Valorization of Inga feuilleei (Pacay) Seeds as a Promising Adsorbent for the Removal of Direct Red 80 Dye in Aqueous Solution—Kinetics, Isotherms, Thermodynamics, and Techno-Economic Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Quantification of Direct Red 80 in Water
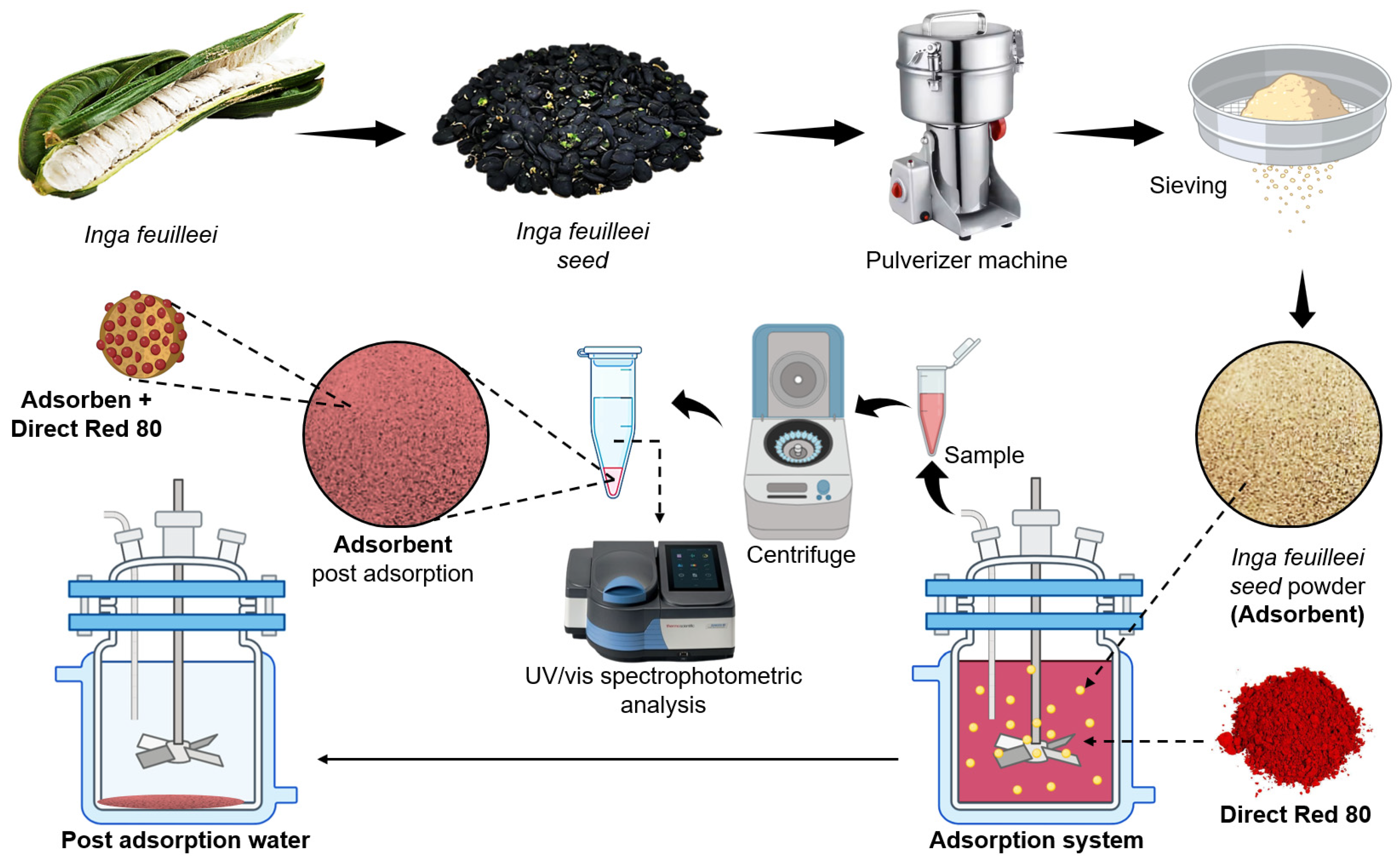
2.3. Adsorption Process
2.4. Characterization
2.5. Particle Size Effect on the Removal Process
2.6. pH Effect on the Removal Process
2.7. Kinetics
2.8. Adsorption Isotherms
2.9. Thermodynamic Study
2.10. Evaluation of Adsorbent Reuse
2.11. Techno-Economic Analysis Process
3. Results and Discussion
3.1. SEM Characterization
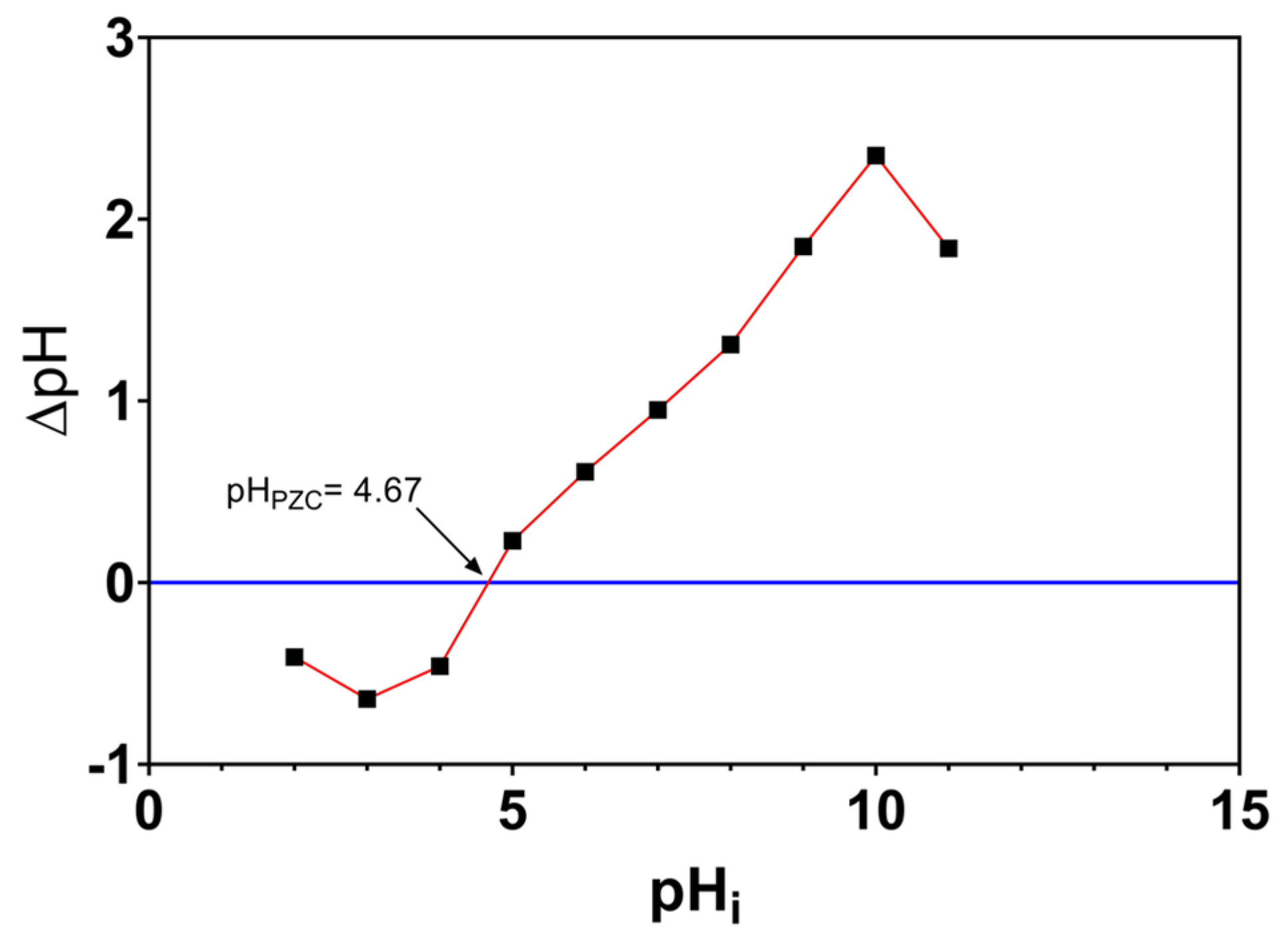
3.2. Point of Zero Charge
3.3. Effect of the Particle Size
3.4. pH Effect
3.5. FTIR Characterization
3.6. Kinetic Study
3.7. Isotherms
3.8. Thermodynamics
3.9. Reuse
3.10. Techno-Economic Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| IFS | Inga feuilleei seed |
| DR80 | Direct Red 80 |
| FTIR | Fourier Transform Infrared Spectroscopy |
| ATR | Attenuated Total Reflectance |
| pHPZC | Point of zero charge |
| SEM | Scanning Electron Microscope |
| ANOVA | Analysis of variance |
| R2 | coefficient of determination |
| qt | Adsorption capacity |
| m | Weight |
| V | Volume |
| Ci | Initial concentration |
| Cf | Final concentration |
| k1 | Pseudo-first order constant |
| k2 | Pseudo-second order |
| qe | Adsorption capacity at equilibrium |
| t | Time |
| Ce | Concentration of DR80 at equilibrium |
| qm | Maximum adsorption capacity |
| KL | Langmuir constant |
| n | Freundlich constant |
| ΔS0 | Entropy |
| ΔH0 | Enthalpy |
| ΔG0 | Gibbs energy |
| T | Temperature |
| R | Universal gas constant |
| Kc | Equilibrium constant |
| NPV | Net Present Value |
| IRR | Internal Rate of Return |
| B/C | Benefit/cost |
| IRP | Investment Recovery Period |
| CF | Cash Flow |
| I0 | Initial investment |
| i | Discount rate |
| A | Immediate previous year of recovery |
| B | Initial investment |
| C | Accumulated cash flow |
| D | Economic cash |
| COK | Cost of Opportunity of Capital |
References
- Rathinam, A.; Jaiswal, R.; Chellam, P.V. Occurrence and Distribution of Emerging Contaminants: Ozonolytic Removal in Aqueous Matrices. In Bioremediation of Emerging Contaminants in Water. Volume 1; ACS Symposium Series; American Chemical Society: Washington, DC, USA, 2024; Volume 1475, pp. 111–127. [Google Scholar] [CrossRef]
- Patel, D.; Jain, K.R.; Madamwar, D.; Desai, C. Phytotechnologies for Bioremediation of Textile Dye Wastewater. In Phytoremediation for Environmental Sustainability; Prasad, R., Ed.; Springer Nature: Singapore, 2021; pp. 251–272. [Google Scholar] [CrossRef]
- Kaur, J.; Tewari, S.; Kaur, A.; Malik, R. Dye and Dye-Containing Hazardous Waste in Water Resource. In Emerging Contaminants in Water and Wastewater: Sources and Substances; Gupta, H., Madhav, S., Dhiman, S., Mishra, R., Eds.; Springer Nature: Cham, Switzerland, 2025; pp. 179–199. [Google Scholar] [CrossRef]
- Lekhak, U.M. 2-Ecotoxicity of synthetic dyes. In Current Developments in Bioengineering and Biotechnology; Govindwar, S.P., Kurade, M.B., Jeon, B.-H., Pandey, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2023; pp. 45–67. [Google Scholar] [CrossRef]
- Chung, K.-T. Azo dyes and human health: A review. J. Environ. Sci. Health Part C Environ. Carcinog. Ecotoxicol. Rev. 2016, 34, 233–261. [Google Scholar] [CrossRef] [PubMed]
- PubChem Direct Red 80. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Direct-Red-80 (accessed on 1 March 2026).
- Song, Y.; Sun, K.; Liu, R. An exploration of the interaction mechanism of Direct Red 80 with α-Amylase at the molecular level. J. Mol. Recognit. 2021, 34, e2883. [Google Scholar] [CrossRef]
- Kurade, M.B.; Jadhav, U.U.; Phugare, S.S.; Kalyani, D.C.; Govindwar, S.P. 1-Global scenario and technologies for the treatment of textile wastewater. In Current Developments in Bioengineering and Biotechnology; Govindwar, S.P., Kurade, M.B., Jeon, B.-H., Pandey, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2023; pp. 1–43. [Google Scholar] [CrossRef]
- Sridharan, R.; Krishnaswamy, V.G. Bioremediation of Textile Dyes for Sustainable Environment—A Review. In Modern Approaches in Waste Bioremediation: Environmental Microbiology; Shah, M.P., Ed.; Springer International Publishing: Cham, Switzerland, 2023; pp. 447–460. [Google Scholar] [CrossRef]
- Ganguly, A.; Nanda, S.; Das, K.; Ghanty, S.; Biswas, G.; Mandi, M.; Mukherjee, S.; Paramanik, M.; Rajak, P. Micropollutants in Environment. In Ecosystem Management; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2024; pp. 453–492. [Google Scholar] [CrossRef]
- Yazdandoust, M.; Ehrampoush, M.H.; Dalvand, A. Moringa oleifera seed extract assisted electrocoagulation process for efficient direct dye removal from textile wastewater: Modelling, optimisation and techno-economic study. Int. J. Environ. Anal. Chem. 2024, 104, 2485–2505. [Google Scholar] [CrossRef]
- Puiatti, G.A.; Elerate, E.M.; de Carvalho, J.P.; Luciano, V.A.; de Carvalho Teixeira, A.P.; Lopes, R.P.; Teixeira de Matos, A. Reuse of iron ore tailings as an efficient adsorbent to remove dyes from aqueous solution. Environ. Technol. 2024, 45, 2308–2319. [Google Scholar] [CrossRef]
- Sadeghy, S.; Pormazar, S.M.; Ghaneian, M.T.; Ehrampoush, M.H.; Dalvand, A. Modeling and optimization of direct dyes removal from aqueous solutions using activated carbon produced from sesame shell waste. Sci. Rep. 2024, 14, 24867. [Google Scholar] [CrossRef]
- Mazlan, N.A.; Lewis, A.; Chen, Z.; Butt, F.S.; Han, J.; Radacsi, N.; Yang, S.; Huang, Y. Photocatalytic self-cleaning graphene oxide/ZnO hybrid membrane for ultrafast cyclic small organic molecule separation. J. Membr. Sci. 2024, 697, 122539. [Google Scholar] [CrossRef]
- Hamami, Z.; Javanbakht, V. Photocatalytic processes using potato peel extract-mediated CuO nanophotocatalyst for fast and efficient direct red 80 dye removal. Biomass Convers. Biorefin. 2025, 15, 563–573. [Google Scholar] [CrossRef]
- do Carmo Dias, G.; de Souza, N.C.S.; de Souza, E.I.P.; Puiatti, G.A.; Moreira, R.P.L. Enhanced degradation of Direct Red 80 dye via Fenton-like process mediated by cobalt ferrite: Generated superoxide radicals and singlet oxygen. Environ. Sci. Pollut. Res. 2024, 31, 28025–28039. [Google Scholar] [CrossRef] [PubMed]
- Kouzi, Y.; Chafiq Elidrissi, Z.; Achiou, B.; Beqqour, D.; Alami Younssi, S.; Rabiller-Baudry, M.; Bouhria, M.; Ouammou, M. Enhancing stability and performance of graphene oxide membrane grafted on low-cost rich-silica support: A comparative study of two activation approaches. Process Saf. Environ. Prot. 2024, 188, 1574–1583. [Google Scholar] [CrossRef]
- Essate, A.; El Machtani Idrissi, D.; Achiou, B.; Adlane, S.; Breida, M.; Curcio, S.; Chakraborty, S.; Alami Younssi, S.; Ouammou, M. High-performance zeolite-A/polystyrene composite membrane tailored for efficient soluble dyes separation and antifouling. Colloids Surf. Physicochem. Eng. Asp. 2024, 703, 135162. [Google Scholar] [CrossRef]
- Liu, J.; Qin, Z.; Yang, L.; Wei, J.; Feng, C.; Tang, B.; Jiang, W.; Yang, L.; Yao, L.; Zheng, J.; et al. Ultra-fast molecular sieving in ZIF-67 and cellulose nanofibers based thin-film nanocomposite membrane. Sep. Purif. Technol. 2025, 355, 129753. [Google Scholar] [CrossRef]
- Yang, M.; Li, L.; Wu, X.; Tian, H. Preparation of covalent organic framework mixed matrix membranes and their application in liquid separation. J. Environ. Chem. Eng. 2025, 13, 118836. [Google Scholar] [CrossRef]
- Adlane, S.; Essate, A.; Naim, J.; Achiou, B.; Tiraferri, A.; Aaddane, A.; Youssefi, S.; Ouammou, M.; Alami Younssi, S. Development and characterization of a purified pyrophyllite-based ultrafiltration membrane for the treatment of dye-contaminated water. J. Environ. Chem. Eng. 2025, 13, 118579. [Google Scholar] [CrossRef]
- Kouzi, Y.; Chafiq Elidrissi, Z.; Essate, A.; Beqqour, D.; Achiou, B.; Alami Younssi, S.; Rabiller-Baudry, M.; Bouhria, M.; Ouammou, M. Tailored rGO-TiO2-pPD low-cost ceramic membrane for dye wastewater filtration: A synergistic strategy of GO reduction, intercalation and crosslinking. Sep. Purif. Technol. 2025, 378, 134643. [Google Scholar] [CrossRef]
- Barman, F.; Nath, S.; Dey, S.; Ansari, M.T.A.; Kundu, R.; Paul, S. Aquatic Plant-Mediated Bioremediation Strategies. In Toxicity of Aquatic System and Remediation; CRC Press: Boca Raton, FL, USA, 2024. [Google Scholar] [CrossRef]
- Alsharif, M.A. Understanding Adsorption: Theories, Techniques, and Applications; IntechOpen: London, UK, 2025. [Google Scholar] [CrossRef]
- Akhtar, M.; Hasany, S.M.; Bhanger, M.I.; Iqbal, S. Low cost sorbents for the removal of methyl parathion pesticide from aqueous solutions. Chemosphere 2007, 66, 1829–1838. [Google Scholar] [CrossRef]
- Mo, J.; Yang, Q.; Zhang, N.; Zhang, W.; Zheng, Y.; Zhang, Z. A review on agro-industrial waste (AIW) derived adsorbents for water and wastewater treatment. J. Environ. Manag. 2018, 227, 395–405. [Google Scholar] [CrossRef]
- Prance, G.T. FRUITS OF TROPICAL CLIMATES|Fruits of Central and South America. In Encyclopedia of Food Sciences and Nutrition (Second Edition); Caballero, B., Ed.; Academic Press: Oxford, UK, 2003; pp. 2810–2816. [Google Scholar] [CrossRef]
- Lima, L.d.S.; Fernandes, E.P.; Novelli, A.; da Costa, L.P.; Monteiro, M.D.S.; dos Santos, M.V.Q.; Freitas, J.A.; Sussuchi, E.M. Adsorption and ecotoxicology studies with aqueous solution of Cr(VI) ions using adsorbent materials derived from Inga edulis. Chemosphere 2025, 378, 144378. [Google Scholar] [CrossRef]
- Silva, E.M.; Pompeu, D.R.; Larondelle, Y.; Rogez, H. Optimisation of the adsorption of polyphenols from Inga edulis leaves on macroporous resins using an experimental design methodology. Sep. Purif. Technol. 2007, 53, 274–280. [Google Scholar] [CrossRef]
- Georgin, J.; Franco, D.S.P.; Netto, M.S.; de Salomón, Y.L.O.; Piccilli, D.G.A.; Foletto, E.L.; Dotto, G.L. Adsorption and mass transfer studies of methylene blue onto comminuted seedpods from Luehea divaricata and Inga laurina. Environ. Sci. Pollut. Res. 2021, 28, 20854–20868. [Google Scholar] [CrossRef]
- Franco, D.S.P.; Georgin, J.; Netto, M.S.; Fagundez, J.L.S.; Salau, N.P.G.; Allasia, D.; Dotto, G.L. Conversion of the forest species Inga marginata and Tipuana tipu wastes into biosorbents: Dye biosorption study from isotherm to mass transfer. Environ. Technol. Innov. 2021, 22, 101521. [Google Scholar] [CrossRef]
- Correia, L.P.; Peron, A.P.; Sia, N.B.P.; Medeiros, F.V.d.S. Evaluation of Macrolobium acaccifolium and Inga nobilis Seeds in Coagulation/flocculation Processes. Chem. Eng. Trans. 2022, 92, 1–6. [Google Scholar] [CrossRef]
- Brennan, E.B.; Mudge, K.W. Vegetative propagation of Inga feuillei from shoot cuttings and air layering. New For. 1998, 15, 37–51. [Google Scholar] [CrossRef]
- Lim, T.K. Inga feuillei . In Edible Medicinal and Non-Medicinal Plants: Volume 2, Fruits; Lim, T.K., Ed.; Springer: Dordrecht, The Netherlands, 2012; pp. 720–722. [Google Scholar] [CrossRef]
- Choquenaira-Quispe, C.; Villanueva-Salas, J.A.; Yucra-Condori, H.R.; Angulo Vargas, S.J.; Rojas Tamata, K.; Gonzales-Condori, E.G. “Chacco” clay from the Peruvian highlands as a potential adsorbent of heavy metals in water. Energy Nexus 2024, 16, 100330. [Google Scholar] [CrossRef]
- Gonzales-Condori, E.G.; Avalos-López, G.; Gonzales-Condori, J.; Mujica-Guzmán, A.; Terán-Hilares, R.; Briceño, G.; Quispe-Avilés, J.M.; Parra-Ocampo, P.J.; Villanueva-Salas, J.A. Universidad Tecnológica del Perú Avocado seed powder residues as a promising bio-adsorbent for color removal from textile wastewater. Rev. Mex. Ing. Quím. 2023, 22, 1–23. [Google Scholar] [CrossRef]
- Gonzales-Condori, E.G.; Bejar-Aramburu, D.G.; Gutierrez-Delgado, J.A.; Villanueva-Salas, J.A.; Gonzales-Condori, J.; Salazar-Pinto, B.M. Valorization of grape seed residues post-oil extraction as an adsorbent for hexavalent chromium in water. Kinetics, isotherms, thermodynamics, and techno-economic analysis. Energy Nexus 2025, 18, 100437. [Google Scholar] [CrossRef]
- Inyinbor, A.A.; Adekola, F.A.; Olatunji, G.A. Copper scavenging efficiency of adsorbents prepared from Raphia hookeri fruit waste. Sustain. Chem. Pharm. 2019, 12, 100141. [Google Scholar] [CrossRef]
- Macedo, M.C.C.; Correia, V.T.d.V.; Silva, V.D.M.; Pereira, D.T.V.; Augusti, R.; Melo, J.O.F.; Pires, C.V.; de Paula, A.C.C.F.F.; Fante, C.A. Development and Characterization of Yellow Passion Fruit Peel Flour (Passiflora edulis f. flavicarpa). Metabolites 2023, 13, 684. [Google Scholar] [CrossRef] [PubMed]
- Montalvo Andia, J.; Larrea, A.; Salcedo, J.; Reyes, J.; Lopez, L.; Yokoyama, L. Synthesis and characterization of chemically activated carbon from Passiflora ligularis, Inga feuilleei and native plants of South America. J. Environ. Chem. Eng. 2020, 8, 103892. [Google Scholar] [CrossRef]
- Ait Haki, M.; Imgharn, A.; Aarab, N.; Hsini, A.; Essekri, A.; Laabd, M.; El Jazouli, H.; Elamine, M.; Lakhmiri, R.; Albourine, A. Efficient removal of crystal violet dye from aqueous solutions using sodium hydroxide-modified avocado shells: Kinetics and isotherms modeling. Water Sci. Technol. 2021, 85, 433–448. [Google Scholar] [CrossRef]
- Liu, Y.; Miao, J.; Han, H.; Xu, P. Differences in Influence of Particle Size on the Adsorption Capacity between Deformed and Undeformed Coal. ACS Omega 2021, 6, 5886–5897. [Google Scholar] [CrossRef] [PubMed]
- Pirillo, S.; Ferreira, M.L.; Rueda, E.H. The effect of pH in the adsorption of Alizarin and Eriochrome Blue Black R onto iron oxides. J. Hazard. Mater. 2009, 168, 168–178. [Google Scholar] [CrossRef] [PubMed]
- Sathya, K.; Jayalakshmi, H.; Reddy, S.N.; Ratnam, M.V.; Bandhu, D. Effective removal of Congo red dye using adsorbent prepared from bio-waste: Isotherm, kinetic, and thermodynamic studies. Biomass Convers. Biorefin. 2025, 15, 3557–3569. [Google Scholar] [CrossRef]
- Akhouairi, S.; Ouachtak, H.; Addi, A.A.; Jada, A.; Douch, J. Natural Sawdust as Adsorbent for the Eriochrome Black T Dye Removal from Aqueous Solution. Water Air Soil Pollut. 2019, 230, 181. [Google Scholar] [CrossRef]
- Reddy, Y.S.; Jose, T.J.; Dinesh, B.; Kumar, R.N.; Kumar, P.S.; Kaviyarasu, K. Equilibrium, kinetic, and thermodynamic study of Direct Yellow 12 dye adsorption by biomass-derived porous graphitic activated carbon. Biomass Convers. Biorefin. 2025, 15, 6817–6833. [Google Scholar] [CrossRef]
- Serban, G.V.; Iancu, V.I.; Dinu, C.; Tenea, A.; Vasilache, N.; Cristea, I.; Niculescu, M.; Ionescu, I.; Chiriac, F.L. Removal Efficiency and Adsorption Kinetics of Methyl Orange from Wastewater by Commercial Activated Carbon. Sustainability 2023, 15, 12939. [Google Scholar] [CrossRef]
- Arami, M.; Limaee, N.Y.; Mahmoodi, N.M.; Tabrizi, N.S. Removal of dyes from colored textile wastewater by orange peel adsorbent: Equilibrium and kinetic studies. J. Colloid Interface Sci. 2005, 288, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Doulati Ardejani, F.; Badii, K.; Yousefi Limaee, N.; Mahmoodi, N.M.; Arami, M.; Shafaei, S.Z.; Mirhabibi, A.R. Numerical modelling and laboratory studies on the removal of Direct Red 23 and Direct Red 80 dyes from textile effluents using orange peel, a low-cost adsorbent. Dye. Pigment. 2007, 73, 178–185. [Google Scholar] [CrossRef]
- Doulati Ardejani, F.; Badii, K.; Limaee, N.Y.; Shafaei, S.Z.; Mirhabibi, A.R. Adsorption of Direct Red 80 dye from aqueous solution onto almond shells: Effect of pH, initial concentration and shell type. J. Hazard. Mater. 2008, 151, 730–737. [Google Scholar] [CrossRef]
- Ben Jeddou, K.; Bouaziz, F.; Ben Taheur, F.; Nouri-Ellouz, O.; Ellouz-Ghorbel, R.; Ellouz-Chaabouni, S. Adsorptive removal of direct red 80 and methylene blue from aqueous solution by potato peels: A comparison of anionic and cationic dyes. Water Sci. Technol. 2021, 83, 1384–1398. [Google Scholar] [CrossRef]
- Arami, M.; Limaee, N.Y.; Mahmoodi, N.M.; Tabrizi, N.S. Equilibrium and kinetics studies for the adsorption of direct and acid dyes from aqueous solution by soy meal hull. J. Hazard. Mater. 2006, 135, 171–179. [Google Scholar] [CrossRef]
- Aljeboree, A.M.; Alshirifi, A.N.; Alkaim, A.F. Kinetics and equilibrium study for the adsorption of textile dyes on coconut shell activated carbon. Arab. J. Chem. 2017, 10, S3381–S3393. [Google Scholar] [CrossRef]
- Matemáticas Financieras y Valoración-Google Books. Available online: https://www.google.com.pe/books/edition/Matem%C3%A1ticas_financieras_y_valoraci%C3%B3n/jCF1EAAAQBAJ?hl=es-419&gbpv=0 (accessed on 28 February 2026).
- cajaarequipa.pe Tarifario de Tasas de Interés Activas. Caja Arequipa. Available online: https://www.cajaarequipa.pe/tasas-de-interes/tarifario-de-tasas-de-interes-activas/ (accessed on 28 February 2026).
- Magueyal Salas, J.L. Análisis y Reflexiones Sobre los Costos de Producción y Ventas; Editorial Digital del Tecnológico de Monterrey: Monterrey, Mexico, 2020. [Google Scholar]
- Fernández Espinoza, S. Los Proyectos de Inversión: Evaluación Financiera; Editorial Tecnologica de Costa Rica: Cartago, Costa Rica, 2007. [Google Scholar]
- Guzman Castro, F. El Estudio Económico-Financiero y la Evaluación en Proyectos de la Industria Química; Universidad Nacional de Colombia: Bogota, Colombia, 2001. [Google Scholar]
- Torrents, A.S.; Vilda, F.G.; Postils, I.A.; Postils, I.A. Manual Práctico de Diseño de Sistemas Productivos; Ediciones Díaz de Santos: Madrid, Spain, 2004. [Google Scholar]







| T (°C) | Ci (mg/L) | Removal % | qexp (mg/g) | Pseudo-First Order | Pseudo-Second Order | ||||
|---|---|---|---|---|---|---|---|---|---|
| k1 (min−1) | qe, cal (mg/g) | R2 | k2 (g mg−1 min−1) | qe, cal (mg/g) | R2 | ||||
| 20 | 100 | 77.55 | 80.68 | 0.0821 | 74.13 | 0.9279 | 0.0013 | 81.21 | 0.9796 |
| 200 | 55.59 | 104.38 | 0.0882 | 95.37 | 0.9330 | 0.0011 | 104.29 | 0.9822 | |
| 300 | 41.86 | 120.84 | 0.0775 | 111.74 | 0.9652 | 0.0008 | 122.75 | 0.9932 | |
| 400 | 26.08 | 105.66 | 0.0606 | 117.22 | 0.9669 | 0.0007 | 108.71 | 0.9947 | |
| 30 | 100 | 88.37 | 92.41 | 0.0919 | 85.05 | 0.9372 | 0.0013 | 92.82 | 0.9842 |
| 200 | 61.14 | 114.81 | 0.1066 | 105.11 | 0.9438 | 0.0015 | 112.82 | 0.9847 | |
| 300 | 50.02 | 144.40 | 0.1309 | 136.14 | 0.9656 | 0.0013 | 145.25 | 0.9942 | |
| 400 | 33.15 | 130.88 | 0.1269 | 142.35 | 0.9379 | 0.0013 | 151.17 | 0.9825 | |
| 40 | 100 | 94.44 | 98.76 | 0.1021 | 91.32 | 0.9370 | 0.0014 | 98.98 | 0.9839 |
| 200 | 67.22 | 126.22 | 0.1838 | 118.17 | 0.9652 | 0.0024 | 123.91 | 0.9915 | |
| 300 | 54.95 | 158.62 | 0.1969 | 148.32 | 0.9619 | 0.0021 | 155.15 | 0.9894 | |
| 400 | 40.31 | 163.32 | 0.1921 | 155.45 | 0.9747 | 0.0020 | 162.38 | 0.9956 | |
| 50 | 100 | 99.39 | 103.93 | 0.1192 | 97.67 | 0.9465 | 0.0016 | 104.73 | 0.9873 |
| 200 | 78.05 | 146.56 | 0.1850 | 137.81 | 0.9645 | 0.0021 | 144.45 | 0.9915 | |
| 300 | 63.90 | 184.44 | 0.2064 | 175.37 | 0.9795 | 0.0021 | 182.34 | 0.9975 | |
| 400 | 48.64 | 195.38 | 0.2326 | 187.42 | 0.9815 | 0.0023 | 194.12 | 0.9951 | |
| Model | Parameters | Value | |||
|---|---|---|---|---|---|
| 20 °C | 30 °C | 40 °C | 50 °C | ||
| Langmuir | qm (mg/g) | 133.11 | 146.78 | 152.78 | 183.51 |
| KL (L/mg) | 0.0601 | 0.1229 | 0.2901 | 2.065 | |
| R2 | 0.9924 | 0.9544 | 0.9520 | 0.9862 | |
| Freundlich | 1/n | 0.1840 | 0.1703 | 0.1417 | 0.1019 |
| KF | 46.13 | 58.81 | 74.55 | 113.51 | |
| R2 | 0.9669 | 0.8931 | 0.9133 | 0.9466 | |
| Adsorbent | T (K) | pH | C0 (mg/L) | Adsorbent Dosage (g/L) | Stirring Time (min)/Stirring Speed (rpm) | Particle Size (μm) | qmax (mg/g) | Reference |
|---|---|---|---|---|---|---|---|---|
| Orange peel | 298 | 2 | 50–125 | 4 | 25/200 | -- | 21.05 | [48,49] |
| Mixture shells | 293 | 6 | 50–150 | 3.2 | 300/200 | <106 | 28.50 | [50] |
| External shells | 293 | 6 | 50–150 | 3.2 | 300/200 | <106 | 23.753 | [50] |
| Internal shells | 293 | 6 | 50–150 | 3.2 | 300/200 | <106 | 22.00 | [50] |
| Potato peels | 303.16 | 2 | 100 | 20 | 60/100 | 100 | 27.778 | [51] |
| Potato peels | 313.16 | 2 | 100 | 20 | 60/100 | 100 | 45.45 | [51] |
| Potato peels | 323.16 | 2 | 100 | 20 | 60/100 | 100 | 32.258 | [51] |
| Soy Meal Hull | 293 | 2 | 50–150 | 0.3 | 120/200 | <125 | 178.57 | [52] |
| Inga feuilleei seed powder | 293 | 2 | 100–400 | 1 | 240/350 | 150 | 133.11 | This research |
| 303 | 2 | 100–400 | 1 | 240/350 | 150 | 146.78 | This research | |
| 313 | 2 | 100–400 | 1 | 240/350 | 150 | 152.78 | This research | |
| 323 | 2 | 100–400 | 1 | 240/350 | 150 | 183.51 | This research |
| T (K) | ΔH° (kJ/mol) | ΔS° (J/mol·K) | ΔG° (kJ/mol) |
|---|---|---|---|
| 293 | 67.85 | 241.21 | −2.98 |
| 303 | −5.12 | ||
| 313 | −7.38 | ||
| 323 | −10.30 |
| Parameter | Value |
|---|---|
| NPV (USD) | 35,701.46 |
| IRR (%) | 25.47 |
| B/C | 1.49 |
| IRP | 3.6289 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Gonzales-Condori, E.G.; Sotomayor-Asencio, M.L.; Mayhuire-Centeno, M.F.; Paz-Corrales, O.A.; Gonzales-Condori, J.; Villanueva-Salas, J.A. Valorization of Inga feuilleei (Pacay) Seeds as a Promising Adsorbent for the Removal of Direct Red 80 Dye in Aqueous Solution—Kinetics, Isotherms, Thermodynamics, and Techno-Economic Analysis. Processes 2026, 14, 968. https://doi.org/10.3390/pr14060968
Gonzales-Condori EG, Sotomayor-Asencio ML, Mayhuire-Centeno MF, Paz-Corrales OA, Gonzales-Condori J, Villanueva-Salas JA. Valorization of Inga feuilleei (Pacay) Seeds as a Promising Adsorbent for the Removal of Direct Red 80 Dye in Aqueous Solution—Kinetics, Isotherms, Thermodynamics, and Techno-Economic Analysis. Processes. 2026; 14(6):968. https://doi.org/10.3390/pr14060968
Chicago/Turabian StyleGonzales-Condori, Elvis G., Madelin L. Sotomayor-Asencio, Marycielo F. Mayhuire-Centeno, Olivia A. Paz-Corrales, Jonathan Gonzales-Condori, and José A. Villanueva-Salas. 2026. "Valorization of Inga feuilleei (Pacay) Seeds as a Promising Adsorbent for the Removal of Direct Red 80 Dye in Aqueous Solution—Kinetics, Isotherms, Thermodynamics, and Techno-Economic Analysis" Processes 14, no. 6: 968. https://doi.org/10.3390/pr14060968
APA StyleGonzales-Condori, E. G., Sotomayor-Asencio, M. L., Mayhuire-Centeno, M. F., Paz-Corrales, O. A., Gonzales-Condori, J., & Villanueva-Salas, J. A. (2026). Valorization of Inga feuilleei (Pacay) Seeds as a Promising Adsorbent for the Removal of Direct Red 80 Dye in Aqueous Solution—Kinetics, Isotherms, Thermodynamics, and Techno-Economic Analysis. Processes, 14(6), 968. https://doi.org/10.3390/pr14060968







