2.1. Drying Kinetics
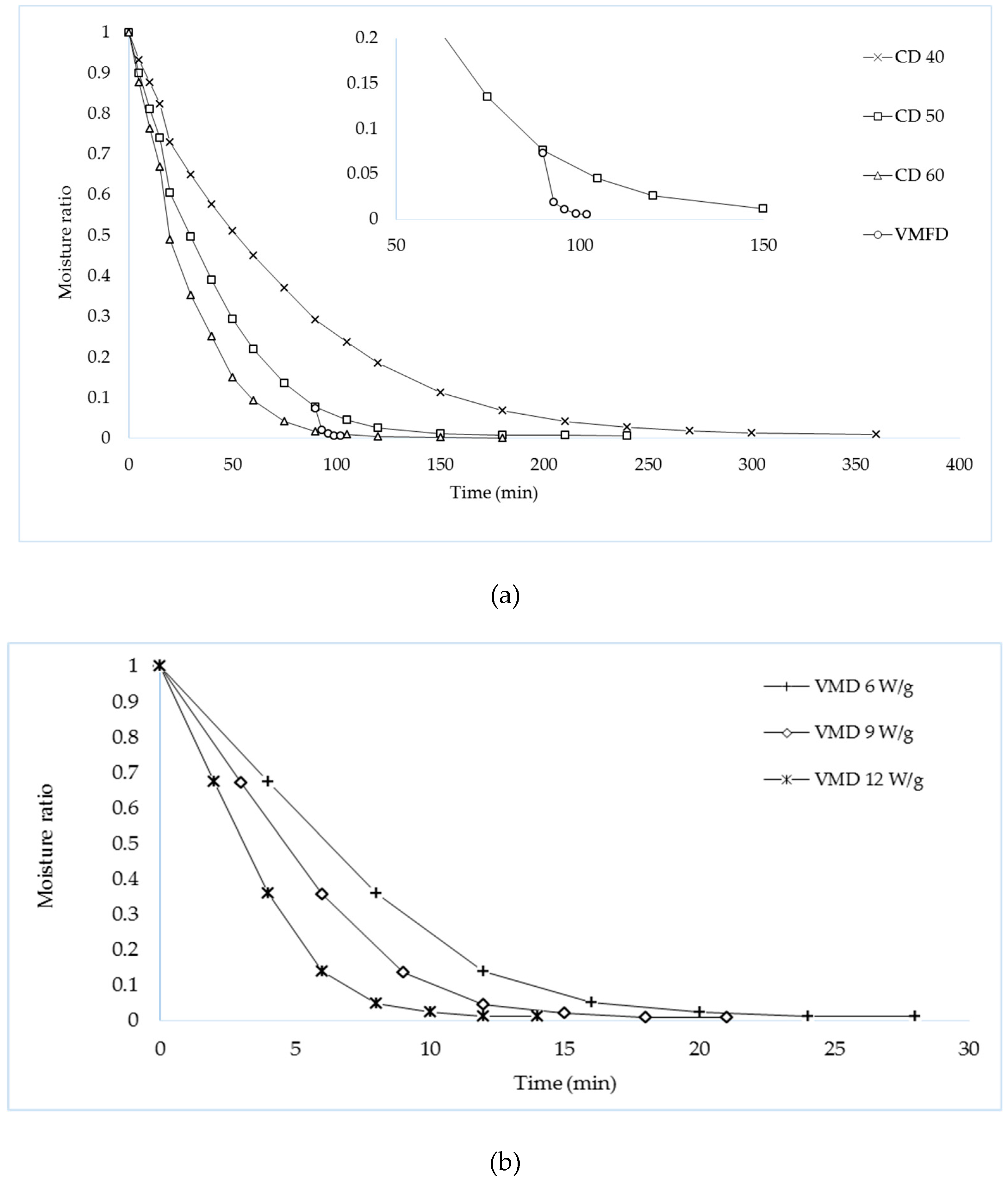
Figure 1a shows the drying kinetics of
P. nodiflora leaves dried with CD and CPD–VMFD, while
Figure 1b shows the drying kinetics of
P. nodiflora dried using VMD. Drying kinetics of all drying methods were best described by an exponential function, namely, the modified Page model (Equation (4)).
Table 1 shows the values of both statistical parameters and mathematical constants of the Lewis, modified Page and Henderson and Pabis model. Selection of the best model was based on the highest Coefficient of determination (
R2) and the lowest root-mean-square error (RMSE), thereby suggesting goodness of fit. The values,
R2 and RMSE, obtained suggest a good agreement between the experimental values obtained and the modified Page model.
Drying duration of CD was the longest compared with VMD and CPD–VMFD. At the initial stage of CD, moisture loss was fast; however, successive moisture loss reduced with time. This phenomenon occurs because CD uses surface heating; the evaporation of moisture from the surface of plant material was efficient but moisture diffusion from internal region to the surface is slow and rate-limiting [
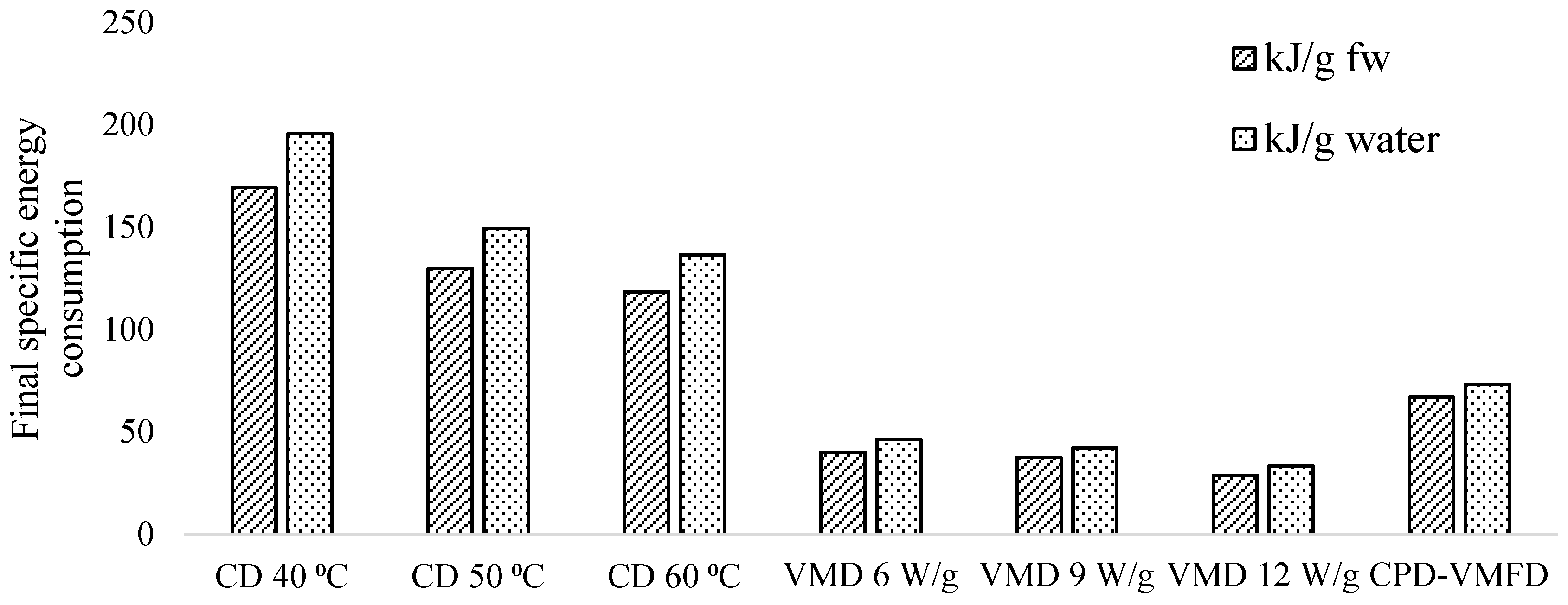
19]. However, when VMFD at 9 W/g was applied to convective pre-dried leaves, drying time was 105 min, as opposed to 240 min of CD, thereby achieving 2.3 times shorter drying duration.
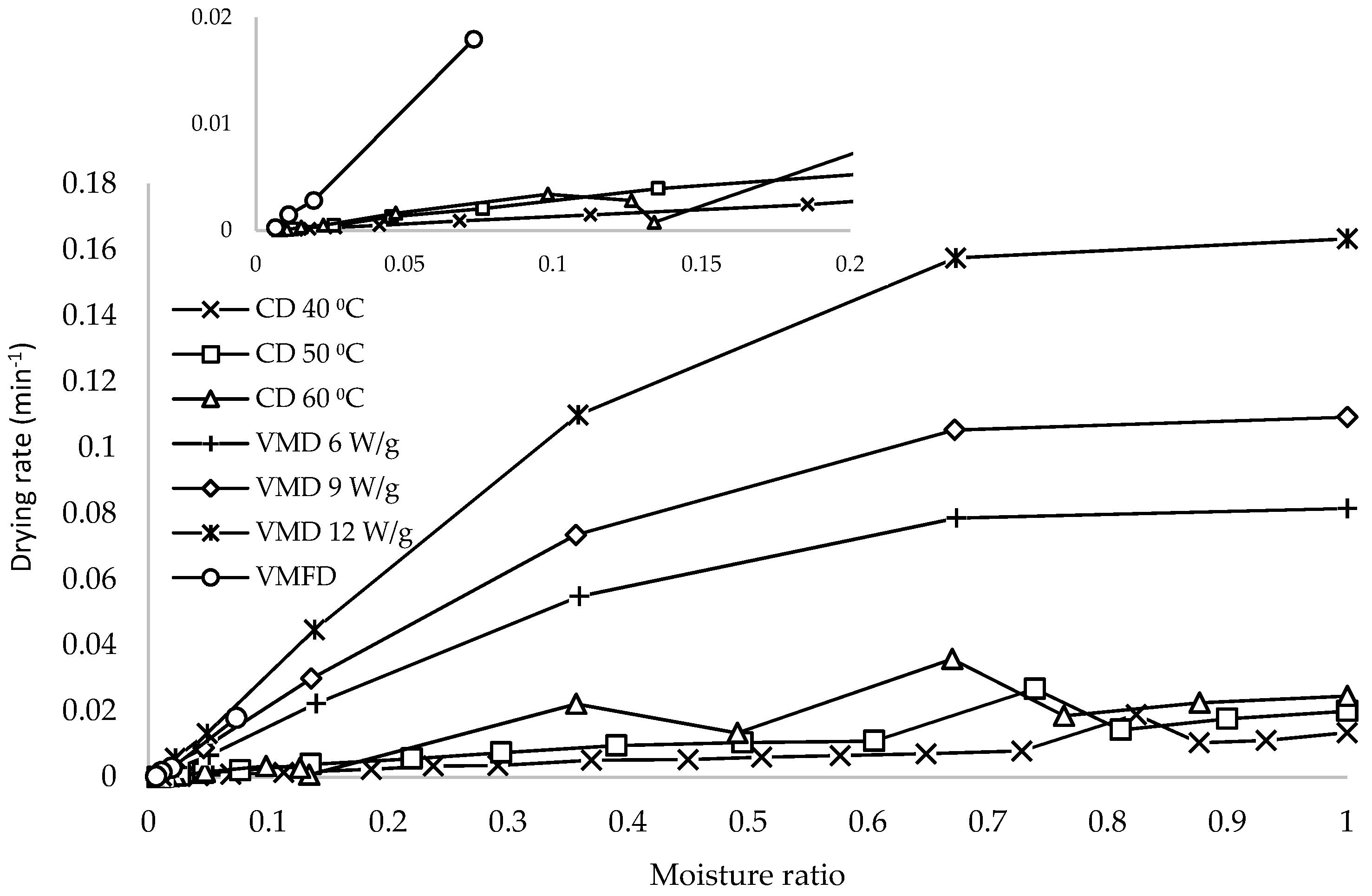
The relationship between drying rate and moisture ratio is shown in
Figure 2. The drying rate of VMD was the highest, which was attributed to the volumetric heating of microwave drying. Microwaves penetrate inner regions of leaf tissue, thereby producing heat by the absorption of microwave energy by water molecules. Increased positive steam pressure in leaf structure pushes vapor to the surface to be removed [
20]. CD recorded the lowest drying rates, but these values increased with increased temperatures. An increase from 40 to 50 °C showed an average increase of 0.003 min
−1, whereas in the case of VMD, an increase from 6 to 9 W/g showed a higher average drying rate of 0.012 min
−1. The low drying rate of CD at 50 °C can be compensated for by the introduction of VMFD, which showed a considerable improvement in drying rate at low moisture ratios.
The drying rate also decreased with decreased moisture ratio. This falling rate period is characterized by a continuous decrease in drying rate [
17]. With VMD, a higher drying rate is typically observed at the initial stage as moisture content was initially higher in leaves, and this finding corresponded to a higher dielectric constant. Larger microwave radiation was absorbed by the sample, generating more heat, thereby increasing the drying rate. As drying progressed, the reduced amount of moisture reduced the microwave absorption, resulting in decreased drying rates.
2.3. Antioxidant Activity and Total Phenolic Content Analysis
Analysis of antioxidant activity showed that the highest antioxidant activity was observed for VMD 6 W/g samples 2,2′-Azinobis (3-ethylbenzthiazoline-6-sulfonic acid (ABTS), 11.00 µM Trolox/100 g dw; ferric reducing antioxidant power (FRAP), 15.99 µM Trolox/100 g dw). The lowest antioxidant activity was obtained with VMD 12 W/g with an ABTS value of 6.62 µM Trolox/100 g dw, whereas the lowest FRAP value of 8.26 µM Trolox/100 g dw was shown by CD 40 °C samples (
Table 2).
Active compounds of thermally dried herbs were assumed to have undergone thermal degradation and oxidative degradation in the case of CD and CPD–VMFD, thereby leading to the lower antioxidant activities observed. The results clearly indicate that a suitable application of hot-air temperature and microwave power, during CD and VMD, have the potential in yielding high antioxidant activities. A low microwave power of 6 W/g (VMD) and a higher temperature of 60 °C in CD were beneficial in yielding high antioxidant activities.
The intensity of drying treatment also affected antioxidant activity. The increase in temperature of CD, from 40 to 60 °C, shortened the drying length within 360 to 180 min. At 50 and 60 °C, the degradation of heat-sensitive compounds was considerably lower compared to CD at 40 °C, which was confirmed by relatively high antioxidant activity. These results indicate that the increase in temperature during CD is effective in accelerating the drying process, thereby shortening sample’s exposure to elevated drying temperature in a highly oxygenated condition. With increased microwave power of VMD, from 6 to 12 W/g, antioxidant activity decreased. The increase in microwave power from 6 to 12 W/g decreased the drying time from 28 to 21 min, which; however, did not contribute to the preservation of antioxidant activity in the same way as observed in the case of CD. Evidently, the reduction in drying time did not compensate for the degradation of the antioxidant compounds resulting from the greater amount of heat generated inside the material by increased microwave radiation.
The lengthy CD process can be shortened with microwave heating, as seen in CPD–VMFD. Results of FRAP assay showed that CPD–VMFD produced samples of higher or not significantly different antioxidant activity from samples dried exclusively with CD or VMD. The higher antioxidant activity of CPD–VMFD samples compared to CD at 40 °C (for FRAP assay) can be explained by the shortened drying time of the former. The presence of high moisture, oxygen, and heat during the lengthy drying process of CD 40 °C may have facilitated the degradation of antioxidant compounds by enzymes [
23]. As for the higher antioxidant activity of CPD–VMFD samples compared with VMD at 9 and 12 W/g, the oxygen exposure during CPD–VMFD did not cause a large reduction in antioxidant activity. Several previous studies have indicated that phenolic content and antioxidant activity increase after heat treatment of peppermint leaves,
Artemisia annua leaves [
24], and culinary herbs, namely basil, rosemary, oregano, sage, thyme, and marjoram [
25]. Wojdylo et al. suggested that polyphenols found in strawberry fruits that were exposed to oxygenated condition may have high antioxidant activity, although the increase is temporary [
26].
Unexpectedly, the total phenolic content (TPC) values of fresh samples were lower than those of dried samples. The thermal treatment may have promoted an easier release of cell constituents from plant cells [
27], as heat exerts modifications to plant tissue microstructure, thereby reducing cell integrity [
28] and allowing for the easy exit of phenolic compounds from plant cells. Dried leaves have the tendency to exhibit increased bioactivity, as reported in past studies [
29,
30]. Moisture loss in leaves during drying, an abiotic stress, may have triggered the production and accumulation of phenolic compounds, thereby increasing the overall antioxidant activity [
31]. Furthermore, the increase in antioxidant activity may have resulted from the Maillard reaction and the formation of Maillard reaction products (MRPs) with antioxidant power [
26].
Although the TPC of samples dried with CPD–VMFD was high, antioxidant activity was relatively low. This finding implies that TPC of
P. nodiflora did not correlate with the antioxidant activity. There are three major classes of chemical compounds in plants: phenolic metabolites, alkaloids, and terpenoids [
32]. Phenolic compounds are the most researched and regarded as potent antioxidants; thus, the correlation between TPC and antioxidant activity has often been made. Terpenoids have also been established to exhibit strong antioxidant properties [
33]. Therefore, in the case of
P. nodiflora, it was assumed that terpenes and terpenoids present, which accounted for 36.5% of total volatiles, may have contributed to the overall antioxidant activity.
The overall antioxidant activity of
P. nodiflora may be contributed by the major constituents in the extract and essential oil. For instance, 1-octen-3-ol, the major volatile compound (
Table 3), β-sitosterol (
Table 4) and α-linolenic acid, accounting for 48.3% of total fatty acids (
Table 5), are among the antioxidants considered potent [
34]. Terpenoids are known to exert antioxidant effect because of their ability to interact with free radicals. The presence of various terpenoids in the volatile fraction of
P. nodiflora could also contribute to the antioxidant effect. With the presence of a wide variety of constituents in the extract of
P. nodiflora, it can be assumed that the synergistic interactions between these molecules have effectively led to the observed antioxidant activities.
Eighty-three compounds were identified in
P. nodiflora leaves, amounting to 176.81 mg 100 g
−1 db of volatile compounds in fresh leaves (
Table 3).
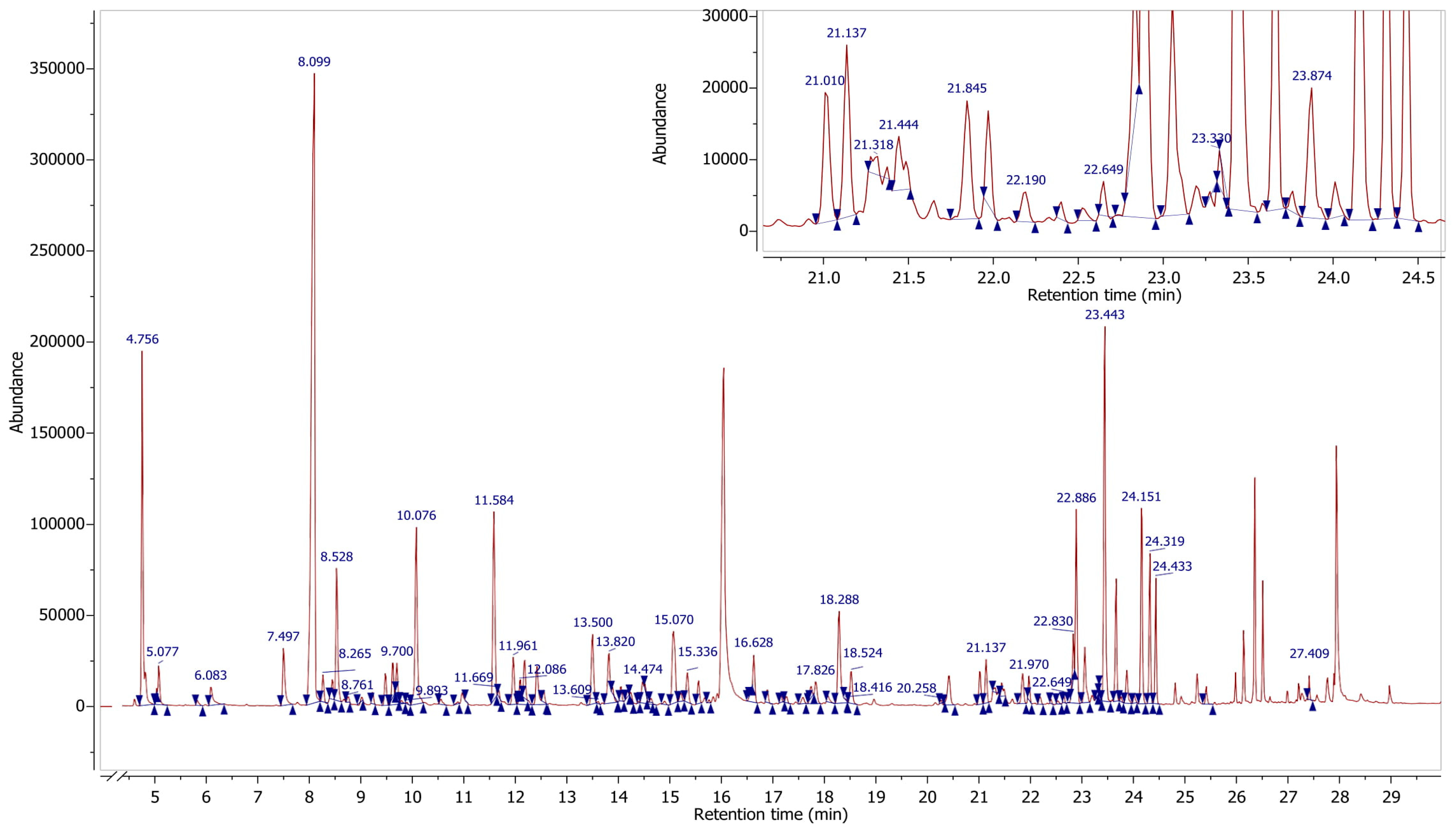
Figure 6 shows a typical chromatogram of the volatile compounds identified. The major volatiles present in fresh
P. nodilfora were 1-octen-3-ol (60.34 mg 100 g
−1 db), 2-hexenal (20.42 mg 100 g
−1 db), and α-amorphene (10.55 mg 100 g
−1 db). The volatiles present in
P. nodilfora can be are categorized under the following groups; alcohols (42.5%), sesquiterpenes (21.8%), aldehydes (16.2%), sesquiterpenoids (8.4%), monoterpenoids (3.2%), esters (3.0%), monoterpenes (2.5%) and diterpenoids (0.6%), thereby representing 98.2% of total volatiles. Other volatile compounds, phenylpropenes and ketones, were present in small concentrations.
Leaves dried using CPD–VMFD (179.02 mg 100 g
−1 db) achieved higher retention of volatiles compared with fresh leaves (176.81 mg 100 g
−1 db), but the differences in values were marginal (
Table 3). Thermal treatment of leaves during CPD–VMFD may have inactivated polyphenol oxidase (PPO), thereby reducing enzymatic degradation of volatiles [
27]. In addition, short thermal treatment, achieved by the introduction of VMFD assured that volatiles were not exposed to high temperature for an excessively long period.
Consistent with the literature, CD and VMD led to considerable losses of volatiles, evidenced by the drying of thyme [
17], basil [
13], and rosemary [
35]. In this study, CD retained a higher amount of volatiles than did VMD. A similar result was reported in the drying of rosemary [
35]. In another study, VMD was shown to retain higher total volatiles because of the fast drying process at a mild temperature [
18]. However, in this study, average temperature throughout VMD, irrespective of microwave power, was 46 °C. Slightly higher temperatures of CD at 50 and 60 °C and CPD–VMFD at 50 °C were advantageous in forming a partially dried surface layer on leaves that function as a barrier, thereby limiting the diffusion of volatiles and minimizing their loss [
36]. Furthermore, the formation of a crust layer is characteristic of products dried using CD. Parameters in CD and VMD also affect the total concentration of volatiles. A moderate temperature of 50 °C and microwave power of 9 W/g were preferable in yielding higher total volatiles. At these conditions, leaves were subjected to moderate temperature at a relatively short time. It was suggested that VMD led to higher amount of volatiles loss as microwave treatment causes a more porous structure in plant tissue because of the high internal vapor formed during the drying process. The higher porosity of tissue structure of vacuum microwave-dried leaves, compared to convective-dried leaves, could be a possible reason that a higher amount of volatiles could escape to the surrounding medium [
37,
38].
2.6. Color Analysis
Color values of fresh and dried
P. nodiflora leaves are shown in
Table 6. Parameters investigated include lightness (
L*), 0 (black) to 100 (white); greenness or redness (
a*), −60 (green) to 60 (red); and blueness or yellowness (
b*), −60 (blue) to 60 (yellow). Color changes in herbs subjected to drying are usually due to the loss of green pigments and carotenoids or enzymatic browning [
48]. However, the shaping of the color profile of the dried product depends not only on the time and drying conditions but also on the amount of residual water, which can be estimated by the water activity values.
Fresh leaves recorded the highest
L* value, whereas VMD at 12 W/g resulted in the darkest colored leaves with lowest
L* value, but these values were not significantly different from those of VMD 6 and 9 W/g and CD 40 °C. These conditions promote PPO activity, which results in browning characteristics of dried products [
49,
50]. An increase in microwave power, from 6 to 12 W/g, associated with increased maximal temperature of dried material from 52 to 57 °C, decreased the
L* value from 45.62 to 43.94. At higher microwave powers, increased amounts of absorbed energy used to vaporize water were observed, which may have been sufficiently large to increase the sample temperatures further during the falling rate period, thereby producing darker leaves [
38]. This finding was evidenced in a previous study, in which burnt spots were observed on mint leaves after VMD [
38].
The loss in green color was assumed to be related to the loss of chlorophyll [
48], as indicated by an increase in
a* values (
Table 6) which is thought to be associated with decreased a
w (
Table 7). This relationship was confirmed for CD and VMD samples because the decrease in a
w from 0.2252 to 0.1081 and from 0.2780 to 0.2353 resulted in increased
a* values from −6.35 to −5.21 and from −5.96 to −5.86, respectively. The lowest values of
a* were −6.35 and −7.02, which were found for CD 40 °C and CPD–VMFD samples, respectively. In the case of CD 40 °C, the processing temperature was the lowest, whereas combined drying CPD–VMFD underwent at a relatively short time and moderate conditions in terms of temperature and microwave power, thereby leading to lower extents of chlorophyll degradation.
Carotenoids, which tend to have yellowish color, were far better preserved in VMD 6 W/g, as indicated by a higher
b* value. High vacuum conditions and lower temperatures of VMD compared with CD methods guaranteed improved preservation of carotenoids. In the presence of oxygen, the susceptibility of carotenoids to oxidation is high [
51]; therefore, we can assume that higher oxidation occurred during CD. However, shortening drying time of CD by applying vacuum microwaves increased the value of
b* coordinate, thereby indicating the effectiveness of CPD–VMFD in preserving natural colorants.