Apple Fermented Products: An Overview of Technology, Properties and Health Effects
Abstract
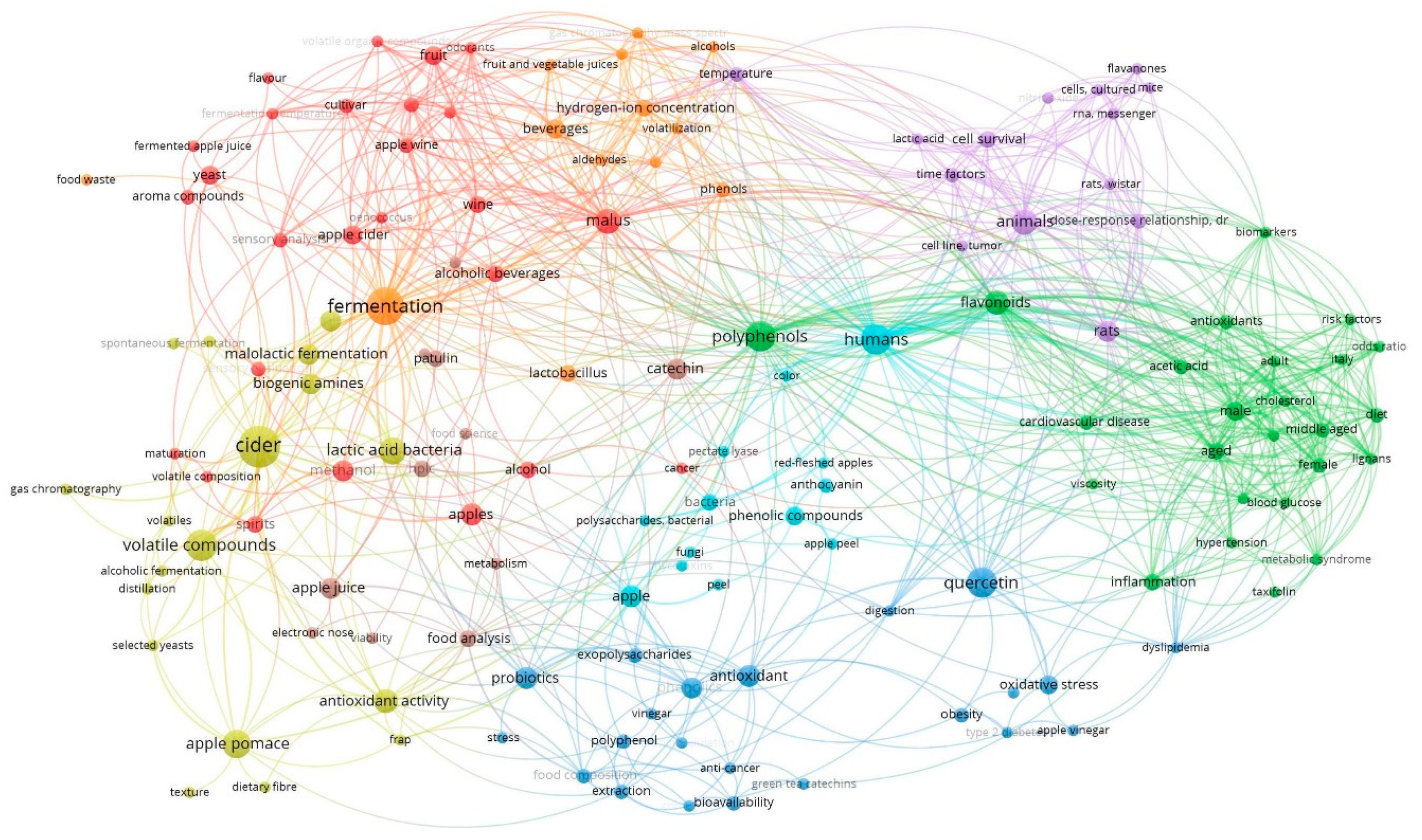
1. Introduction
2. Methodology
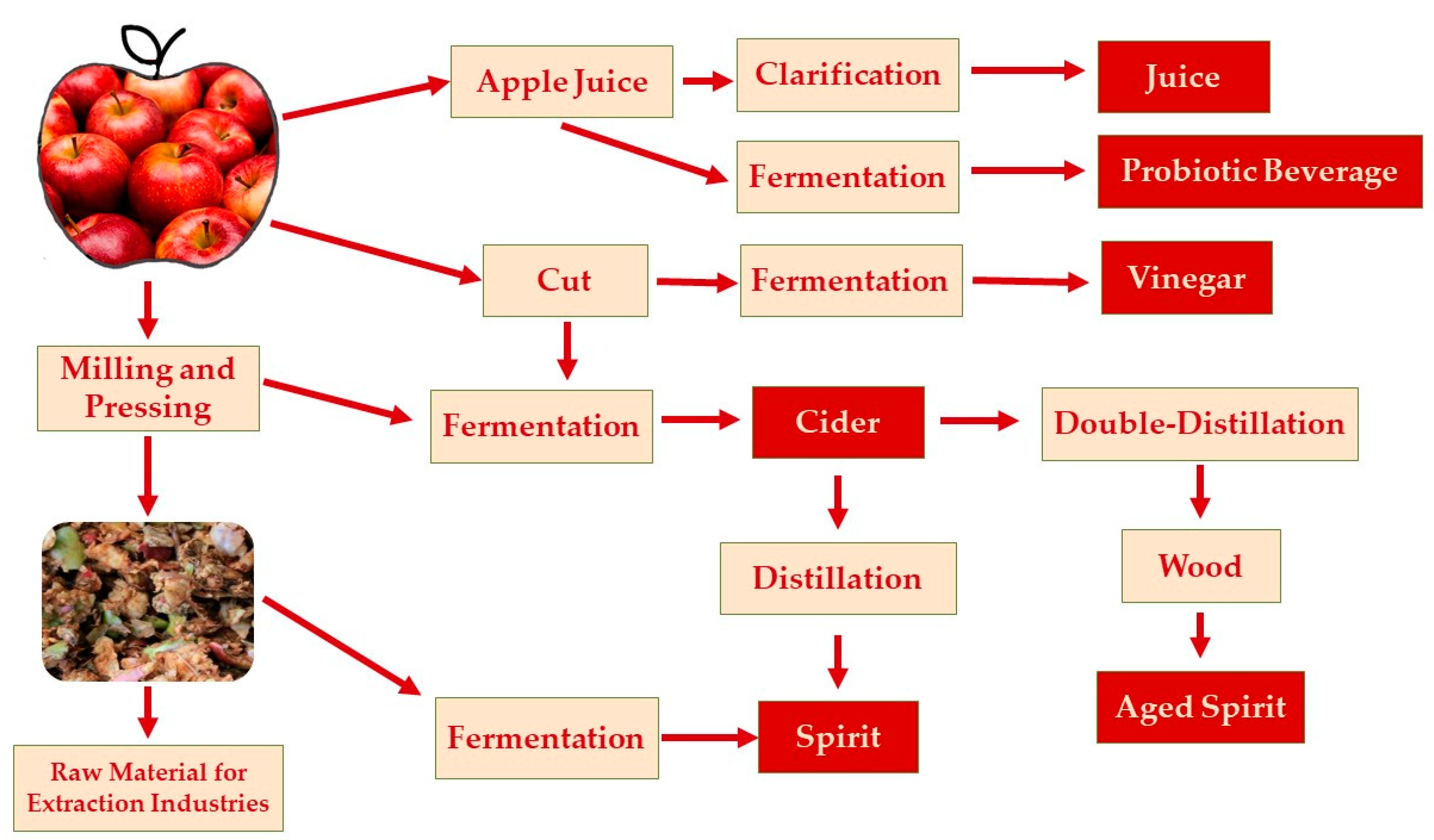
3. Fermentation Technology Applied to Apple Products
3.1. Apple Pomace
3.2. Cider
3.3. Vinegar
3.4. Apple Spirit
3.5. Probiotic Fermented Apples
4. Properties of Fermented Apple Products
5. Health Effects of Fermented Apple Products
6. Final Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Li, Y.; Zhang, X.; Nie, J.; Bacha, S.A.S.; Yan, Z.; Gao, G. Occurrence and co-occurrence of mycotoxins in apple and apple products from China. Food Control. 2020, 118, 107354. [Google Scholar] [CrossRef]
- Zhang, L.; Hu, J.; Han, X.; Li, J.; Gao, Y.; Richards, C.; Zhang, C.; Tian, Y.; Liu, G.; Gul, H.; et al. A high-quality apple genome assembly reveals the association of a retrotransposon and red fruit colour. Nat. Commun. 2019, 10, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Food and Agriculture Organization. FAOSTAT. Available online: http://www.fao.org/faostat/en/#data/QL (accessed on 5 November 2020).
- Bars-Cortina, D.; Macià, A.; Iglesias, I.; Garanto, X.; Badiella, L.; Motilva, M.J. Seasonal Variability of the Phytochemical Composition of New Red-Fleshed Apple Varieties Compared with Traditional and New White-Fleshed Varieties. J. Agric. Food Chem. 2018, 66, 10011–10025. [Google Scholar] [CrossRef] [PubMed]
- Bars-Cortina, D.; Martínez-Bardají, A.; Macià, A.; Motilva, M.J.; Piñol-Felis, C. Consumption evaluation of one apple flesh a day in the initial phases prior to adenoma/adenocarcinoma in an azoxymethane rat colon carcinogenesis model. J. Nutr. Biochem. 2020, 83, 108418. [Google Scholar] [CrossRef] [PubMed]
- Mohebbi, S.; Babalar, M.; Zamani, Z.; Askari, M.A. Influence of early season boron spraying and postharvest calcium dip treatment on cell-wall degrading enzymes and fruit firmness in ‘Starking Delicious’ apple during storage. Sci. Hortic. 2020, 259, 108822. [Google Scholar] [CrossRef]
- Bílková, A.; Baďurová, K.; Svobodová, P.; Vávra, R.; Jakubec, P.; Chocholouš, P.; Švec, F.; Sklenářová, H. Content of major phenolic compounds in apples: Benefits of ultra-low oxygen conditions in long-term storage. J. Food Compos. Anal. 2020, 92, 103587. [Google Scholar] [CrossRef]
- Pruksasri, S.; Lanner, B.; Novalin, S. Nanofiltration as a potential process for the reduction of sugar in apple juices on an industrial scale. LWT 2020, 133, 110118. [Google Scholar] [CrossRef]
- Sauceda-Gálvez, J.; Codina-Torrella, I.; Martinez-Garcia, M.; Hernández-Herrero, M.; Gervilla, R.; Roig-Sagués, A. Combined effects of ultra-high pressure homogenization and short-wave ultraviolet radiation on the properties of cloudy apple juice. LWT 2021, 136, 110286. [Google Scholar] [CrossRef]
- Kidoń, M.; Grabowska, J. Bioactive compounds, antioxidant activity, and sensory qualities of red-fleshed apples dried by different methods. LWT 2021, 136, 110302. [Google Scholar] [CrossRef]
- Feng, L.; Xu, Y.; Xiao, Y.; Song, J.; Li, D.; Zhang, Z.; Liu, C.; Liu, C.; Jiang, N.; Zhang, M.; et al. Effects of pre-drying treatments combined with explosion puffing drying on the physicochemical properties, antioxidant activities and flavor characteristics of apples. Food Chem. 2021, 338, 128015. [Google Scholar] [CrossRef]
- Cruz, A.C.; Guiné, R.P.; Gonçalves, J.C. Drying Kinetics and Product Quality for Convective Drying of Apples (cvs. Golden Delicious and Granny Smith). Int. J. Fruit Sci. 2015, 15, 54–78. [Google Scholar] [CrossRef]
- Guiné, R.P.; Cruz, A.C.; Mendes, M. Convective Drying of Apples: Kinetic Study, Evaluation of Mass Transfer Properties and Data Analysis using Artificial Neural Networks. Int. J. Food Eng. 2014, 10, 281–299. [Google Scholar] [CrossRef]
- Dobiáš, J.; Voldřich, M.; Čurda, D. Heating of canned fruits and vegetables: Deaeration and texture changes. J. Food Eng. 2006, 77, 421–425. [Google Scholar] [CrossRef]
- Lan, W.; Bureau, S.; Chen, S.; Leca, A.; Renard, C.M.; Jaillais, B. Visible, near- and mid-infrared spectroscopy coupled with an innovative chemometric strategy to control apple puree quality. Food Control. 2021, 120, 107546. [Google Scholar] [CrossRef]
- Lan, W.; Jaillais, B.; Leca, A.; Renard, C.M.; Bureau, S. A new application of NIR spectroscopy to describe and predict purees quality from the non-destructive apple measurements. Food Chem. 2020, 310, 125944. [Google Scholar] [CrossRef] [PubMed]
- Peng, W.; Meng, D.; Yue, T.; Wang, Z.; Gao, Z. Effect of the apple cultivar on cloudy apple juice fermented by a mixture of Lactobacillus acidophilus, Lactobacillus plantarum, and Lactobacillus fermentum. Food Chem. 2020, 340, 127922. [Google Scholar] [CrossRef]
- Roberts, D.; Reyes, V.; Bonilla, F.; Dzandu, B.; Liu, C.; Chouljenko, A.; Sathivel, S. Viability of Lactobacillus plantarum NCIMB 8826 in fermented apple juice under simulated gastric and intestinal conditions. LWT 2018, 97, 144–150. [Google Scholar] [CrossRef]
- Chen, C.; Lu, Y.; Yu, H.; Chen, Z.; Tian, H. Influence of 4 lactic acid bacteria on the flavor profile of fermented apple juice. Food Biosci. 2019, 27, 30–36. [Google Scholar] [CrossRef]
- Sousa, A.; Vareda, J.; Pereira, R.; Silva, C.; Câmara, J.S.; Câmara, J.S.; Perestrelo, R. Geographical differentiation of apple ciders based on volatile fingerprint. Food Res. Int. 2020, 137, 109550. [Google Scholar] [CrossRef]
- Lobo, A.P.; Bedriñana, R.P.; Madrera, R.R.; Valles, B.S. Aromatic, olfactometric and consumer description of sweet ciders obtained by cryo-extraction. Food Chem. 2021, 338, 127829. [Google Scholar] [CrossRef]
- Cusano, E.; Cagliani, L.R.; Consonni, R.; Simonato, B.; Zapparoli, G. NMR-based metabolic profiling of different yeast fermented apple juices. LWT 2020, 118, 108771. [Google Scholar] [CrossRef]
- Bortolini, D.G.; Benvenutti, L.; Demiate, I.M.; Nogueira, A.; Alberti, A.; Zielinski, A.A.F. A new approach to the use of apple pomace in cider making for the recovery of phenolic compounds. LWT 2020, 126, 109316. [Google Scholar] [CrossRef]
- Madrera, R.R.; Bedriñana, R.P.; Valles, B.S. Enhancement of the nutritional properties of apple pomace by fermentation with autochthonous yeasts. LWT-Food Sci. Technol. 2017, 79, 27–33. [Google Scholar] [CrossRef]
- Jin, Q.; Qureshi, N.; Wang, H.; Huang, H. Acetone-butanol-ethanol (ABE) fermentation of soluble and hydrolyzed sugars in apple pomace by Clostridium beijerinckii P. Fuel 2019, 244, 536–544. [Google Scholar] [CrossRef]
- Zheng, Z.; Shetty, K. Enhancement of pea (Pisum sativum) seedling vigour and associated phenolic content by extracts of apple pomace fermented with Trichoderma spp. Process. Biochem. 2000, 36, 79–84. [Google Scholar] [CrossRef]
- Katz, S.E.; Pollan, M. The Art of Fermentation: An In-Depth Exploration of Essential Concepts and Processes from around the World; Worth.it: White River Junction, VT, USA, 2012. [Google Scholar]
- Couto, S.R.; Sanromán, M. Ángeles Application of solid-state fermentation to food industry—A review. J. Food Eng. 2006, 76, 291–302. [Google Scholar] [CrossRef]
- García, C.; Rendueles, M.; Díaz, L.A.G. Liquid-phase food fermentations with microbial consortia involving lactic acid bacteria: A review. Food Res. Int. 2019, 119, 207–220. [Google Scholar] [CrossRef]
- Garro, M.S.; Rivas, F.P.; Garro, O.A. 3.10 - Solid State Fermentation in Food Processing: Advances in Reactor Design and Novel Applications. In Innovative Food Processing Technologies; Knoerzer, K., Muthukumarappan, K., Eds.; Elsevier: Oxford, UK, 2021; pp. 165–182. [Google Scholar]
- Hassan, A.; Nelson, B. Invited review: Anaerobic fermentation of dairy food wastewater. J. Dairy Sci. 2012, 95, 6188–6203. [Google Scholar] [CrossRef]
- Reihani, S.F.S.; Khosravi-Darani, K. Influencing factors on single-cell protein production by submerged fermentation: A review. Electron. J. Biotechnol. 2019, 37, 34–40. [Google Scholar] [CrossRef]
- Gavala, H.N.; Gavala, H.N.; Skiadas, I.V. Reactor systems for syngas fermentation processes: A review. Chem. Eng. J. 2018, 348, 732–744. [Google Scholar] [CrossRef]
- Zhou, M.; Yan, B.; Wong, J.; Zhang, Y. Enhanced volatile fatty acids production from anaerobic fermentation of food waste: A mini-review focusing on acidogenic metabolic pathways. Bioresour. Technol. 2018, 248, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Peris, M.; Escuder-Gilabert, L. On-line monitoring of food fermentation processes using electronic noses and electronic tongues: A review. Anal. Chim. Acta 2013, 804, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Herranz, B.; Fernández-Jalao, I.; Álvarez, M.D.; Quiles, A.; Sánchez-Moreno, C.; Hernando, I.; De Ancos, B. Phenolic compounds, microstructure and viscosity of onion and apple products subjected to in vitro gastrointestinal digestion. Innov. Food Sci. Emerg. Technol. 2019, 51, 114–125. [Google Scholar] [CrossRef]
- Wang, D.; Jiang, Y.; Sun-Waterhouse, D.-X.; Zhai, H.; Guan, H.; Rong, X.; Li, F.; Yu, J.-C.; Li, D. MicroRNA-based regulatory mechanisms underlying the synergistic antioxidant action of quercetin and catechin in H2O2-stimulated HepG2 cells: Roles of BACH1 in Nrf2-dependent pathways. Free. Radic. Biol. Med. 2020, 153, 122–131. [Google Scholar] [CrossRef] [PubMed]
- Tian, C.; Liu, X.; Chang, Y.; Wang, R.; Lv, T.; Cui, C.; Liu, M. Investigation of the anti-inflammatory and antioxidant activities of luteolin, kaempferol, apigenin and quercetin. South Afr. J. Bot. 2021, 137, 257–264. [Google Scholar] [CrossRef]
- Yong, H.; Bai, R.; Bi, F.; Liu, J.; Qin, Y.; Liu, J. Synthesis, characterization, antioxidant and antimicrobial activities of starch aldehyde-quercetin conjugate. Int. J. Biol. Macromol. 2020, 156, 462–470. [Google Scholar] [CrossRef]
- Dimpfel, W. Rat electropharmacograms of the flavonoids rutin and quercetin in comparison to those of moclobemide and clinically used reference drugs suggest antidepressive and/or neuroprotective action. Phytomedicine 2009, 16, 287–294. [Google Scholar] [CrossRef]
- Li, S.; Pei, Y.; Wang, W.; Liu, F.; Zheng, K.; Zhang, X. Quercetin suppresses the proliferation and metastasis of metastatic osteosarcoma cells by inhibiting parathyroid hormone receptor. Biomed. Pharmacother. 2019, 114, 108839. [Google Scholar] [CrossRef]
- Imran, M.; Iqubal, M.K.; Imtiyaz, K.; Saleem, S.; Mittal, S.; Rizvi, M.M.A.; Ali, J.; Baboota, S. Topical nanostructured lipid carrier gel of quercetin and resveratrol: Formulation, optimization, in vitro and ex vivo study for the treatment of skin cancer. Int. J. Pharm. 2020, 587, 119705. [Google Scholar] [CrossRef]
- Ishizawa, K.; Yoshizumi, M.; Kawai, Y.; Terao, J.; Kihira, Y.; Ikeda, Y.; Tomita, S.; Minakuchi, K.; Tsuchiya, K.; Tamaki, T. Pharmacology in Health Food: Metabolism of Quercetin In Vivo and Its Protective Effect Against Arteriosclerosis. J. Pharmacol. Sci. 2011, 115, 466–470. [Google Scholar] [CrossRef]
- Ebrahimpour, S.; Zakeri, M.; Esmaeili, A. Crosstalk between obesity, diabetes, and alzheimer’s disease: Introducing quercetin as an effective triple herbal medicine. Ageing Res. Rev. 2020, 62, 101095. [Google Scholar] [CrossRef] [PubMed]
- Sahoo, P.K.; Pradhan, L.K.; Aparna, S.; Agarwal, K.; Banerjee, A.; Das, S.K. Quercetin abrogates bisphenol A induced altered neurobehavioral response and oxidative stress in zebrafish by modulating brain antioxidant defence system. Environ. Toxicol. Pharmacol. 2020, 80, 103483. [Google Scholar] [CrossRef] [PubMed]
- DiNicolantonio, J.J.; Mccarty, M.F. Targeting Casein kinase 2 with quercetin or enzymatically modified isoquercitrin as a strategy for boosting the type 1 interferon response to viruses and promoting cardiovascular health. Med. Hypotheses 2020, 142, 109800. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Tao, B.; Wan, Y.; Sun, Y.; Wangab, L.; Sun, J.; Li, C. Drug delivery based pharmacological enhancement and current insights of quercetin with therapeutic potential against oral diseases. Biomed. Pharmacother. 2020, 128, 110372. [Google Scholar] [CrossRef] [PubMed]
- Boots, A.W.; Haenen, G.R.; Bast, A. Health effects of quercetin: From antioxidant to nutraceutical. Eur. J. Pharmacol. 2008, 585, 325–337. [Google Scholar] [CrossRef] [PubMed]
- Veeriah, S.; Miene, C.; Habermann, N.; Hofmann, T.; Klenow, S.; Sauer, J.; Böhmer, F.; Wölfl, S.; Pool-Zobel, B.L. Apple polyphenols modulate expression of selected genes related to toxicological defence and stress response in human colon adenoma cells. Int. J. Cancer 2008, 122, 2647–2655. [Google Scholar] [CrossRef]
- Chang, H.; Lei, L.; Zhou, Y.; Ye, F.; Zhao, G. Dietary Flavonoids and the Risk of Colorectal Cancer: An Updated Meta-Analysis of Epidemiological Studies. Nutrients 2018, 10, 950. [Google Scholar] [CrossRef]
- He, X.; Wang, Y.; Hu, H.; Zhang, Z. In Vitro and in Vivo Antimammary Tumor Activities and Mechanisms of the Apple Total Triterpenoids. J. Agric. Food Chem. 2012, 60, 9430–9436. [Google Scholar] [CrossRef]
- Chung, W.S.F.; Meijerink, M.; Zeuner, B.; Holck, J.; Louis, P.; Meyer, A.S.; Wells, J.M.; Flint, H.J.; Duncan, S.H. Prebiotic potential of pectin and pectic oligosaccharides to promote anti-inflammatory commensal bacteria in the human colon. FEMS Microbiol. Ecol. 2017, 93. [Google Scholar] [CrossRef]
- Li, Y.; Wang, S.; Sun, Y.; Xu, W.; Zheng, H.; Wang, Y.; Tang, Y.; Gao, X.; Song, C.; Long, Y.; et al. Apple polysaccharide protects ICR mice against colitis associated colorectal cancer through the regulation of microbial dysbiosis. Carbohydr. Polym. 2020, 230, 115726. [Google Scholar] [CrossRef]
- Williams, D.J.; Edwards, D.; Hamernig, I.; Jian, L.; James, A.P.; Johnson, S.K.; Tapsell, L.C. Vegetables containing phytochemicals with potential anti-obesity properties: A review. Food Res. Int. 2013, 52, 323–333. [Google Scholar] [CrossRef]
- Guiné, R.P.; Florença, S.G.; Barroca, M.J.; Anjos, O. The duality of innovation and food development versus purely traditional foods. Trends Food Sci. Technol. 2021, 109, 16–24. [Google Scholar] [CrossRef]
- Lyu, F.; Luiz, S.F.; Azeredo, D.R.P.; Cruz, A.G.; Ajlouni, S.; Ranadheera, C.S. Apple Pomace as a Functional and Healthy Ingredient in Food Products: A Review. Processes 2020, 8, 319. [Google Scholar] [CrossRef]
- Perussello, A.C.; Zhang, Z.; Marzocchella, A.; Tiwari, B.K. Valorization of Apple Pomace by Extraction of Valuable Compounds. Compr. Rev. Food Sci. Food Saf. 2017, 16, 776–796. [Google Scholar] [CrossRef]
- Roberto, R.M.; Bedriñana, R.P.; Hevia, A.G.; Arce, M.B.; Valles, B.S. Production of spirits from dry apple pomace and selected yeasts. Food Bioprod. Process. 2013, 91, 623–631. [Google Scholar] [CrossRef]
- May, C.D. Industrial pectins: Sources, production and applications. Carbohydr. Polym. 1990, 12, 79–99. [Google Scholar] [CrossRef]
- Medeiros, A.B.; Pandey, A.; Freitas, R.J.; Christen, P.; Soccol, C.R. Optimization of the production of aroma compounds by Kluyveromyces marxianus in solid-state fermentation using factorial design and response surface methodology. Biochem. Eng. J. 2000, 6, 33–39. [Google Scholar] [CrossRef]
- Grigelmo-Miguel, N.; Martín-Belloso, O. Comparison of Dietary Fibre from By-products of Processing Fruits and Greens and from Cereals. LWT Food Sci. Technol. 1999, 32, 503–508. [Google Scholar] [CrossRef]
- Masoodi, F.; Sharma, B.; Chauhan, G. Use of apple pomace as a source of dietary fiber in cakes. Plant Foods Hum. Nutr. 2002, 57, 121–128. [Google Scholar] [CrossRef]
- García, Y.D.; Valles, B.S.; Lobo, A.P. Phenolic and antioxidant composition of by-products from the cider industry: Apple pomace. Food Chem. 2009, 117, 731–738. [Google Scholar] [CrossRef]
- Alibés, X.; Muñoz, F.; Rodriguez, J. Feeding value of apple pomace silage for sheep. Anim. Feed. Sci. Technol. 1984, 11, 189–197. [Google Scholar] [CrossRef]
- Berovič, M.; Ostroveršnik, H. Production of Aspergillus niger pectolytic enzymes by solid state bioprocessing of apple pomace. J. Biotechnol. 1997, 53, 47–53. [Google Scholar] [CrossRef]
- Daigle, P.; Gélinas, P.; Leblanc, D.; Morin, A. Production of aroma compounds by Geotrichum candidum on waste bread crumb. Food Microbiol. 1999, 16, 517–522. [Google Scholar] [CrossRef]
- Villas-Boas, S.G.; Esposito, E.; De Mendonça, M.M. Novel lignocellulolytic ability of Candida utilis during solid-substrate cultivation on apple pomace. World J. Microbiol. Biotechnol. 2002, 18, 541–545. [Google Scholar] [CrossRef]
- Jin, H.; Kim, H.-S.; Kim, S.-K.; Shin, M.-K.; Kim, J.-H.; Lee, J.-W. Production of heteropolysaccharide-7 by Beijerinckia indica from agro-industrial byproducts. Enzym. Microb. Technol. 2002, 30, 822–827. [Google Scholar] [CrossRef]
- Canteri-Schemin, M.H.; Fertonani, H.C.R.; Waszczynskyj, N.; Wosiacki, G. Extraction of pectin from apple pomace. Braz. Arch. Biol. Technol. 2005, 48, 259–266. [Google Scholar] [CrossRef]
- Bhushan, S.; Joshi, V. Baker’s Yeast Production under Fed Batch Culture from Apple Pomace. J. Sci. Ind. Res. 2006, 65, 72–76. [Google Scholar]
- Wang, X.; Kristo, E.; Lapointe, G. The effect of apple pomace on the texture, rheology and microstructure of set type yogurt. Food Hydrocoll. 2019, 91, 83–91. [Google Scholar] [CrossRef]
- Dhillon, G.S.; Kaur, S.; Brar, S.K. Perspective of apple processing wastes as low-cost substrates for bioproduction of high value products: A review. Renew. Sustain. Energy Rev. 2013, 27, 789–805. [Google Scholar] [CrossRef]
- Ricci, A.; Cirlini, M.; Guido, A.; Liberatore, C.M.; Ganino, T.; Lazzi, C.; Chiancone, B. From Byproduct to Resource: Fermented Apple Pomace as Beer Flavoring. Foods 2019, 8, 309. [Google Scholar] [CrossRef]
- Li, S.; Nie, Y.; Ding, Y.; Zhao, J.; Tang, X. Effects of Pure and Mixed Koji Cultures with S accharomyces cerevisiae on Apple Homogenate Cider Fermentation. J. Food Process. Preserv. 2015, 39, 2421–2430. [Google Scholar] [CrossRef]
- Madrera, R.R.; Bedriñana, R.P.; Valles, B.S. Production and characterization of aroma compounds from apple pomace by solid-state fermentation with selected yeasts. LWT 2015, 64, 1342–1353. [Google Scholar] [CrossRef]
- Silva, M.; Macedo, A.; Malcata, F. Review: Steam distilled spirits from fermented grape pomace Revision: Bebidas destiladas obtenidas de la fermentación del orujo de uva. Food Sci. Technol. Int. 2000, 6, 285–300. [Google Scholar] [CrossRef]
- Botelho, G.; Anjos, O.; Estevinho, L.M.; Caldeira, I. Methanol in Grape Derived, Fruit and Honey Spirits: A Critical Review on Source, Quality Control, and Legal Limits. Processes 2020, 8, 1609. [Google Scholar] [CrossRef]
- AICV. European Cider Trends 2019; The European Cider & Fruit Wine Association: Brussels, Belgium, 2019. [Google Scholar]
- Nicolini, G.; Roman, T.; Carlin, S.; Malacarne, M.; Nardin, T.; Cossignani, L.; Larcher, R. Characterisation of single-variety still ciders produced with dessert apples in the Italian Alps. J. Inst. Brew. 2018, 124, 457–466. [Google Scholar] [CrossRef]
- Ye, M.; Yue, T.; Yuan, Y. Changes in the profile of volatile compounds and amino acids during cider fermentation using dessert variety of apples. Eur. Food Res. Technol. 2014, 239, 67–77. [Google Scholar] [CrossRef]
- Valles, B.S.; Bedriñana, R.P.; Queipo, A.L.; Alonso, J.J.M. Screening of cider yeasts for sparkling cider production (Champenoise method). Food Microbiol. 2008, 25, 690–697. [Google Scholar] [CrossRef]
- Lobo, A.P.; Tascón, N.F.; Roberto, R.M.; Valles, B.S. Sensory and Foaming Properties of Sparkling Cider. J. Agric. Food Chem. 2005, 53, 10051–10056. [Google Scholar] [CrossRef]
- Gomis, D.B.; Tamayo, D.M.; Valles, B.S.; Alonso, J.J.M. Detection of Apple Juice Concentrate in the Manufacture of Natural and Sparkling Cider by Means of HPLC Chemometric Sugar Analyses. J. Agric. Food Chem. 2004, 52, 201–203. [Google Scholar] [CrossRef][Green Version]
- Rosend, J.; Kaleda, A.; Kuldjärv, R.; Arju, G.; Nisamedtinov, I. The Effect of Apple Juice Concentration on Cider Fermentation and Properties of the Final Product. Foods 2020, 9, 1401. [Google Scholar] [CrossRef]
- Tarko, T.; Duda-Chodak, A.; Sroka, P.; Januszek, M. Effect of Musts Oxygenation at Various Stages of Cider Production on Oenological Parameters, Antioxidant Activity, and Profile of Volatile Cider Compounds. Biomolecules 2020, 10, 890. [Google Scholar] [CrossRef] [PubMed]
- Guyot, S.; Marnet, N.; Sanoner, P.; Drilleau, J.-F. Variability of the Polyphenolic Composition of Cider Apple (Malus domestica) Fruits and Juices. J. Agric. Food Chem. 2003, 51, 6240–6247. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, A.; Mongruel, C.; Simões, D.R.S.; Waszczynskyj, N.; Wosiacki, G. Effect of biomass reduction on the fermentation of cider. Braz. Arch. Biol. Technol. 2007, 50, 1083–1092. [Google Scholar] [CrossRef]
- Al Daccache, M.; Koubaa, M.; Salameh, D.; Maroun, R.G.; Louka, N.; Vorobiev, E. Ultrasound-assisted fermentation for cider production from Lebanese apples. Ultrason. Sonochem. 2020, 63, 104952. [Google Scholar] [CrossRef]
- Lorenzini, M.; Simonato, B.; Slaghenaufi, D.; Ugliano, M.; Zapparoli, G. Assessment of yeasts for apple juice fermentation and production of cider volatile compounds. LWT 2019, 99, 224–230. [Google Scholar] [CrossRef]
- Laaksonen, O.; Kuldjärv, R.; Paalme, T.; Virkki, M.; Yang, B. Impact of apple cultivar, ripening stage, fermentation type and yeast strain on phenolic composition of apple ciders. Food Chem. 2017, 233, 29–37. [Google Scholar] [CrossRef]
- Cousin, F.J.; Le Guellec, R.; Schlusselhuber, M.; Dalmasso, M.; LaPlace, J.M.; Cretenet, M. Microorganisms in Fermented Apple Beverages: Current Knowledge and Future Directions. Microorganisms 2017, 5, 39. [Google Scholar] [CrossRef]
- Abrahamse, C.E.; Bartowsky, E. Timing of malolactic fermentation inoculation in Shiraz grape must and wine: Influence on chemical composition. World J. Microbiol. Biotechnol. 2011, 28, 255–265. [Google Scholar] [CrossRef]
- Sumby, K.M.; Grbin, P.R.; Jiranek, V. Implications of new research and technologies for malolactic fermentation in wine. Appl. Microbiol. Biotechnol. 2014, 98, 8111–8132. [Google Scholar] [CrossRef]
- Sumby, K.M.; Bartle, L.; Grbin, P.R.; Jiranek, V. Measures to improve wine malolactic fermentation. Appl. Microbiol. Biotechnol. 2019, 103, 2033–2051. [Google Scholar] [CrossRef]
- Betteridge, A.; Grbin, P.; Jiranek, V. Improving Oenococcus oeni to overcome challenges of wine malolactic fermentation. Trends Biotechnol. 2015, 33, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Bartowsky, E.; Costello, P.; Chambers, P. Emerging trends in the application of malolactic fermentation. Aust. J. Grape Wine Res. 2015, 21, 663–669. [Google Scholar] [CrossRef]
- Sánchez, A.; Coton, M.; Coton, E.; Herrero, M.; García, L.A.; Díaz, M. Prevalent lactic acid bacteria in cider cellars and efficiency of Oenococcus oeni strains. Food Microbiol. 2012, 32, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, A.; Rodríguez, R.; Coton, M.; Coton, E.; Herrero, M.; García, L.A.; Díaz, M. Population dynamics of lactic acid bacteria during spontaneous malolactic fermentation in industrial cider. Food Res. Int. 2010, 43, 2101–2107. [Google Scholar] [CrossRef]
- Reuss, R.; Stratton, J.; Smith, D.; Read, P.; Cuppett, S.; Parkhurst, A. Malolactic Fermentation as a Technique for the Deacidification of Hard Apple Cider. J. Food Sci. 2010, 75, C74–C78. [Google Scholar] [CrossRef]
- Li, C.X.; Zhao, X.H.; Zuo, W.F.; Zhang, T.L.; Zhang, Z.Y.; Chen, X. The effects of simultaneous and sequential inoculation of yeast and autochthonous Oenococcus oeni on the chemical composition of red-fleshed apple cider. LWT 2020, 124, 109184. [Google Scholar] [CrossRef]
- Fan, W.; Xu, Y.; Yu, A. Influence of Oak Chips Geographical Origin, Toast Level, Dosage and Aging Time on Volatile Compounds of Apple Cider. J. Inst. Brew. 2006, 112, 255–263. [Google Scholar] [CrossRef]
- Budak, H.N.; Guzel-Seydim, Z.B. Antioxidant activity and phenolic content of wine vinegars produced by two different techniques. J. Sci. Food Agric. 2010, 90, 2021–2026. [Google Scholar] [CrossRef]
- Mudura, E.; Coldea, T.; Socaciu, C.; Ranga, F.; Pop, C.; Rotar, A.; Pasqualone, A. Brown beer vinegar: A potentially functional product based on its phenolic profile and antioxidant activity. J. Serbian Chem. Soc. 2018, 83, 19–30. [Google Scholar] [CrossRef]
- Bakir, S.; Toydemir, G.; Boyacioglu, D.; Beekwilder, J.; Capanoglu, E. Fruit Antioxidants during Vinegar Processing: Changes in Content and in Vitro Bio-Accessibility. Int. J. Mol. Sci. 2016, 17, 1658. [Google Scholar] [CrossRef] [PubMed]
- Joshi, V.K.; Sharma, S. Cider Vinegar: Microbiology, Technology and QualityVinegars of the World. In Vinegars of the World; Springer: Milan, Italy, 2009; pp. 196–207. ISBN 978-88-470-0866-3. [Google Scholar]
- Alkorta, I.; Garbisu, C.; Llama, M.J.; Serra, J.L. Industrial applications of pectic enzymes: A review. Process. Biochem. 1998, 33, 21–28. [Google Scholar] [CrossRef]
- Budak, N.H.; Aykin, E.; Seydim, A.C.; Greene, A.K.; Guzel-Seydim, Z.B. Functional Properties of Vinegar. J. Food Sci. 2014, 79, R757–R764. [Google Scholar] [CrossRef] [PubMed]
- EC. Regulation (EC) No 110/2008 of the European Parliament and of the Council of 15 January 2008 on the Definition, Description, Presentation, Labelling and the Protection of Geographical Indications of Spirit Drinks and Repealing Council Regulation (EEC) No 1576/89; European Commission: Brussels, Belgium, 2008. [Google Scholar]
- Januszek, M.; Satora, P.; Tarko, T. Oenological Characteristics of Fermented Apple Musts and Volatile Profile of Brandies Obtained from Different Apple Cultivars. Biomolecules 2020, 10, 853. [Google Scholar] [CrossRef] [PubMed]
- Hang, Y.D.; Woodams, E.E. Influence of apple cultivar and juice pasteurization on hard cider and eau-de-vie methanol content. Bioresour. Technol. 2010, 101, 1396–1398. [Google Scholar] [CrossRef] [PubMed]
- Versini, G.; Franco, M.; Moser, S.; Barchetti, P.; Manca, G. Characterisation of apple distillates from native varieties of Sardinia island and comparison with other Italian products. Food Chem. 2009, 113, 1176–1183. [Google Scholar] [CrossRef]
- Coldea, T.E.; Socaciu, C.; Mudura, E.; Socaci, S.A.; Ranga, F.; Pop, C.R.; Vriesekoop, F.; Pasqualone, A. Volatile and phenolic profiles of traditional Romanian apple brandy after rapid ageing with different wood chips. Food Chem. 2020, 320, 126643. [Google Scholar] [CrossRef]
- Small, R.W.; Couturier, M.; Godfrey, M. Beverage Basics: Understanding and Appreciating Wine, Beer, and Spirits, 1st ed.; Wiley: Hoboken, NJ, USA, 2011. [Google Scholar]
- Roberto, R.M.; Lobo, A.P.; Alonso, J.J.M. Effect of cider maturation on the chemical and sensory characteristics of fresh cider spirits. Food Res. Int. 2010, 43, 70–78. [Google Scholar] [CrossRef]
- Zhang, H.; Woodams, E.E.; Hang, Y.D. Influence of pectinase treatment on fruit spirits from apple mash, juice and pomace. Process. Biochem. 2011, 46, 1909–1913. [Google Scholar] [CrossRef]
- Anjos, O.; Santos, A.J.; Estevinho, L.M.; Caldeira, I. FTIR–ATR spectroscopy applied to quality control of grape-derived spirits. Food Chem. 2016, 205, 28–35. [Google Scholar] [CrossRef]
- Iyer, B.K.; Ananthanarayan, L. Effect of α-amylase addition on fermentation of idli—A popular south Indian cereal—Legume-based snack food. LWT 2008, 41, 1053–1059. [Google Scholar] [CrossRef]
- Daeschel, M.A. Antimicrobial Substances from Lactic Acid Bacteria for Use as Food Preservatives. Food Technol. 1989, 43, 164–167. [Google Scholar]
- Leroy, F.; De Vuyst, L. Lactic acid bacteria as functional starter cultures for the food fermentation industry. Trends Food Sci. Technol. 2004, 15, 67–78. [Google Scholar] [CrossRef]
- Gomes, A.M.P.; Malcata, F.X. Bifidobacterium spp. and Lactobacillus acidophilus: Biological, biochemical, technological and therapeutical properties relevant for use as probiotics. Trends Food Sci. Technol. 1999, 10, 139–157. [Google Scholar] [CrossRef]
- Ellendersen, L.D.S.N.; Granato, D.; Guergoletto, K.B.; Wosiacki, G. Development and sensory profile of a probiotic beverage from apple fermented with Lactobacillus casei. Eng. Life Sci. 2012, 12, 475–485. [Google Scholar] [CrossRef]
- Dimitrovski, D.; Velickova, E.; Langerholc, T.; Winkelhausen, E. Apple juice as a medium for fermentation by the probiotic Lactobacillus plantarum PCS 26 strain. Ann. Microbiol. 2015, 65, 2161–2170. [Google Scholar] [CrossRef]
- Braga, C.M.; Zielinski, A.A.F.; Da Silva, K.M.; De Souza, F.K.F.; Pietrowski, G.D.A.M.; Couto, M.; Granato, D.; Wosiacki, G.; Nogueira, A. Classification of juices and fermented beverages made from unripe, ripe and senescent apples based on the aromatic profile using chemometrics. Food Chem. 2013, 141, 967–974. [Google Scholar] [CrossRef] [PubMed]
- Costa, M.G.M.; Fonteles, T.V.; De Jesus, A.L.T.; Rodrigues, S. Sonicated pineapple juice as substrate for L. casei cultivation for probiotic beverage development: Process optimisation and product stability. Food Chem. 2013, 139, 261–266. [Google Scholar] [CrossRef]
- Pereira, A.L.F.; Maciel, T.C.; Rodrigues, S. Probiotic beverage from cashew apple juice fermented with Lactobacillus casei. Food Res. Int. 2011, 44, 1276–1283. [Google Scholar] [CrossRef]
- Veron Ponce, H.E.; Gauffin Cano, M.P.; Fabersani Marrades, M.E.; Sanz, Y.; Isla, M.I.; Fernández Espinar, M.T.; Gil Ponce, J.V.; Torres Castaños, S. Cactus Pear (Opuntia Ficus-Indica) Juice Fermented with Autochthonous Lactobacillus Plantarum S-811. Food Funct. 2019. [Google Scholar] [CrossRef]
- Wu, C.; Li, T.; Qi, J.; Jiang, T.; Xu, H.; Lei, H. Effects of lactic acid fermentation-based biotransformation on phenolic profiles, antioxidant capacity and flavor volatiles of apple juice. LWT 2020, 122, 109064. [Google Scholar] [CrossRef]
- Filannino, P.; Azzi, L.; Cavoski, I.; Vincentini, O.; Rizzello, C.G.; Gobbetti, M.; Cagno, R. Exploitation of the health-promoting and sensory properties of organic pomegranate (Punica granatum L.) juice through lactic acid fermentation. Int. J. Food Microbiol. 2013, 163, 184–192. [Google Scholar] [CrossRef] [PubMed]
- Kaprasob, R.; Kerdchoechuen, O.; Laohakunjit, N.; Sarkar, D.; Shetty, K. Fermentation-based biotransformation of bioactive phenolics and volatile compounds from cashew apple juice by select lactic acid bacteria. Process. Biochem. 2017, 59, 141–149. [Google Scholar] [CrossRef]
- Jaros, D.; Thamke, I.; Raddatz, H.; Rohm, H. Single-cultivar cloudy juice made from table apples: An attempt to identify the driving force for sensory preference. Eur. Food Res. Technol. 2009, 229, 51–61. [Google Scholar] [CrossRef]
- Gao, H.; Wen, J.-J.; Hu, J.-L.; Nie, Q.-X.; Chen, H.-H.; Nie, S.-P.; Xiong, T.; Xie, M.-Y. Momordica charantia juice with Lactobacillus plantarum fermentation: Chemical composition, antioxidant properties and aroma profile. Food Biosci. 2019, 29, 62–72. [Google Scholar] [CrossRef]
- Qin, Z.; Petersen, M.A.; Bredie, W.L. Flavor profiling of apple ciders from the UK and Scandinavian region. Food Res. Int. 2018, 105, 713–723. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Deng, J.; Bi, J.; Wu, X.; Zhang, B. Cultivar classification of cloudy apple juices from substandard fruits in China based on aroma profile analyzed by HS-SPME/GC-MS. LWT 2019, 102, 304–309. [Google Scholar] [CrossRef]
- Ricci, A.; Cirlini, M.; Levante, A.; Dall’Asta, C.; Galaverna, G.; Lazzi, C. Volatile profile of elderberry juice: Effect of lactic acid fermentation using L. plantarum, L. rhamnosus and L. casei strains. Food Res. Int. 2018, 105, 412–422. [Google Scholar] [CrossRef]
- Ziadi, M.; Wathelet, J.P.; Marlier, M.; Hamdi, M.; Thonart, P. Analysis of Volatile Compounds Produced by 2 Strains ofLactococcus lactisIsolated from Leben (Tunisian Fermented Milk) Using Solid-Phase Microextraction-Gas Chromatography. J. Food Sci. 2008, 73, S247–S252. [Google Scholar] [CrossRef]
- Andreu-Sevilla, A.J.; Mena, P.; Martí, N.; Viguera, C.G.; Carbonell-Barrachina, A. Volatile composition and descriptive sensory analysis of pomegranate juice and wine. Food Res. Int. 2013, 54, 246–254. [Google Scholar] [CrossRef]
- Leguerinel, I.; Cleret, J.J.; Bourgeois, C.; Mafart, P. Yeast Strain and the Formation of Flavour Components In Cider. J. Inst. Brew. 1988, 94, 391–395. [Google Scholar] [CrossRef]
- Mangas, J.J.; González, M.P.; Rodriguez, R.; Blanco, D. Solid-phase extraction and determination of trace aroma and flavour components in cider by GC-MS. Chromatographia 1996, 42, 101–105. [Google Scholar] [CrossRef]
- Peng, B.; Li, F.; Cui, L.; Guo, Y. Effects of Fermentation Temperature on Key Aroma Compounds and Sensory Properties of Apple Wine. J. Food Sci. 2015, 80, S2937–S2943. [Google Scholar] [CrossRef] [PubMed]
- Peng, B.; Yue, T.; Yuan, Y. Analysis of key aroma components in cider from Shaanxi (China)Fujiapple. Int. J. Food Sci. Technol. 2009, 44, 610–615. [Google Scholar] [CrossRef]
- Riekstina-Dolge, R.; Kruma, Z.; Karklina, D.; Seglina, D. Influence of Different Yeast Strains on the Production of Volatile Compounds in Fermented Apple Juice. Res. Rural. Dev. Int. Sci. Conf. 2011, 1, 133–139. [Google Scholar]
- Al Daccache, M.; Koubaa, M.; Maroun, R.G.; Salameh, D.; Louka, N.; Vorobiev, E. Impact of the Physicochemical Composition and Microbial Diversity in Apple Juice Fermentation Process: A Review. Molecules 2020, 25, 3698. [Google Scholar] [CrossRef] [PubMed]
- Aung, M.T.; Lee, P.-R.; Yu, B.; Liu, S.-Q. Cider fermentation with three Williopsis saturnus yeast strains and volatile changes. Ann. Microbiol. 2014, 65, 921–928. [Google Scholar] [CrossRef]
- Peng, B.; Yue, T.; Yuan, Y. A fuzzy comprehensive evaluation for selecting yeast for cider making. Int. J. Food Sci. Technol. 2008, 43, 140–144. [Google Scholar] [CrossRef]
- Wang, L.; Xu, Y.; Zhao, G.; Li, J. Rapid Analysis of Flavor Volatiles in Apple Wine Using Headspace Solid-Phase Microextraction. J. Inst. Brew. 2004, 110, 57–65. [Google Scholar] [CrossRef]
- Rita, R.-D.; Zanda, K.; Daina, K.; Dalija, S. Composition of aroma compounds in fermented apple juice: Effect of apple variety, fermentation temperature and inoculated yeast concentration. Procedia Food Sci. 2011, 1, 1709–1716. [Google Scholar] [CrossRef]
- Haider, W.; Barillier, D.; Hayat, A.; Gaillard, J.-L.; Ledauphin, J. Rapid quantification and comparison of major volatile compounds of ciders from France (Normandy and Brittany) using microextraction by packed sorbent (MEPS). Anal. Methods 2014, 6, 1364–1376. [Google Scholar] [CrossRef]
- Lobo, A.P.; Antón-Díaz, M.J.; Alonso, J.J.M.; Valles, B.S. Characterization of Spanish ciders by means of chemical and olfactometric profiles and chemometrics. Food Chem. 2016, 213, 505–513. [Google Scholar] [CrossRef] [PubMed]
- Vidrih, R.; Hribar, J. Synthesis of higher alcohols during cider processing. Food Chem. 1999, 67, 287–294. [Google Scholar] [CrossRef]
- Picinelli, A.; Suárez, B.; Moreno, J.; Rodríguez, R.; Caso-García, L.M.; Mangas, J.J. Chemical Characterization of Asturian Cider. J. Agric. Food Chem. 2000, 48, 3997–4002. [Google Scholar] [CrossRef] [PubMed]
- Ye, M.; Yue, T.; Yuan, Y. Evolution of polyphenols and organic acids during the fermentation of apple cider. J. Sci. Food Agric. 2014, 94, 2951–2957. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Yue, T.; Yuan, Y. Impact of polyphenols on the headspace concentration of aroma compounds in apple cider. J. Sci. Food Agric. 2019, 99, 1635–1642. [Google Scholar] [CrossRef] [PubMed]
- Lea, A.G.H.; Arnold, G.M. The phenolics of ciders: Bitterness and astringency. J. Sci. Food Agric. 1978, 29, 478–483. [Google Scholar] [CrossRef]
- Symoneaux, R.; Baron, A.; Marnet, N.; Bauduin, R.; Chollet, S. Impact of apple procyanidins on sensory perception in model cider (part 1): Polymerisation degree and concentration. LWT Food Sci. Technol. 2014, 57, 22–27. [Google Scholar] [CrossRef]
- Verdu, C.F.; Childebrand, N.; Marnet, N.; LeBail, G.; Dupuis, F.; Laurens, F.; Guilet, D.; Guyot, S. Polyphenol variability in the fruits and juices of a cider apple progeny. J. Sci. Food Agric. 2013, 94, 1305–1314. [Google Scholar] [CrossRef]
- Lachowicz, S.; Oszmiański, J.; Uździcka, M.; Chmielewska, J. The Influence of Yeast Strain, β-Cyclodextrin, and Storage Time on Concentrations of Phytochemical Components, Sensory Attributes, and Antioxidative Activity of Novel Red Apple Ciders. Molecules 2019, 24, 2477. [Google Scholar] [CrossRef]
- Marks, S.C.; Mullen, W.; Crozier, A. Flavonoid and Hydroxycinnamate Profiles of English Apple Ciders. J. Agric. Food Chem. 2007, 55, 8723–8730. [Google Scholar] [CrossRef]
- Lobo, A.P.; García, Y.D.; Mangas-Sanchez, J.; Roberto, R.M.; Valles, B.S. Phenolic and antioxidant composition of cider. J. Food Compos. Anal. 2009, 22, 644–648. [Google Scholar] [CrossRef]
- Buron, N.; Guichard, H.; Coton, E.; Ledauphin, J.; Barillier, D. Evidence of 4-ethylcatechol as one of the main phenolic off-flavour markers in French ciders. Food Chem. 2011, 125, 542–548. [Google Scholar] [CrossRef]
- Buron, N.; Coton, M.; Legendre, P.; Ledauphin, J.; Kientz-Bouchart, V.; Guichard, H.; Barillier, D.; Coton, E. Implications of Lactobacillus collinoides and Brettanomyces/Dekkera anomala in phenolic off-flavour defects of ciders. Int. J. Food Microbiol. 2012, 153, 159–165. [Google Scholar] [CrossRef]
- Tripathi, S.; Mazumder, P.M. Apple Cider Vinegar (ACV) and their Pharmacological Approach towards Alzheimer’s Disease (AD): A Review. Indian J. Pharm. Educ. Res. 2020, 54, s67–s74. [Google Scholar] [CrossRef]
- Qi, Z.; Dong, D.; Yang, H.; Xia, X. Improving fermented quality of cider vinegar via rational nutrient feeding strategy. Food Chem. 2017, 224, 312–319. [Google Scholar] [CrossRef] [PubMed]
- Saadat, Y.R.; Khosroushahi, A.Y.; Gargari, B.P. A comprehensive review of anticancer, immunomodulatory and health beneficial effects of the lactic acid bacteria exopolysaccharides. Carbohydr. Polym. 2019, 217, 79–89. [Google Scholar] [CrossRef] [PubMed]
- Abid, Y.; Casillo, A.; Gharsallah, H.; Joulak, I.; Lanzetta, R.; Corsaro, M.M.; Attia, H.; Azabou, S. Production and structural characterization of exopolysaccharides from newly isolated probiotic lactic acid bacteria. Int. J. Biol. Macromol. 2018, 108, 719–728. [Google Scholar] [CrossRef] [PubMed]
- Oleksy, M.; Klewicka, E. Exopolysaccharides produced byLactobacillussp.: Biosynthesis and applications. Crit. Rev. Food Sci. Nutr. 2016, 58, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Nwodo, U.U.; Green, E.; Okoh, A.I. Bacterial Exopolysaccharides: Functionality and Prospects. Int. J. Mol. Sci. 2012, 13, 14002–14015. [Google Scholar] [CrossRef]
- Cirrincione, S.; Breuer, Y.; Mangiapane, E.; Mazzoli, R.; Pessione, E. ‘Ropy’ phenotype, exopolysaccharides and metabolism: Study on food isolated potential probiotics LAB. Microbiol. Res. 2018, 214, 137–145. [Google Scholar] [CrossRef]
- Halima, B.H.; Sonia, G.; Sarra, K.; Houda, B.J.; Fethi, B.S.; Abdallah, A. Apple Cider Vinegar Attenuates Oxidative Stress and Reduces the Risk of Obesity in High-Fat-Fed Male Wistar Rats. J. Med. Food 2018, 21, 70–80. [Google Scholar] [CrossRef] [PubMed]
- Mota, A.C.L.G.; De Castro, R.D.; Oliveira, J.D.A.; Lima, E.D.O. Antifungal Activity of Apple Cider Vinegar on Candida Species Involved in Denture Stomatitis. J. Prosthodont. 2015, 24, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Budak, N.H.; Doguc, D.K.; Savas, C.M.; Seydim, A.C.; Tas, T.K.; Ciris, M.I.; Guzel-Seydim, Z.B. Effects of Apple Cider Vinegars Produced with Different Techniques on Blood Lipids in High-Cholesterol-Fed Rats. J. Agric. Food Chem. 2011, 59, 6638–6644. [Google Scholar] [CrossRef] [PubMed]
- Khezri, S.S.; Saidpour, A.; Hosseinzadeh, N.; Amiri, Z. Beneficial effects of Apple Cider Vinegar on weight management, Visceral Adiposity Index and lipid profile in overweight or obese subjects receiving restricted calorie diet: A randomized clinical trial. J. Funct. Foods 2018, 43, 95–102. [Google Scholar] [CrossRef]
- Grosso, G.; Stepaniak, U.; Micek, A.; Stefler, D.; Bobak, M.; Pająk, A. Dietary polyphenols are inversely associated with metabolic syndrome in Polish adults of the HAPIEE study. Eur. J. Nutr. 2017, 56, 1409–1420. [Google Scholar] [CrossRef] [PubMed]
- Pounis, G.; Bonaccio, M.; Di Castelnuovo, A.; Costanzo, S.; De Curtis, A.; Persichillo, M.; Sieri, S.; Donati, M.B.; Cerletti, C.; De Gaetano, G.; et al. Polyphenol intake is associated with low-grade inflammation, using a novel data analysis from the Moli-sani study. Thromb. Haemost. 2016, 115, 344–352. [Google Scholar] [CrossRef]
- Salau, V.F.; Erukainure, O.L.; Islam, M.S. Chapter 29-Phenolics: Therapeutic applications against oxidative injury in obesity and type 2 diabetes pathology. In Pathology; Preedy, V.R., Ed.; Academic Press: London, UK, 2020; pp. 297–307. [Google Scholar]
- Sugiyama, S.; Kishi, M.; Fushimi, T.; Oshima, Y.; Kaga, T.; Kajimoto, O. Hypotensive Effect and Safety of Brown Rice Vinegar with High Concentration of GABA on Mild Hypertensive Subjects. Jpn. Pharmacol. Ther. 2008, 36, 429–444. [Google Scholar]
- Kajimoto, O.; Ueno, H.; Nagata, Y.; Yabune, M.; Kajimoto, Y. Hypotensive Effects of Tablet Containing γ-Amino Butyric Acid (GABA) on High Normal Blood Pressure and Mild Hypertensive Subjects. Jpn. Pharmacol. Ther. 2004, 32, 929–944. [Google Scholar]
- Zhang, S.; Hu, C.Y.; Guo, Y.; Wang, X.; Meng, Y. Polyphenols in fermented apple juice: Beneficial effects on human health. J. Funct. Foods 2021, 76, 104294. [Google Scholar] [CrossRef]
- Gheflati, A.; Bashiri, R.; Ghadiri-Anari, A.; Reza, J.Z.; Kord, M.T.; Nadjarzadeh, A.; Bashiri, R. The effect of apple vinegar consumption on glycemic indices, blood pressure, oxidative stress, and homocysteine in patients with type 2 diabetes and dyslipidemia: A randomized controlled clinical trial. Clin. Nutr. ESPEN 2019, 33, 132–138. [Google Scholar] [CrossRef]
- Shishehbor, F.; Mansoori, A.; Sarkaki, A.; Jalali, M.; Latifi, S. Apple Cider Vinegar Attenuates Lipid Profile in Normal and Diabetic Rats. Pak. J. Biol. Sci. 2008, 11, 2634–2638. [Google Scholar] [CrossRef] [PubMed]
- Johnston, C.S.; Steplewska, I.; Long, C.A.; Harris, L.N.; Ryals, R.H. Examination of the Antiglycemic Properties of Vinegar in Healthy Adults. Ann. Nutr. Metab. 2010, 56, 74–79. [Google Scholar] [CrossRef] [PubMed]
- Leeman, M.; Östman, E.; Björck, I. Vinegar dressing and cold storage of potatoes lowers postprandial glycaemic and insulinaemic responses in healthy subjects. Eur. J. Clin. Nutr. 2005, 59, 1266–1271. [Google Scholar] [CrossRef] [PubMed]
- White, A.M.; Johnston, C.S. Vinegar Ingestion at Bedtime Moderates Waking Glucose Concentrations in Adults with Well-Controlled Type 2 Diabetes. Diabetes Care 2007, 30, 2814–2815. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Santos, H.O.; De Moraes, W.M.; Da Silva, G.A.; Prestes, J.; Schoenfeld, B.J. Vinegar (acetic acid) intake on glucose metabolism: A narrative review. Clin. Nutr. ESPEN 2019, 32, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Asejeje, F.; Ighodaro, O.; Asejeje, G.; Adeosun, A. Protective role of apple cider vinegar (APCV) in CCl4-induced renal damage in wistar rats. Metab. Open 2020, 8, 100063. [Google Scholar] [CrossRef]
- Chiu, H.-F.; Chiang, M.; Liao, H.-J.; Shen, Y.-C.; Venkatakrishnan, K.; Cheng, I.-S.; Wang, C.-K. The ergogenic activity of cider vinegar: A randomized cross-over, double-blind, clinical trial. Sports Med. Heal. Sci. 2020, 2, 38–43. [Google Scholar] [CrossRef]
- Wang, L.; Cen, S.; Wang, G.; Lee, Y.-K.; Zhao, J.; Zhang, H.; Chen, W. Acetic acid and butyric acid released in large intestine play different roles in the alleviation of constipation. J. Funct. Foods 2020, 69, 103953. [Google Scholar] [CrossRef]
- Tyakht, A.; Kopeliovich, A.; Klimenko, N.; Efimova, D.; Dovidchenko, N.; Odintsova, V.; Kleimenov, M.; Toshchakov, S.; Popova, A.; Khomyakova, M.; et al. Characteristics of bacterial and yeast microbiomes in spontaneous and mixed-fermentation beer and cider. Food Microbiol. 2021, 94, 103658. [Google Scholar] [CrossRef]
- Jarvis, B. Cider (Cyder; Hard Cider). In Encyclopedia of Food Microbiology; Academic Press: Oxford, UK, 2014; pp. 437–443. [Google Scholar]
- Lee, E.; Lee, J.-E. Impact of drinking alcohol on gut microbiota: Recent perspectives on ethanol and alcoholic beverage. Curr. Opin. Food Sci. 2021, 37, 91–97. [Google Scholar] [CrossRef]
- Salas-Salvadó, J.; Casas-Agustench, P.; Salas-Huetos, A. Cultural and Historical Aspects of Mediterranean Nuts with Emphasis on Their Attributed Healthy and Nutritional Properties. Nutr. Metab. Cardiovasc. Dis. 2011, 21 (Suppl. 1), S1–S6. [Google Scholar] [CrossRef] [PubMed]
- Salaspuro, M. Key role of local acetaldehyde in upper GI tract carcinogenesis. Best Pract. Res. Clin. Gastroenterol. 2017, 31, 491–499. [Google Scholar] [CrossRef] [PubMed]
- Agnoli, L.; Boeri, M.; Scarpa, R.; Capitello, R.; Begalli, D. Behavioural patterns in Mediterranean-style drinking: Generation Y preferences in alcoholic beverage consumption. J. Behav. Exp. Econ. 2018, 75, 117–125. [Google Scholar] [CrossRef]
- He, W.; Liu, S.; Heponiemi, P.; Heinonen, M.; Marsol-Vall, A.; Ma, X.; Yang, B.; Laaksonen, O. Effect of Saccharomyces cerevisiae and Schizosaccharomyces pombe strains on chemical composition and sensory quality of ciders made from Finnish apple cultivars. Food Chem. 2021, 345, 128833. [Google Scholar] [CrossRef] [PubMed]
- Bedriñana, R.P.; Alonso, J.M.; Valles, B.S. Evaluation of autochthonous Saccharomyces bayanus strains under stress conditions for making ice ciders. LWT 2017, 81, 217–225. [Google Scholar] [CrossRef]
- Al Daccache, M.; Koubaa, M.; Salameh, D.; Vorobiev, E.; Maroun, R.G.; Louka, N. Control of the sugar/ethanol conversion rate during moderate pulsed electric field-assisted fermentation of a Hanseniaspora sp. strain to produce low-alcohol cider. Innov. Food Sci. Emerg. Technol. 2020, 59, 102258. [Google Scholar] [CrossRef]
- Coldea, T.E.; Socaciu, C.; Moldovan, Z.; Mudura, E. Minor Volatile Compounds in Traditional Homemade Fruit Brandies from Transylvania-Romania, as Determined by GC-MS Analysis. Not. Bot. Horti Agrobot. Cluj-Napoca 2014, 42, 530–537. [Google Scholar] [CrossRef]
- Ledauphin, J.; Lefrancois, A.; Marquet, N.; Beljean-Leymarie, M.; Barillier, D. Development of an accurate and sensitive gas chromatographic method for the determination of acrolein content in Calvados and cider. LWT 2006, 39, 1045–1052. [Google Scholar] [CrossRef]
- EC. Regulation (EEC) No 1014/90 of 24 April 1990 Laying down Detailed Implementing Rules on the Definition, Description and Presentation of Spirit Drinks; European Commission: Brussels, Belgium, 1990. [Google Scholar]
- Cederbaum, A.I. Alcohol Metabolism. Clin. Liver Dis. 2012, 16, 667–685. [Google Scholar] [CrossRef]
- Tulpule, K.; Dringen, R. Formate generated by cellular oxidation of formaldehyde accelerates the glycolytic flux in cultured astrocytes. Glia 2012, 60, 582–593. [Google Scholar] [CrossRef]
- Choi, J.-H.; Lee, S.K.; Gil, Y.-E.; Ryu, J.; Jung-Choi, K.; Kim, H.; Choi, J.Y.; Park, S.A.; Lee, H.W.; Yun, J.Y. Neurological Complications Resulting from Non-Oral Occupational Methanol Poisoning. J. Korean Med Sci. 2017, 32, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Elkhamary, S.M.; Fahmy, D.M.; Galvez-Ruiz, A.; Asghar, N.; Bosley, T.M. Spectrum of MRI findings in 58 patients with methanol intoxication: Long-term visual and neurological correlation. Egypt. J. Radiol. Nucl. Med. 2016, 47, 1049–1055. [Google Scholar] [CrossRef][Green Version]
- Yoon, Y.-H.; Chen, C.M.; Slater, M.E.; Jung, M.K.; White, A.M. Trends in Premature Deaths From Alcoholic Liver Disease in the U.S., 1999–2018. Am. J. Prev. Med. 2020, 59, 469–480. [Google Scholar] [CrossRef] [PubMed]
- Askgaard, G.; Gronbaek, M.; Kjær, M.S.; Tjønneland, A.; Tolstrup, J.S. Alcohol drinking pattern and risk of alcoholic liver cirrhosis: A prospective cohort study. J. Hepatol. 2015, 62, 1061–1067. [Google Scholar] [CrossRef]
- Schutte, R.; Papageorgiou, M.; Najlah, M.; Huisman, H.W.; Ricci, C.; Zhang, J.; Milner, N.; Schutte, A.E. Drink types unmask the health risks associated with alcohol intake–Prospective evidence from the general population. Clin. Nutr. 2020, 39, 3168–3174. [Google Scholar] [CrossRef] [PubMed]
- Sauvageot, N.; Gouffi, K.; LaPlace, J.-M.; Auffray, Y. Glycerol metabolism in Lactobacillus collinoides: Production of 3-hydroxypropionaldehyde, a precursor of acrolein. Int. J. Food Microbiol. 2000, 55, 167–170. [Google Scholar] [CrossRef]
- Muguruma, K.; Pradipta, A.R.; Ode, Y.; Terashima, K.; Michiba, H.; Fujii, M.; Tanaka, K. Disease-associated acrolein: A possible diagnostic and therapeutic substrate for in vivo synthetic chemistry. Bioorganic Med. Chem. 2020, 28, 115831. [Google Scholar] [CrossRef]
- Keshava, C.; Davis, J.A.; Stanek, J.; Thayer, K.A.; Galizia, A.; Keshava, N.; Gift, J.S.; Vulimiri, S.V.; Woodall, G.M.; Gigot, C.; et al. Application of systematic evidence mapping to assess the impact of new research when updating health reference values: A case example using acrolein. Environ. Int. 2020, 143, 105956. [Google Scholar] [CrossRef]
- Woodruff, T.J.; Axelrad, D.A. Ambient Concentrations of Acrolein: Health Risks. In Encyclopedia of Environmental Health (Second Edition); Nriagu, J., Ed.; Elsevier: Oxford, UK, 2019; pp. 82–86. [Google Scholar]
- Hong, J.-H.; Lee, P.A.H.; Lu, Y.-C.; Huang, C.-Y.; Chen, C.-H.; Chiang, C.-H.; Chow, P.-M.; Jaw, F.-S.; Wang, C.-C.; Huang, C.-Y.; et al. Acrolein contributes to urothelial carcinomas in patients with chronic kidney disease. Urol. Oncol. Semin. Orig. Investig. 2020, 38, 465–475. [Google Scholar] [CrossRef]
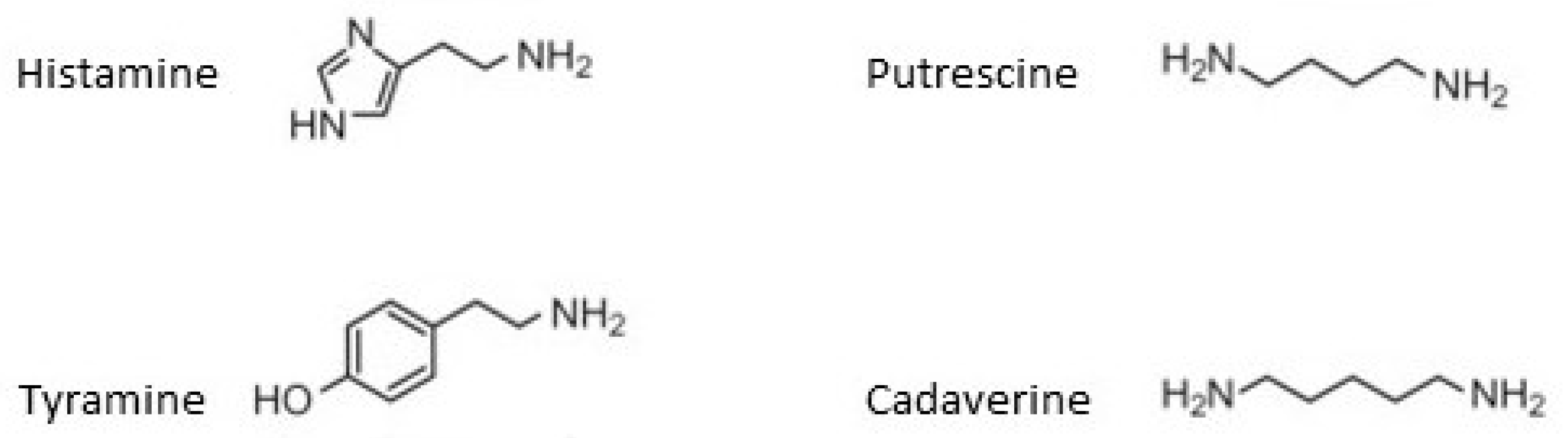
- Costantini, A.; Pietroniro, R.; Doria, F.; Pessione, E.; García-Moruno, E. Putrescine production from different amino acid precursors by lactic acid bacteria from wine and cider. Int. J. Food Microbiol. 2013, 165, 11–17. [Google Scholar] [CrossRef]
- Ladero, V.; Coton, M.; Fernández, M.; Buron, N.; Martín, M.C.; Guichard, H.; Coton, E.; Alvarez, M.A. Biogenic amines content in Spanish and French natural ciders: Application of qPCR for quantitative detection of biogenic amine-producers. Food Microbiol. 2011, 28, 554–561. [Google Scholar] [CrossRef] [PubMed]
- Ladero, V.; Calles-Enriquez, M.; Fernandez, M.; Alvarez, M.A. Toxicological Effects of Dietary Biogenic Amines. Curr. Nutr. Food Sci. 2010, 6, 145–156. [Google Scholar] [CrossRef]
- Coton, M.; Romano, A.; Spano, G.; Ziegler, K.; Vetrana, C.; Desmarais, C.; Lonvaud-Funel, A.; Lucas, P.; Coton, E. Occurrence of biogenic amine-forming lactic acid bacteria in wine and cider. Food Microbiol. 2010, 27, 1078–1085. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, M.A.; Moreno-Arribas, M.V. The problem of biogenic amines in fermented foods and the use of potential biogenic amine-degrading microorganisms as a solution. Trends Food Sci. Technol. 2014, 39, 146–155. [Google Scholar] [CrossRef]
- Garai, G.; Dueñas, M.; Irastorza, A.; Moreno-Arribas, M.V. Biogenic amine production by lactic acid bacteria isolated from cider. Lett. Appl. Microbiol. 2007, 45, 473–478. [Google Scholar] [CrossRef]
- Garai-Ibabe, G.; Irastorza, A.; Dueñas, M.T.; Martín-Álvarez, P.J.; Moreno-Arribas, V.M. Evolution of amino acids and biogenic amines in natural ciders as a function of the year and the manufacture steps. Int. J. Food Sci. Technol. 2012, 48, 375–381. [Google Scholar] [CrossRef]
- Bullerman, L.B. MYCOTOXINS | Classifications. In Encyclopedia of Food Sciences and Nutrition (Second Edition); Caballero, B., Ed.; Academic Press: Oxford, UK, 2003; pp. 4080–4089. [Google Scholar]
- Kharayat, B.S.; Singh, Y. Chapter 13-Mycotoxins in Foods: Mycotoxicoses, Detection, and Management. In Microbial Contamination and Food Degradation; Holban, A.M., Grumezescu, A.M., Eds.; Handbook of Food Bioengineering; Academic Press: London, UK, 2018; pp. 395–421. [Google Scholar]
- Saleh, I.; Goktepe, I. Health risk assessment of Patulin intake through apples and apple-based foods sold in Qatar. Heliyon 2019, 5, e02754. [Google Scholar] [CrossRef]
- Wei, C.; Yu, L.; Qiao, N.; Zhao, J.; Zhang, H.; Zhai, Q.; Tian, F.; Chen, W. Progress in the distribution, toxicity, control, and detoxification of patulin: A review. Toxicon 2020, 184, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Yang, X.; Nisar, T.; Tian, Y.; Sun, L.; Zhang, X.; Guo, Y. Patulin biodegradation and quality improvement of apple puree fermented with Byssochlamys nivea FF1-2. Food Biosci. 2018, 21, 45–52. [Google Scholar] [CrossRef]
- Zhang, X.; Guo, Y.; Ma, Y.; Chai, Y.; Li, Y. Biodegradation of patulin by a Byssochlamys nivea strain. Food Control. 2016, 64, 142–150. [Google Scholar] [CrossRef]
- Gulsunoglu, Z.; Purves, R.W.; Karbancioglu-Guler, F.; Kilic-Akyilmaz, M. Enhancement of phenolic antioxidants in industrial apple waste by fermentation with Aspergillus spp. Biocatal. Agric. Biotechnol. 2020, 25, 101562. [Google Scholar] [CrossRef]
- Marks, S.C.; Mullen, W.; Crozier, A. Flavonoid and chlorogenic acid profiles of English cider apples. J. Sci. Food Agric. 2007, 87, 719–728. [Google Scholar] [CrossRef]
- Raudone, L.; Raudonis, R.; Liaudanskas, M.; Janulis, V.; Viskelis, P. Phenolic antioxidant profiles in the whole fruit, flesh and peel of apple cultivars grown in Lithuania. Sci. Hortic. 2017, 216, 186–192. [Google Scholar] [CrossRef]
- Mihailović, N.R.; Mihailović, V.B.; Kreft, S.; Ćirić, A.R.; Joksović, L.G.; Đurđević, P.T. Analysis of phenolics in the peel and pulp of wild apples (Malus sylvestris (L.) Mill.). J. Food Compos. Anal. 2018, 67, 1–9. [Google Scholar] [CrossRef]
- Lee, J.; Chan, B.L.S.; Mitchell, A.E. Identification/quantification of free and bound phenolic acids in peel and pulp of apples (Malus domestica) using high resolution mass spectrometry (HRMS). Food Chem. 2017, 215, 301–310. [Google Scholar] [CrossRef] [PubMed]
- Bi, X.; Zhang, J.; Chen, C.; Zhang, D.; Li, P.; Ma, F. Anthocyanin contributes more to hydrogen peroxide scavenging than other phenolics in apple peel. Food Chem. 2014, 152, 205–209. [Google Scholar] [CrossRef]
- Zhang, M.; Zhang, G.; You, Y.; Yang, C.; Li, P.; Ma, F. Effects of relative air humidity on the phenolic compounds contents and coloration in the ‘Fuji’ apple (Malus domestica Borkh.) peel. Sci. Hortic. 2016, 201, 18–23. [Google Scholar] [CrossRef]
- Khlifi, R.; Dhaouefi, Z.; Maatouk, M.; Sassi, A.; Boudhiba, N.; Ioannou, I.; Ghedira, K.; Ghedira, L.C.; Kilani-Jaziri, S. Heat treatment improves the immunomodulatory and cellular antioxidant behavior of a natural flavanone: Eriodictyol. Int. Immunopharmacol. 2018, 61, 317–324. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, Y.; Chen, Y.; Zhou, B.; Shan, X.; Yang, G. Eriodictyol inhibits IL-1β-induced inflammatory response in human osteoarthritis chondrocytes. Biomed. Pharmacother. 2018, 107, 1128–1134. [Google Scholar] [CrossRef]
- Khan, M.K.; Huma, Z.E.; Dangles, O. A comprehensive review on flavanones, the major citrus polyphenols. J. Food Compos. Anal. 2014, 33, 85–104. [Google Scholar] [CrossRef]
- Lee, J.K. Anti-inflammatory effects of eriodictyol in lipopolysaccharidestimulated raw 264.7 murine macrophages. Arch. Pharmacal Res. 2011, 34, 671–679. [Google Scholar] [CrossRef] [PubMed]
- Yoo, J.-M.; Kim, J.-H.; Park, S.-J.; Kang, Y.-J.; Kim, T.-J. Inhibitory Effect of Eriodictyol on IgE/Ag-Induced Type I Hypersensitivity. Biosci. Biotechnol. Biochem. 2012, 76, 1285–1290. [Google Scholar] [CrossRef] [PubMed]
- Rogovskiĭ, V.S.; Matiushin, A.I.; Shimanovskiĭ, N.L.; Semeĭkin, A.V.; Kukhareva, T.S.; Koroteev, A.M.; Koroteev, M.P.; Nifant’ev, E.E. Antiproliferative and antioxidant activity of new dihydroquercetin derivatives. Eksp Klin Farm. 2010, 73, 39–42. [Google Scholar]
- Salama, S.A.; Kabel, A.M. Taxifolin ameliorates iron overload-induced hepatocellular injury: Modulating PI3K/AKT and p38 MAPK signaling, inflammatory response, and hepatocellular regeneration. Chem. Interact. 2020, 330, 109230. [Google Scholar] [CrossRef] [PubMed]
- Galochkina, A.V.; Zarubaev, V.V.; Kiselev, I.O.; Babkin, B.A.; Ostroukhova, L.A. Antiviral Activity of The Dihydroquercetin During The Coxsackievirus B4 Replication In Vitro. Vopr. Virusol. 2016, 61, 27–31. [Google Scholar] [PubMed]
- Gao, L.; Yuan, P.; Zhang, Q.; Fu, Y.; Hou, Y.; Wei, Y.; Zheng, X.; Feng, W. Taxifolin improves disorders of glucose metabolism and water-salt metabolism in kidney via PI3K/AKT signaling pathway in metabolic syndrome rats. Life Sci. 2020, 263, 118713. [Google Scholar] [CrossRef] [PubMed]
- Plotnikov, M.B.; Aliev, O.I.; Sidekhmenova, A.V.; Shamanaev, A.Y.; Anishchenko, A.M.; Nosarev, A.V.; Pushkina, E.A. Modes of Hypotensive Action of Dihydroquercetin in Arterial Hypertension. Bull. Exp. Biol. Med. 2017, 162, 353–356. [Google Scholar] [CrossRef]
- Zanwar, A.A.; Badole, S.L.; Shende, P.S.; Hegde, M.V.; Bodhankar, S.L. Chapter 21-Antioxidant Role of Catechin in Health and Disease. In Polyphenols in Human Health and Disease; Watson, R.R., Preedy, V.R., Zibadi, S., Eds.; Academic Press: San Diego, CA, USA, 2014; pp. 267–271. [Google Scholar]
- Chen, W.; Zhu, X.; Lu, Q.; Zhang, L.; Wang, X.; Liu, R. C-ring cleavage metabolites of catechin and epicatechin enhanced antioxidant activities through intestinal microbiota. Food Res. Int. 2020, 135, 109271. [Google Scholar] [CrossRef]
- Vechi, G.; de Cássia Melo Vilhena de Andrade Fonsecada Silva, R.; De Souza, P.; Da Silva, L.M.; De Andrade, S.F.; Filho, V.C. Cryptostrobin and catechin isolated from Eugenia mattosii D. Legrand leaves induce endothelium-dependent and independent relaxation in spontaneously hypertensive rat aorta. Pharmacol. Rep. 2019, 71, 950–957. [Google Scholar] [CrossRef]
- Hodges, J.K.; Sasaki, G.Y.; Bruno, R.S. Anti-inflammatory Activities of Green Tea Catechins along the Gut-liver Axis in Nonalcoholic Fatty Liver Disease: Lessons Learned from Preclinical and Human Studies. J. Nutr. Biochem. 2020, 85, 108478. [Google Scholar] [CrossRef]
- Cheng, Z.; Zhang, Z.; Han, Y.; Liu, J.; Wang, Y.; Chen, X.; Shao, Y.; Cheng, Y.; Zhou, W.; Lu, X.; et al. A review on anti-cancer effect of green tea catechins. J. Funct. Foods 2020, 74, 104172. [Google Scholar] [CrossRef]
- Hui, C.K.; Majid, N.I.; Yusof, H.M.; Zainol, K.M.; Mohamad, H.; Zin, Z.M. Catechin profile and hypolipidemic activity of Morinda citrifolia leaf water extract. Heliyon 2020, 6, e04337. [Google Scholar] [CrossRef]
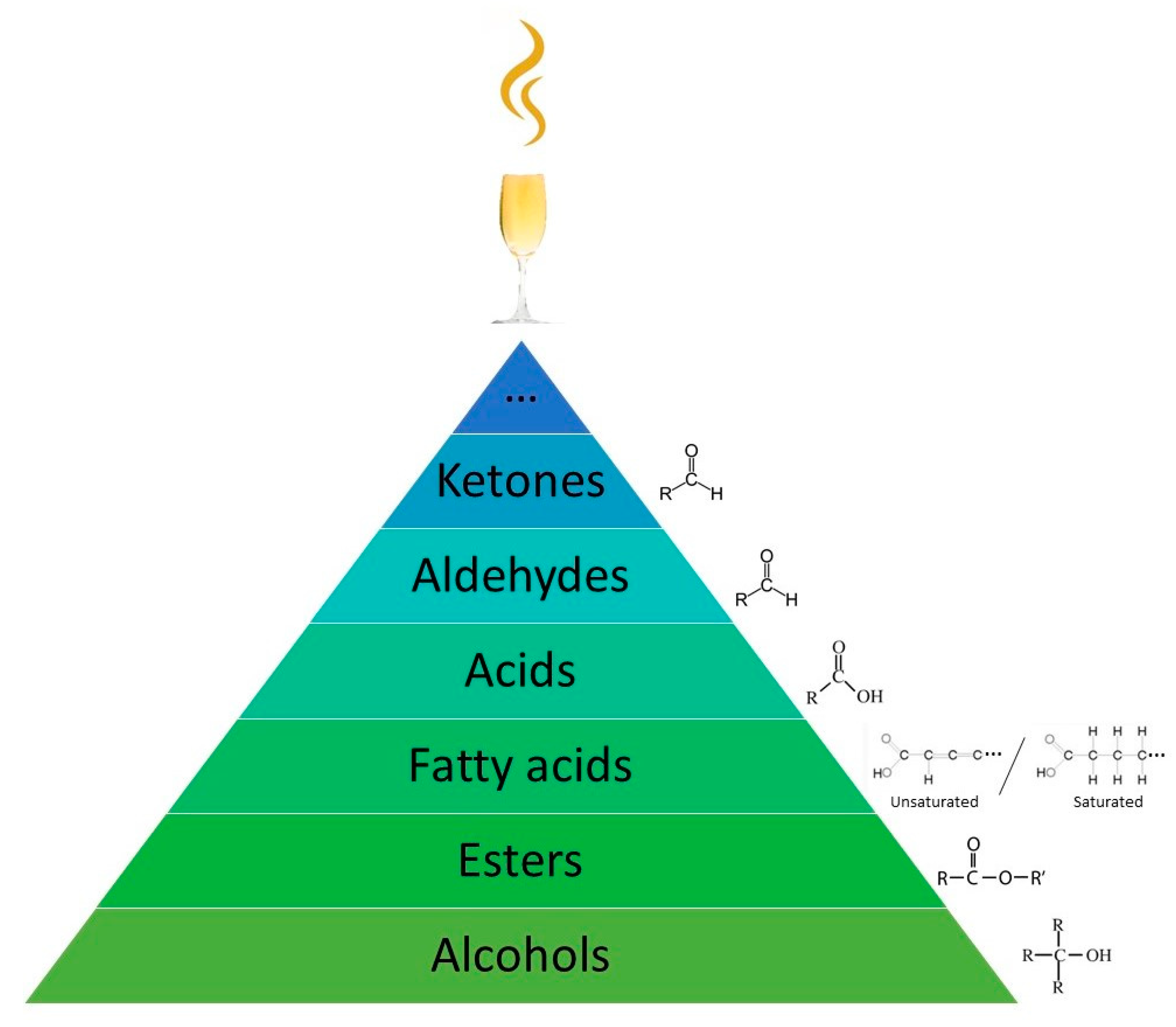
| Classes | Compounds |
|---|---|
| Esters | hexyl acetate, ethyl butyrate, ethyl hexanoate, butyl acetate, ethyl 2-methylbutyrate, hexyl butyrate, ethyl acetate, methyl isovalerate, isoamyl isovalerate, methyl salicylate |
| Alcohols | 1-butanol, 1-hexanol, 2-methyl-1-butanol, 2-ethylhexanol, (E)-2-hexen-1-ol, 2-methyl-1-propanol, isobutenylcarbinol, trans-2-hexen-1-ol, benzyl alcohol, 1-octanol, β-citronellol, geraniol, 2-methyl-1-pentanol, 3-hexen-1-ol (Z) and 2-hexen-1-ol (E) |
| Aldehydes | hexanal, (E)-2-hexenal, octanal, decanal, 2-nonanal, (Z)-2-hexenal, acetaldehyde |
| Acids | acetic acid, butanoic acid, lactic acid, isovaleric acid, tartaric acid |
| Ketones | 6-methyl-5-hepten-2-one, β-Damascenone, 2-undecanone, 2-heptanone, 2-nonanone, 4-heptanone, 4-cyclopentene-1,3-dione |
| Others | D-limonene, eugenol, linalool |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guiné, R.P.F.; Barroca, M.J.; Coldea, T.E.; Bartkiene, E.; Anjos, O. Apple Fermented Products: An Overview of Technology, Properties and Health Effects. Processes 2021, 9, 223. https://doi.org/10.3390/pr9020223
Guiné RPF, Barroca MJ, Coldea TE, Bartkiene E, Anjos O. Apple Fermented Products: An Overview of Technology, Properties and Health Effects. Processes. 2021; 9(2):223. https://doi.org/10.3390/pr9020223
Chicago/Turabian StyleGuiné, Raquel P. F., Maria João Barroca, Teodora Emilia Coldea, Elena Bartkiene, and Ofélia Anjos. 2021. "Apple Fermented Products: An Overview of Technology, Properties and Health Effects" Processes 9, no. 2: 223. https://doi.org/10.3390/pr9020223
APA StyleGuiné, R. P. F., Barroca, M. J., Coldea, T. E., Bartkiene, E., & Anjos, O. (2021). Apple Fermented Products: An Overview of Technology, Properties and Health Effects. Processes, 9(2), 223. https://doi.org/10.3390/pr9020223











