Hydrogenation of Trans,Trans-Muconic Acid to Bio-Adipic Acid: Mechanism Identification and Kinetic Modelling
Abstract
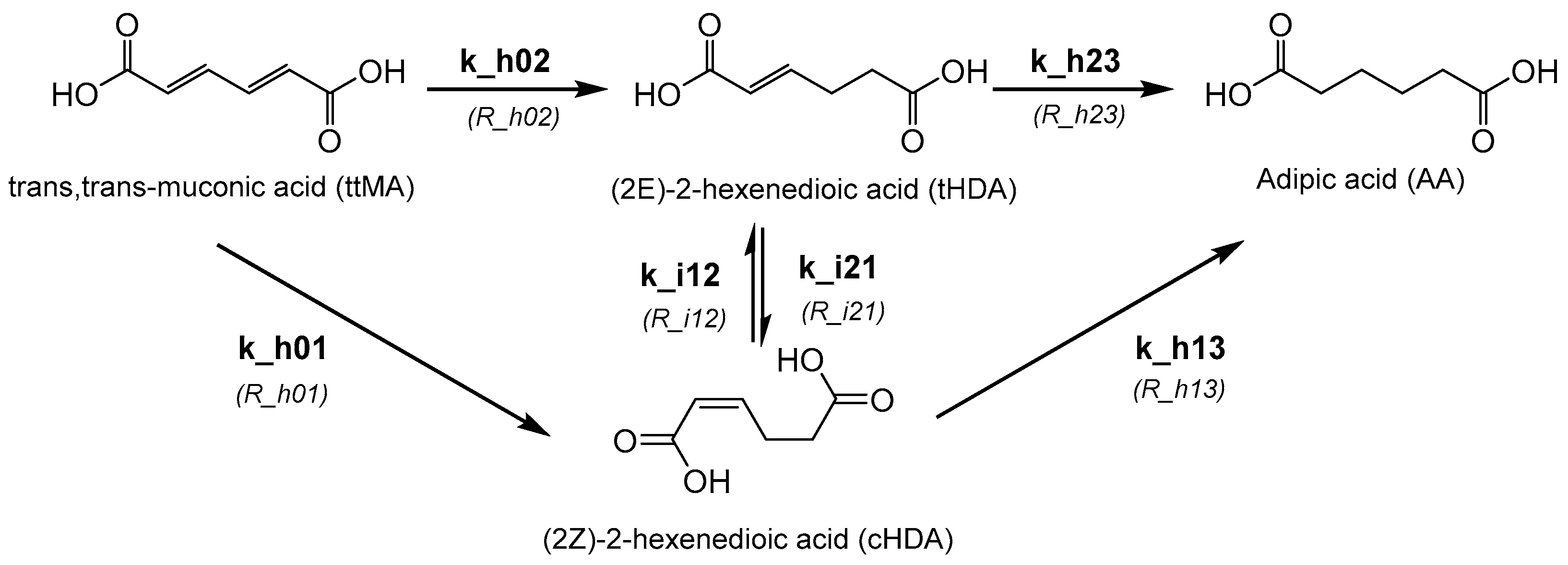
1. Introduction
2. Materials and Methods
2.1. Experimental
2.2. Kinetic Modelling
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ttMA | trans,trans-muconic acid |
| tHDA | (2E)-2-hexenedioic acid |
| cHDA | (2Z)-2-hexenedioic acid |
| AA | adipic acid |
| LHHW | Langmuir–Hinshelwood–Hougen–Watson model |
| SSE | sum of squared errors |
| R2 | coefficient of determination |
References
- Bart, J.C.J.; Cavallaro, S. Transiting from adipic acid to bioadipic acid. 1, Petroleum-based processes. Ind. Eng. Chem. Res. 2015, 54, 1–46. [Google Scholar] [CrossRef]
- Becker, J.; Lange, A.; Fabarius, C.; Wittmann, C. Top value platform chemicals: Bio-based production of organic acids. Curr. Opin. Biotechnol. 2015, 36, 168–175. [Google Scholar] [CrossRef]
- Collias, D.I.; Harris, A.M.; Nagpal, V.; Cottrell, L.W.; Schultheis, M.W. Biobased terephthalic acid technologies: A literature review. Ind. Biotechnol. 2014, 10, 91–105. [Google Scholar] [CrossRef]
- Xie, N.-Z.; Liang, H.; Huang, R.-B.; Xu, P. Biotechnological production of muconic acid: Current status and future prospects. Biotechnol. Adv. 2014, 32, 615–622. [Google Scholar] [CrossRef] [PubMed]
- Kruyer, N.S.; Peralta-Yahya, P. Metabolic engineering strategies to bio-adipic acid production. Curr. Opin. Biotechnol. 2017, 45, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Polen, T.; Spelberg, M.; Bott, M. Toward biotechnological production of adipic acid and precursors from biorenewables. J. Biotechnol. 2013, 167, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Bui, V.; Lau, M.K.; Macrare, D.; Schweitzer, D. Methods for Producing Isomers of Muconic Acid and Muconate Salts. U.S. Patent 20130030215 A1, 1 January 2013. [Google Scholar]
- Niu, W.; Draths, K.M.M.; Frost, J.W.W. Benzene-free synthesis of adipic acid. Biotechnol. Prog. 2002, 1, 201–211. [Google Scholar] [CrossRef] [PubMed]
- Johnson, C.W.; Salvachúa, D.; Khanna, P.; Smith, H.; Peterson, D.J.; Beckham, G.T.; Salvachua, D.; Khanna, P.; Smith, H.; Peterson, D.J.; et al. Enhancing muconic acid production from glucose and lignin-derived aromatic compounds via increased protocatechuate decarboxylase activity. Metab. Eng. Commun. 2016, 3, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Vardon, D.R.; Franden, M.A.; Johnson, C.W.; Karp, E.M.; Guarnieri, M.T.; Linger, J.G.; Salm, M.J.; Strathmann, T.J.; Beckham, G.T. Adipic acid production from lignin. Energy Environ. Sci. 2015, 8, 617–628. [Google Scholar] [CrossRef]
- Tuck, C.O.; Perez, E.; Horváth, I.T.; Sheldon, R.A.; Poliakoff, M. Valorization of biomass: Deriving more value from waste. Science 2012, 337, 695–699. [Google Scholar] [CrossRef]
- Averesch, N.J.H.; Krömer, J.O. Tailoring strain construction strategies for muconic acid production in S. Cerevisiae and E. Coli. Metab. Eng. Commun. 2014, 1, 19–28. [Google Scholar] [CrossRef]
- Suástegui, M.; Yu Ng, C.; Chowdhury, A.; Sun, W.; Cao, M.; House, E.; Maranas, C.D.; Shao, Z. Multilevel engineering of the upstream module of aromatic amino acid biosynthesis in saccharomyces cerevisiae for high production of polymer and drug precursors. Metab. Eng. 2017, 42, 134–144. [Google Scholar] [CrossRef] [PubMed]
- Rorrer, N.A.; Dorgan, J.R.; Vardon, D.R.; Martinez, C.R.; Yang, Y.; Beckham, G.T. Renewable unsaturated polyesters from muconic acid. ACS Sustain. Chem. Eng. 2016, 4, 6867–6876. [Google Scholar] [CrossRef]
- Rorrer, N.A.; Vardon, D.R.; Dorgan, J.R.; Gjersing, E.J.; Beckham, G.T. Biomass-derived monomers for performance-differentiated fiber reinforced polymer composites. Green Chem. 2017, 19, 2812–2825. [Google Scholar] [CrossRef]
- Matthiesen, J.E.; Suàstegui, M.; Wu, Y.; Viswanathan, M.; Qu, Y.; Cao, M.; Rodriguez-Quiroz, N.; Okerlund, A.; Kraus, G.; Raman, D.R.; et al. Electrochemical conversion of biologically produced muconic acid: Key considerations for scale-up and corresponding technoeconomic analysis. ACS Sustain. Chem. Eng. 2016, 4, 7098–7109. [Google Scholar] [CrossRef]
- Lu, R.; Lu, F.; Chen, J.; Yu, W.; Huang, Q.; Zhang, J.; Xu, J.; Rui, L.; Lu, F.; Chen, J.; et al. Production of diethyl terephthalate from biomass-derived muconic acid. Angew. Chemie Int. Ed. 2015, 55, 249–253. [Google Scholar] [CrossRef]
- Frost, J.W.; Miermont, A.; Schweitzer, D.; Bui, V.; Wicks, D.A. Terephthalic and Trimellitic Based Acids and Carboxylate Derivatives Thereof. U.S. Patent US8367858 B2, 5 February 2013. [Google Scholar]
- Capelli, S.; Motta, D.; Evangelisti, C.; Dimitratos, N.; Prati, L.; Pirola, C.; Villa, A. Bio Adipic Acid Production from Sodium Muconate and Muconic Acid: A Comparison of Two Systems. ChemCatChem 2019, 11, 3075–3084. [Google Scholar] [CrossRef]
- Bart, J.C.J.; Cavallaro, S. Transiting from adipic acid to bioadipic acid. part II. Biosynthetic pathways. Ind. Eng. Chem. Res. 2015, 54, 567–576. [Google Scholar] [CrossRef]
- Vardon, D.R.; Rorrer, N.A.; Salvachua, D.; Settle, A.E.; Johnson, C.W.; Menart, M.J.; Cleveland, N.S.; Ciesielski, P.N.; Steirer, K.X.; Dorgan, J.R.; et al. Cis,cis-muconic acid: Separation and catalysis to bio-adipic acid for nylon-6,6 polymerization. Green Chem. 2016, 18, 3397–3413. [Google Scholar] [CrossRef]
- Scelfo, S.; Pirone, R.; Russo, N. Highly efficient catalysts for the synthesis of adipic acid from cis,cis-muconic acid. Catal. Commun. 2016, 84, 98–102. [Google Scholar] [CrossRef]
- Capelli, S.; Rosengart, A.; Villa, A.; Citterio, A.; Di Michele, A.; Bianchi, C.L.L.; Prati, L.; Pirola, C. Bio-adipic acid production by catalysed hydrogenation of muconic acid in mild operating conditions. Appl. Catal. B Environ. 2017, 218, 220–229. [Google Scholar] [CrossRef]
- Matthiesen, J.E.; Carraher, J.M.; Vasiliu, M.; Dixon, D.A.; Tessonnier, J.P. Electrochemical conversion of muconic acid to biobased diacid monomers. ACS Sustain. Chem. Eng. 2016, 4, 3575–3585. [Google Scholar] [CrossRef]
- Draths, K.M.; Frost, J.W. Environmentally compatible synthesis of adipic acid from D-glucose. J. Am. Chem. Soc. 1994, 116, 399–400. [Google Scholar] [CrossRef]
- Thomas, J.M.; Raja, R.; Johnson, B.F.G.; O’Connell, T.J.; Sankar, G.; Khimyak, T. Bimetallic nanocatalysts for the conversion of muconic acid to adipic acid. Chem. Commun. 2003, 1126–1127. [Google Scholar] [CrossRef]
- Li, X.; Wu, D.; Lu, T.; Yi, G.; Su, H.; Zhang, Y. Highly efficient chemical process to convert mucic acid into adipic acid and DFT studies of the mechanism of the rhenium-catalyzed deoxydehydration. Angew. Chemie -Int. Ed. 2014, 53, 4200–4204. [Google Scholar] [CrossRef]
- She, X.; Brown, H.M.; Zhang, X.; Ahring, B.K.; Wang, Y. Selective hydrogenation of trans,trans-muconic acid to adipic acid over a titania-supported rhenium catalyst. ChemSusChem 2011, 4, 1071–1073. [Google Scholar] [CrossRef]
- Shiramizu, M.; Toste, F.D. Expanding the scope of biomass-derived chemicals through tandem reactions based on oxorhenium-catalyzed deoxydehydration. Angew. Chemie Int. Ed. 2013, 52, 12905–12909. [Google Scholar] [CrossRef]
- Sirasani, G.; Tong, L.; Balskus, E.P. A biocompatible alkene hydrogenation merges organic synthesis with microbial metabolism. Angew. Chemie-Int. Ed. 2014, 53, 7785–7788. [Google Scholar] [CrossRef]
- Neri, G.; Musolino, M.G.; Milone, C.; Galvagno, S. Kinetic modeling of 2,4-dinitrotoluene hydrogenation over Pd/C. Ind. Eng. Chem. Res. 1995, 34, 2226–2231. [Google Scholar] [CrossRef]
- Carraher, J.M.; Pfennig, T.; Rao, R.G.; Shanks, B.H.; Tessonnier, J.-P. Cis,cis-muconic acid isomerization and catalytic conversion to biobased Cyclic-C6-1,4-Diacid monomers. Green Chem. 2017, 21–25. [Google Scholar] [CrossRef]
- Rosengart, A.; Capelli, S.; Pirola, C.; Citterio, A.; Bianchi, C.L.; Prati, L.; Villa, A. Renewable adipic acid from the hydrogenation of trans, trans-muconic acid: Selection of a three phases kinetic model. Chem. Eng. Trans. 2017, 57, 931–936. [Google Scholar] [CrossRef]
- Froment, G.F.; Bischoff, K.B.; De Wilde, J. Chemical Reactor Analysis and Design, 3rd ed.; Wiley: New York, NY, USA, 2011. [Google Scholar]
- Lylykangas, M.S.; Rautanen, P.A.; Krause, A.O.I. Hydrogenation and deactivation kinetics in the liquid-phase hydrogenation of isooctenes on Pt/Al2O3. Ind. Eng. Chem. Res. 2004, 43, 1641–1648. [Google Scholar] [CrossRef]
- Buzzi-Ferraris, G.; Manenti, F. Interpolation and Regression Models for the Chemical Engineer: Solving Numerical Problems; John Wiley & Sons: Hoboken, NJ, USA, 2010. [Google Scholar]
- Veldsink, J.W.; Bouma, M.J.; Schöön, N.H.; Beenackers, A.C.M. Heterogeneous hydrogenation of vegetable oils: A literature review. Catal. Rev. 1997, 39, 253–318. [Google Scholar] [CrossRef]
- Rajashekharam, M.V.; Nikalje, D.D.; Jaganathan, R.; Chaudhari, R.V. Hydrogenation of 2,4-dinitrotoluene using a Pd/Al2O3 catalyst in a slurry reactor: A molecular level approach to kinetic modeling and nonisothermal effects. Ind. Eng. Chem. Res. 1997, 36, 592–604. [Google Scholar] [CrossRef]
- Jones, W.H. Catalysis in Organic Syntheses; Science, E., Ed.; Academic Press: Cambridge, MA, USA, 2013. [Google Scholar] [CrossRef]
- Chaudhari, R.V.; Rode, C.V.; Deshpande, R.M.; Jaganathan, R.; Leib, T.M.; Mills, P.L. Kinetics of hydrogenation of maleic acid in a batch slurry reactor using a bimetallic Ru-Re/C catalyst. Chem. Eng. Sci. 2003, 58, 627–632. [Google Scholar] [CrossRef]
- Zhan, Z.; Jackson, J.E.; Miller, D.J.; Zhang, Z. Kinetics of aqueous-phase hydrogenation of lactic acid to propylene glycol. Ind. Eng. Chem. Res. 2002, 41, 691–696. [Google Scholar] [CrossRef]
- Feng, Y.; Zhou, L.; Wan, Q.; Lin, S.; Guo, H. Selective Hydrogenation of 1,3-Butadiene Catalyzed by a Single Pd Atom Anchored on Graphene: The Importance of Dynamics. Chem. Sci. 2018, 9, 5890–5896. [Google Scholar] [CrossRef]

| Catalyst | T (°C) | P (bar) | Time (h) | Conversion (%) | AA yield (%) | Solvent | Ref. |
|---|---|---|---|---|---|---|---|
| Pt/C 10% | 25 | 3.5 | 3 | 100 | 90 | Water | [25] |
| Pt/C 10% | 25 | 34 | 2.5 | 100 | 97 | Water | [8] |
| Ru10Pt2/SiO2 | 80 | 30 | 5 | 91 | 96 | Ethanol | [26] |
| Pt/C 5% | 160 | n.a. | 12 | n.a. | 99 | Pentanol | [27] |
| Re/TiO2 | 210 | 68 | 5 | 100 | 90 | Methanol | [28] |
| Pd/C 10% | 25 | 7 | 4 | n.a. | 62 | n-butanol | [29] |
| Royer | 37 | 25 | 18 | n.a. | 75 | [30] | |
| Pd/C 1% | 24 | 24 | 0.3 | >97 | >97 | Water | [10] |
| Ni/Al2O3 14.2% | 60 | 10 | 5 | 100 | >99 | [22] | |
| Ni electrode | 25 | 25 | 1 | 50 | <5 | Acidic solution | [24] |
| Pt/C 5% | 60 | 4 | 2.5 | 100 | 100 | Water | [23] |
| Species | Kinetic Equation | |
|---|---|---|
| ttMA | (7) | |
| cHDA | (8) | |
| tHDA | (9) | |
| AA | (10) | |
| H2 | (11) |
| LHHW_13P | Case 1 | Case 2 | Case 3 | ||||
|---|---|---|---|---|---|---|---|
| Reference Parameters | Calculated Parameters | Divergence | Calculated Parameters | Divergence | Calculated Parameters | Divergence | |
| KttMA | 9.40 × 100 | 9.40 × 100 | 0.00% | 9.41 × 100 | +0.11% | 9.85 × 100 | +4.79% |
| KcHDA | 3.44 × 104 | 3.21 × 104 | −6.69% | 3.59 × 104 | +4.36% | 4.95 × 104 | +43.90% |
| KtHDA | 2.28 × 101 | 2.27 × 101 | −0.44% | 2.28 × 101 | 0.00% | 2.52 × 101 | +10.53% |
| Ah02 | 8.86 × 10−6 | 1.00 × 10−5 | limited | 1.00 × 10−3 | limited | 1.00 × 10−1 | limited |
| Eah02 | 2.75 × 104 | 2.75 × 104 | 0.00% | 2.75 × 104 | 0.00% | 2.73 × 104 | −0.73% |
| Ah23 | 1.39 × 10−1 | 1.39 × 10−1 | 0.00% | 1.40 × 10−1 | +0.72% | 2.40 × 10−1 | +72.66% |
| Eah23 | 4.01 × 104 | 4.01 × 104 | 0.00% | 4.01 × 104 | 0.00% | 3.97 × 104 | −1.00% |
| Ai12 | 2.23 × 101 | 2.23 × 101 | 0.00% | 2.23 × 101 | 0.00% | 2.30 × 101 | +3.14% |
| Ai21 | 5.17 × 100 | 5.23 × 100 | +1.16% | 5.24 × 100 | +1.35% | 5.02 × 100 | −2.90% |
| Eai12 | 2.95 × 104 | 2.05 × 104 | −30.51% | 2.92 × 104 | −1.02% | 2.60 × 104 | −11.86% |
| Eai21 | 7.63 × 105 | 7.53 × 105 | −1.31% | 7.51 × 105 | −1.57% | 8.25 × 105 | +8.13% |
| Ah13 | 8.66 × 100 | 8.66 × 100 | 0.00% | 8.72 × 100 | +0.69% | 8.89 × 100 | −2.66% |
| Eah13 | 3.89 × 104 | 3.29 × 104 | −15.42% | 3.80 × 104 | −2.31% | 4.67 × 104 | +20.05% |
| RR | 7.76 × 10−4 | 7.69 × 10−4 | 7.69 × 10−4 | 7.71 × 10−4 | |||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rosengart, A.; Pirola, C.; Capelli, S. Hydrogenation of Trans,Trans-Muconic Acid to Bio-Adipic Acid: Mechanism Identification and Kinetic Modelling. Processes 2020, 8, 929. https://doi.org/10.3390/pr8080929
Rosengart A, Pirola C, Capelli S. Hydrogenation of Trans,Trans-Muconic Acid to Bio-Adipic Acid: Mechanism Identification and Kinetic Modelling. Processes. 2020; 8(8):929. https://doi.org/10.3390/pr8080929
Chicago/Turabian StyleRosengart, Alessandro, Carlo Pirola, and Sofia Capelli. 2020. "Hydrogenation of Trans,Trans-Muconic Acid to Bio-Adipic Acid: Mechanism Identification and Kinetic Modelling" Processes 8, no. 8: 929. https://doi.org/10.3390/pr8080929
APA StyleRosengart, A., Pirola, C., & Capelli, S. (2020). Hydrogenation of Trans,Trans-Muconic Acid to Bio-Adipic Acid: Mechanism Identification and Kinetic Modelling. Processes, 8(8), 929. https://doi.org/10.3390/pr8080929







