Plant and Biomass Extraction and Valorisation under Hydrodynamic Cavitation
Abstract
1. Introduction
2. Advantages of HC Extraction
3. HC Reactors: Technical Aspects
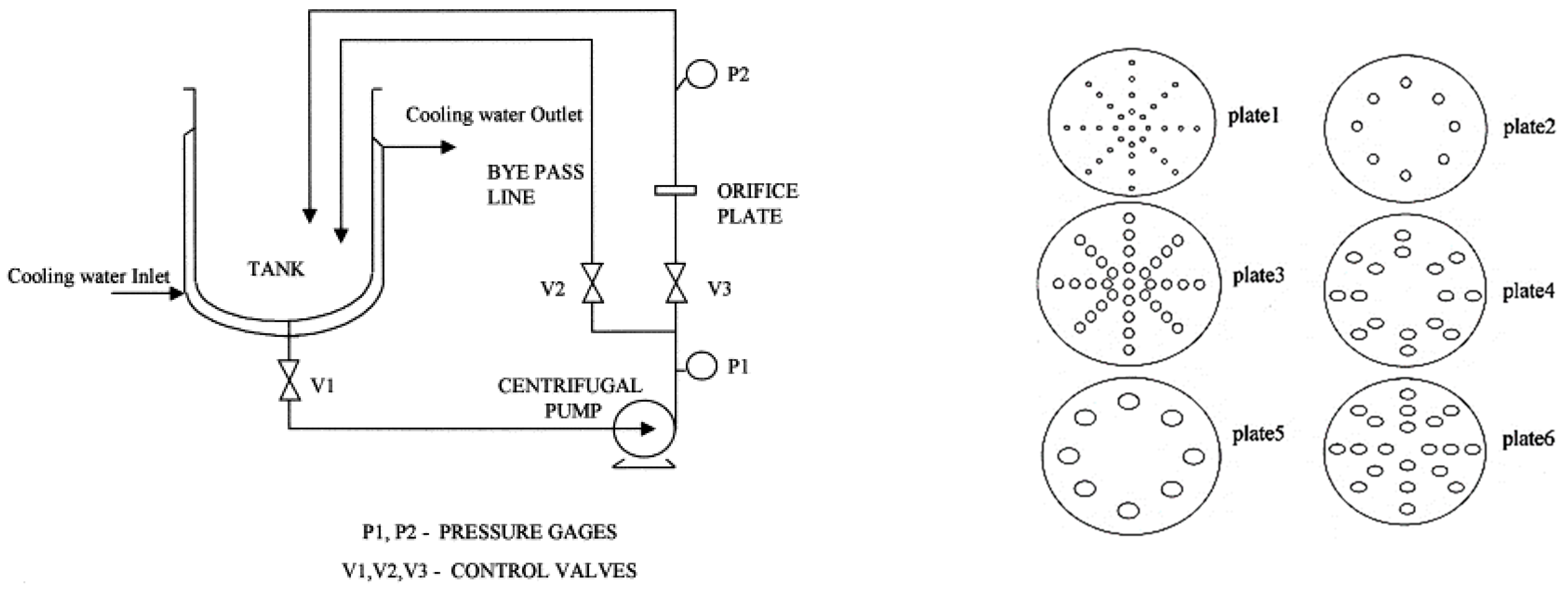
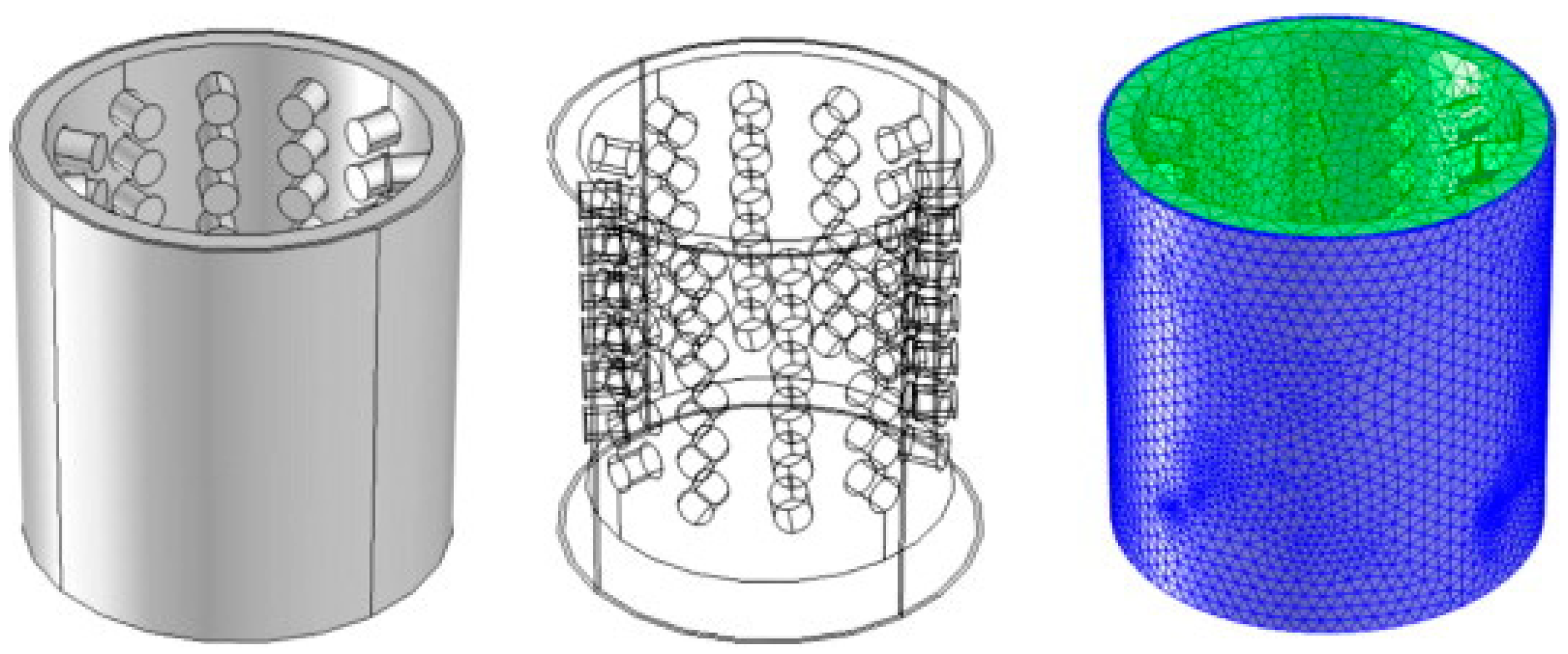
3.1. Orifice-Structure HCRs
3.1.1. Orifice Plates
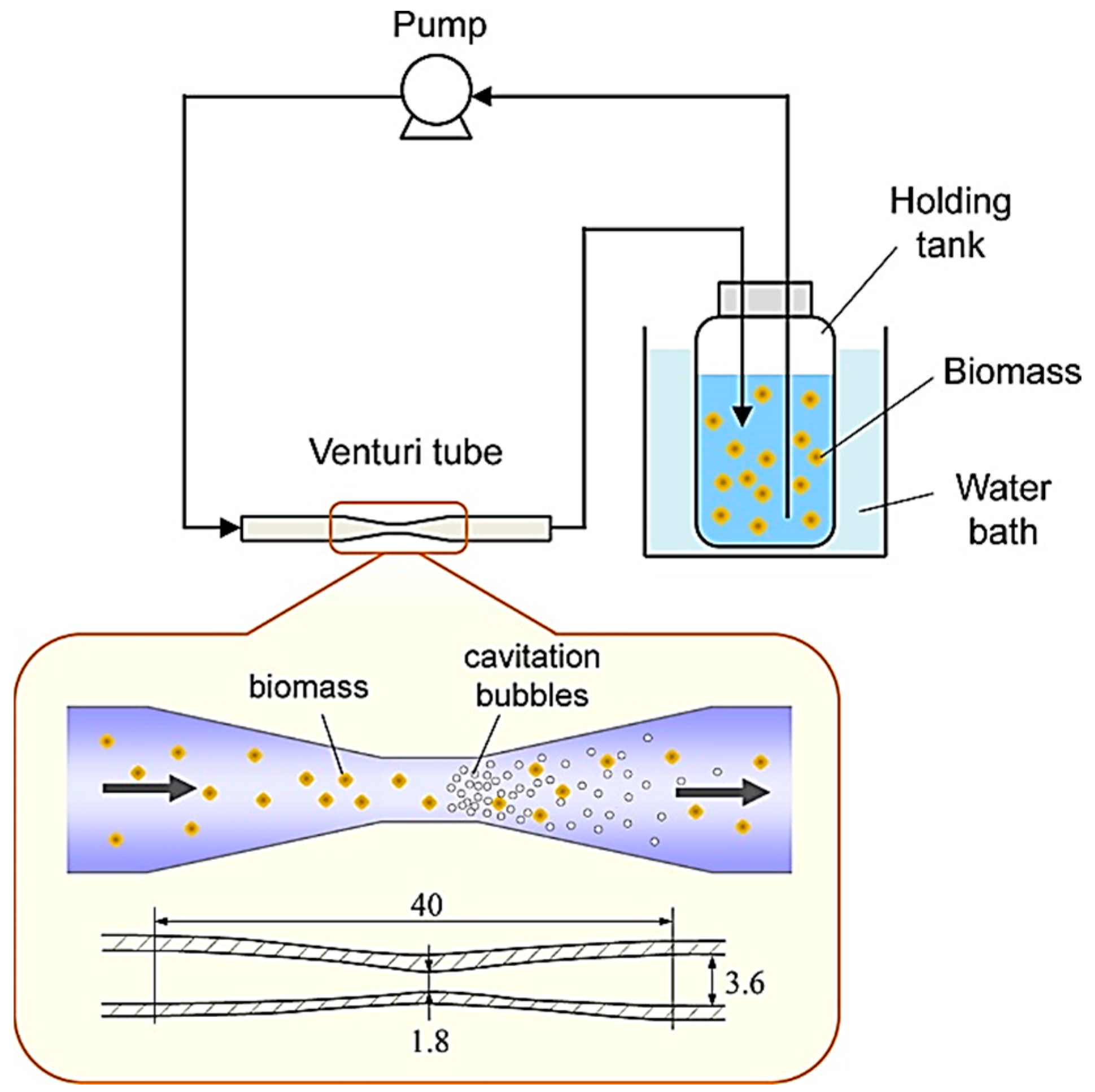
3.1.2. Venturi Tubes
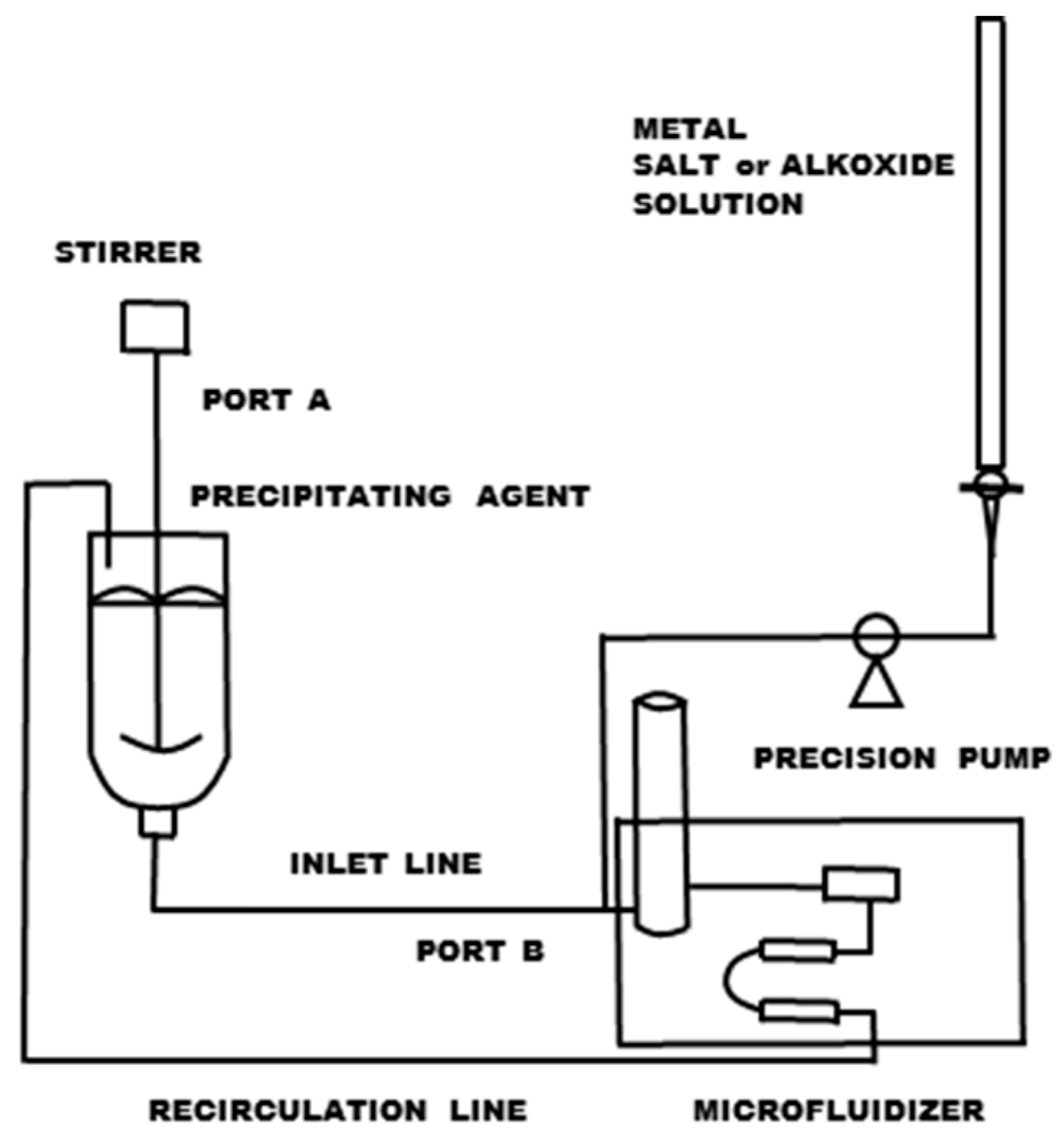
3.1.3. High-Pressure Nozzles (Microfluidizers)
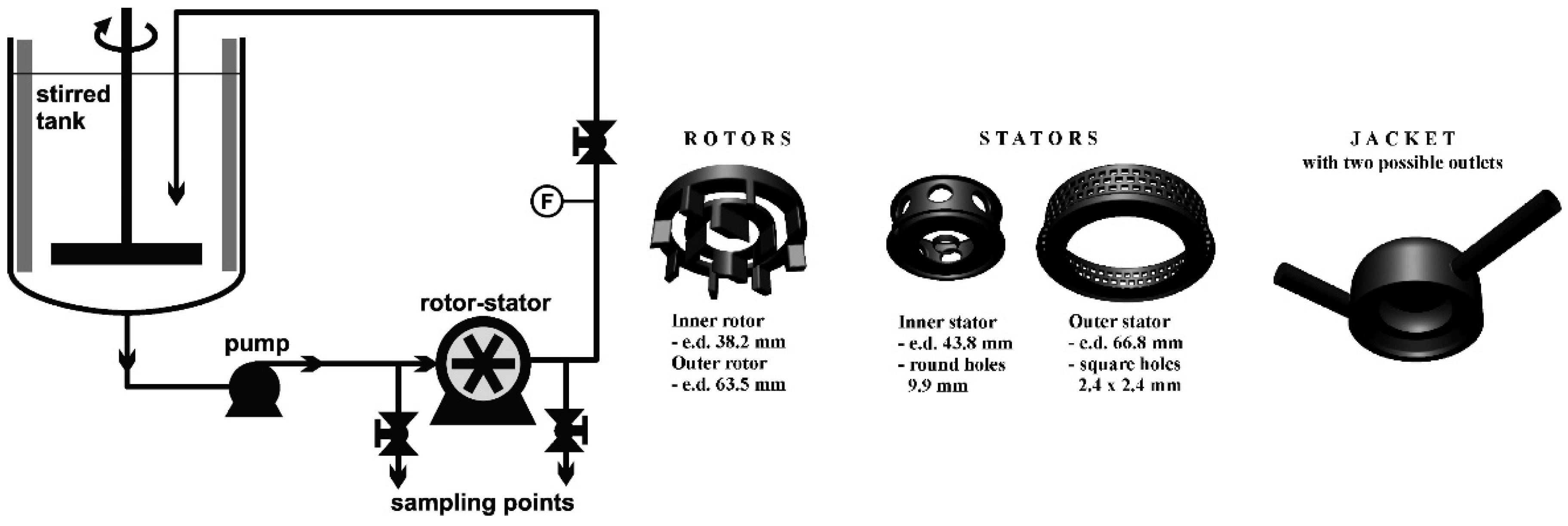
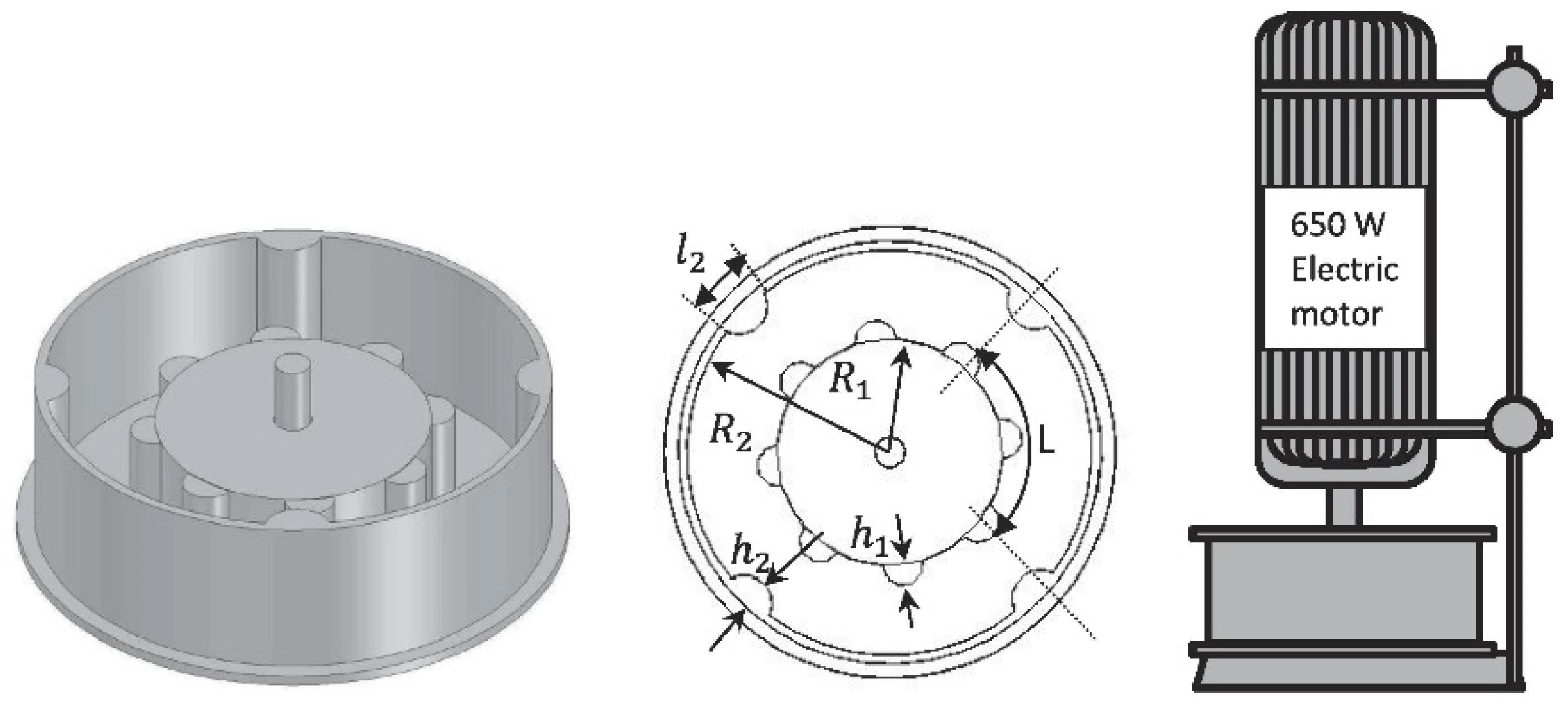
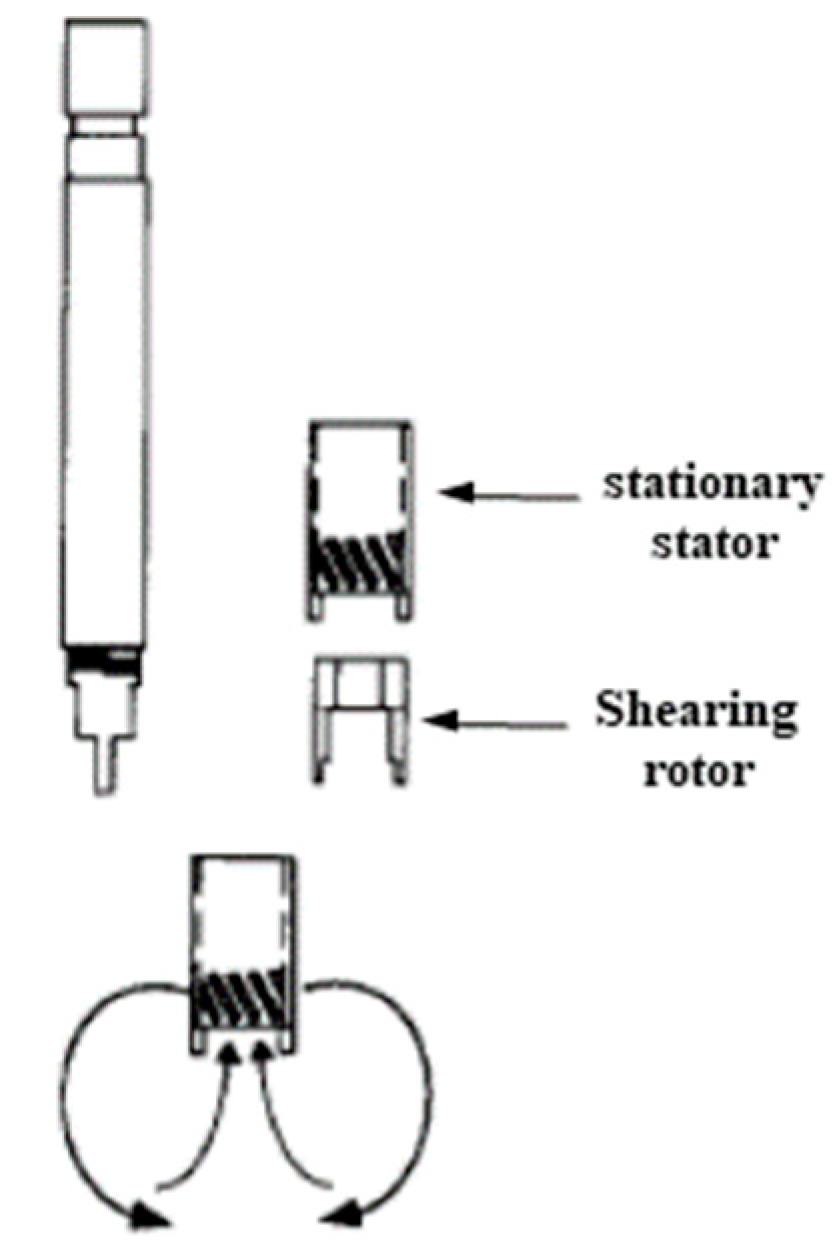
3.2. Rotor-Stator HCRs
3.2.1. Breakage of Nanoparticle Clusters
3.2.2. Microorganism Cell Disruption
3.2.3. Emulsification of Immiscible Mixtures
4. Extraction with HC Reactors
4.1. Extraction of Bioactive Compounds and Antioxidants from Plants
4.2. Lipid Extraction from Microorganisms
4.3. Delignification of Lignocellulosic Biomass
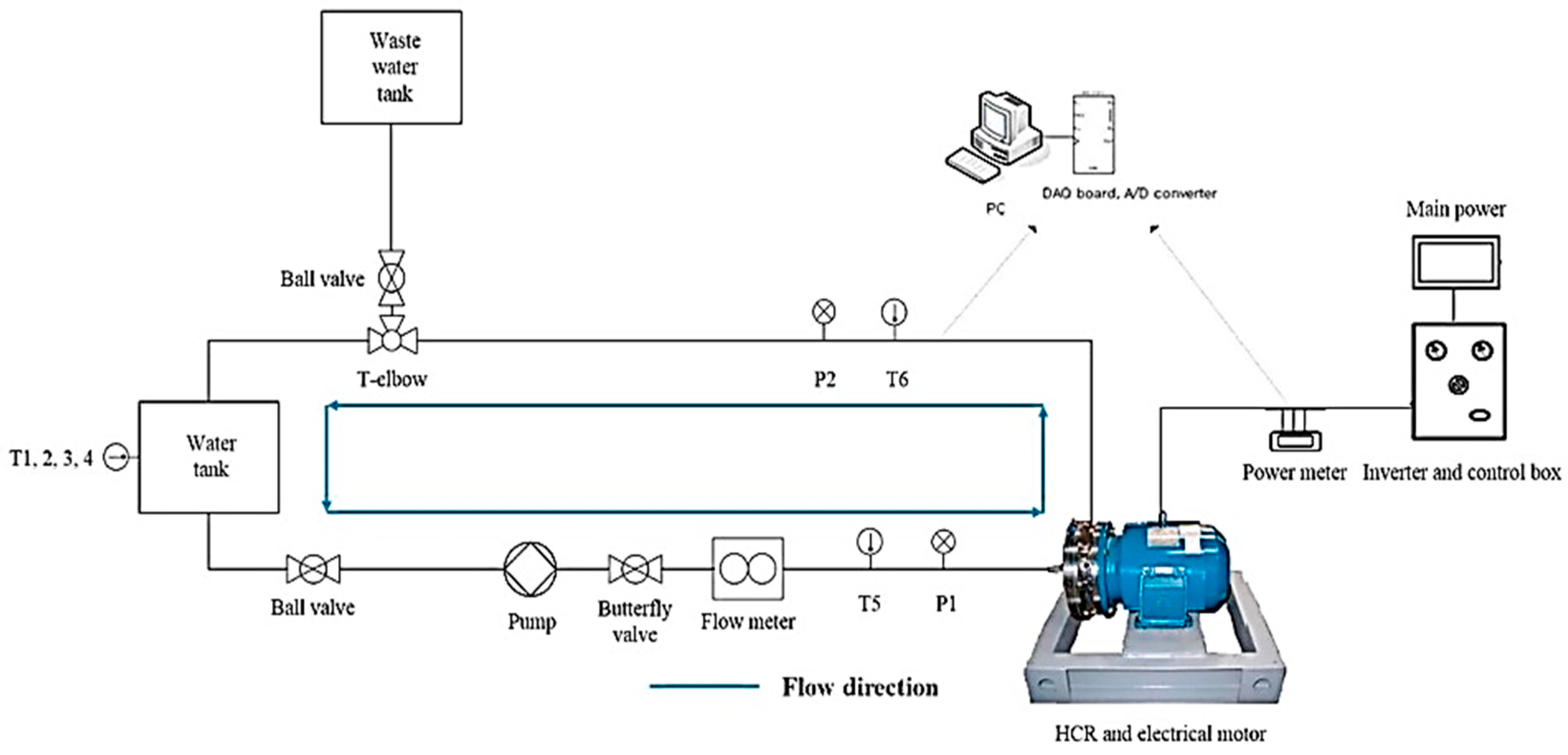
5. Case Study for Extracting Polyphenols from Basil
Author Contributions
Funding
Conflicts of Interest
References
- Joshi, S.M.; Gogate, P.R. Waste Biomass Management–A Holistic Approach. In Waste Biomass Management—A Holistic Approach; Singh, V.C., Kalia, L., Eds.; Springer: Cham, Switzerland, 2017; pp. 251–287. [Google Scholar]
- Ragauskas, A.J.; Beckham, G.T.; Biddy, M.J.; Chandra, R.; Chen, F.; Davis, M.F.; Davison, B.H.; Dixon, R.A.; Gilna, P.; Keller, M.; et al. Lignin valorization: Improving lignin processing in the biorefinery. Science 2014, 344. [Google Scholar] [CrossRef]
- Stevens, C.; Verhé, R. Renewable Bioresources: Scope and Modification for Non-Food Applications; John Wiley & Sons: New York, NY, USA, 2004. [Google Scholar]
- Grillo, G.; Boffa, L.; Binello, A.; Mantegna, S.; Cravotto, G.; Chemat, F.; Dizhbite, T.; Lauberte, L.; Telysheva, G. Cocoa bean shell waste valorisation; extraction from lab to pilot-scale cavitational reactors. Food Res. Int. 2019, 115, 200–208. [Google Scholar] [CrossRef] [PubMed]
- Albanese, L.; Bonetti, A.; D’Acqui, L.P.; Meneguzzo, F.; Zabini, F. Affordable production of antioxidant aqueous solutions by hydrodynamic cavitation processing of silver fir (Abies alba Mill.) needles. Foods 2019, 8, 65. [Google Scholar] [CrossRef] [PubMed]
- Panda, D.; Manickam, S. Cavitation technology-the future of greener extraction method: A review on the extraction of natural products and process intensification mechanism and perspectives. Appl. Sci. 2019, 9, 766. [Google Scholar] [CrossRef]
- Seo, Y.H.; Lee, I.G.; Han, J.I. Cultivation and lipid production of yeast Cryptococcus curvatus using pretreated waste active sludge supernatant. Bioresour. Technol. 2013, 135, 304–308. [Google Scholar] [CrossRef] [PubMed]
- Setyawan, M.; Mulyono, P.; Sutijan, S.; Budiman, A. Comparison of Nannochloropsis sp. cells disruption between hydrodynamic cavitation and conventional extraction. In MATEC Web of Conferences, Proceedings of the 2nd International Conference on Engineering and Technology for Sustainable Development (ICET4SD 2017), Yogyakarta, Indonesia, 13–14 September 2017; EDP Sciences: Les Ulis, France, 2018; Volume 154, p. 01023. [Google Scholar]
- Lee, A.K.; Lewis, D.M.; Ashman, P.J. Microalgal cell disruption by hydrodynamic cavitation for the production of biofuels. J. Appl. Phycol. 2015, 27, 1881–1889. [Google Scholar] [CrossRef]
- Lee, A.K.; Lewis, D.M.; Ashman, P.J. Disruption of microalgal cells for the extraction of lipids for biofuels: Processes and specific energy requirements. Biomass Bioenergy 2012, 46, 89–101. [Google Scholar] [CrossRef]
- Setyawan, M.; Budiman, A.; Mulyono, P. Sutijan Optimum extraction of algae-oil from microalgae using hydrodynamic cavitation. Int. J. Renew. Energy Res. 2018, 8, 451–458. [Google Scholar]
- Yusaf, T.; Al-Juboori, R.A. Alternative methods of microorganism disruption for agricultural applications. Appl. Energy 2014, 114, 909–923. [Google Scholar] [CrossRef]
- Qian, E.W. Research approaches to sustainable biomass systems. In Research Approaches to Sustainable Biomass Systems; Tojo, T.S., Hirasawa, T., Eds.; Academic Press: Cambridge, MA, USA, 2014; pp. 181–204. [Google Scholar]
- Binod, P.; Pandey, A. Chapter 1—Introduction. In Pretreatment of Biomass; Pandey, A., Negi, S., Binod, P., Larroche, C., Eds.; Elsevier: Amsterdam, The Netherlands, 2015; pp. 3–6. ISBN 978-0-12-800080-9. [Google Scholar]
- Ishizaki, H.; Hasumi, K. Chapter 10—Ethanol Production from Biomass. In Research Approaches to Sustainable Biomass Systems; Tojo, S., Hirasawa, T., Eds.; Academic Press: Boston, MA, USA, 2014; pp. 243–258. ISBN 978-0-12-404609-2. [Google Scholar]
- Mosier, N.; Wyman, C.; Dale, B.; Elander, R.; Lee, Y.Y.; Holtzapple, M.; Ladisch, M. Features of promising technologies for pretreatment of lignocellulosic biomass. Bioresour. Technol. 2005, 96, 673–686. [Google Scholar] [CrossRef]
- Kumar, P.; Barrett, D.M.; Delwiche, M.J.; Stroeve, P. Methods for pretreatment of lignocellulosic biomass for efficient hydrolysis and biofuel production. Ind. Eng. Chem. Res. 2009, 48, 3713–3729. [Google Scholar] [CrossRef]
- Kuna, E.; Behling, R.; Valange, S.; Chatel, G.; Colmenares, J.C. Sonocatalysis: A potential sustainable pathway for the valorization of lignocellulosic biomass and derivatives. Top. Curr. Chem. 2017, 375, 41. [Google Scholar] [CrossRef] [PubMed]
- Roohinejad, S.; Koubaa, M.; Barba, F.J.; Greiner, R.; Orlien, V.; Lebovka, N.I. Negative pressure cavitation extraction: A novel method for extraction of food bioactive compounds from plant materials. Trends Food Sci. Technol. 2016, 52, 98–108. [Google Scholar] [CrossRef]
- Cheng, H.-T.; Shen, X.-S. The technology of extracting tea polyphenols by cavitation. Mod. Chem. Ind. 2016, 36, 157–160. [Google Scholar]
- Lee, I.; Han, J.-I. Simultaneous treatment (cell disruption and lipid extraction) of wet microalgae using hydrodynamic cavitation for enhancing the lipid yield. Bioresour. Technol. 2015, 186, 246–251. [Google Scholar] [CrossRef]
- Lee, I.; Oh, Y.-K.; Han, J.-I. Design optimization of hydrodynamic cavitation for effectual lipid extraction from wet microalgae. J. Environ. Chem. Eng. 2019, 7, 102942. [Google Scholar] [CrossRef]
- Iskalieva, A.; Yimmou, B.M.; Gogate, P.R.; Horvath, M.; Horvath, P.G.; Csoka, L. Cavitation assisted delignification of wheat straw: A review. Ultrason. Sonochem. 2012, 19, 984–993. [Google Scholar] [CrossRef]
- Thangavelu, K.; Desikan, R.; Taran, O.P.; Uthandi, S. Delignification of corncob via combined hydrodynamic cavitation and enzymatic pretreatment: Process optimization by response surface methodology. Biotechnol. Biofuels 2018, 11, 203. [Google Scholar] [CrossRef]
- Badve, M.P.; Gogate, P.R.; Pandit, A.B.; Csoka, L. Hydrodynamic cavitation as a novel approach for delignification of wheat straw for paper manufacturing. Ultrason. Sonochem. 2014, 21, 162–168. [Google Scholar] [CrossRef]
- Baxi, P.B.; Pandit, A.B. Using cavitation for delignification of wood. Bioresour. Technol. 2012, 110, 697–700. [Google Scholar] [CrossRef]
- Asaithambi, N.; Singha, P.; Dwivedi, M.; Singh, S.K. Hydrodynamic cavitation and its application in food and beverage industry: A review. J. Food Process Eng. 2019, 42, e13144. [Google Scholar] [CrossRef]
- Mishra, K.P.; Gogate, P.R. Intensification of degradation of Rhodamine B using hydrodynamic cavitation in the presence of additives. Sep. Purif. Technol. 2010, 75, 385–391. [Google Scholar] [CrossRef]
- Hilares, R.T.; Ramos, L.; da Silva, S.S.; Dragone, G.; Mussatto, S.I.; Santos, J.C.D. Hydrodynamic cavitation as a strategy to enhance the efficiency of lignocellulosic biomass pretreatment. Crit. Rev. Biotechnol. 2018, 38, 483–493. [Google Scholar] [CrossRef]
- Gogate, P.R. Alternative Energy Sources for Green Chemistry; Stefanidis, A.G., Stankiewicz, A., Eds.; Royal Society of Chemistry: London, UK, 2016; pp. 126–160. [Google Scholar]
- Gogate, P.R. Hydrodynamic cavitation for food and water processing. Food Bioprocess Technol. 2011, 4, 996–1011. [Google Scholar] [CrossRef]
- Albanese, L.; Ciriminna, R.; Meneguzzo, F.; Pagliaro, M. Beer-brewing powered by controlled hydrodynamic cavitation: Theory and real-scale experiments. J. Clean. Prod. 2017, 142, 1457–1470. [Google Scholar] [CrossRef]
- Capocelli, M.; Prisciandaro, M.; Lancia, A.; Musmarra, D. Comparison between hydrodynamic and acoustic cavitation in microbial cell disruption. Chem. Eng. Trans. 2014, 38, 13–18. [Google Scholar]
- Askarian, M.; Vatani, A.; Edalat, M. Heavy oil upgrading via hydrodynamic cavitation in the presence of an appropriate hydrogen donor. J. Pet. Sci. Eng. 2017, 151, 55–61. [Google Scholar] [CrossRef]
- Gaikwad, V.; Gaikwad, S.; Ranade, V. Effect of orifice shape on water disinfection efficacy. Pollut. Res. 2016, 35, 765–771. [Google Scholar]
- Agrawal, N.; Maddikeri, G.L.; Pandit, A.B. Sustained release formulations of citronella oil nanoemulsion using cavitational techniques. Ultrason. Sonochem. 2017, 36, 367–374. [Google Scholar] [CrossRef]
- Dong, Z.; Zhao, W. Killing rate of colony count by hydrodynamic cavitation due to square multi-orifice plates. In IOP Conference Series: Earth and Environmental Science, Proceedings of the International Conference on Energy Engineering and Environmental Protection (EEEP2017), Sanya, China, 20–22 November 2017; IOP Science: Bristol, UK, 2018; Volume 121, p. 022004. [Google Scholar]
- Terán Hilares, R.; de Almeida, G.F.; Ahmed, M.A.; Antunes, F.A.F.; da Silva, S.S.; Han, J.-I.; Santos, J.C.D. Hydrodynamic cavitation as an efficient pretreatment method for lignocellulosic biomassA parametric study. Bioresour. Technol. 2017, 235, 301–308. [Google Scholar] [CrossRef]
- Sawant, S.S.; Anil, A.C.; Krishnamurthy, V.; Gaonkar, C.; Kolwalkar, J.; Khandeparker, L.; Desai, D.; Mahulkar, A.V.; Ranade, V.V.; Pandit, A.B. Effect of hydrodynamic cavitation on zooplankton: A tool for disinfection. Chem. Eng. J. 2008, 42, 320–328. [Google Scholar] [CrossRef]
- Nakashima, K.; Ebi, Y.; Shibasaki-Kitakawa, N.; Soyama, H.; Yonemoto, T. Hydrodynamic cavitation reactor for efficient pretreatment of lignocellulosic biomass. Ind. Eng. Chem. Res. 2016, 55, 1866–1871. [Google Scholar] [CrossRef]
- More, N.S.; Gogate, P.R. Intensified degumming of crude soybean oil using cavitational reactors. J. Food Eng. 2018, 218, 33–43. [Google Scholar] [CrossRef]
- Nemchin, A. New Technological cavitation-induced effects of heat and mass transfer. Heat Transf. Res. 1999, 30, 275–288. [Google Scholar] [CrossRef]
- Bałdyga, J.; Makowski, Ł.; Orciuch, W.; Sauter, C.; Schuchmann, H.P. Deagglomeration processes in high-shear devices. Chem. Eng. Res. Des. 2008, 86, 1369–1381. [Google Scholar] [CrossRef]
- Bałdyga, J.; Orciuch, W.; Makowski, Ł.; Malski-Brodzicki, M.; Malik, K. Break up of nano-particle clusters in high-shear devices. Chem. Eng. Process. Process Intensif. 2007, 46, 851–861. [Google Scholar] [CrossRef]
- Find, J.; Emerson, S.C.; Krausz, I.M.; Moser, W.R. Hydrodynamic cavitation as a tool to control macro-, micro-, and nano-properties of inorganic materials. J. Mater. Res. 2001, 16, 3503–3513. [Google Scholar] [CrossRef]
- Moser, W.R.; Find, J.; Emerson, S.C.; Krausz, I.M. Engineered synthesis of nanostructured materials and catalysts. Adv. Chem. Eng. 2001, 27, 1–48. [Google Scholar]
- Sunstrom IV, J.E.; Moser, W.R.; Marshik-Guerts, B. General route to nanocrystalline oxides by hydrodynamic cavitation. Chem. Mater. 1996, 8, 2061–2067. [Google Scholar] [CrossRef]
- Moser, W.R.; Marshik, B.J.; Kingsley, J.; Lemberger, M.; Willette, R.; Chan, A.; Sunstrom IV, J.E.; Boye, A. The synthesis and characterization of solid-state materials produced by high shear-hydrodynamic cavitation. J. Mater. Res. 1995, 10, 2322–2335. [Google Scholar] [CrossRef]
- Kuldeep; Saharan, V.K. Computational study of different venturi and orifice type hydrodynamic cavitating devices. J. Hydrodyn. 2016, 28, 293–305. [Google Scholar] [CrossRef]
- Cerecedo, L.M.; Dopazo, C.; Gomez-Lus, R. Water disinfection by hydrodynamic cavitation in a rotor-stator device. Ultrason. Sonochem. 2018, 48, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Park, J.J.; Kim, H.S.; Lee, S.H.; Seong, S.J.; Om, A.S.; Yoon, J.Y. Experimental investigation of the thermal and disinfection performances of a novel hydrodynamic cavitation reactor. Ultrason. Sonochem. 2018, 49, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Badve, M.P.; Alpar, T.; Pandit, A.B.; Gogate, P.R.; Csoka, L. Modeling the shear rate and pressure drop in a hydrodynamic cavitation reactor with experimental validation based on KI decomposition studies. Ultrason. Sonochem. 2015, 22, 272–277. [Google Scholar] [CrossRef] [PubMed]
- Rinaldi, L.; Wu, Z.; Giovando, S.; Bracco, M.; Crudo, D.; Bosco, V.; Cravotto, G. Oxidative polymerization of waste cooking oil with air under hydrodynamic cavitation. Green Process. Synth. 2017, 6, 425–432. [Google Scholar] [CrossRef]
- Crudo, D.; Bosco, V.; Cavaglià, G.; Grillo, G.; Mantegna, S.; Cravotto, G. Biodiesel production process intensification using a rotor-stator type generator of hydrodynamic cavitation. Ultrason. Sonochem. 2016, 33, 220–225. [Google Scholar] [CrossRef]
- Kim, H.; Sun, X.; Koo, B.; Yoon, J.Y. Experimental investigation of sludge treatment using a rotor-stator type hydrodynamic cavitation reactor and an ultrasonic bath. Processes 2019, 7, 790. [Google Scholar] [CrossRef]
- Maa, Y.-F.; Hsu, C. Liquid-liquid emulsification by rotor/stator homogenization. J. Controlled Release 1996, 38, 219–228. [Google Scholar] [CrossRef]
- Burmistrov, V.A.; Lipatova, I.M.; Losev, N.V.; Rodicheva, J.A.; Koifman, O.I. Influence of the composition and high shear stresses on the structure and properties of hybrid materials based on starch and synthetic copolymer. Carbohydr. Polym. 2018, 196, 368–375. [Google Scholar] [CrossRef]
- Petkovšek, M.; Zupanc, M.; Dular, M.; Kosjek, T.; Heath, E.; Kompare, B.; Širok, B. Rotation generator of hydrodynamic cavitation for water treatment. Sep. Purif. Technol. 2013, 118, 415–423. [Google Scholar] [CrossRef]
- Petkovšek, M.; Mlakar, M.; Levstek, M.; Stražar, M.; Širok, B.; Dular, M. A novel rotation generator of hydrodynamic cavitation for waste-activated sludge disintegration. Ultrason. Sonochem. 2015, 26, 408–414. [Google Scholar] [CrossRef] [PubMed]
- Gogate, P.R. Cavitation: An auxiliary technique in wastewater treatment schemes. Adv. Environ. Res. 2002, 6, 335–358. [Google Scholar] [CrossRef]
- Gogate, P.R.; Shirgaonkar, I.Z.; Sivakumar, M.; Senthilkumar, P.; Vichare, N.P.; Pandit, A.B. Cavitation reactors: Efficiency assessment using a model reaction. AIChE J. 2001, 47, 2526–2538. [Google Scholar] [CrossRef]
- Wu, Z.; Franke, M.; Ondruschka, B.; Zhang, Y.; Ren, Y.; Braeutigam, P.; Wang, W. Enhanced effect of suction-cavitation on the ozonation of phenol. J. Hazard. Mater. 2011, 190, 375–380. [Google Scholar] [CrossRef]
- Wu, Z.; Ondruschka, B.; Zhang, Y.; Bremner, D.H.; Shen, H.; Franke, M. Chemistry driven by suction. Green Chem. 2009, 11, 1026. [Google Scholar] [CrossRef]
- Wu, Z.; Shen, H.; Ondruschka, B.; Zhang, Y.; Wang, W.; Bremner, D.H. Removal of blue-green algae using the hybrid method of hydrodynamic cavitation and ozonation. J. Hazard. Mater. 2012, 235–236, 152–158. [Google Scholar] [CrossRef]
- Kumar, P.S.; Pandit, A.B. Modeling hydrodynamic cavitation. Chem. Eng. Technol. 1999, 22, 1017–1027. [Google Scholar] [CrossRef]
- Li, P.; Song, Y.; Yu, S.; Park, H.-D. The effect of hydrodynamic cavitation on Microcystis aeruginosa: Physical and chemical factors. Chemosphere 2015, 136, 245–251. [Google Scholar] [CrossRef]
- Gogate, P.R. Application of cavitational reactors for water disinfection: Current status and path forward. J. Environ. Manag. 2007, 85, 801–815. [Google Scholar] [CrossRef]
- Sivakumar, M.; Pandit, A.B. Wastewater treatment: A novel energy efficient hydrodynamic cavitational technique. Ultrason. Sonochem. 2002, 9, 123–131. [Google Scholar] [CrossRef]
- Zupanc, M.; Pandur, Z.; Stepišnik Perdih, T.; Stopar, D.; Petkovšek, M.; Dular, M. Effects of cavitation on different microorganisms: The current understanding of the mechanisms taking place behind the phenomenon. A review and proposals for further research. Ultrason. Sonochem. 2019, 57, 147–165. [Google Scholar] [CrossRef] [PubMed]
- Sayyaadi, H. Assessment of tandem Venturi on enhancement of cavitational chemical reaction. J. Fluid Eng.-Trans. ASME 2009, 131, 113011–113017. [Google Scholar] [CrossRef]
- Capocelli, M.; Musmarra, D.; Prisciandaro, M.; Lancia, A. Chemical effect of hydrodynamic cavitation: Simulation and experimental comparison. AIChE J. 2014, 60, 2566–2572. [Google Scholar] [CrossRef]
- Zhang, X.; Fu, Y.; Li, Z.; Zhao, Z. The collapse intensity of cavities and the concentration of free hydroxyl radical released in cavitation flow. Chin. J. Chem. Eng. 2008, 16, 547–551. [Google Scholar] [CrossRef]
- Geng, K.; Dong, Z.-Y.; Zhang, K.; Ju, W.-J.; Zhao, W.-Q.; Li, Y.-R.; Qin, Z.-Y.; Wang, L. Experimental study of Escherichia coli killed by hydrodynamic cavitation due to venturi tube. J. Environ. Sci. 2017, 37, 3385–3391. [Google Scholar]
- Karamah, E.F.; Sunarko, I. Disinfection of bacteria Escherichia coli using hydrodynamic cavitation. Int. J. Technol. 2013, 4, 209–216. [Google Scholar] [CrossRef][Green Version]
- Maddikeri, G.L.; Gogate, P.R.; Pandit, A.B. Intensified synthesis of biodiesel using hydrodynamic cavitation reactors based on the interesterification of waste cooking oil. Fuel 2014, 137, 285–292. [Google Scholar] [CrossRef]
- Pawar, S.K.; Mahulkar, A.V.; Pandit, A.B.; Roy, K.; Moholkar, V.S. Sonochemical effect induced by hydrodynamic cavitation: Comparison of venturi/orifice flow geometries. AIChE J. 2017, 63, 4705–4716. [Google Scholar] [CrossRef]
- Rajoriya, S.; Bargole, S.; Saharan, V.K. Degradation of reactive blue 13 using hydrodynamic cavitation: Effect of geometrical parameters and different oxidizing additives. Ultrason. Sonochem. 2017, 37, 192–202. [Google Scholar] [CrossRef]
- Šarc, A.; Stepišnik-Perdih, T.; Petkovšek, M.; Dular, M. The issue of cavitation number value in studies of water treatment by hydrodynamic cavitation. Ultrason. Sonochem. 2017, 34, 51–59. [Google Scholar] [CrossRef]
- Pradhan, A.A.; Gogate, P.R. Removal of p-nitrophenol using hydrodynamic cavitation and Fenton chemistry at pilot scale operation. Chem. Eng. J. 2010, 156, 77–82. [Google Scholar] [CrossRef]
- Suslick, K.S.; Mdleleni, M.M.; Ries, J.T. Chemistry induced by hydrodynamic cavitation. J. Am. Chem. Soc. 1997, 119, 9303–9304. [Google Scholar] [CrossRef]
- Zhang, J.; Xu, S.; Li, W. High shear mixers: A review of typical applications and studies on power draw, flow pattern, energy dissipation and transfer properties. Chem. Eng. Process. Process Intensif. 2012, 57–58, 25–41. [Google Scholar] [CrossRef]
- Bałdyga, J.; Orciuch, W.; Makowski, Ł.; Malik, K.; Özcan-Taşkin, G.; Eagles, W.; Padron, G. Dispersion of nanoparticle clusters in a rotor-stator mixer. Ind. Eng. Chem. Res. 2008, 47, 3652–3663. [Google Scholar] [CrossRef]
- Moussa, M.; Perrier-Cornet, J.-M.; Gervais, P. Damage in Escherichia coli cells treated with a combination of high hydrostatic pressure and subzero temperature. Appl. Environ. Microbiol. 2007, 73, 6508–6518. [Google Scholar] [CrossRef]
- Hewitt, C.J.; Boon, L.A.; McFarlane, C.M.; Nienow, A.W. The use of flow cytometry to study the impact of fluid mechanical stress on Escherichia coli W3110 during continuous cultivation in an agitated bioreactor. Biotechnol. Bioeng. 1998, 59, 612–620. [Google Scholar] [CrossRef]
- Croughan, M.S.; Sayre, E.S.; Wang, D.I.C. Viscous reduction of turbulent damage in animal cell culture. Biotechnol. Bioeng. 1989, 33, 862–872. [Google Scholar] [CrossRef]
- Lakhotia, S.; Papoutsakis, E.T. Agitation induced cell injury in microcarrier cultures. Protective effect of viscosity is agitation intensity dependent: Experiments and modeling. Biotechnol. Bioeng. 1992, 39, 95–107. [Google Scholar] [CrossRef]
- Dreosti, I.E. Bioactive ingredients: Antioxidants and polyphenols in tea. Nutr. Rev. 1996, 54, S51–S58. [Google Scholar] [CrossRef]
- Pan, X.; Niu, G.; Liu, H. Microwave-assisted extraction of tea polyphenols and tea caffeine from green tea leaves. Chem. Eng. Process. 2003, 42, 129–133. [Google Scholar] [CrossRef]
- Chemat, F.; Zill-E-Huma; Khan, M.K. Applications of ultrasound in food technology: Processing, preservation and extraction. Ultrason. Sonochem. 2011, 18, 813–835. [Google Scholar] [CrossRef] [PubMed]
- Vilkhu, K.; Mawson, R.; Simons, L.; Bates, D. Applications and opportunities for ultrasound assisted extraction in the food industry—A review. Innov. Food Sci. Emerg. Technol. 2008, 9, 161–169. [Google Scholar] [CrossRef]
- Miao, S.-F.; Yu, J.-P.; Du, Z.; Guan, Y.-X.; Yao, S.-J.; Zhu, Z.-Q. Supercritical fluid extraction and micronization of ginkgo flavonoids from ginkgo biloba leaves. Ind. Eng. Chem. Res. 2010, 49, 5461–5466. [Google Scholar] [CrossRef]
- Malovanyy, M.; Nikiforov, V.; Kharlamova, O.; Synelnikov, O. Production of renewable energy resources via complex treatment of cyanobacteria biomass. Chem. Chem. Technol. 2016, 10, 251–254. [Google Scholar] [CrossRef]
- Dong, Y.-Y.; Li, S.-M.; Ma, M.-G.; Zhao, J.-J.; Sun, R.-C.; Wang, S.-P. Environmentally-friendly sonochemistry synthesis of hybrids from lignocelluloses and silver. Carbohydr. Polym. 2014, 102, 445–452. [Google Scholar] [CrossRef] [PubMed]
- Terán Hilares, R.; dos Santos, J.C.; Ahmed, M.A.; Jeon, S.H.; da Silva, S.S.; Han, J.-I. Hydrodynamic cavitation-assisted alkaline pretreatment as a new approach for sugarcane bagasse biorefineries. Bioresour. Technol. 2016, 214, 609–614. [Google Scholar] [CrossRef] [PubMed]
- Terán Hilares, R.; Kamoei, D.V.; Ahmed, M.A.; da Silva, S.S.; Han, J.-I.; Santos, J.C.D. A new approach for bioethanol production from sugarcane bagasse using hydrodynamic cavitation assisted-pretreatment and column reactors. Ultrason. Sonochem. 2018, 43, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Ancza, E.; Diószegi, M.B.; Horváth, M. Hydrodynamic cavitation device that makes straw cuts suitable for efficient biogas production. Appl. Mech. Mater. 2014, 564, 572–576. [Google Scholar] [CrossRef]
- Zieliński, M.; Dębowski, M.; Kisielewska, M.; Nowicka, A.; Rokicka, M.; Szwarc, K. Comparison of ultrasonic and hydrothermal cavitation pretreatments of cattle manure mixed with straw wheat on fermentative biogas production. Waste Biomass Valori. 2017, 10, 747–754. [Google Scholar] [CrossRef]
- 2018—Italian Patent n. 102016000007489. Available online: https://www.epic-srl.com (accessed on 14 December 2019).
- Calcio Gaudino, E.; Grillo, G.; Cravotto, G.; Daniele, C.; Thomas, D.; Gerhard, S.; Liga, L.; Galina, T.; Ochoa-gómez, J.R. High-intensity ultrasound and hydrodynamic cavitation as powerful treament for biomass conversion. In Proceedings of the 4th International Congress on Catalysis for Biorefineries, Catbior 2017, Université Lyon, Lyon, France, 11–15 December 2017; Volume 2, p. 168. [Google Scholar]

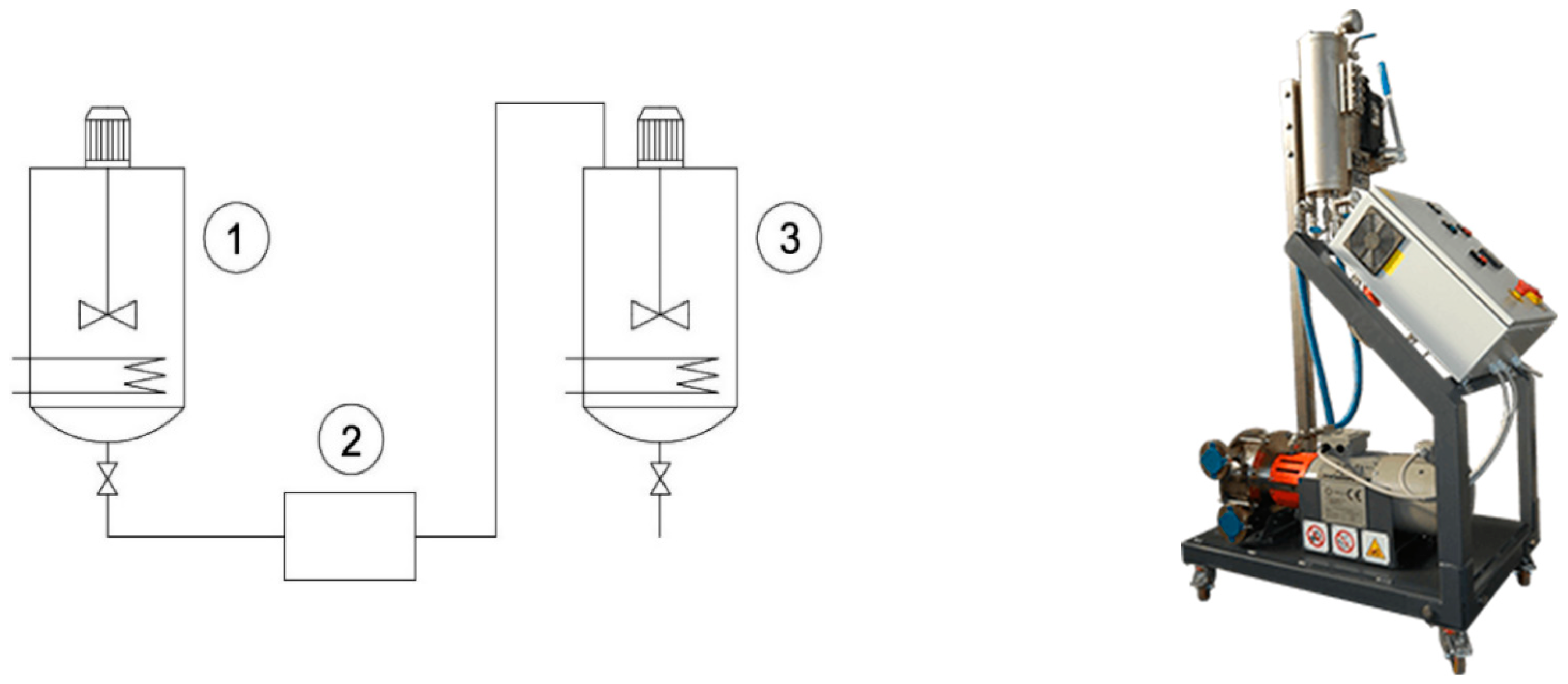
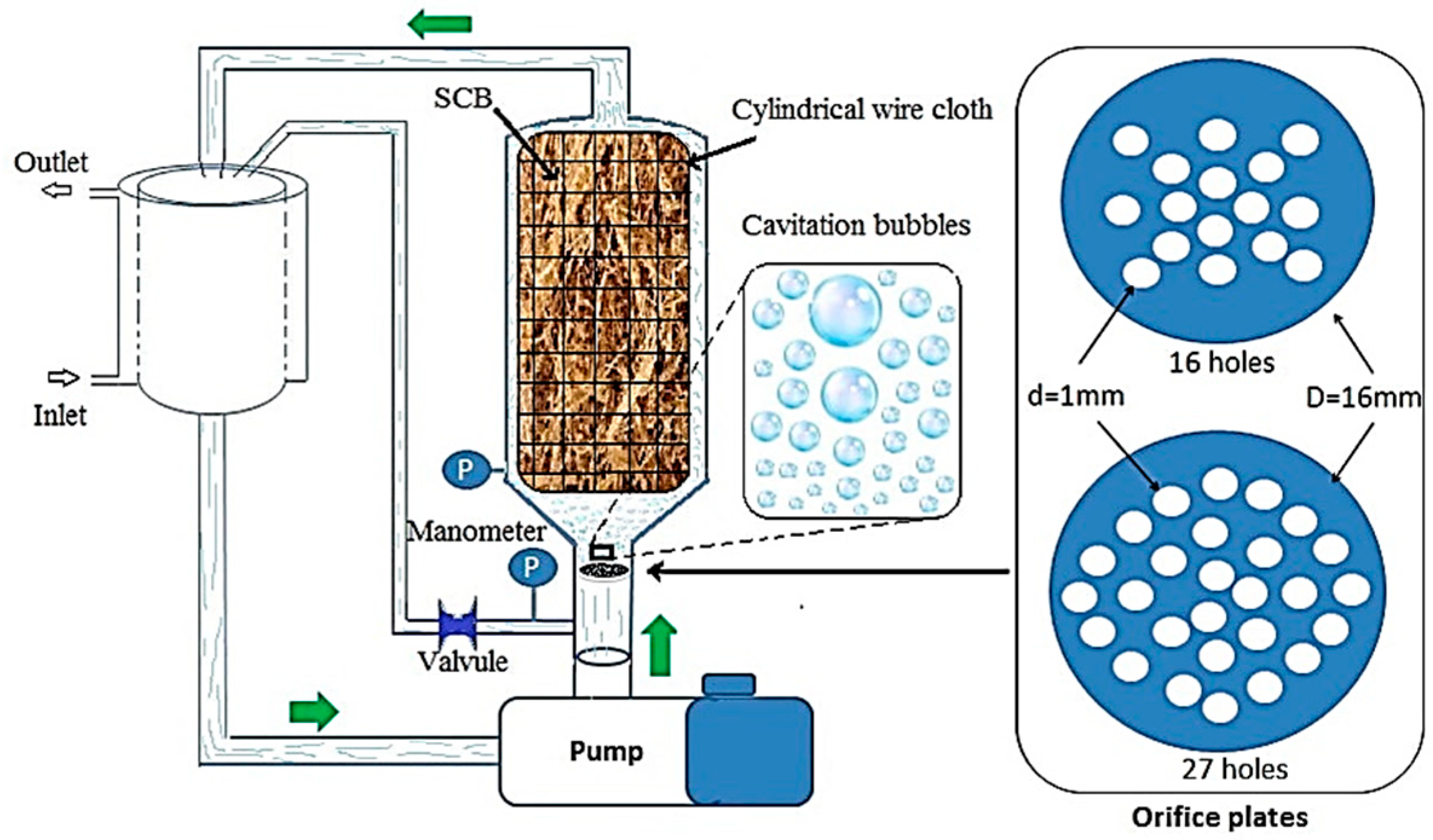
| Hydro-Alcoholic Phase | Hexane Phase | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Theobromine | Caffeine | TPC | DPPH EC50 | Trolox Eq. | Fats | |||||
| Method | w/w% extract | mg/g shells | w/w% extract | mg/g shells | mg/g extract | mg/g shells | μg/mL | μmol/g extract | w/w% extract | mg/g shells |
| UAE | 5.04 | 7.02 ± 0.11 | 0.81 | 1.13 ± 0.09 | 51.1 | 7.1 | 76.9 ± 3.6 | 204.7 ± 9.6 | 91.7 | 23.2 |
| RSD | 9.25 | 13.5 ± 0.16 | 0.75 | 1.09 ± 0.12 | 79.9 | 11.7 | 72.1 ± 4.1 | 218.3 ± 12.4 | 94.3 | 95.0 |
| Sample | Extraction Method | Total phenolic Content | Antioxidant Capacity | |
|---|---|---|---|---|
| mg/g Extract | mg/g Matrix | μmol ET/g Matrix | ||
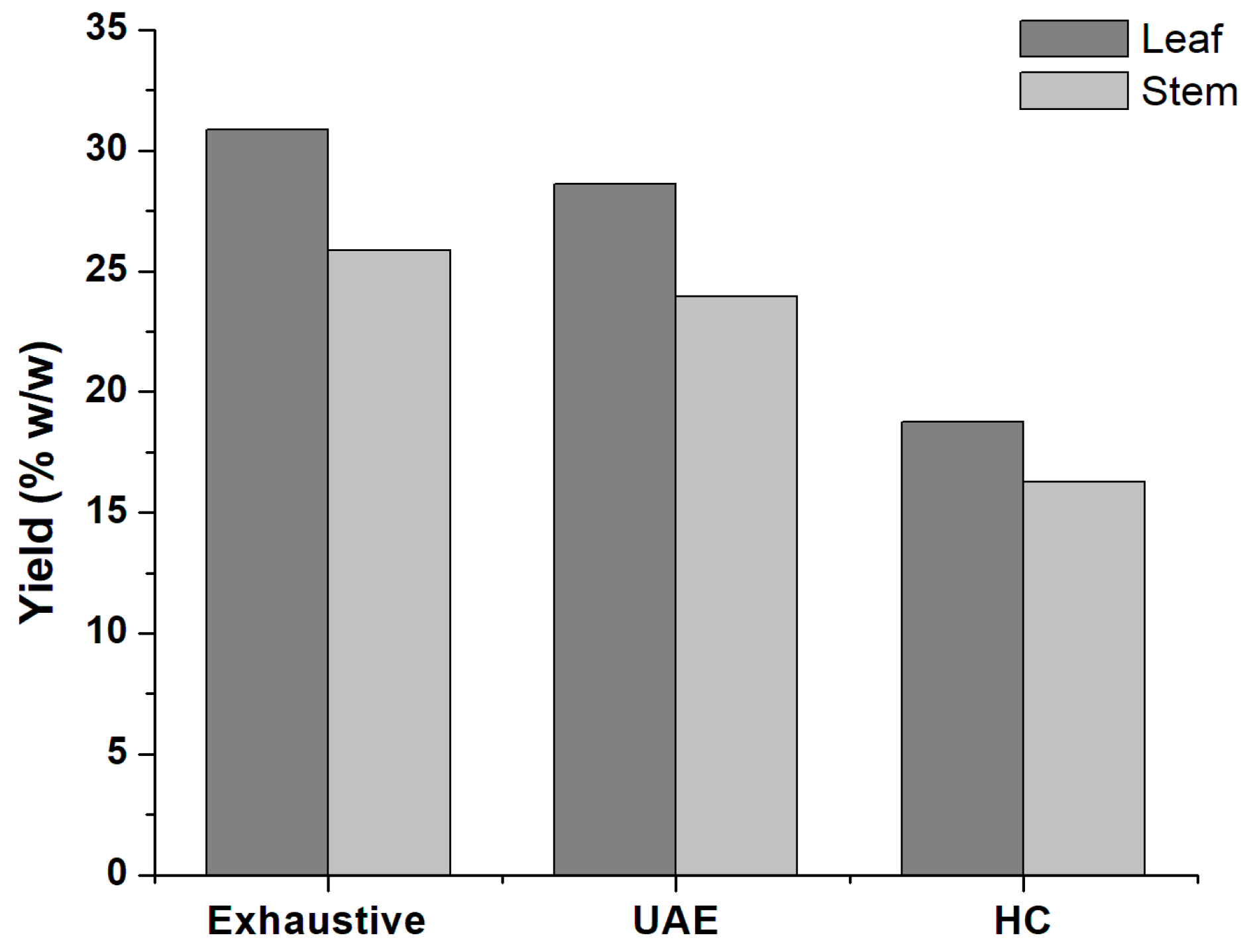
| Leaf | Exhaustive | 220.92 | 68.24 | 96.55 |
| UAE | 232.30 | 66.44 | 79.15 | |
| HCE | 234.78 | 44.01 | 79.51 | |
| Exhaustive | 199.19 | 50.23 | 72.02 | |
| Stem | UAE | 207.31 | 49.65 | 71.39 |
| HCE | 238.80 | 40.43 | 69.09 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, Z.; Ferreira, D.F.; Crudo, D.; Bosco, V.; Stevanato, L.; Costale, A.; Cravotto, G. Plant and Biomass Extraction and Valorisation under Hydrodynamic Cavitation. Processes 2019, 7, 965. https://doi.org/10.3390/pr7120965
Wu Z, Ferreira DF, Crudo D, Bosco V, Stevanato L, Costale A, Cravotto G. Plant and Biomass Extraction and Valorisation under Hydrodynamic Cavitation. Processes. 2019; 7(12):965. https://doi.org/10.3390/pr7120965
Chicago/Turabian StyleWu, Zhilin, Daniele F. Ferreira, Daniele Crudo, Valentina Bosco, Livio Stevanato, Annalisa Costale, and Giancarlo Cravotto. 2019. "Plant and Biomass Extraction and Valorisation under Hydrodynamic Cavitation" Processes 7, no. 12: 965. https://doi.org/10.3390/pr7120965
APA StyleWu, Z., Ferreira, D. F., Crudo, D., Bosco, V., Stevanato, L., Costale, A., & Cravotto, G. (2019). Plant and Biomass Extraction and Valorisation under Hydrodynamic Cavitation. Processes, 7(12), 965. https://doi.org/10.3390/pr7120965






