, 9(5), 867. https://doi.org/10.3390/pr9050867








