Kinetic control of aqueous polymerization using radicals generated in different spin states
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Polymer Synthesis
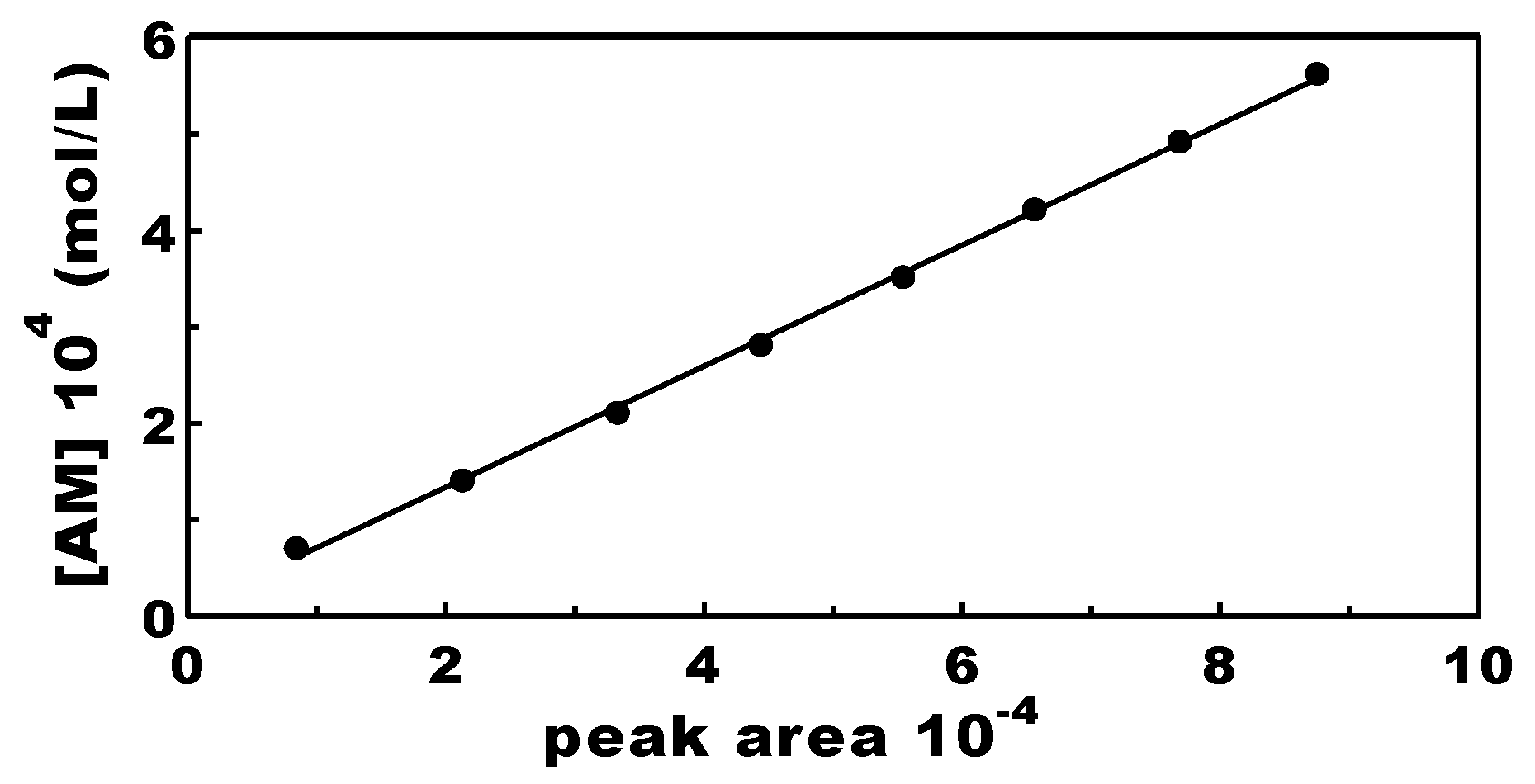
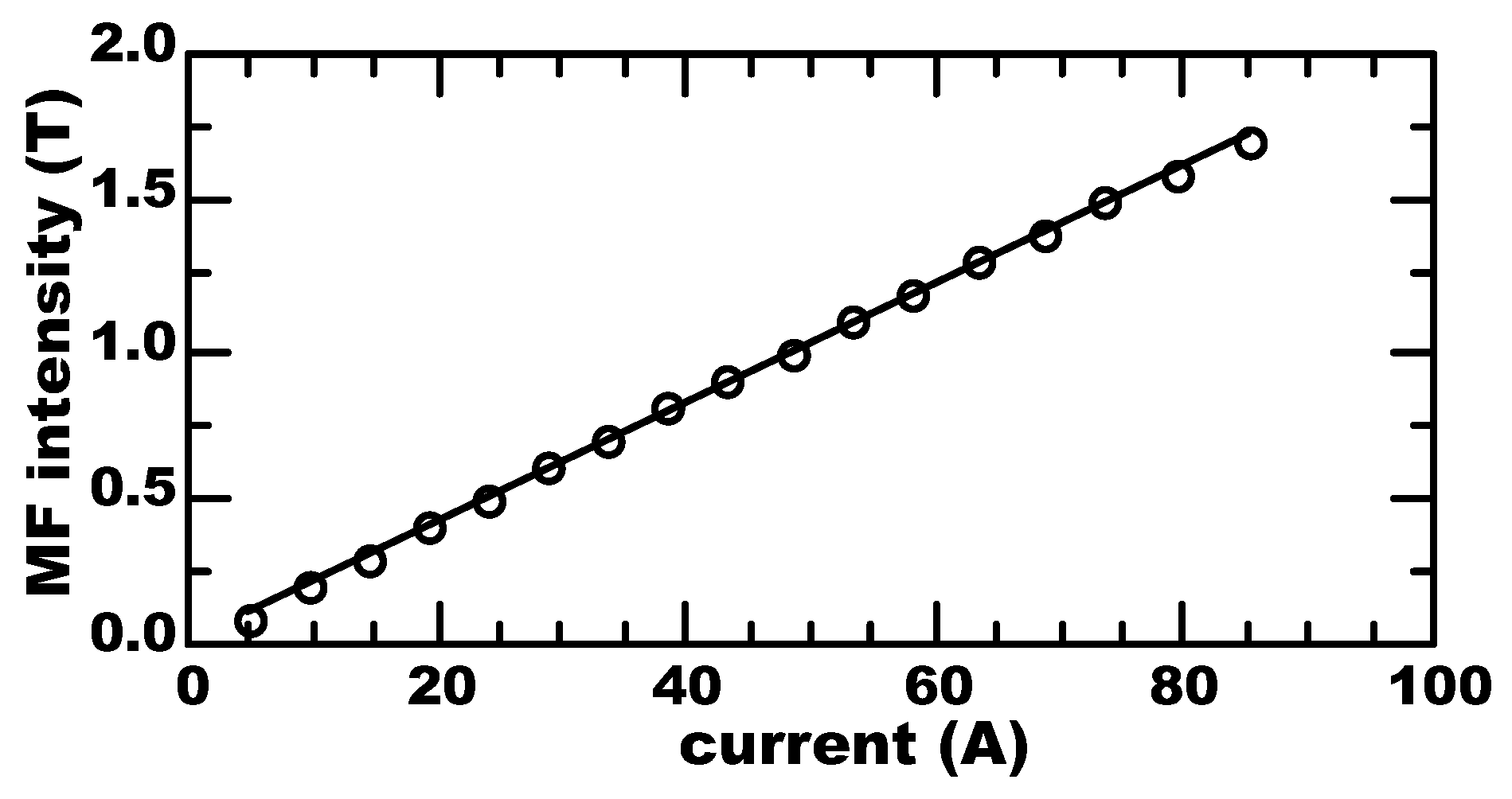
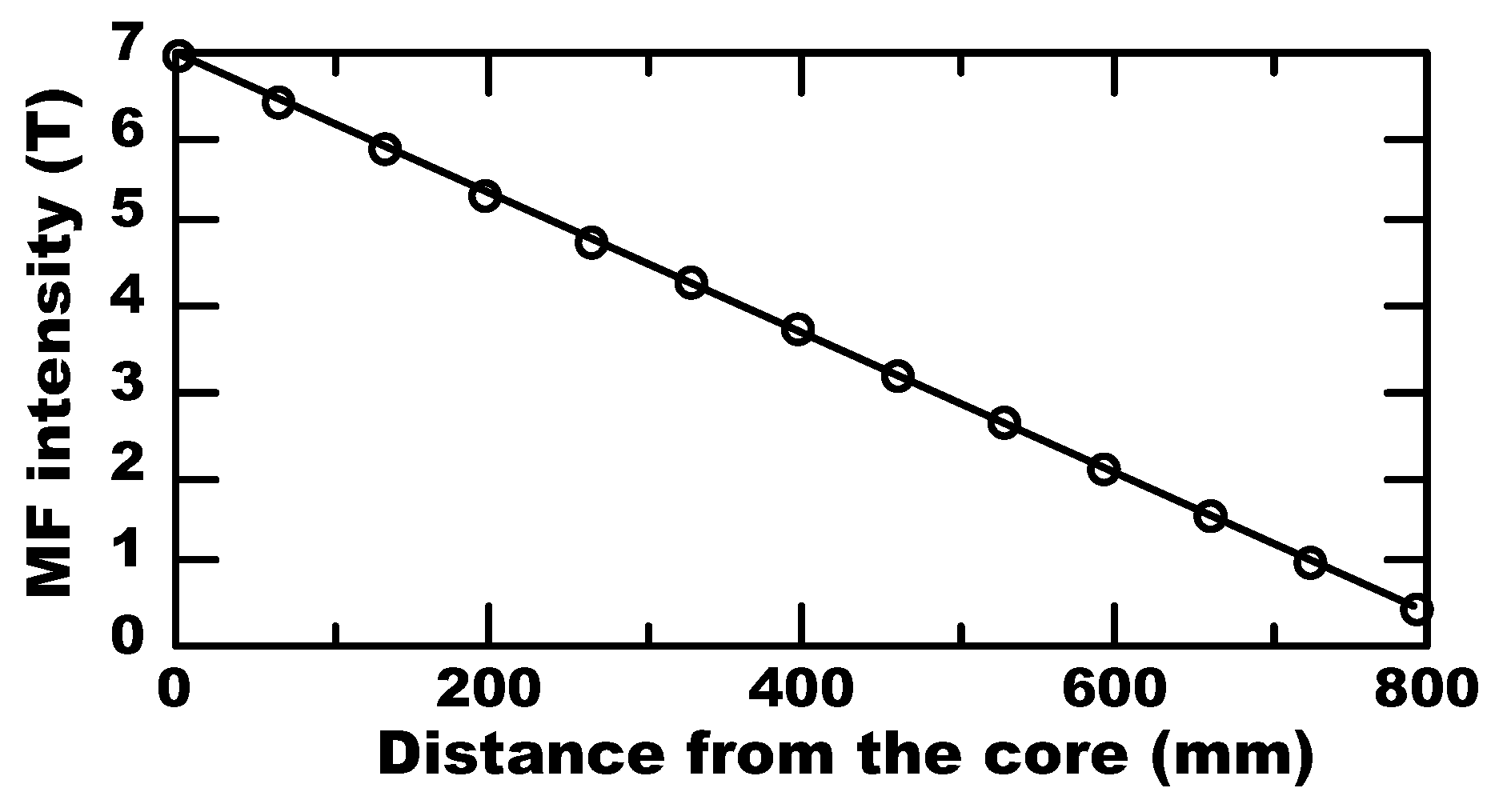
2.3. Analytics and Instruments Calibration
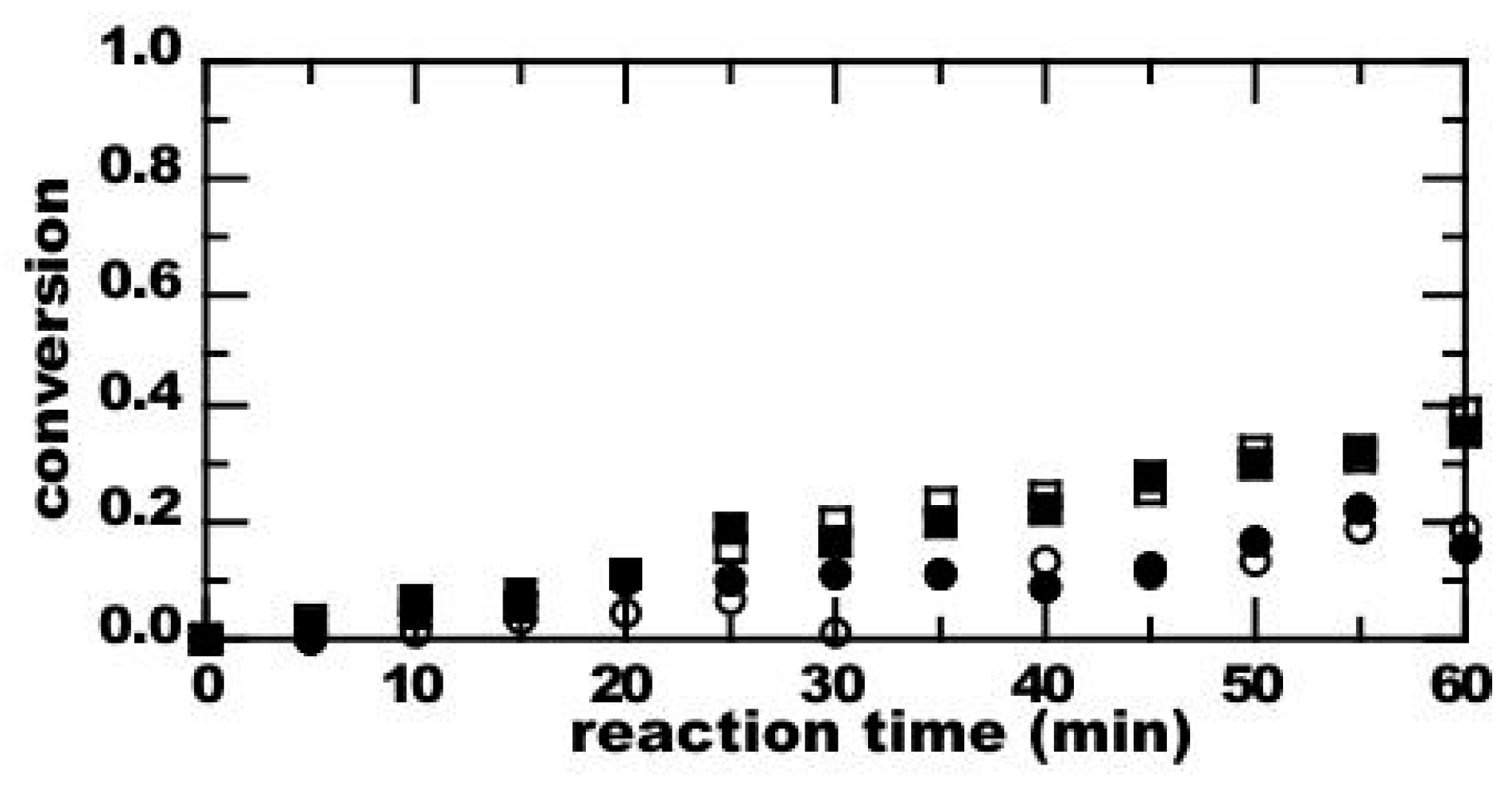
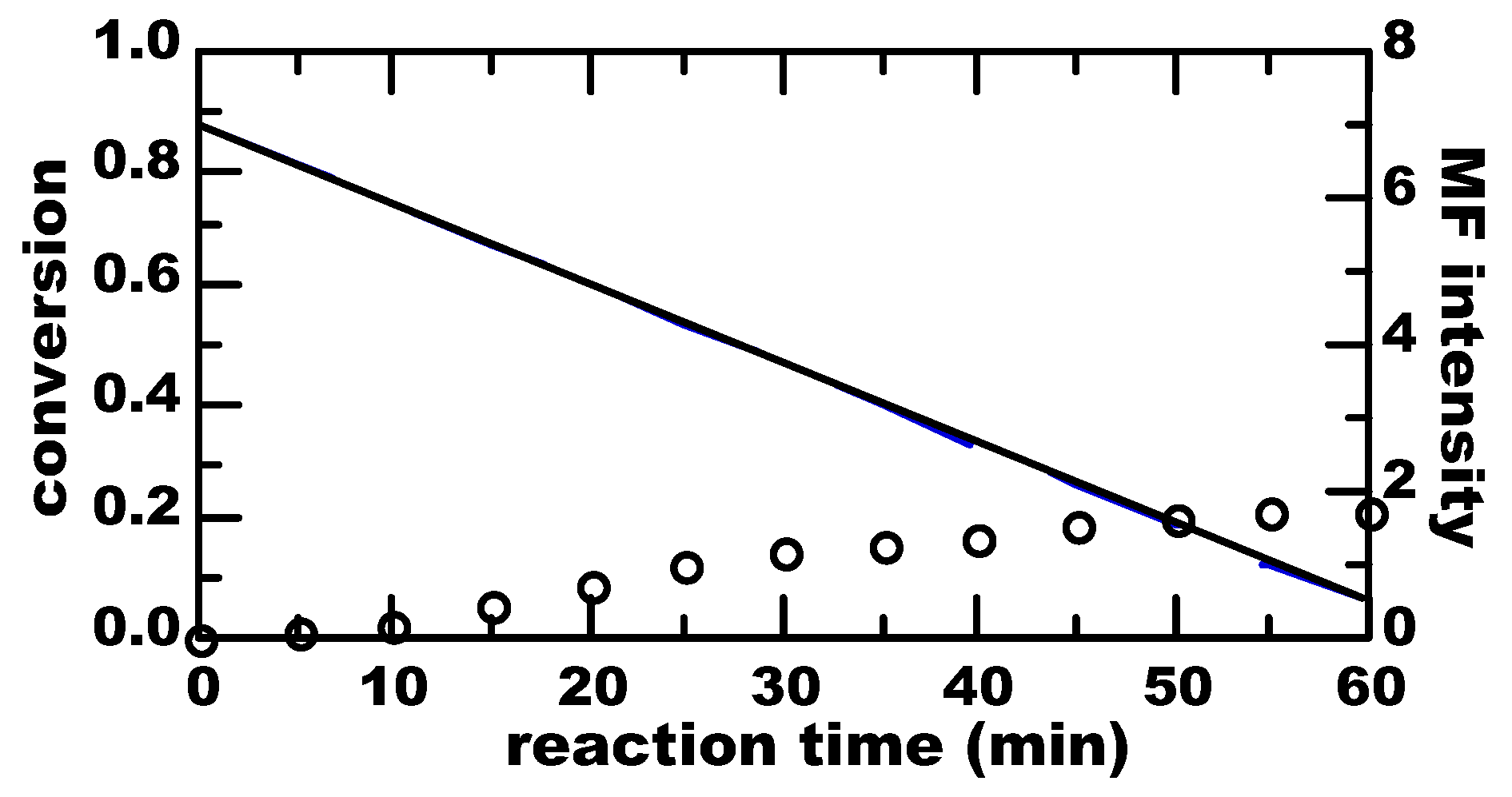
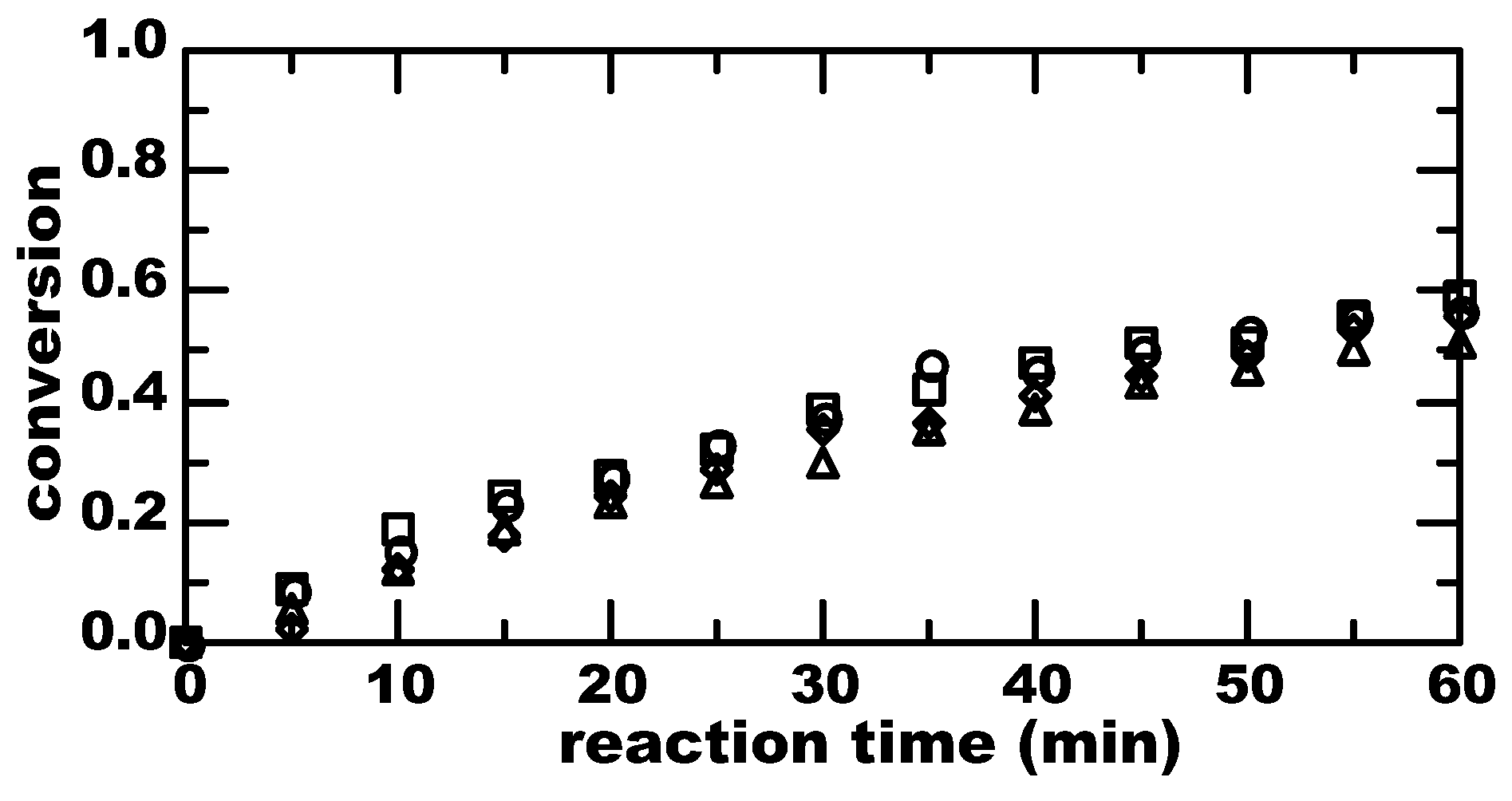
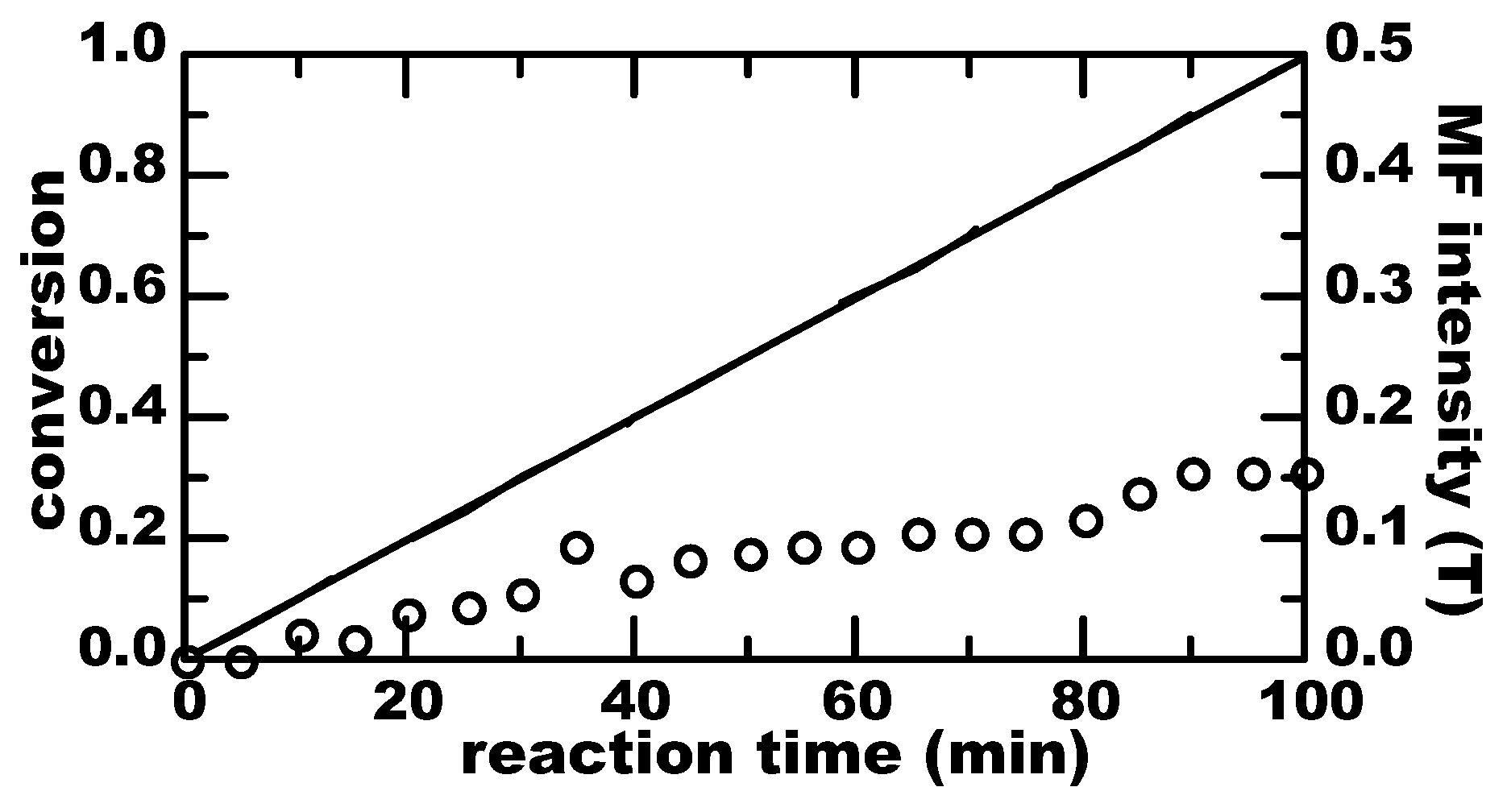
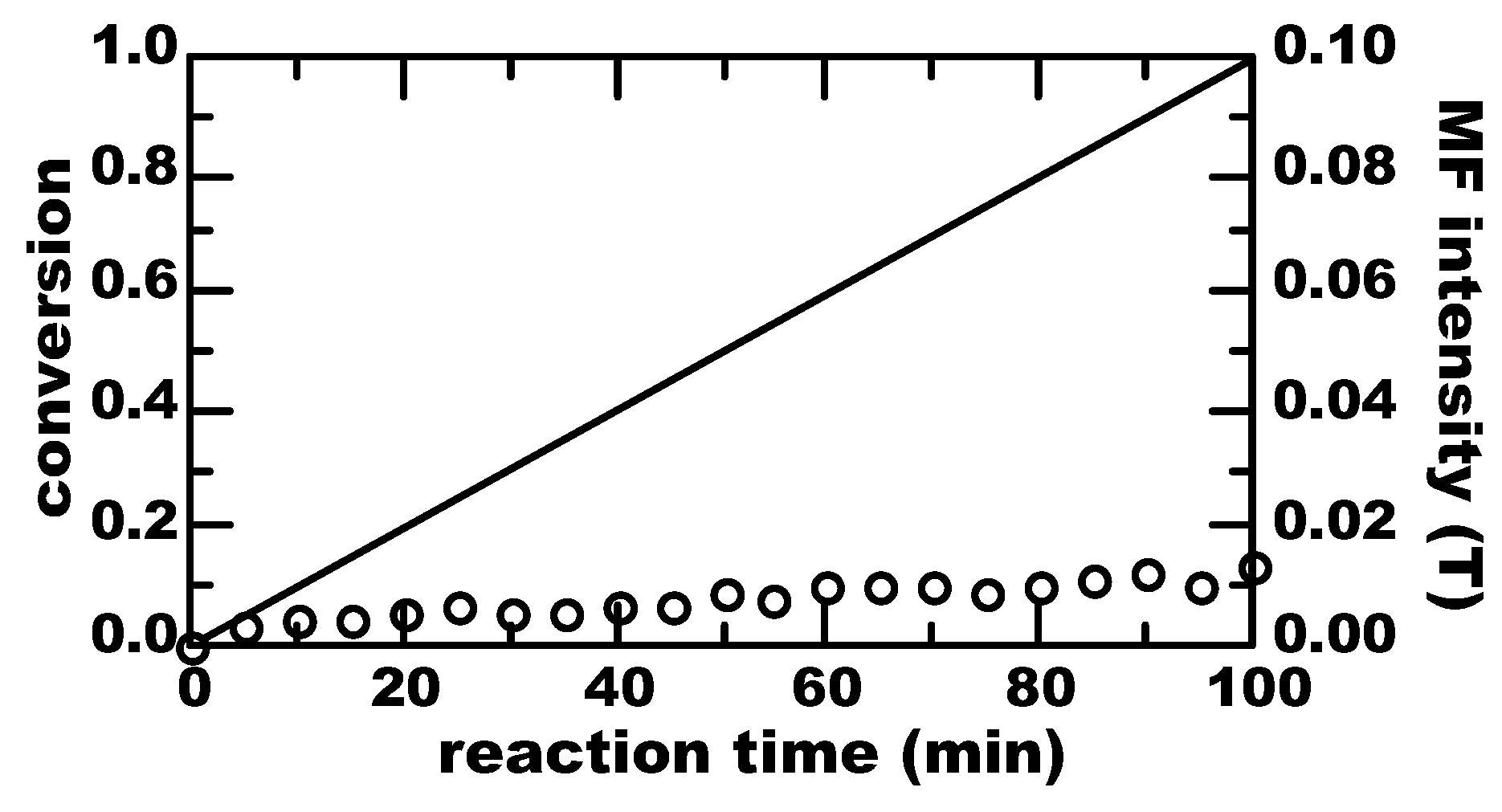
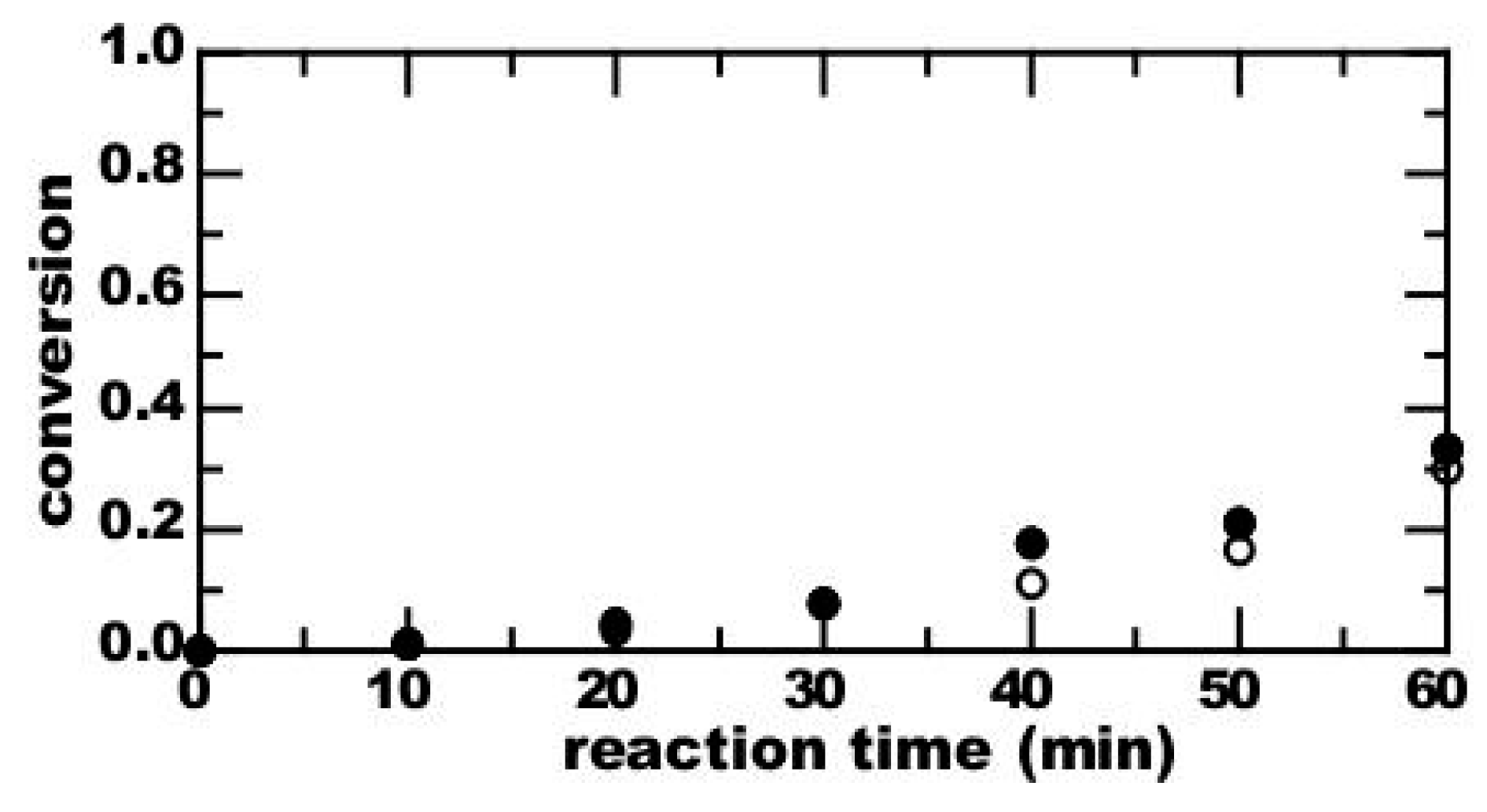
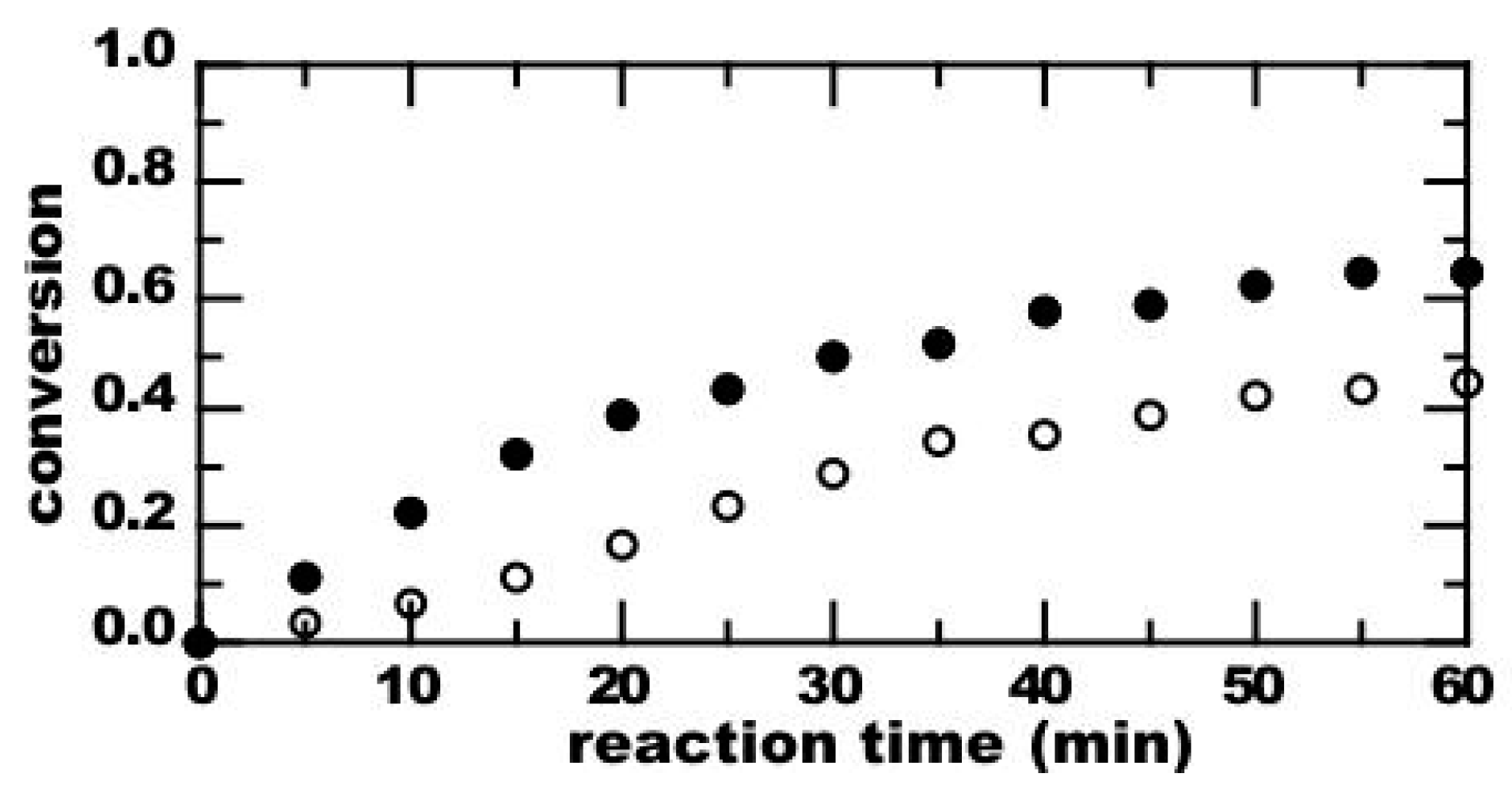
3. Results
4. Discussion
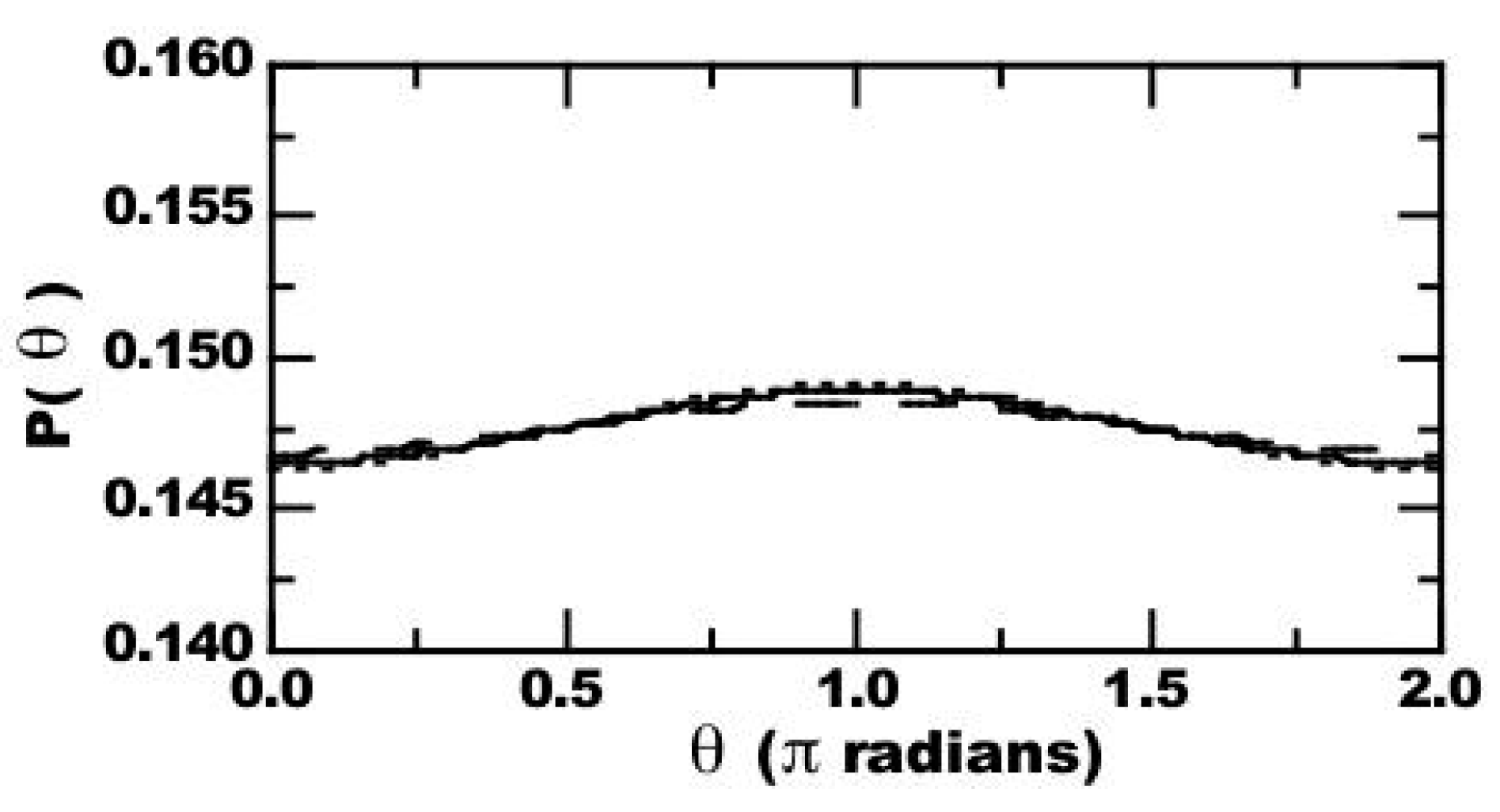
4.1. Thermal Equilibrium of Spin States
4.2. Magnetically-Induced Molecular Orientation
4.3. Radical Pair Mechanism
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bhatagnar, S.S.; Mathur, R.N.; Kapur, R.N. The effects of a magnetic field on certain chemical reactions. Philos. Mag. 1929, 8, 457–473. [Google Scholar] [CrossRef]
- Selwood, P.W. Magnetism and catalysis. Chem. Rev. 1946, 38, 41–82. [Google Scholar] [CrossRef] [PubMed]
- Khudyakov, I.V.; Arsu, N.; Jockusch, S.; Turro, N.J. Magnetic and spin effects in the photoinitiation of polymerization. Des. Mon. Polym. 2003, 6, 91–101. [Google Scholar] [CrossRef]
- Bag, D.S.; Maiti, S. Polymerization under magnetic field—II. Radical polymerization of acrylonitrile, styrene and methyl methacrylate. Polymer 1998, 39, 525–531. [Google Scholar] [CrossRef]
- Bag, D.S.; Maiti, S. Polymerization under a magnetic field. VI. Triplet dye-sensitized photopolymerization of acrylamide and methyl methacrylate. J. Polym. Sci. Part A Polym. Chem. 1998, 36, 1509–1513. [Google Scholar] [CrossRef]
- Chiriac, A.P. Polymerization in magnetic field. XVI. Kinetic aspects regarding methyl methacrylate polymerization in high magnetic field. J. Polym. Sci. Part A Polym. Chem. 2004, 42, 5678–5686. [Google Scholar] [CrossRef]
- Chiriac, A.P.; Simionescu, C.I. Some properties of vinyl acetate/methyl methacrylate/acrylamide copolymer synthesized in a magnetic field. Polym. Test. 1997, 16, 185–192. [Google Scholar] [CrossRef]
- Chiriac, A.P.; Simionescu, C.I. Aspects regarding the characteristics of some acrylic and methacrylic polyesters synthesized in a magnetic field. Polym. Test. 1996, 15, 537–548. [Google Scholar] [CrossRef]
- Huang, J.; Song, Q. Effect of polyethylene glycol with sensitizer groups at both ends on the photoinitiated polymerization of styrene in the water phase in the presence of a magnetic field. Macromolecules 1993, 26, 1359–1362. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, R.; Li, H.; Han, B.; Liu, Z.; Jiang, T.; He, J.; Zhang, X.; Yang, G. How does magnetic field affect polymerization in supercritical fluids? Study of radical polymerization in supercritical CO2. New J. Chem. 2002, 26, 958–961. [Google Scholar] [CrossRef]
- Simionescu, C.I.; Chiriac, A.P.; Chiriac, M.V. Polymerization in a magnetic field: 1. Influence of esteric chain length on the synthesis of various poly(methacrylate)s. Polymer 1993, 34, 3917–3920. [Google Scholar] [CrossRef]
- Turro, N.J.; Chow, M.F.; Chung, C.J.; Tung, C.H. An efficient, high conversion photoinduced emulsion polymerization. Magnetic field effects on polymerization efficiency and polymer molecular weight. J. Am. Chem. Soc. 1980, 102, 7391–7393. [Google Scholar] [CrossRef]
- Turro, N.J. Application of weak magnetic fields to influence rates and molecular weight distributions of styrene polymerization. Ind. Eng. Chem. Prod. Res. Dev. 1983, 22, 272–276. [Google Scholar] [CrossRef]
- Turro, N.J.; Chow, M.F.; Chung, C.J.; Tung, C.H. Magnetic field and magnetic isotope effects on photoinduced emulsion polymerization. J. Am. Chem. Soc. 1983, 105, 1572–1577. [Google Scholar] [CrossRef]
- Huang, J.; Hu, Y.; Song, Q. Effect of magnetic field on block copolymerization of styrene and methyl methacrylate by photochemical initiation in micellar solution of poly(ethylene glycol) with sensitizer end group. Polymer 1994, 35, 1105–1108. [Google Scholar] [CrossRef]
- Chiriac, A. Polymerization in a magnetic field. 13 Influence of the reaction conditions in the styrene polymerization. Rev. Roum. Chim. 2000, 45, 689–695. [Google Scholar]
- Chiriac, A.P.; Simionescu, C.I. Polymerization in a magnetic field. X. Solvent effect in poly(methyl methacrylate) synthesis. J. Polym. Sci. Part A Polym. Chem. 1996, 34, 567–573. [Google Scholar] [CrossRef]
- Keskin, S.; Aydin, M.; Khudyakov, I.; Arsu, N. Study of the polymerization of methyl methacrylate initiated by thioxanthone derivatives: a magnetic field effect. Turk. J. Chem. 2009, 33, 201–207. [Google Scholar]
- Vedeneev, A.A.; Khudyakov, I.V.; Golubkova, N.A.; Kuzmin, V.A. External magnetic field effect on the dye-photoinitiated polymerization of acrylamide. J. Chem. Soc. Faraday Trans. 1990, 86, 3545–3549. [Google Scholar] [CrossRef]
- Rintoul, I.; Wandrey, C. Magnetic field effects on the copolymerization of water-soluble and ionic monomers. J. Polym. Sci. Part A Polym. Chem. 2009, 47, 373–383. [Google Scholar] [CrossRef]
- Rintoul, I.; Wandrey, C. Radical homo- and copolymerization of acrylamide and ionic monomers in weak magnetic field. Macromol. Symp. 2008, 261, 121–129. [Google Scholar] [CrossRef]
- Rintoul, I.; Wandrey, C. Magnetic field effects on the free radical solution polymerization of acrylamide. Polymer 2007, 48, 1903–1914. [Google Scholar] [CrossRef]
- Odian, G. Radical Chain Polymerization. In Principles of Polymerization, 4th ed.; Wiley-Interscience: Hoboken, NJ, USA, 2004; Chapter 3; pp. 198–346. [Google Scholar]
- Steiner, U.; Ulrich, T. Magnetic field effects in chemical kinetics and related phenomena. Chem. Rev. 1989, 89, 51–147. [Google Scholar] [CrossRef]
- Carrington, A.; McLachlan, A.D. Introduction to Magnetic Resonance, with Applications to Chemistry and Chemical Physics; Harper International editions: London, UK, 1979. [Google Scholar]
- Ayscough, P.B. Electron Spin Resonance in Chemistry; Methuen & Co. Ltd.: London, UK, 1967. [Google Scholar]
- Sobhi, H.F. Synthesis and Characterization of Acylphospine Oxide Photoinitiators. Ph.D. Thesis, Cleveland State University, Cleveland, OH, USA, May 2008. [Google Scholar]
- Rintoul, I.; Wandrey, C. Limit of applicability of the monomer-enhanced mechanism for radical generation in persulfate initiated polymerization of acrylamide. Lat. Am. Appl. Res. 2010, 40, 365–372. [Google Scholar]
- Rintoul, I.; Wandrey, C. Polymerization of ionic monomers in polar solvents: kinetics and mechanism of the free radical copolymerization of acrylamide/acrylic acid. Polymer 2005, 46, 4525–4532. [Google Scholar] [CrossRef]
- Atkins, P.W.; de Paula, J. Statistical thermodynamics. In Elements of Physical Chemistry, 6th ed.; Oxford University Press: Oxford, UK, 2013. [Google Scholar]
| Monomer | Initiator | System | MF Effect | Ref. |
|---|---|---|---|---|
| AN | AIBN | Bulk | Increase of Rp, polymer yield, molar mass, syndiotacticity, crystallinity, and thermal stability of resulting polymers | [4] |
| MMA, ST | AIBN | Bulk | No effect | [4] |
| MMA | MB | H2O-MeOH mixtures | Decrease of the polymer yield and increase of the molar mass polymers | [5] |
| MMA | MB | Aqueous solution | No effect | [5] |
| MMA | BP, AMP, APA, AHC | Aqueous solution | Increase of the initiator efficiency and decrease of the monomer exponent and molar mass | [6] |
| AC, MMA, AM | BP, AMP, APA, AHC | Several solvents | Increase of the initiator efficiency and thermal stability of polymers | [7] |
| MMA, EMA, BMA | AIBN | Bulk | Increase of molar mass and thermal stability of products | [8] |
| ST | AIBN | H2O-EG mixtures | Increase of molar mass and homogeneity of polymers | [9] |
| MMA, ST | BP | Liquid CO2 | Increase of conversion and molar mass | [10] |
| MMA, ST | BP | Cyclohexane | No effect | [10] |
| MMA, EMA, BMA | BP | Bulk | Increase of Rp and molar mass | [11] |
| ST | BK | Emulsion | Increase of Rp and molar mass | [12,13] |
| ST, MMA, AA | BK | Emulsion | Increase of Rp and molar mass | [14] |
| ST, MMA | AIBN | Emulsion | Increase of molar mass and decrease of molar mass distribution | [15] |
| ST | K2S2O8 | Emulsion | Decrease of Rp | [16] |
| MMA | BP | 10 different organic solvents | Increase of Rp and conversion and decrease of the induction period for initiation | [17] |
| MMA | TX | Dimethylformamide | Increase of conversion and molar mass | [18] |
| AM | MB | H2O-EG mixtures | Increase of Rp | [19] |
| AM, AA, DADMAC and combinations | C26H27O3P | H2O-EG mixtures | Increase of Rp of all monomers in homo and copolymerizations. Increase of molar mass of polyAA. No effect in the molar mass of polyAM and copolymer compositions | [20,21] |
| Series | MF Tesla | [AM] mol/L | [Initiator] mol/L | Solvent | Temp. K |
|---|---|---|---|---|---|
| 1 | 0.0 | 0.20 | - | H2O | 313 |
| 2 | 0.0 | 0.20 | [C26H27O3P] = 2 × 10−6 | H2O | 323 |
| 3 | 0.0 | 0.20 | [K2S2O8] = 2.3 × 10−3 | H2O | 273 |
| 4 | 0.0 7.0 | 0.15 | [K2S2O8] = 2.3 × 10−3 | H2O | 308 |
| 5 | 0.0 7.0 | 0.15 | [K2S2O8] = 2.3 × 10−3 | 50% EG | 308 |
| 6 | 7.0 < MF < 0.5 | 0.10 | [K2S2O8] = 2.3 × 10−3 | H2O | 308 |
| 7 | 0.00 0.11 0.35 0.50 | 0.20 | [K2S2O8] = 1.2 × 10−2 | H2O | 313 |
| 8A 8B | 0.0 < MF < 0.50.0 < MF < 0.1 | 0.20 0.10 | [K2S2O8] = 1.2 × 10−2 [K2S2O8] = 1.2 × 10−3 | H2O | 313 |
| 9 | 0.0 0.1 | 0.20 | [C26H27O3P] = 1 × 10−6 | H2O | 313 |
| 10 | 0.0 0.1 | 0.20 | [C26H27O3P] = 1 × 10−6 | 50% EG | 313 |
© 2017 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rintoul, I. Kinetic control of aqueous polymerization using radicals generated in different spin states. Processes 2017, 5, 15. https://doi.org/10.3390/pr5020015
Rintoul I. Kinetic control of aqueous polymerization using radicals generated in different spin states. Processes. 2017; 5(2):15. https://doi.org/10.3390/pr5020015
Chicago/Turabian StyleRintoul, Ignacio. 2017. "Kinetic control of aqueous polymerization using radicals generated in different spin states" Processes 5, no. 2: 15. https://doi.org/10.3390/pr5020015
APA StyleRintoul, I. (2017). Kinetic control of aqueous polymerization using radicals generated in different spin states. Processes, 5(2), 15. https://doi.org/10.3390/pr5020015





