Sustainable Green Procedure for Extraction of Hesperidin from Selected Croatian Mandarin Peels
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Plant Material
2.2. Preparation of Deep Eutectic Solvents (DESs)
2.3. Extraction of Hesperidin from Mandarin Peels with DESs
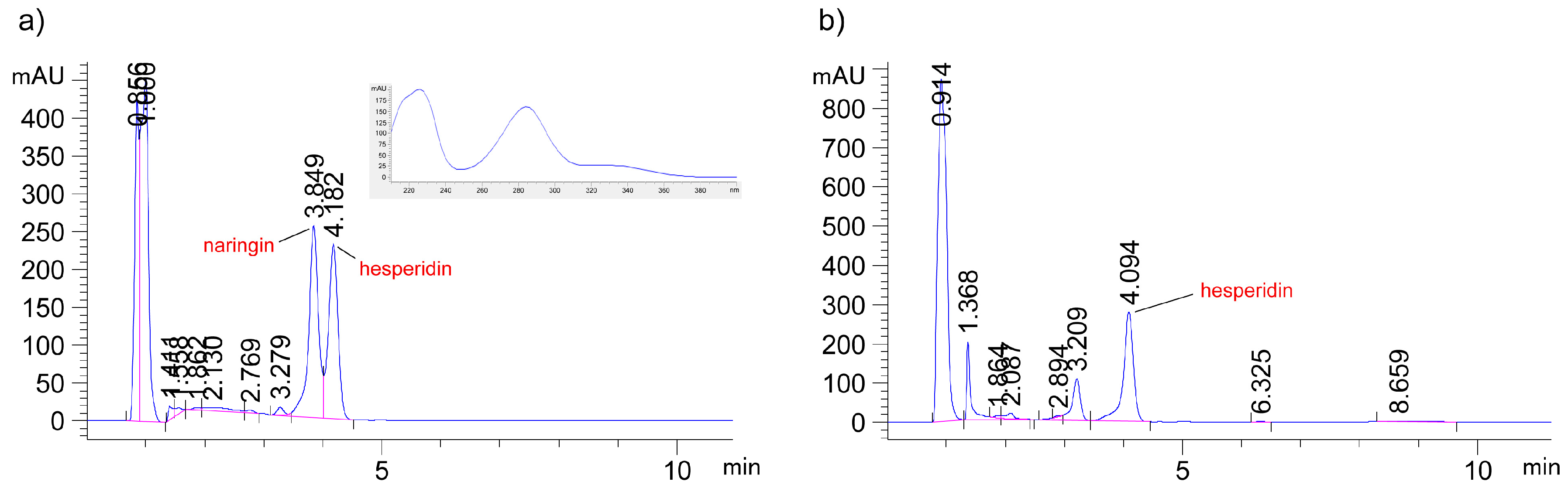
2.4. HPLC Analysis of Hesperidin in the Extracts
2.5. Experimental Design
2.6. Development of the RSM Model
3. Results and Discussion
3.1. Screening of DES for Hesperidin Extraction
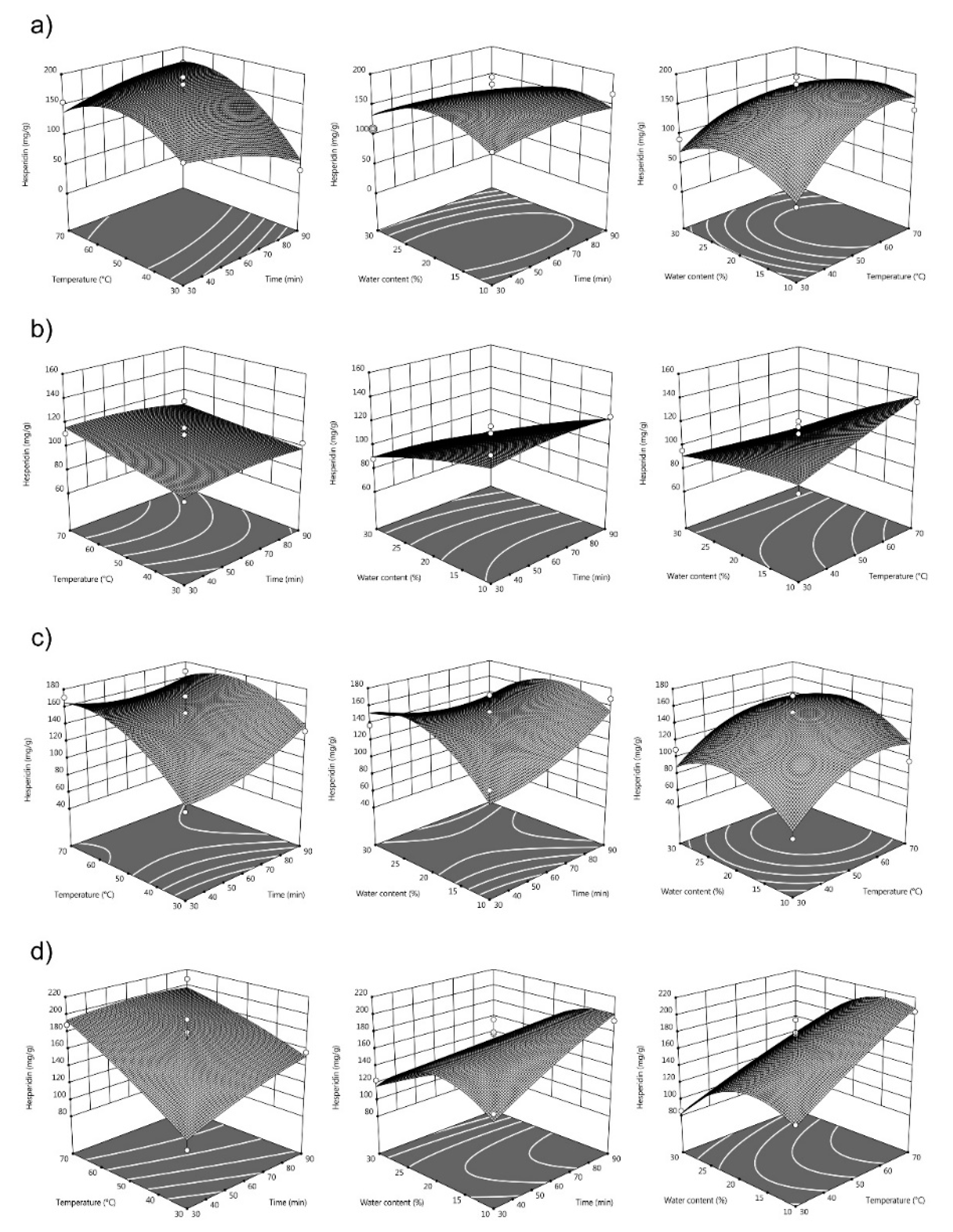
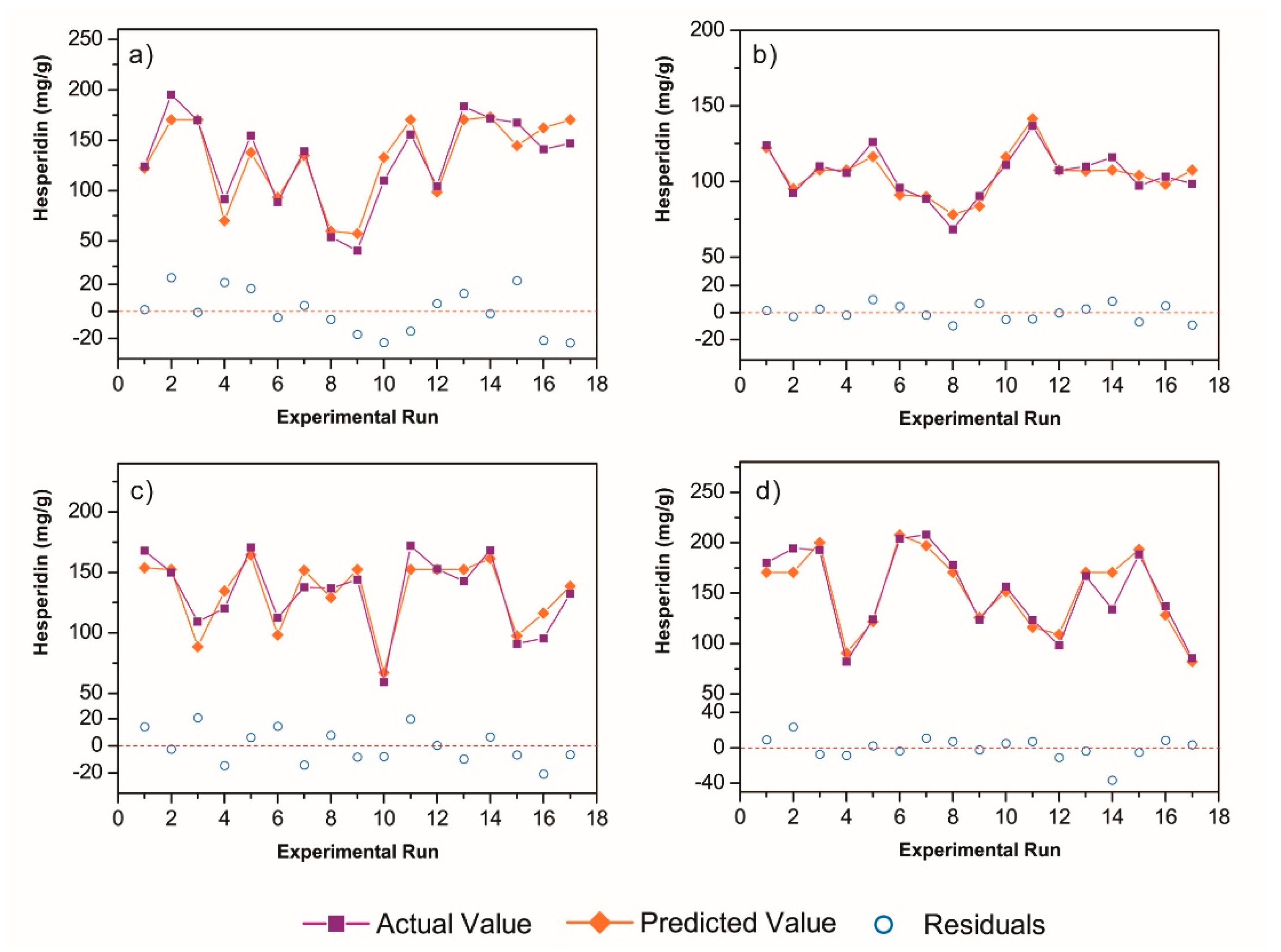
3.2. Response Surface Analysis and Process Optimization
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Moulehi, I.; Bourgou, S.; Ourghemmi, I.; Saidani Tounsi, M. Variety and ripening impact on phenolic composition and antioxidant activity of mandarin (Citrus reticulate Blanco) and bitter orange (Citrus aurantium L.) seeds extracts. Ind. Crops Prod. 2012, 39, 74–80. [Google Scholar] [CrossRef]
- Mamma, D.; Christakopoulos, P. Biotransformation of Citrus By-Products into Value Added Products. Waste Biomass Valorization 2014, 5, 529–549. [Google Scholar] [CrossRef]
- Lu, Y.; Zhang, C.; Bucheli, P.; Wei, D. Citrus Flavonoids in Fruit and Traditional Chinese Medicinal Food Ingredients in China. Plant Foods Hum. Nutr. 2006, 61, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.S.; Ho, S.C. Polymethoxy flavones are responsible for the anti-inflammatory activity of citrus fruit peel. Food Chem. 2010, 119, 868–873. [Google Scholar] [CrossRef]
- Tumbas, V.T.; Ćetković, G.S.; Djilas, S.M.; Čanadanović-Brunet, J.M.; Vulić, J.J.; Knez, Ž.; Škerget, M. Antioxidant activity of mandarin (Citrus reticulata) peel. Acta Period. Technol. 2010, 41, 195–203. [Google Scholar] [CrossRef]
- Jung, U.J.; Lee, M.K.; Jeong, K.S.; Choi, M.S. The Hypoglycemic Effects of Hesperidin and Naringin Are Partly Mediated by Hepatic Glucose-Regulating Enzymes in C57BL/KsJ-db/db Mice. J. Nutr. 2004, 134, 2499–2503. [Google Scholar] [CrossRef] [Green Version]
- Al-Ashaal, H.A.; El-Sheltawy, S.T. Antioxidant capacity of hesperidin from Citrus peel using electron spin resonance and cytotoxic activity against human carcinoma cell lines. Pharm. Biol. 2011, 49, 276–282. [Google Scholar] [CrossRef] [PubMed]
- Guardia, T.; Rotelli, A.E.; Juarez, A.O.; Pelzer, L.E. Anti-inflammatory properties of plant flavonoids. Effects of rutin, quercetin and hesperidin on adjuvant arthritis in rat. II Farm. 2001, 56, 683–687. [Google Scholar] [CrossRef]
- Manthey, J.A.; Guthrie, N. Antiproliferative Activities of Citrus Flavonoids against Six Human Cancer Cell Lines. J. Agric. Food Chem. 2002, 50, 5837–5843. [Google Scholar] [CrossRef]
- Brglez Mojzer, E.; Knez Hrnčič, M.; Škerget, M.; Knez, Ž.; Bren, U. Polyphenols: Extraction Methods, Antioxidative Action, Bioavailability and Anticarcinogenic Effects. Molecules 2016, 21, 901. [Google Scholar] [CrossRef]
- Garg, A.; Garg, S.; Zaneveld, L.J.D.; Singla, A.K. Chemistry and Pharmacology of the Citrus Bioflavonoid Hesperidin. Phytother. Res. 2011, 15, 655–669. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Ye, X.; Hao, Y.; Xu, G.; Xu, G.; Liu, D. Ultrasound-assisted extraction of hesperidin from Penggan (Citrus reticulata) peel. Ultrason. Sonochem. 2008, 15, 227–232. [Google Scholar] [CrossRef] [PubMed]
- Safdar, M.N.; Kausar, T.; Jabbar, S.; Mumtaz, A.; Ahad, K.; Saddozai, A.A. Extraction and quantification of polyphenols from kinnow (Citrus reticulate L.) peel using ultrasound and maceration techniques. J. Food Drug Anal. 2016, 25, 488–500. [Google Scholar] [CrossRef] [PubMed]
- Zainal-Abidin, M.H.; Hayyan, M.; Hayyan, A.; Jayakumar, N.S. New horizons in the extraction of bioactive compounds using deep eutectic solvents: A review. Anal. Chim. Acta 2017, 979, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.; van Spronsen, J.; Witkamp, G.J.; Verpoorte, R.; Choi, Y.H. Natural deep eutectic solvents as new potential media for green technology. Anal. Chim. Acta 2013, 766, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Bajkacz, S.; Adamek, J. Development of a Method Based on Natural Deep Eutectic Solvents for Extraction of Flavonoids from Food Samples. Food Anal. Methods 2018, 11, 1330–1344. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, H.; Yu, H.; Guo, S.; Chen, D. Deep eutectic solvent as a green solvent for enhanced extraction of narirutin, naringin, hesperidin and neohesperidin from Aurantii Fructus. Phytochem. Anal. 2018, 30, 156–163. [Google Scholar] [CrossRef]
- Van den Bruinhorst, A.; Kouris, P.; Timmer, J.M.K.; de Croon, M.H.J.M.; Kroon, M.C. Exploring orange peel treatment with deep eutectic solvents and diluted organic acids. Nat. Prod. Chem Res. 2016, 4, 1–5. [Google Scholar] [CrossRef]
- Lota, M.L.; de Rocca Serra, D.; Tomi, F.; Casanova, J. Chemical variability of peel and leaf essential oils of mandarins from Citrus reticulata Blanco. Biochem. Syst. Ecol. 2000, 28, 61–78. [Google Scholar] [CrossRef]
- Sun, Y.; Wang, J.; Gu, S.; Liu, Z.; Zhang, Y.; Zhang, X. Simultaneous Determination of Flavonoids in Different Parts of Citrus reticulata ‘Chachi’ Fruit by High Performance Liquid Chromatography—Photodiode Array Detection. Molecules 2010, 15, 5378–5388. [Google Scholar] [CrossRef]
- Budavari, S. (Ed.) The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals, 12th ed.; Merck & Co, Inc.: Whitehouse Station, NJ, USA, 1996. [Google Scholar]
- Bi, W.; Tian, M.; Row, K.H. Evaluation of alcohol based deep eutectic solvent in extraction and determination of flavonoids with response surface methodology optimization. J. Chromatogr. A 2013, 1285, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Majumdar, S.; Srirangam, R. Solubility, stability, physicochemical characteristics and in vitro ocular tissue permeability of hesperidin: A natural bioflavonoid. Pharm. Res. 2009, 26, 1217–1225. [Google Scholar] [CrossRef]
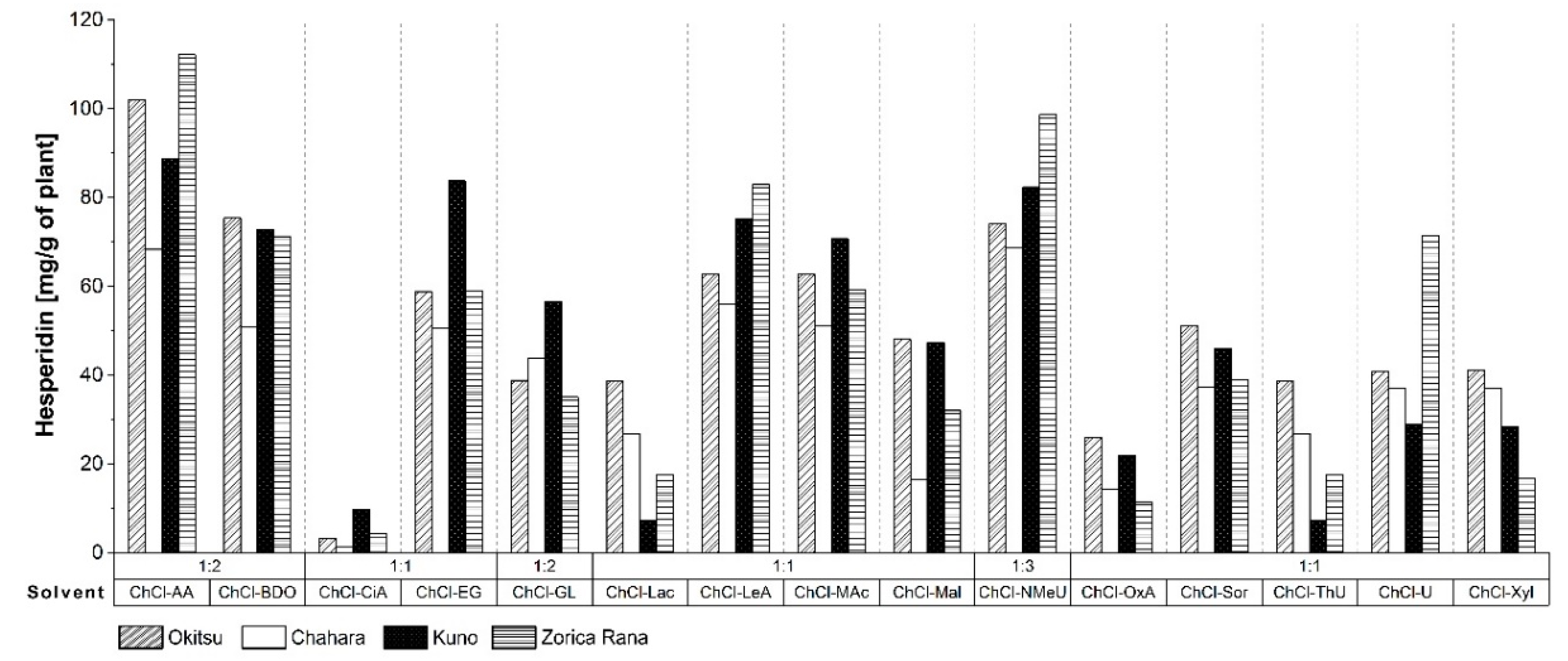
| Abbreviation | Components | Mole Ratio (HBA: HBD) | |
|---|---|---|---|
| Hydrogen Bond Acceptors (HBAs) | Hydrogen Bond Donors (HBDs) | ||
| ChCl-AA | Choline chloride | Acetamide | 1:2 |
| ChCl-BDO | Butane-1,4-diol | 1:2 | |
| ChCl-CiA | Citric acid | 1:1 | |
| ChCl-EG | Ethylene glycol | 1:1 | |
| ChCl-GL | Glycerol | 1:2 | |
| ChCl-Lac | Lactic acid | 1:1 | |
| ChCl-LeA | Levulinic acid | 1:1 | |
| ChCl-MAc | Malonic acid | 1:1 | |
| ChCl-Mal | Malic acid | 1:1 | |
| ChCl-NMeU | N-methyl urea | 1:3 | |
| ChCl-OxA | Oxalic acid | 1:1 | |
| ChCl-Sor | Sorbitol | 1:1 | |
| ChCl-ThU | Thiourea | 1:1 | |
| ChCl-U | Urea | 1:1 | |
| ChCl-Xyl | Xylitol | 1:1 | |
| Independent Variable | Symbol | Level | |||||
|---|---|---|---|---|---|---|---|
| Low (−1) | Middle (0) | High (+1) | |||||
| Time (min) | X1 | 30 | 60 | 90 | |||
| Temperature (°C) | X2 | 30 | 50 | 70 | |||
| Water content (%) | X3 | 10 | 20 | 30 | |||
| Okitsu | Chahara | Kuno | Zorica Rana | ||||
| Run | X1 | X2 | X3 | Hesperidin (mg/g of plant) | |||
| 1 | 90 | 30 | 20 | 40.45 | 103.17 | 132.35 | 156.47 |
| 2 | 30 | 30 | 20 | 124.01 | 92.35 | 91.03 | 98.12 |
| 3 | 30 | 50 | 30 | 110.03 | 88.36 | 137.66 | 123.48 |
| 4 | 90 | 50 | 30 | 88.89 | 68.41 | 120.14 | 81.99 |
| 5 | 60 | 50 | 20 | 195.32 | 110.15 | 189.84 | 179.96 |
| 6 | 60 | 50 | 20 | 169.81 | 105.70 | 144.08 | 194.50 |
| 7 | 60 | 70 | 30 | 104.39 | 90.40 | 136.88 | 123.59 |
| 8 | 60 | 50 | 20 | 155.82 | 107.45 | 172.13 | 177.85 |
| 9 | 60 | 50 | 20 | 183.55 | 116.02 | 152.80 | 167.06 |
| 10 | 60 | 30 | 10 | 53.83 | 97.27 | 59.33 | 124.05 |
| 11 | 30 | 50 | 10 | 139.29 | 126.11 | 112.67 | 136.91 |
| 12 | 60 | 70 | 10 | 140.94 | 136.86 | 95.45 | 204.05 |
| 13 | 60 | 30 | 30 | 91.46 | 95.76 | 109.34 | 85.46 |
| 14 | 60 | 50 | 20 | 147.15 | 98.43 | 142.74 | 134.05 |
| 15 | 90 | 70 | 20 | 171.56 | 109.91 | 168.11 | 207.88 |
| 16 | 90 | 50 | 10 | 167.48 | 124.03 | 167.89 | 192.86 |
| 17 | 30 | 70 | 20 | 154.67 | 111.06 | 170.67 | 188.36 |
| Source | Sum of Squares | Degree of Freedom | Mean Square | F Value | p-Value a |
|---|---|---|---|---|---|
| Okitsu | |||||
| Model | 27478.88 | 9 | 3053.21 | 5.11 | 0.0215 * |
| Residual | 4186.19 | 7 | 598.03 | ||
| Lack of fit | 2638.68 | 3 | 879.56 | 2.27 | 0.2221 |
| Pure error | 1547.52 | 4 | 386.88 | ||
| Total | 31665.07 | 16 | |||
| R2 = 0.8678 | |||||
| Chahara | |||||
| Model | 3659.79 | 9 | 406.64 | 5.10 | 0.0215 * |
| Residual | 557.94 | 7 | 79.71 | ||
| Lack of fit | 392.92 | 3 | 130.97 | 3.17 | 0.1468 |
| Pure error | 165.02 | 4 | 41.25 | ||
| Total | 4217.73 | 16 | |||
| R2 = 0.8677 | |||||
| Kuno | |||||
| Model | 13543.90 | 9 | 1504.88 | 4.17 | 0.0364 * |
| Residual | 2523.63 | 7 | 360.52 | ||
| Lack of fit | 1965.27 | 3 | 655.09 | 4.69 | 0.0847 |
| Pure error | 558.36 | 4 | 139.59 | ||
| Total | 16067.53 | 16 | |||
| R2 = 0.8429 | |||||
| Zorica rana | |||||
| Model | 24872.12 | 9 | 2763.57 | 7.34 | 0.0077 ** |
| Residual | 2635.01 | 7 | 376.43 | ||
| Lack of fit | 575.09 | 3 | 191.70 | 0.3722 | 0.7785 |
| Pure error | 2059.92 | 4 | 514.98 | ||
| Total | 27507.13 | 16 | |||
| R2 = 0.9042 |
| Source | Coefficients | Standard Error | F-Value | p-Value a |
|---|---|---|---|---|
| Okitsu | ||||
| Intercept | ||||
| β0 | 170.33 | 10.94 | ||
| Linear | ||||
| β1 | −7.45 | 8.65 | 0.74 | 0.4173 |
| β2 | 32.73 | 8.65 | 14.33 | 0.0068 ** |
| β3 | −13.35 | 8.65 | 2.38 | 0.1666 |
| Cross product | ||||
| β12 | 25.11 | 12.23 | 4.22 | 0.0791 |
| β13 | −12.33 | 12.23 | 1.02 | 0.3468 |
| β23 | −18.55 | 12.23 | 2.30 | 0.1731 |
| Quadratic | ||||
| β11 | −9.44 | 11.92 | 0.63 | 0.4541 |
| β22 | −38.21 | 11.92 | 10.28 | 0.0149 * |
| β33 | −34.46 | 11.92 | 8.36 | 0.0233 * |
| C.V. % | 18.57 | |||
| Chahara | ||||
| Intercept | ||||
| β0 | 107.55 | 3.99 | ||
| Linear | ||||
| β1 | −1.54 | 3.16 | 0.24 | 0.6396 |
| β2 | 7.46 | 3.16 | 5.58 | 0.0501 |
| β3 | −17.67 | 3.16 | 31.33 | 0.0008 ** |
| Cross product | ||||
| β12 | −2.99 | 4.46 | 0.45 | 0.5241 |
| β13 | −4.47 | 4.46 | 1.00 | 0.3505 |
| β23 | −11.24 | 4.46 | 6.34 | 0.0399 * |
| Quadratic | ||||
| β11 | −3.39 | 4.35 | 0.61 | 0.4619 |
| β22 | −0.0400 | 4.35 | 0.0001 | 0.9929 |
| β33 | −2.44 | 4.35 | 0.31 | 0.5929 |
| C.V. % | 8.52 | |||
| Kuno | ||||
| Intercept | ||||
| β0 | 152.32 | 8.49 | ||
| Linear | ||||
| β1 | 9.56 | 6.71 | 2.03 | 0.1976 |
| β2 | 22.38 | 6.71 | 11.12 | 0.0125 * |
| β3 | 8.58 | 6.71 | 1.64 | 0.2417 |
| Cross product | ||||
| β12 | −10.97 | 9.49 | 1.34 | 0.2858 |
| β13 | −18.18 | 9.49 | 3.67 | 0.0970 |
| β23 | −2.14 | 9.49 | 0.0509 | 0.8279 |
| Quadratic | ||||
| β11 | 11.28 | 9.25 | 1.49 | 0.2623 |
| β22 | −23.06 | 9.25 | 6.21 | 0.0415 * |
| β33 | −29.01 | 9.25 | 9.83 | 0.0165 * |
| C.V. % | 14.26 | |||
| Zorica rana | ||||
| Intercept | ||||
| β0 | 170.69 | 8.68 | ||
| Linear | ||||
| β1 | 11.54 | 6.86 | 2.83 | 0.1364 |
| β2 | 32.47 | 6.86 | 22.41 | 0.0021 ** |
| β3 | −30.42 | 6.86 | 19.66 | 0.0030 ** |
| Cross product | ||||
| β12 | −9.71 | 9.70 | 1.00 | 0.3503 |
| β13 | −24.36 | 9.70 | 6.31 | 0.0403 * |
| β23 | −10.47 | 9.70 | 1.16 | 0.3163 |
| Quadratic | ||||
| β11 | −4.23 | 9.46 | 0.2 | 0.6682 |
| β22 | −3.75 | 9.46 | 0.16 | 0.7034 |
| β33 | −32.65 | 9.46 | 11.92 | 0.0106 * |
| C.V. % | 12.80 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jokić, S.; Šafranko, S.; Jakovljević, M.; Cikoš, A.-M.; Kajić, N.; Kolarević, F.; Babić, J.; Molnar, M. Sustainable Green Procedure for Extraction of Hesperidin from Selected Croatian Mandarin Peels. Processes 2019, 7, 469. https://doi.org/10.3390/pr7070469
Jokić S, Šafranko S, Jakovljević M, Cikoš A-M, Kajić N, Kolarević F, Babić J, Molnar M. Sustainable Green Procedure for Extraction of Hesperidin from Selected Croatian Mandarin Peels. Processes. 2019; 7(7):469. https://doi.org/10.3390/pr7070469
Chicago/Turabian StyleJokić, Stela, Silvija Šafranko, Martina Jakovljević, Ana-Marija Cikoš, Nikolina Kajić, Filip Kolarević, Jurislav Babić, and Maja Molnar. 2019. "Sustainable Green Procedure for Extraction of Hesperidin from Selected Croatian Mandarin Peels" Processes 7, no. 7: 469. https://doi.org/10.3390/pr7070469