Measurement of Biomass in Small-Scale Microalgal and Microalgal–Bacterial Systems for Wastewater Treatment: Mini Review and Experimental Evaluation
Abstract
1. Introduction
2. Common Methods for Biomass Quantification in Small-Scale Studies
2.1. Direct Quantification Methods
2.1.1. Gravimetric Methods (Dry Cell Weight)
2.1.2. Cell Counts
2.1.3. Packed Cell Volume
2.2. Indirect Quantification Methods
2.2.1. Turbidimetry (Optical Density)
The Importance of Choosing the Correct Turbidimetric Wavelength/s
Inter- and Intra-Strain Inaccuracies in Measuring Microalgal Biomass Using Turbidimetry
2.2.2. Chlorophyll-a Quantification
3. Gravimetric Biomass Measurements
3.1. Introduction
3.2. Materials and Methods
3.2.1. Microalgal Culture
3.2.2. Tannery Wastewater
3.2.3. Experimental Set-Up and Procedures
3.2.4. Analytical Procedures
Chemical Analyses
Gravimetric Analysis
Reactors Total Suspended Solids and Total Volatile Solids
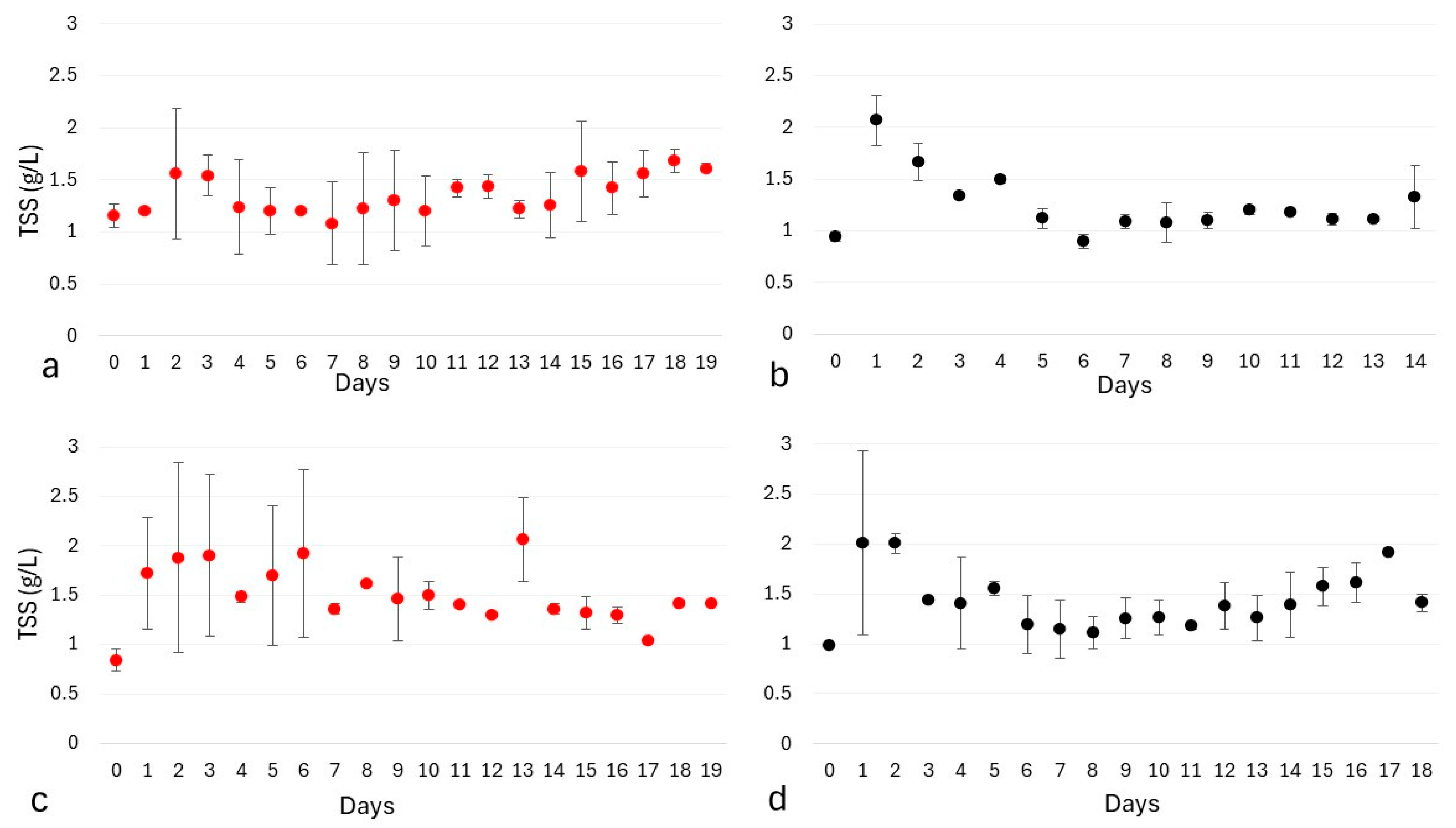
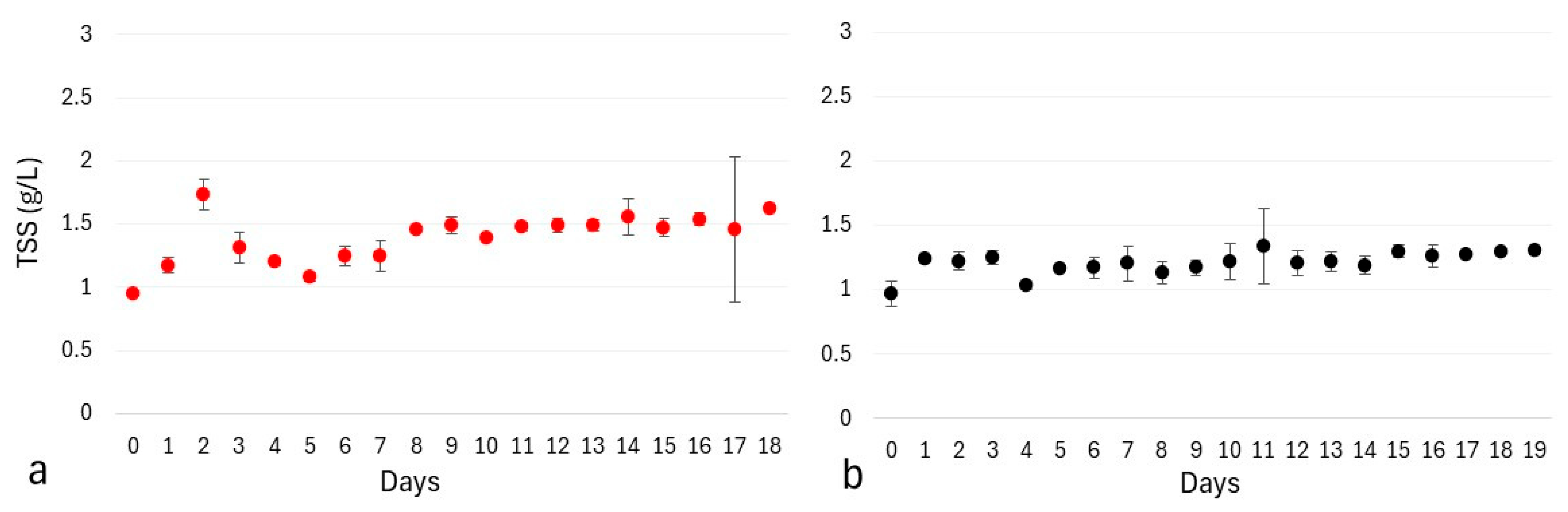
3.3. Results and Discussion
3.3.1. Total Suspended Solids Measurements with Different Working and Sampling Volumes
3.3.2. Influence of Wastewater Pretreatment on Dry Cell Weight Determination
3.4. Conclusions and Recommendations
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhou, Y.; Cui, X.; Wu, B.; Wang, Z.; Liu, Y.; Ren, T.; Xia, S.; Rittmann, B.E. Microalgal extracellular polymeric substances (EPS) and their roles in cultivation, biomass harvesting, and bioproducts extraction. Bioresour. Technol. 2024, 406, 131054. [Google Scholar] [CrossRef]
- Felz, S.; Vermeulen, P.; van Loosdrecht, M.C.M.; Lin, Y.M. Chemical characterization methods for the analysis of structural extracellular polymeric substances (EPS). Water Res. 2019, 157, 201–208. [Google Scholar] [CrossRef]
- Hammes, F.; Egli, T. Cytometric methods for measuring bacteria in water: Advantages, pitfalls and applications. Anal. Bioanal. Chem. 2010, 397, 1083–1095. [Google Scholar] [CrossRef]
- Satiro, J.; Gomes, A.; Florencio, L.; Simões, R.; Albuquerque, A. Effect of microalgae and bacteria inoculation on the startup of bioreactors for paper pulp wastewater and biofuel production. J. Environ. Manag. 2024, 362, 121305. [Google Scholar] [CrossRef]
- Cosenza, A.; Lima, S.; Gurreri, L.; Mancini, G.; Scargiali, F. Microalgae in the Mediterranean area: A geographical survey outlining the diversity and technological potential. Algal Res. 2024, 82, 103669. [Google Scholar] [CrossRef]
- Salbitani, G.; Carfagna, S. Ammonium Utilization in Microalgae: A Sustainable Method for Wastewater Treatment. Sustainability 2021, 13, 956. [Google Scholar] [CrossRef]
- Waqas, S.; Harun, N.Y.; Sambudi, N.S.; Abioye, K.J.; Zeeshan, M.H.; Ali, A.; Abdulrahman, A.; Alkhattabi, L.; Alsaadi, A.S. Effect of Operating Parameters on the Performance of Integrated Fixed-Film Activated Sludge for Wastewater Treatment. Membranes 2023, 13, 704. [Google Scholar] [CrossRef] [PubMed]
- APHA (American Public Health Association). American Water Works Association Standard Methods for Examination of Water and Wastewater, 24th ed.; Water Environment Federation: Alexandria, VA, USA, 2025. [Google Scholar]
- Queenan, K.; Burton, C.H.; Bechir, C. Development of a centrifuge-based procedure to analyse agricultural effluents for total and volatile suspended solids. Bioresour. Technol. 1996, 57, 259–263. [Google Scholar] [CrossRef]
- De Souza, M.F.; Rodrigues, M.A.; da Silva Bon, E.P.; Freitas, S.P. Interference of starch accumulation in microalgal cell growth measurement. J. Appl. Phycol. 2019, 31, 249–254. [Google Scholar] [CrossRef]
- Battula, S.R.; Sen, R. Leveraging the dynamice of microalgal CO2 capture to estimate the maximum inherent photosynthetic potential. J. Chem. Technol. Biotechnol. 2024, 99, 2320–2325. [Google Scholar] [CrossRef]
- Ranjan, A.; Lilly, M.; Deepnarain, N.; Welz, P.J. Valorisation of tannery wastewater with indigenous microalgal strains: Acclimation, identification, biomass characterization and remediation. Algal Res. 2025, 86, 103953. [Google Scholar] [CrossRef]
- Fekete, G.; Sebok, A.; Klatyik, S.; Varga, Z.I.; Grosz, J.; Czinkota, I.; Aleksza, L. Comparative analysis of laboratory-based and spectroscopic methods used to estimate the algal density of Chlorella vulgaris. Microorganisms 2024, 12, 1050. [Google Scholar] [CrossRef]
- Carneiro, M.; Cicchi, B.; Maia, I.B.; Pereira, H.; Zitelli, G.C.; Varela, J.; Malcata, F.X.; Torzillo, G. Effect of temperature on growth, photosynthesis and biochemical composition of Nannochloropsis oceanica, grown outdoors in tubular photobioreactors. Algal Res. 2020, 49, 101923. [Google Scholar] [CrossRef]
- Griffiths, M.J.; Garcin, C.; van Hille, R.P.; Harrison, S.T.L. Interferences by pigment in the estimation of microalgal biomass concentration by optical density. Microbiol. Methods 2011, 85, 119–123. [Google Scholar] [CrossRef]
- Chiu, S.Y.; Tsai, M.T.; Kao, C.Y.; Ong, S.C.; Lin, C.S. The air-lift photobioreactors with flow patterning for high density culture of microalgae and carbon dioxide removal. Eng. Life Sci. 2009, 9, 254–260. [Google Scholar] [CrossRef]
- Dube, A.; Cowan, A.K. Algal settling ponds contribute to final effluent quality of integrated algal pond systems for municipal sewage treatment. Water Environ. J. 2023, 37, 232–241. [Google Scholar] [CrossRef]
- McGinn, P.J.; MacQuarrie, S.P.; Choi, J.; Tartakovsky, B. Maximizing the productivity of the microalgae Scenedesmus AMDD cultivated in a continuous photobioreactor using flow rate control. Bioprocess Biosyst. Eng. 2017, 40, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Choe, H.W.; Kim, D.; Choi, Y.J.; Koh, H.G.; Park, W.K. Influence of Microalgal Cell Division Tendency on OD to DCW Conversion Factor and Chlorophyll Contents. J. Microbiol. Biotechnol. 2025, 9, e2412049. [Google Scholar] [CrossRef]
- Calmes, B.; Strittmatter, M.; Jacquemin, B.; Perrineau, M.M.; Rosseau, C.; Badis, Y.; Cock, J.M.; Destombe, C.; Valero, M.; Gachon, C.M.M. Parallelisable non-invasive biomass, fitness and growth measurement of macroalgae and other protists with nephelometry. Algal Res. 2020, 46, 101762. [Google Scholar] [CrossRef]
- Lopes, A.P.; Santos, F.M.; Silva, T.E.C.V.; Vilar, V.J.P.; Pires, J.C.M. Outdoor cultivation of the microalga Chlorella vulgaris in a new photobioreactor configuration: The effect of ultraviolet and visible radiation. Energies 2020, 13, 1962. [Google Scholar] [CrossRef]
- Pahija, E.; Hui, C.W. A systematic study on the effect of dynamic environments on microalgae concentration. Algal Res. 2019, 42, 101599. [Google Scholar] [CrossRef]
- Nielsen, S.L.; Hansen, B.W. Evaluation of the robustness of optical density as a tool for estimation of biomass in microalgal cultivation: The effects of growth condition and physiological state. Agric. Res. 2019, 50, 2698–2706. [Google Scholar] [CrossRef]
- Čertnerová, D.; Galbraith, D.W. Best practices in the flow cytometry of microalgae. Cytometry 2021, 99, 359–364. [Google Scholar] [CrossRef]
- Schagerl, M.; Siedler, R.; Konopácová, E.; Ali, S.S. Estimating biomass and vitality of microalgae for monitoring cultures: A roadmap for reliable measurements. Cells 2022, 11, 2455. [Google Scholar] [CrossRef] [PubMed]
- Almomani, F.A.; Örmeci, B. Monitoring and measurement of microalgae using the first derivative of absorbance and comparison with chlorophyll extraction method. Environ. Monit. Assess. 2018, 190, 90. [Google Scholar] [CrossRef] [PubMed]
- Ranjan, A.; Welz, P.J. Influence of season, inoculum density and ammonia concentration on biomass abundance and ammonia utilization of a microalgal consortium for tertiary remediation of tannery wastewater and valorization of biomass. Algal Res. 2025, 90, 104137. [Google Scholar] [CrossRef]
- Liang, F.; Ya, Q.; Du, W.C.; Wen, X.B.; Geng, Y.H.; Li, Y.G. The relationships between optical density, cell number, and biomass (dry weight) of four microalgae. Acta Ecol. Sin. 2014, 34, 6156–6163. [Google Scholar] [CrossRef]
- Tang, C.D.; Hu, Y.R.; Zhang, M.; Chen, S.L.; He, Z.W.; Li, Z.H.; Tian, Y.; Wang, X.C. Role of phosphate in microalgal-bacterial symbiosis system treating wastewater containing heavy metals. Environ. Pollut. 2024, 349, 123951. [Google Scholar] [CrossRef]
- Ritchie, R.J. Universal chlorophyll equations for estimating chlorophylls a, b, c, and d and total chlorophylls in natural assemblages of photosynthetic organisms using acetone, methanol or ethanol solvents. Phototsynthetica 2008, 46, 115–126. [Google Scholar] [CrossRef]
- Ramaraj, R.; Tsai, D.D.W.; Chen, P.H. Chlorophyll is not accurate measurement for algal biomass. Chiang Mai J. Sci. 2013, 40, 547–555. [Google Scholar]
- Patil, S.P.; Anil, A.C. Simulation experiments to elucidate variable fluorescence as a potential proxy for bulk microalgal viability from natural water, sediments and biofilms: Implication in ships ballast water management. J. Environ. Manag. 2018, 222, 242–249. [Google Scholar] [CrossRef]
- Serôdio, J.; da Silva, J.; Catarino, F. Use of in vivo chlorophyll a fluorescence to quantify short-term variations in the productive biomass of intertidal microphytobenthos. Mar. Ecol. Prog. Ser. 2001, 218, 45–61. [Google Scholar] [CrossRef]
- Adams, H.; Ye, J.; Persaud, B.; Slowinski, S.; Pour, H.K.; Van-Cappellen, P. Chlorophyll-a growth rates and related envrionmental variable in global temperate and cold-temperate lakes. Earth Syst. Sci. Data 2022, 14, 5139–5156. [Google Scholar] [CrossRef]
- Chao, Q.; Wang, T.Y.; Tang, C.C.; Wang, R.; He, Z.W.; Li, Z.H.; Tian, Y. Effects of triclosan on microalgal-bacterial symbiosis system operating in stable phase: Performances and mechanisms. J. Water Process Eng. 2025, 72, 107568. [Google Scholar] [CrossRef]
- Lewis, K.M.; Arntsen, A.E.; Coupel, P.; Joy-Warren, H.; Lowry, K.E.; Matsouka, A.; Mills, M.M.; Dijken, G.L.; Selz, V.; Arrigo, K.R. Photoacclimation of Arctic Ocean phytoplankton to shifting light and nutrient limitation. Limnol. Oceanogr. 2018, 64, 284–301. [Google Scholar] [CrossRef]
- Inomura, K.; Omta, A.W.; Talmy, D.; Bragg, J.; Deutsch, C.; Follows, M.J. A Mechanistic Model of Macromolecular Allocation, Elemental Stoichiometry, and Growth Rate in Phytoplankton. Front. Microbiol. 2020, 11, 86. [Google Scholar] [CrossRef] [PubMed]
- Gan, K.; Mou, X.; Xu, Y.; Want, H. Application of ozonated piggery wastewater for cultivation of oil-rich Chlorella pyrenoidosa. Bioresour. Technol. 2014, 171, 285–290. [Google Scholar] [CrossRef] [PubMed]
- Kozik, V.; Barbusinski, K.; Thomas, M.; Sroda, A.; Jampilek, J.; Sochanik, A.; Smolinski, A.; Bak, A. Taguchi Method and Response Surface Methodology in the Treatment of Highly Contaminated Tannery Wastewater Using Commercial Potassium Ferrate. Materials 2019, 12, 3784. [Google Scholar] [CrossRef] [PubMed]
- Ngobeni, P.V.; Mpofu, A.B.; Ranjan, A.; Welz, P.J. A Critical Review of Systems for Bioremediation of Tannery Effluent with a Focus on Nitrogenous and Sulfurous Species Removal and Resource Recovery. Processes 2024, 12, 1527. [Google Scholar] [CrossRef]
- Ali, S.K.; Abdou, M.M.; Emara, M.M.; Farag, R.S.; Mubarak, M.F. Eco-friendly solutions: A comprehensive review of natural coagulants for sustainable water treatment. Environ. Geochem. Health 2025, 47, 535. [Google Scholar] [CrossRef]
- Ma, L.; Mao, G.; Liu, J.; Yu, H.; Gao, G.; Wang, Y. Rapid quantification of bacteria and viruses in influent, settled water, activated sludge and effluent from a wastewater treatment plant using flow cytometry. Water Sci. Technol. 2013, 68, 1763–1769. [Google Scholar] [CrossRef]
- Nayana, K.; Vidya, D.; Soorya, K.; Dineshan, A.; Menon, A.S.; Mambad, R.; Arunkumar, K. Effect of Volume and Surface Area on Growth and Productivity of Microalgae in Culture System. Bioenergy Res. 2023, 16, 1013–1025. [Google Scholar] [CrossRef]
- Benner, P.; Meier, L.; Pfeffer, A.; Krüger, K.; Vargas, J.E.O.; Weuster-Botz, D. Lab-scale photobioreactor systems: Principles, applications, and scalability. Bioprocess Biosyst. Eng. 2022, 45, 791–813. [Google Scholar] [CrossRef]
- Marchese, A.; Lima, S.; Cosenza, A.; Giambalvo, F.; Scargiali, F. Effects of Light Quality Adjustment in Microalgal Cultivation: Flashing Light and Wavelength Shifts in Photobioreactor Design. Processes 2025, 13, 1159. [Google Scholar] [CrossRef]
- Sibisi, S.; Mogany, T.; Bux, F.; Rawat, I. Development and performance of microalgae-based symbiotic systems for high-strength chemical oxygen demand wastewater treatment from the sugar mills. Algal Res. 2024, 84, 103773. [Google Scholar] [CrossRef]
- Vo, T.P.; Danaee, S.; Chaiwong, C.; Pham, B.T.; Poddar, N.; Kim, M.; Kuzhiumparambil, U.; Songsomboon, C.; Pernice, M.; Ngo, H.H.; et al. Microalgae-bacteria consortia for organic pollutants remediation from wastewater: A critical review. J. Environ. Chem. Eng. 2024, 12, 114213. [Google Scholar] [CrossRef]
- Chen, H.; Chu, Z.; Bai, Z.; Hu, J.; Ma, J. The phototactic collection of microalgae and its implications for the comprehensive utilization of microalgal biomass. Ind. Crops Prod. 2025, 233, 121353. [Google Scholar] [CrossRef]
- Gawali, S.; Alam, S.N.; Tilva, J.; Jagadale, K.; Singh, B.; Guldhe, A. Development of scalable cultivation strategy for microalgal biomass generation using wastewater and integrated biorefinery approach for biofuel production. J. Environ. Chem. Eng. 2025, 13, 115441. [Google Scholar] [CrossRef]
- Kang, D.; Kim, K.T.; Heo, T.Y.; Kwon, G.; Lim, C.; Park, J. Inhibition of Photosynthetic Activity in Wastewater-Borne Microalgal–Bacterial Consortia Under Various Light Conditions. Sustainability 2019, 11, 2951. [Google Scholar] [CrossRef]
- Horn, E.J.; van Hille, R.P.; Oyekola, O.O.; Welz, P.J. Functional microbial communities in hybrid linear flow channel reactors for desulfurization of tannery effluent. Microorganisms 2022, 10, 2305. [Google Scholar] [CrossRef] [PubMed]
- Welz, P.J.; De Jonge, N.; Lilly, M.; Kaira, W.M.; Mpofu, A.B. Integrated biological system for remediation and valorization of tannery wastewater: Focus on microbial communities responsible for methanogenesis and sulfidogenesis. Bioresour. Technol. 2024, 395, 130411. [Google Scholar] [CrossRef] [PubMed]



| Method | Advantages | Disadvantages |
|---|---|---|
| Direct Methods | ||
| Dry cell weight: Centrifugation | Absolute measurement Can be accurate Current gold standard method Can be conducted with standard laboratory equipment | Time consuming May require larger volumes depending on biomass density Can over-estimate in presence of non-biomass solids |
| Dry cell weight: Filtration | As per centrifugation method | As per centrifugation method Biomass can clog filters Losses can occur with small volumes |
| Microscopic cell counts | Absolute measurement Small sample volumes Good for assessing changes in cell numbers Can combine with digital software to determine cell counts and sizes Expertise required | Inaccuracies due to differences in size, volume, density of cells Cannot measure if cells are clumped together |
| Automated cell counter | Absolute measurement Can accurately measure cell numbers and cell volumes Small sample volumes | Specialized equipment required Clumps of cells, granules or flocs reduce accuracy Interference if other solids are present Cannot account for cell density |
| Packed cell volume | Absolute measurement Simple and rapid Small sample volumes | Inaccuracies due to various factors affecting cell packing density Only wet weight determined |
| Indirect Methods | ||
| Turbidimetry | Rapid (once standard curve has been constructed) Small sample volumes | Indirect method Careful consideration of applicable wavelength for each situation Not suitable for turbid culture media or wastewater Notable differences for different strains can be found Notable differences in same strain under different conditions can be found Need for standard graphs for each microalgal strain Only suitable for pure cultures Often used ‘blindly’ at sub-optimal wavelengths |
| Chlorophyll-a UV-vis and fluorimetry | Rapid (once standard curve has been constructed) Relatively small sample volumes | Indirect method Highly unreliable for biomass Careful consideration of applicable wavelength for each situation Notable differences for different strains can be found Notable differences in same strain under different conditions can be found Need for standard graphs for each microalgal strain Only suitable for pure cultures Need to account for interference by other chlorophylls and pigments |
| Parameter | Raw | Settled | Filtered |
|---|---|---|---|
| pH | 6.6 | 6.84 | 6.83 |
| CODt (mg/L) | 5190 ± 20 | 1150 ± 11 | 882 ± 4 |
| CODs (mg/L) | 5000 ± 39 | 2553 ± 169 | 2153 ± 50 |
| VOA (mg/L) | 1246 ± 43 | 98.5 ± 6.4 | 98.5 ± 6.4 |
| NH3-N (mg/L) | 40.7 ± 1.1 | 30.6 ± 0.5 | 30.6 ± 0.5 |
| NO3−-N (mg/L) | 4.8 ± 0.4 | 2.65 ± 0.1 | 2.65 ± 0.1 |
| NO2−-N (mg/L) | 0.97 ± 0.06 | 0.85 ± 0.2 | 0.85 ± 0.2 |
| T.Alk (mgCaCO3/L) | 782 ± 68 | 276 ± 8.5 | 276 ± 8.5 |
| TP (mg/L) | 8.4 ± 0.06 | 1.25 ± 0.07 | 1.25 ± 0.07 |
| SO42− (mg/L) | 1657 ± 80 | 487.5 ± 3.5 | 487.5 ± 3.5 |
| Cl (mg/L) | 1373 ± 23 | 745 ± 7.07 | 745 ± 7.07 |
| S2− (mg/L) | 5.4 ± 0.4 | 0.28 | 0.28 |
| Turbidity | Highly turbid | Partially turbid | Clear |
| Working/Flask Volumes (mL) | TSS Concentration (g/L) | Absolute TSS (mg) | ||
|---|---|---|---|---|
| 2.5 mL Aliquot | 10 mL Aliquot | 2.5 mL Aliquot | 10 mL Aliquot | |
| 150/250 | 1.37 * | 1.23 | 3.43 | 12.3 |
| 200/250 | 1.50 * | 1.45 | 3.75 | 14.5 |
| 300/500 | - | 1.39 | - | 13.9 |
| 600/1000 | - | 1.21 | - | 12.1 |
| Raw | Settled | Filtered | |
|---|---|---|---|
| CODs day 0 (mg/L) | 2735 ± 308 | 2553 ± 169 | 2153 ± 50.1 |
| CODs day 5 (mg/L) | 1140 ± 32.8 | 1418 ± 34.0 | 1441 ± 119 |
| Removal (%) | 58.3 | 44.5 | 33.1 |
| NH3-N day 0 (mgN/L) | 66 ± 8.8 | 65 ± 11 | 75 ± 0.4 |
| NH3-N day 5 (mgN/L) | 52 ± 2.0 | 59 ± 5.7 | 49 ± 6.6 |
| Removal (%) | 21.2 | 9.23 | 34.7 |
| pH day 0 (units) | 6.8 | 6.8 | 7.5 |
| pH day 5 (units) | 9.1 | 8.9 | 9.1 |
| TSS (mg/mL) | Working Vol. (mL) | Minimum Aliquot Volume (mL) | Maximum Number of Aliquots (n) |
|---|---|---|---|
| 0.10 | 150 | 100 | 1 |
| 200 | 1 | ||
| 250 | 2 | ||
| 300 | 2 | ||
| 0.20 | 150 | 50 | 2 |
| 200 | 3 | ||
| 250 | 4 | ||
| 300 | 4 | ||
| 1.00 | 150 | 10 | 12 |
| 200 | 16 | ||
| 250 | 20 | ||
| 300 | 24 | ||
| 2.00 | 150 | 5 | 24 |
| 200 | 32 | ||
| 250 | 40 | ||
| 300 | 48 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Ranjan, A.; Ngobeni, P.V.; Welz, P.J. Measurement of Biomass in Small-Scale Microalgal and Microalgal–Bacterial Systems for Wastewater Treatment: Mini Review and Experimental Evaluation. Processes 2026, 14, 1145. https://doi.org/10.3390/pr14071145
Ranjan A, Ngobeni PV, Welz PJ. Measurement of Biomass in Small-Scale Microalgal and Microalgal–Bacterial Systems for Wastewater Treatment: Mini Review and Experimental Evaluation. Processes. 2026; 14(7):1145. https://doi.org/10.3390/pr14071145
Chicago/Turabian StyleRanjan, Amrita, Philadelphia V. Ngobeni, and Pamela Jean Welz. 2026. "Measurement of Biomass in Small-Scale Microalgal and Microalgal–Bacterial Systems for Wastewater Treatment: Mini Review and Experimental Evaluation" Processes 14, no. 7: 1145. https://doi.org/10.3390/pr14071145
APA StyleRanjan, A., Ngobeni, P. V., & Welz, P. J. (2026). Measurement of Biomass in Small-Scale Microalgal and Microalgal–Bacterial Systems for Wastewater Treatment: Mini Review and Experimental Evaluation. Processes, 14(7), 1145. https://doi.org/10.3390/pr14071145








