1. Introduction
At the present, there is a remarkable global interest to identify and analyze antioxidant compounds from different food sources [
1]. Many studies have revealed that these phytochemicals may prevent the development of diabetes and cardiovascular diseases, having favorable impacts on cardiometabolic health, including lowering blood pressure, improving lipid profiles and decreasing markers of inflammation [
2,
3]. Antioxidant compounds can prevent these diseases by reducing or inhibiting the adverse effects of free radicals. These radicals are very reactive and can attack biologically relevant molecules, leading to cell damage [
2]. Consequently, there has been an increasing interest in determining the antioxidant potential of natural products such as phenolic compounds [
2,
4,
5].
Between the phenolic compounds, anthocyanin pigments and flavonoids [
3,
6] are powerful antioxidants that protect cells from various forms of cancer. According to some nutritionists, the human food should be rich in antioxidant compounds (especially in the areas with high pollution degree) [
7]. The only source for the production of anthocyanin pigments are fruits and vegetables. Anthocyanins are found in blueberries, blackberries, raspberries, cherries, cranberries, black currant, elderberry, eggplant, purple corn, black beans, black carrot and purple potatoes [
8,
9].
The phenolics contained in cider beverage have a great importance from the sensorial point of view [
10,
11]. They may affect the antioxidant activity, the taste, the visual aspect and the preservation of ciders. No study has been performed on the antioxidant properties and protein-precipitating capacity (PPC) of the ciders enriched with blueberry and black carrot extracts and treated with ultrasounds. The present work is focused to present just these issues, being focused on the effects of the extracts and ultrasounds treatments on the antioxidant properties, TPC (total phenolic compounds), TFC (total flavonoids compounds) and PPC of the cider variants.
The application of ultrasound-assisted extraction for different materials and its opportunities in the food processing industry has proved many advantages [
8,
12,
13,
14,
15,
16,
17,
18,
19]. The ultrasound is used in the extraction of biologically compounds from biologic matrices such as in the extraction of phenolics from plants and waste food materials [
17,
18]. Many studies on several bioactive phenolic compounds derived from plants reported the importance of these compounds and the huge demand increase of these natural products on the commercial market [
8,
12,
13,
14,
15,
16]. In food processing, ultrasound treatment may be used as an efficient and environmentally friendly treatment for enhancing the nutritional status of products in terms of increasing the bioactive compounds, especially the phenolics [
20,
21]. It was reported that ultrasound was applied in extraction of plant materials because of increased sugar content, total acid content, phenolics content as well as color density of juices [
22]. Moreover, the ultrasound treatment can influence the quality parameters of food products in terms of aromatic profile and sensory properties [
23,
24]. In the last years, many research studies explored the effects of sonication time and amplitude on the total phenolic content in order to optimize the UAE conditions for phenolic compounds [
25,
26,
27,
28].
In certain conditions, depending to amplitude, frequency, time of treatment, the use of ultrasound is considered a nonthermal processing method, numerous advantages deriving from it (food nutritional quality improvement, thermo sensitive compounds protection, etc.) [
14,
19]. The application of ultrasound in the case of liquids, as in the case of the present work, is based on the phenomenon of cavitation, which involves the formation, development and implosion of bubbles along with the generation and propagation of vibrations in the mass it crosses. The implosion, finally, causes mechanical (turbulent, shear), thermal and also chemical effects [
19].
The experimental studies of this research were focused on the following aspects:
- -
to evaluate the effects of several ultrasound treatments (different amplitudes and time) on TPC, TFC, antioxidant power and protein precipitation capacity of cider enriched with black carrot and blueberry extracts;
- -
to evaluate the antioxidant capacity of the cider variants using: FRAP (ferric-reducing antioxidant power) assay, ABTS (2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid)) assay and DPPH (2,2-diphenyl-1-picrylhydrazyl) radical scavenging activity. These assays were chosen because they work under different mechanisms: FRAP and DPPH methods use electron transfer mechanism, while the ABTS method uses both hydrogen atom transfer and single electron transfer mechanisms, depending on the antioxidant present [
29]. In addition, they are simple and fast, which is very important when many measurements must be made on many samples.
Phenolic compounds also take part in the formation of haze and sediments because of protein–phenolic interactions, which are the most frequent cause of haze in beverages [
30]. The PPC was determined in order to study the tendency of the phenolic compounds of ciders to form solid compounds with the proteins contained in them. It was measured following Hagerman’s protocol, a robust method that works well with virtually all plant extracts [
31] and is still being used today [
32]. The presence of some haze and solids is a characteristic of cider and thus, bottles are usually shaken before opening to distribute the turbidity evenly. However, excessive haze or solid quantity is negatively perceived by consumers.
The musts were obtained from a single apple variety. After this, the material was enriched with extracts concentrated in antioxidant compounds (obtained in laboratory conditions [
33] and ultrasound-treated using different amplitudes values and times). By spontaneous fermentation, 18 experimental variants of ciders (two extracts additions and ultrasounds treatment parameters) were obtained. The evolution of the musts was followed for six months by measuring the mentioned parameters throughout three samplings. This study presents the results of the final products obtained (the last sampling).
The purpose of this study was to estimate the effect of ultrasound treatments and of two natural extracts additions on the level of TPC and TFC on antioxidant activity of different cider variants. In addition, the PPC of the ciders variants was determined because this indicator is one the indicators of the quality of this beverage.
2. Materials and Methods
2.1. Chemicals and Reagents
Folin–Ciocâlteu reagent, aluminum chloride were purchased from Sigma-Aldrich (St. Louis, MO, USA). DPPH (cat. no. D9132, purity 97%) and ABTS (cat. no. A1888, purity 98.6%) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Quercetin dihydrate and gallic acid were purchased from Karl Roth GmbH (Karlsruhe, Germany). Hydrochloric acid, sodium acetate, sodium carbonate, sodium chloride and sodium hydroxide were purchased from POCH BBASIC (Glivice, Poland). Total Antioxidant Status assay kit was obtained from Rel Assay Diagnostics (Gaziantep, Turkey). Albumin from bovine serum (BSA), iron(III) chloride hexahydrate, tannic acid, triethanolamine, 2,4,6-tripyridyl-s-triazine, sodium dodecyl sulfate (SDS), (trans)-ferulic acid, trolox were purchased from Sigma-Aldrich (St. Louis, MO, USA). Solutions were prepared with double-distilled water.
2.2. Cider Preparation
The apples used to make cider were from Royal Gala variety, Voineasa origin (Valcea region, Romania, latitude 45°24′56′′ N, longitude 23°57′49′′ E) harvested in September 2018 from private orchards. The variety of apple used in the study is recognized as a good variant for obtaining apple juices in Romania, given that it has complex organoleptic properties (especially more mild and sweet aroma), comparing to the varieties studied in 2013 season, when another varieties Golden, Jonathan and Renet were used [
34]. Fresh apples were crushed with crusher–destemmer (Enoventa Technologies Enologiche, Piazzola sul Brenta, Italy) followed by a pressing process with a hydraulic press machine (L.U.C.M.E. Elettromeccanica, Verona, Italy). Apple must was obtained in three repetitions (30 kg of apples/repetition). The juice was collected into glass containers for starting fermentation. The temperature during fermentation was maintained at 19 °C. Alcoholic and malolactic fermentations took place spontaneously by indigenous microflora. The development of alcoholic fermentation was followed by the decrease in density and the malolactic fermentation by the decrease in malic acid content.
Vitamin C (0.01 g/L) was added to the must before starting fermentation. Because of the presence of this compound (that could produce a degradation of the natural yeast present in the crude juice and a delay of the fermentation), it was necessary to use fermentation yeast of exogenous nature, added to the wort after about two days. The Saccharomyces cerevisiae yeast added to the wort had also the role of removing harmful bacteria from the wort (which can affect the taste of the drink). The added yeast was necessary to ensure the start of fermentation, since in some cases the use of addition may lead to the cessation of the activity of indigenous microflora naturally present in the apple must. It was then desired to stop fermentation when the alcoholic concentration reached just over 5% by volume (5.13% v/v). The cider was subsequently racked to remove the sediments and stored at refrigeration temperature. Sulfur dioxide 50 ppm was added to each decantation (but not exceeding the total quantity of SO2 200 ppm in any cider, by summarizing all additions). Post-fermentation handling is also crucial for cider quality.
2.3. Extracts Added to the Cider
The extracts used for improving the antioxidant capacity of the cider were the following:
- -
black carrot colorant (commercial extract added to the cider variants in proportion of 0.3%, v/v);
- -
blueberry extracts (extract obtained in laboratory, added to the cider variants in proportion of 5%, v/v)
The black carrot colorants used at industrial level, it is named in international language “colored concentrate black carrot HC red–blue”, manufactured by Dohler GMBH company in Darmstadt, Germany.
The blueberry extract was prepared in laboratory, using fruits from plant growing in spontaneous flora (
Vaccinium) and 2% acidulated water as solvent [
33]. The citric acid was used for acidification. The fruits samples were freeze-dried with a 0.12 mBa vacuum at −50 °C (ScanVac CoolSafe 55–9 Pro freeze dryer, Denmark), ground to a fine powder (using a coffee grinder). After this, the sample was homogenized for 30 min. at a temperature of 30 °C with the solvent (report sample: solvent = 1:20,
w/
v), using the extraction solid–liquid. This blueberry extract was ultrasound-treated (200 mL placed in glass beaker, the ultrasound probe fixed at 35 mm depth in the mixture, amplitude A20%, 5 min) using a 750 W ultrasonic processor for small and medium volume applications (VCX 750, Sonics & Materials, Inc., Newtown, CT, USA) with a 13 mm probe, at a constant frequency of 20 kHz [
33].
Both extracts were added before ultrasound treatments.
2.4. Ultrasound Treatment of Enriched-Cider Variants
The cider variants enriched with the extracts were treated with ultrasound (VCX-750, Sonics & Materials, Inc., Newtown, CT, USA) at 750 W, with constant frequency of 20 kHz at amplitude A20%, A30% and A40% for time periods of 2, 5 or 7 min using a probe of 19 mm and a sample of 500 mL cider. This combination of amplitude and time was established according results of previous study, where a release of antioxidant compounds was obtained [
16]. We used low values for amplitude and time for not improving the values of PPC.
2.5. Sample Preparation for Analysis
Cider samples of about 200 mL were taken at the end of alcoholic fermentation (density ≈ 1000 g/L). Each sample was homogenized by manual shaking and degassed by using a stirrer. Immediately afterwards, density and malic acid content were measured. A 100 mL portion was centrifuged at 8200 rpm (9000× g) for 20 min in a Hettich EBA21 centrifuge (Thermo Fisher Scientific, Walthan, MA, USA). This portion was used for determination TPC, TFC and to perform FRAP, ABTS, DPPH assays and PPC. The sampling procedure described was performed in one day to avoid oxidation of phenolic compounds and continuation of fermentation.
2.6. Total Phenolic Content (TPC) Analysis
The TPC was determined spectrophotometrically by Folin–Ciocâlteu method [
35] with several modifications. Fifty microliters of samples were mixed with 100 μL distilled water in a 96 well flat bottom assay plate (NUNC, Roskilde, Denmark). Fifty microliters Folin–Ciocâlteu reagent was added and mixed for 1 min in the plate reader (Tecan, Sun Rise™, software Magellan™, Männedorf, Switzerland). After 5 min, 80 μL of a 20% solution (
w/
v) of Na
2CO
3 were added and mixed; the microplates were shaken for 5 min. in the plate reader. After this, the plates were incubated at room temperature in the dark, agitating at 150 rpm on a MicroPlate Shaker (Biosan PST-60HL-4, Riga, Latvia) for 90 min. The absorbance of the samples was determined at 725 nm with a spectrophotometer plate reader (Tecan, SunRise™, software Magellan™, Männedorf, Switzerland). Gallic acid was used as standard and total phenolic content was expressed as milligrams GAE (gallic acid equivalents) per liter of cider.
2.7. Total Flavonoid Content (TFC) Analysis
The method is based on a nitration reaction followed by the formation of an aluminum complex which turns to red in a basic medium [
36]. The TFC was measured by a colorimetric method [
36] with several minor changes [
37]. So, in 1.5 mL tubes, 150 μL cider samples were mixed with 600 μL distilled water and 45 μL sodium nitrite 7.5%. The mixture was left to react for 5 min. A volume of 45 μL aluminum chloride 10% solution was added in the tubes, mixed by inversion and allowed to react for 1min. Finally, 300 μL of a 1 N sodium hydroxide solution and 360 μL distilled water were added, and the tubes were vortex mixed (Vortex TopMix Stirrer, Heidolph™, Schwabach, Germany). The absorbance at 510 nm was measured using a spectrophotometer DR2800 (Hach Lange, Loveland, CO, USA). Values of the absorbance samples were interpolated into a least squares regression equation (a 5-point calibration curve with an R
2 value of 0.998), which was calculated with the absorbance and the corresponding concentration of each quercetin standard [
37]. Final results were calculated taking into account the dilution factors applied and were expressed as mg quercetin equivalents (QE) per liter of cider [
37].
2.8. FRAP Assay
This assay was performed according to the Benzie and Strain procedure [
38] using Trolox as a standard instead of ascorbic acid and with some changes to adapt the method to the automatic analyzer. The following solutions were prepared: (a) acetate buffer 300 mM pH 3.6; (b) 2,4,6-tripyridyl-
s-triazine 10 mM in HCl 40 mM; (c) FeCl
3·6H
2O 20 mM; (d) working FRAP reagent prepared at the time of use (mixture of the previous solutions in the ratio of 10:1:1, respectively).
The assay was achieved as follows. Sample or standard (3 μL) was mixed with 300 μL of working FRAP reagent and absorbance at 600 nm was measured after 4min. Trolox standards (0 to 5 mmol/L) were used to construct the calibration curve (absorbance versus Trolox mmol/L). Results are given in mmol Trolox Equivalents per liter of cider (mmol TE/L).
2.9. ABTS Assay
This assay was measured according to the procedure described by Erel [
39] and performed with the Total Antioxidant Status assay kit [
40]. The commercial kit provides a 0.4 mmol/L acetic/acetate buffer solution of pH 5.8 and a 30 mmol/L solution of ABTS radical cation.
The assay was achieved as follows. Sample or standard (4 μL) was mixed with 200 μL of acetic/acetate buffer and 25 μL of ABTS radical cation reagent and absorbance at 670 nm was measured after 5 min. Trolox standards (0 to 5 mmol/L) were used to construct the calibration curve (absorbance versus Trolox mmol/L). Results are given in mmol Trolox Equivalents per liter of cider (mmol TE/L).
2.10. DPPH Assay
Volumes 20-μ Lof samples were added to 20 mL of distilled water in a 96-well bottom microplate. Two hundred microliters of 120mg/L DPPH radical solution (using ethanol as a solvent) was then added and mixed thoroughly. The absorbance was measured using a plate reader (Tecan, SunRise™, software Magellan™, Männedorf, Switzerland) at 515 nm after keeping the plates in the dark for 30 min. A control with 20 μL of ethanol was also included in each plate.
The DPPH radical scavenging activity was calculated with the formula:
where A control is the absorbance of the control at 515 nm and A sample is the absorbance of the sample at 515 nm.
2.11. PPC
According to Hagerman’s method [
31], the following solutions were prepared: (a) 0.20 M acetate buffer of pH 4.9 in 0.17 M NaCl; (b) 1 mg/mL bovine serum albumin (BSA) dissolved in the previous buffer; (c) 50 mL/L triethanolamine (TEA) in 10 g/L sodium dodecyl sulfate (SDS); 0.01 M FeCl
3 in 0.01 M HCl. The assay was performed as follows. A total of 2.00 mL BSA solution was dispensed into 15 mL centrifuge tubes, and 1 mL of sample solution was added to it. Both the solutions were mixed immediately and allowed to sit for 24 h at 4 °C. Then the mixture was centrifuged for 15 min at 8200 rpm and poured off the supernatant. The pellet was dissolved in 4.00 mL SDS/TEA solution, followed by the addition of 1.00 mL FeCl
3 solution; the mixture was vortexed immediately. After about 15 min, the absorbance was measured at 510 nm using a spectrophotometer DR2800 (Hach Lange, Loveland, CO, USA).
2.12. Statistical Analysis
Data were performed by using SPSS (statistical package for the social sciences) software and analyzed by ANOVA and Duncan’s multiple range test (scored as significant if p < 0.05). It was analyzed the correlation between the variables, using the Pearson correlation coefficients.
3. Results
3.1. Cider Variants and Classification
The cider experimental variants obtained and tested in this study are presented in
Table 1.
According to the English classification of cider apple varieties, they are divided into four groups: bittersharp (>4.5 g/L malic acid, >2 g/L tannic acid), sharp (>4.5 g/L malic acid, <2 g/L tannic acid), bittersweet (<4.5 g/L malic acid, >2 g/L tannic acid) and sweet (<4.5 g/L malic acid, <2 g/L tannic acid) [
41]. All the cider variants obtained in this research were bitter sweet [data not shown]. Compared with the products obtained in 2013 [
34], a more aromatic cider variants were obtained.
3.2. Total Phenolic Content (TPC)
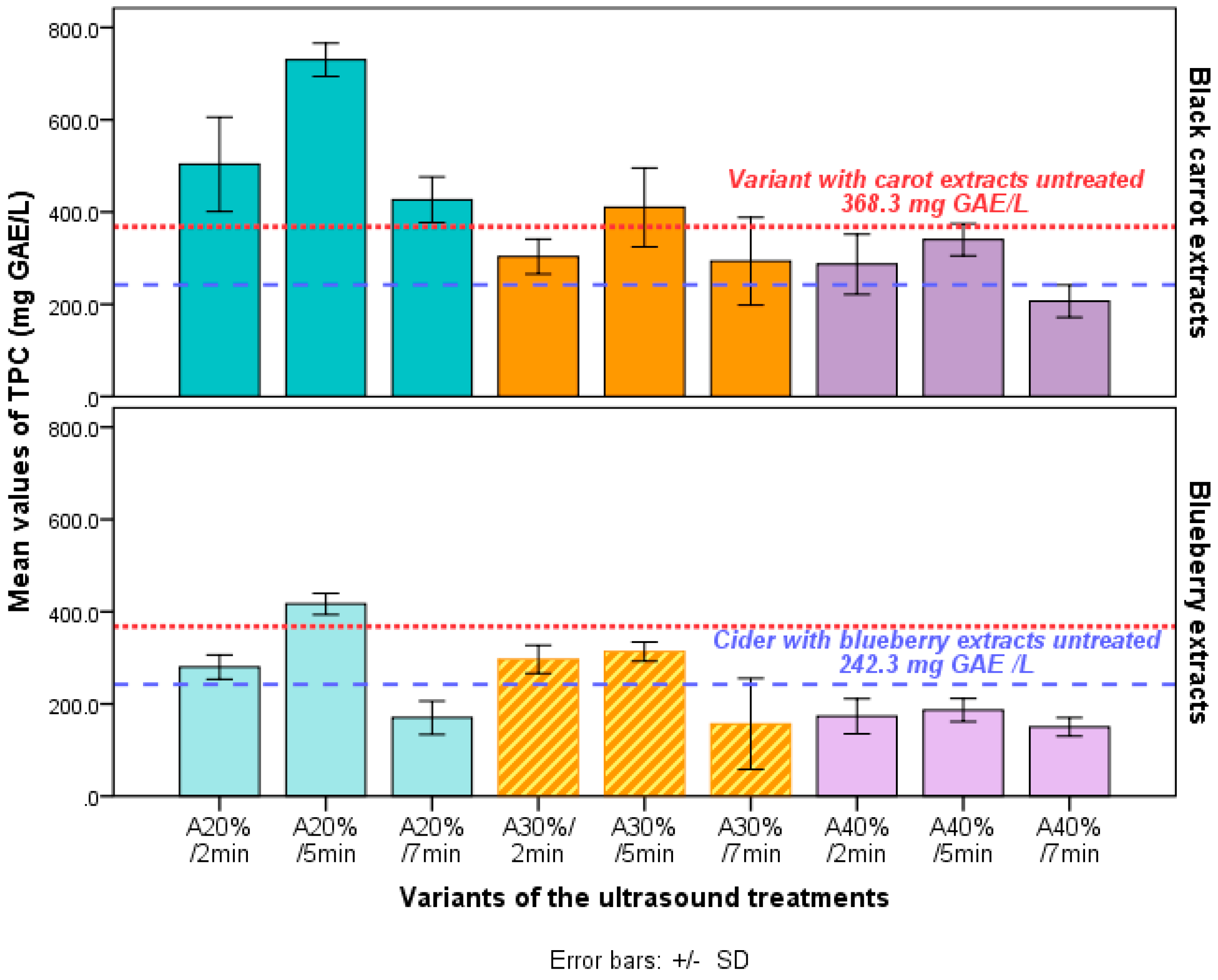
Table 2 shows the total phenolic content of cider variants analyzed. There were significant differences between the variants, samples enriched with black carrot and treated using the amplitude A20% having the highest TPC values (from 423.6 to 730.5 mg GAE/L). The lowest TPC was found for the cider samples with blueberry extracts and treated using the highest amplitude A40% (from 152 to 184 mg GAE/L).
As shown in
Figure 1,
Table 2 and
Table 3 the sonication conditions of the cider with extracts added had a significant influence of the TPC.
The results show that TPC of the cider enriched with black carrot extracts was significant higher (
p < 0.05) than the cider untreated (
Figure 1,
Table 3a). It was an increase of TPC values from control M (191.1 ± 32.7 mg GAE/L) for both extracts added (
Figure 1,
Table 3a).
Regarding the sonication parameters, the highest TPC of the ciders was obtained using amplitude A20% (
Table 3b) and 5 min as time period. Hence, the ultrasonic treatments using this parameters provide positive improvements such that the controls. The effectiveness of the ultrasound treatments could result from the economical prices of the process (by reducing the amplitude and time periods) and from the improvement of phenolic compound yield.
Similar results (regarding the high value of TPC observed after samples ultrasonic treatments at low values of amplitude) has also been reported by Lieu et al. for grape [
42], Adekunte et al. [
43] for tomato juice and Bhat et al. [
44] for lime juice which presented higher total phenolic compound content after sonication. Phenolic compounds are found in soluble form in the vacuole or bound to the pectin, hemicellulose traces of the wall [
45]. It is possible that ultrasound enhances the release of these compounds from the cell wall [
46], through the collapse via cavitation in the surroundings of colloidal particles [
47]. During the cider storage, the senescence and decomposition of the cell structure could take place and thus, free phenolic acids and free amino acids are released, contributing to increasement of total phenolic compounds. This change may be attributed to cell wall degradation caused by microbial growth during first days of storage in control samples.
3.3. TFC of the Cider Variants
Figure 2 shows the results of total flavonoid content. Ultrasonic treated samples exhibited higher total flavonoid content values compared to the controls in both case of extracts additions. With the advantage of extracts added (black carrot extracts) the untreated sample had an increase of these compounds, while the variants with blueberry extracts added presented slight variability.
Compared with the control C (30.42 ± 3.154 mg QE/L), the highest values for TFC in case of the ciders enriched with black carrot were obtained for the lowest amplitude of ultrasonic sonication treatments (A20%) (43.82 ± 7.843 mg QE/L for 2 min, 45.13 ± 1.655 mg QE/L for 5 min and, respectively 40.62 ± 1.450 mg QE/L for 5 min) (
Figure 2,
Table 4).
Regarding the samples with blueberry extracts, only two treatments variants lead to highest values of TFC compared with the control B. These values were obtained using the lowest amplitude A20% for 2 min (31.99 ± 3.387 mg QE/L) and for 5 min (35.85 ± 1.618 mg QE/L).
The results show that TFC of the cider enriched with black carrot extracts was significantly higher than the variant C (
Table 5). It was a significant increase of TFC values and for variant C305 if the comparison was made using the experience mean.
3.4. PPC of the Cider Variants
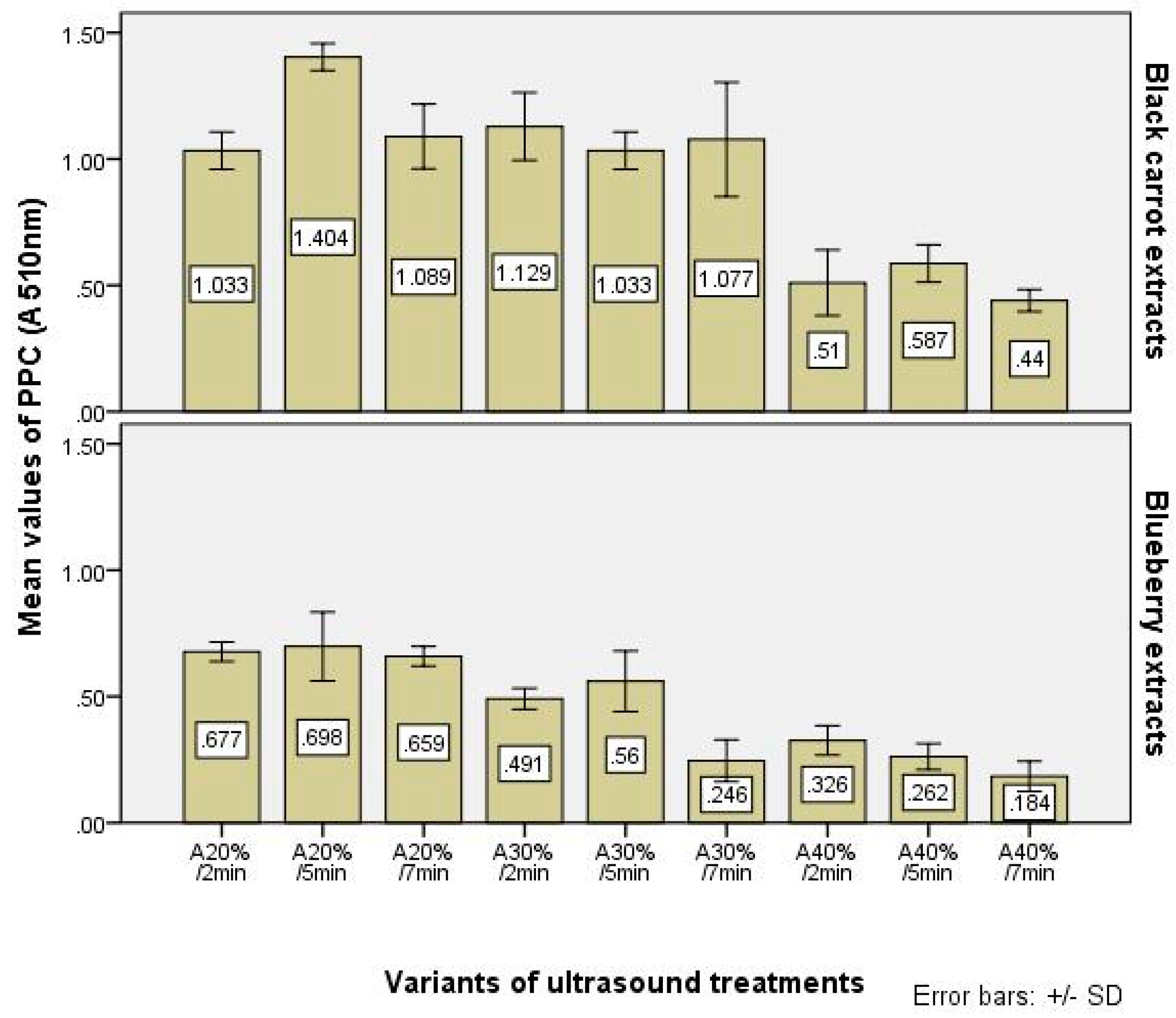
In this research, for estimate the PPC, we used only the values of absorbance values (at 510 nm) of samples treated as described in the previous paragraph. The values of this parameter are presented in
Table 6 and
Figure 3. They are correlated with the values of TPC and TFC. Because of the treatments, the cider variants with the highest TPC and TFC values had higher values of PPC.
Many papers were published about the interaction between phenolics and proteins to form haze or sediments in beverages, mostly beers, wines and fruit juices, including apple juices [
30,
48,
49,
50,
51]. According to them, protein–phenolic interactions are the most frequent cause of hazes in beverages. Flavonols—especially catechins and procyanidins—are considered as main contributors to haze.
However, there is a single work that tries to find relations between haze or solids formed in ciders and individual phenolics [
52]. Milet et al. (2017) reported that the phenolics found in haze were, in decreasing concentration order, chlorogenic acid > 4-
p-coumaroylquinic acid > phloridzin > phloretin 2′-O-xyloglucoside > epicatechin. The authors are not sure about the procyanidins content. Apparently, phenolics in ciders do not have the same behavior pattern as reported in other beverages, including apple juices. In the next work of the same team about this subject [
53], they demonstrated that not procyanidins, but their oxidized derivatives are responsible for haze formation.
3.5. Antioxidant Capacity of the Cider Variants
Table 7 shows the antioxidant capacity of the experimental cider variants (evaluated using FRAP, ABTS and DPPH assay). As shown in this table, the values of FRAP, ABTS and DPPH inhibition were significantly different between the ciders tested (
p < 0.05).
Regarding the antioxidant capacity expressed using all the assay, there were significantly differences between the variants, the cider with black carrot colorant ultrasound-treated (A20%, 5 min (C205) having the highest values (14.69 ± 0.09-mmol TE/L for FRAP assay, 25.79 ± 0.89-mmol TE/L for ABTS assay, respectively 65.35% ± 2.59% for DPPH radicals inhibition %). The lowest values for all these antioxidant assay were found for the cider with blueberry extracts added and treated at the highest amplitude and time period (B407), (2.16 ± 0.18-mmol TE/L for FRAP assay, 5.48 ± 1.75-mmol TE/L for ABTS assay, respectively 25.94% ± 4.309% for DPPH radicals inhibition%).
The antioxidant capacity reported in our study are higher than those reported by Picinelli (2009) [
54]. In comparison with other alcoholic and non-alcoholic beverages, the antioxidant power of many cider variants obtained using the extracts and ultrasound treatments was more power full, as you can see in
Table 8.
In
Table 8, only ABTS values are displayed in order to make comparisons, since they are given in most of the articles. FRAP values appear less often. The antioxidant activity range of the ciders between 5.48 ± 0.89 and 25.79 ± 1.75-mmol TE/L, lies over that the treatments and extracts added and is a little higher to that of red wines. This is an important fact because the great antioxidant activity of wines is often quoted as a beneficial aspect. Moreover, the antioxidant activity of our ciders variants is greater than that of other alcoholic beverages like beer, Cognac, Armagnac and rum. With respect to non-alcoholic beverages, the antioxidant activity of the ciders is similar to that of coffee and is greater than that of tea (excepting the variant B407), orange juice, olive oil, sunflower oil and cola. The comparison with orange juice is particularly interesting because this drink is always quoted as model of healthy and antioxidant beverage.
Evaluation of antioxidant activities of a selected antioxidant needs multiple test systems. DPPH free radical is a stable group which is broadly used in evaluating the scavenging activity of antioxidant on the free radical. In DPPH assay, the DPPH radical inhibition% by antioxidants was attributed to their hydrogen-donating capability.
Figure 4A shows the scavenging activities for DPPH radicals, the DPPH free radical inhibition% increased from 37.24% ± 6.416% to 65.9% ± 2.587% for the cider enriched with black carrot extracts and from 25.94% ± 4.309% to 56.29% ± 2.884% for the variants with blueberry extracts.
The results indicate that the extracts added have good percent inhibition activity on the DPPH radicals, but lower than that of the other radicals used in our research (
Table 7).
As shown in
Figure 4, the ultrasonic sonication conditions of the cider with extracts added had a significant influence of the antioxidant activity tested by FRAP and ABTS assay.
The results show that a good antioxidant capacity is preferably to obtain using a lower amplitude (in our case A20%) and a period time 5 min and this is most probably due to the high sensitivity of phenolic compounds to ultrasonic amplitude power. This observation is in agreement with the previous finding by Nayak and Rastogi (2011) [
64] where they found that an amplitude of 10%–14% was optimum for improving the antioxidant capacity.
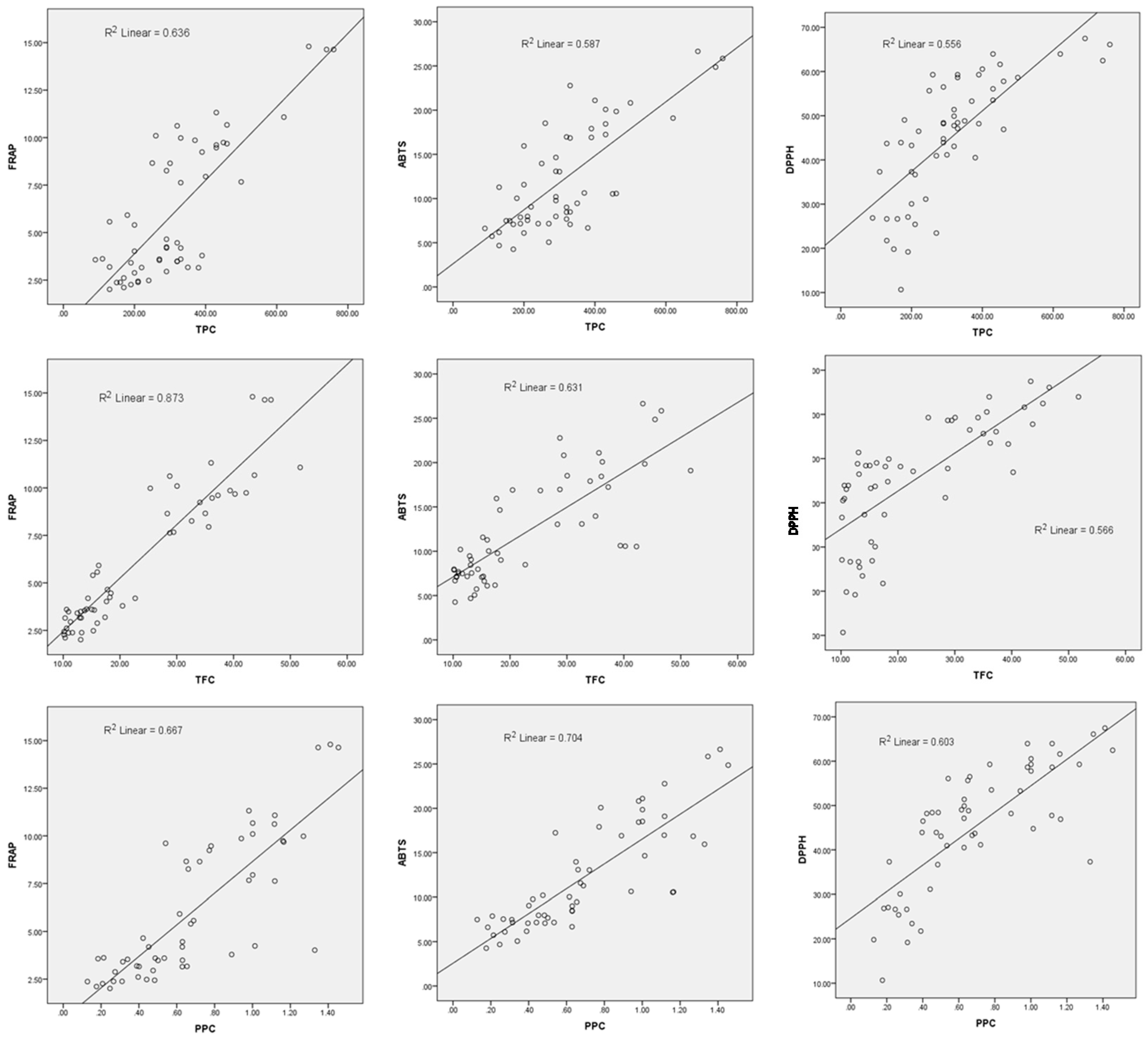
In our study, the TPC and TFC values were correlated with the antioxidant activity estimated using FRAP, ABTS and DPPH inhibition percent. The lowest value of the correlation coefficient (R2) was obtained in case of DPPH.
Our study show that the FRAP assay values were good correlated with the TFC (R2 = 0.873). In all cases the best correlations were identify in case of TFC, compared with TPC. It is possible that some differences between phenolic content influenced the antioxidant capacity because the complex samples (extracts) could content some phenolic non antioxidants.
A good correlation was found and between TFC and FRAP values (R
2 = 0.873) and between PPC and ABTS values (R
2 = 0.704) (
Figure 5).
Table 9 presents the Pearson correlation coefficients (
p < 0.01) for the interaction between TPC, TFC, PPC and the antioxidant capacity of the cider variants (expressed by ABTS, FRAP values and DPPH percent inhibition values). The results reveal a positive correlation between the antioxidant activity and all the chemical compounds tested.
All experiments were used without experimental planning (multivariate). We intend to achieve the optimum condition for obtaining the best cider variant in a next more complex article. Therefore, we could go further in the experiments to optimization the process, we intent to add to the results presented in this study the content of other compounds that gives the nutritional value of our cider variants (vitamin C, tannic acid, malic acid, carbohydrates).
4. Conclusions
The results presented in this study highlight the level of total phenolic and flavonoids content correlated with the antioxidant capacity (expressed by three types of assay) of several cider variants enriched by adding extract from black carrot and from blueberry fruits and ultrasonic sonication treated.
The FRAP, ABTS and DPPH scavenging activity is due among others to the content in phenolics and flavonoids (which were detected in significant amount in the variants of cider ultrasonic treated using frequency of 20 kHz, amplitude of 20%, time period five minutes). The highest mean values for these compounds were found in cider sampled from variant enriched with black carrot extracts (0.3%, v/v) treated in the conditions presented above. Regarding the cider enriched with blueberry extracts (5%, v/v) the highest values of TPC and TFC values were obtained for the samples treated in similar conditions presented above. Unfortunately, the PPC of the variants very rich in polyphenols had high values and could damage the quality of the ciders. However, the intensive color of these variants cover up the haze of the beverage.
The values of FRAP-, ABTS- and DPPH-scavenging activity were significantly different between the cider variants tested (p < 0.05), the variant with the highest TPC and TFC values having the highest percentage of radical inhibition.
The results indicated that the cider variants enriched using the carrot and blueberry extracts exhibited good antioxidant capacity, similar or highest to that of red wines and greater than that of orange juices.
The cider enriched with antioxidants could contribute to the daily intake of phenolics and flavonoids (phytochemicals with functional and antioxidant potential) and their consumption thereby may have positive effects on the human health. We could go further in the experiments to identify the beneficial compounds “hidden” in the cider variants obtained in our study.