Liquid Biphasic System: A Recent Bioseparation Technology
Abstract
1. Introduction
2. Liquid Biphasic System
2.1. Polymer-Based LBS
2.2. Organic Solvent-Based LBS
2.3. Ionic Liquid-Based LBS
2.4. Deep-Eutectic-Solvent-Based LBS
2.5. Surfactant/Detergent-Based LBS
3. Advance Technologies Integrated with LBS
3.1. Bubble-Assisted LBS
3.2. Ultrasound-Assisted LBS
3.3. Electricity-Assisted LBS
4. Key Parameters Affecting LBS
4.1. Type and Molecular Weight of Polymer
4.2. Type and Concentration of Alcohol
4.3. Type and Concentration of Salt
4.4. pH System
4.5. Temperature
5. Future Prospect and Challenges of LBS Application
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Khoo, K.S.; Lee, S.Y.; Ooi, C.W.; Fu, X.; Miao, X.; Ling, T.C.; Show, P.L. Recent Advances in Biorefinery of Astaxanthin from Haematococcus pluvialis. Bioresour. Technol. 2019, 288, 121606. [Google Scholar] [CrossRef] [PubMed]
- Tham, P.E.; Ng, Y.J.; Sankaran, R.; Khoo, K.S.; Chew, K.W.; Yap, Y.J.; Malahubban, M.; Aziz Zakry, F.A.; Show, P.L. Recovery of Protein from Dairy Milk Waste Product Using Alcohol-Salt Liquid Biphasic Flotation. Processes 2019, 7, 875. [Google Scholar] [CrossRef]
- Azevedo, A.M.; Rosa, P.A.; Ferreira, I.F.; Aires-Barros, M.R. Chromatography-free recovery of biopharmaceuticals through aqueous two-phase processing. Trends Biotechnol. 2009, 27, 240–247. [Google Scholar] [CrossRef] [PubMed]
- Drexler, I.L.; Yeh, D.H. Membrane applications for microalgae cultivation and harvesting: A review. Rev. Environ. Sci. Bio 2014, 13, 487–504. [Google Scholar] [CrossRef]
- Grossmann, L.; Ebert, S.; Hinrichs, J.; Weiss, J. Effect of precipitation, lyophilization, and organic solvent extraction on preparation of protein-rich powders from the microalgae Chlorella protothecoides. Algal. Res. 2018, 29, 266–276. [Google Scholar] [CrossRef]
- Sankaran, R.; Manickam, S.; Yap, Y.J.; Ling, T.C.; Chang, J.-S.; Show, P.L. Extraction of proteins from microalgae using integrated method of sugaring-out assisted liquid biphasic flotation (LBF) and ultrasound. Ultrason. Sonochem. 2018, 48, 231–239. [Google Scholar] [CrossRef]
- Khoo, K.S.; Chew, K.W.; Ooi, C.W.; Ong, H.C.; Ling, T.C.; Show, P.L. Extraction of natural astaxanthin from Haematococcus pluvialis using liquid biphasic flotation system. Bioresour. Technol. 2019, 290, 121794. [Google Scholar] [CrossRef]
- Leong, H.Y.; Ooi, C.W.; Law, C.L.; Julkifle, A.L.; Ling, T.C.; Show, P.L. Application of liquid biphasic flotation for betacyanins extraction from peel and flesh of Hylocereus polyrhizus and antioxidant activity evaluation. Sep. Purif. Technol. 2018, 201, 156–166. [Google Scholar] [CrossRef]
- Phong, W.N.; Le, C.F.; Show, P.L.; Chang, J.S.; Ling, T.C. Extractive disruption process integration using ultrasonication and an aqueous two-phase system for protein recovery from Chlorella sorokiniana. Eng. Life Sci. 2017, 17, 357–369. [Google Scholar] [CrossRef]
- Phong, W.N.; Show, P.L.; Teh, W.H.; Teh, T.X.; Lim, H.M.Y.; binti Nazri, N.S.; Tan, C.H.; Chang, J.-S.; Ling, T.C. Proteins recovery from wet microalgae using liquid biphasic flotation (LBF). Bioresour. Technol. 2017, 244, 1329–1336. [Google Scholar] [CrossRef]
- Lee, S.Y.; Khoiroh, I.; Ling, T.C.; Show, P.L. Aqueous Two-Phase Flotation for the Recovery of Biomolecules. Sep. Purif. Rev. 2016, 45, 81–92. [Google Scholar] [CrossRef]
- Sankaran, R.; Show, P.L.; Cheng, Y.-S.; Tao, Y.; Ao, X.; Nguyen, T.D.P.; Van Quyen, D. Integration Process for Protein Extraction from Microalgae Using Liquid Biphasic Electric Flotation (LBEF) System. Mol. Biotechnol. 2018, 60, 749–761. [Google Scholar] [CrossRef] [PubMed]
- Koyande, A.K.; Chew, K.W.; Rambabu, K.; Tao, Y.; Chu, D.T.; Show, P.L. Microalgae: A potential alternative to health supplementation for humans. Food Science and Human Wellness. 2019, 8, 16–24. [Google Scholar] [CrossRef]
- Chew, K.W.; Chia, S.R.; Krishnamoorthy, R.; Tao, Y.; Chu, D.-T.; Show, P.L. Liquid biphasic flotation for the purification of C-phycocyanin from Spirulina platensis microalga. Bioresour. Technol. 2019, 288, 121519. [Google Scholar] [CrossRef] [PubMed]
- Van Berlo, M.; Luyben, K.C.A.; van der Wielen, L.A. Poly (ethylene glycol)–salt aqueous two-phase systems with easily recyclable volatile salts. J. Chromatogr. B Biomed. Sci. Appl. 1998, 711, 61–68. [Google Scholar] [CrossRef]
- Grilo, A.L.; Raquel Aires-Barros, M.; Azevedo, A.M. Partitioning in aqueous two-phase systems: Fundamentals, applications and trends. Sep. Purif. Rev. 2016, 45, 68–80. [Google Scholar] [CrossRef]
- Albertsson, P. Fractionation of particles and macromolecules in aqueous two-phase systems. Biochem. Pharmcol. 1961, 5, 351–358. [Google Scholar] [CrossRef]
- Albertsson, P.A. Partitioning of Cell Particles and Macromolecules; Wiley: New York, NY, USA, 1986. [Google Scholar]
- Asenjo, J.A.; Andrews, B.A. Aqueous two-phase systems for protein separation: Phase separation and applications. J. Chromatogr. A 2012, 1238, 1–10. [Google Scholar] [CrossRef]
- Ooi, C.W.; Tey, B.T.; Hii, S.L.; Kamal, S.M.M.; Lan, J.C.W.; Ariff, A.; Ling, T.C. Purification of lipase derived from Burkholderia pseudomallei with alcohol/salt-based aqueous two-phase systems. Process Biochem. 2009, 44, 1083–1087. [Google Scholar] [CrossRef]
- Zhao, L.; Peng, Y.-L.; Gao, J.-M.; Cai, W.-M. Bioprocess intensification: An aqueous two-phase process for the purification of C-phycocyanin from dry Spirulina platensis. Eur. Food Res. Technol. 2014, 238, 451–457. [Google Scholar] [CrossRef]
- Rosa, P.A.J.; Ferreira, I.F.; Azevedo, A.M.; Aires-Barros, M.R. Aqueous two-phase systems: A viable platform in the manufacturing of biopharmaceuticals. J. Chromatogr. A 2010, 1217. [Google Scholar] [CrossRef] [PubMed]
- Raja, S.; Murty, V.R.; Thivaharan, V.; Rajasekar, V.; Ramesh, V. Aqueous two phase systems for the recovery of biomolecules–a review. Sci. Technol. 2011, 1, 7–16. [Google Scholar] [CrossRef]
- Iqbal, M.; Tao, Y.; Xie, S.; Zhu, Y.; Chen, D.; Wang, X.; Huang, L.; Peng, D.; Sattar, A.; Shabbir, M.A.B. Aqueous two-phase system (ATPS): An overview and advances in its applications. Biol. Proced. Online 2016, 18, 18. [Google Scholar] [CrossRef] [PubMed]
- Hatti-Kaul, R. Aqueous Two-Phase Systems: Methods and Protocols; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2000; Volume 11. [Google Scholar]
- Raja, S.; Murty, V.R. Development and evaluation of environmentally benign aqueous two phase systems for the recovery of proteins from tannery waste water. ISRN Chem. Eng. 2012. [Google Scholar] [CrossRef]
- Johansson, H.-O.; Feitosa, E.; Junior, A.P. Phase diagrams of the aqueous two-phase systems of poly (ethylene glycol)/sodium polyacrylate/salts. Polymers 2011, 3, 587–601. [Google Scholar] [CrossRef]
- Hou, D.; Li, Y.; Cao, X. Synthesis of two thermo-sensitive copolymers forming aqueous two-phase systems. Sep. Purif. Technol. 2014, 122, 217–224. [Google Scholar] [CrossRef]
- Tan, Z.; Li, F.; Zhao, C.; Teng, Y.; Liu, Y. Chiral separation of mandelic acid enantiomers using an aqueous two-phase system based on a thermo-sensitive polymer and dextran. Sep. Purif. Technol. 2017, 172, 382–387. [Google Scholar] [CrossRef]
- Leong, Y.K.; Lan, J.C.W.; Loh, H.S.; Ling, T.C.; Ooi, C.W.; Show, P.L. Thermoseparating aqueous two-phase systems: Recent trends and mechanisms. J. Sep. Sci. 2016, 39, 640–647. [Google Scholar] [CrossRef]
- Show, P.L.; Tan, C.P.; Shamsul Anuar, M.; Ariff, A.; Yusof, Y.A.; Chen, S.K.; Ling, T.C. Extractive fermentation for improved production and recovery of lipase derived from Burkholderia cepacia using a thermoseparating polymer in aqueous two-phase systems. Bioresour. Technol. 2012, 116. [Google Scholar] [CrossRef]
- Ng, H.S.; Tan, C.P.; Mokhtar, M.N.; Ibrahim, S.; Ariff, A.; Ooi, C.W.; Ling, T.C. Recovery of Bacillus cereus cyclodextrin glycosyltransferase and recycling of phase components in an aqueous two-phase system using thermo-separating polymer. Sep. Purif. Technol. 2012, 89, 9–15. [Google Scholar] [CrossRef]
- Lin, Y.K.; Show, P.L.; Yap, Y.J.; Tan, C.P.; Ng, E.-P.; Ariff, A.B.; Annuar, M.S.B.M.; Ling, T.C. Direct recovery of cyclodextringlycosyltransferase from Bacillus cereus using aqueous two-phase flotation. J. Biosci. Bioeng. 2015, 120, 684–689. [Google Scholar] [CrossRef] [PubMed]
- Jong, W.Y.L.; Show, P.L.; Ling, T.C.; Tan, Y.S. Recovery of lignin peroxidase from submerged liquid fermentation of Amauroderma rugosum (Blume & T. Nees) Torrend using polyethylene glycol/salt aqueous two-phase system. J. Biosci. Bioeng. 2017, 124, 91–98. [Google Scholar] [PubMed]
- Lin, Y.K.; Ooi, C.W.; Tan, J.S.; Show, P.L.; Ariff, A.; Ling, T.C. Recovery of human interferon alpha-2b from recombinant Escherichia coli using alcohol/salt-based aqueous two-phase systems. Sep. Purif. Technol. 2013, 120, 362–366. [Google Scholar] [CrossRef]
- Chia, S.R.; Show, P.L.; Phang, S.-M.; Ling, T.C.; Ong, H.C. Sustainable approach in phlorotannin recovery from macroalgae. J. Biosci. Bioeng. 2018, 126, 220–225. [Google Scholar] [CrossRef]
- Smiglak, M.; Metlen, A.; Rogers, R.D. The Second Evolution of Ionic Liquids: From Solvents and Separations to Advanced Materials Energetic Examples from the Ionic Liquid Cookbook. Acc. Chem. Res. 2007, 40, 1182–1192. [Google Scholar] [CrossRef]
- Lee, S.Y.; Vicente, F.A.; e Silva, F.A.; Sintra, T.E.; Taha, M.; Khoiroh, I.; Coutinho, J.O.A.; Show, P.L.; Ventura, S.P. Evaluating self-buffering ionic liquids for biotechnological applications. ACS Sustain. Chem. Eng. 2015, 3, 3420–3428. [Google Scholar] [CrossRef]
- Zhang, S.; Sun, N.; He, X.; Lu, X.; Zhang, X. Physical properties of ionic liquids: Database and evaluation. J. Phys. Chem. Ref. Data 2006, 35, 1475–1517. [Google Scholar] [CrossRef]
- Lee, S.Y.; Khoiroh, I.; Ooi, C.W.; Ling, T.C.; Show, P.L. Recent advances in protein extraction using ionic liquid-based aqueous two-phase systems. Sep. Purif. Rev. 2017, 46, 291–304. [Google Scholar] [CrossRef]
- Ostadjoo, S.; Berton, P.; Shamshina, J.L.; Rogers, R.D. Scaling-up ionic liquid-based technologies: How much do we care about their toxicity? Prima facie information on 1-ethyl-3-methylimidazolium acetate. Toxicol. Sci. 2017, 161, 249–265. [Google Scholar] [CrossRef]
- Sun, N.; Rahman, M.; Qin, Y.; Maxim, M.L.; Rodríguez, H.; Rogers, R.D. Complete dissolution and partial delignification of wood in the ionic liquid 1-ethyl-3-methylimidazolium acetate. Green Chem. 2009, 11, 646–655. [Google Scholar] [CrossRef]
- Swatloski, R.P.; Spear, S.K.; Holbrey, J.D.; Rogers, R.D. Dissolution of cellose with ionic liquids. J. Am. Chem. Soc. 2002, 124, 4974–4975. [Google Scholar] [CrossRef] [PubMed]
- Gutowski, K.E.; Broker, G.A.; Willauer, H.D.; Huddleston, J.G.; Swatloski, R.P.; Holbrey, J.D.; Rogers, R.D. Controlling the aqueous miscibility of ionic liquids: Aqueous biphasic systems of water-miscible ionic liquids and water-structuring salts for recycle, metathesis, and separations. J. Am. Chem. Soc. 2003, 125, 6632–6633. [Google Scholar] [CrossRef] [PubMed]
- Du, Z.; Yu, Y.L.; Wang, J.H. Extraction of proteins from biological fluids by use of an ionic liquid/aqueous two-phase system. Chem. A Eur. J. 2007, 13, 2130–2137. [Google Scholar] [CrossRef] [PubMed]
- Ng, H.S.; Ooi, C.W.; Show, P.L.; Tan, C.P.; Ariff, A.; Moktar, M.N.; Ng, E.-P.; Ling, T.C. Recovery of Bacillus cereus cyclodextrin glycosyltransferase using ionic liquid-based aqueous two-phase system. Sep. Purif. Technol. 2014, 138, 28–33. [Google Scholar] [CrossRef]
- Chang, Y.-K.; Show, P.-L.; Lan, J.C.-W.; Tsai, J.-C.; Huang, C.-R. Isolation of C-phycocyanin from Spirulina platensis microalga using Ionic liquid based aqueous two-phase system. Bioresour. Technol. 2018, 270, 320–327. [Google Scholar] [CrossRef]
- Garcia, E.S.; Ruiz, C.A.S.; Tilaye, T.; Eppink, M.H.; Wijffels, R.H.; van den Berg, C. Fractionation of proteins and carbohydrates from crude microalgae extracts using an ionic liquid based-aqueous two phase system. Sep. Purif. Technol. 2018, 204, 56–65. [Google Scholar] [CrossRef]
- Abbott, A.P.; Boothby, D.; Capper, G.; Davies, D.L.; Rasheed, R.K. Deep eutectic solvents formed between choline chloride and carboxylic acids: Versatile alternatives to ionic liquids. J. Am. Chem. Soc. 2004, 126, 9142–9147. [Google Scholar] [CrossRef]
- Shishov, A.; Bulatov, A.; Locatelli, M.; Carradori, S.; Andruch, V. Application of deep eutectic solvents in analytical chemistry. A review. Microchem. J. 2017, 135, 33–38. [Google Scholar] [CrossRef]
- Paiva, A.; Craveiro, R.; Aroso, I.; Martins, M.; Reis, R.L.; Duarte, A.R.C. Natural deep eutectic solvents–solvents for the 21st century. ACS Sustain. Chem. Eng. 2014, 2, 1063–1071. [Google Scholar] [CrossRef]
- Dai, Y.; van Spronsen, J.; Witkamp, G.-J.; Verpoorte, R.; Choi, Y.H. Natural deep eutectic solvents as new potential media for green technology. Anal. Chim. Acta 2013, 766, 61–68. [Google Scholar] [CrossRef]
- Pang, J.; Sha, X.; Chao, Y.; Chen, G.; Han, C.; Zhu, W.; Li, H.; Zhang, Q. Green aqueous biphasic systems containing deep eutectic solvents and sodium salts for the extraction of protein. RSC Adv. 2017, 7, 49361–49367. [Google Scholar] [CrossRef]
- Zeng, Q.; Wang, Y.; Huang, Y.; Ding, X.; Chen, J.; Xu, K. Deep eutectic solvents as novel extraction media for protein partitioning. Analyst 2014, 139, 2565–2573. [Google Scholar] [CrossRef]
- Zhang, X.; Teng, G.; Zhang, J. Deep eutectic solvents aqueous two-phase system based ultrasonically assisted extraction of ursolic acid (UA) from Cynomorium songaricum Rupr. Chem. Eng. Commun. 2019, 206, 419–431. [Google Scholar] [CrossRef]
- Weschayanwiwat, P.; Kunanupap, O.; Scamehorn, J.F. Benzene removal from waste water using aqueous surfactant two-phase extraction with cationic and anionic surfactant mixtures. Chemosphere 2008, 72, 1043–1048. [Google Scholar] [CrossRef] [PubMed]
- Selber, K.; Tjerneld, F.; Collén, A.; Hyytiä, T.; Nakari-Setälä, T.; Bailey, M.; Fagerström, R.; Kan, J.; Van Der Laan, J.; Penttilä, M. Large-scale separation and production of engineered proteins, designed for facilitated recovery in detergent-based aqueous two-phase extraction systems. Process Biochem. 2004, 39, 889–896. [Google Scholar] [CrossRef]
- Amid, M.; Manap, M.; Hussin, M.; Mustafa, S. A novel aqueous two phase system composed of surfactant and xylitol for the purification of lipase from pumpkin (Cucurbita moschata) seeds and recycling of phase components. Molecules 2015, 20, 11184–11201. [Google Scholar] [CrossRef] [PubMed]
- Sankaran, R.; Show, P.L.; Yap, Y.J.; Tao, Y.; Ling, T.C.; Tomohisa, K. Green technology of liquid biphasic flotation for enzyme recovery utilizing recycling surfactant and sorbitol. Clean Technol. Environ. Policy 2018, 20, 2001–2012. [Google Scholar] [CrossRef]
- Sebba, F. Ion Flotation; Elsevier: Amsterdam, The Netherlands, 1962; Volume 30. [Google Scholar]
- Sankaran, R.; Show, P.L.; Lee, S.Y.; Yap, Y.J.; Ling, T.C. Integration process of fermentation and liquid biphasic flotation for lipase separation from Burkholderia cepacia. Bioresour. Technol. 2018, 250, 306–316. [Google Scholar] [CrossRef]
- Mathiazakan, P.; Shing, S.Y.; Ying, S.S.; Kek, H.K.; Tang, M.S.; Show, P.L.; Ooi, C.-W.; Ling, T.C. Pilot-scale aqueous two-phase floatation for direct recovery of lipase derived from Burkholderia cepacia strain ST8. Sep. Purif. Technol. 2016, 171, 206–213. [Google Scholar] [CrossRef]
- Bi, P.-y.; Dong, H.-r.; Yuan, Y.-c. Application of aqueous two-phase flotation in the separation and concentration of puerarin from Puerariae extract. Sep. Purif. Technol. 2010, 75, 402–406. [Google Scholar] [CrossRef]
- Jiang, B.; Wang, L.; Na, J.; Zhang, X.; Yuan, Y.; Liu, C.; Feng, Z. Environmentally-friendly strategy for separation of α-lactalbumin from whey by aqueous two phase flotation. Arab. J. Chem. 2018, 13, 3391–3402. [Google Scholar] [CrossRef]
- Wu, X.; Joyce, E.M.; Mason, T.J. The effects of ultrasound on cyanobacteria. Harmful Algae 2011, 10, 738–743. [Google Scholar] [CrossRef]
- Wang, M.; Yuan, W.; Jiang, X.; Jing, Y.; Wang, Z. Disruption of microalgal cells using high-frequency focused ultrasound. Bioresour. Technol. 2014, 153, 315–321. [Google Scholar] [CrossRef]
- Gerde, J.A.; Montalbo-Lomboy, M.; Yao, L.; Grewell, D.; Wang, T. Evaluation of microalgae cell disruption by ultrasonic treatment. Bioresour. Technol. 2012, 125, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Chemat, F.; Rombaut, N.; Sicaire, A.; Meullemiestre, A.; Abert-vian, M.; Fabiano-Tixier, A.; Abert-vian, M. Ultrasonics Sonochemistry Ultrasound assisted extraction of food and natural products. Mechanisms, techniques, combinations, protocols and applications. A review. Ultrason. Sonochem. 2017, 34, 540–560. [Google Scholar]
- Pakhale, S.V.; Vetal, M.D.; Rathod, V.K. Separation of bromelain by aqueous two phase flotation. Sep. Sci. Technol. 2013, 48, 984–989. [Google Scholar] [CrossRef]
- Rahim, A.H.A.; Khoo, K.S.; Yunus, N.M.; Hamzah, W.S.W. Ether-Functionalized Ionic Liquids as Solvent for Gigantochloa Scortechini Dissolution. Proceedings of AIP Conference Proceedings, Cesme-Izmir, Turkey, 26–30 May 2019; p. 020025. [Google Scholar]
- Dong, B.; Yuan, X.; Zhao, Q.; Feng, Q.; Liu, B.; Guo, Y.; Zhao, B. Ultrasound-assisted aqueous two-phase extraction of phenylethanoid glycosides from Cistanche deserticola YC Ma stems. J. Sep. Sci. 2015, 38, 1194–1203. [Google Scholar] [CrossRef] [PubMed]
- Đorđević, T.; Antov, M. Ultrasound assisted extraction in aqueous two-phase system for the integrated extraction and separation of antioxidants from wheat chaff. Sep. Purif. Technol. 2017, 182, 52–58. [Google Scholar] [CrossRef]
- Eing, C.J.; Bonnet, S.; Pacher, M.; Puchta, H.; Frey, W. Effects of nanosecond pulsed electric field exposure on Arabidopsis thaliana. IEEE Trans. Dielectr. Electr. Insul. 2009, 16, 1322–1328. [Google Scholar] [CrossRef]
- Kotnik, T.; Frey, W.; Sack, M.; Meglič, S.H.; Peterka, M.; Miklavčič, D. Electroporation-based applications in biotechnology. Trends Biotechnol. 2015, 33, 480–488. [Google Scholar] [CrossRef]
- Luengo, E.; Condón-Abanto, S.; Álvarez, I.; Raso, J. Effect of pulsed electric field treatments on permeabilization and extraction of pigments from Chlorella vulgaris. J. Membr. Biol. 2014, 247, 1269–1277. [Google Scholar] [CrossRef]
- Leong, H.Y.; Ooi, C.W.; Law, C.L.; Julkifle, A.L.; Katsuda, T.; Show, P.L. Integration process for betacyanins extraction from peel and flesh of Hylocereus polyrhizus using liquid biphasic electric flotation system and antioxidant activity evaluation. Sep. Purif. Technol. 2019, 209, 193–201. [Google Scholar] [CrossRef]
- ‘t Lam, G.P.; van der Kolk, J.A.; Chordia, A.; Vermuë, M.H.; Olivieri, G.; Eppink, M.H.; Wijffels, R.H. Mild and selective protein release of cell wall deficient microalgae with pulsed electric field. ACS Sustain. Chem. Eng. 2017, 5, 6046–6053. [Google Scholar] [CrossRef]
- Phong, W.N.; Show, P.L.; Chow, Y.H.; Ling, T.C. Recovery of biotechnological products using aqueous two phase systems. J. Biosci. Bioeng. 2018, 126, 273–281. [Google Scholar] [CrossRef] [PubMed]
- Santos, S.B.; Reis, I.A.; Silva, C.P.; Campos, A.F.; Ventura, S.P.; Soares, C.M.; Lima, Á.S. Selective partition of caffeine from coffee bean and guaraná seed extracts using alcohol–salt aqueous two-phase systems. Sep. Sci. Technol. 2016, 51, 2008–2019. [Google Scholar] [CrossRef]
- Huang, L.; Li, W.; Feng, Y.; Fang, X.; Li, J.; Gao, Z.; Li, H. Simultaneous recovery of glycyrrhizic acid and liquiritin from Chinese licorice root (Glycyrrhiza uralensis Fisch) by aqueous two-phase system and evaluation biological activities of extracts. Sep. Sci. Technol. 2018, 53, 1342–1350. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, Y.; Han, J.; Hu, S. Application of water-miscible alcohol-based aqueous two-phase systems for extraction of dyes. Sep. Sci. Technol. 2011, 46, 1283–1288. [Google Scholar] [CrossRef]
- Wingfield, P. Protein precipitation using ammonium sulfate. Curr. Protoc. Protein Sci. 1998, 13, A. 3F. 1–A. 3F. 8. [Google Scholar]
- Lu, Y.; Yu, M.; Tan, Z.; Yan, Y. Phase equilibria and salt effect on the aqueous two-phase system of polyoxyethylene cetyl ether and sulfate salt at three temperatures. J. Chem. Eng. Data 2016, 61, 2135–2143. [Google Scholar] [CrossRef]
- Yang, L.; Huo, D.; Hou, C.; He, K.; Lv, F.; Fa, H.; Luo, X. Purification of plant-esterase in PEG1000/NaH2PO4 aqueous two-phase system by a two-step extraction. Process Biochem. 2010, 45, 1664–1671. [Google Scholar] [CrossRef]
- Goja, A.M.; Yang, H.; Cui, M.; Li, C. Aqueous two-phase extraction advances for bioseparation. J. Bioprocess. Biotechnol. 2013, 4, 1–8. [Google Scholar]
- Karr, L.J.; Shafer, S.G.; Harris, J.M.; Van Alstine, J.M.; Snyder, R.S. Immuno-affinity partition of cells in aqueous polymer two-phase systems. J. Chromatogr. A 1986, 354, 269–282. [Google Scholar] [CrossRef]
- Andrews, B.; Schmidt, A.; Asenjo, J. Correlation for the partition behavior of proteins in aqueous two-phase systems: Effect of surface hydrophobicity and charge. Biotechnol. Bioeng. 2005, 90, 380–390. [Google Scholar] [CrossRef] [PubMed]
- Olivera-Nappa, A.; Lagomarsino, G.; Andrews, B.A.; Asenjo, J.A. Effect of electrostatic energy on partitioning of proteins in aqueous two-phase systems. J. Chromatogr. B 2004, 807, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, A.; Sen, K. Impact of pH and temperature on phase diagrams of different aqueous biphasic systems. J. Chromatogr. A 2016, 1433, 41–55. [Google Scholar] [CrossRef] [PubMed]
- Chow, Y.H.; Yap, Y.J.; Tan, C.P.; Anuar, M.S.; Tejo, B.A.; Show, P.L.; Ariff, A.B.; Ng, E.-P.; Ling, T.C. Characterization of bovine serum albumin partitioning behaviors in polymer-salt aqueous two-phase systems. J. Biosci. Bioeng. 2015, 120, 85–90. [Google Scholar] [CrossRef] [PubMed]
- Torres-Acosta, M.A.; Mayolo-Deloisa, K.; González-Valdez, J.; Rito-Palomares, M. Aqueous Two-Phase Systems at Large Scale: Challenges and Opportunities. Biotechnol. J. 2019, 14, 1800117. [Google Scholar] [CrossRef] [PubMed]
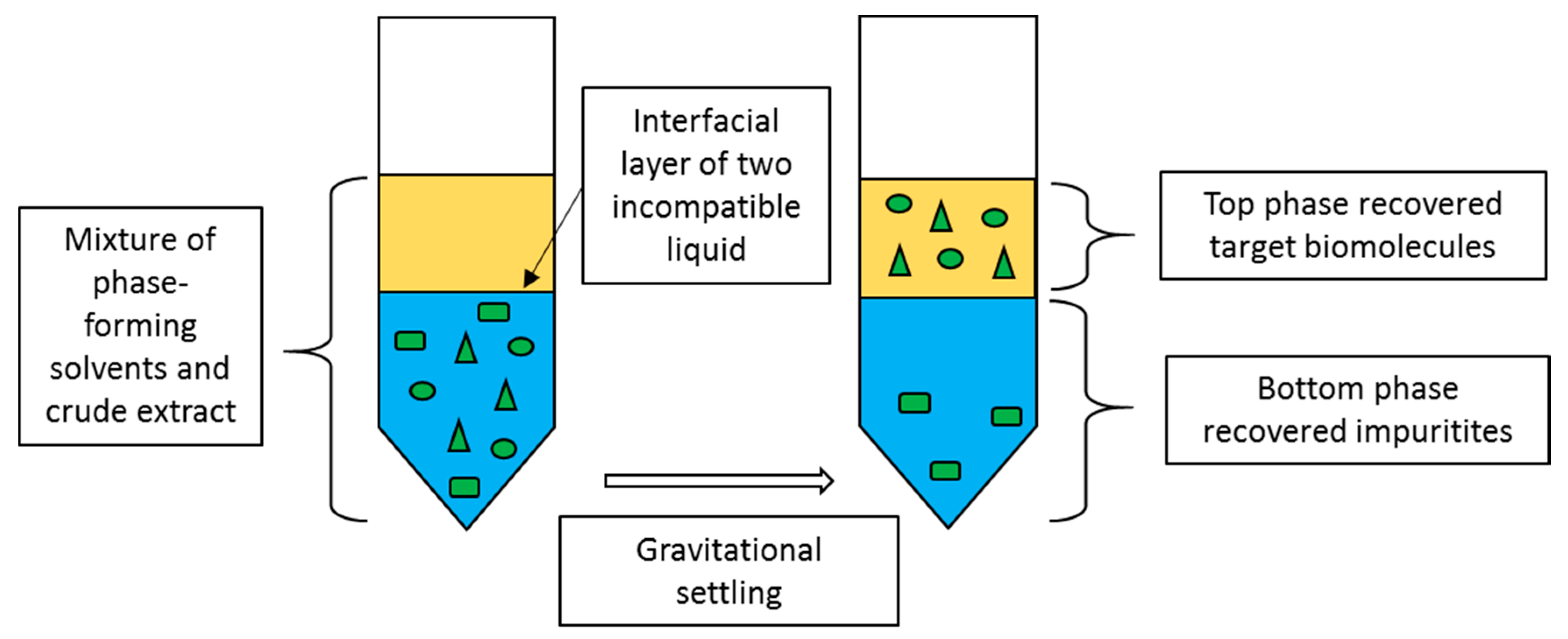
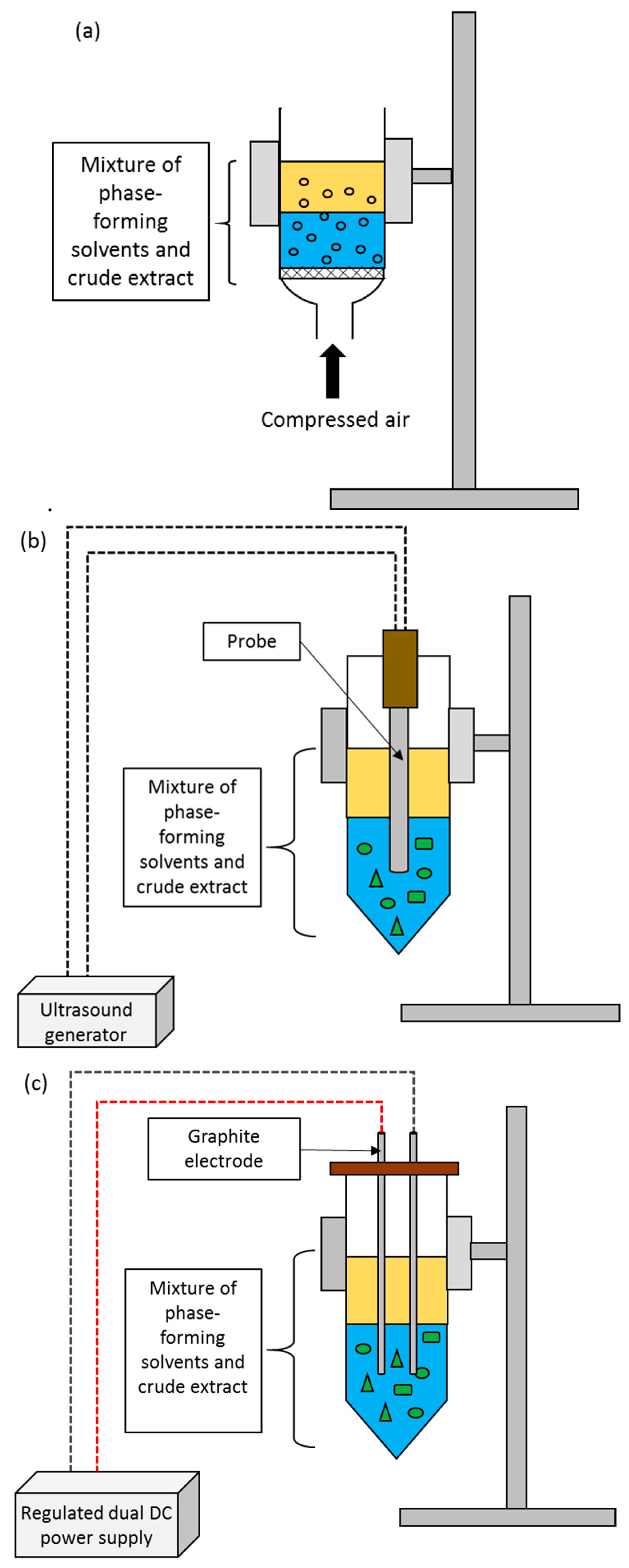
| Type of LBS | Composition of LBS | Type of Feedstock | Biomolecule | Selectivity | Partition Coefficient, K | Purification Factor, PFT | Recovery Yield (%) | Ref. |
|---|---|---|---|---|---|---|---|---|
| Polymer/salt-based | EOPO 3900 and two phosphate salts | Bacillus cereus cyclodextrin glycosyltransferase | Cyclodextringlycosyltransferase (CGTase) | 3.19 | 17.54 | 5.30 | 87.0 | [32] |
| 18% (w/w) PEG 8000 and 7.0% (w/w) potassium phosphate salts | Bacillus cereus cyclodextrin glycosyltransferase | Cyclodextringlycosyltransferase (CGTase) | - | - | 21.8 | 97.1 | [33] | |
| 15% (w/w) PEG 600 and 16% (w/w) dipotassium phosphate | Amauroderma rugosum | Lignin peroxidase | - | - | 1.33 ± 0.62 | 2.18 ± 8.50 | [34] | |
| Alcohol/salt-based | 18% (w/w) 2-propanol and 22% (w/w) ammonium sulfate, (NH4)2SO4 | Escherichia coli | Interferon (IFN)/ Glycoproteins | - | 0.82 | 16.24 | 74.64 | [35] |
| 16% (w/w) 2-propanol and 16% (w/w) potassium phosphate | Burkholderia pseudomallei | Lipase | 287.5 | - | 13.5 | 99.3 | [20] | |
| 33.5% (w/w) of 2-propanol and 10% (w/w) ammonium sulfate | Padina australis | Phlorotannin | - | - | 2.49 | 76.1 | [36] | |
| 25% (w/w) of 2-propanol and 12.5% (w/w) ammonium sulfate | Sargassum binderi | Phlorotannin | - | - | 1.59 | 91.67 | [36] | |
| Ionic-liquid based | 35% (w/w) of (Emim)BF4 and 18% (w/w) of sodium carbonate Na2CO3 | Fermentation broth | Bacillus cereus cyclodextrin glycosyltransferase (CGTase) | 9.66 | - | 51.0 | 96.00 | [46] |
| C8MIM-Br and tri-potassium phosphate | Spirulina platensis | C-phycocyanin (CPC) | 5.8 | 36.6 | - | 99.00 | [47] | |
| Deep-eutectic solvent based | 0.7 g mL−1 ChCl-urea and 2.0 mL dipotassium phosphate, K2HPO4 | Protein | Bovine serum albumin (BSA) | - | - | - | 99.6 99.7 and 100.0 BSA | [54] |
| Choline chloride and PEG 2000, molar ratio of 20:1 | Protein | Bovine serum albumin and papain | - | - | - | Bovine serum albumin (95.16), papain (90.95) | [53] | |
| 36% (w/w) ChCl-glucose and 25% (w/w) dipotassium phosphate, K2HPO4 | Ursolic acid | Cynomorium songaricum Rupr. | - | - | 42.41 ± 0.84 | 22.10 ± 0.44 mg/g | [55] | |
| Surfactant/detergent based | 24% (w/w) Triton X-100 and 20% (w/w) xylitol | Cucurbita moschata | Lipase | - | - | 16.4 | 97.0 | [58] |
| 25% (w/w) of xylitol concentration, 15% (w/w) Triton X-100 | Burkholderia cepacia | Lipase | 2.62 | - | 2.56 | 86.46 | [59] |
| Assisted Technology | Composition of LBS | Type of Assisted Employed | Type of Feedstock | Biomolecule | Time | Extraction Efficiency, E (%) | Partition Coefficient, K | Recovery Yield (%) | Ref |
|---|---|---|---|---|---|---|---|---|---|
| Bubble-assisted LBS or Liquid biphasic flotation (LBF) | 50% (w/w) of 1-propanol and 250 g/L ammonium sulfate salt, (NH4)2SO4 | Flotation system (compressed air 0.5 bar) | Burkholderia cepacian | Lipase | 30 min | 88.0 | - | 93.27 | [62] |
| 100% ethanol, 20 mL of 200 g/L dipotassium phosphate K2HPO4 | Flotation system (compressed air 0.5 bar) | Hylocereous polyrhizus | Lipase | 15 min | E for peel and flesh were 88.361 ± 1.708%, 94.886 ± 0.060%. | K value of peel and flesh were 24.168 ± 2.949, 21.195 ± 1.030. | Recovery for peel and flesh were 95.488 ± 0.213, 94.886 ± 0.060. | [8] | |
| 0.5 g/mL PEG 1000, 35 mL of 0.40 g/mL trisodium citrate Na3C6H5O7 | Flotation system (30 mL/min flow velocity) | Whey | a-lactalbumin | 42 min | 87.54 | - | - | [64] | |
| Ultrasound-assisted LBS | 100% (w/w) acetonitrile and 200 g/L glucose solution. | Ultrasound irradiated for 5 min of 5 s ON/10 s OFF pulse mode and flotation system | Chlorella vulgaris FSP-E | Protein | 5 min | 86.38 | - | 93.33 of protein recovered | [6] |
| 20% (w/w) ethanol and 23.5% ammonium sulfate | Ultrasound irradiated (300 W, 37 min) | Cistanche deserticola Y. C. Ma stems | Phenylethanoid glycosides | 37 min | Echinacoside and acteoside were 5.35 and 6.22 mg/g dry weight | - | Echinacoside and acteoside were 27.56 and 30.23 mg/g dry weight | [71] | |
| 24.3% (w/w) ethanol and 23.8% (w/w) ammonium sulfate | Ultrasound irradiated (30 Hz, 500 W, 10 min), | Wheat chaff | Xylooligosaccharides (sugar) and phenolic compound | 10 min | 72.79 ± 3.98 | 3.91 | Recovery of sugar and phenols were 16 mg/g and 2.67 mg/g | [72] | |
| Electricity-assisted LBS | Without LBS | PEF treatment (5–7.5 kV/cm, 1–10 pulses and a pulse length of 0.05–0.2 ms) | Cell wall C. reinhardtii strain (cc-124) and cell wall deficient mutant strain (cc-400) | Protein | 10 min/pulse | - | - | Cell wall strain (cc-124) and cell deficient (cc-400) with average protein yield of 31 ± 6 protein and 11 ± 3 protein. | [77] |
| 100% (w/w) ethanol, 200 g/L of dipotassium hydrogen phosphate (K2HPO4) | PEF treatment (3 V of voltage using graphitic electrodes) and 15 min flotation system | Peel and flesh of Hylocereus polyrhizus | Betacyanins | 15 min | E for peel and flesh were 98.383 ± 0.215 and 96.576 ± 0.083 | K for peel and flesh were 100.814 ± 7.324 and 24.883 ± 1.052 | Betacyanins concentration (98.383 ± 0.215 for peel and 96.576 ± 0.0083 for flesh | [76] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khoo, K.S.; Leong, H.Y.; Chew, K.W.; Lim, J.-W.; Ling, T.C.; Show, P.L.; Yen, H.-W. Liquid Biphasic System: A Recent Bioseparation Technology. Processes 2020, 8, 149. https://doi.org/10.3390/pr8020149
Khoo KS, Leong HY, Chew KW, Lim J-W, Ling TC, Show PL, Yen H-W. Liquid Biphasic System: A Recent Bioseparation Technology. Processes. 2020; 8(2):149. https://doi.org/10.3390/pr8020149
Chicago/Turabian StyleKhoo, Kuan Shiong, Hui Yi Leong, Kit Wayne Chew, Jun-Wei Lim, Tau Chuan Ling, Pau Loke Show, and Hong-Wei Yen. 2020. "Liquid Biphasic System: A Recent Bioseparation Technology" Processes 8, no. 2: 149. https://doi.org/10.3390/pr8020149
APA StyleKhoo, K. S., Leong, H. Y., Chew, K. W., Lim, J.-W., Ling, T. C., Show, P. L., & Yen, H.-W. (2020). Liquid Biphasic System: A Recent Bioseparation Technology. Processes, 8(2), 149. https://doi.org/10.3390/pr8020149









