The Engineering of Porous Silica and Hollow Silica Nanoparticles to Enhance Drug-loading Capacity
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
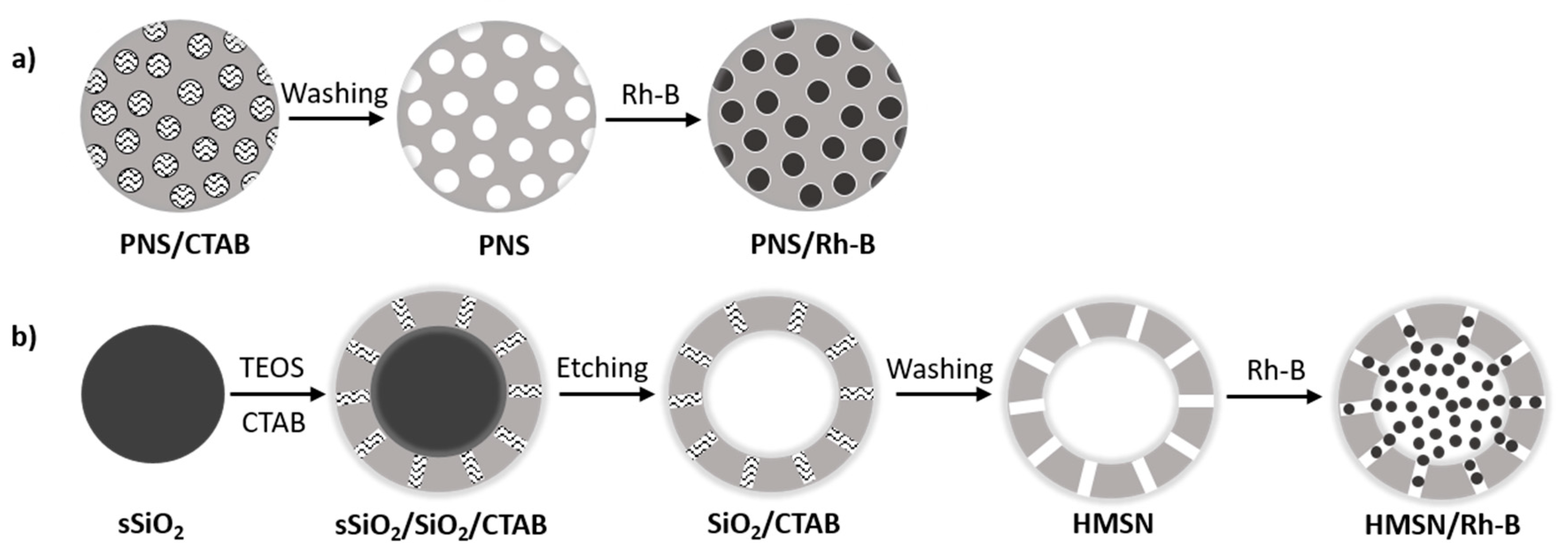
2.2. Preparation of PNS
2.3. Preparation of HMSNs
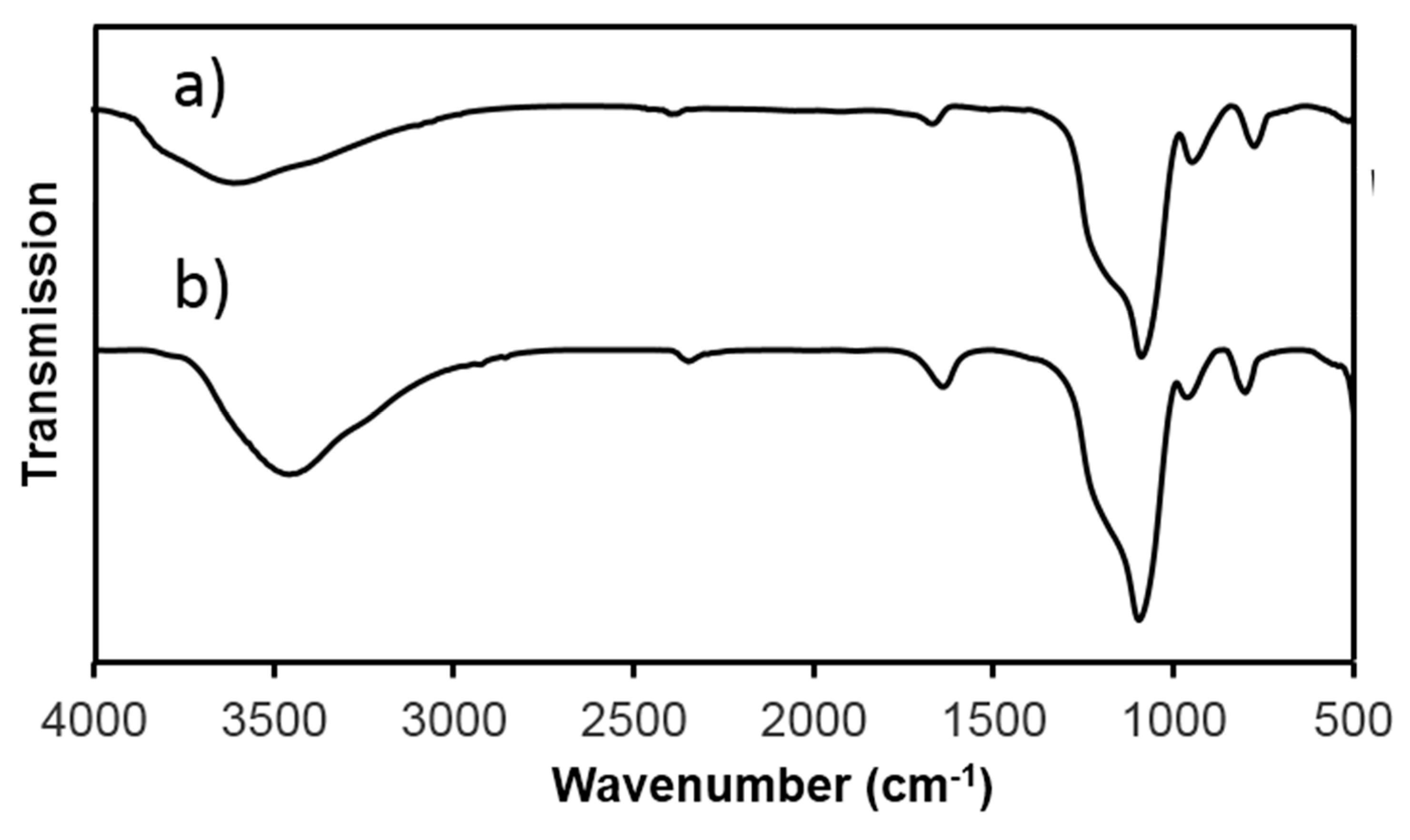
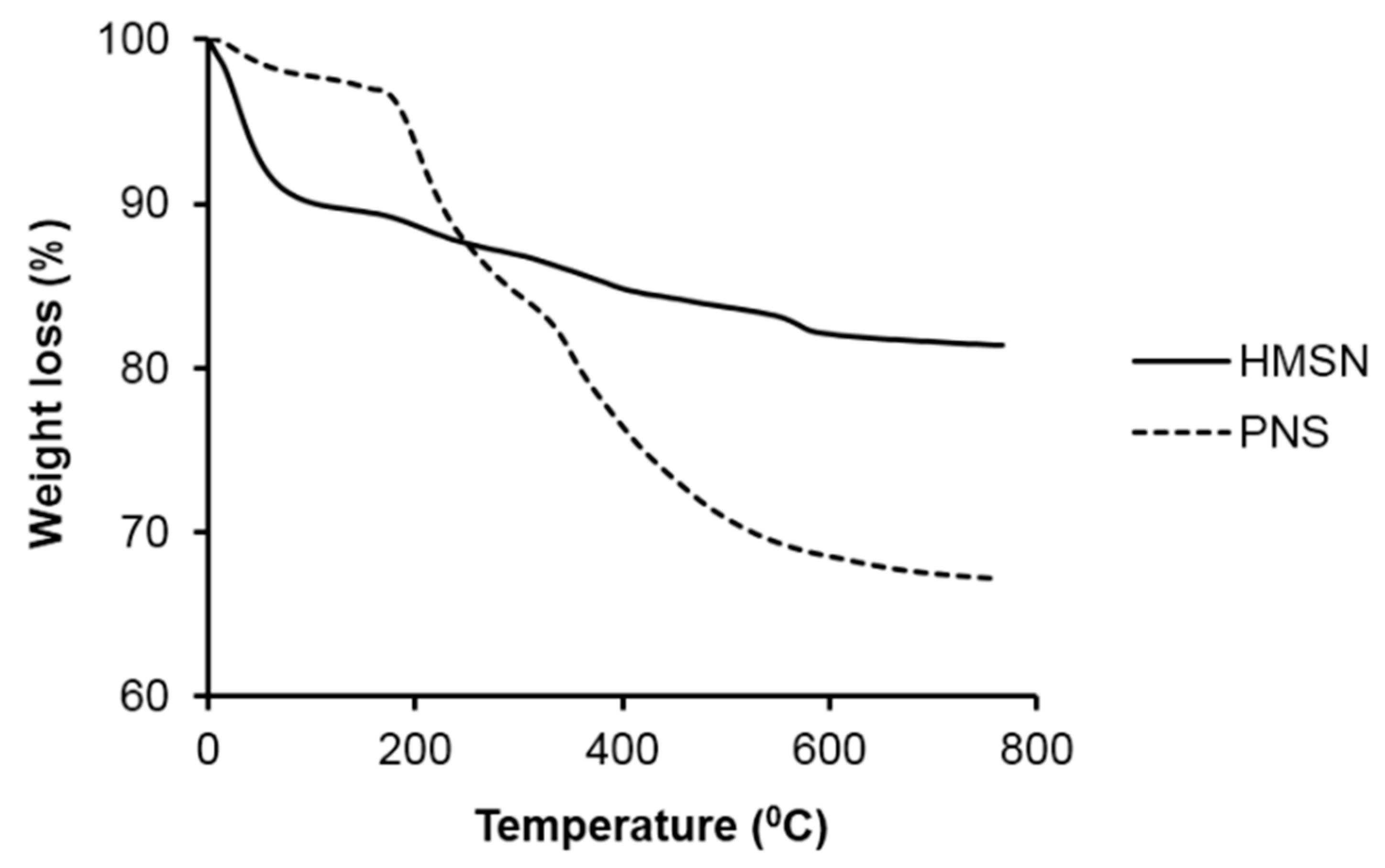
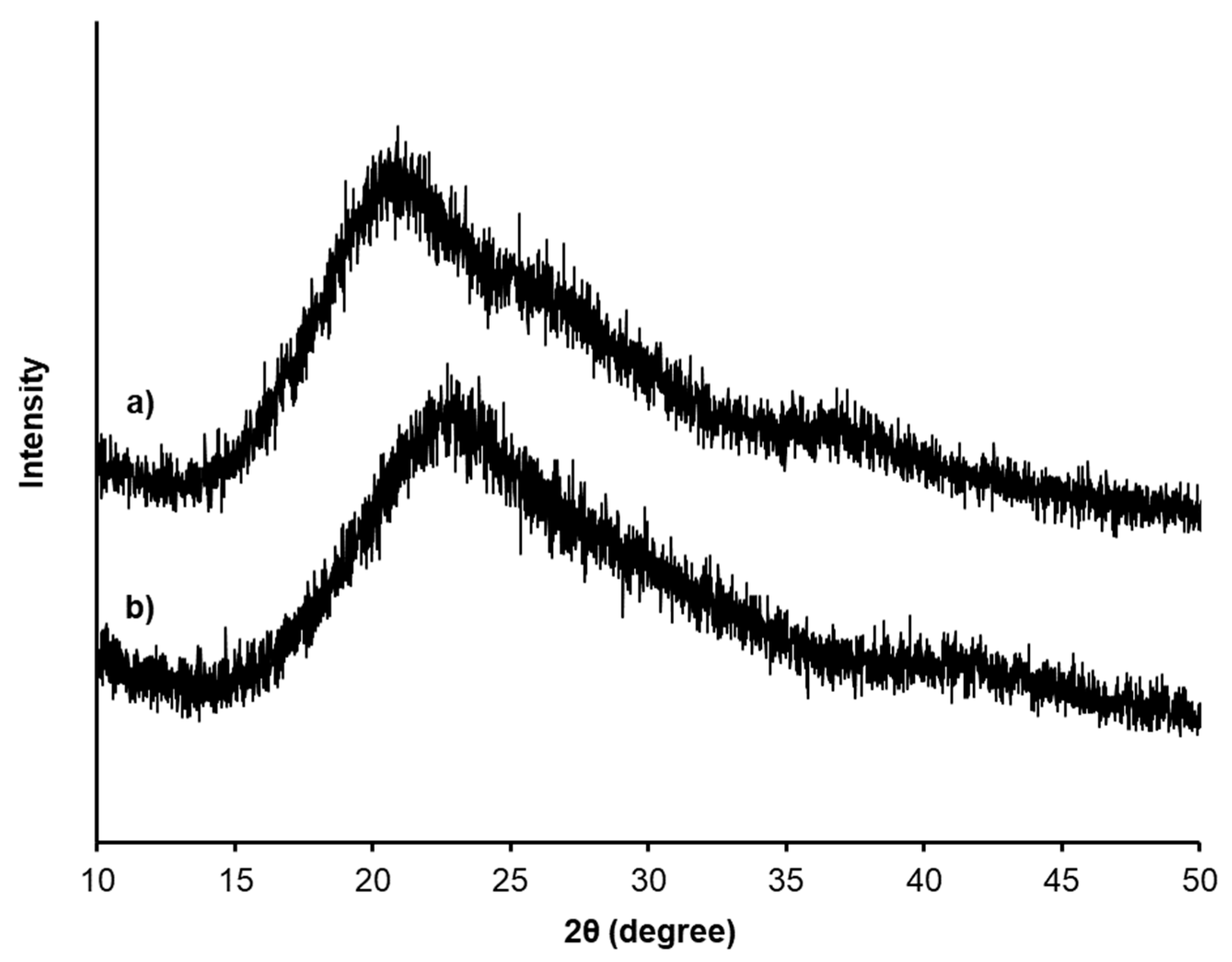
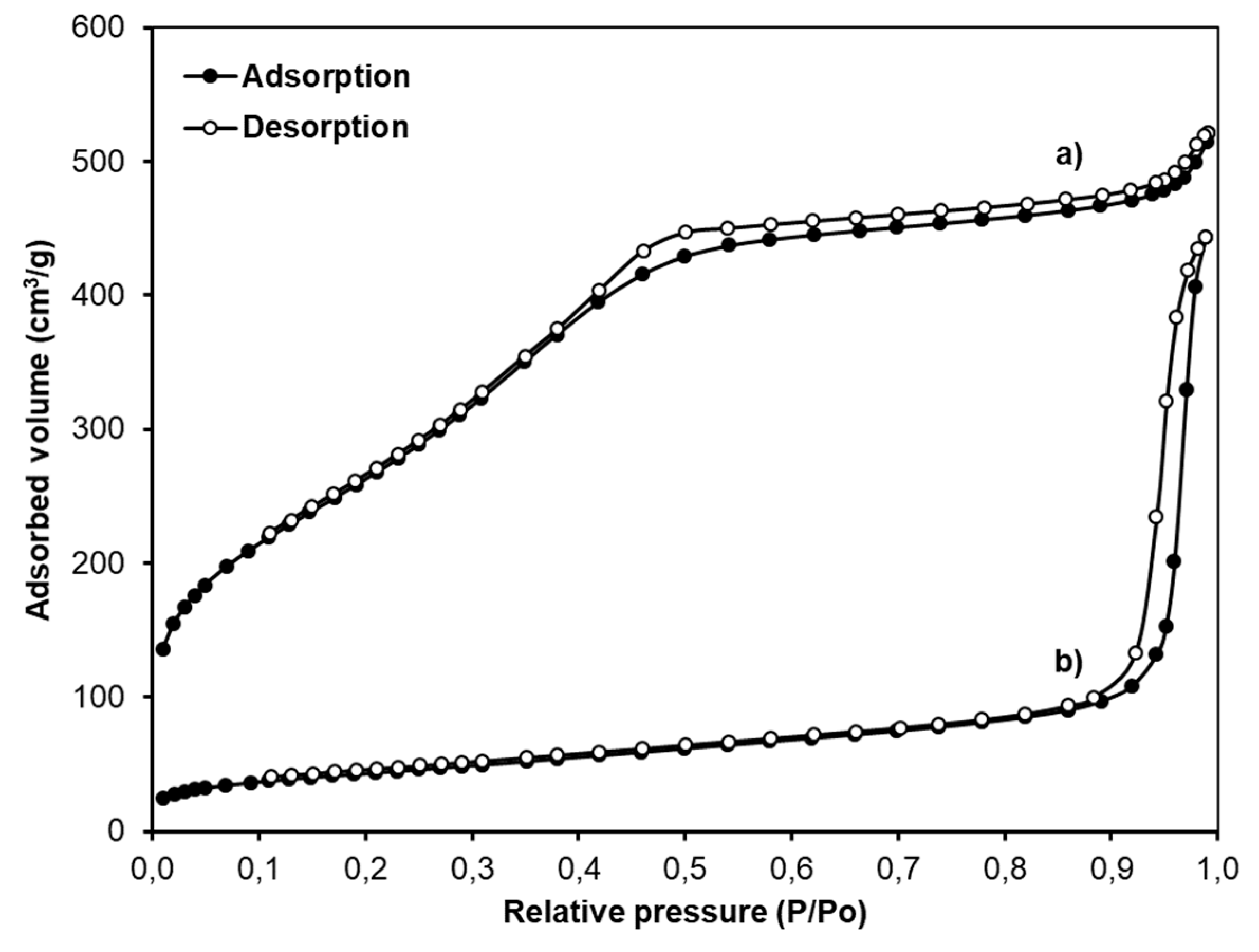
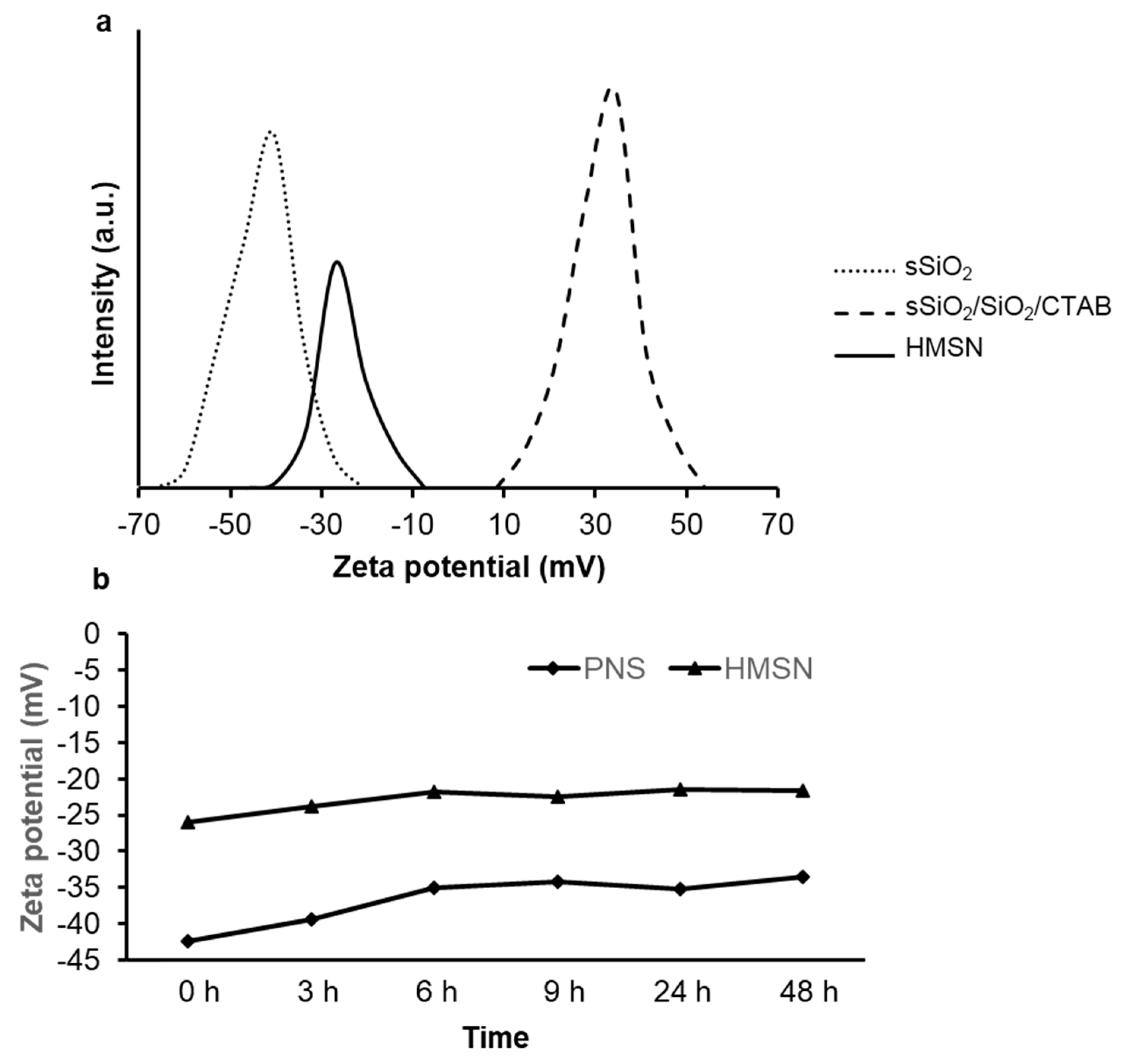
2.4. Characterization
2.5. Preparation of RhB/PNS and RhB/HMSN
2.6. Statistical Analysis
3. Result and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Vo, U.V.; Nguyen, C.K.; Nguyen, V.C.; Tran, T.V.; Thi, B.Y.T.; Nguyen, D.H. Gelatin-poly (ethylene glycol) methyl ether-functionalized porous Nanosilica for efficient doxorubicin delivery. J. Polym. Res. 2019, 26, 6. [Google Scholar] [CrossRef]
- Lu, J.; Liong, M.; Zink, J.; Tamanoi, F. Mesoporous Silica Nanoparticles as a Delivery System for Hydrophobic Anticancer Drugs. Small 2007, 3, 1341–1346. [Google Scholar] [CrossRef]
- Slowing, I.I.; Trewyn, B.; Giri, S.; Lin, V.S.Y. Mesoporous silica nanoparticles for drug delivery and biosensing applications. Adv. Funct. Mater. 2007, 17, 1225–1236. [Google Scholar] [CrossRef]
- Thi, T.T.H.; Cao, V.D.; Nguyen, T.N.Q.; Hoang, D.T.; Ngo, V.C.; Nguyen, D.H. Functionalized mesoporous silica nanoparticles and biomedical applications. Mater. Sci. Eng. C 2019, 99, 631–656. [Google Scholar]
- Bao, B.Q.; Le, N.H.; Nguyen, D.H.T.; Tran, T.V.; Pham, L.P.T.; Bach, L.G.; Ho, H.M.; Nguyen, T.H.; Nguyen, D.H. Evolution and present scenario of multifunctionalized mesoporous nanosilica platform: A mini review. Mater. Sci. Eng. C 2018, 91, 912–928. [Google Scholar] [CrossRef] [PubMed]
- Slowing, I.I.; Vivero-Escoto, J.; Wu, C.W.; Lin, V.S.Y. Mesoporous silica nanoparticles as controlled release drug delivery and gene transfection carriers. Adv. Drug Deliv. Rev. 2008, 60, 1278–1288. [Google Scholar] [CrossRef] [PubMed]
- Tang, F.; Li, L.; Chen, D. Mesoporous silica nanoparticles: Synthesis, biocompatibility and drug delivery. Adv. Mater. 2012, 24, 1504–1534. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, S.; Manivasagam, G.; Kumar, P.; Ambasta, R.K. Cellular Toxicity of Mesoporous Silica Nanoparticle in SHSY5Y and BMMNCs Cell. Pharm. Nanotechnol. 2018, 6, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Murugadoss, S.; Lison, D.; Godderis, L.; Brule, S.V.D.; Mast, J.; Brassinne, F.; Sebaihi, N.; Hoet, P.H. Toxicology of silica nanoparticles: An update. Arch. Toxicol. 2017, 91, 2967–3010. [Google Scholar] [CrossRef] [PubMed]
- Tsou, C.J.; Hung, Y.; Mou, C.Y. Hollow mesoporous silica nanoparticles with tunable shell thickness and pore size distribution for application as broad-ranging pH nanosensor. Microporous Mesoporous Mater. 2014, 190, 181–188. [Google Scholar] [CrossRef]
- Chen, F.; Hong, H.; Shi, S.; Goel, S.; Valdovinos, H.F.; Hernandez, R.; Theuer, C.P.; Barnhart, T.E.; Cai, W. Engineering of Hollow Mesoporous Silica Nanoparticles for Remarkably Enhanced Tumor Active Targeting Efficacy. Sci. Rep. 2014, 4, 5080. [Google Scholar] [CrossRef] [PubMed]
- Farsangi, Z.J.; Rezayat, S.M.; Beitollahi, A.; Sarkar, S.; Jaafari, M.; Amani, A. Hollow Mesoporous Silica Nanoparticles (HMSNs) Synthesis and in vitro Evaluation of Cisplatin Delivery. J. Nanoanal. 2016, 3, 120–127. [Google Scholar]
- Ghasemi, S.; Farsangi, Z.; Beitollahi, A.; Mirkazemi, S.M.; Rezayat, M.; Sarkar, S. Synthesis of Hollow Mesoporous Silica (HMS) nanoparticles as a candidate for sulfasalazine drug loading. Ceram. Int. 2017, 43, 11225–11232. [Google Scholar] [CrossRef]
- Zhu, Y.; Fang, Y.; Borchardt, L.; Kaskel, S. PEGylated hollow mesoporous silica nanoparticles as potential drug delivery vehicles. Microporous Mesoporous Mater. 2011, 141, 199–206. [Google Scholar] [CrossRef]
- Shi, S.; Chen, F.; Cai, W. Biomedical applications of functionalized hollow mesoporous silica nanoparticles: Focusing on molecular imaging. Nanomedicine 2013, 8, 2027–2039. [Google Scholar] [CrossRef]
- Zhu, Y.; Shi, J.; Shen, W.; Dong, X.; Feng, J.; Ruan, M.; Li, Y. Stimuli-Responsive Controlled Drug Release from a Hollow Mesoporous Silica Sphere/Polyelectrolyte Multilayer Core–Shell Structure. Angew. Chem. Int. Edit. 2005, 44, 5213–5217. [Google Scholar] [CrossRef]
- Mei, X.; Yang, S.; Chen, D.; Li, N.; Li, H.; Xu, Q.; Ge, J.; Lu, J. Light-triggered reversible assemblies of azobenzene-containing amphiphilic copolymer with β-cyclodextrin-modified hollow mesoporous silica nanoparticles for controlled drug release. Chem. Commun. 2012, 48, 10010–10012. [Google Scholar] [CrossRef]
- Ma, X.; Zhao, Y.; Ng, K.W.; Zhao, Y. Integrated hollow mesoporous silica nanoparticles for target drug/siRNA co-delivery. Chem. A Eur. J. 2013, 19, 15593–15603. [Google Scholar] [CrossRef]
- Yang, Y.; Guo, X.; Wei, K.; Wang, L.; Yang, D.; Lai, L.; Cheng, M.; Liu, Q. Synthesis and drug-loading properties of folic acid-modified superparamagnetic Fe3O4 hollow microsphere core/mesoporous SiO2 shell composite particles. J. Nanopart. Res. 2014, 16, 2210. [Google Scholar] [CrossRef]
- Patil, P.B.; Karade, V.; Waifalkar, P.P.; Sahoo, S.C.; Kollu, P.; Nimbalkar, M.; Chougale, A.D.; Patil, P.S. Functionalization of magnetic hollow spheres with 3-Aminopropyl) triethoxysilane (APTES) for controlled drug release. IEEE Trans. Magn. 2017, 53, 1–4. [Google Scholar] [CrossRef]
- Vo, U.V.; Tran, T.V.; Nguyen, D.M.T.; Nguyen, C.K.; Thi, N.T.N.; Nguyen, D.H. Effective pH-responsive hydrazine-modified silica for doxorubicin delivery. Asian J. Med. Health 2017, 4, 1–7. [Google Scholar] [CrossRef]
- Liu, J.; Luo, Z.; Zhang, J.; Luo, T.; Zhou, J.; Zhao, X.; Cai, K. Hollow mesoporous silica nanoparticles facilitated drug delivery via cascade pH stimuli in tumor microenvironment for tumor therapy. Biomaterials 2016, 83, 51–65. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Wang, S.; Yang, Y.; Li, X.; Di, D.; Zhang, C.; Jiang, T.; Wang, S. Hyaluronic acid and carbon dots-gated hollow mesoporous silica for redox and enzyme-triggered targeted drug delivery and bioimaging. Mater. Sci. Eng. C 2017, 78, 475–484. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Ma, W.; Shangguan, J.; Jiang, W.; Zhong, Q. Controllable synthesis of hollow mesoporous silica spheres and application as support of nano-gold. J. Solid State Chem. 2014, 215, 67–73. [Google Scholar] [CrossRef]
- Chen, Y.; Xu, P.; Chen, H.; Li, Y.; Bu, W.; Shu, Z.; Li, Y.; Zhang, J.; Zhang, L.; Pan, L.; et al. Colloidal HPMO Nanoparticles: Silica-Etching Chemistry Tailoring, Topological Transformation, and Nano-Biomedical Applications. Adv. Mater. 2013, 25, 3100–3105. [Google Scholar] [CrossRef]
- Chen, Y. Design, Synthesis, Multifunctionalization and Biomedical Applications of Multifunctional Mesoporous Silica-Based Drug Delivery Nanosystems; Springer: Berlin/Heidelberg, Germany, 2016. [Google Scholar]
- Masalov, V.M.; Sukhinina, N.S.; Kudrenko, E.; Emelchenko, G.A. Mechanism of formation and nanostructure of Stöber silica particles. Nanotechnology 2011, 22, 275718. [Google Scholar] [CrossRef]
- Zhou, X.; Cheng, X.; Feng, W.; Qiu, K.; Chen, L.; Nie, W.; Yin, Z.; Mo, X.; Wang, H.; He, C. Synthesis of hollow mesoporous silica nanoparticles with tunable shell thickness and pore size using amphiphilic block copolymers as core templates. Dalton Trans. 2014, 43, 11834. [Google Scholar] [CrossRef]
- Nguyen, D.H.; Lee, J.S.; Choi, J.H.; Lee, Y.; Son, J.Y.; Bae, J.W.; Lee, K.; Park, K.D. Heparin nanogel-containing liposomes for intracellular RNase delivery. Macromol. Res. 2015, 23, 765–769. [Google Scholar] [CrossRef]
- Tran, D.H.N.; Nguyen, T.H.; Vo, T.N.N.; Pham, L.P.T.; Vo, D.M.H.; Nguyen, C.K.; Bach, L.G.; Nguyen, D.H. Self-assembled poly(ethylene glycol) methyl ether-grafted gelatin nanogels for efficient delivery of curcumin in cancer treatment. J. Appl. Polym. Sci. 2019, 136, 20. [Google Scholar] [CrossRef]
- Nguyen, T.N.; Huynh, T.N.; Hoang, D.; Nguyen, D.H.; Nguyen, Q.H.; Tran, T.H. Functional Nanostructured Oligochitosan–Silica/Carboxymethyl Cellulose Hybrid Materials: Synthesis and Investigation of Their Antifungal Abilities. Polymers 2019, 11, 628. [Google Scholar] [CrossRef]
- Sing, K.S.W.; Everett, D.H.; Haul, R.A.W.; Moscou, L.; Pierotti, R.A.; Rouquerol, J.; Siemieniewska, T. Reporting Physisorption Data for Gas/Solid Systems. Int. Union Pure Appl. Chem. 1985, 57, 603–619. [Google Scholar] [CrossRef]
- Wang, W.; Liu, P.; Zhang, M.; Hu, J.; Xing, F. The Pore Structure of Phosphoaluminate Cement. Open J. Compos. Mater. 2012, 2, 104–112. [Google Scholar] [CrossRef]
- Nguyen, A.K.; Nguyen, T.H.; Bao, B.Q.; Bach, L.G.; Nguyen, D.H. Efficient Self-Assembly of mPEG End-Capped Porous Silica as a Redox-Sensitive Nanocarrier for Controlled Doxorubicin Delivery. Int. J. Biomater. 2018, 2018, 1575438. [Google Scholar] [CrossRef] [PubMed]
- Blanco, E.; Shen, H.; Ferrari, M. Principles of nanoparticle design for overcoming biological barriers to drug delivery. Nat. Biotechnol. 2015, 33, 941–951. [Google Scholar] [CrossRef]
- Shen, S.; Wu, Y.; Liu, Y.; Wu, D. High drug-loading nanomedicines: Progress, current status, and prospects. Int. J. Nanomed. 2017, 12, 4085–4109. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen-Thi, N.-T.; Pham Tran, L.P.; Le, N.T.T.; Cao, M.-T.; Tran, T.-N.; Nguyen, N.T.; Nguyen, C.H.; Nguyen, D.-H.; Than, V.T.; Le, Q.T.; et al. The Engineering of Porous Silica and Hollow Silica Nanoparticles to Enhance Drug-loading Capacity. Processes 2019, 7, 805. https://doi.org/10.3390/pr7110805
Nguyen-Thi N-T, Pham Tran LP, Le NTT, Cao M-T, Tran T-N, Nguyen NT, Nguyen CH, Nguyen D-H, Than VT, Le QT, et al. The Engineering of Porous Silica and Hollow Silica Nanoparticles to Enhance Drug-loading Capacity. Processes. 2019; 7(11):805. https://doi.org/10.3390/pr7110805
Chicago/Turabian StyleNguyen-Thi, Ngoc-Tram, Linh Phuong Pham Tran, Ngoc Thuy Trang Le, Minh-Tri Cao, The-Nam Tran, Ngoc Tung Nguyen, Cong Hao Nguyen, Dai-Hai Nguyen, Van Thai Than, Quang Tri Le, and et al. 2019. "The Engineering of Porous Silica and Hollow Silica Nanoparticles to Enhance Drug-loading Capacity" Processes 7, no. 11: 805. https://doi.org/10.3390/pr7110805
APA StyleNguyen-Thi, N.-T., Pham Tran, L. P., Le, N. T. T., Cao, M.-T., Tran, T.-N., Nguyen, N. T., Nguyen, C. H., Nguyen, D.-H., Than, V. T., Le, Q. T., & Trung, N. Q. (2019). The Engineering of Porous Silica and Hollow Silica Nanoparticles to Enhance Drug-loading Capacity. Processes, 7(11), 805. https://doi.org/10.3390/pr7110805




