Time of Flight Size Control of Carbon Nanoparticles Using Ar+CH4 Multi-Hollow Discharge Plasma Chemical Vapor Deposition Method
Abstract
1. Introduction
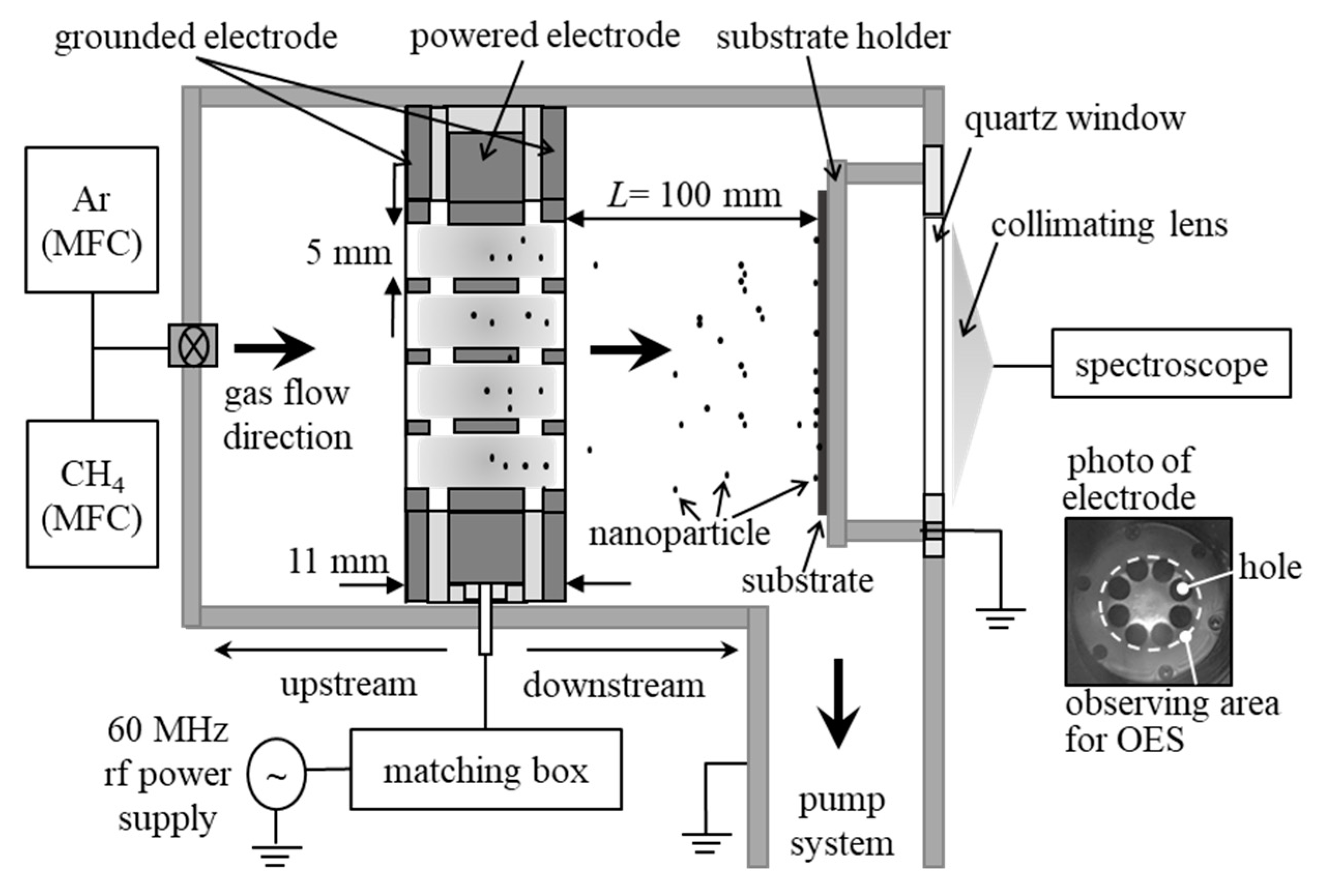
2. Materials and Methods
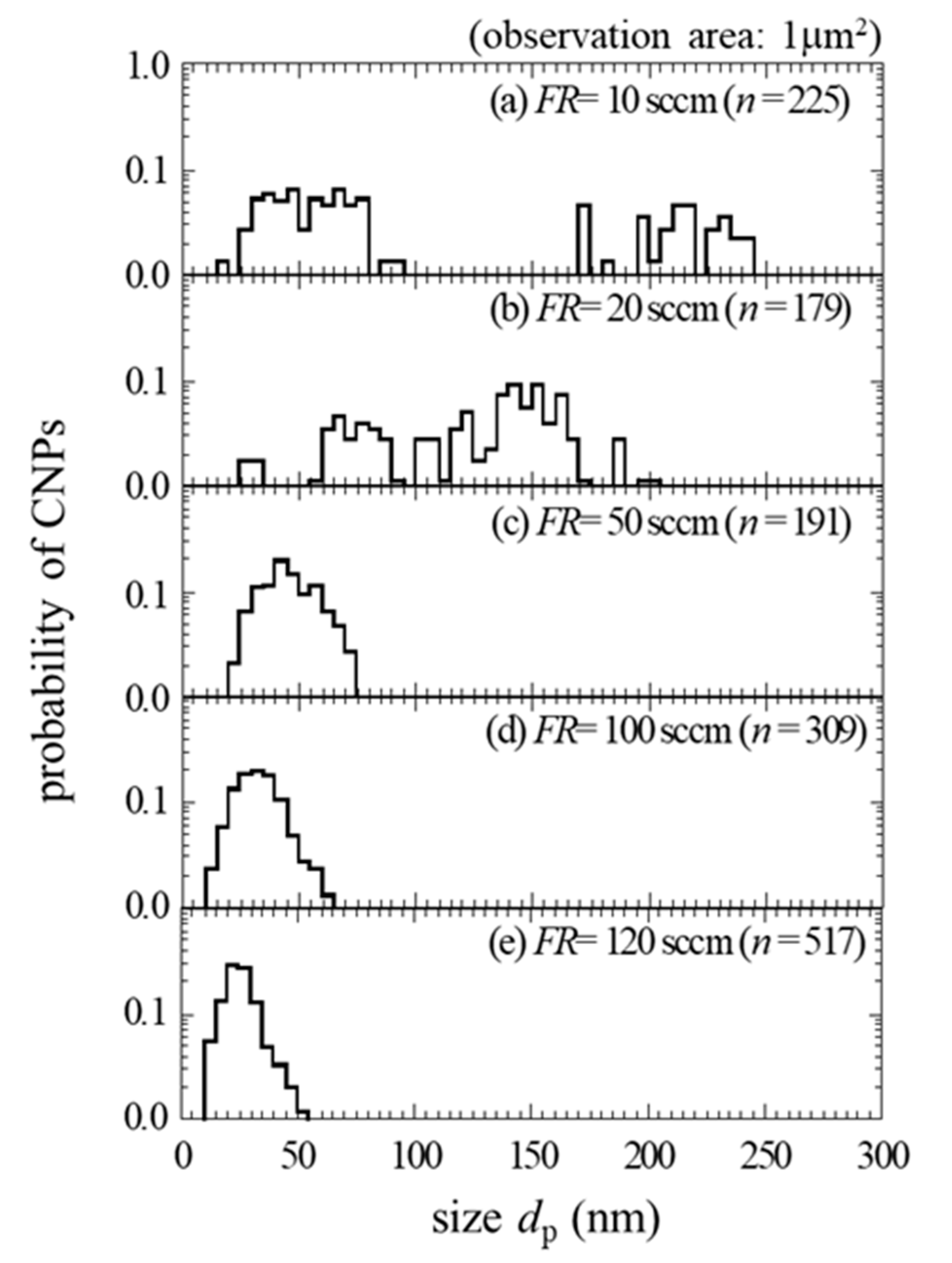
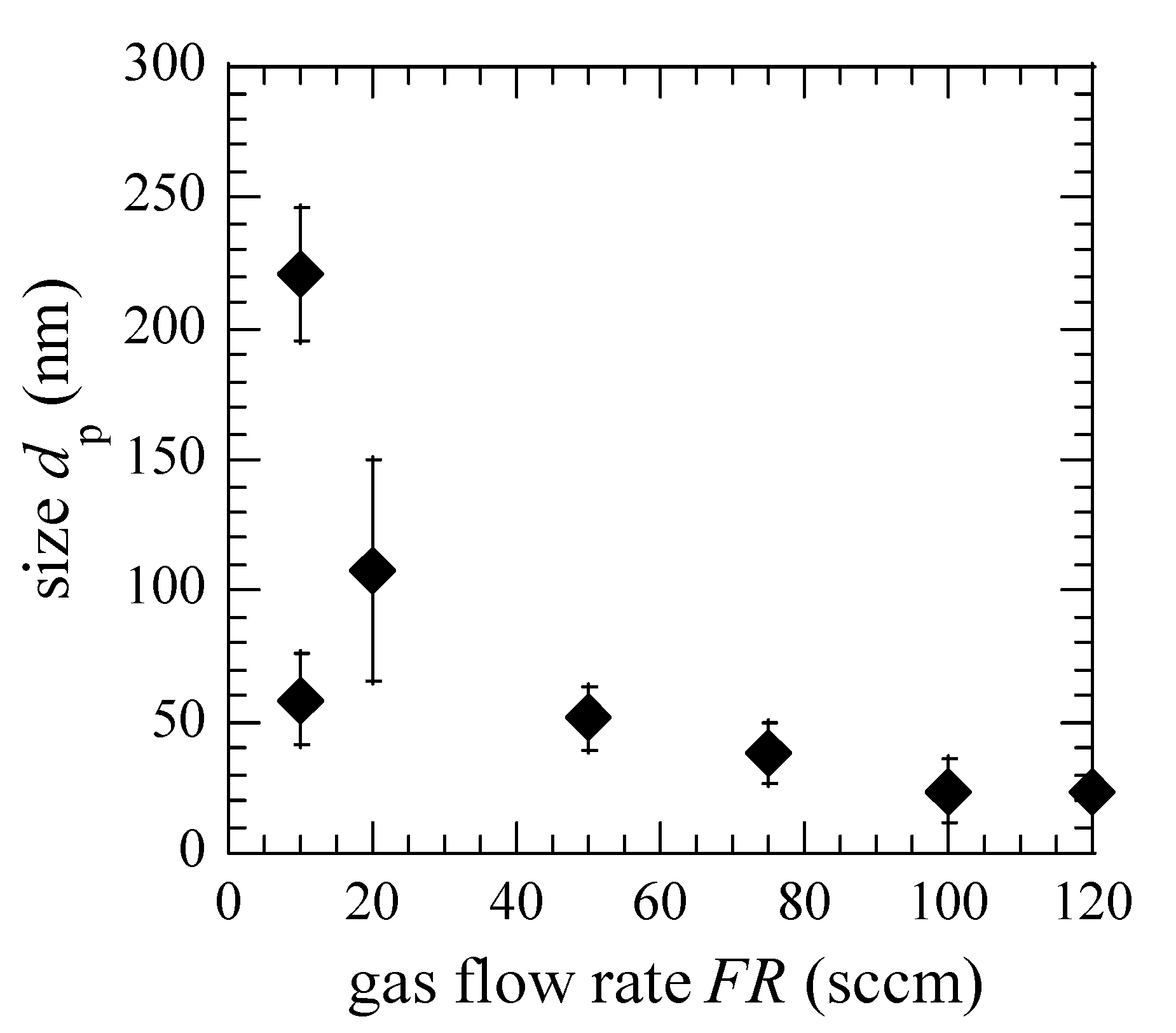
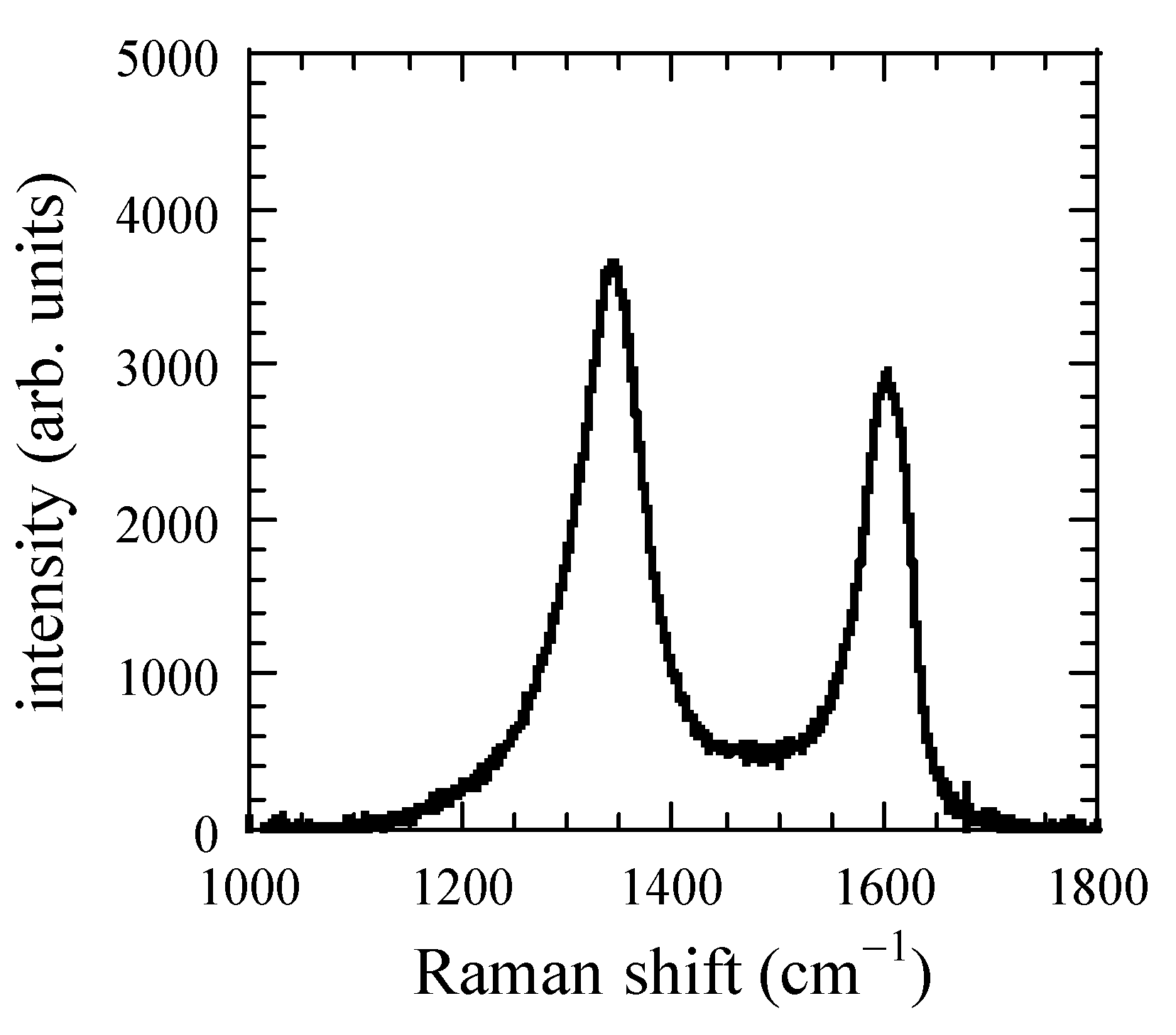
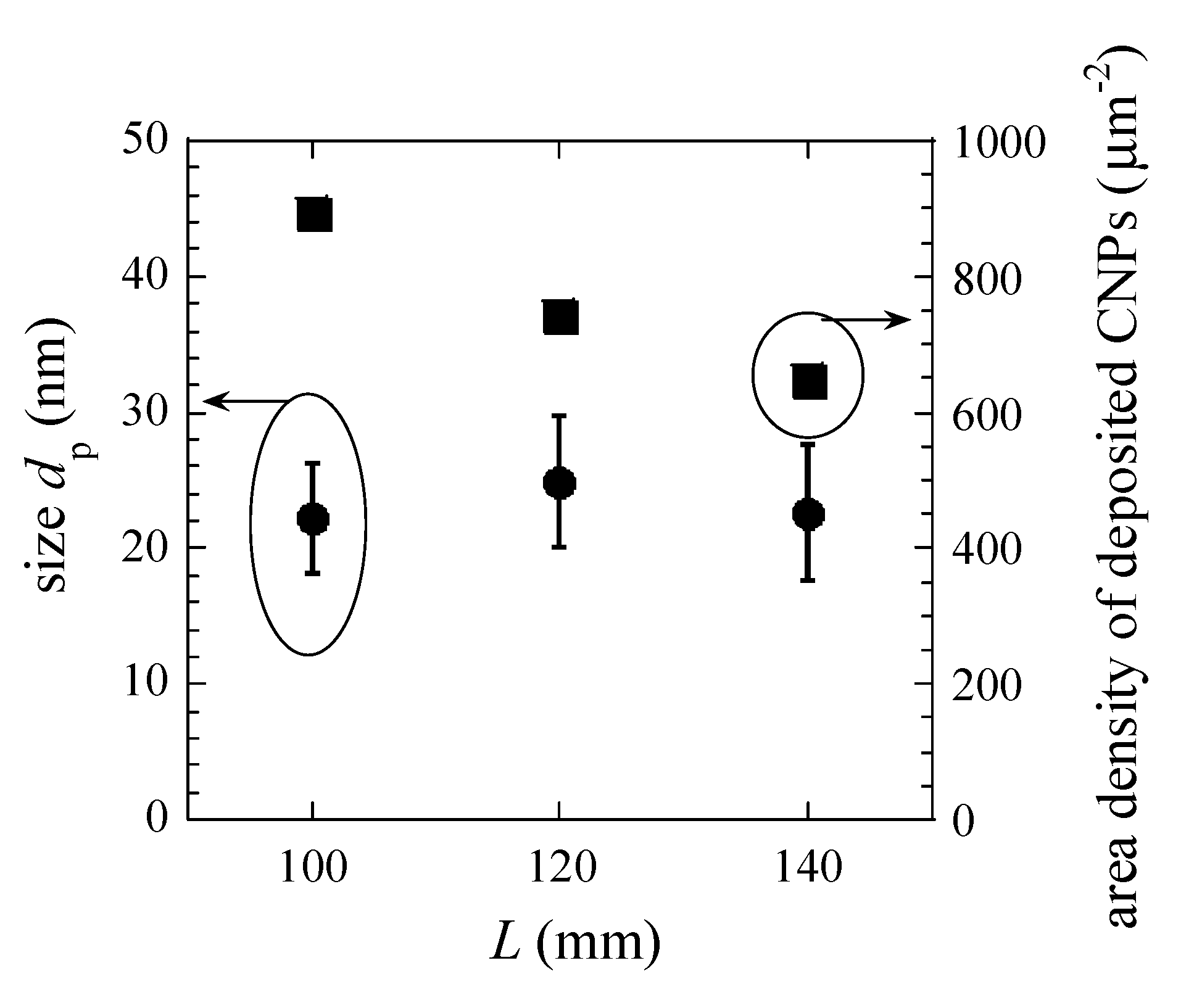
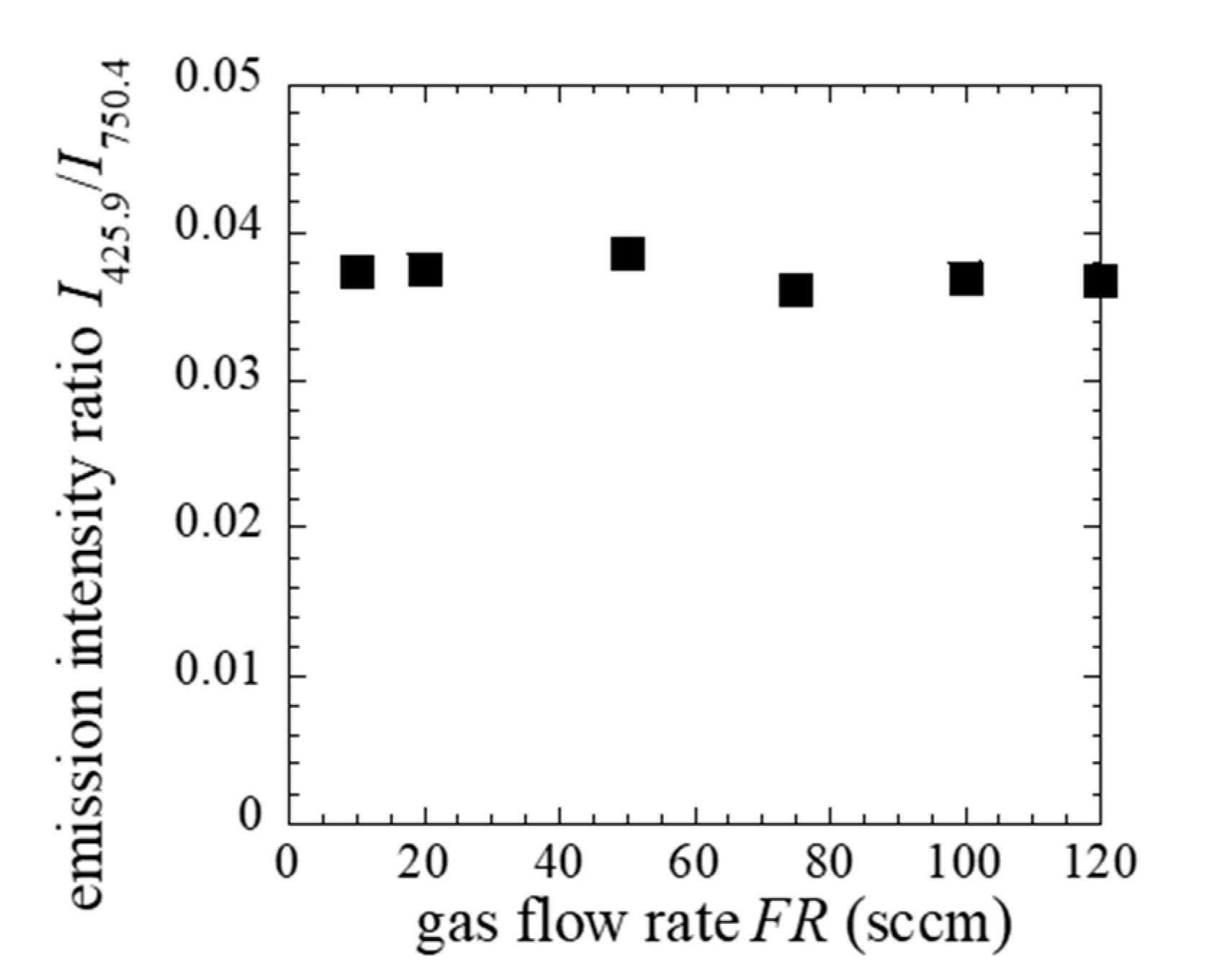
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statements
Acknowledgments
Conflicts of Interest
References
- Kumar, S.; Nehra, M.; Kedia, D.; Dilbaghi, N.; Tankeshwar, K.; Kim, K.H. Nanodiamonds: Emerging face of future nanotechnology. Carbon N. Y. 2019, 143, 678–699. [Google Scholar] [CrossRef]
- Li, S.; Pasc, A.; Fierro, V.; Celzard, A. Hollow carbon spheres, synthesis and applications-a review. J. Mater. Chem. A 2016, 4, 12686–12713. [Google Scholar] [CrossRef]
- Bian, X.; Chen, Q.; Zhang, Y.; Sang, L.; Tang, W. Deposition of nano-diamond-like carbon films by an atmospheric pressure plasma gun and diagnostic by optical emission spectrum on the process. Surf. Coat. Technol. 2008, 202, 5383–5385. [Google Scholar] [CrossRef]
- Tian, F.; He, C.N. Fabrication and growth mechanism of carbon nanospheres by chemical vapor deposition. Mater. Chem. Phys. 2010, 123, 351–355. [Google Scholar] [CrossRef]
- Wang, F.; Hong, R. Continuous preparation of structure-controlled carbon nanoparticle via arc plasma and the reinforcement of polymeric composites. Chem. Eng. J. 2017, 328, 1098–1111. [Google Scholar] [CrossRef]
- Estes, C.S.; Gerard, A.Y.; Godward, J.D.; Hayes, S.B.; Liles, S.H.; Shelton, J.L.; Stewart, T.S.; Webster, R.I.; Webster, H.F. Preparation of highly functionalized carbon nanoparticles using a one-step acid dehydration of glycerol. Carbon N. Y. 2019, 142, 547–557. [Google Scholar] [CrossRef]
- Li, Z.; Wang, L.; Li, Y.; Feng, Y.; Feng, W. Carbon-based functional nanomaterials: Preparation, properties and applications. Compos. Sci. Technol. 2019, 179, 10–40. [Google Scholar] [CrossRef]
- Kang, J.; Li, O.L.; Saito, N. Synthesis of structure-controlled carbon nano spheres by solution plasma process. Carbon N. Y. 2013, 60, 292–298. [Google Scholar] [CrossRef]
- Miao, J.Y.; Hwang, D.W.; Narasimhulu, K.V.; Lin, P.I.; Chen, Y.T.; Lin, S.H.; Hwang, L.P. Synthesis and properties of carbon nanospheres grown by CVD using Kaolin supported transition metal catalysts. Carbon N. Y. 2004, 42, 813–822. [Google Scholar] [CrossRef]
- Pottathara, Y.B.; Grohens, Y.; Kokol, V.; Kalarikkal, N.; Thomas, S. Synthesis and Processing of Emerging Two-Dimensional Nanomaterials; Elsevier Inc.: Amsterdam, The Netherlands, 2019; ISBN 9780128157510. [Google Scholar]
- Kumar, M.; Ando, Y. Chemical Vapor Deposition of Carbon Nanotubes: A Review on Growth Mechanism and Mass Production. J. Nanosci. Nanotechnol. 2010, 10, 3739–3758. [Google Scholar] [CrossRef]
- Koga, K.; Matsuoka, Y.; Tanaka, K.; Shiratani, M.; Watanabe, Y. In situ observation of nucleation and subsequent growth of clusters in silane radio frequency discharges. Appl. Phys. Lett. 2000, 77, 196–198. [Google Scholar] [CrossRef]
- Nunomura, S.; Shiratani, M.; Koga, K.; Kondo, M.; Watanabe, Y. Nanoparticle coagulation in fractionally charged and charge fluctuating dusty plasmas. Phys. Plasmas 2008, 15, 080703. [Google Scholar] [CrossRef]
- Kim, K.; Park, J.H.; Doo, S.G.; Nam, J.D.; Kim, T. Generation of size and structure controlled Si nanoparticles using pulse plasma for energy devices. Thin Solid Films 2009, 517, 4184–4187. [Google Scholar] [CrossRef]
- Nakamura, W.M.; Shimokawa, D.; Miyahara, H.; Koga, K.; Shiratani, M. Spatial Profile of Deposition Rate of a-Si:H Films in Multi-Hollow Discharge Plasma Chemical Vapor Deposition. Trans. Mater. Res. Soc. Jpn. 2007, 32, 469–472. [Google Scholar] [CrossRef]
- Shiratani, M.; Koga, K.; Iwashita, S.; Uchida, G.; Itagaki, N.; Kamataki, K. Nano-factories in plasma: Present status and outlook. J. Phys. D Appl. Phys. 2011, 44, 1–8. [Google Scholar] [CrossRef]
- Koga, K.; Nakahara, K.; Kim, Y.-W.; Kawashima, Y.; Matsunaga, T.; Sato, M.; Yamashita, D.; Matsuzaki, H.; Uchida, G.; Kamataki, K.; et al. Deposition of cluster-free P-doped a-Si:H films using SiH4+PH3 multi-hollow discharge plasma CVD. Phys. Status Solidi 2011, 8, 3013–3016. [Google Scholar] [CrossRef]
- Uchida, G.; Yamamoto, K.; Kawashima, Y.; Sato, M.; Nakahara, K.; Kamataki, K.; Itagaki, N.; Koga, K.; Kondo, M.; Shiratani, M. Surface nitridation of silicon nano-particles using double multi-hollow discharge plasma CVD. Phys. Status Solidi Curr. Top. Solid State Phys. 2011, 8, 3017–3020. [Google Scholar] [CrossRef]
- Koga, K.; Matsunaga, T.; Kim, Y.; Nakahara, K.; Yamashita, D.; Matsuzaki, H.; Kamataki, K.; Uchida, G.; Itagaki, N.; Shiratani, M. Combinatorial deposition of microcrystalline silicon films using multihollow discharge plasma chemical vapor deposition. Jpn. J. Appl. Phys. 2012, 51, 1–4. [Google Scholar] [CrossRef]
- Kim, Y.; Matsunaga, T.; Nakahara, K.; Seo, H.; Kamataki, K.; Uchida, G.; Itagaki, N.; Koga, K.; Shiratani, M. Effects of nanoparticle incorporation on properties of microcrystalline films deposited using multi-hollow discharge plasma CVD. Surf. Coat. Technol. 2013, 228, S550–S553. [Google Scholar] [CrossRef]
- Seo, H.; Ichida, D.; Hashimoto, S.; Itagaki, N.; Koga, K.; Shiratani, M.; Nam, S.H.; Boo, J.H. Improvement of charge transportation in Si quantum dot-sensitized solar cells using vanadium doped TiO2. J. Nanosci. Nanotechnol. 2016, 16, 4875–4879. [Google Scholar] [CrossRef]
- Toko, S.; Kanemitsu, Y.; Seo, H.; Itagaki, N.; Koga, K.; Shiratani, M. Optical bandgap energy of Si nanoparticle composite films deposited by a multi-hollow discharge plasma chemical vapor deposition method. J. Nanosci. Nanotechnol. 2016, 16, 10753–10757. [Google Scholar] [CrossRef]
- Hwang, S.H.; Kamataki, K.; Itagaki, N.; Koga, K.; Shiratani, M. Effects of gas pressure on the size distribution and structure of carbon nanoparticles using Ar + CH4 multi-hollow discharged plasma chemical vapor deposition. Plasma Fusion Res. 2019, 14, 1–5. [Google Scholar] [CrossRef]
- Hwang, S.H.; Okumura, T.; Kamataki, K.; Itagaki, N.; Koga, K.; Shiratani, M. Size and Flux of Carbon Nanoparticles Synthesized by Ar+CH4 Multi-hollow Plasma Chemical Vapor Deposition. Diam. Relat. Mater. 2020, 108050. [Google Scholar] [CrossRef]
- Hwang, S.H.; Okumura, T.; Kamataki, K.; Itagaki, N.; Koga, K.; Nakatani, T.; Shiratani, M. Low stress diamond-like carbon films containing carbon nanoparticles fabricated by combining rf sputtering and plasma chemical vapor deposition. Jpn. J. Appl. Phys. 2020, 59, 100906. [Google Scholar] [CrossRef]
- Watanabe, Y. Formation and behaviour of nano/micro-particles in low pressure plasmas. J. Phys. D Appl. Phys. 2006, 39, R329. [Google Scholar] [CrossRef]
- Siddique, A.B.; Pramanick, A.K.; Chatterjee, S.; Ray, M. Amorphous Carbon Dots and their Remarkable Ability to Detect 2,4,6-Trinitrophenol. Sci. Rep. 2018, 8, 9700. [Google Scholar] [CrossRef]
- Ferrari, A.C.; Robertson, J. Raman spectroscopy of amorphous, nanostructured, diamond like carbon, and nanodiamond. Philos. Trans. R. Soc. Lond. A 2004, 362, 2477–2512. [Google Scholar] [CrossRef]
- Merlen, A.; Buijnsters, J.G.; Pardanaud, C. A guide to and review of the use of multiwavelength Raman spectroscopy for characterizing defective aromatic carbon solids: From graphene to amorphous carbons. Coatings 2017, 7, 153. [Google Scholar] [CrossRef]
- Dong, X.; Koga, K.; Yamashita, D.; Seo, H.; Itagaki, N.; Shiratani, M.; Setsuhara, Y.; Sekine, M.; Hori, M. Effects of deposition rate and ion bombardment on properties of a-C:H films deposited by H-assisted plasma CVD method. Jpn. J. Appl. Phys. 2016, 55. [Google Scholar] [CrossRef]
- Nunomura, S.; Kita, M.; Koga, K.; Shiratani, M.; Watanabe, Y. In situ simple method for measuring size and density of nanoparticles in reactive plasmas. J. Appl. Phys. 2006, 99, 083302. [Google Scholar] [CrossRef]
- Shiratani, M.; Koga, K.; Kamataki, K.; Iwashita, S. Theory for correlation between plasma fluctuation and fluctuation of nanoparticle growth in reactive plasmas Theory for correlation between plasma fl uctuation and fl uctuation of nanoparticle growth in reactive plasmas. Jpn. J. Appl. Phys. 2014, 53, 010201. [Google Scholar] [CrossRef]
- Moshkalyov, S.A. Deposition of silicon nitride by low-pressure electron cyclotron resonance plasma enhanced chemical vapor deposition in N2/Ar/SiH4. J. Vac. Sci. Technol. B Microelectron. Nanom. Struct. 1997, 15, 2682. [Google Scholar] [CrossRef]
- Daltrini, A.M.; Moshkalev, S.A.; Monteiro, M.J.R.; Besseler, E.; Kostryukov, A.; Machida, M. Mode transitions and hysteresis in inductively coupled plasmas. J. Appl. Phys. 2007, 101, 073309. [Google Scholar] [CrossRef]
- Toyoda, H.; Kojima, H.; Sugai, H. Mass spectroscopic investigation of the CH3 radicals in a methane rf discharge. Appl. Phys. Lett. 1989, 54, 1507–1509. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hwang, S.H.; Koga, K.; Hao, Y.; Attri, P.; Okumura, T.; Kamataki, K.; Itagaki, N.; Shiratani, M.; Oh, J.-S.; Takabayashi, S.; et al. Time of Flight Size Control of Carbon Nanoparticles Using Ar+CH4 Multi-Hollow Discharge Plasma Chemical Vapor Deposition Method. Processes 2021, 9, 2. https://doi.org/10.3390/pr9010002
Hwang SH, Koga K, Hao Y, Attri P, Okumura T, Kamataki K, Itagaki N, Shiratani M, Oh J-S, Takabayashi S, et al. Time of Flight Size Control of Carbon Nanoparticles Using Ar+CH4 Multi-Hollow Discharge Plasma Chemical Vapor Deposition Method. Processes. 2021; 9(1):2. https://doi.org/10.3390/pr9010002
Chicago/Turabian StyleHwang, Sung Hwa, Kazunori Koga, Yuan Hao, Pankaj Attri, Takamasa Okumura, Kunihiro Kamataki, Naho Itagaki, Masaharu Shiratani, Jun-Seok Oh, Susumu Takabayashi, and et al. 2021. "Time of Flight Size Control of Carbon Nanoparticles Using Ar+CH4 Multi-Hollow Discharge Plasma Chemical Vapor Deposition Method" Processes 9, no. 1: 2. https://doi.org/10.3390/pr9010002
APA StyleHwang, S. H., Koga, K., Hao, Y., Attri, P., Okumura, T., Kamataki, K., Itagaki, N., Shiratani, M., Oh, J.-S., Takabayashi, S., & Nakatani, T. (2021). Time of Flight Size Control of Carbon Nanoparticles Using Ar+CH4 Multi-Hollow Discharge Plasma Chemical Vapor Deposition Method. Processes, 9(1), 2. https://doi.org/10.3390/pr9010002






