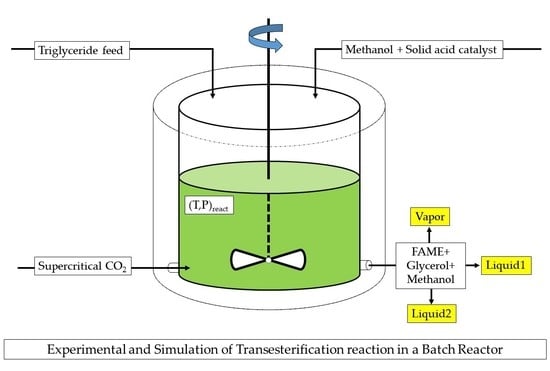
Supercritical CO2 Transesterification of Triolein to Methyl-Oleate in a Batch Reactor: Experimental and Simulation Results
Abstract
:1. Introduction
2. Materials and Methods
- (i)
- Chemicals: For calibration of the supercritical fluid chromatography-mass spectroscopy (SFC-MS) unit, all oleate species standards were purchased from Sigma-Aldrich (purity ≥ 99%, St. Louis, MO, USA) except diolein (1,2 and 1,3 DG isomers, 2:1 isomeric ratio) from MP Biomedicals, LLC (purity ≥ 99%, Santa Ana, CA, USA). ACS grade methanol was obtained from J.T. Baker (Radnor, PA, USA). High Performance Liquid Chromatography (HPLC) grade heptane and ultrapure isopropanol were obtained from Alfa Aesar (Haverhill, MA, USA) and Sigma-Aldrich, Inc. Bone-dry CO2 with a siphon tube and nitrogen gas were supplied by Airgas, Inc. (Radnor, PA, USA). Corn oil was obtained from a local market and was analyzed for fatty-acid content using standard methods (10.4 wt% C16:0, 30.8 wt% C18:1, 58.8 wt% C18:2, others in trace quantities) [13]. Note that, for experiments at low methanol/triolein molar ratios, inexpensive corn oil was purchased.
- (ii)
- Catalyst Characteristics: Nafion NR50 was purchased from Ion Power, Inc. (New Castle, DE, USA), and stored in a desiccator [14]. For all of the experiments reported, the catalyst concentration was 0.00379 mol/L (based upon the number of active sites per μmol). To assess CO2’s effect on particle swelling, all Nafion NR50 was presoaked in methanol for at least 72 h before reaction.
- (iii)
- Reactor and reaction conditions: Nearly all of the reactions were performed in a stainless-steel stirred reactor (Supercritical Fluid Technologies, Inc. (Newark, DE, USA), High-Pressure Reactor, 100 mL). For each reaction, the catalyst and substrates were added directly into the reactor that was then sealed and heated to the desired temperature using the built-in heating jacket and controlled by an RXTrol Jr. integrated processor (Newark, DE, USA). The reactor was then pressurized with CO2 and stirred at 300 rpm to increase the interfacial area between triglyceride and methanol phases. Preliminary experiments indicate that this mixing speed is sufficient to minimize mass-transfer limitations within the reactor [15]. The conditions were maintained for 4 h when CO2 was vented through a restrictor valve. After the reaction, the venting CO2 was slowly sparged through isopropanol liquid to dissolve the reaction products. Then, this isopropanol was added to the liquids remaining in the reactor, which were dissolved in it. The resulting isopropanol was analyzed to determine concentrations of the reactor products. An internal standard was used to analytically compensate for any loss of isopropanol during sparging. All reactions were performed in at least duplicate with an initial substrate (corn oil) at loadings, depending on the methanol/triolein ratio. Note that for all loadings, the combined volumes of methanol and corn oil was 5.22 mL, giving a fixed volume of CO2.
- (iv)
- Analysis: Samples were analyzed by supercritical fluid chromatography-mass spectrometry (Waters® Acquity UPC2 with Xevo TQD Triple Quadrupole Mass Spectrometer (Milford, MA, USA) with an Acquity HSS C18 column (100 Å, 1.8 µm, 2.1 mm × 100 mm) and using a 1 µL sample volume. The column was held at 45 °C with a back pressure of 1500 psi. The mobile phase consisted of CO2 (A) and 90:10 acetonitrile: methanol (B). The elution gradient started at 15% B and increased linearly to 35% B in 3.5 min where it was held for 1 min before return to the starting conditions. The mass spectrometer was run in Atmospheric pressure chemical ionization (APCI+) mode with a desolvation temperature of 600 °C and N2 flow rate of 1000 L/h and cone flow of 40 L/h. The APCI voltages were 3.5 kV (corona) and 50 V (cone). Each FAME was identified using its [M-H]+ adduct and quantified using a calibration curve and analyzed in its linear range.
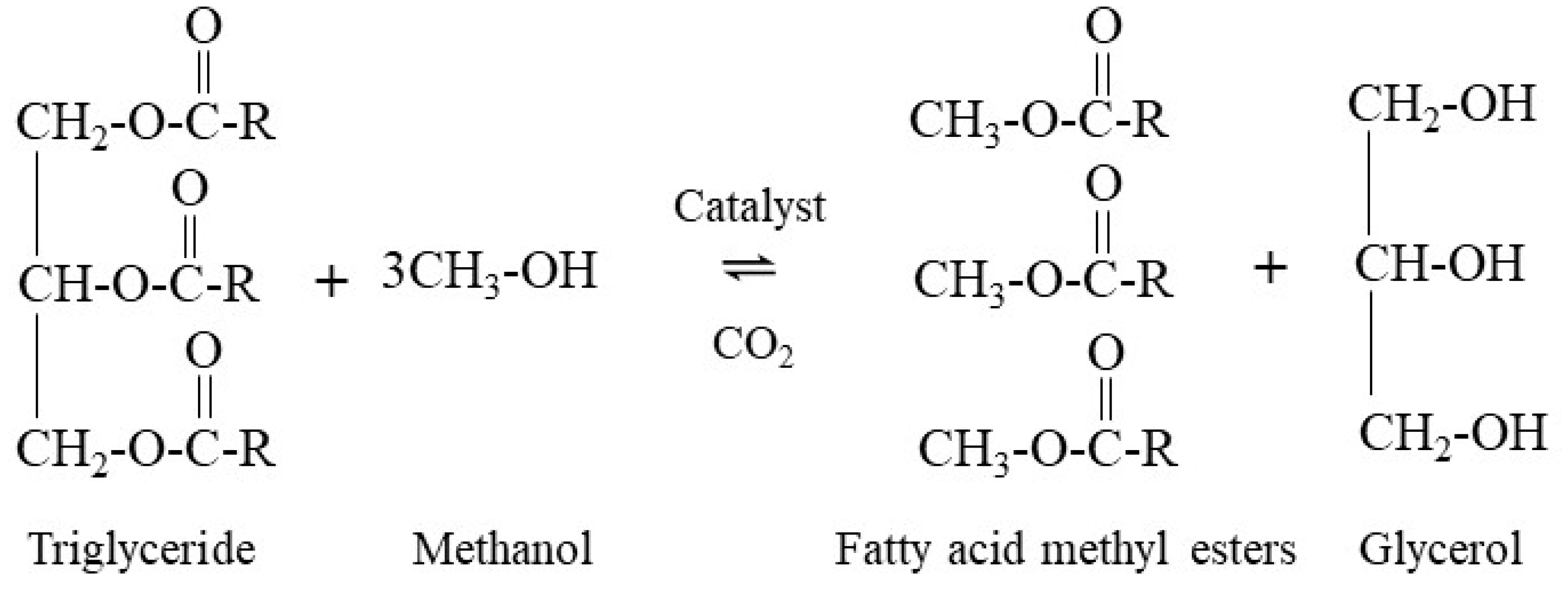
3. Multiphase Chemical Kinetics Modeling
3.1. Batch Reactor Model
3.2. RK-ASPEN Equations-of-State
4. Results and Discussion
4.1. Experimental Data with Simulated Results
4.2. Simulated Temperature and Pressure Variations
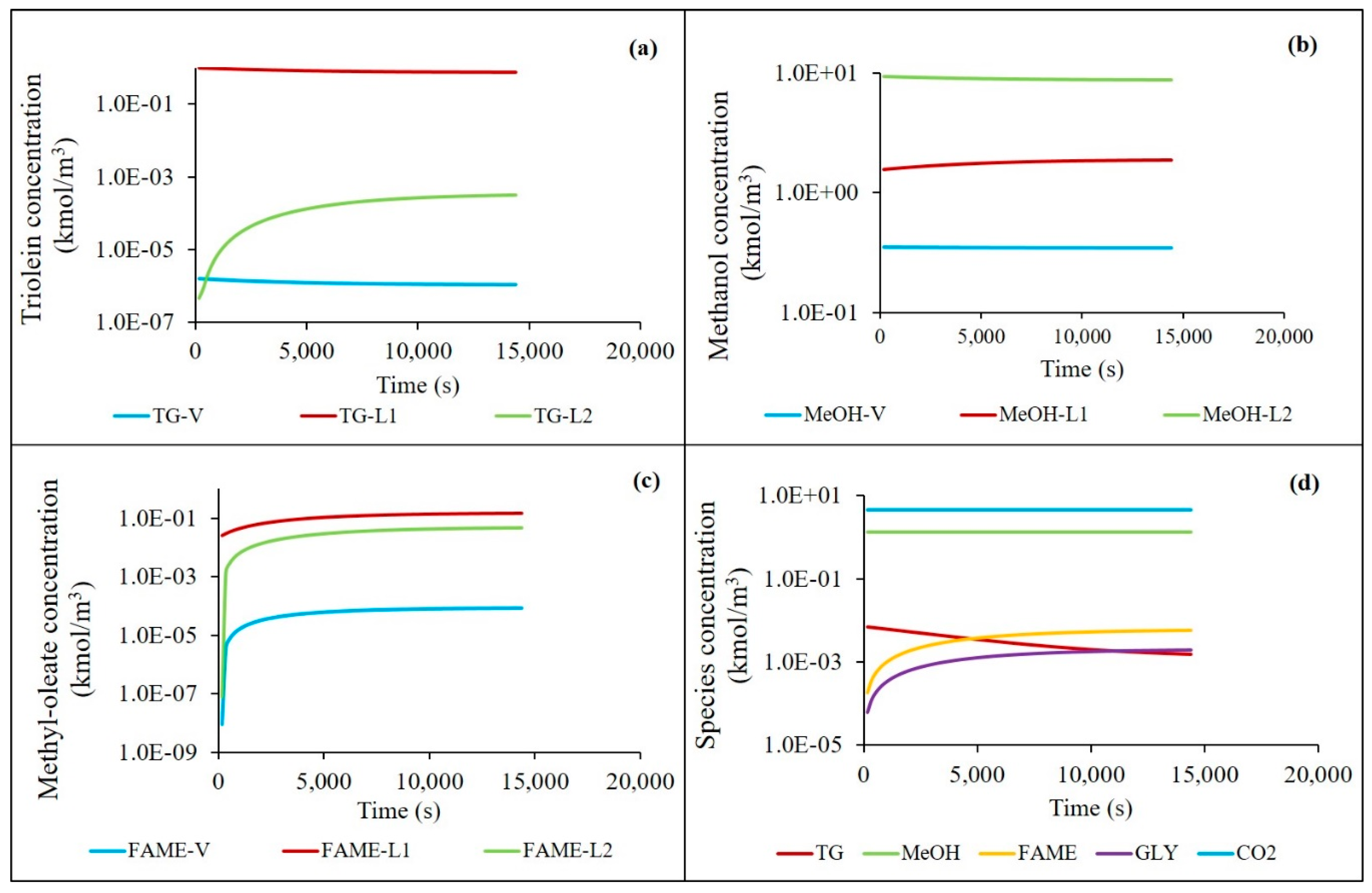
4.3. Simulated Batch Reactor Concentration Profiles
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Knothe, G. “Designer” Biodiesel: Optimizing Fatty Ester Composition to Improve Fuel Properties. Energy Fuels 2008, 22, 1358–1364. [Google Scholar] [CrossRef]
- Tian, Y.; Xiang, J.; Verni, C.C.; Soh, L. Fatty Acid Methyl Ester Production via Ferric Sulfate Catalyzed Interesterification. Biomass Bioenergy 2018, 115, 82–87. [Google Scholar] [CrossRef]
- Chang, A.-F.; Liu, Y.A. Integrated Process Modeling and Product Design of Biodiesel Manufacturing. Ind. Eng. Chem. Res. 2010, 49, 1197–1213. [Google Scholar] [CrossRef]
- Portha, J.; Allain, F.; Coupard, V.; Dandeu, A.; Girot, E.; Schaer, E.; Falk, L. Simulation and Kinetic Study of Transesterification of Triolein to Biodiesel Using Modular Reactors. Chem. Eng. J. 2012, 207–208, 285–298. [Google Scholar] [CrossRef]
- Kusdiana, D.; Saka, S. Kinetics of Transesterification in Rapeseed Oil to Biodiesel Fuel as Treated in Supercritical Methanol. Fuel 2001, 80, 693–698. [Google Scholar] [CrossRef]
- Hegel, P.; Mabe, G.; Pereda, S.; Brignole, E.A. Phase Transitions in a Biodiesel Reactor Using Supercritical Methanol. Ind. Eng. Chem. Res. 2007, 46, 6360–6365. [Google Scholar] [CrossRef]
- Maçaira, J.; Santana, A.; Costa, A.; Ramirez, E.; Larrayoz, M.A. Process Intensification Using CO2 as Cosolvent under Supercritical Conditions Applied to the Design of Biodiesel Production. Ind. Eng. Chem. Res. 2014, 53, 3985–3995. [Google Scholar] [CrossRef]
- Maçaira, J.; Santana, A.; Recasens, F.; Larrayoz, M.A. Biodiesel Production Using Supercritical Methanol/Carbon Dioxide Mixtures in a Continuous Reactor. Fuel 2011, 90, 2280–2288. [Google Scholar] [CrossRef]
- Soh, L.; Curry, J.; Beckman, E.J.; Zimmerman, J.B. Effect of System Conditions for Biodiesel Production via Transesterification Using Carbon Dioxide–Methanol Mixtures in the Presence of a Heterogeneous Catalyst. ACS Sustain. Chem. Eng. 2014, 2, 387–395. [Google Scholar] [CrossRef]
- Beckman, E.J. Oxidation Reactions in CO2: Academic Exercise or Future Green Processes? Environ. Sci. Technol. 2003, 37, 5289–5296. [Google Scholar] [CrossRef] [PubMed]
- Soh, L.; Chen, C.-C.; Kwan, T.A.; Zimmerman, J.B. Role of CO2 in Mass Transfer, Reaction Kinetics, and Interphase Partitioning for the Transesterification of Triolein in an Expanded Methanol System with Heterogeneous Acid Catalyst. ACS Sustain. Chem. Eng. 2015, 3, 2669–2677. [Google Scholar] [CrossRef]
- Silva, C.; Soh, L.; Barberio, A.; Zimmerman, J.; Seider, W.D. Phase Equilibria of Triolein to Biodiesel Reactor Systems. Fluid Phase Equilib. 2016, 409, 171–192. [Google Scholar] [CrossRef]
- Lepage, G.; Roy, C.C. Improved Recovery of Fatty Acid through Direct Transesterification without Prior Extraction or Purification. J. Lipid Res. 1984, 25, 1391–1396. [Google Scholar] [PubMed]
- Soh, L.; Montazeri, M.; Haznedaroglu, B.Z.; Kelly, C.; Peccia, J.; Eckelman, M.J.; Zimmerman, J.B. Evaluating Microalgal Integrated Biorefinery Schemes: Empirical Controlled Growth Studies and Life Cycle Assessment. Bioresour. Technol. 2014, 151, 19–27. [Google Scholar] [CrossRef]
- Soh, L.; Lane, M.K.M.; Xiang, J.; Kwan, T.A.; Zimmerman, J.B. Carbon Dioxide Mediated Transesterification of Mixed Triacylglyceride Substrates. Energy Fuels 2018, 32, 9624–9632. [Google Scholar] [CrossRef]
- Soave, G. Equilibrium Constants from a Modified Redlich-Kwong Equation of State. Chem. Eng. Sci. 1972, 27, 1197–1203. [Google Scholar] [CrossRef]
- Peng, D.-Y.; Robinson, D.B. A New Two-Constant Equation of State. Ind. Eng. Chem. Fundam. 1976, 15, 59–64. [Google Scholar] [CrossRef]
- Farobie, O.; Matsumura, Y. Biodiesel Production in Supercritical Methanol Using a Novel Spiral Reactor. Procedia Environ. Sci. 2015, 28, 204–213. [Google Scholar] [CrossRef]
- Rathore, V.; Madras, G. Synthesis of Biodiesel from Edible and Non-Edible Oils in Supercritical Alcohols and Enzymatic Synthesis in Supercritical Carbon Dioxide. Fuel 2007, 86, 2650–2659. [Google Scholar] [CrossRef]
- Bunyakiat, K.; Makmee, S.; Sawangkeaw, R.; Ngamprasertsith, S. Continuous Production of Biodiesel via Transesterification from Vegetable Oils in Supercritical Methanol. Energy Fuels 2006, 20, 812–817. [Google Scholar] [CrossRef]
- Tsai, Y.-T.; Lin, H.; Lee, M.-J. Biodiesel Production with Continuous Supercritical Process: Non-Catalytic Transesterification and Esterification with or without Carbon Dioxide. Bioresour. Technol. 2013, 145, 362–369. [Google Scholar] [CrossRef] [PubMed]
- Pollardo, A.A.; Lee, H.; Lee, D.; Kim, S.; Kim, J. Effect of Supercritical Carbon Dioxide on the Enzymatic Production of Biodiesel from Waste Animal Fat Using Immobilized Candida Antarctica Lipase B Variant. BMC Biotechnol. 2017, 17, 70. [Google Scholar] [CrossRef] [PubMed]
| Corn Oil (g) | MeOH (mL) | Molar Ratio MeOH/Corn Oil | Experiment 95 °C, 9.65 MPa FAME (g) | Simulation 95 °C, 9.65 MPa FAME (g) |
|---|---|---|---|---|
| 1.551 | 3.52 | 50X | 0.10 | 0.17 |
| 0.918 | 4.21 | 100X | 0.13 | 0.19 |
| 0.352 | 4.83 | 300X | 0.16 | 0.24 |
| 0.218 | 5 | 550X | 0.13 | 0.17 |
| Corn Oil (g) | MeOH (mL) | Molar Ratio MeOH/Triolein | Experiment 95 °C, 9.65 MPa Percent Yield [(FAME/Corn Oil) × 100] | Simulation 95 °C, 9.65 MPa Percent Yield [(FAME/Triolein) × 100] |
|---|---|---|---|---|
| 1.551 | 3.52 | 50X | 6.4 | 11.10 |
| 0.918 | 4.21 | 100X | 14.11 | 20.42 |
| 0.352 | 4.83 | 300X | 44.08 | 66.82 |
| 0.218 | 5 | 550X | 61.5 | 79.78 |
| Temp (°C) | Press (MPa) | Molar Ratio MeOH/Triolein 50× (FAME/triolein) × 100 | Molar Ratio MeOH/Triolein 100× (FAME/triolein) × 100 | Molar Ratio MeOH/Triolein 300× (FAME/triolein) × 100 | Molar Ratio MeOH/Triolein 550× (FAME/triolein) × 100 |
|---|---|---|---|---|---|
| 80 | 8 | 9.09 | 19.34 | 49.45 | 57.38 |
| 80 | 9.65 | 14.71 | 16.24 | 42.68 | 52.88 |
| 95 | 8 | 6.01 | 11.88 | 32.05 | 35.46 |
| 95 | 9.65 | 11.11 | 20.42 | 66.83 | 79.79 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yadav, G.; Fabiano, L.A.; Soh, L.; Zimmerman, J.; Sen, R.; Seider, W.D. Supercritical CO2 Transesterification of Triolein to Methyl-Oleate in a Batch Reactor: Experimental and Simulation Results. Processes 2019, 7, 16. https://doi.org/10.3390/pr7010016
Yadav G, Fabiano LA, Soh L, Zimmerman J, Sen R, Seider WD. Supercritical CO2 Transesterification of Triolein to Methyl-Oleate in a Batch Reactor: Experimental and Simulation Results. Processes. 2019; 7(1):16. https://doi.org/10.3390/pr7010016
Chicago/Turabian StyleYadav, Geetanjali, Leonard A. Fabiano, Lindsay Soh, Julie Zimmerman, Ramkrishna Sen, and Warren D. Seider. 2019. "Supercritical CO2 Transesterification of Triolein to Methyl-Oleate in a Batch Reactor: Experimental and Simulation Results" Processes 7, no. 1: 16. https://doi.org/10.3390/pr7010016
APA StyleYadav, G., Fabiano, L. A., Soh, L., Zimmerman, J., Sen, R., & Seider, W. D. (2019). Supercritical CO2 Transesterification of Triolein to Methyl-Oleate in a Batch Reactor: Experimental and Simulation Results. Processes, 7(1), 16. https://doi.org/10.3390/pr7010016