Optimization of Total Anthocyanin Content, Stability and Antioxidant Evaluation of the Anthocyanin Extract from Vietnamese Carissa Carandas L. Fruits
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and the Extraction Method
2.2. Determination of the Moisture Content of Fresh Fruits or Crude Extracts
2.3. Determination of Total Anthocyanin Content
2.4. Evaluation of Colorants
2.5. Determination of the Antioxidant Capacity
3. Results and Discussion
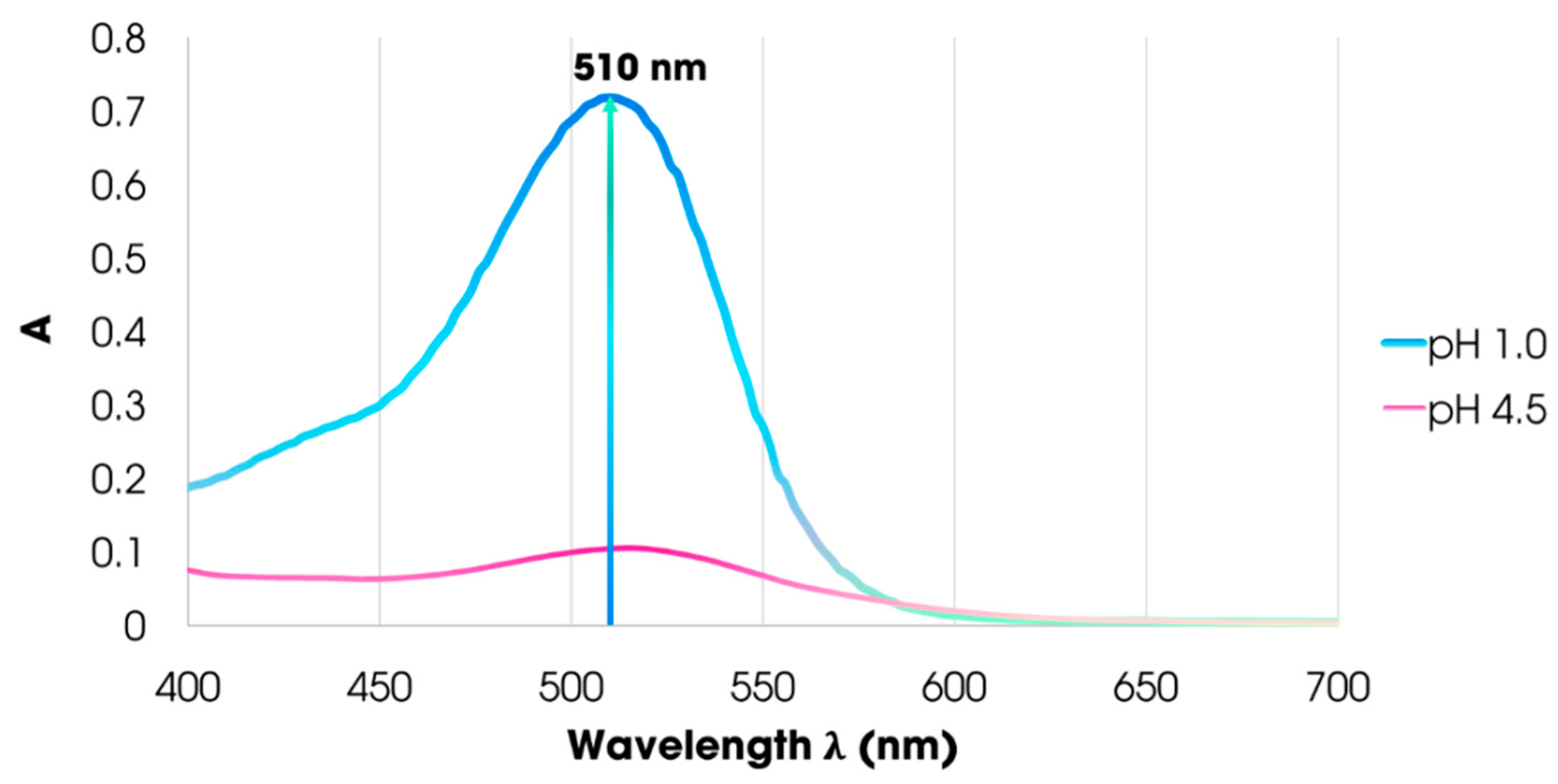
3.1. Maximum Absorption Wavelength of the Carissa Carandas Extract
3.2. Moisture of Carissa Carandas Fruit
3.3. The Effect of Extraction Conditions on Total Anthocyanin Content
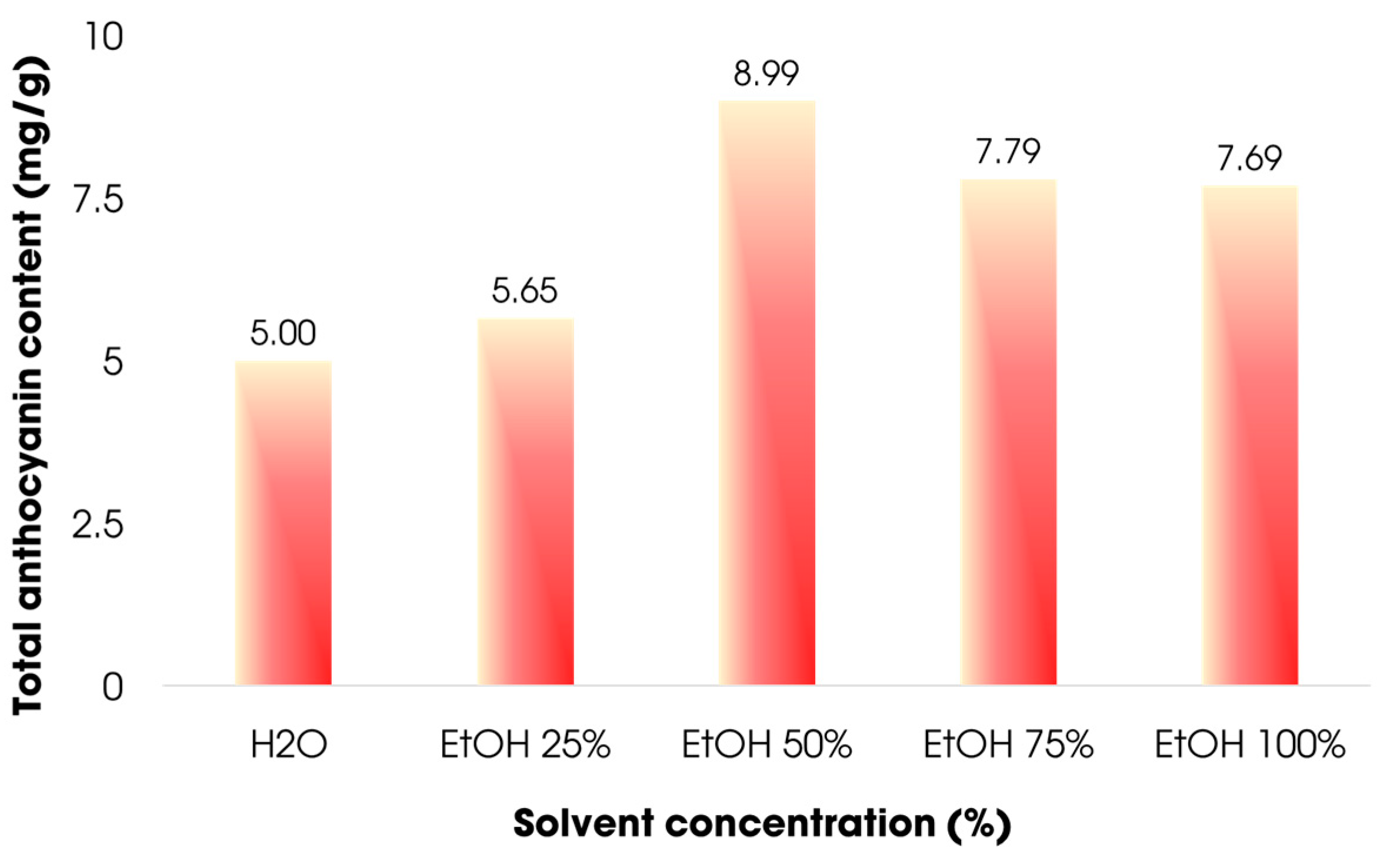
3.3.1. Effect of Solvent Concentration
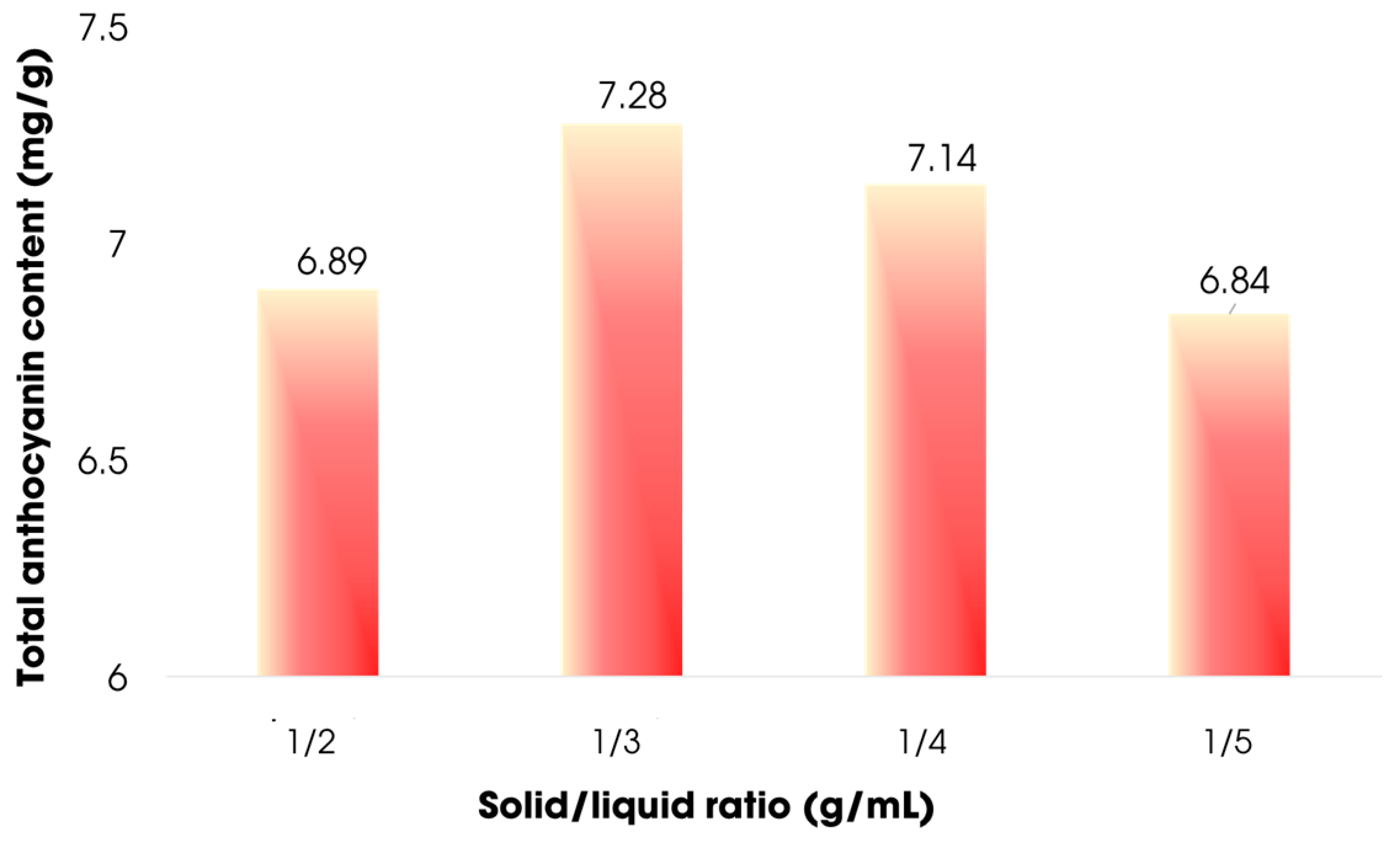
3.3.2. Effect of Solid:Liquid Ratio
3.3.3. Effect of Temperature of Extraction
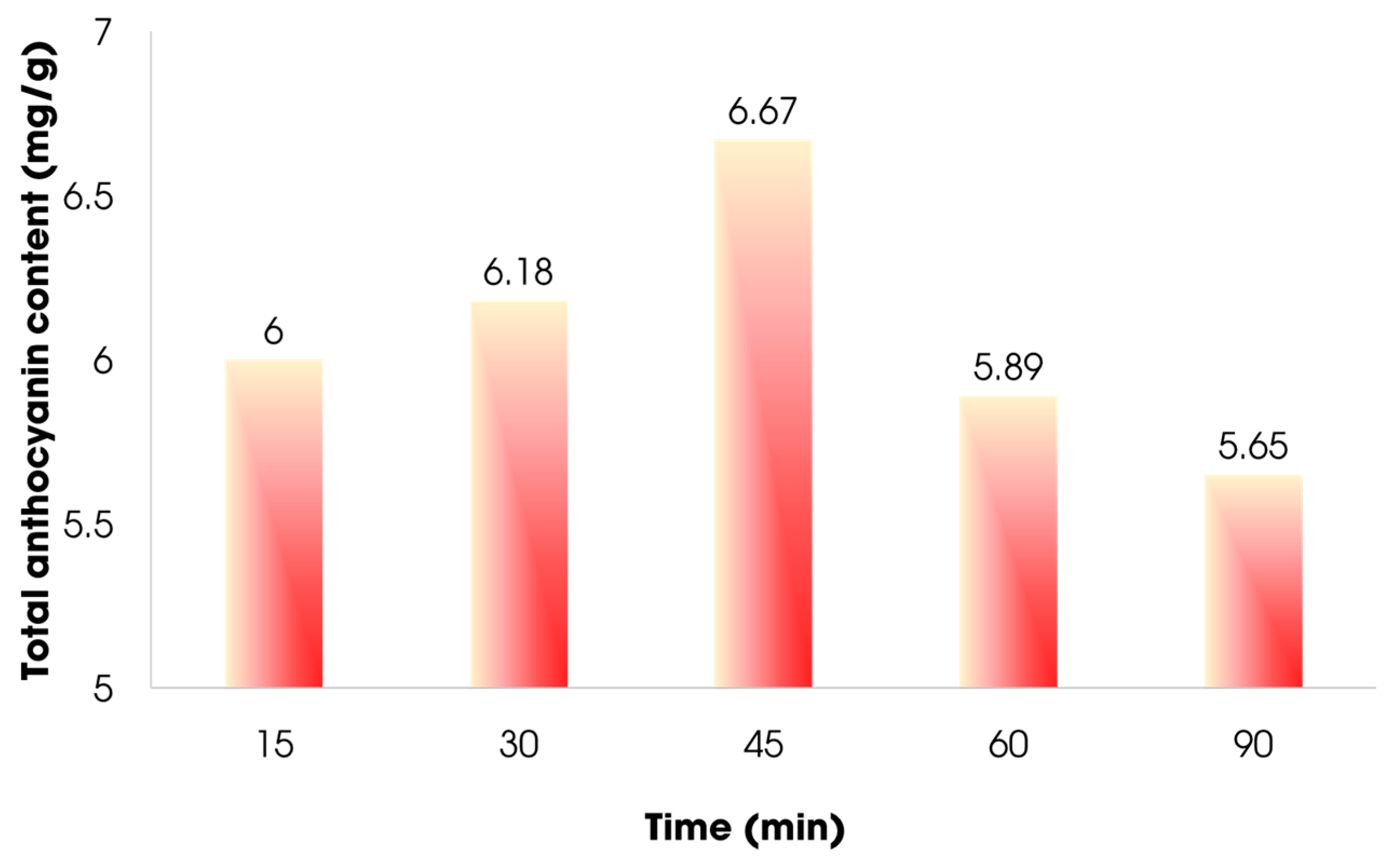
3.3.4. Effect of Time in Extraction Process
3.3.5. Effect of fruit ripeness
3.3.6. Effect of Extraction Cycle and Material Size
3.4. Stability Evaluation of Aqueous Solutions of Extract from Carissa Carandas Fruit
3.5. Stability and Antioxidant Evaluation of the Dried Extract from Carissa Carandas Fruit
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Francisco, D.V.; Octavio, P.L. Natural colorants for food and nutraceutical uses, 1st ed.; CRC Press: Boca Raton, FL, USA, 2002. [Google Scholar]
- Gould, K.; Davies, K.M.; Winefield, C. Anthocyanins Biosynthesis, Functions, and Applications; Springer: New York, NY, USA, 2009. [Google Scholar]
- Hubbermann, E.M.; Heins, A.; Stockmann, H.; Schwarz, K. Influence of acids, salt, sugars and hydrocolloids on the colour stability of anthocyanin rich black currant and elderberry concentrates. Eur. Food Res. Technol. 2006, 223, 83–90. [Google Scholar] [CrossRef]
- Singh, A.; Uppal, G.K. A review on Carissa carandas: Phytochemistry, ethno-pharmacology, and micropropagation as conservation strategy. Asian. J. Pharm. Clin. Res. 2015, 8, 26–30. [Google Scholar]
- Ozela, E.F.; Stringheta, P.C.; Chauca, M.C. Stability of anthocyanin in spinach vine (Basella rubra A.) fruits. Cien. Inv. Agr. 2007, 34, 115–120. [Google Scholar]
- Francis, F.J. A new group of food colorants. Trends. Food Sci. Technol. 1992, 3, 27–30. [Google Scholar] [CrossRef]
- Chai, X.F.; Ding, P. Postharvest Quality and Antioxidant Activity of Karanda (Carissa carandas) Fruit. Acta. Hortic. 2013, 1012, 177–182. [Google Scholar] [CrossRef]
- Weerawatanakorn, M.; Pan, M.H. Phytochemical components of Carissa carandas and the inhibitory effects of fruit juice on inducible nitric oxide synthase and cyclooxygenase-2. J. Food Biochem. 2017, 41. [Google Scholar] [CrossRef]
- Djilali, A.B.; Besombes, C.; Allaf, K.; Nabiev, M. Technological aptitudes and pharmaceutical applications of schinus terebinthifolius raddi leaf and seed oil and fruit powders of Ziziphus jujuba mill. Phytothérapie 2018. [Google Scholar] [CrossRef]
- Egbuna, C.; Ifemeje, J.C.; Maduako, M.C.; Tijjani, H.; Udedi, S.C.; Nwaka, A.C.; Ifemeje, M.O. Phytochemical test methods: Qualitative, quantitative and proximate analysis. In Phytochemistry: Volume 1: Fundamentals, Modern Techniques, and Applications, 1st ed.; Egbuna, C., Ifemeje, J.C., Udedi, S.C., Kumar, S., Eds.; Apple Academic Press: New York, NY, USA, 2018. [Google Scholar]
- Zou, T.B.; Wang, M.; Gan, R.Y.; Ling, W.H. Optimization of ultrasound-assisted extraction of anthocyanins from mulberry, using response surface methodology. Int. J. Mol. Sci. 2011, 12, 3006–3017. [Google Scholar] [CrossRef] [PubMed]
- Maran, J.P.; Sivakumar, V.; Thirugnanasambandham, K.; Sridhar, R. Extraction of natural anthocyanin and colors from pulp of jamun fruit. J. Food Sci. Technol. 2014, 52, 3617–3626. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yang, L.; Cao, Y.L.; Jiang, J.G.; Lin, Q.S.; Chen, J.; Zhu, L. Response surface optimization of ultrasound-assisted flavonoids extraction from the flower of Citrus aurantium L. var. amara Engl. J. Sep. Sci. 2010, 33, 1349–1355. [Google Scholar] [PubMed]
- Anuar, N.; Mohd, A.F.A.; Saat, N.; Aziz, N.; Mat, R.T. Optimization of extraction parameters by using response surface methodology, purification, and identification of anthocyanin pigments in melastoma malabathricum fruit. Sci. World J. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Barbour, M.E.; Parker, D.M.; Allen, G.C.; Jandt, K.D. Human enamel dissolution in citric acid as a function of pH in the range 2.30 ≤ pH ≤ 6.30—A nanoindentation study. Eur. J. Oral. Sci. 2003, 111, 258–262. [Google Scholar] [CrossRef] [PubMed]
- Braca, A.; Sortino, C.; Politi, M.; Morelli, I.; Mendez, J. Antioxidant activity of flavonoids from Licania licaniaeflora. J. Ethnopharmacol. 2002, 79, 379–381. [Google Scholar] [CrossRef]
- Wrolstad, R.E.; Durst, R.W.; Lee, J. Tracking color and pigment changes in anthocyanin products. Trends Food Sci. Technol. 2005, 16, 423–428. [Google Scholar] [CrossRef]
- Giusti, M.M.; Wrolstad, R.E. Characterization and measurement of anthocyanins by UV-visible spectroscopy. Curr. Protoc. Food Anal. Chem. 2001, 00, F1.2.1–F1.2.13. [Google Scholar] [CrossRef]
- Harborne, J.B. Spectral methods of characterizing anthocyanins. Biochem. J. 1958, 70, 22–28. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Liang, N.N.; Mu, L.; Pan, Q.H.; Wang, J.; Reeves, M.J.; Duan, C.Q. Anthocyanins and their variation in red wines ii. anthocyanin derived pigments and their color evolution. Molecules 2012, 17, 1483–1519. [Google Scholar] [CrossRef] [PubMed]
- Danışman, G.; Arslan, E.; Toklucu, A.K. Kinetic analysis of anthocyanin degradation and polymeric colour formation in grape juice during heating. Czech. J. Food Sci. 2016, 33, 103–108. [Google Scholar] [CrossRef]

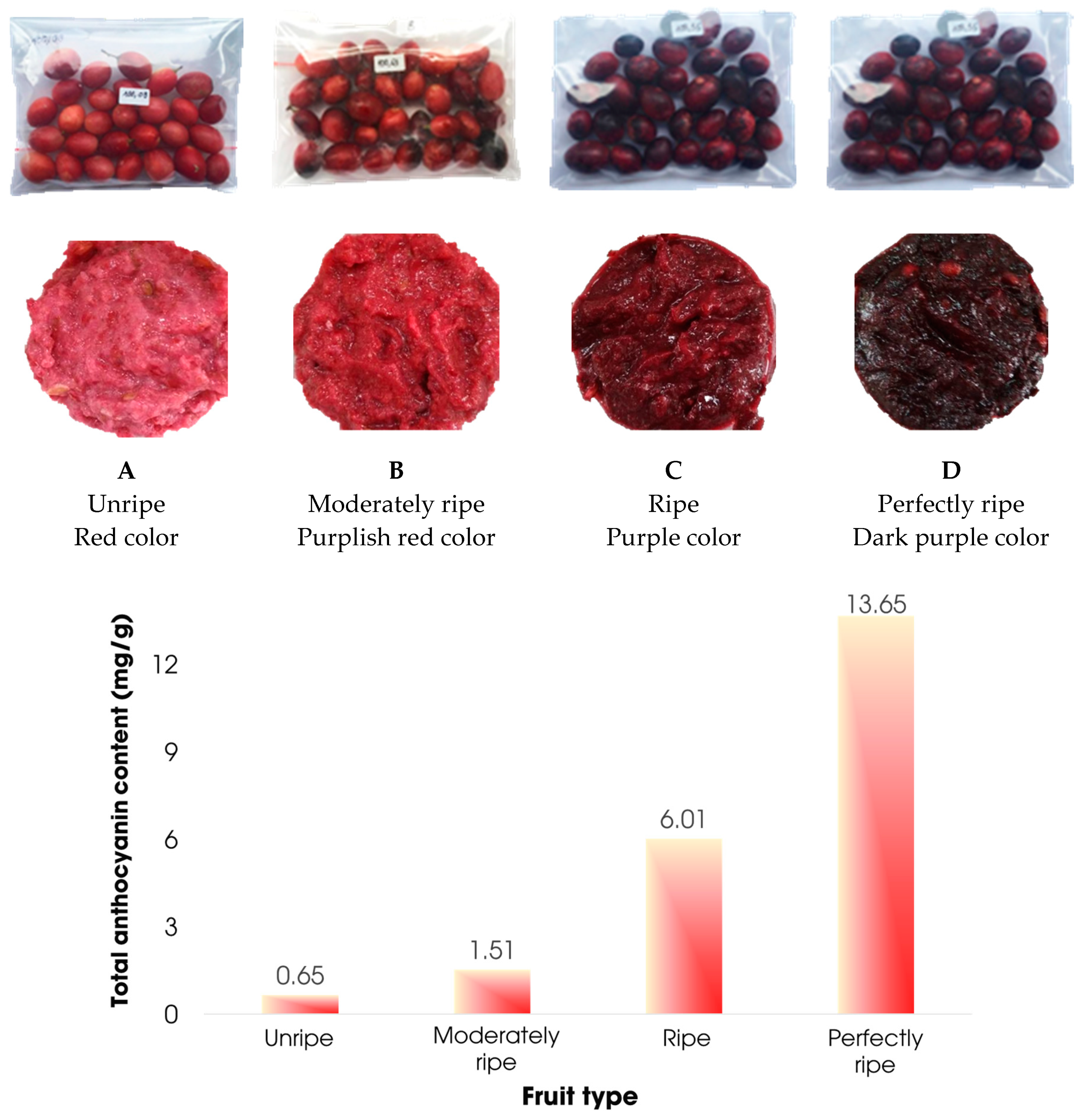



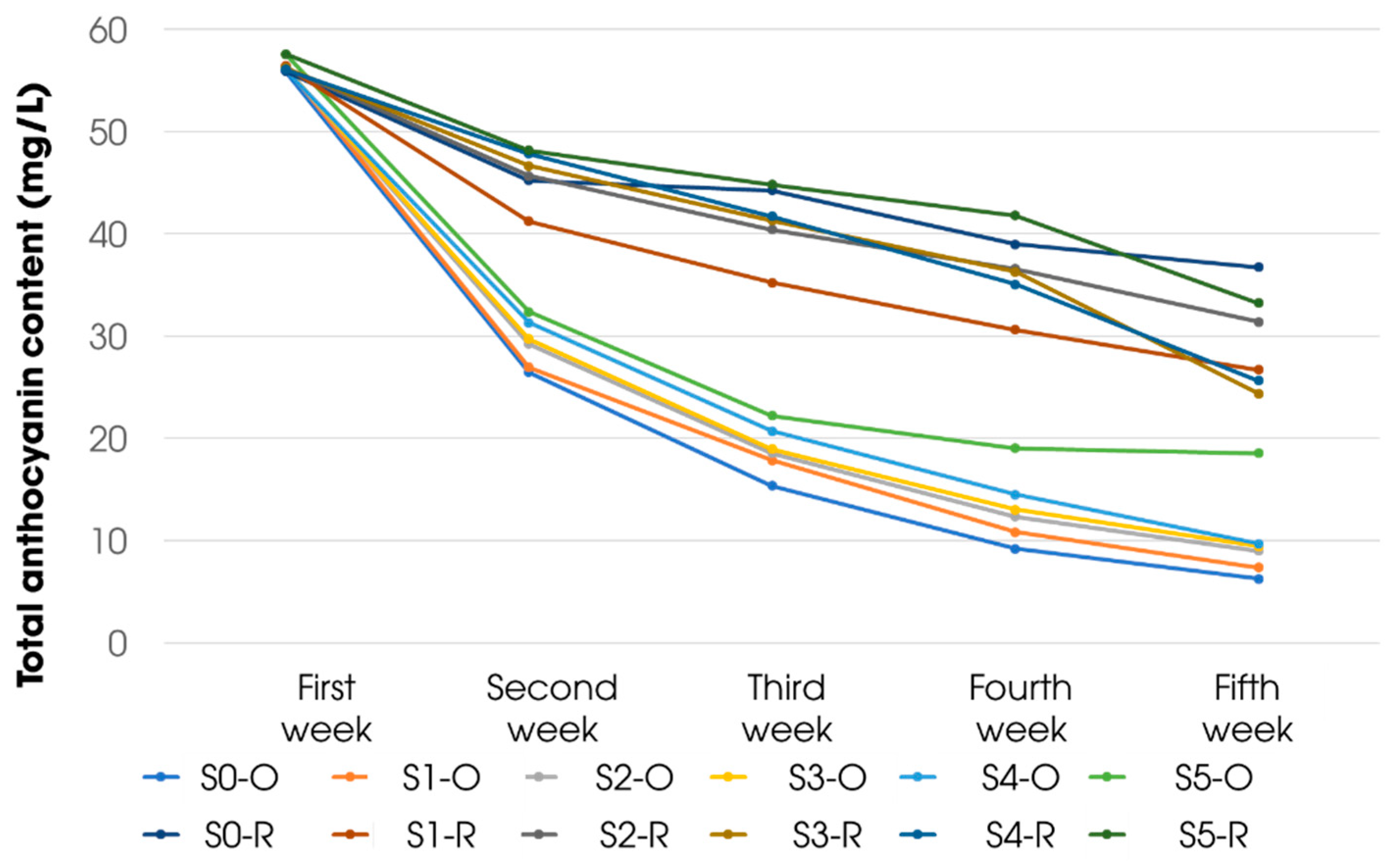




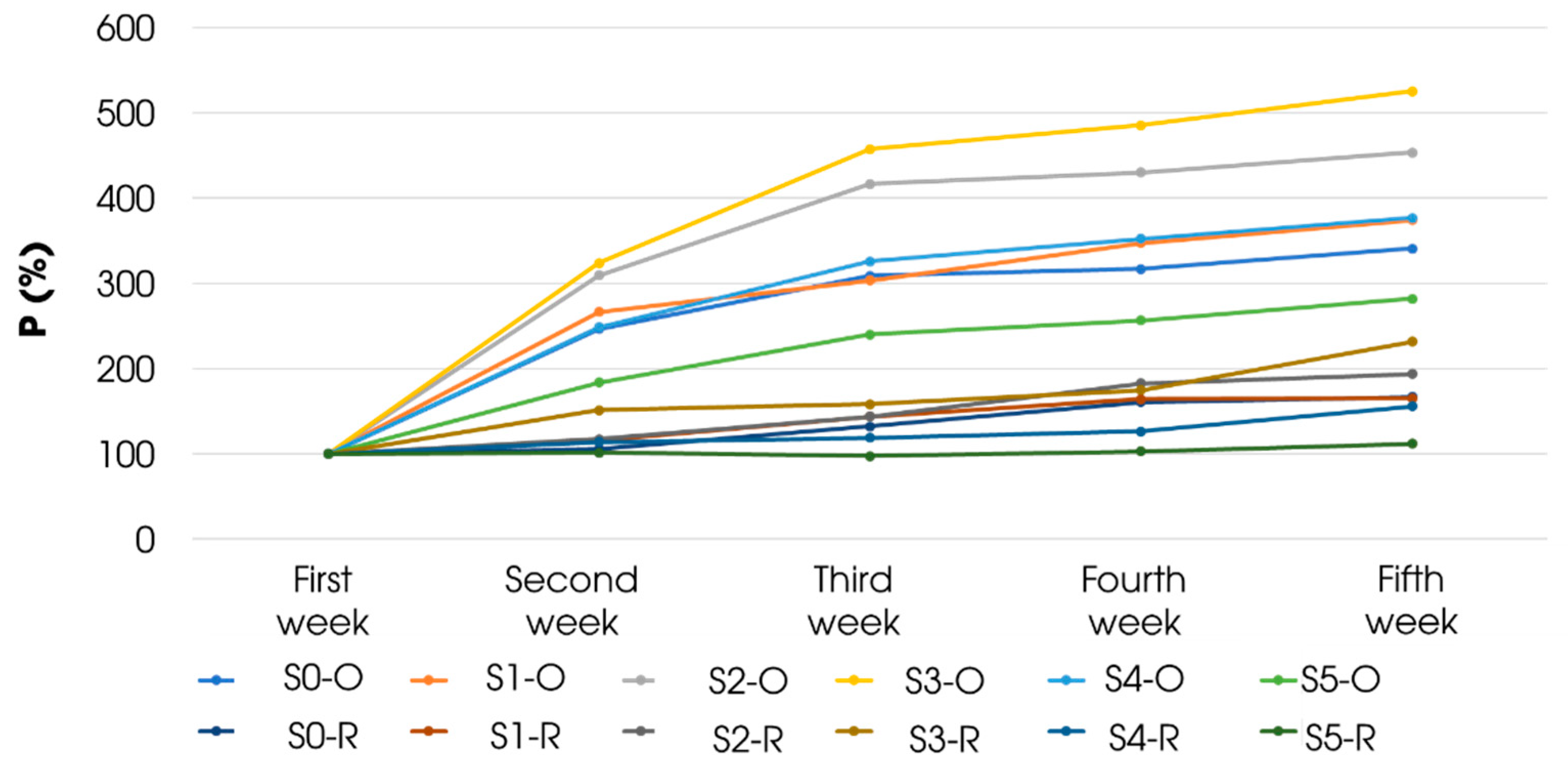
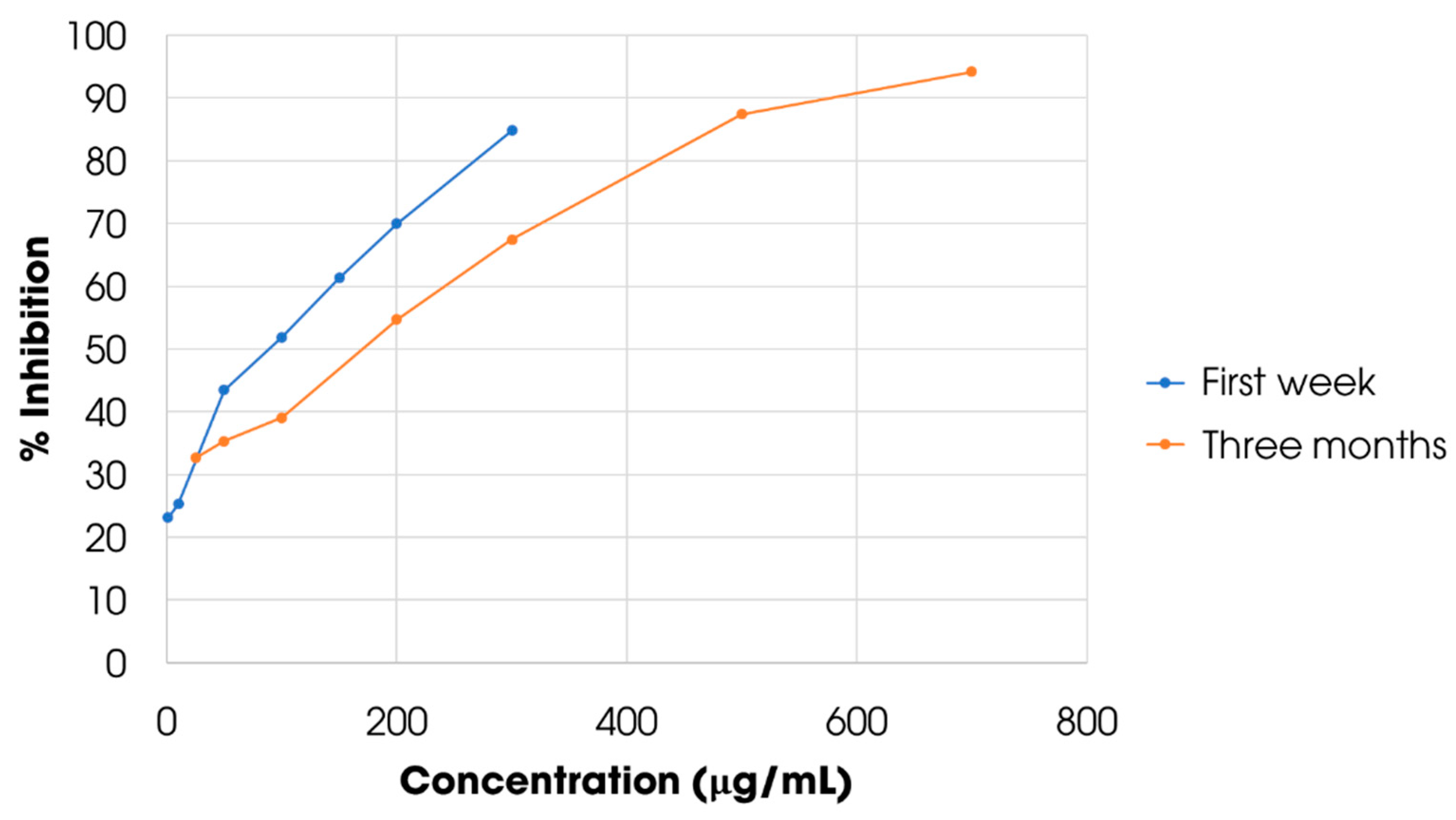
| Sample | Sample 0 | Sample 1 | Sample 2 | Sample 3 | Sample 4 | Sample 5 |
|---|---|---|---|---|---|---|
| m Citric acid (g/L) | 0 | 1 | 2 | 3 | 4 | 5 |
| pH | 3.25 | 2.83 | 2.66 | 2.57 | 2.44 | 2.39 |
| Sample | Sample 0 | Sample 1 | Sample 2 | Sample 3 | Sample 4 | Sample 5 |
|---|---|---|---|---|---|---|
| m Citric acid (g/L) | 0 | 1 | 2 | 3 | 4 | 5 |
| pH | 3.91 | 3.71 | 3.6 | 3.52 | 3.4 | 3.31 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Le, X.T.; Huynh, M.T.; Pham, T.N.; Than, V.T.; Toan, T.Q.; Bach, L.G.; Trung, N.Q. Optimization of Total Anthocyanin Content, Stability and Antioxidant Evaluation of the Anthocyanin Extract from Vietnamese Carissa Carandas L. Fruits. Processes 2019, 7, 468. https://doi.org/10.3390/pr7070468
Le XT, Huynh MT, Pham TN, Than VT, Toan TQ, Bach LG, Trung NQ. Optimization of Total Anthocyanin Content, Stability and Antioxidant Evaluation of the Anthocyanin Extract from Vietnamese Carissa Carandas L. Fruits. Processes. 2019; 7(7):468. https://doi.org/10.3390/pr7070468
Chicago/Turabian StyleLe, Xuan Tien, Minh Thuan Huynh, Tri Nhut Pham, Van Thai Than, Tran Quoc Toan, Long Giang Bach, and Nguyen Quang Trung. 2019. "Optimization of Total Anthocyanin Content, Stability and Antioxidant Evaluation of the Anthocyanin Extract from Vietnamese Carissa Carandas L. Fruits" Processes 7, no. 7: 468. https://doi.org/10.3390/pr7070468
APA StyleLe, X. T., Huynh, M. T., Pham, T. N., Than, V. T., Toan, T. Q., Bach, L. G., & Trung, N. Q. (2019). Optimization of Total Anthocyanin Content, Stability and Antioxidant Evaluation of the Anthocyanin Extract from Vietnamese Carissa Carandas L. Fruits. Processes, 7(7), 468. https://doi.org/10.3390/pr7070468





