A Review on the Separation of Lithium Ion from Leach Liquors of Primary and Secondary Resources by Solvent Extraction with Commercial Extractants
Abstract
:1. Introduction
1.1. Applications and Resources
1.2. Lithium Recovery
2. Pretreatment and Leaching of Primary and Secondary Resources
3. Separation of Li(I) from Leach Liquors of Primary and Secondary Resources by Solvent Extraction
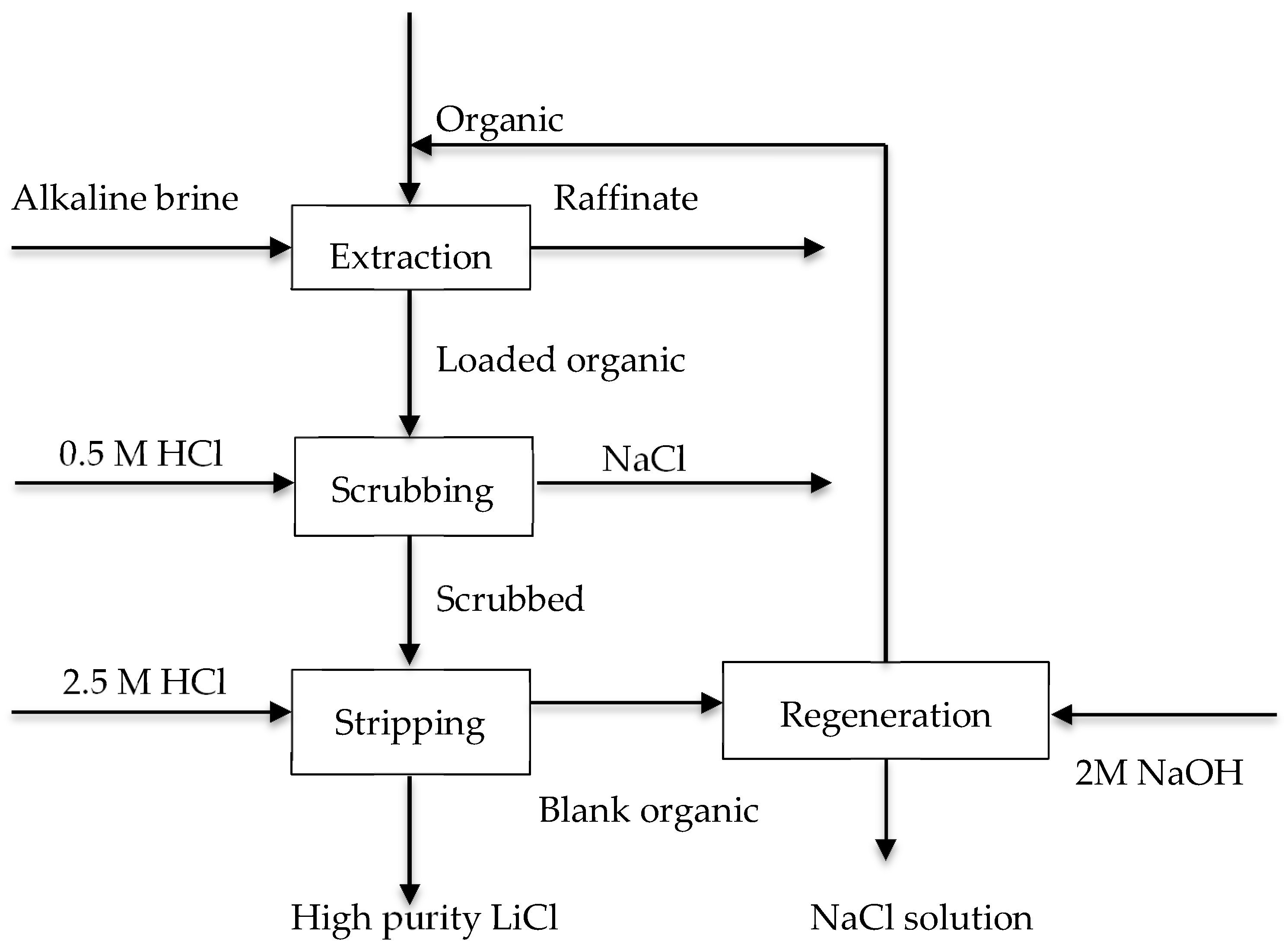
3.1. Selective Extraction of Li(I) from Brines/Alkaline Solutions
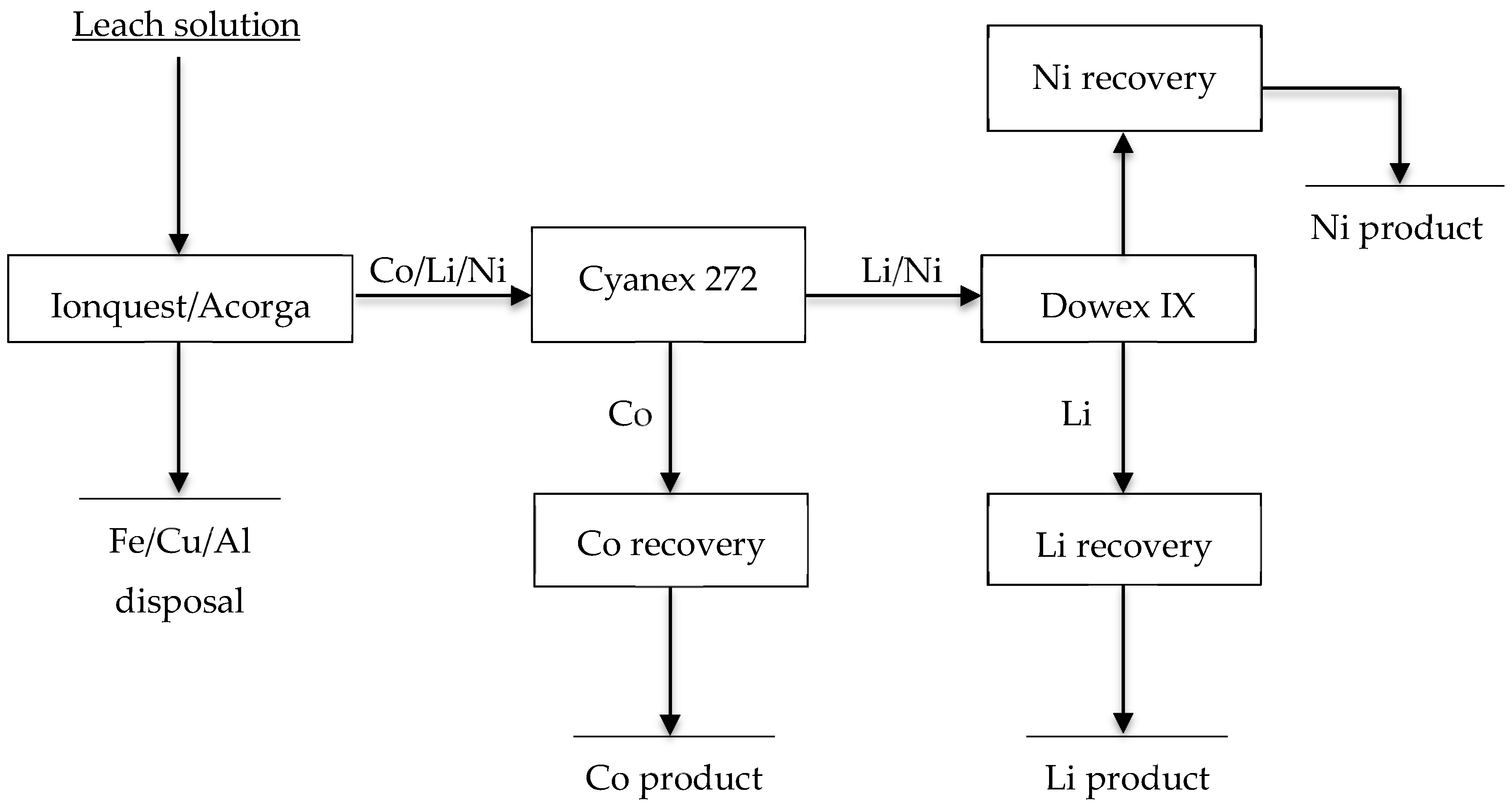
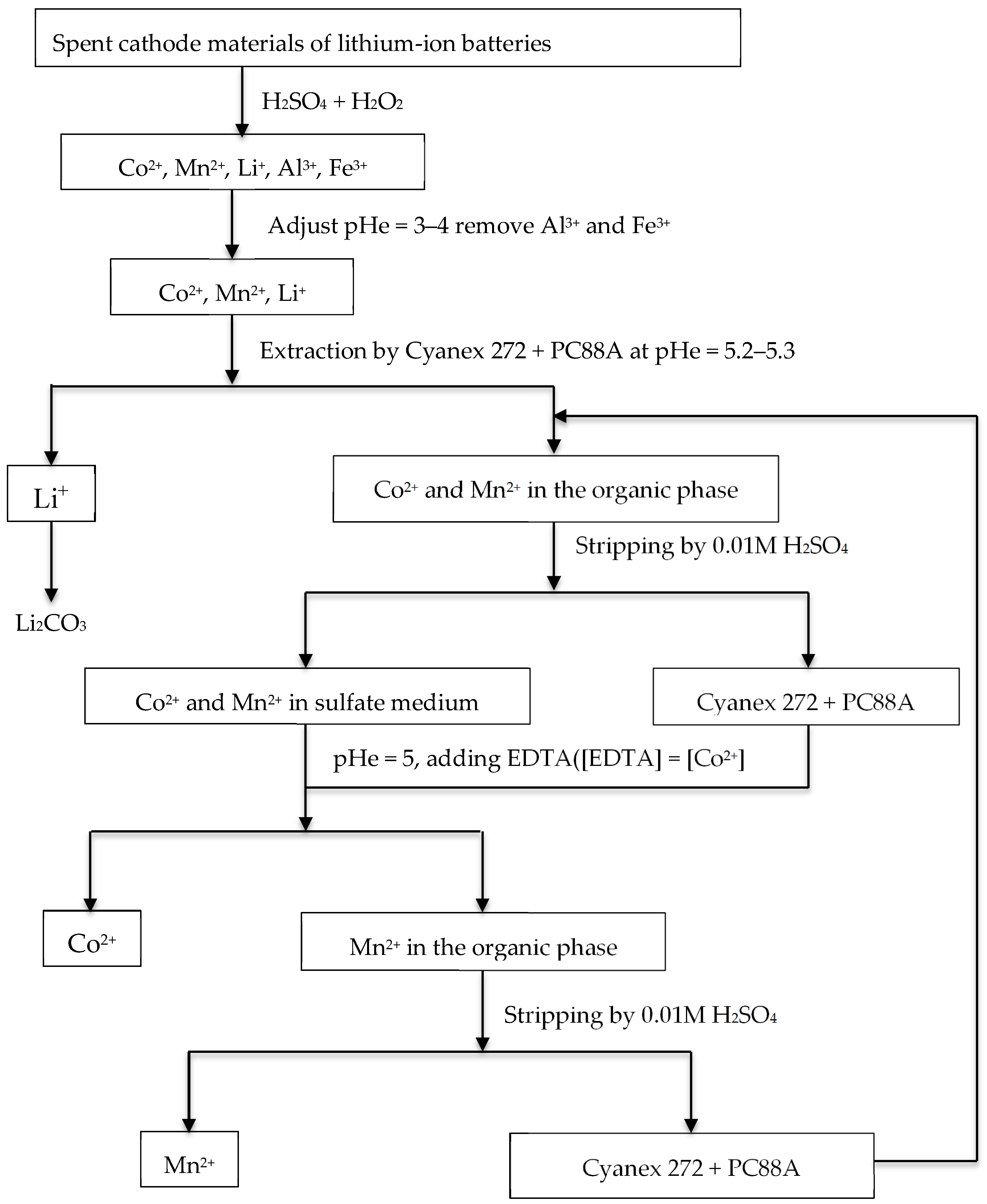
3.2. Selective Extraction and Recovery of Li(I) from Leach Liquors of Secondary Resources
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
Appendix A
| Extractants | Structure of the Compound |
|---|---|
| D2EHPA Di-2-ethylhexyl phosphoric acid | 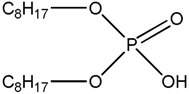 |
| PC88A 2-Ethylhexyl phosphonic acid mono-2-ethylhexyl ester | 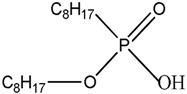 |
| MEHPA Mono-2-ethylhexyl phosphoric acid | 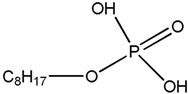 |
| Cyanex 272 Bis-2,4,4-trimethyl pentyl phosphinic acid | 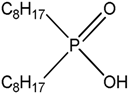 |
| LIX 54 -acetyl-m-dodecylacetophenone | 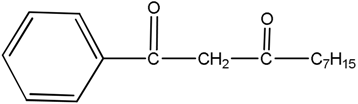 |
| MIBK Methyl isobutyl ketone | 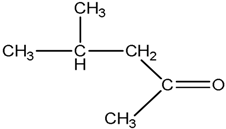 |
| TBP Tri-n-butyl phosphate | 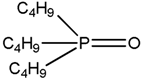 |
| TOPO Tri-n-octyl phosphine oxide | 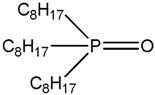 |
| Cyanex 923 Mixture of main trialkyl phosphine oxides |  R,R’ = [CH3(CH2)7]−normal octyl |
References
- Swain, B. Separation and purification of lithium by solvent extraction and supported liquid membrane, analysis of their mechanism: A review. J. Chem. Technol. Biotechnol. 2016, 91, 2549–2562. [Google Scholar] [CrossRef]
- Albema. Global Lithium Market Outlook. In Proceedings of the Goldman Sachs Houston Chemical Intensity Days Conference, Houston, TX, USA, 15 March 2016; Available online: https://www.scribd.com/document/341213533/HCID-Conference-ALB-Lithium-Presentation-v1-1. (accessed on 15 March 2016).
- Zhou, Z.; Qin, W.; Liu, Y.; Fei, W. Extraction equilibria of lithium with tributyl phosphate in kerosene and FeCl3. J. Chem. Eng. Data 2011, 57, 82–86. [Google Scholar] [CrossRef]
- Swain, B. Recovery and recycling of lithium: A review. Sep. Purif. Technol. 2017, 172, 88–403. [Google Scholar] [CrossRef]
- Liu, X.; Zhong, M.; Chen, X.; Zhao, Z. Separating lithium and magnesium in brine by aluminum-based materials. Hydrometallurgy 2018, 176, 73–77. [Google Scholar] [CrossRef]
- Wietelmann, U.; Steinbild, M. Lithium and Lithium Compounds. Ullmann’s Encycl. Ind. Chem. 2014, 1–38. [Google Scholar] [CrossRef]
- Kesler, S.E.; Gruber, P.W.; Medina, P.A.; Keoleian, G.A.; Everson, M.P.; Wallington, T.J. Global lithium resources: Relative importance of pegmatite, brine and other deposits. Ore Geol. Rev. 2012, 48, 55–69. [Google Scholar] [CrossRef]
- Swain, B.; Jeong, J.; Lee, J.C.; Lee, G.H.; Sohn, J.S. Hydrometallurgical process for recovery of cobalt from waste cathodic active material generated during manufacturing of lithium ion batteries. J. Power Sources 2007, 167, 536–544. [Google Scholar] [CrossRef]
- Nan, J.; Han, D.; Zuo, X. Recovery of metal values from spent lithium-ion batteries with chemical deposition and solvent extraction. J. Power Sources 2005, 152, 278–284. [Google Scholar] [CrossRef]
- Nayl, A.A.; Hamed, M.M.; Rizk, S.E. Selective extraction and separation of metal values from leach liquor of mixed spent Li-ion batteries. J. Taiwan Inst. Chem. Eng. 2015, 55, 119–125. [Google Scholar] [CrossRef]
- Ordoñez, J.; Gago, E.J.; Girardm, A. Processes and technologies for the recycling and recovery of spent lithium-ion batteries. Renew. Sustain. Energy Rev. 2016, 60, 195–205. [Google Scholar] [CrossRef]
- Meshram, P.; Pandey, B.D.; Mankhand, T.R. Extraction of lithium from primary and secondary sources by pre-treatment, leaching and separation: A comprehensive review. Hydrometallurgy 2014, 150, 192–208. [Google Scholar] [CrossRef]
- Maraghechi, H.; Rajabipour, F.; Pantano, C.G.; Burgos, W.D. Effect of calcium on dissolution and precipitation reactions of amorphous silica at high alkalinity. Cem. Concr. Res. 2016, 87, 1–13. [Google Scholar] [CrossRef]
- Harvianto, G.R.; Kim, S.H.; Ju, C.S. Solvent extraction and stripping of lithium ion from aqueous solution and its application to seawater. Rare Met. 2016, 35, 948–953. [Google Scholar] [CrossRef]
- Ji, L.; Hu, Y.; Li, L.; Shi, D.; Li, J.; Nie, F. Lithium Extraction with a Synergistic System of Dioctyl Phthalate and Tributyl Phosphate in Kerosene and FeCl3. Hydrometallurgy 2016, 162, 71–78. [Google Scholar] [CrossRef]
- An, J.W.; Kang, D.J.; Tran, K.T.; Kim, M.J.; Lim, T.; Tran, T. Recovery of lithium from Uyuni salar brine. Hydrometallurgy 2012, 117, 64–70. [Google Scholar] [CrossRef]
- Bukowsky, H.; Uhlemann, E.; Steinborn, D. The recovery of pure lithium chloride from “brines” containing higher contents of calcium chloride and magnesium chloride. Hydrometallurgy 1991, 27, 317–325. [Google Scholar] [CrossRef]
- Chitrakar, R.; Makita, Y.; Ooi, K.; Sonoda, A. Lithium recovery from salt lake brine by H2TiO3. Dalt. Trans. 2014, 43, 8933–8939. [Google Scholar] [CrossRef] [PubMed]
- Works, D.S. Extraction of lithium from the Dead Sea. Hydrometallurgy 1981, 6, 269–275. [Google Scholar]
- El-Eswed, B.; Sunjuk, M.; Al-Degs, Y.S.; Shtaiwi, A. Solvent Extraction of Li+ using Organophosphorus Ligands in the Presence of Ammonia. Sep. Sci. Technol. 2014, 49, 1342–1348. [Google Scholar] [CrossRef]
- Hano, T.; Matsumoto, M.; Ohtake, T.; Egashira, N.; Hori, F. Recovery of lithium from geothermal water by solvent extraction technique. Solvent Extr. Ion Exch. 1992, 10, 195–206. [Google Scholar] [CrossRef]
- Xiang, W.; Liang, S.; Zhou, Z.; Qin, W.; Fei, W. Lithium recovery from salt lake brine by counter-current extraction using tributyl phosphate/FeCl3 in methyl isobutyl ketone. Hydrometallurgy 2017, 171, 27–32. [Google Scholar] [CrossRef]
- Jandová, J.; Vu, H.N.; Belková, T.; Dvorák, P.; Kondás, J. Obtaining Li2CO3 from Zinnwaldite Wastes. Ceram-Silikáty 2009, 53, 108–112. [Google Scholar]
- Kondás, J.; Jandová, J. Lithium extraction from zinnwaldite wastes after gravity dressing of Sn-W ore. Acta Metall. Slovaca 2006, 12, 197–202. [Google Scholar]
- Siame, E.; Pascoe, R.D. Extraction of lithium from micaceous waste from china clay production. Miner. Eng. 2011, 24, 1595–1602. [Google Scholar] [CrossRef]
- Yan, Q.; Li, X.; Wang, Z.; Wu, X.; Wang, J.; Guo, H. Extraction of lithium from lepidolite by sulfation roasting and water leaching. Int. J. Miner. Process. 2012, 110, 1–5. [Google Scholar] [CrossRef]
- Sitando, O.; Crouse, P.L. Processing of a Zimbabwean petalite to obtain lithium carbonate. Int. J. Miner. Process. 2012, 102, 45–50. [Google Scholar] [CrossRef]
- Clarke, G.M. Lithium-ion batteries: Raw material considerations. Amer. Inst. Chem. Eng. 2013, 44–52. [Google Scholar]
- Jandová, J.; Dvorák, P.; Vu, H.N. Processing of zinnwaldite waste to obtain Li2CO3. Hydrometallurgy 2010, 103, 12–18. [Google Scholar] [CrossRef]
- Vu, H.; Bernardi, J.; Jandová, J.; Vaculíková, L.; Goliáš, V. Lithium and rubidium extraction from zinnwaldite by alkali digestion process: Sintering mechanism and leaching kinetics. Int. J. Miner. Process. 2013, 123, 9–17. [Google Scholar] [CrossRef]
- Chen, L.; Tang, X.; Zhang, Y.; Li, L.; Zeng, Z.; Zhang, Y. Process for the recovery of cobalt oxalate from spent lithium-ion batteries. Hydrometallurgy 2011, 108, 80–86. [Google Scholar] [CrossRef]
- Golmohammadzadeh, R.; Rashchi, F.; Vahidi, E. Recovery of lithium and cobalt from spent lithium-ion batteries using organic acids: Process optimization and kinetic aspects. Waste Manag. 2017, 64, 244–254. [Google Scholar] [CrossRef] [PubMed]
- Joulié, M.; Laucournet, R.; Billy, E. Hydrometallurgical process for the recovery of high value metals from spent lithium nickel cobalt aluminum oxide based lithium-ion batteries. J. Power Sources 2014, 247, 551–555. [Google Scholar] [CrossRef]
- Li, L.; Zhai, L.; Zhang, X.; Lu, J.; Chen, R.; Wu, F. Recovery of valuable metals from spent lithium-ion batteries by ultrasonic-assisted leaching process. J. Power Sources 2014, 262, 380–385. [Google Scholar] [CrossRef]
- Meshram, P.; Pandey, B.D.; Mankhand, T.R. Hydrometallurgical processing of spent lithium ion batteries (LIBs) in the presence of a reducing agent with emphasis on kinetics of leaching. Chem. Eng. J. 2015, 281, 418–427. [Google Scholar] [CrossRef]
- Nayl, A.A.; Elkhashab, R.A.; Badawy, S.M.; El-Khateeb, M.A. Acid leaching of mixed spent Li-ion batteries. Arab. J. Chem. 2017, 10, S3632–S3639. [Google Scholar] [CrossRef]
- Wang, R.C.; Lin, Y.C.; Wu, S.H. A novel recovery process of metal values from the cathode active materials of the lithium-ion secondary batteries. Hydrometallurgy 2009, 99, 194–201. [Google Scholar] [CrossRef]
- Shuva, M.A.H.; Kurny, A. Hydrometallurgical Recovery of Value Metals from Spent Lithium Ion Batteries. Am. J. Mater. Eng. Technol. 2013, 1, 8–12. [Google Scholar] [CrossRef]
- Yong-jia, L.; Ting, L. Hydrometallurgical Process for Recovery and Synthesis of LiCoO2 from Spent Lithium-ion Batteries. In Proceedings of the 2011 International Conference on Electric Technology and Civil Engineering (ICETCE), Lushan, China, 22–24 April 2011; pp. 6009–6011. [Google Scholar]
- Kang, J.; Senanayake, G.; Sohn, J.; Shin, S.M. Recovery of cobalt sulfate from spent lithium ion batteries by reductive leaching and solvent extraction with Cyanex 272. Hydrometallurgy 2010, 100, 168–171. [Google Scholar] [CrossRef]
- Dorella, G.; Mansur, M.B. A study of the separation of cobalt from spent Li-ion battery residues. J. Power Sources 2007, 170, 210–215. [Google Scholar] [CrossRef]
- Shin, S.M.; Kim, N.H.; Sohn, J.S.; Yang, D.H.; Kim, Y.H. Development of a metal recovery process from Li-ion battery wastes. Hydrometallurgy 2005, 79, 172–181. [Google Scholar] [CrossRef]
- Lee, C.K.; Rhee, K.I. Reductive leaching of cathodic active materials from lithium ion battery wastes. Hydrometallurgy 2003, 68, 5–10. [Google Scholar] [CrossRef]
- Li, L.; Ge, J.; Wu, F.; Chen, R.; Chen, S.; Wu, B. Recovery of cobalt and lithium from spent lithium ion batteries using organic citric acid as leachant. J. Hazard. Mater. 2010, 176, 288–293. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Lu, J.; Ren, Y.; Zhang, X.X.; Chen, R.J.; Wu, F. Ascorbic-acid-assisted recovery of cobalt and lithium from spent Li-ion batteries. J. Power Sources 2012, 218, 21–27. [Google Scholar] [CrossRef]
- Li, L.; Ge, J.; Chen, R.; Wu, F.; Chen, S.; Zhang, X. Environmental friendly leaching reagent for cobalt and lithium recovery from spent lithium-ion batteries. J. Hazard. Mater. 2010, 30, 2615–2621. [Google Scholar] [CrossRef] [PubMed]
- Zeng, X.; Li, J.; Shen, B. Novel approach to recover cobalt and lithium from spent lithium-ion battery using oxalic acid. J. Hazard. Mater. 2015, 295, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Li, L.; Shi, D.; Li, J.; Peng, X.; Nie, F. Selective extraction of lithium from alkaline brine using HBTA-TOPO synergistic extraction system. Sep. Purif. Technol. 2017, 188, 167–173. [Google Scholar] [CrossRef]
- Ishimori, K.; Imura, H.; Ohashi, K. Effect of 1,10-phenanthroline on the extraction and separation of lithium(I), sodium(I) and potassium(I) with thenoyltrifluoroacetone. Anal. Chim. Acta 2002, 454, 241–247. [Google Scholar] [CrossRef]
- Kim, Y.S.; In, G.; Choi, J.M. Chemical Equilibrium and Synergism for Solvent Extraction of Trace Lithium with Thenoyltrifluoroacetone in the Presence of Trioctylphosphine Oxide. Bull. Korean Chem. Soc. 2003, 24, 1495–1500. [Google Scholar] [CrossRef]
- Ji, L.; Li, L.; Shi, D.; Li, J.; Liu, Z.; Xu, D. Extraction equilibria of lithium with N,N-bis(2-ethylhexyl)-3-oxobutanamide and tributyl phosphate in kerosene and FeCl3. Hydrometallurgy 2016, 164, 304–312. [Google Scholar] [CrossRef]
- Zhou, Z.; Qin, W.; Fei, W.; Li, Y. A study on stoichiometry of complexes of tributyl phosphate and methyl isobutyl ketone with lithium in the presence of FeCl3. Chin. J. Chem. Eng. 2012, 20, 36–39. [Google Scholar] [CrossRef]
- Zhou, Z.; Qin, W.; Liu, Y.; Fei, W. Extraction equilibria of lithium with tributyl phosphate in Three Diluents. J. Chem. Eng. Data 2011, 56, 3518–3522. [Google Scholar] [CrossRef]
- Pranolo, Y.; Zhu, Z.; Cheng, C.Y. Separation of lithium from sodium in chloride solutions using SSX systems with LIX 54 and Cyanex 923. Hydrometallurgy 2015, 154, 33–39. [Google Scholar] [CrossRef]
- Kinugasa, T.; Nishibara, H.; Murao, Y.; Kawamura, Y.; Watanabe, K.; Takeuchi, H. Equilibrium and Kinetics of Lithium Extraction by a Mixture of LIX54 and TOPO. J. Chem. Eng. Jpn. 1994, 27, 815–818. [Google Scholar] [CrossRef]
- Zhou, Z.; Qin, W.; Chu, Y.; Fei, W. Elucidation of the structures of tributyl phosphate/Li complexes in the presence of FeCl3 via UV-visible, Raman and IR spectroscopy and the method of continuous variation. Chem. Eng. Sci. 2013, 101, 577–585. [Google Scholar] [CrossRef]
- Zhou, Z.; Qin, W.; Fei, W.; Liu, Y. A study on stoichiometry of complexes of tributyl phosphate and metyl isobutyl ketone with lithium in the presence of FeCl3. Chin. J. Chem. Eng. 2012, 20, 36–39. [Google Scholar] [CrossRef]
- Harvianto, G.R.; Jeong, S.G.; Ju, C.S. The effect of dominant ions on solvent extraction of lithium ion from aqueous solution. Korean J. Chem. Eng. 2014, 31, 828–833. [Google Scholar] [CrossRef]
- Lupi, C.; Pasquali, M.; Dell’Era, A. Nickel and cobalt recycling from lithium-ion batteries by electrochemical processes. Waste Manag. 2005, 25, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Mantuano, D.P.; Dorella, G.; Elias, R.C.A.; Mansur, M.B. Analysis of a hydrometallurgical route to recover base metals from spent rechargeable batteries by liquid-liquid extraction with Cyanex 272. J. Power Sources 2006, 159, 1510–1518. [Google Scholar] [CrossRef]
- Swain, B.; Mishra, C.; Jeong, J.; Lee, J.C.; Hong, H.S.; Pandey, B.D. Separation of Co(II) and Li(I) with Cyanex 272 using hollow fiber supported liquid membrane: A comparison with flat sheet supported liquid membrane and dispersive solvent extraction process. Chem. Eng. J. 2015, 271, 61–70. [Google Scholar] [CrossRef]
- Devi, N.B.; Nathsarma, K.C.; Chakravortty, V. Sodium salts of D2EHPA, PC-88A and Cyanex-272 and their mixtures as extractants for cobalt(II). Hydrometallurgy 1994, 34, 331–342. [Google Scholar] [CrossRef]
- Zhao, J.M.; Shen, X.Y.; Deng, F.L.; Wang, F.C.; Wu, Y.; Liu, H.Z. Synergistic extraction and separation of valuable metals from waste cathodic material of lithium ion batteries using Cyanex272 and PC-88A. Sep. Purif. Technol. 2011, 78, 345–351. [Google Scholar] [CrossRef]
- Chen, X.; Chen, Y.; Zhou, T.; Liu, D.; Hu, H.; Fan, S. Hydrometallurgical recovery of metal values from sulfuric acid leaching liquor of spent lithium-ion batteries. Waste Manag. 2015, 38, 349–356. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Yokoyama, T.; Itabashi, O.; Wakui, Y.; Suzuki, T.M.; Inoue, K. Hydrometallurical process for recovery of metal values from spent nickel-metal hydride secondary batteries. Hydrometallurgy 1998, 50, 61–75. [Google Scholar] [CrossRef]
- Pranolo, Y.; Zhang, W.; Cheng, C.Y. Hydrometallurgy Recovery of metals from spent lithium-ion battery leach solutions with a mixed solvent extractant system. Hydrometallurgy 2010, 102, 37–42. [Google Scholar] [CrossRef]
| Applications | Market Size | Lithium and Lithium Compounds | |
|---|---|---|---|
| Traditional uses | Glass/ceramics | 46 kt | • Spodumene • Li2CO3 |
| Greases/lubricants | 18 kt | • LiOH | |
| Chemical synthesis | 11 kt | • Li organometallics fed by Li metal LiCl | |
| Energy | Portable electronics and other handheld devices | 48 kt | • BG Li2CO3 • BG LiOH • BG Li metal • BG electrolyte salts • BG LiCl • BG alloys • BG specialty compounds |
| Plug-in hybrid and hybrid electric vehicles | |||
| Battery electric vehicles (BEVs) | |||
| Grid and other power storage applications | |||
| Ores/Clays | Pretreatment and Leaching Condition | Li Leaching, % | Ref. | |
|---|---|---|---|---|
| Calcination and Roasting | Water Leaching | |||
| Petalite | Calcination: 1050–1100 °C Roasting: 93% H2SO4; 250 °C; 1 h | - | 85 | [6] |
| Calcination: 1100 °C; 2 h Roasting: H2SO4; 300 °C; 1 h | S/L = 1/7.5; 320 rpm; 1 h; 50 °C | 97 | [27] | |
| Spodumene | Calcination: 1050–1090 °C, 0.5 h Roasting: H2SO4 | S/L = 4; 225 °C; 1 h | 96 | [28] |
| Zinnwaldite | Roasting: CaSO4 + Ca(OH)2; 950 °C; 1 h | S/L:1/10; 10 min; 90 °C | 96 | [23] |
| Roasting: CaCO3; 825 °C; 1 h | S/L: 1/5; 1 h; 90–95 °C | 85 | [29] | |
| Roasting: CaSO4 + Ca(OH)2; 975 °C | S/L:1/5; 1 h; 90 °C | 93 | [24] | |
| Roasting: CaCO3; 825 °C; 1 h | S/L: 1/10, 400 rpm; 4 h, 95 °C | 84 | [30] | |
| Clay | Roasting: CaSO4; 1050 °C, 1 h Roasting: Na2SO4; 850 °C, 1 h | S/L: 1/10; 10 min; 85 °C | 8497 | [25] |
| Lepidolite | Roasting: Na2SO4 + K2SO4 + CaO; 850 °C, 0.5 h | S/L: 1/2.5; 0.5 h; room temperature | 92 | [26] |
| Type of Acid | Leaching Condition | Leaching Efficiency, % | Ref. | |
|---|---|---|---|---|
| Li(I) | Others | |||
| Inorganic acids | Ultrasonic power: 90 W 2M HCl/H2SO4; S/L: 1/40; 5 h; 60 °C | 97 (H2SO4) 98 (HCl) | Co: 48 (H2SO4) Co: 76 (HCl) | [34] |
| 2M HCl/H2SO4/HNO3; S/L:5%w/v; 18h; 25 °C | 80 (HCl) <80 (H2SO4) >80 (HNO3) | Co, Ni, Al: >60 (HCl) Co, Ni, Al: 40 (H2SO4) Co, Ni, Al: 40 (HNO3) | [33] | |
| 4M HCl; S/L: 1/50; 1 h; 80 °C | >99% | Co, Mn, Ni: >99 | [37] | |
| 3M HCl + 3.5%v/v H2O2; S/L: 1/20; 1 h; 80 °C | 89% | Co: 89% | [38] | |
| 1M H2SO4 + 30%v/v H2O2; S/L: 1/1.4; 2 h; 80 °C | - | Co: 88 | [39] | |
| 2M H2SO4 + 6%v/v H2O2; S/L:1:10; 1 h; 60 °C; 300 rpm | - | Co: >99% | [40] | |
| 2M H2SO4 + 5%v/v H2O2; S/L: 1/10; 0.5 h; 75 °C | 94 | Co: 93 | [8] | |
| 6%v/v H2SO4 + 5%v/v H2O2; S/L: 3/10; 1 h; 65 °C | 95 | Co: 80 Al: 55 | [41] | |
| 2M H2SO4 + 15%v/v H2O2; S/L: 1:20; 10 min; 75 °C; 300 rpm | 100 | Co: 95 | [42] | |
| 4M H2SO4 + 10%v/v H2O2; S/L: 1/10; 2 h; 85 °C | 96 | Co: 95 | [31] | |
| 2M H2SO4 + 4%v/v H2O2; S/L: 1:10; 2 h; 70 °C | 99 | Co: 100 Ni: 99 Mn: 98 | [36] | |
| 1M H2SO4 + 0.0075M NaHSO3; S/L: 1/50; 4 h; 95 °C | 97 | Co: 92 Ni:96 Mn:88 | [35] | |
| 1M HNO3 + 1.7%v/v H2O2; S/L: 1:50; 1 h; 75 °C | 95 | Co: 95 | [43] | |
| Organic acids | 2M citric acid + 1.25%v/v H2O2 S/L: 3/100; 2 h; 60 °C | 92 | Co: 81 | [32] |
| Ultrasonic power: 90 W 2M citric acid + 0.55M H2O2; S/L: 1/40; 5 h; 60 °C | 98 | Co: 96 | [34] | |
| 1.25M citric acid + 1%v/v H2O2; S/L: 1/40; 0.5 h; 90 °C; 300 rpm | 100 | Co: >90 | [44] | |
| 1.25M ascorbic acid; S/L: 1/40; 20 min; 70 °C | 99 | Co: 95 | [45] | |
| 1.5M DL-malic acid+ 2%v/v H2O2; S/L: 1/40; 40 min; 90 °C | 100 | Co: 90 | [46] | |
| 1M oxalic acid; S/L: 1/66.7; 2.5 h; 95 °C; 400 rpm | 98 | Co: 97 | [47] | |
| Extractants | Condition | Remarks | Ref. |
|---|---|---|---|
| HBTA-TOPO | Li(I): 0.14 g/L pH = 11.2 | 97% of Li(I) was extracted with form complexes of Li.2BTA.TOPO; scrubbing with 0.5M HCl; stripping of Li(I) with 2.5M HCl; regeneration of the organic phase was achieved by washing with NaOH | [48] |
| Thenoyltrifluoracetone (TTA)-TOPO in kerosene | Li(I): 1 mg/L pH = 10.6 | Mg2+ had a strong effect on Li(I) extraction; 70% Li(I) was extracted from Mg(II)-free aqueous solution | [48] |
| TTA-1, 10-phenanthroline (Phen) in chlorobenzene | Li(I): 0.01–0.1 mol/L pH = 6.5–11.6 | Li(I) was extracted in the wide phen concentration while the extraction of K(I) and Na(I) was only possible in a high phen concentration | [49] |
| TTA-TOPO in m-xylene/MIBK/n-henxane/benzene/chloroform | Li(I): 5.8.10−4M NH4Cl: 0.1M, pH = 9 | Extraction efficiency of Li(I) followed the sequence: m-xylene > benzene > MIBK > n-hexane > chlorofrom; extracted sepecies were Li.TTA.2TOPO | [50] |
| -carbonyl amide (NB2EHOTA)-TBP-FeCl3 | HCl: 0.05M; Li(I): 2 g/L Fe(III)/Li(I):1.3 MgCl2: 4.8M | Separation factor of Li(I)/Mg(II) was higher than 450; extracted sepecies were (LiFeCl4.2TBP.NB2EHOTA).4TBP.NB2EHOTA | [51] |
| Dioctyl phthalate (DOP)/ acetyl tributyl citrate(ATBC)/ tri-n-butyl citrate(TBC)-TBP-FeCl3 | HCl: 0.05 Li(I): 1.86 g/L Fe(III)/Li(I):1.3 MgCl2: 4.8 | Li(I) extraction efficiency was in the order of DOP > ATBC > TBP;after three stages, 99.5% Li was extracted with extracted species of LiFeCl4.2TBP.0.1DOP; separation factors of Li(I)/Mg(II), Li(I)/Na(I) and Li(I)/K(I) were 31,458, 1259 and 16,508, respectively | [15] |
| TBP/MIBK-FeCl3-keosene | LiCl: 0.025–0.05 mol/L MgCl2: 3.5–4 mol/L FeCl3: 0.025–0.09 mol/L | Extracted species were LiFeCl4.TBP and LiFeCl4.2MIBK | [52] |
| TBP -FeCl3-keosene | Li(I): 0.2 mol/L Fe(III)/Li(I): 1.0–1.9 MgCl2/CaCl2/NH4Cl | Fe(III) extraction was a precondition of Li(I) extraction; the extraction efficiency of Li(I) followed the sequence: MgCl2 > CaCl2 > NH4Cl; MgCl2 at Fe(III)/Li(I) = 1.9 was the optimum condition for Li(I) extraction | [3] |
| TBP-MIBK-FeCl3 | Li(I): 0.05 mol/L Mg(II): 4.74 mol/L SO42−: 0.12 mol/L Cl– 9.43 mol/L | 98% Li(I) was extracted at a high Mg(II)/Li(I) molar ratio; Mg(II) scrubbing with LiCl/NaCl; Li(I) stripping with HCl/NaCl; regeneration of the organic phase was obtained by washing with NaOH/NaCl | [53] |
| α-acetyl-m-dodecylacetophenone (LIX 54) - a mixture of four trialkylphosphine oxides (Cyanex 923) | pH = 11 Li(I): 1 g/L Na(I): 20–80 g/L | High separation of Li(I) at a high Na(I) concentration (SF = 110–1500); 95% of Li(I) was extracted after three stages; extracted species was LiR Cyanex 923 | [54] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen, T.H.; Lee, M.S. A Review on the Separation of Lithium Ion from Leach Liquors of Primary and Secondary Resources by Solvent Extraction with Commercial Extractants. Processes 2018, 6, 55. https://doi.org/10.3390/pr6050055
Nguyen TH, Lee MS. A Review on the Separation of Lithium Ion from Leach Liquors of Primary and Secondary Resources by Solvent Extraction with Commercial Extractants. Processes. 2018; 6(5):55. https://doi.org/10.3390/pr6050055
Chicago/Turabian StyleNguyen, Thi Hong, and Man Seung Lee. 2018. "A Review on the Separation of Lithium Ion from Leach Liquors of Primary and Secondary Resources by Solvent Extraction with Commercial Extractants" Processes 6, no. 5: 55. https://doi.org/10.3390/pr6050055
APA StyleNguyen, T. H., & Lee, M. S. (2018). A Review on the Separation of Lithium Ion from Leach Liquors of Primary and Secondary Resources by Solvent Extraction with Commercial Extractants. Processes, 6(5), 55. https://doi.org/10.3390/pr6050055






