Biomaterial Implants in Abdominal Wall Hernia Repair: A Review on the Importance of the Peritoneal Interface
Abstract
1. Introduction
2. Biomaterials in Abdominal Wall Repair
Mesh Positioning in the Abdominal Wall
3. The Abdominal Cavity
3.1. The Peritoneum
3.2. The Omentum
3.3. Host Tissue and Cell Response
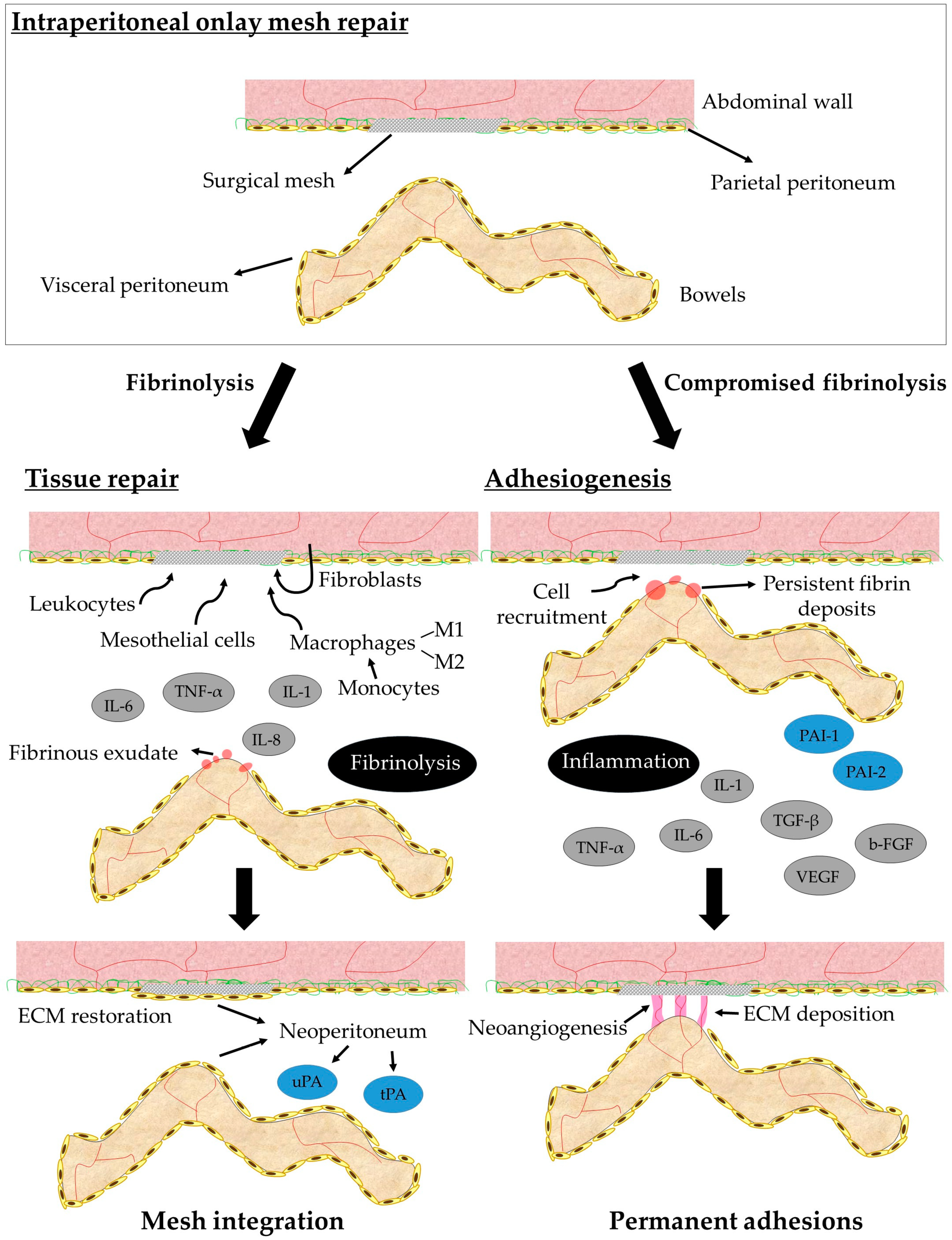
4. Peritoneal Adhesions
5. Available Biomaterials for Abdominal Surgery
5.1. Synthetic Meshes
5.1.1. Permanent Reticular Materials
5.1.2. Permanent Laminar Materials
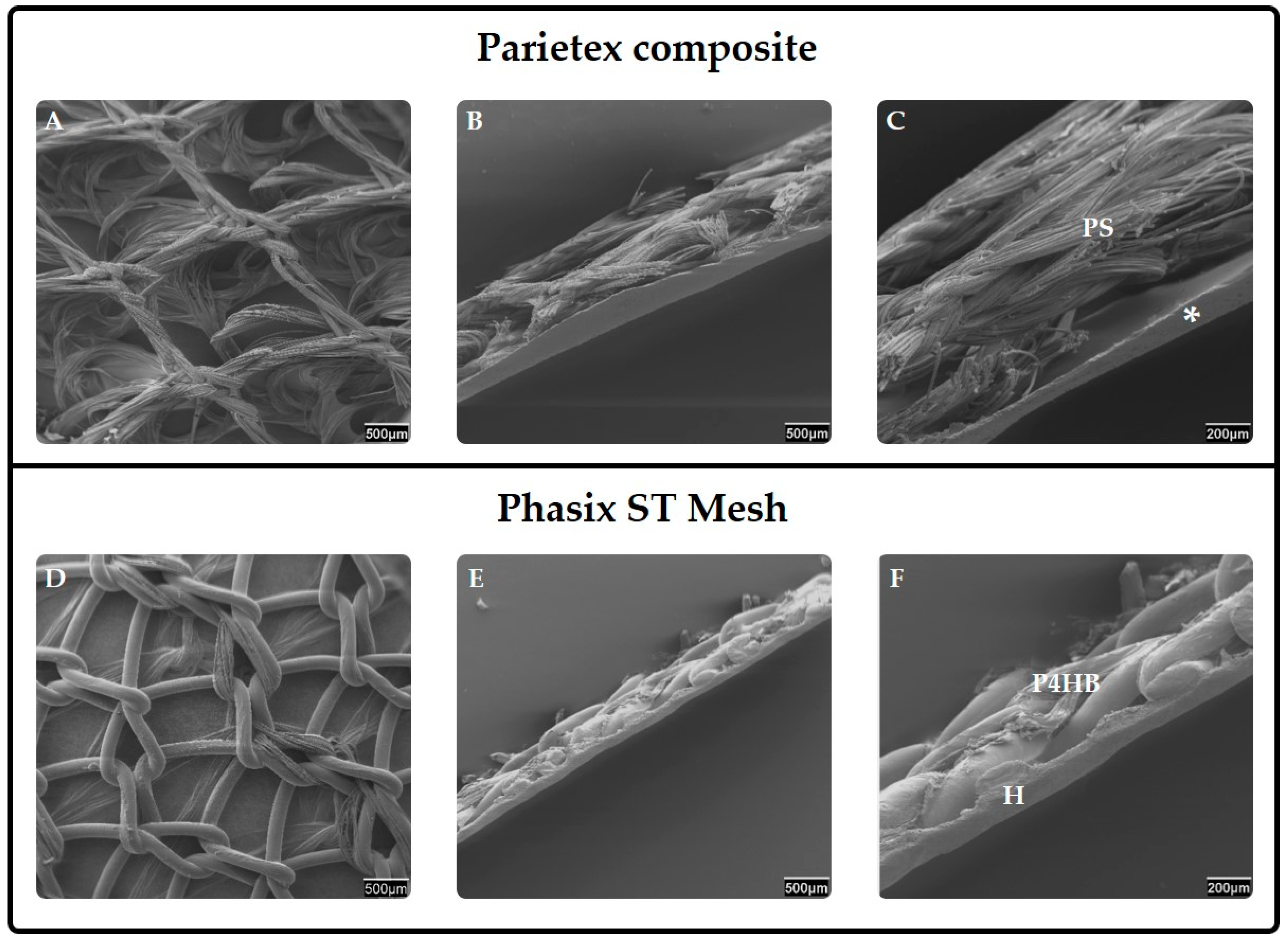
5.1.3. Composites
5.1.4. Absorbable Materials
5.1.5. Hybrid Meshes
5.2. Biological Meshes
5.3. Cell-Coated Meshes
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Williams, D.F. On the mechanisms of biocompatibility. Biomaterials 2008, 29, 2941–2953. [Google Scholar] [CrossRef] [PubMed]
- Kingsnorth, A.; LeBlanc, K. Hernias: Inguinal and incisional. Lancet 2003, 362, 1561–1571. [Google Scholar] [CrossRef]
- Hidalgo, M.; Castellón, C.; Figueroa, J.; Eymar, J.; Moreno González, E. Complicaciones de la cirugía de las hernias. Cir. Esp. 2001, 69, 217–223. [Google Scholar] [CrossRef]
- Bisgaard, T.; Bay-Nielsen, M.; Kehlet, H. Groin hernia repair in young males: Mesh or sutured repair? Hernia 2010, 14, 467–469. [Google Scholar] [CrossRef] [PubMed]
- Poulose, B.K.; Shelton, J.; Phillips, S.; Moore, D.; Nealon, W.; Penson, D.; Beck, W.; Holzman, M.D. Epidemiology and cost of ventral hernia repair: Making the case for hernia research. Hernia 2012, 16, 179–183. [Google Scholar] [CrossRef] [PubMed]
- Luijendijk, R.W.; Hop, W.C.; van den Tol, M.P.; de Lange, D.C.; Braaksma, M.M.; IJzermans, J.N.; Boelhouwer, R.U.; de Vries, B.C.; Salu, M.K.; Wereldsma, J.C.; et al. A Comparison of Suture Repair with Mesh Repair for Incisional Hernia. N. Engl. J. Med. 2000, 343, 392–398. [Google Scholar] [CrossRef] [PubMed]
- Petersson, P.; Montgomery, A.; Petersson, U. Wound dehiscence: Outcome comparison for sutured and mesh reconstructed patients. Hernia 2014, 18, 681–689. [Google Scholar] [CrossRef] [PubMed]
- Burger, J.W.A.; Luijendijk, R.W.; Hop, W.C.J.; Halm, J.A.; Verdaasdonk, E.G.G.; Jeekel, J. Long-term Follow-up of a Randomized Controlled Trial of Suture Versus Mesh Repair of Incisional Hernia. Ann. Surg. 2004, CXXII, 176–183. [Google Scholar] [CrossRef]
- Matthews, B.D.; Paton, L. Updates in Mesh and Biomaterials. Surg. Clin. N. Am. 2018, 98, 463–470. [Google Scholar] [CrossRef]
- Todros, S.; Pavan, P.G.; Natali, A.N. Synthetic surgical meshes used in abdominal wall surgery: Part I-materials and structural conformation. J. Biomed. Mater. Res. B Appl. Biomater. 2017, 105, 689–699. [Google Scholar] [CrossRef]
- Todros, S.; Pavan, P.G.; Pachera, P.; Natali, A.N. Synthetic surgical meshes used in abdominal wall surgery: Part II-Biomechanical aspects. J. Biomed. Mater. Res. B Appl. Biomater. 2017, 105, 892–903. [Google Scholar] [CrossRef] [PubMed]
- Trippoli, S.; Caccese, E.; Tulli, G.; Ipponi, P.; Marinai, C.; Messori, A. Biological meshes for abdominal hernia: Lack of evidence-based recommendations for clinical use. Int. J. Surg. 2018, 52, 278–284. [Google Scholar] [CrossRef] [PubMed]
- Guillaume, O.; Teuschl, A.H.; Gruber-Blum, S.; Fortelny, R.H.; Redl, H.; Petter-Puchner, A. Emerging Trends in Abdominal Wall Reinforcement: Bringing Bio-Functionality to Meshes. Adv. Healthc. Mater. 2015, 4, 1763–1789. [Google Scholar] [CrossRef] [PubMed]
- Savioz, D.; Ludwig, C.; Leissing, C.; Bolle, J.F.; Bühler, L.H.; Morel, P.M. Repeated macroscopic haematuria caused by intravesical migration of a preperitoneal prosthesis. Eur. J. Surg. = Acta Chir. 1997, 163, 631–632. [Google Scholar]
- Yamamoto, S.; Kubota, T.; Abe, T. A rare case of mechanical bowel obstruction caused by mesh plug migration. Hernia 2015, 19, 983–985. [Google Scholar] [CrossRef] [PubMed]
- Al-Subaie, S.; Al-Haddad, M.; Al-Yaqout, W.; Al-Hajeri, M.; Claus, C. A case of a colocutaneous fistula: A rare complication of mesh migration into the sigmoid colon after open tension-free hernia repair. Int. J. Surg. Case Rep. 2015, 14, 26–29. [Google Scholar] [CrossRef] [PubMed]
- Aziz, F.; Zaeem, M. Chronic Abdominal Pain Secondary to Mesh Erosion into Ceacum Following Incisional Hernia Repair: A Case Report and Literature Review. J. Clin. Med. Res. 2014. [Google Scholar] [CrossRef]
- Ceci, F.; D’Amore, L.; Annesi, E.; Bambi, L.; Grimaldi, M.R.; Gossetti, F.; Negro, P. Chronic anemia due to transmural e-PTFE anti-adhesive barrier mesh migration in the small bowel after open incisional hernia repair: A case report. Int. J. Surg. Case Rep. 2018, 53, 54–57. [Google Scholar] [CrossRef]
- Chuback, J.A.; Singh, R.S.; Sills, C.; Dick, L.S. Small bowel obstruction resulting from mesh plug migration after open inguinal hernia repair. Surgery 2000, 127, 475–476. [Google Scholar] [CrossRef]
- Shrivastava, A.; Gupta, A.; Gupta, A.; Shrivastava, J. Erosion of small intestine with necrotising fasciitis of over lying abdominal wall after expanded poly-tetrafluoroethylene mesh implantation: A rare complication after laparoscopic incisional hernia repair. J. Minim. Access Surg. 2013, 9, 138. [Google Scholar] [CrossRef]
- Chew, D.K.W.; Choi, L.H.; Rogers, A.M. Enterocutaneous fistula 14 years after prosthetic mesh repair of a ventral incisional hernia: A life-long risk? Surgery 2000, 127, 352–353. [Google Scholar] [CrossRef] [PubMed]
- Morin, B.; Bonnamy, C.; Maurel, J.; Samama, G.; Gignoux, M. Fistules intestinales tardives après implantation de prothèse pariétale abdominale. Annales de Chirurgie 2001, 126, 876–880. [Google Scholar] [CrossRef]
- Moussi, A.; Daldoul, S.; Bourguiba, B.; Othmani, D.; Zaouche, A. Gas gangrene of the abdominal wall due to late-onset enteric fistula after polyester mesh repair of an incisional hernia. Hernia 2012, 16, 215–217. [Google Scholar] [CrossRef] [PubMed]
- Ott, V.; Groebli, Y.; Schneider, R. Late intestinal fistula formation after incisional hernia using intraperitoneal mesh. Hernia 2005, 9, 103–104. [Google Scholar] [CrossRef] [PubMed]
- Almeida, J.A.; Franklin, M.E. Laparoscopic Repair for Inguinal Hernias: Is there a place for IPOM technique? Indications, technique and results. In Laparoscopic Ventral Hernia Repair, 1st ed.; Morales-Conde, S., Morales-Méndez, S., Eds.; Springer: Paris, France, 2003; p. 484. [Google Scholar] [CrossRef]
- Van Baal, J.O.A.M.; de Vijver, K.K.V.; Nieuwland, R.; van Noorden, C.J.F.; van Driel, W.J.; Sturk, A.; Kenter, G.G.; Rikkert, L.G.; Lok, C.A.R. The histophysiology and pathophysiology of the peritoneum. Tissue Cell 2017, 49, 95–105. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.B.; Li, M.; Li, J.C. Recent Advances in the Research of Lymphatic Stomata. Anat. Rec. (Hoboken) 2010, 293, 754–761. [Google Scholar] [CrossRef] [PubMed]
- Mutsaers, S.E. Mesothelial cells: Their structure, function and role in serosal repair. Respirology 2002, 7, 171–191. [Google Scholar] [CrossRef] [PubMed]
- Mutsaers, S.E. The mesothelial cell. Int. J. Biochem. Cell Biol. 2004, 36, 9–16. [Google Scholar] [CrossRef]
- Kastelein, A.W.; Vos, L.M.C.; de Jong, K.H.; van Baal, J.O.A.M.; Nieuwland, R.; van Noorden, C.J.F.; Roovers, J.P.W.R.; Lok, C.A.R. Embryology, anatomy, physiology and pathophysiology of the peritoneum and the peritoneal vasculature. Semin. Cell Dev. Biol. 2018. [Google Scholar] [CrossRef]
- Witz, C. Composition of the extracellular matrix of the peritoneum. J. Soc. Gynecol. Investig. 2001, 8, 299–304. [Google Scholar] [CrossRef]
- Holmdahl, L.; Ivarsson, M. The role of cytokines, coagulation, and fibrinolysis in peritoneal tissue repair. Eur. J. Surg. 1999, 165, 1012–1019. [Google Scholar] [PubMed]
- Aroeira, L.S.; Aguilera, A.; Sánchez-Tomero, J.A.; Bajo, M.A.; del Peso, G.; Jiménez-Heffernan, J.A.; Selgas, R.; López-Cabrera, M. Epithelial to mesenchymal transition and peritoneal membrane failure in peritoneal dialysis patients: Pathologic significance and potential therapeutic interventions. J. Am. Soc. Nephrol. 2007, 18, 2004–2013. [Google Scholar] [CrossRef]
- Ghellai, A.M.; Stucchi, A.F.; Chegini, N.; Ma, C.; Andry, C.D.; Kaseta, J.M.; Burns, J.W.; Skinner, K.C.; Becker, J.M. Role of transforming growth factor beta-l in peritonitis-induced adhesions. J. Gastrointest. Surg. 2000, 4, 316–323. [Google Scholar] [CrossRef]
- Fraser, D.; Wakefield, L.; Phillips, A. Independent regulation of transforming growth factor-β1 transcription and translation by glucose and platelet-derived growth factor. Am. J. Pathol. 2002, 161, 1039–1049. [Google Scholar] [CrossRef]
- Margetts, P.J.; Bonniaud, P.; Liu, L.; Hoff, C.M.; Holmes, C.J.; West-Mays, J.A.; Kelly, M.M. Transient overexpression of TGF-β1 induces epithelial mesenchymal transition in the rodent peritoneum. J. Am. Soc. Nephrol. 2005, 16, 425–436. [Google Scholar] [CrossRef] [PubMed]
- Sandoval, P.; Jiménez-Heffernan, J.A.; Guerra-Azcona, G.; Pérez-Lozano, M.L.; Rynne-Vidal, Á.; Albar-Vizcaíno, P.; Gil-Vera, F.; Martín, P.; Coronado, M.J.; Barcena, C.; Dotor, J.; et al. Mesothelial-to-mesenchymal transition in the pathogenesis of post-surgical peritoneal adhesions. J. Pathol. 2016, 239, 48–59. [Google Scholar] [CrossRef]
- Yang, A.H.; Chen, J.Y.; Lin, J.K. Myofibroblastic conversion of mesothelial cells. Kidney Int. 2003, 63, 1530–1539. [Google Scholar] [CrossRef] [PubMed]
- Bajo, M.A.; del Peso, G.; Teitelbaum, I. Peritoneal Membrane Preservation. Semin. Nephrol. 2017, 37, 77–92. [Google Scholar] [CrossRef] [PubMed]
- Aroeira, L.S.; Aguilera, A.; Selgas, R.; Ramírez-Huesca, M.; Pérez-Lozano, M.L.; Cirugeda, A.; Bajo, M.A.; del Peso, G.; Sánchez-Tomero, J.A.; Jiménez-Heffernan, J.A.; et al. Mesenchymal Conversion of Mesothelial Cells as a Mechanism Responsible for High Solute Transport Rate in Peritoneal Dialysis: Role of Vascular Endothelial Growth Factor. Am. J. Kidney Dis. 2005, 46, 938–948. [Google Scholar] [CrossRef]
- Capobianco, A.; Cottone, L.; Monno, A.; Manfredi, A.A.; Rovere-Querini, P. The peritoneum: Healing, immunity, and diseases. J. Pathol. 2017, 243, 137–147. [Google Scholar] [CrossRef]
- Vita, G.D.; Patti, R.; D’Agostino, P.; Caruso, G.; Arcara, M.; Buscemi, S.; Bonventre, S.; Ferlazzo, V.; Arcoleo, F.; Cillari, E. Cytokines and growth factors in wound drainage fluid from patients undergoing incisional hernia repair. Wound Repair Regen. 2006, 14, 259–264. [Google Scholar] [CrossRef] [PubMed]
- Mazzaferro, D.; Song, P.; Massand, S.; Jaiswal, R.; Pu, L.; Mirmanesh, M. The Omental Free Flap—A Review of Usage and Physiology. J. Reconstr. Microsurg. 2018, 34, 151–169. [Google Scholar] [CrossRef]
- Weibel, M.A.; Majno, G. Peritoneal adhesions and their relation to abdominal surgery. Am. J. Surg. 1973, 126, 345–353. [Google Scholar] [CrossRef]
- Gómez-Gil, V.; García-Honduvilla, N.; Pascual, G.; Rodríguez, M.; Buján, J.; Bellón, J.M. Peritoneal adhesion formation and reformation tracked by sequential laparoscopy: Optimizing the time point for adhesiolysis. Surgery 2010, 147, 378–391. [Google Scholar] [CrossRef] [PubMed]
- Keating, J.H.; Melidone, R.; Garcia-Polite, F. Preclinical Evaluation of Mesh Implants: The Pathologist’s Perspective. Toxicol. Pathol. 2018, 20. [Google Scholar] [CrossRef]
- Rangel-Moreno, J.; Moyron-Quiroz, J.E.; Carragher, D.M.; Kusser, K.; Hartson, L.; Moquin, A.; Randall, T.D. Omental Milky Spots Develop in the Absence of Lymphoid Tissue-Inducer Cells and Support B and T Cell Responses to Peritoneal Antigens. Immunity 2009, 30, 731–743. [Google Scholar] [CrossRef] [PubMed]
- Mebius, R.E. Lymphoid Organs for Peritoneal Cavity Immune Response: Milky Spots. Immunity 2009, 30, 670–672. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Gil, V.; Pascual, G.; Pérez-Köhler, B.; Cifuentes, A.; Buján, J.; Bellón, J.M. Involvement of transforming growth factor-β3 and betaglycan in the cytoarchitecture of postoperative omental adhesions. J. Surg. Res. 2014, 187, 699–711. [Google Scholar] [CrossRef] [PubMed]
- Isaza-Restrepo, A.; Martin-Saavedra, J.S.; Velez-Leal, J.L.; Vargas-Barato, F.; Riveros-Dueñas, R. The Peritoneum: Beyond the Tissue—A Review. Front. Physiol. 2018, 9. [Google Scholar] [CrossRef]
- Junge, K.; Binnebösel, M.; von Trotha, K.T.; Rosch, R.; Klinge, U.; Neumann, U.P.; Jansen, P.L. Mesh biocompatibility: Effects of cellular inflammation and tissue remodelling. Langenbecks Arch. Surg. 2012, 397, 255–270. [Google Scholar] [CrossRef]
- Mutsaers, S.E.; Prêle, C.M.A.; Pengelly, S.; Herrick, S.E. Mesothelial cells and peritoneal homeostasis. Fertil. Steril. 2016, 106, 1018–1024. [Google Scholar] [CrossRef] [PubMed]
- Matthews, B.D.; Mostafa, G.; Carbonell, A.M.; Joels, C.S.; Kercher, K.W.; Austin, C.; Norton, H.J.; Heniford, B.T. Evaluation of adhesion formation and host tissue response to intra-abdominal polytetrafluoroethylene mesh and composite prosthetic mesh. J. Surg. Res. 2005, 123, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Stommel, M.W.J.; Strik, C.; ten Broek, R.P.G.; de Wilt, J.H.W.; van Goor, H. Impact of Adhesiolysis on Outcome of Colorectal Surgery. Dig. Surg. 2016, 33, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Vrijland, W.W.; Jeekel, J.; van Geldorp, H.J.; Swank, D.J.; Bonjer, H.J. Abdominal adhesions: Intestinal obstruction, pain, and infertility. Surg. Endosc. 2003, 17, 1017–1022. [Google Scholar] [CrossRef] [PubMed]
- Diamond, M.P.; Freeman, M.L. Clinical implications of postsurgical adhesions. Hum. Reprod. Update 2001, 7, 567–576. [Google Scholar] [CrossRef] [PubMed]
- Menzies, D.; Ellis, H. Intestinal obstruction from adhesions—How big is the problem? Ann. R. Coll. Surg. Engl. 1990, 72, 60–63. [Google Scholar] [PubMed]
- Menzies, D. Postoperative adhesions: Their treatment and relevance in clinical practice. Ann. R. Coll. Surg. Engl. 1993, 75, 147–153. [Google Scholar]
- Moris, D.; Chakedis, J.; Rahnemai-Azar, A.A.; Wilson, A.; Hennessy, M.M.; Athanasiou, A.; Beal, E.W.; Argyrou, C.; Felekouras, E.; Pawlik, T.M. Postoperative Abdominal Adhesions: Clinical Significance and Advances in Prevention and Management. J. Gastrointest. Surg. 2017, 21, 1713–1722. [Google Scholar] [CrossRef]
- Turza, K.C.; Butler, C.E. Adhesions and Meshes. Plast. Reconstr. Surg. 2012, 130, 206S–213S. [Google Scholar] [CrossRef]
- Ergul, E.; Korukluoglu, B. Peritoneal adhesions: Facing the enemy. Int. J. Surg. 2008, 6, 253–260. [Google Scholar] [CrossRef]
- Maciver, A.H.; McCall, M.; Shapiro, A.M.J. Intra-abdominal adhesions: Cellular mechanisms and strategies for prevention. Int. J. Surg. 2011, 9, 589–594. [Google Scholar] [CrossRef] [PubMed]
- Bellón, J.M.; Contreras, L.A.; Buján, J.; Jurado, F. Effect of phosphatidylcholine on the process of peritoneal adhesion following implantation of a polypropylene mesh prosthesis. Biomaterials 1996, 17, 1369–1372. [Google Scholar] [CrossRef]
- Diamond, M.P.; Daniell, J.F.; Feste, J.; Surrey, M.W.; McLaughlin, D.S.; Friedman, S.; Vaughn, W.K.; Martin, D.C. Adhesion reformation and de novo adhesion formation after reproductive pelvic surgery. Fertil. Steril. 1987, 47, 864–866. [Google Scholar] [CrossRef]
- Zuhlke, H.V.; Lorenz, E.M.; Straub, E.M.; Savvas, V. Pathophysiology and Classification of Adhesions. In Langenbecks Archiv für Chirurgie Supplement II, Verhandlungen der Deutschen Gesellschaft für Chirurgie Deutsche Gesellschaft für Chirurgie Kongress; Springer: Berlin, Germany, 1990; Volume 190, pp. 1009–1016. [Google Scholar] [CrossRef]
- Coccolini, F.; Ansaloni, L.; Manfredi, R.; Campanati, L.; Poiasina, E.; Bertoli, P.; Capponi, M.G.; Sartelli, M.; Saverio, S.D.; Cucchi, M.; et al. Peritoneal adhesion index (PAI): Proposal of a score for the “ignored iceberg” of medicine and surgery. World J. Emerg. Surg. 2013, 8, 6. [Google Scholar] [CrossRef] [PubMed]
- Rossi, L.F.; Trindade, M.R.M.; D‘Acampora, A.J.; Meurer, L. Peritoneal adhesions type I, III and total collagen on polypropylene and coated polypropylene meshes: Experimental study in rats. Arq. Bras. Cir. Dig. 2017, 30, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Deeken, C.R.; Lake, S.P. Mechanical properties of the abdominal wall and biomaterials utilized for hernia repair. J. Mech. Behav. Biomed. Mater. 2017, 74, 411–427. [Google Scholar] [CrossRef] [PubMed]
- Baylón, K.; Rodríguez-Camarillo, P.; Elías-Zúñiga, A.; Díaz-Elizondo, J.A.; Gilkerson, R.; Lozano, K. Past, Present and Future of Surgical Meshes: A Review. Membranes 2017, 7. [Google Scholar] [CrossRef]
- Usher, F.; Fries, J.; Ochsner, J.; Tuttle, L.D. Marlex mesh, a new plastic mesh for replacing tissue defects: II. Clinical studies. AMA Arch. Surg. 1959, 78, 138–145. [Google Scholar] [CrossRef]
- Usher, F.C. Hernia repair with knitted polypropylene mesh. Surg. Gynecol. Obstet. 1963, 117, 239–240. [Google Scholar]
- Stoppa, R.; Petit, J.; Henry, X. Unsutured Dacron prosthesis in groin hernias. Int. Surg. 1975, 60, 411–412. [Google Scholar]
- Brown, R.H.; Subramanian, A.; Hwang, C.S.; Chang, S.; Awad, S.S. Comparison of infectious complications with synthetic mesh in ventral hernia repair. Am. J. Surg. 2013, 205, 182–187. [Google Scholar] [CrossRef] [PubMed]
- Klinge, U.; Klosterhalfen, B.; Müller, M.; Öttinger, A.P.; Schumpelick, V. Shrinking of polypropylene mesh in vivo: An experimental study in dogs. Eur. J. Surg. 1998, 164, 965–969. [Google Scholar] [CrossRef] [PubMed]
- Bauer, JJ.; Salky, BA.; Gelernt, IM.; Kreel, I. Repair of Large Abdominal Wall Defects with Expanded Polytetrafluoroethylene (PTFE). Ann. Surg. 1987, 206, 765–769. [Google Scholar] [CrossRef]
- Read, R.C. Milestones in the history of hernia surgery: Prosthetic repair. Hernia 2004, 8, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Bellón, J.M. Mallas y cirugía. ¿Cuáles y cuándo? Cir. Esp. 2003, 74, 1–3. [Google Scholar] [CrossRef]
- Byrd, J.F.; Agee, N.; Nguyen, P.H.; Heath, J.J.; Lau, K.N.; Mckillop, I.H.; Sindram, D.; Martinie, J.B.; Iannitti, D.A. Evaluation of Composite Mesh for Ventral Hernia Repair. JSLS J. Soc. Laparoendosc. Surg. Soc. Laparoendosc. Surg. 2011, 15, 298–304. [Google Scholar] [CrossRef] [PubMed]
- Diaz, J.J.; Gray, B.; Dobson, J.; Grogan, E.; May, A.; Miller, R.; Guy, J.; O’Neill, P.; Morris, J.J. Repair of giant abdominal hernias: Does the type of prosthesis matter? Am. Surg. 2004, 70, 396–402. [Google Scholar]
- Bellón, J.M.; Jurado, F.; García-Honduvilla, N.; López, R.; Carrera-San Martín, A.; Buján, J. The structure of a biomaterial rather than its chemical composition modulates the repair process at the peritoneal level. Am. J. Surg. 2002, 184, 154–159. [Google Scholar] [CrossRef]
- Eriksen, J.R.; Gögenur, I.; Rosenberg, J. Choice of mesh for laparoscopic ventral hernia repair. Hernia 2007, 11, 481–492. [Google Scholar] [CrossRef]
- Deeken, C.R.; Faucher, K.M.; Matthews, B.D. A review of the composition, characteristics, and effectiveness of barrier mesh prostheses utilized for laparoscopic ventral hernia repair. Surg. Endosc. 2012, 26, 566–575. [Google Scholar] [CrossRef]
- Koehler, R.H.; Begos, D.; Berger, D.; Carey, S.; LeBlanc, K.; Park, A.; Ramshaw, B.; Smoot, R.; Voeller, G. Minimal adhesions to ePTFE mesh after laparoscopic ventral incisional hernia repair: Reoperative findings in 65 cases. Zentralblatt für Chirurgie 2003, 128, 625–630. [Google Scholar] [CrossRef] [PubMed]
- Wassenaar, E.B.; Schoenmaeckers, E.J.P.; Raymakers, J.T.F.J.; Rakic, S. Subsequent abdominal surgery after laparoscopic ventral and incisional hernia repair with an expanded polytetrafluoroethylene mesh: A single institution experience with 72 reoperations. Hernia 2010, 14, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Bellón, J.M.; Serrano, N.; Rodríguez, M.; García-Honduvilla, N.; Pascual, G.; Buján, J. Composite prostheses used to repair abdominal wall defects: Physical or chemical adhesion barriers? J. Biomed. Mater. Res. B Appl. Biomater. 2005, 74B, 718–724. [Google Scholar] [CrossRef] [PubMed]
- Schreinemacher, M.H.F.; Emans, P.J.; Gijbels, M.J.J.; Greve, J.W.M.; Beets, G.L.; Bouvy, N.D. Degradation of mesh coatings and intraperitoneal adhesion formation in an experimental model. Br. J. Surg. 2009, 96, 305–313. [Google Scholar] [CrossRef] [PubMed]
- Robinson, T.N.; Clarke, J.H.; Schoen, J.; Walsh, M.D. Major mesh-related complications following hernia repair. Surg. Endosc. 2005, 19, 1556–1560. [Google Scholar] [CrossRef] [PubMed]
- Greenawalt, K.E.; Butler, T.J.; Rowe, E.A.; Finneral, A.C.; Garlick, D.S.; Burns, J.W. Evaluation of Sepramesh Biosurgical Composite in a Rabbit Hernia Repair Model. J. Surg. Res. 2000, 94, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Bohmer, R.; Byrne, P.; Maddern, G. A peeling mesh. Hernia 2002, 6, 86–87. [Google Scholar] [CrossRef]
- Konerding, M.A.; Chantereau, P.; Delventhal, V.; Holste, J.L.; Ackermann, M. Biomechanical and histological evaluation of abdominal wall compliance with intraperitoneal onlay mesh implants in rabbits: A comparison of six different state-of-the-art meshes. Med. Eng. Phys. 2012, 34, 806–816. [Google Scholar] [CrossRef]
- Judge, T.W.; Parker, D.M.; Dinsmore, R.C. Abdominal Wall Hernia Repair: A Comparison of Sepramesh and Parietex Composite Mesh in a Rabbit Hernia Model. J. Am. Coll. Surg. 2007, 204, 276–281. [Google Scholar] [CrossRef]
- Bittner, J.G.; El-Hayek, K.; Strong, A.T.; LaPinska, M.P.; Yoo, J.S.; Pauli, E.M.; Kroh, M. First human use of hybrid synthetic/biologic mesh in ventral hernia repair: A multicenter trial. Surg. Endosc. 2018, 32, 1123–1130. [Google Scholar] [CrossRef]
- Abid, S.; El-Hayek, K. Which mesh or graft? Prosthetic devices for abdominal wall reconstruction. Br. J. Hosp. Med. (Lond.) 2016, 77, 157–161. [Google Scholar] [CrossRef] [PubMed]
- Bellón, J.M.; Rodríguez, M.; García-Honduvilla, N.; Pascual, G.; Gómez Gil, V.; Buján, J. Peritoneal Effects of Prosthetic Meshes Used to Repair Abdominal Wall Defects: Monitoring Adhesions by Sequential Laparoscopy. J. Laparoendosc. Adv. Surg. Tech. A 2007, 17, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Jasbon, F.; Norrby, J.; Ivarsson, M.L.; Björck, S. Inguinal hernia repair using a synthetic long-term resorbable mesh: Results from a 3-year prospective safety and performance study. Hernia 2014, 18, 723–730. [Google Scholar] [CrossRef] [PubMed]
- De Vries Reilingh, T.S.; van Goor, H.; Koppe, M.J.; Bodegom, M.E.; Hendriks, T.; Bleichrodt, R.P. Interposition of Polyglactin Mesh Does Not Prevent Adhesion Formation Between Viscera and Polypropylene Mesh. J. Surg. Res. 2007, 140, 27–30. [Google Scholar] [CrossRef] [PubMed]
- Tyrell, J.; Silberman, H.; Chandrasoma, P.; Niland, J.; Shull, J. Absorbable Versus Permanent Mesh in Abdominal Operations. Surg. Gynecol. Obstet. 1989, 168, 227–232. [Google Scholar] [PubMed]
- Junge, K.; Rosch, R.; Krones, C.J.; Klinge, U.; Mertens, P.R.; Lynen, P.; Schumpelick, V.; Klosterhalfen, B. Influence of polyglecaprone 25 (Monocryl) supplementation on the biocompatibility of a polypropylene mesh for hernia repair. Hernia 2005, 9, 212–217. [Google Scholar] [CrossRef] [PubMed]
- Fortelny, R.H.; Petter-Puchner, A.H.; Glaser, K.S.; Offner, F.; Benesch, T.; Rohr, M. Adverse effects of polyvinylidene fluoride-coated polypropylene mesh used for laparoscopic intraperitoneal onlay repair of incisional hernia. Br. J. Surg. 2010, 97, 1140–1145. [Google Scholar] [CrossRef]
- Jamry, A.; Jałyński, M.; Piskorz, L.; Brocki, M. Assessment of adhesion formation after laparoscopic intraperitoneal implantation of Dynamesh IPOM mesh. Arch. Med. Sci. 2013, 9, 487–492. [Google Scholar] [CrossRef]
- D’Amore, L.; Ceci, F.; Mattia, S.; Fabbi, M.; Negro, P.; Gossetti, F. Adhesion prevention in ventral hernia repair: An experimental study comparing three lightweight porous meshes recommended for intraperitoneal use. Hernia 2017, 21, 115–123. [Google Scholar] [CrossRef]
- Burger, J.W.; Halm, J.A.; Wijsmuller, A.R.; ten Raa, S.; Jeekel, J. Evaluation of new prosthetic meshes for ventral hernia repair. Surg. Endosc. 2006, 20, 1320–1325. [Google Scholar] [CrossRef]
- Franklin, M.; Gonzalez, J.; Michaelson, R.; Glass, J.; Chock, D. Preliminary experience with new bioactive prosthetic material for repair of hernias in infected fields. Hernia 2002, 6, 171–174. [Google Scholar] [CrossRef] [PubMed]
- Chand, B.; Indeck, M.; Needleman, B.; Finnegan, M.; Van Sickle, KR.; Ystgaard, B.; Gossetti, F.; Pullan, R.D.; Giordano, P.; McKinley, A. A retrospective study evaluating the use of PermacolTM surgical implant in incisional and ventral hernia repair. Int. J. Surg. 2014, 12, 296–303. [Google Scholar] [CrossRef] [PubMed]
- Pavan, P.G.; Pachera, P.; Todros, S. Mechanical characterization of animal derived grafts for surgical implantation. J. Mech. Med. Biol. 2016, 16. [Google Scholar] [CrossRef]
- Chavarriaga, L.F.; Lin, E.; Losken, A.; Cook, M.W.; Jeansonne, L.O.; White, B.C.; Sweeney, J.F.; Galloway, J.R.; Davis, S.S., Jr. Management of complex abdominal wall defects using acellular porcine dermal collagen. Am. Surg. 2010, 76, 96–100. [Google Scholar] [PubMed]
- Pascual, G.; Pérez-Köhler, B.; Rodríguez, M.; Sotomayor, S.; Bellón, J.M. Postimplantation host tissue response and biodegradation of biologic versus polymer meshes implanted in an intraperitoneal position. Surg. Endoc. 2014, 28, 559–569. [Google Scholar] [CrossRef] [PubMed]
- Beale, E.W.; Hoxworth, R.E.; Livingston, E.H.; Trussler, A.P. The role of biologic mesh in abdominal wall reconstruction: A systematic review of the current literature. Am. J. Surg. 2012, 204, 510–517. [Google Scholar] [CrossRef] [PubMed]
- Dunn, R.M. Cross-Linking in Biomaterials. Plast. Reconstr. Surg. 2012, 130, 18S–26S. [Google Scholar] [CrossRef] [PubMed]
- Novitsky, Y. Biology of Biological Meshes Used in Hernia Repair. Surg. Clin. N. Am. 2013, 93, 1211–1215. [Google Scholar] [CrossRef] [PubMed]
- Carbonell, A.M.; Criss, C.N.; Cobb, W.S.; Novitsky, Y.W.; Rosen, M.J. Outcomes of Synthetic Mesh in Contaminated Ventral Hernia Repairs. J. Am. Coll. Surg. 2013, 217, 991–998. [Google Scholar] [CrossRef] [PubMed]
- Bellows, C.F.; Smith, A.; Malsbury, J.; Helton, W.S. Repair of incisional hernias with biological prosthesis: A systematic review of current evidence. Am. J. Surg. 2013, 205, 85–101. [Google Scholar] [CrossRef] [PubMed]
- Slater, N.J.; van der Kolk, M.; Hendriks, T.; van Goor, H.; Bleichrodt, R.P. Biologic grafts for ventral hernia repair: A systematic review. Am. J. Surg. 2013, 205, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Rosen, M.J.; DeNoto, G.; Itani, K.M.F.; Butler, C.; Vargo, D.; Smiell, J.; Rutan, R. Evaluation of surgical outcomes of retro-rectus versus intraperitoneal reinforcement with bio-prosthetic mesh in the repair of contaminated ventral hernias. Hernia 2013, 17, 31–35. [Google Scholar] [CrossRef] [PubMed]
- Bellón, J.M.; Rodríguez, M.; Gómez-Gil, V.; Sotomayor, S.; Buján, J.; Pascual, G. Postimplant intraperitoneal behavior of collagen-based meshes followed by laparoscopy. Surg. Endosc. 2012, 26, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Majumder, A.; Gao, Y.; Sadava, E.E.; Anderson, J.M.; Novitsky, Y.W. Cell-coating affects tissue integration of synthetic and biologic meshes: Comparative analysis of the onlay and underlay mesh positioning in rats. Surg. Endosc. 2016, 30, 4445–4453. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Krpata, D.M.; Criss, C.N.; Liu, L.; Posielski, N.; Rosen, M.J.; Novitsky, Y.W. Effects of mesenchymal stem cell and fibroblast coating on immunogenic potential of prosthetic meshes in vitro. Surg. Endosc. 2014, 28, 2357–2367. [Google Scholar] [CrossRef] [PubMed]
- Dolce, C.J.; Stefanidis, D.; Keller, J.E.; Walters, K.C.; Newcomb, W.L.; Heath, J.J.; Norton, H.J.; Lincourt, A.E.; Kercher, K.W.; Heniford, B.T. Pushing the envelope in biomaterial research: Initial results of prosthetic coating with stem cells in a rat model. Surg. Endosc. 2010, 24, 2687–2693. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Zhang, Z.; Wang, J.; Yin, P.; Zhou, J.; Zhen, M.; Cui, W.; Xu, G.; Yang, D.; Liu, Z. Abdominal Hernia Repair with a Decellularized Dermal Scaffold Seeded with Autologous Bone Marrow-Derived Mesenchymal Stem Cells. Artif. Organs 2012, 36, 247–255. [Google Scholar] [CrossRef]
- Cheng, H.; Zhang, Y.; Zhang, B.; Cheng, J.; Wang, W.; Tang, X.; Teng, P.; Li, Y. Biocompatibility of polypropylene mesh scaffold with adipose-derived stem cells. Exp. Ther. Med. 2017, 13, 2922–2926. [Google Scholar] [CrossRef]
- Petter-Puchner, A.H.; Fortelny, R.H.; Gruber-Blum, S.; Redl, H.; Dietz, U. The future of stem cell therapy in hernia and abdominal wall repair. Hernia 2015, 19, 25–31. [Google Scholar] [CrossRef]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gómez-Gil, V.; Pascual, G.; Bellón, J.M. Biomaterial Implants in Abdominal Wall Hernia Repair: A Review on the Importance of the Peritoneal Interface. Processes 2019, 7, 105. https://doi.org/10.3390/pr7020105
Gómez-Gil V, Pascual G, Bellón JM. Biomaterial Implants in Abdominal Wall Hernia Repair: A Review on the Importance of the Peritoneal Interface. Processes. 2019; 7(2):105. https://doi.org/10.3390/pr7020105
Chicago/Turabian StyleGómez-Gil, Verónica, Gemma Pascual, and Juan M. Bellón. 2019. "Biomaterial Implants in Abdominal Wall Hernia Repair: A Review on the Importance of the Peritoneal Interface" Processes 7, no. 2: 105. https://doi.org/10.3390/pr7020105
APA StyleGómez-Gil, V., Pascual, G., & Bellón, J. M. (2019). Biomaterial Implants in Abdominal Wall Hernia Repair: A Review on the Importance of the Peritoneal Interface. Processes, 7(2), 105. https://doi.org/10.3390/pr7020105