Separation of Protein-Binding Anthraquinones from Semen Cassiae Using Two-Stage Foam Fractionation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Aqueous Extract of Semen Cassiae
2.3. Preparation of Semen Cassiae Proteins
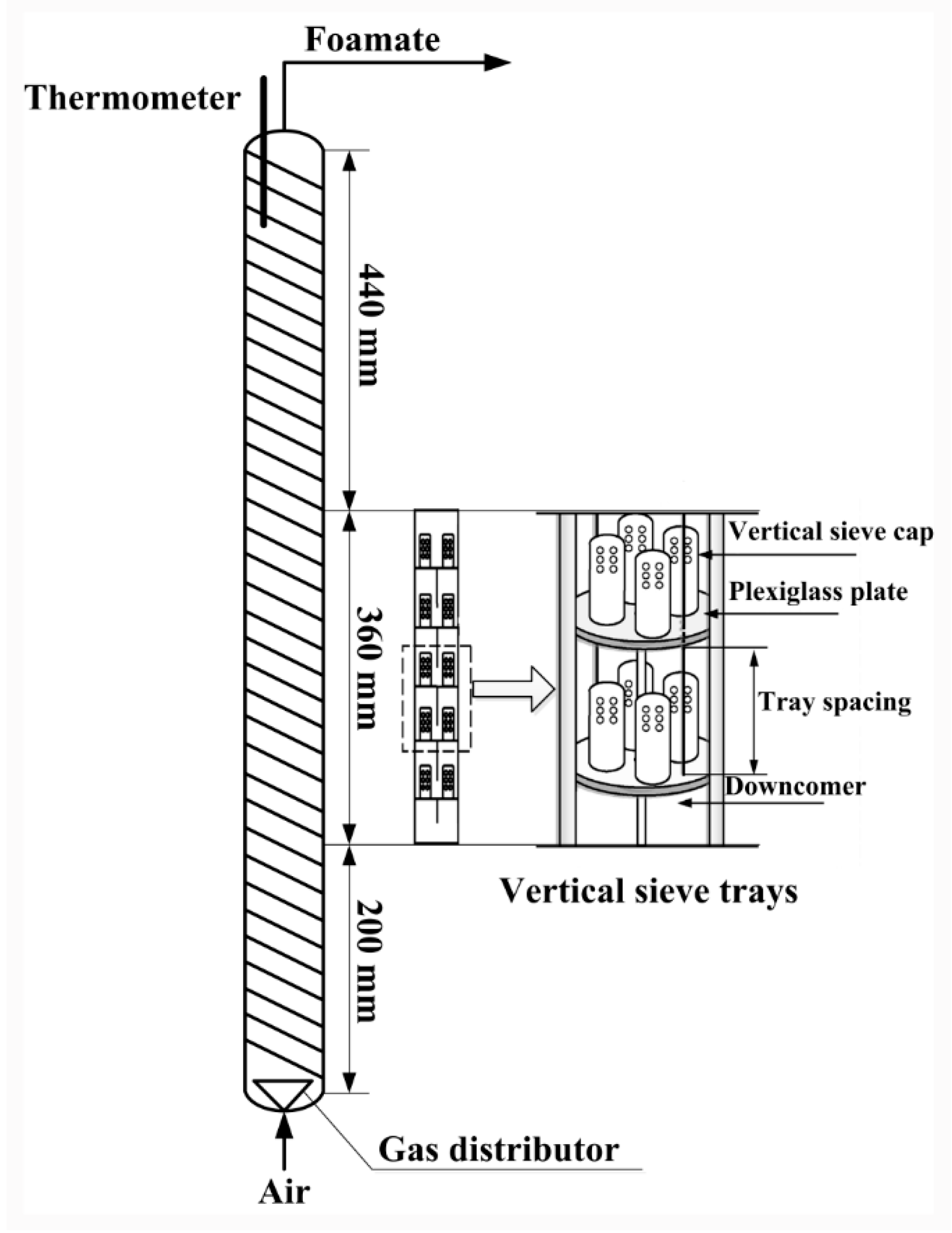
2.4. Separation of Total Anthraquinones from the Aqueous Extract of Semen Cassiae Using Foam Fractionation
2.5. Measurement of Intrinsic Fluorescence Spectrum of Semen Cassiae Proteins
2.6. Measurement of the Concentration of Total Anthraquinones
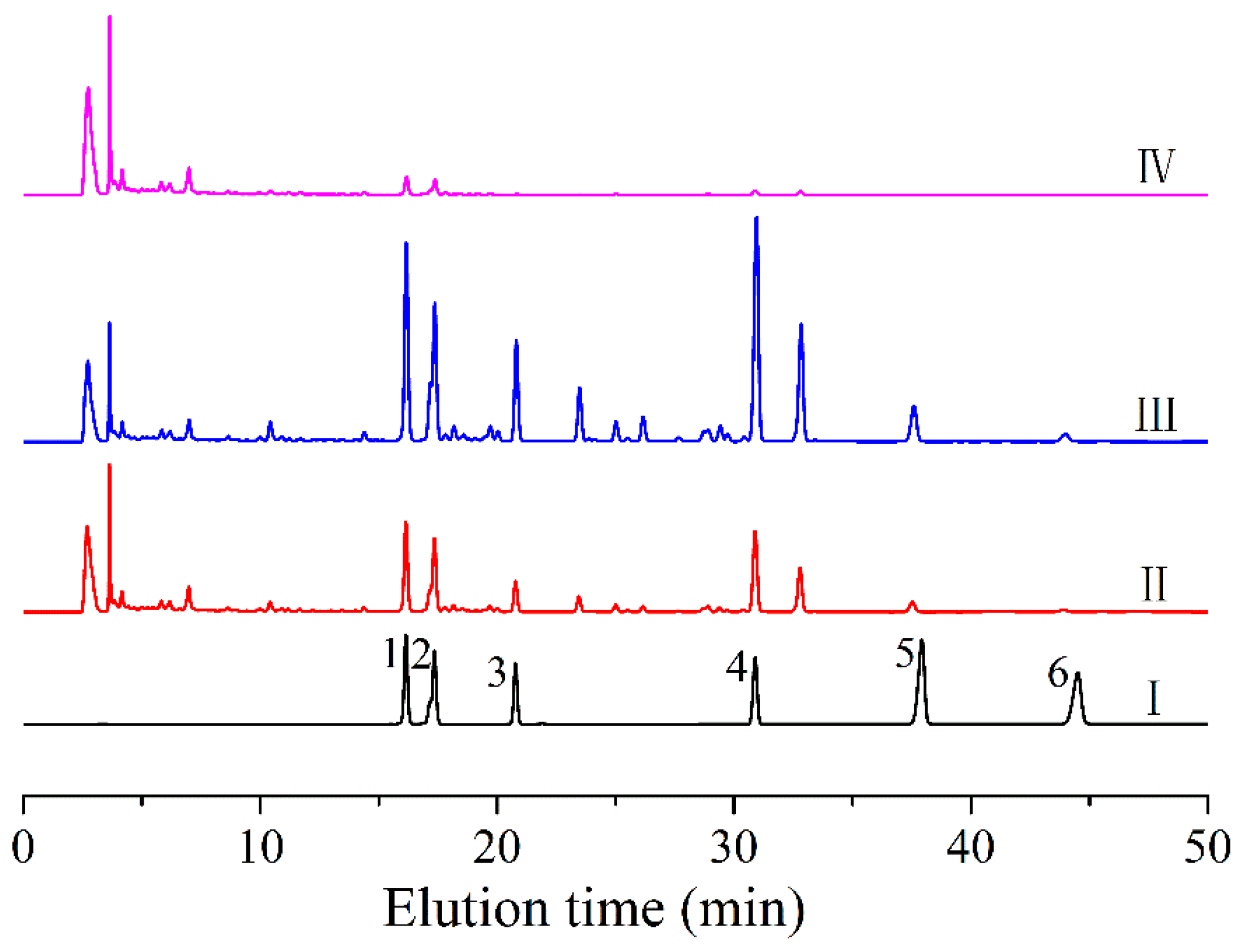
2.7. HPLC Analysis of Semen Cassiae Anthraquinones
2.8. Hydroxyl Radical Scavenging Activity
2.9. Evaluation of the Foam Fractionation Performances
2.10. Statistical Analysis
3. Results and Discussion
3.1. Stem–Volmer Analysis of Interaction between Physcion and Proteins Derived from Semen Cassiae
3.1.1. Fluorescence Quenching Spectra of Semen Cassiae Proteins with Physcion
3.1.2. Binding Constants and Binding Sites of Physcion and Semen Cassiae Proteins
3.1.3. Binding Mode and Nature of the Binding Force between Physcion and Semen Cassiae Proteins
3.2. HPLC Analysis of Foam Fractionation of Anthraquinones from Semen Cassiae Extract
3.3. Two-Stage Foam Fractionation of the Protein-Binding Anthraquinones from Aqueous Extract of Semen Cassiae
3.3.1. Optimization of the First-Stage Foam Fractionation
Effects of VSTs on E and R
Effects of Initial Feed Concentration of Total Anthraquinones on E, R and Cf
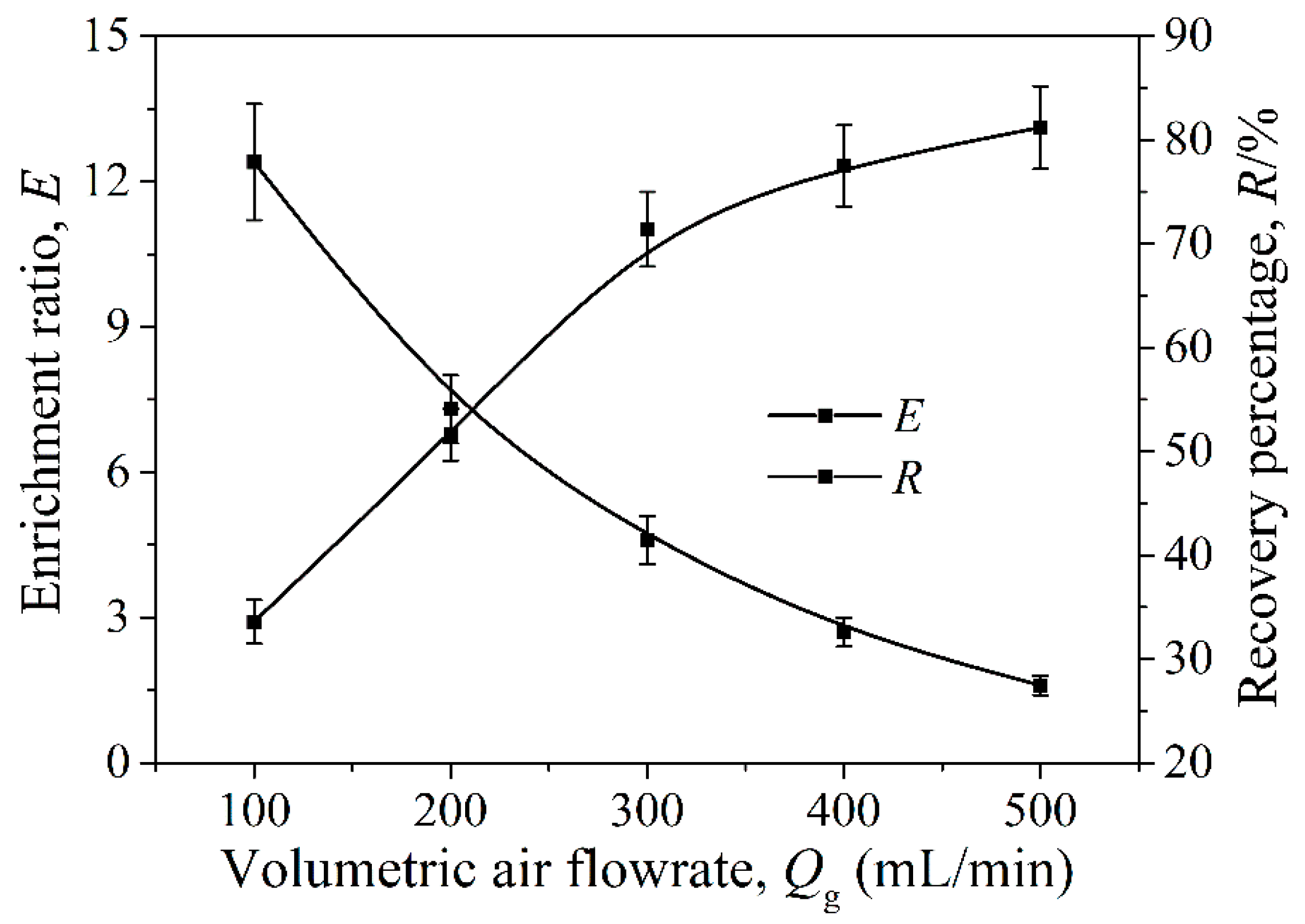
Effects of Volumetric Air Flow Rate on E and R
Effects of Pore Diameter of Gas Distributor on E and R
3.3.2. Optimization of the Second-Stage Foam Fractionation
Effects of Temperature on E and R
Optimization of the Second-Stage Foam Fractionation Using Orthogonal Test
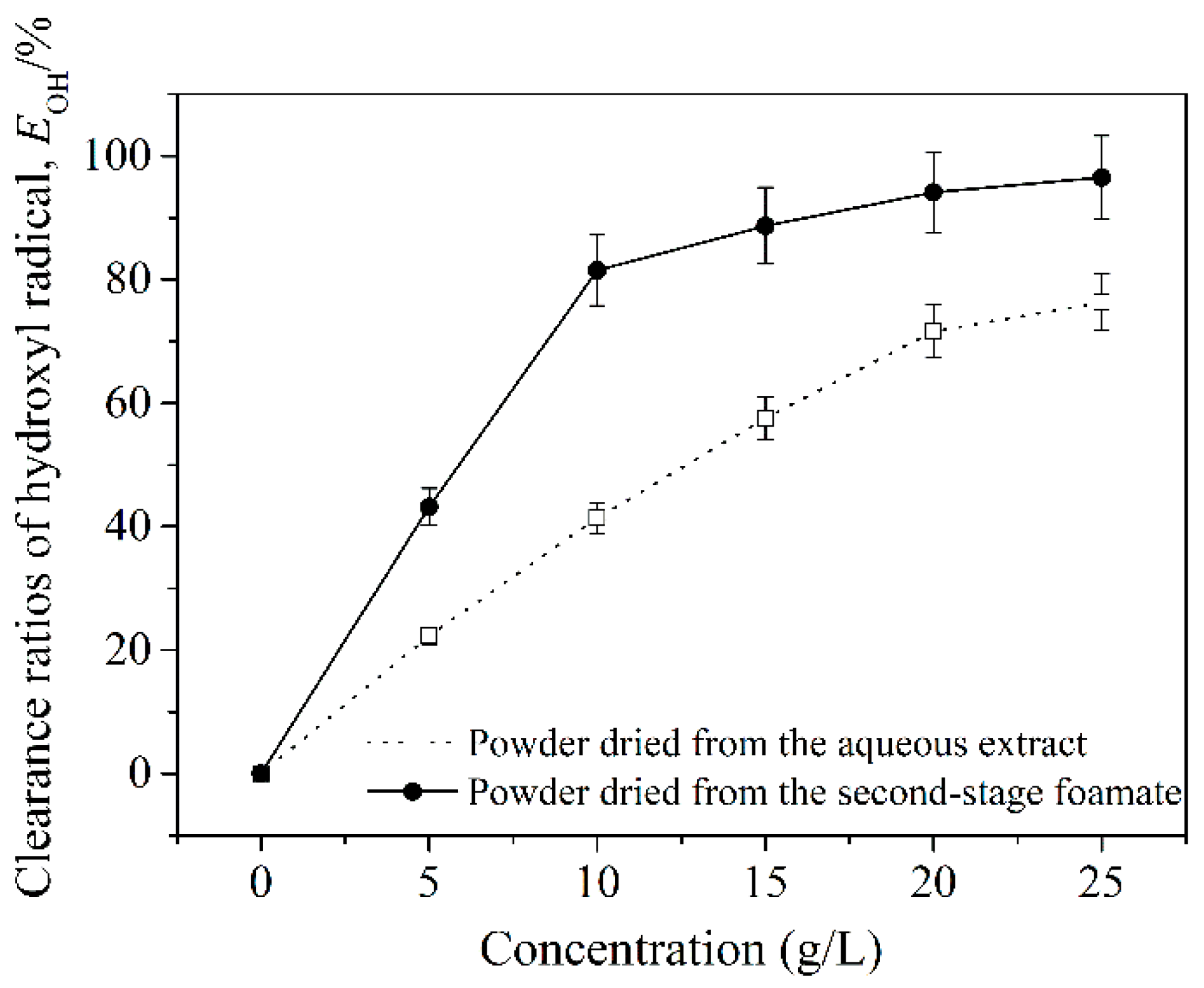
3.4. Hydroxyl Radical Scavenging Activity of Semen Cassiae Anthraquinones in Aqueous Extract and Foamate
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Yang, B.; Hu, J.; Zhu, X.; Zhuang, Y.; Yin, F.; Qin, K.; Cai, B. Qualitative analysis of multiple compounds in raw and prepared Semen Cassiae coupled with multiple statistical strategies. J. Sep. Sci. 2017, 40, 4718–4729. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.D.; Wang, Y.; Wang, Q.; Yang, W.J.; Gu, Y.; Wang, R.; Song, X.M.; Wang, X.J. Quality evaluation of Semen Cassiae (Cassia obtusifolia L.) by using ultra-high performance liquid chromatography coupled with mass spectrometry. J. Sep. Sci. 2012, 35, 2054–2062. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Wang, S.; Guo, X.; Sun, J.; Liu, L.; Wu, L. Simultaneous determination of seven anthraquinones in rat plasma by ultra high performance liquid chromatography-tandem mass spectrometry and pharmacokinetic study after oral administration of Semen Cassiae extract. J. Ethnopharmacol. 2015, 169, 305–313. [Google Scholar] [CrossRef] [PubMed]
- Kwon, K.S.; Lee, J.H.; So, K.S.; Park, B.K.; Lim, H.; Choi, J.S.; Kim, H.P. Aurantio-btusin, an anthraquinone from cassiae semen, ameliorates lung inflammatory responses. Phytother. Res. 2018, 32, 1537–1545. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Chan, C.O.; Lau, C.C.; Yu, Z.; Mok, D.K.; Chen, S. Simultaneous determination of eight anthraquinones in Semen Cassiae by HPLC-DAD. Phytochem. Anal. 2012, 23, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.L.; Tang, L.Y.; Zhou, X.D.; Zhou, G.H.; Wang, Z.J. Five new anthraquinones from the seed of Cassia obtusifolia. Arch. Pharmacal Res. 2015, 38, 1054–1058. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Su, M.; Liang, S.; Sun, H. Investigation of six bioactive anthraquinones in slimming tea by accelerated solvent extraction and high performance capillary electrophoresis with diode-array detection. Food Chem. 2016, 199, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Duval, J.; Pecher, V.; Poujol, M.; Lesellier, E. Research advances for the extraction, analysis and uses of anthraquinones: A review. Ind. Crop. Prod. 2016, 94, 812–833. [Google Scholar] [CrossRef]
- Weng, W.C.; Sheu, S.J. Separation of anthraquinones by capillary electrophoresis and high-performance liquid chromatography. J. High Resolut. Chromatogr. 2000, 23, 143–148. [Google Scholar] [CrossRef]
- Ghosh, R.; Sahu, A.; Pushpavanam, S. Removal of trace hexavalent chromium from aqueous solutions by ion foam fractionation. J. Hazard. Mater. 2019, 367, 589–598. [Google Scholar] [CrossRef]
- Wanschura, R.; Windeisen, E.; Richter, K. Application of foam fractionation to wood. Wood Sci. Technol. 2019, 53, 349–371. [Google Scholar] [CrossRef]
- Liu, W.; Lv, Y.; Tian, S.; Zhang, M.; Shu, T.; Hu, N.; Wu, Z. Foam fractionation for effective recovery of resveratrol from the leaching liquor of Polygonum cuspidatum by using partially ethylated β-cyclodextrin as collector and frother. Ind. Crop. Prod. 2018, 112, 420–426. [Google Scholar] [CrossRef]
- Wu, Z.; Li, N.; Zhang, X.; Xu, Y.; Shu, T.; Liu, W.; Hu, B. Effective recovery of trans-resveratrol from the leaching solution of muscat grape pomace by developing a novel technology of foam fractionation. J. Food Eng. 2019, 241, 41–50. [Google Scholar] [CrossRef]
- Huang, J.; Zhu, L.; Zeng, G.; Shi, L.; Shi, Y.; Yi, K.; Li, X. Recovery of Cd (II) and surfactant in permeate from MEUF by foam fractionation with anionic-nonionic surfactant mixtures. Coll. Surf. A Hysicochem. Eng. Asp. 2019, 570, 81–88. [Google Scholar] [CrossRef]
- Eksborg, S.; Ehrsson, H.; Ekqvist, B. Protein binding of anthraquinone glycosides, with special reference to adriamycin. Cancer Chemother. Pharmacol. 1982, 10, 7–10. [Google Scholar] [CrossRef] [PubMed]
- Bi, S.; Song, D.; Kan, Y.; Xu, D.; Tian, Y.; Zhou, X.; Zhang, H. Spectroscopic characterization of effective components anthraquinones in Chinese medicinal herbs binding with serum albumins. Spectrochim. Acta Part. A Mol. Biomol. Spectrosc. 2005, 62, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Lu, Y.; Zhang, T.; Liu, K.; Liu, L.; He, Z.; Xu, B.; Wu, X. Characterization of binding interactions of anthraquinones and bovine β-lactoglobulin. Food Chem. 2019, 281, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Ding, L.; Wu, Z.; Wang, Y.; Liu, W.; Wang, Y. β-cyclodextrin assisted two-stage foam fractionation of bromelain from the crude extract of pineapple peels. Ind. Crop. Prod. 2016, 94, 233–239. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, Y.; Wu, Z.; Zhang, Q.; Wang, Y. Intensification of the interfacial adsorption of whey soy protein in the liquid phase using a foam separation column with the vertical sieve tray internal. Ind. Crop. Prod. 2014, 53, 308–313. [Google Scholar] [CrossRef]
- Anjusha, S.; Gangaprasad, A. Callus culture and in vitro production of anthraquinone in Gynochthodes umbellata (L.) Razafim. & B. Bremer (Rubiaceae). Ind. Crop. Prod. 2017, 95, 608–614. [Google Scholar]
- Husain, S.R.; Cillard, J.; Cillard, P. Hydroxyl radical scavenging activity of flavonoids. Phytochemistry 1987, 26, 2489–2491. [Google Scholar] [CrossRef]
- Yan, H.; Hider, R.C.; Ma, Y. Synthesis, characterisation and quantum chemical studies of a new series of iron chelatable fluorescent sensors. Mol. Phys. 2019, 117, 661–671. [Google Scholar] [CrossRef]
- Sedmak, J.J.; Grossberg, S.E. A rapid, sensitive, and versatile assay for protein using Coomassie brilliant blue G250. Anal. Biochem. 1977, 79, 544–552. [Google Scholar] [CrossRef]
- Qin, Y.; Zhang, Y.; Yan, S.; Ye, L. A comparison study on the interaction of hyperoside and bovine serum albumin with Tachiya model and Stern-Volmer equation. Spectrochim. Acta Part. A Mol. Biomol. Spectrosc 2010, 75, 1506–1510. [Google Scholar] [CrossRef]
- Qi, J.; Li, B.; Wang, X.; Zhang, Z.; Wang, Z.; Han, J.; Chen, L. Three-dimensional paper-based microfluidic chip device for multiplexed fluorescence detection of Cu2+ and Hg2+ ions based on ion imprinting technology. Sens. Actuators B Chem. 2017, 251, 224–233. [Google Scholar] [CrossRef]
- Panigrahi, G.K.; Suthar, M.K.; Verma, N.; Asthana, S.; Tripathi, A.; Gupta, S.K.; Saxena, J.K.; Das, M. Investigation of the interaction of anthraquinones of Cassia occidentalis seeds with bovine serum albumin by molecular docking and spectroscopic analysis: Correlation to their in vitro cytotoxic potential. Food Res. Int. 2015, 77, 368–377. [Google Scholar] [CrossRef]
- Asghari, A.K.; Norton, I.; Mills, T.; Sadd, P.; Spyropoulos, F. Interfacial and foaming characterisation of mixed protein-starch particle systems for food-foam applications. Food Hydrocoll. 2016, 53, 311–319. [Google Scholar] [CrossRef] [Green Version]
- Li, R.; Xintong, J.; Youshuang, Z.; Yuran, Z.; Jing, G. Precipitation of proteins from soybean whey wastewater by successive foaming and defoaming. Chem. Eng. Process. 2018, 128, 124–131. [Google Scholar] [CrossRef]
- Chen, Y.C.; Parlar, H. Enrichment behavior of immunoglobulin by foam fractionation using response surface methodology. Sep. Purif. Technol. 2013, 107, 102–108. [Google Scholar] [CrossRef] [Green Version]
- Li, R.; Fu, N.; Wu, Z.; Wang, Y.; Wang, Y. Protein aggregation in foam fractionation of bovine serum albumin: Effect of protein concentration. Biochem. Eng. J. 2015, 103, 234–241. [Google Scholar] [CrossRef]
- Firmani Perna, R.; Pereira Gonçalves, M.C.; Costapinto Santana, C. Ascertainment of surfactin concentration in bubbles and foam column operated in semi-batch. Processes 2019, 7, 154. [Google Scholar] [CrossRef]
- Huang, Y.; Zhao, S.; Ding, Y.D.; Liao, Q.; Huang, Y.; Zhu, X. Optimizing the gas distributor based on CO2 bubble dynamic behaviors to improve microalgal biomass production in an air-lift photo-bioreactor. Bioresour. Technol. 2017, 233, 84–91. [Google Scholar] [CrossRef]
- Li, R.; Chen, X.E.; Chang, Y.; Zhang, L.; Zhang, Y.; Zhu, Y.; Wang, T. Increase of bubble size playing a critical role in foam-induced protein aggregation: Aggregation of BSA in foam fractionation. Chem. Eng. Sci. 2017, 174, 387–395. [Google Scholar] [CrossRef]
- Liu, W.; Wu, Z.; Wang, Y.; Li, R.; Huang, D. Isolation of soy whey proteins from isoflavones in the concentrated solution using foam fractionation. Sep. Purif. Technol. 2015, 149, 31–37. [Google Scholar] [CrossRef]
- Delahaije, R.J.; Lech, F.J.; Wierenga, P.A. Investigating the effect of temperature on the formation and stabilization of ovalbumin foams. Food Hydrocoll. 2019, 91, 263–274. [Google Scholar] [CrossRef]




| Anthraquinonyl | R1 | R2 | R3 | R6 | R7 | R8 | |
|---|---|---|---|---|---|---|---|
| Emodin |  | H | OH | H | H | H | H |
| Chrysophanol | OH | H | CH3 | H | H | OH | |
| Rhein | OH | H | H | COOH | H | OH | |
| Physcion | OH | H | OCH3 | CH3 | H | OH | |
| Obtusin | OCH3 | OH | CH3 | OCH3 | OCH3 | OH | |
| Physcion-8-O-β-glucoside | OH | H | CH3 | OCH3 | H | Glu |
| Temperature (°C) | Ka (mol−1) | n | Linear Correlation Coefficient, R2 |
|---|---|---|---|
| 26.0 | 1.17 × 103 | 1.060 | 0.9981 |
| 31.0 | 1.33 × 103 | 0.983 | 0.9990 |
| 37.0 | 1.63 × 103 | 0.925 | 0.9971 |
| Temperature (°C) | ∆G (kJ/mol) | ∆H (kJ/mol) | ∆S (J/mol·K) |
|---|---|---|---|
| 26.0 | −17.45 | 23.3 | 136.3 |
| 31.0 | −18.13 | ||
| 37.0 | 18.95 |
| Aurantio-Obtusifolin | Aloe-Emodin | Rhein | Emodin | Chrysophanol | Physcion | |
|---|---|---|---|---|---|---|
| E | 2.2 | 2.0 | 3.2 | 2.8 | 3.4 | 2.7 |
| R/% | 56.4 | 51.3 | 82.0 | 71.8 | 87.1 | 69.2 |
| E | R/% | |||
|---|---|---|---|---|
| Temperature | With VST | Without VST | With VST | Without VST |
| 25 ± 1 °C | 1.4 ± 0.1 | 1.5 ± 0.1 | 73.6 ± 3.5 | 56.4 ± 2.7 |
| 40 ± 2 °C | 6.6 ± 0.6 | 5.2 ± 0.5 | 41.8 ± 2.1 | 25.4 ± 1.6 |
| Pore Diameter of Gas Distributor (μm) | E | R/% |
|---|---|---|
| 100 ± 20 | 4.1 ± 0.5 | 74.5 ± 3.4 |
| 200 ± 20 | 4.6 ± 0.5 | 71.4 ± 3.6 |
| 400 ± 20 | 6.1 ± 0.6 | 65.2 ± 3.2 |
| 600 ± 20 | 7.4 ± 0.7 | 58.4 ± 2.9 |
| Factors | Levels | ||
|---|---|---|---|
| 1 | 2 | 3 | |
| A, temperature (°C) | 40 | 45 | 50 |
| B, volumetric air flow rate (mL/min) | 100 | 200 | 300 |
| C, pore diameter of gas distributor (μm) | 200 ± 20 μm | 400 ± 20 μm | 600 ± 20 μm |
| No. | A | B | C | E | R/% |
|---|---|---|---|---|---|
| 1 | 40 | 100 | 200 ± 20 | 6.4 ± 0.6 | 63.4 ± 3.2 |
| 2 | 40 | 200 | 400 ± 20 | 4.7 ± 0.5 | 71.6 ± 3.5 |
| 3 | 40 | 300 | 600 ± 20 | 3.9 ± 0.4 | 85.7 ± 4.7 |
| 4 | 45 | 100 | 400 ± 20 | 9.3 ± 0.7 | 43.5 ± 2.6 |
| 5 | 45 | 200 | 600 ± 20 | 7.7 ± 0.8 | 58.4 ± 3.1 |
| 6 | 45 | 300 | 200 ± 20 | 4.1 ± 0.5 | 64.9 ± 3.3 |
| 7 | 50 | 100 | 600 ± 20 | 10.4 ± 1.1 | 23.5 ± 1.5 |
| 8 | 50 | 200 | 200 ± 20 | 7.3 ± 0.6 | 51.7 ± 2.8 |
| 9 | 50 | 300 | 400 ± 20 | 5.5 ± 0.5 | 56.8 ± 2.5 |
| M1E | 5.00 | 8.70 | 5.93 | ||
| M2E | 7.03 | 6.56 | 6.50 | ||
| M3E | 7.73 | 4.50 | 7.33 | ||
| M1R | 73.57 | 43.47 | 60.00 | ||
| M2R | 55.60 | 60.57 | 57.30 | ||
| M3R | 44.00 | 69.13 | 55.87 | ||
| KE | 2.73 | 4.20 | 1.40 | ||
| KR | 29.57 | 25.66 | 4.13 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ding, L.; Wang, Y.; Yue, C.; Wu, Z.; Sun, Y.; Wang, M.; Li, R. Separation of Protein-Binding Anthraquinones from Semen Cassiae Using Two-Stage Foam Fractionation. Processes 2019, 7, 463. https://doi.org/10.3390/pr7070463
Ding L, Wang Y, Yue C, Wu Z, Sun Y, Wang M, Li R. Separation of Protein-Binding Anthraquinones from Semen Cassiae Using Two-Stage Foam Fractionation. Processes. 2019; 7(7):463. https://doi.org/10.3390/pr7070463
Chicago/Turabian StyleDing, Linlin, Yanyan Wang, Chuanjun Yue, Zeying Wu, Ye Sun, Min Wang, and Rui Li. 2019. "Separation of Protein-Binding Anthraquinones from Semen Cassiae Using Two-Stage Foam Fractionation" Processes 7, no. 7: 463. https://doi.org/10.3390/pr7070463
APA StyleDing, L., Wang, Y., Yue, C., Wu, Z., Sun, Y., Wang, M., & Li, R. (2019). Separation of Protein-Binding Anthraquinones from Semen Cassiae Using Two-Stage Foam Fractionation. Processes, 7(7), 463. https://doi.org/10.3390/pr7070463





