Antifungal and Antibacterial Activities of Musa paradisiaca L. Peel Extract: HPLC Analysis of Phenolic and Flavonoid Contents
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Extract and Wood Blocks
2.2. Antimicrobial Activity of Wood Treated with Methanol Extract
2.3. HPLC Conditions for Phenolic and Flavonoid Compounds
2.4. Statistical Analysis
3. Results
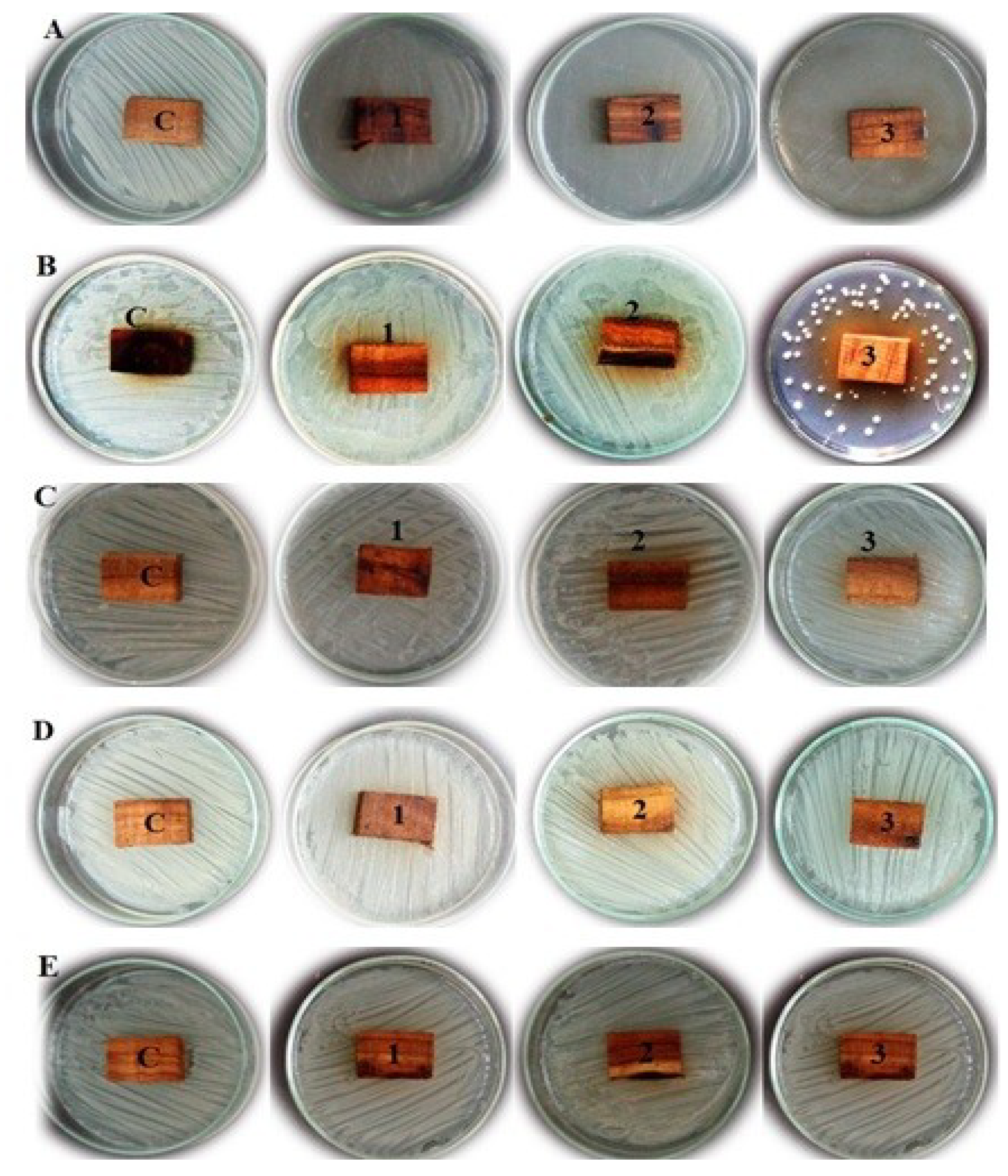
3.1. Visual Observations of Antibacterial Activity on Extract-Treated Wood
3.2. Antifungal Activity of the Extract
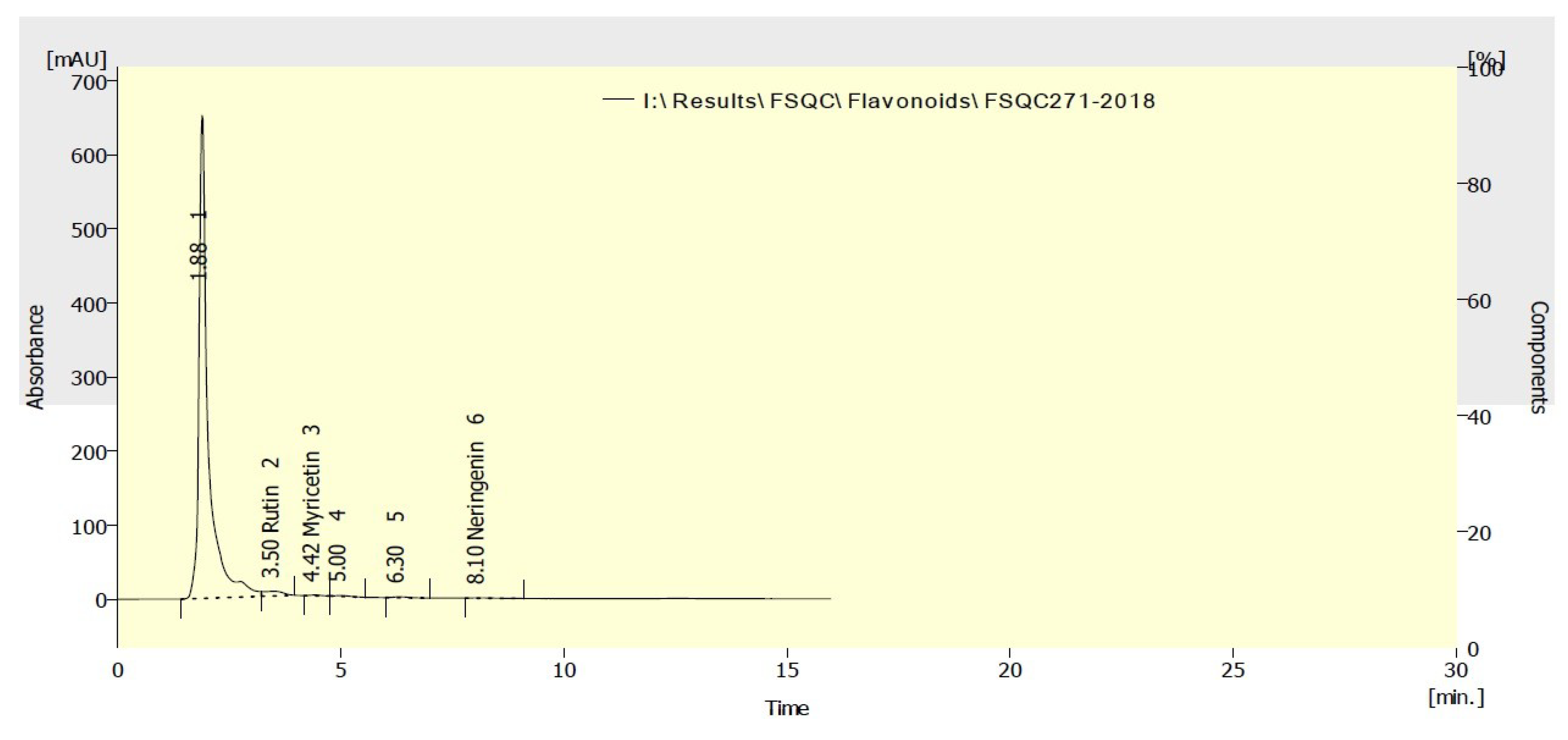
3.3. Phenolic and Flavonoid Compounds of the Methanol Extract
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Okorondu, S.I.; Mepba, H.D.; Okorondu, M.M.O.; Aririatu, L.E. Antibacterial properties of Musa paradisiaca peel extract. Curr. Trends Microbiol. 2010, 6, 21–26. [Google Scholar]
- Asoso, O.S.; Akharaiyi, F.C.; Animba, L.S. Anti-Fungal Activity and Mineral Compositions of Ethanol Extract of Plantain (Musa paradisiaca). Available online: https://afribary.com/works/anti-fungal-activity-and-mineral-compositions-of-ethanol-extract-of-plantain-musa-paradisiaca-1309 (accessed on 9 September 2018).
- Darsini, D.T.P.; Maheshu, V.; Vishnupriya, M.; Sasikumar, J.M. In vitro antioxidant activity of banana (Musa spp. ABB cv. Pisang Awak). Indian J. Biochem. Biophys. 2012, 49, 124–129. [Google Scholar] [PubMed]
- Mokbel, M.S.; Hashinaga, F. Antibacterial and antioxidant activities of Banana (Musa, AAA cv. Cavendish) Fruits Peel. Am. J. Biochem. Biotechnol. 2005, 1, 125–131. [Google Scholar] [CrossRef]
- Nagarajaiah, S.B.; Prakash, J. Chemical composition and antioxidant potential of peels from three varieties of banana. Asian J. Food Agro Ind. 2011, 4, 31–46. [Google Scholar]
- Sulaiman, S.F.; Yusoff, N.A.M.; Eldeen, I.M.; Seow, E.M.; Sajak, A.A.B.; Ooi, K.L. Correlation between total phenolic and mineral contents with antioxidant activity of eight Malaysian bananas (Musa sp.). J. Food Compos. Anal. 2011, 24, 1–10. [Google Scholar] [CrossRef]
- Fidrianny, I.; Rizki, K.; Insanu, M. In vitro antioxidant activities from various extracts of banana peels using ABTS, DPPH assays and correlation with phenolic, flavonoid, carotenoid content. J. Pharm. Pharm. Sci. 2014, 6, 299–303. [Google Scholar]
- Agama-Acevedo, E.; Sañudo-Barajas, J.A.; Vélez De La Rocha, R.; González-Aguilar, G.A.; Bello-Peréz, L.A. Potential of plantain peels flour (Musa paradisiaca L.) as a source of dietary fiber and antioxidant compound. CYTA J. Food 2016, 14, 117–123. [Google Scholar] [CrossRef]
- Fidrianny, I.; Anggraeni, N.A.S.; Insanu, M. Antioxidant properties of peels extracts from three varieties of banana (Musa sp.) grown in West Java-Indonesia. Int. Food Res. J. 2018, 25, 57–64. [Google Scholar]
- Brooks, A.A. Ethanol production potential of local yeast strains isolated from ripe banana peels. Afr. J. Biotechnol. 2008, 7, 3749–3752. [Google Scholar]
- Girish, H.V.; Satish, S. Antibacterial activity of important medicinal plants on human pathogenic bacteria—A comparative analysis. World Appl. Sci. J. 2008, 5, 267–271. [Google Scholar]
- Andersen, B.; Frisvad, J.C.; Søndergaard, I.; Rasmussen, I.S.; Larsen, L.S. Associations between fungal species and water-damaged building materials. Appl. Environ. Microb. 2011, 77, 4180–4188. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Lee, S.; Wu, Y.; Wu, Q. Borate-treated strand board from southern wood species: Resistance against decay and mold fungi. BioResources 2013, 8, 104–114. [Google Scholar] [CrossRef]
- Lee, Y.M.; Lee, H.; Jang, Y.; Cho, Y.; Kim, G.-H.; Kim, J.-J. Phylogenetic analysis of major molds inhabiting woods. Part 4. Genus Alternaria. Holzforschung 2014, 68, 247–251. [Google Scholar] [CrossRef]
- Salem, M.Z.M. EDX measurements and SEM examination of surface of some imported woods inoculated by three mold fungi. Measurement 2016, 86, 301–309. [Google Scholar] [CrossRef]
- Mohamed, W.A.; Mansour, M.M.A.; Salem, M.Z.M. Lemna gibba and Eichhornia crassipes extracts: Clean alternatives for deacidification, antioxidation and fungicidal treatment of historical paper. J. Clean. Prod. 2019, 219, 846–855. [Google Scholar] [CrossRef]
- Salem, M.Z.M.; Mansour, M.M.A.; Elansary, H.O. Evaluation of the effect of inner and outer bark extracts of Sugar Maple (Acer saccharum var. saccharum) in combination with citric acid against the growth of three common molds. J. Wood Chem. Technol. 2019, 16, 1–12. [Google Scholar] [CrossRef]
- Ochoa, J.L.; Hernández-Montiel, L.G.; Latisnere-Barragán, H.; León de La Luz, J.L.; Larralde-Corona, C.P. Isolation and identification of pathogenic fungi from orange Citrus sinensis L. Osbeck cultured in Baja California Sur, Mexico. Cienc. Tecnol. Aliment. 2007, 5, 352–359. [Google Scholar] [CrossRef]
- Abdel-Monaim, M.F.; El-Morsi, M.E.A.; Hassan, M.A.E. Control of root rot and wilt disease complex of some evergreen fruit transplants by using plant growth promoting rhizobacteria in the New Valley Governorate, Egypt. J. Phytopathol. Pest Manag. 2014, 1, 23–33. [Google Scholar]
- Restuccia, C.; Giusino, F.; Licciardello, F.; Randazzo, C.; Caggia, C.; Muratore, G. Biological control of peach fungal pathogens by commercial products and indigenous yeasts. J. Food Protect. 2006, 69, 2465–2470. [Google Scholar] [CrossRef]
- Hernández-Montiel, L.G.; Ochoa, J.L.; Troyo-Diéguez, E.; Larralde-Corona, C.P. Biocontrol of postharvest blue mold (Penicillium italicum Wehmer) on Mexican lime by marine and citrus Debaryomyces hansenii isolates. Postharvest Biol. Technol. 2010, 56, 181–187. [Google Scholar] [CrossRef]
- Pérombelon, M.C.M. Potato diseases caused by soft rot erwinias: An overview of pathogenesis. Plant Pathol. 2002, 51, 1–12. [Google Scholar]
- Meyer, V. A small protein that fights fungi: AFP as a new promising antifungal agent of biotechnological value. Appl. Microbiol. Biot. 2008, 78, 17–28. [Google Scholar] [CrossRef] [PubMed]
- El-Hefny, M.; Mohamed, A.A.; Salem, M.Z.M.; Abd El-Kareem, M.S.M.; Ali, H.M. Chemical composition, antioxidant capacity and antibacterial activity against some potato bacterial pathogens of fruit extracts from Phytolacca dioica and Ziziphus spina-christi grown in Egypt. Sci. Hortic. 2018, 233, 225–232. [Google Scholar] [CrossRef]
- Salem, M.Z.M.; Elansary, H.O.; Elkelish, A.A.; Zeidler, A.; Ali, H.M.; Hefny, M.E.L.; Yessoufou, K. In vitro bioactivity and antimicrobial activity of Picea abies and Larix decidua wood and bark extracts. BioResources 2016, 11, 9421–9437. [Google Scholar] [CrossRef]
- Salem, M.Z.M.; Behiry, S.I.; Salem, A.Z.M. Effectiveness of root-bark extract from Salvadora persica against the growth of certain molecularly identified pathogenic bacteria. Microb. Pathogen. 2018, 117, 320–326. [Google Scholar] [CrossRef]
- Salem, M.Z.M.; Ali, H.M.; El-Shanhorey, N.A.; Abdel-Megeed, A. Evaluation of extracts and essential oil from Callistemon viminalis leaves: Antibacterial and antioxidant activities, total phenolic and flavonoid contents. Asian Pac. J. Trop. Med. 2013, 6, 785–791. [Google Scholar] [CrossRef]
- Mansour, M.M.A.; Salem, M.Z.M. Evaluation of wood treated with some natural extracts and Paraloid B-72 against the fungus Trichoderma harzianum: Wood elemental composition, in-vitro and application evidence. Int. Biodeterior. Biodegr. 2015, 100, 62–69. [Google Scholar] [CrossRef]
- NCCLS–National Committee for Clinical Laboratory Standards. Performance Standards for Antimicrobial Disk Susceptibility Tests Sixth Edition: Approved Standard M2-A6; NCCLS: Villanova, PA, USA, 1997. [Google Scholar]
- Mansour, M.M.A.; Abdel-Megeed, A.; Nasser, R.A.; Salem, M.Z.M. Comparative evaluation of some woody tree methanolic extracts and Paraloid B-72 against phytopathogenic mold fungi Alternaria tenuissima and Fusarium culmorum. BioResources 2015, 10, 2570–2584. [Google Scholar] [CrossRef]
- Salem, M.Z.M.; Zidan, Y.E.; Mansour, M.M.A.; El Hadidi, N.M.N.; Abo Elgat, W.A.A. Antifungal activities of two essential oils used in the treatment of three commercial woods deteriorated by five common mold fungi. Int. Biodeterior. Biodegradation 2016, 106, 88–96. [Google Scholar] [CrossRef]
- Salem, M.Z.M.; Zidan, Y.E.; Mansour, M.M.A.; El Hadidi, N.M.N.; Abo Elgat, W.A.A. Evaluation of usage three natural extracts applied to three commercial wood species against five common molds. Int. Biodeterior. Biodegrad. 2016, 110, 206–226. [Google Scholar] [CrossRef]
- Al-Huqail, A.A.; Behiry, S.I.; Salem, M.Z.M.; Ali, H.M.; Siddiqui, M.H.; Salem, A.Z.M. Antifungal, antibacterial, and antioxidant activities of Acacia saligna (Labill.) H. L. Wendl. flower extract: HPLC analysis of phenolic and flavonoid compounds. Molecules 2019, 24, 700. [Google Scholar] [CrossRef] [PubMed]
- SAS. Users Guide: Statistics (Release 8.02); SAS Institute Inc.: Cary, NC, USA, 2001. [Google Scholar]
- Dall’Acqua, S.; Minesso, P.; Shresta, B.B.; Comai, S.; Jha, P.K.; Gewali, M.B.; Greco, E.; Cervellati, R.; Innocenti, G. Phytochemical and antioxidant-related investigations on bark of Abies spectabilis (D. don) Spach. from Nepal. Molecules 2012, 17, 1686–1697. [Google Scholar] [CrossRef] [PubMed]
- Fawole, O.A.; Makunga, N.P.; Opara, U.L. Antibacterial, antioxidant and tyrosinase-inhibition activities of pomegranate fruit peel methanolic extract. BMC Complement. Altern. Med. 2012, 12, 200–218. [Google Scholar] [CrossRef] [PubMed]
- Baldan, V.; Sut, S.; Faggian, M.; Gassa, E.D.; Ferrari, S.; De Nadai, G.; Francescato, S.; Baratto, G.; Dall’Acqua, S. Larix decidua bark as a source of phytoconstituents: An LC-MS study. Molecules 2017, 22, 1974. [Google Scholar] [CrossRef] [PubMed]
- Aquino, C.F.; Salomão, L.C.C.; Ribeiro, S.M.R.; de Siqueira, D.L.; Cecon, P.R. Carbohydrates, phenolic compounds and antioxidant activity in pulp and peel of 15 banana cultivars. Rev. Bras. Frutic. 2016, 38. [Google Scholar] [CrossRef]
- Mahmood, A.; Ngah, N.; Omar, M. Phytochemicals constituent and antioxidant activities in musa x paradisiaca flower. Eur. J. Sci. Res. 2011, 66, 311–318. [Google Scholar]
- Vipa, S.; Chidchom, H. Extraction of tannin from banana peel. Kasetsart J. 1994, 28, 578–586. [Google Scholar]
- Anal, A.K.; Jaisanti, S.; Noomhorm, A. Enhanced yield of phenolic extracts from banana peels (Musa acuminata Colla AAA) and cinnamon barks (Cinnamomum varum) and their antioxidative potentials in fish oil. J. Food. Sci. Technol. 2014, 51, 2632–2639. [Google Scholar] [CrossRef]
- Kanazawa, K.; Sakakibara, H. High content of dopamine, a strong antioxidant, in Cavendish banana. J. Agric. Food Chem. 2000, 48, 844–848. [Google Scholar] [CrossRef]
- Subagio, A.; Morita, N.; Sawada, S. Carotenoids and their fatty-acid esters in banana peel. J. Nutr. Sci. Vitaminol. 1996, 42, 553–566. [Google Scholar] [CrossRef]
- Corona, M.A.G.; Gómez-Patiño, M.B.; de Flores, M.J.P.; Ruiz, L.A.M.; Martinez, B.M.B.; Arrieta-Baez, D. An integrated analysis of the Musa paradisiaca peel, using UHPLC-ESI, FTIR and confocal microscopy techniques. Ann. Chromatogr. Sep. Tech. 2015, 1, 1005. [Google Scholar]
- Someya, S.; Yoshiki, Y.; Okubo, K. Antioxidant compounds from bananas (Musa Cavendish). Food Chem. 2002, 79, 351–354. [Google Scholar] [CrossRef]
- Mierziak, J.; Kostyn, K.; Kulma, A. Flavonoids as important molecules of plant interactions with the environment. Molecules 2014, 19, 16240–16265. [Google Scholar] [CrossRef]
- Orhan, D.D.; Özçelik, B.; Özgen, S.; Ergun, F. Antibacterial, antifungal, and antiviral activities of some flavonoids. Microbiol. Res. 2010, 165, 496–504. [Google Scholar] [CrossRef]
- Okorondu, S.I.; Akujobi, C.O.; Nwachukwu, I.N. Antifungal properties of Musa paradisiaca (Plantain) peel and stalk extracts. Int. J. Biol. Chem. Sci. 2012, 6, 1527–1534. [Google Scholar] [CrossRef]
- Prakash, B.; Sumangala, C.H.; Melappa, G.; Gavimath, C. Evaluation of antifungal activity of Banana peel against scalp fungi. Mater. Today Proc. 2017, 4, 11977–11983. [Google Scholar] [CrossRef]
- Carvalho, R.S.; Carollo, C.A.; de Magalhães, J.C.; Palumbo, J.M.C.; Boaretto, A.G.; Nunes e Sá, I.C.; Ferraz, A.C.; Lima, W.G.; de Siqueira, J.M.; Ferreira, J.M.S. Antibacterial and antifungal activities of phenolic compound-enriched ethyl acetate fraction from Cochlospermum regium (mart. Et. Schr.) Pilger roots: Mechanisms of action and synergism with tannin and gallic acid. S. Afr. J. Bot. 2018, 114, 181–187. [Google Scholar] [CrossRef]
- Oliveira, C.C.; Siqueira, J.M.; Souza, K.C.B.; Resende, U.M. Antibacterial activity of rhizomes from Cochlospermum regium preliminary results. Fitoterapia 1996, 67, 176–177. [Google Scholar]
- Sólon, S.; Carollo, C.A.; Brandão, L.F.G.; Macedo, C.S.; Klein, A.; Dias-Junior, C.A.; Siqueira, J.M. Phenolic derivatives and other chemical compounds from Cochlospermum regium. Quím. Nova 2012, 35, 1169–1172. [Google Scholar] [CrossRef]
- Borges, A.; Ferreira, C.; Saavedra, M.J.; Simões, M. Antibacterial Activity and Mode of Action of Ferulic and Gallic Acids Against Pathogenic Bacteria. Microb. Drug Resist. 2013, 19, 256–265. [Google Scholar] [CrossRef] [PubMed]
- Alves, C.T.; Ferreira, I.C.; Barros, L.; Silva, S.; Azeredo, J.; Henriques, M. Antifungal activity of phenolic compounds identified in flowers from North Eastern Portugal against Candida species. Future Microbiol. 2014, 9, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Singulani, J.L.; Scorzoni, L.; Gomes, P.C.; Nazaré, A.C.; Polaquini, C.R.; Regasini, L.O.; Fusco-Almeida, A.M.; Mendes-Giannini, M.J.S. Activity of gallic acid and its ester derivatives in Caenorhabditis elegans and zebrafish (Danio rerio) models. Future Med. Chem. 2017, 9, 1863–1872. [Google Scholar] [CrossRef]
- Onaran, A.; Bayram, M. Determination of Antifungal Activity and Phenolic Compounds of Endemic Muscari aucheri (Boiss.) Baker Extract. J. Agric. Fac. Gaziosmanpasa Univ. 2018, 35, 60–67. [Google Scholar] [CrossRef]
- Johann, S.; Mendes, B.G.; Missau, F.C.; de Resende, M.A.; Pizzolatti, M.G. Antifungal activity of five species of Polygala. Braz. J. Microbiol. 2011, 42, 1065–1075. [Google Scholar] [CrossRef] [PubMed]
- Cushnie, T.P.T.; Lamb, A.J. Antimicrobial activity of flavonoids. Int. J. Antimicrob. Agents 2005, 26, 343–356. [Google Scholar] [CrossRef] [PubMed]
- Hendra, R.; Ahmad, S.; Sukari, A.; Shukor, M.Y.; Oskoueian, E. Flavonoid analyses and antimicrobial Activity of various parts of Phaleria macrocarpa (Scheff.) Boerl Fruit. Int. J. Mol. Sci. 2011, 12, 3422–3431. [Google Scholar] [CrossRef]


| Conc. (%) | Inhibition Zone (mm) | ||||
|---|---|---|---|---|---|
| A. tumefaciens | D. solani | E. amylovora | P. cichorii | S. pylmuthica | |
| 0 | 0.00 b | 0.00 | 0.00 | 0.00 | 0.00 |
| 1 | 90.00 a | 0.00 | 0.00 | 0.00 | 0.00 |
| 2 | 90.00 a | 0.00 | 0.00 | 0.00 | 0.00 |
| 3 | 90.00 a | 0.00 | 0.00 | 0.00 | 0.00 |
| Significant | *** | ns | ns | ns | ns |
| Conc. (%) | Inhibition Percentage (%) | |
|---|---|---|
| F. culmorum | R. solani | |
| 0 | 0.00 d | 0.00 c |
| 1 | 37.03 c ± 2.79 | 50.00 b ± 0.00 |
| 2 | 55.18 b ± 1.69 | 93.33 a ± 1.11 |
| 3 | 68.88 a ± 2.22 | 94.07 a ± 0.64 |
| LSD0.05 | 3.722 | 1.207 |
| Compound | Conc. (mg/100 g DE *) |
|---|---|
| Phenolic compounds | |
| Gallic acid | 7.73 |
| Catechol | 0.82 |
| Ferulic acid | 1.63 |
| Ellagic acid | 16.19 |
| o-Coumaric acid | 1.12 |
| Salicylic acid | 0.27 |
| Cinnamic acid | 0.07 |
| Flavonoid compounds | |
| Rutin | 973.08 |
| Myricetin | 11.52 |
| Naringenin | 8.47 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Behiry, S.I.; Okla, M.K.; Alamri, S.A.; EL-Hefny, M.; Salem, M.Z.M.; Alaraidh, I.A.; Ali, H.M.; Al-Ghtani, S.M.; Monroy, J.C.; Salem, A.Z.M. Antifungal and Antibacterial Activities of Musa paradisiaca L. Peel Extract: HPLC Analysis of Phenolic and Flavonoid Contents. Processes 2019, 7, 215. https://doi.org/10.3390/pr7040215
Behiry SI, Okla MK, Alamri SA, EL-Hefny M, Salem MZM, Alaraidh IA, Ali HM, Al-Ghtani SM, Monroy JC, Salem AZM. Antifungal and Antibacterial Activities of Musa paradisiaca L. Peel Extract: HPLC Analysis of Phenolic and Flavonoid Contents. Processes. 2019; 7(4):215. https://doi.org/10.3390/pr7040215
Chicago/Turabian StyleBehiry, Said I., Mohmmad K. Okla, Saud A. Alamri, Mervat EL-Hefny, Mohamed Z. M. Salem, Ibrahim A. Alaraidh, Hayssam M. Ali, Salem M. Al-Ghtani, José C. Monroy, and Abdelfattah Z. M. Salem. 2019. "Antifungal and Antibacterial Activities of Musa paradisiaca L. Peel Extract: HPLC Analysis of Phenolic and Flavonoid Contents" Processes 7, no. 4: 215. https://doi.org/10.3390/pr7040215
APA StyleBehiry, S. I., Okla, M. K., Alamri, S. A., EL-Hefny, M., Salem, M. Z. M., Alaraidh, I. A., Ali, H. M., Al-Ghtani, S. M., Monroy, J. C., & Salem, A. Z. M. (2019). Antifungal and Antibacterial Activities of Musa paradisiaca L. Peel Extract: HPLC Analysis of Phenolic and Flavonoid Contents. Processes, 7(4), 215. https://doi.org/10.3390/pr7040215









