High-Performance Iontronic Pressure Sensor with a Multi-Level Conoid-like Structure Fabricated via Direct Laser Writing
Abstract
1. Introduction
2. Materials and Methods
2.1. Fabrication of the PI Templates
2.2. Preparation of the Ionic Gel Film
2.3. Characterization and Device Assembly
3. Results and Discussion
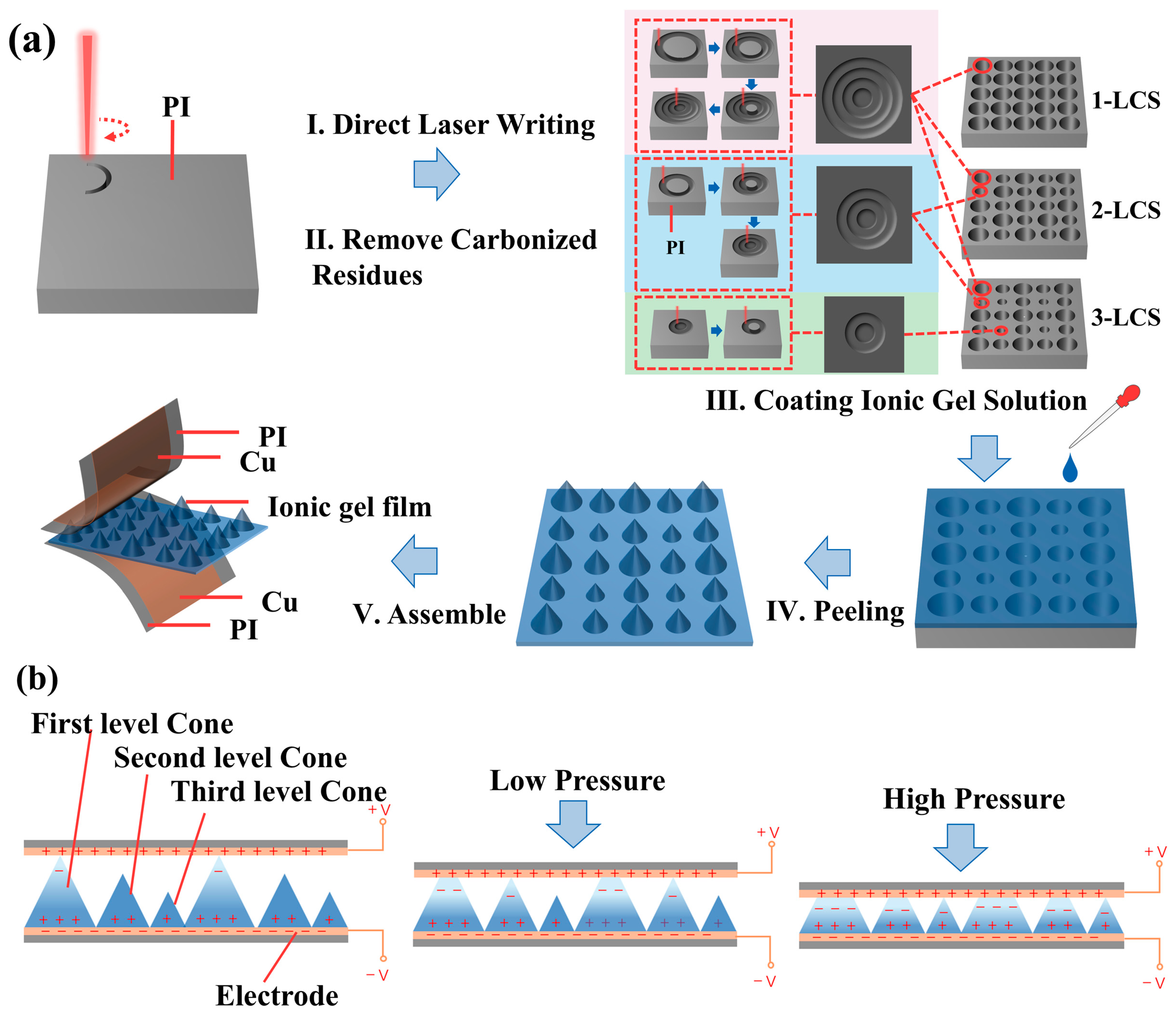
3.1. Sensing Mechanism and Fabrication of the Iontronic Sensor
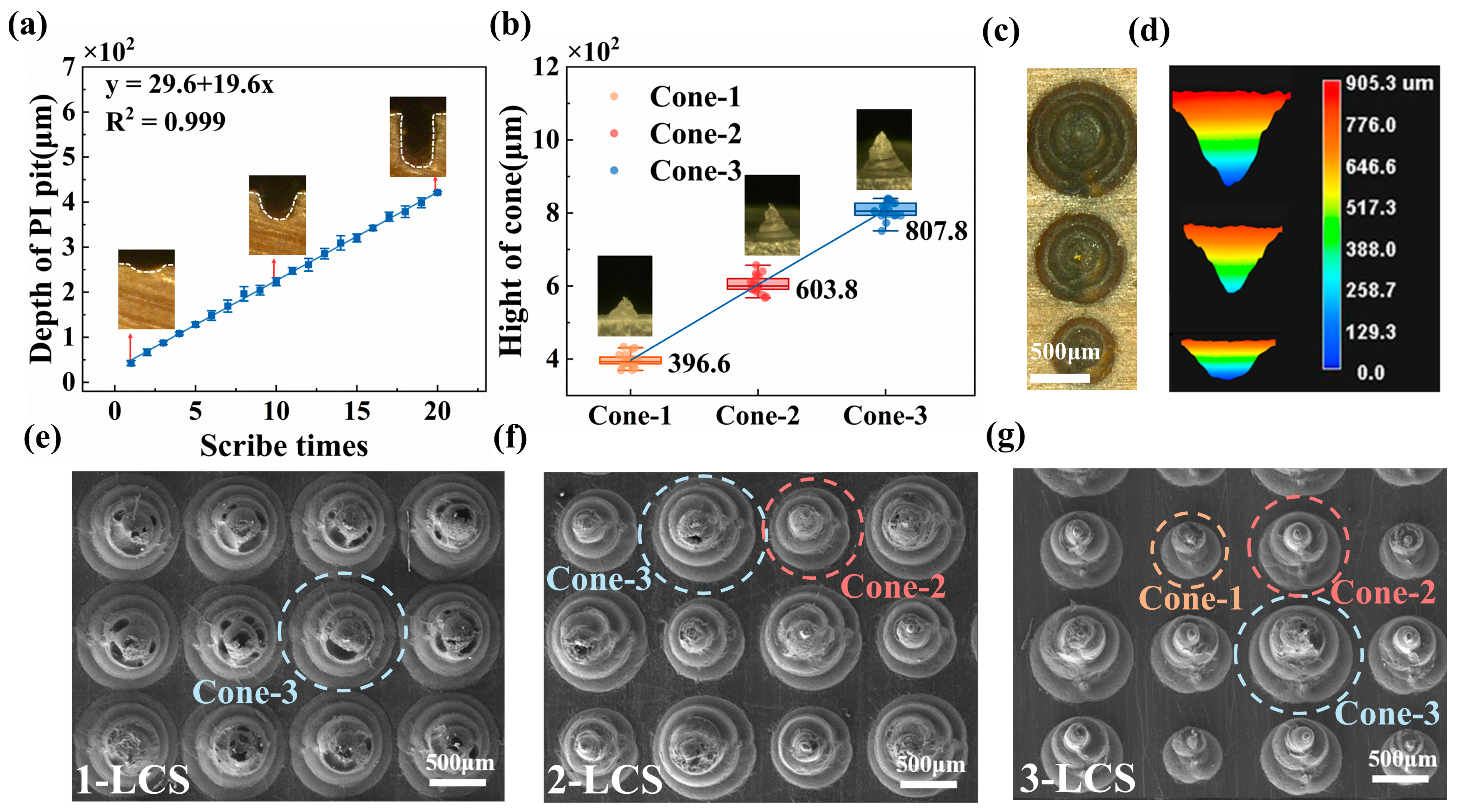
3.2. Characterization and Application of Iontronic Pressure Sensor
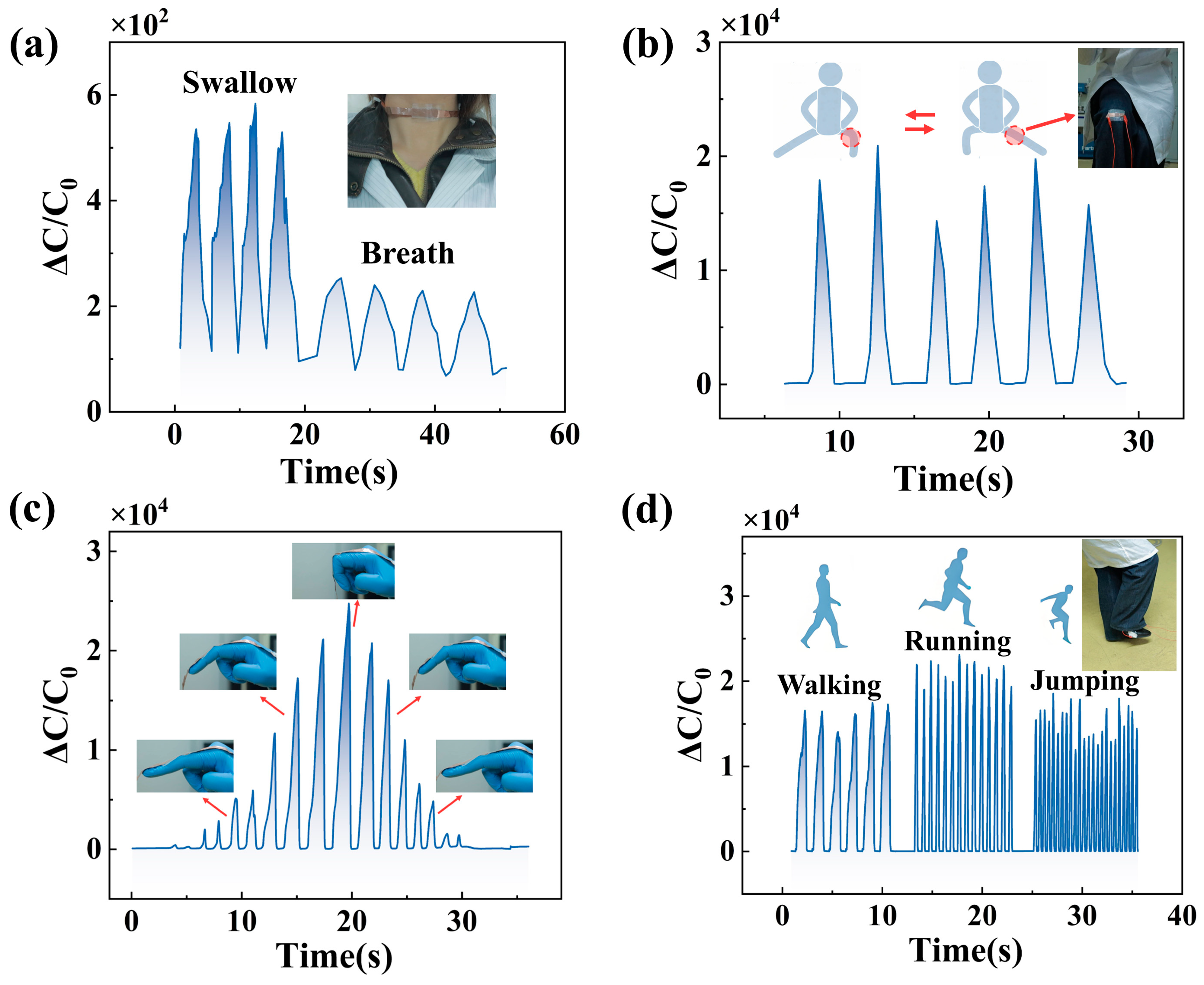
3.3. Application in Human Physiological Monitoring
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Cui, X.; Chen, J.; Wu, W.; Liu, Y.; Li, H.; Xu, Z.; Zhu, Y. Flexible and breathable all-nanofiber iontronic pressure sensors with ultraviolet shielding and antibacterial performances for wearable electronics. Nano Energy 2022, 95, 107022. [Google Scholar] [CrossRef]
- Liang, T.; Liu, Z.; Zhang, H.; Zhou, X.; Liang, Y. The structure, material and performance of multi-functional tactile sensor and its application in robot field: A review. Mater. Today 2025, 86, 452–481. [Google Scholar] [CrossRef]
- Deng, W.-J.; Wang, L.-F.; Dong, L.; Huang, Q.-A. LC Wireless Sensitive Pressure Sensors with Microstructured PDMS Dielectric Layers for Wound Monitoring. IEEE Sens. J. 2018, 18, 4886–4892. [Google Scholar] [CrossRef]
- Huang, Y.-C.; Liu, Y.; Ma, C.; Cheng, H.-C.; He, Q.; Wu, H.; Wang, C.; Lin, C.-Y.; Huang, Y.; Duan, X. Sensitive pressure sensors based on conductive microstructured air-gap gates and two-dimensional semiconductor transistors. Nat. Electron. 2020, 3, 59–69. [Google Scholar] [CrossRef]
- Long, H.; Li, Q.; Peng, S.; Chen, S.; Zhang, T.; Zhang, M.; Li, M.; Chen, L. Flexible, Strong and Multifunctional Anf@Ag Nanocomposite Film for Human Physiology and Motion Monitoring. Processes 2022, 10, 961. [Google Scholar] [CrossRef]
- Kim, J.; Kim, J. Deep Learning Integration for Normal Breathing Classification Using a Flexible Fiber Sensor. Processes 2024, 12, 2644. [Google Scholar] [CrossRef]
- Liu, Q.; Liu, Y.; Shi, J.; Liu, Z.; Wang, Q.; Guo, C.F. High-Porosity Foam-Based Iontronic Pressure Sensor with Superhigh Sensitivity of 9280 kPa−1. Nano-Micro Lett. 2021, 14, 21. [Google Scholar] [CrossRef] [PubMed]
- Ha, K.H.; Zhang, W.; Jang, H.; Kang, S.; Wang, L.; Tan, P.; Hwang, H.; Lu, N. Highly Sensitive Capacitive Pressure Sensors over a Wide Pressure Range Enabled by the Hybrid Responses of a Highly Porous Nanocomposite. Adv. Mater. 2021, 33, 2103320. [Google Scholar] [CrossRef]
- Wang, W. Flexible Pressure Sensors: Classification, Cutting-Edge Advances and Applications. In Proceedings of the 2025 2nd International Conference on Electrical Engineering and Intelligent Control (EEIC 2025), Auckland, New Zealand, 3–4 July 2025; Advances in Engineering Research; Atlantis Press: Dordrecht, The Netherlands, 2025; pp. 639–654. [Google Scholar] [CrossRef]
- Li, Z.; Zhao, K.; Wang, J.; Wang, B.; Lu, J.; Jia, B.; Ji, T.; Han, X.; Luo, G.; Yu, Y.; et al. Sensitive, Robust, Wide-Range, and High-Consistency Capacitive Tactile Sensors with Ordered Porous Dielectric Microstructures. ACS Appl. Mater. Interfaces 2024, 16, 7384–7398. [Google Scholar] [CrossRef]
- Bijender; Kumar, A. Flexible and wearable capacitive pressure sensor for blood pressure monitoring. Sens. Bio-Sens. Res. 2021, 33, 100434. [Google Scholar] [CrossRef]
- Ray, T.R.; Choi, J.; Bandodkar, A.J.; Krishnan, S.; Gutruf, P.; Tian, L.; Ghaffari, R.; Rogers, J.A. Bio-Integrated Wearable Systems: A Comprehensive Review. Chem. Rev. 2019, 119, 5461–5533. [Google Scholar] [CrossRef]
- Luo, Z.; Huang, J.; Song, Q.; Xia, B.; Li, H.; Zeng, G. High-sensitivity and wide-dynamic-range parameter estimation via weak-value amplification. Phys. Rev. Appl. 2024, 22, 064070. [Google Scholar] [CrossRef]
- Mathew, R.; Ravi Sankar, A. A Review on Surface Stress-Based Miniaturized Piezoresistive SU-8 Polymeric Cantilever Sensors. Nano-Micro Lett. 2018, 10, 35. [Google Scholar] [CrossRef]
- Chang, Y.; Wang, L.; Li, R.; Zhang, Z.; Wang, Q.; Yang, J.; Guo, C.F.; Pan, T. First Decade of Interfacial Iontronic Sensing: From Droplet Sensors to Artificial Skins. Adv. Mater. 2020, 33, 2003464. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Lv, B.; Wang, H.; Yang, B.; Li, Z.; Junfang, R.; Gui, G.; Liu, W.; Yang, S.; Li, L. Ionogel-based flexible stress and strain sensors. Int. J. Smart Nano Mater. 2021, 12, 307–336. [Google Scholar] [CrossRef]
- Cui, X.; Xi, Y.; Tu, S.; Zhu, Y. An overview of flexible sensors from ionic liquid-based gels. TrAC Trends Anal. Chem. 2024, 174, 117662. [Google Scholar] [CrossRef]
- Song, J.; Yang, R.; Shi, J.; Chen, X.; Xie, S.; Liao, Z.; Zou, R.; Feng, Y.; Ye, T.T.; Guo, C.F. Polyelectrolyte-based wireless and drift-free iontronic sensors for orthodontic sensing. Sci. Adv. 2025, 11, eadu6086. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Qu, C.; Yao, J.; Zhang, Y.; Xu, Y. Two-Stage Micropyramids Enhanced Flexible Piezoresistive Sensor for Health Monitoring and Human–Computer Interaction. ACS Appl. Mater. Interfaces 2024, 16, 7640–7649. [Google Scholar] [CrossRef]
- Wang, Y.; Jing, R.; Jiang, J.; Wang, H.; Qian, L.; Yu, B.; Zhao, Z.-J. Coral-Inspired Capacitive Pressure Sensor with High Sensitivity and Wide Range for Human–Computer Interaction. ACS Appl. Electron. Mater. 2024, 6, 6726–6737. [Google Scholar] [CrossRef]
- Zhang, H.; Chen, X.; Liu, Y.; Yang, C.; Liu, W.; Qi, M.; Zhang, D. PDMS Film-Based Flexible Pressure Sensor Array with Surface Protruding Structure for Human Motion Detection and Wrist Posture Recognition. ACS Appl. Mater. Interfaces 2024, 16, 2554–2563. [Google Scholar] [CrossRef]
- Ding, Z.; Li, W.; Wang, W.; Zhao, Z.; Zhu, Y.; Hou, B.; Zhu, L.; Chen, M.; Che, L. Highly Sensitive Iontronic Pressure Sensor with Side-by-Side Package Based on Alveoli and Arch Structure. Adv. Sci. 2024, 11, e2309407. [Google Scholar] [CrossRef]
- Zhong, Y.; Liu, K.; Wu, L.; Ji, W.; Cheng, G.; Ding, J. Flexible Tactile Sensors with Gradient Conformal Dome Structures. ACS Appl. Mater. Interfaces 2024, 16, 52966–52976. [Google Scholar] [CrossRef]
- Wang, D.; Li, B.; Niu, S.; Han, Z.; Ren, L. Novel Iontronic Pressure Sensor Coupling High Sensitivity and Wide-Range for Stiffness Identification and Long-Distance Precise Motion Control. Adv. Funct. Mater. 2024, 35, 2413551. [Google Scholar] [CrossRef]
- Bai, N.; Wang, L.; Xue, Y.; Wang, Y.; Hou, X.; Li, G.; Zhang, Y.; Cai, M.; Zhao, L.; Guan, F.; et al. Graded Interlocks for Iontronic Pressure Sensors with High Sensitivity and High Linearity over a Broad Range. ACS Nano 2022, 16, 4338–4347. [Google Scholar] [CrossRef]
- Yan, Z.; Wang, S.; Huang, F.; Deng, G.; Sui, X.; Wu, Z.; Wang, J. High sensitivity iontronic pressure sensors with wavy structure electrode and two-level raised structures ionic gel film prepared by direct laser writing. Sens. Actuators A Phys. 2023, 363, 114735. [Google Scholar] [CrossRef]
- Mao, L.; Pan, T.; Lin, L.; Ke, Y.; Su, H.; Li, Y.; Huang, W.; Li, T.; Lin, Y. Simultaneously enhancing sensitivity and operation range of flexible pressure sensor by constructing a magnetic-guided microstructure in laser-induced graphene composite. Chem. Eng. J. 2024, 481, 148639. [Google Scholar] [CrossRef]
- Wang, H.; Li, Z.; Liu, Z.; Fu, J.; Shan, T.; Yang, X.; Lei, Q.; Yang, Y.; Li, D. Flexible capacitive pressure sensors for wearable electronics. J. Mater. Chem. C 2022, 10, 1594–1605. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, H.; Liang, J.; Zhao, X.; Li, B.; Zang, J.; Gao, L.; Zhang, Z.; Xue, C. Ti3C2Tx-MXene/PET textile-based flexible pressure sensor for wearable pulse monitoring. J. Mater. Chem. C 2023, 11, 15638–15648. [Google Scholar] [CrossRef]
- Bai, N.; Wang, L.; Wang, Q.; Deng, J.; Wang, Y.; Lu, P.; Huang, J.; Li, G.; Zhang, Y.; Yang, J.; et al. Graded intrafillable architecture-based iontronic pressure sensor with ultra-broad-range high sensitivity. Nat. Commun. 2020, 11, 209. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Wang, F.; Wang, J.; Yang, S. Highly Sensitive and Wide Linear Range Flexible Pressure Sensors with Hierarchical Microstructure and Its Application in Human Signal Detection. In Proceedings of the 2024 8th International Conference on Electrical, Mechanical and Computer Engineering (ICEMCE), Xi’an, China, 25–27 October 2024; IEEE: New York, NY, USA, 2024; pp. 379–382. [Google Scholar] [CrossRef]
- Cui, C.; Qin, Y.; Zeng, Y.; Lu, X.; Wei, E.; Xie, J. A flexible capacitive pressure sensor with dual-layer microstructure for health monitoring. Sens. Actuators A Phys. 2024, 377, 115709. [Google Scholar] [CrossRef]
- Wang, S.; Zong, Q.; Yang, H.; Tan, C.; Huang, Q.; Liu, X.; Zhang, G.; French, P.; Ye, H. Rapid Fabrication of High-Performance Flexible Pressure Sensors Using Laser Pyrolysis Direct Writing. ACS Appl. Mater. Interfaces 2023, 15, 41055–41066. [Google Scholar] [CrossRef]
- Wang, S.; Chang, S.; Song, Y.; Qiao, X.; Li, L.; Zhao, L.; Yang, P.; Yu, S. High sensitivity capacitive flexible pressure sensor based on PDMS double wrinkled microstructure. J. Mater. Sci. Mater. Electron. 2024, 35, 78. [Google Scholar] [CrossRef]
- Mubasshira; Rahman, M.M.; Uddin, M.N.; Rhaman, M.; Roy, S.; Sarker, M.S. Next-Generation Smart Carbon–Polymer Nanocomposites: Advances in Sensing and Actuation Technologies. Processes 2025, 13, 2991. [Google Scholar] [CrossRef]
- Guo, X.; Hong, W.; Hu, B.; Zhang, T.; Jin, C.; Yao, X.; Li, H.; Yan, Z.; Jiao, Z.; Wang, M.; et al. Human touch sensation-inspired, ultrawide-sensing-range, and high-robustness flexible piezoresistive sensor based on CB/MXene/SR/fiber nanocomposites for wearable electronics. Compos. Struct. 2023, 321, 117329. [Google Scholar] [CrossRef]
- Liu, L.; Cai, Y.; Jiang, X.; Wang, J.; Wang, C.; Duan, J.-A. A versatile surface micro structure design strategy for porous-based pressure sensors to enhance electromechanical performance. Chem. Eng. J. 2024, 490, 151529. [Google Scholar] [CrossRef]
- Li, X.; Liang, Q.; Liu, H.; Zhao, L.; Sun, C.; Hou, C. High-Sensitivity MXene/MWCNTs/PDMS Flexible Capacitive Sensor for Wearable Health Monitoring. Adv. Mater. Technol. 2025, 10, e00677. [Google Scholar] [CrossRef]
- Kou, H.; Zhang, Y.; Ye, Y.; Pang, Y.; Zhang, L.; Zhang, X.; Yang, L.; Shang, Z.; Gui, Z.; Chen, Y. Ultrasensitive iontronic pressure sensor based on microstructural electrodes and porous dielectric layer for wearable electronics. Sens. Actuators A Phys. 2025, 394, 116977. [Google Scholar] [CrossRef]
- Li, G.; Chen, D.; Li, C.; Liu, W.; Liu, H. Engineered Microstructure Derived Hierarchical Deformation of Flexible Pressure Sensor Induces a Supersensitive Piezoresistive Property in Broad Pressure Range. Adv. Sci. 2020, 7, 2000154. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Zhao, Y.; Lin, N.; Xie, Y. Skin-inspired flexible pressure sensor with hierarchical interlocked spinosum microstructure by laser direct writing for high sensitivity and large linearity. Sens. Actuators A Phys. 2024, 366, 114988. [Google Scholar] [CrossRef]
- Tu, S.; Xi, Y.; Cui, X.; Xu, Z.; Liu, Z.; Zhu, Y. Skin-inspired interlocked microstructures with soft-hard synergistic effect for high-sensitivity and wide-linear-range pressure sensing. Chem. Eng. J. 2024, 496, 154083. [Google Scholar] [CrossRef]
- Li, G.; Zhang, Y.; Zhang, X.; Zhu, B.; Lei, Y.; Chen, D.; Shui, L.; Wu, G.; Xue, L. Filiform Papillae-Inspired Wearable Pressure Sensor with High Sensitivity and Wide Detection Range. Adv. Funct. Mater. 2024, 35, 2414465. [Google Scholar] [CrossRef]
- Peng, S.; Blanloeuil, P.; Wu, S.; Wang, C.H. Rational Design of Ultrasensitive Pressure Sensors by Tailoring Microscopic Features. Adv. Mater. Interfaces 2018, 5, 1800403. [Google Scholar] [CrossRef]
- An, T.; Zhang, Y.; Wen, J.; Dong, Z.; Du, Q.; Liu, L.; Wang, Y.; Xing, G.; Zhao, X. Multi-Level Pyramidal Microstructure-Based Pressure Sensors with High Sensitivity and Wide Linear Range for Healthcare Monitoring. ACS Sens. 2024, 9, 726–735. [Google Scholar] [CrossRef]
- Li, P.; Lang, S.; Xie, L.; Zhang, Y.; Gou, X.; Zhang, C.; Dong, C.; Li, C.; Yang, J. Skin-Inspired Ultra-Linear Flexible Iontronic Pressure Sensors for Wearable Musculoskeletal Monitoring. Nano-Micro Lett. 2025, 18, 55. [Google Scholar] [CrossRef]
- Zhao, C.; Wang, Y.; Tang, G.; Ru, J.; Zhu, Z.; Li, B.; Guo, C.F.; Li, L.; Zhu, D. Ionic Flexible Sensors: Mechanisms, Materials, Structures, and Applications. Adv. Funct. Mater. 2022, 32, 2110417. [Google Scholar] [CrossRef]
- Kim, Y.; Yang, H.; Oh, J.H. Simple fabrication of highly sensitive capacitive pressure sensors using a porous dielectric layer with cone-shaped patterns. Mater. Des. 2021, 197, 109203. [Google Scholar] [CrossRef]
- Lin, X.; Zhou, Y.; Lei, Z.; Chen, R.; Chen, W.; Meng, X.; Li, Y. Facile Electrochemical Biosensing Platform Based on Laser Induced Graphene/Laccase Electrode for the Effective Determination of Gallic Acid. Processes 2023, 11, 2048. [Google Scholar] [CrossRef]
- Chortos, A.; Bao, Z. Skin-inspired electronic devices. Mater. Today 2014, 17, 321–331. [Google Scholar] [CrossRef]
- Takizawa, C.; Gemmell, E.; Kenworthy, J.; Speyer, R. A Systematic Review of the Prevalence of Oropharyngeal Dysphagia in Stroke, Parkinson’s Disease, Alzheimer’s Disease, Head Injury, and Pneumonia. Dysphagia 2016, 31, 434–441. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Wang, X.; Wang, S.; Zhao, S.; Qi, L.; Chen, Q.; Guo, C.; Deng, G. High-Performance Iontronic Pressure Sensor with a Multi-Level Conoid-like Structure Fabricated via Direct Laser Writing. Processes 2026, 14, 1234. https://doi.org/10.3390/pr14081234
Wang X, Wang S, Zhao S, Qi L, Chen Q, Guo C, Deng G. High-Performance Iontronic Pressure Sensor with a Multi-Level Conoid-like Structure Fabricated via Direct Laser Writing. Processes. 2026; 14(8):1234. https://doi.org/10.3390/pr14081234
Chicago/Turabian StyleWang, Xingyi, Shutong Wang, Shengbin Zhao, Lufan Qi, Quan Chen, Chenyu Guo, and Guoliang Deng. 2026. "High-Performance Iontronic Pressure Sensor with a Multi-Level Conoid-like Structure Fabricated via Direct Laser Writing" Processes 14, no. 8: 1234. https://doi.org/10.3390/pr14081234
APA StyleWang, X., Wang, S., Zhao, S., Qi, L., Chen, Q., Guo, C., & Deng, G. (2026). High-Performance Iontronic Pressure Sensor with a Multi-Level Conoid-like Structure Fabricated via Direct Laser Writing. Processes, 14(8), 1234. https://doi.org/10.3390/pr14081234





