1. Introduction
Poultry farming provides a significant portion of the food necessary for human life, while also contributing to the protection and conservation of bird species at risk of extinction [
1,
2]. Currently, in Romania, poultry farming is one of the most important economic sectors, with poultry meat being the preferred choice for human consumption [
3].
The poultry sector generates a series of residues (manure, wastewater) that present a risk to the environment and human health. The wastewater resulting from this sector has a high load of organic substances (blood, proteins, fats), pathogens, and antibiotic residues or other chemical substances (heavy metals, microplastics, etc.). Advanced treatments of residues (manure, wastewater) by various biological, physicochemical, or combined methods can ensure their safe discharge or reuse according to the principles of the circular economy [
4,
5,
6].
Innovative technologies in poultry farming and the use of antibacterial substances, especially those with bactericidal effects, have contributed to reducing mortality and disease in poultry farms, thereby increasing meat and egg production. However, in many parts of the world, subtherapeutic doses of antibiotics are still used daily to promote rapid growth and prevent disease in poultry [
7,
8]. The use of antibacterial substances in poultry farms should be limited strictly to therapeutic purposes, not for preventive use. According to data published by the World Health Organization (WHO), the number of deaths attributable to antimicrobial resistance is projected to reach approximately 10 million annually by 2050. An immediate consequence of this trend will be a reduction in the global gross domestic product by an estimated 2% to 3.5% [
3,
9,
10].
Within the European Union, such practices are no longer permitted, with the use of antibiotics being strictly regulated due to the risk of emergence and environmental dissemination of resistant bacterial strains [
11,
12,
13]. The primary health concern lies in the transfer of antibiotic resistance genes to humans and the development of so-called “superbugs” exhibiting atypical behavior [
14]. The prevention and control of diseases caused by microbial agents in poultry farming must be approached holistically, emphasizing the importance of implementing preventive measures against pathogenic agents, which include hygiene and disinfection protocols, biosecurity measures, management practices, and vaccination programs [
15,
16,
17,
18,
19].
The regulations imposed by the European Union are stipulated in Article 107 of Regulation (EU) 2019/6, which sets forth three key provisions regarding metaphylaxis and prophylaxis [
20].
In Romania, the official state authority responsible for the use, commercialization, and quality control of veterinary medicinal products is the National Sanitary Veterinary and Food Safety Authority (NSVFSA). The use of antibacterial substances in poultry farming is regulated by Article 107 of Regulation (EU) 2019/6, as well as by a series of orders issued by NSVFSA [
21]. Consequently, the administration of antimicrobial substances, particularly antibiotics, for the control of microbial infections in poultry farms is restricted to authorized personnel. The veterinary practitioner is responsible for determining, based on clinical evaluation, the appropriate treatment and the method of antibiotic administration [
22,
23,
24].
In poultry farming, the criteria for selecting an antibiotic include the animal’s weight, the origin of the medicinal product, its tendency to accumulate in the organism, its elimination profile, and the overall health status of the poultry. Indiscriminate administration or the use of antibiotics at preventive doses in farms promotes bacterial resistance to antibiotics, with associated risks for the environment and consumer health [
18,
23,
24].
Chlorine-based products represent a well-known category of hygiene agents widely used across multiple sectors, with broad domestic and industrial applications, available in various forms (powder, gel, liquid) under different trade names and price ranges [
25]. According to technical specifications, chlorine-based products are primarily employed for disinfection and for limiting the development of microorganisms, aspects that have been analyzed in various contexts [
24,
25,
26,
27].
In the poultry sector, the wastewater treatment techniques employed ensure the sanitary limitation of the potential microbiological risk associated with wastewater. However, the use of such wastewater, sewage sludge, or manure resulting from poultry production in agriculture still requires careful attention, as there remains a risk of soil and groundwater contamination with various pathogenic agents [
25]. The effectiveness of pathogen removal through wastewater treatment techniques applied in the poultry sector represents a critical guarantee for both environmental protection and public health [
26,
27,
28].
Conventional wastewater treatment processes and techniques applied to effluents generated by slaughterhouse activities do not fully ensure the removal of pollutant compounds. In addition, these methods present several disadvantages, such as requiring large land areas for the installation of treatment equipment, involving high maintenance costs, consuming significant amounts of energy, and exhibiting limited adaptability to varying parameters and conditions [
29,
30,
31,
32].
In most cases, advanced treatment technologies do not replace conventional systems; rather, they complement them to achieve a higher level of purification of industrial effluents [
33,
34]. One of the most efficient and advanced methods for retaining pollutant compounds from industrial effluents is membrane technology, applied individually after conventional processes or combined with a disinfection stage [
35,
36,
37,
38,
39,
40]. Treatment techniques using membrane filtration, accompanied by disinfection processes with or without Advanced Oxidation Processes (AOPs), have proven effective in removing both organic and inorganic compounds as well as pathogenic agents from poultry wastewater [
38,
39,
40,
41].
Nevertheless, in treated water subjected to the aforementioned non-conventional processes, pathogenic agents that have developed resistance and subsequently reproduced or regenerated can still be detected [
37,
38,
39,
40,
41].
Recent studies emphasize that conventional anaerobic digestion represents a robust baseline technology for livestock wastewater management, providing simultaneous pathogen reduction, organic matter stabilization, and energy recovery in the form of biogas. Beyond these well-established functions, emerging research highlights the growing importance of digestate valorization pathways, particularly through the recovery of nutrients and humic-like substances with demonstrated agronomic benefits. While comprehensive analyses underline the role of anaerobic digestion in generating humic and fulvic compounds suitable for soil amendment and circular bioeconomy applications, complementary studies point to the potential of integrating post-treatment and valorization strategies, such as microalgae-based systems [
42], to further enhance nutrient recycling and environmental performance. These studies position anaerobic digestion not as an endpoint, but as a central platform within integrated treatment chains, where additional processes can be selectively applied depending on regulatory requirements, reuse objectives, and sustainability targets [
43].
The ability of bacteria to survive wastewater treatment and disinfection techniques is considerable, and the mechanisms enabling bacteria to repair, survive, and reproduce following these processes are diverse [
44]. Understanding the conditions under which these bacteria persist and undergo regrowth in the environment (water, soil) remains an important topic for society, particularly for mitigating negative impacts [
44]. Currently, the range of substances with antibacterial potential available on the market is extensive, and their improper use by untrained personnel in the poultry sector can cause significant harm to both the environment and public health [
11,
14,
20]. The use of disinfectants and antimicrobial products by small-scale farmers in poultry production is a common practice in Romanian society [
21]. The habits of use, the diversity of substances, and their modes of administration constituted one of the objectives of this study. The selection of substances with antimicrobial effect was carried out using a questionnaire [
45,
46,
47].
This article aimed to assess the use of antimicrobial products for evaluating the behavior of bacterial strains isolated from advanced-treated water originating from the poultry sector. Pre-treated wastewater from poultry processing was first subjected to non-conventional treatment processes (UF, UV, RO). These water samples were microbiologically evaluated by determining the total bacterial count (CFU/mL). Furthermore, after culture evaluation, four bacterial strains that exhibited the highest survival and reactivation following additional treatments applied to the pre-treated wastewater (wastewater from a poultry slaughterhouse) were randomly isolated in pure culture using specific microbiological methods. These bacterial strains were subsequently exposed to antimicrobial substances at varying concentrations and contact times. The innovation of our research is the optimization by mathematical modeling of two parameters (dose and action time) of antimicrobial substances on the bacterial optical densities, as a response function.
2. Materials and Methods
2.1. Additional Wastewater Treatment from a Poultry Slaughterhouse
In this study, pre-treated wastewater originating from the poultry sector in the Moldova region of Romania was utilized. The samples were collected in April 2025 and subjected to a series of advanced treatment processes.
According to the literature, the washing and evisceration stages are the main sources of effluent and pollutant loads (blood, proteins, fats/oils, solids). At the site analyzed, the specific water consumption is approximately 6.51 m
3/t carcass (≈15 L/2.3 kg carcass). The poultry slaughterhouse processes up to 160 t/day of carcasses, and the water supply is provided from its own borehole 70% and the public system 30% [
29].
The pre-treated wastewater (PT–W) was sampled after the treatment stage involving screening, bar racks, and Dissolved Air Flotation (DAF). The pre-treatment process applied by the poultry slaughterhouse, as well as the sampling points, are described in accordance with our previous research [
29].
Typically, wastewater from poultry slaughterhouses has high loads: COD (Chemical Oxygen Demand) ≈ 1837.75 mg·L
−1, BOD
5 (Biochemical Oxygen Demand (5 days)) ≈ 738.00 mg·L
−1, TSS (Total Suspended Solids) ≈ 1326.75 mg·L
−1, SO
42− ≈ 617.50 mg·L
−1, TKN (Total Kjeldahl Nitrogen) ≈ 238.29 mg·L
−1, and total bacteria count (TBC) ≈ 1 × 10
7 CFU/mL at 37 °C. In study [
30], the DAF stage led to substantial reductions in pollutants, particularly for NTG (≈99–100%) and TSS (≈97.6%), and TKN (≈86.5%); for COD, BOD
5, and SO
42−, reductions of approximately 80%, 73%, and 78% were achieved [
29].
Following pre-treatment, the water samples were collected and subjected to various treatment configurations, including membrane processes such as UF and RO, as well as UV disinfection combined with AOPs. Accordingly, the samples analyzed in this study were collected after each applied non-conventional treatment configuration.
UF was performed using a polysulfone (PSf) hollow fiber membrane with a pore diameter of ≈0.01 µm and an active surface area of 0.6 m2. The UF process was performed at a TMP (Transmembrane Pressure) of 0.7–1.3 bar, with filtration times of 60 and 75 min, respectively, to ensure a total volume of 50 L of real effluent. In parallel, a disinfection/AOP installation (maximum pressure 10 bar, 2–40 °C, 80 W UV lamp, wavelength 254 nm) was implemented to inactivate pathogens and oxidize residual organic compounds in the treated effluent. All stands were tested on real effluent under different operating conditions to synergistically evaluate the performance of UF, RO, and disinfection/AOP. UV disinfection was performed at exposure doses of 400, 600, and 800 J·m−2, where the logarithmic reduction was followed. Depending on the UV intensity and implicitly the exposure dose, the HRT (Hydraulic Retention Time) fluctuated between 5 and 13 s. UV/AOP was tested at different H2O2 concentrations of 15, 25, 35 mg·L−1. The UV intensity varied in the first part of the experiment, where later, after 10 min, it stabilized depending on the solid load in the pre-treated effluent. The UV intensity fluctuation range was 40–65 W/m. A total of 30 L of pre-treated and stabilized effluent was used in the disinfection system. In the RO stage, a flat module with a polyamide (PA) membrane was used, operated in the range of 6–8 bar, with flow rates ranging between 0.5 and 3 L·min−1 as a purification stage for advanced removal of salts and pathogens in the effluent. The process was carried out at an equal time interval of 210 min, regardless of the operating conditions achieved.
The bacterial strains labeled “B1–B4” originated from water samples treated through advanced methods, and their respective treatment configurations are detailed in
Table 1.
Although additional treatment of wastewater from a poultry slaughterhouse quantitatively reduces the number of bacteria, their recovery is reported differently in all treatment techniques [
44].
2.2. Quantitative Determination of Poultry Wastewater Microbiota and Isolation in Pure Culture of B1–B4 Bacterial Strains
The B1–B4 bacterial strains were isolated in pure culture using the streak plate technique on Peptone Casein Agar medium (PCA) (Merck, Darmstadt, Germany) (
Figure 1).
To obtain B1–B4 strains (
Figure 1), the following steps were performed: quantitative determination of the microbial load in the additionally treated wastewater samples, according to the methods specified in
Table 1, using the colony count technique on solid medium (CFU/mL) (
Figure 1a,b), and isolation of B1–B4 strains in pure culture by the streak plate technique (
Figure 1c,d) [
48,
49,
50].
After the quantitative evaluation of the water samples, the Petri plates were analyzed, and the most representative colonies for each type of wastewater treatment were randomly selected. These colonies were isolated in pure culture using specific microbiological techniques (growth parameters: 37 °C, for 24–48 h) (
Figure 1c,d).
The steps performed for this objective were as follows: transferring the selected colony under aseptic conditions onto Petri plates containing solid PCA agar using the streak plate technique; performing the catalase test; and conducting cultural and microscopic evaluation of the developed bacterial culture after 24 h. The bacterial strain isolated in pure culture was preserved and subsequently used in the experiments (B1–B4) (
Figure 1d) [
48,
49,
50,
51].
2.3. Evaluation of the Antimicrobial Substances on B1–B4 Bacterial Strains
The antimicrobial substances used in this study were as follows: the antibiotic (AB) Avianprotect Ten (Romvac Company S.A., Ilfov, Romania) [
52] and the antiseptic (AS) Clorom (S.C. G&M 2000 S.R.L., Bucharest, Romania) [
53,
54]. Avianprotect Ten contains 100 mg: tylosin tartrate 6.4 mg, erythromycin thiocyanate 1.6 mg, and neomycin sulfate 10 mg. In the case of Clorom, each tablet releases 1.5 g of active chlorine. The selection of these substances was based both on findings from the scientific literature [
55,
56,
57,
58,
59,
60,
61] and on a questionnaire designed to obtain a realistic overview of how antimicrobial agents are used in practice [
62].
To test the action of Clorom and Avianprotect Ten on B1–B4 bacterial strains, the following protocol was followed: the strains were transferred under aseptic conditions into liquid culture medium. From the pure cultures of B1–B4 previously isolated on solid PCA medium (
Figure 1d), an inoculum with a density of 0.5 McFarland units (McF) was prepared by suspending and homogenizing the sample in sterile physiological saline solution.
The liquid medium used had a composition similar to the solid PCA medium, but without agar, consisting of casein peptone 5 g, yeast extract 2.5 g, glucose 1 g, and pH 7.0 ± 0.2.
After transferring the bacterial strains into the liquid medium, 1 mL of each antimicrobial solution was added under aseptic conditions to evaluate their effect on B1–B4 strains. The antimicrobial solutions of Clorom or Avianprotect Ten were freshly prepared by grinding and dissolving the substances in bidistilled water, according to the desired concentrations. Three concentrations of each substance (1, 4, and 7 mg·L−1) were tested in triplicate, with exposure durations of 2, 13, and 24 h. The antimicrobial activity of Clorom or Avianprotect Ten on B1–B4 strains was assessed by measuring bacterial density at the specified time intervals (2, 13, and 24 h).
Avianprotect Ten and chloramine were selected based on convergent evidence of availability and frequent use in the local poultry context. The agents were evaluated independently; their distinct mechanisms of action (antibiotic vs. oxidative antiseptic) were intentionally selected to enable a proof-of-principle RSM optimization of agent-specific dose–time conditions under wastewater treatment.
2.4. Selection of Substances with Antimicrobial Effect Using the Questionnaire
The questionnaire developed for this research aimed to collect general information from the Romanian public regarding the rearing and maintenance of domestic poultry. As a research method, the questionnaire is highly versatile and widely applied across various fields for the purpose of collecting and analyzing information on a specific topic [
45,
63]. The use of questionnaires offers clear advantages, such as low cost, ease of design and analysis, and the ability to generate results that can inform decision-making on the studied subject. Questionnaires can be employed at different stages of the study and can be rapidly adapted [
63]. However, the known and acknowledged disadvantages of using questionnaires include the potential falsity of responses, the possibility that respondents may leave questions unanswered, and the risk of misinterpretation or bias in responses [
45,
46,
47].
In this study, the questionnaire consisted of nine items. The questions addressed to respondents aimed primarily at collecting information regarding backyard poultry farming and the substances used for disease prevention and treatment. A total of 50 respondents completed the questionnaire, of which 39 individuals (private persons) responded via the Google Forms platform over a 7-day period (21–27 March 2025), and 11 respondents were authorized professionals working in veterinary pharmacies. Following the analysis of the questionnaire responses, the most frequently used antibacterial substances were identified, based on both online and in-person responses. Accordingly, for this study, the antiseptic Clorom and the antibiotic Avianprotect Ten were selected. The questionnaire was administered prior to the laboratory experiments, which commenced in April 2025. The questionnaire was conceived to inform substance selection rather than to provide nationally representative estimates; responses were triangulated with field audits of veterinary stores and veterinarian consultations to map commonly used, readily available antimicrobials in the Romanian poultry sector. Accordingly, despite the modest sample (n = 50), the aligned findings supported the choice of Avianprotect Ten and chloramine for subsequent laboratory evaluation.
2.5. Measurement of Bacterial Density
To assess the optical density of the samples (B1–B4), a Biosan DEN-1 densitometer (Riga, Latvia) (1 × 10
8 CFUs/mL) was used [
64,
65]. This device measures sample turbidity within a range of 0.0–6.0 McF units. The optical density of the B1–B4 samples exposed to the antimicrobial substances, Clorom (AS) and Avianprotect Ten (AB), at the previously specified concentrations and exposure times was measured in triplicate.
Bacterial strains that survive and regenerate following wastewater treatments in poultry farming and exposure to antimicrobial substances may pose risks to both the environment and human health. These processes are complex and depend on multiple factors, including temperature, light, nutrient content of the water, and bacterial metabolism.
To assess the regrowth potential of B1–B4 bacterial strains following exposure to antimicrobial substances, the bacterial regrowth ratio was calculated using the equation [
44]:
where:
R = bacterial regrowth ratio;
Ni = CFU/mL in untreated wastewater;
Nf = CFU/mL in treated wastewater.
2.6. Factorial Design
In this paper, the influence of two parameters for the determination of the antibacterial effect of some antimicrobial substances, one with a bacteriostatic effect (chloramine) and the other with a bactericidal effect (AB), on some bacteria isolated from poultry slaughterhouse wastewater, was investigated.
The two parameters were: chloramine or antibiotic concentration (
X1) and the action time between chloramine or antibiotic and B1–B4 bacteria strains (
X2). The response functions investigated were optical densities of B1–B4 bacteria, which were tested with Clorom (
Y1,
Y2,
Y3, and
Y4), and optical densities of B1–B4 bacteria, which were tested with Avianprotect Ten (
Y5,
Y6,
Y7, and
Y8). The variation ranges are summarized in
Table 2.
This section presents the elaboration of the mathematical model, focusing on the specific form of the response function for a 3
2 factorial design (which corresponds to nine experiments), where the model includes the main effects of factors, their quadratic terms, and interaction effects, expressed as:
where
Y is the response,
x1 and
x2 are the coded levels of the factors,
a0 is the intercept,
a1 and
a2 are the linear coefficients,
a11 and
a22 are the quadratic coefficients, and
a12 represents the interaction between the two factors.
In order to determine the significance of the polynomial coefficients of the eight response functions, the t-Student test was used. The average values of the response function and the average values of the measurement errors were calculated according to the algorithm presented in the literature [
66,
67,
68,
69,
70]. The graphs were made with the Mathlab V6.1 software.
3. Results
3.1. Microbial Load of Wastewater Samples from the Poultry Sector Subjected to Additional Treatment
The microbial load of the wastewater samples (CFU/mL), from which the B1–B4 bacterial strains were isolated, is presented in
Table 3.
As shown in
Table 3, sample No. 4, corresponding to the pre-treated wastewater, contains a markedly elevated microbial load, whereas sample No. 1, treated through the UF–UV–RO configuration, displays a substantially reduced microbial load. Thus, a reduction of approximately 13 times was obtained in the case of sample No. 1, compared to sample No. 4, which was pre-treated.
3.2. Questionnaire-Based Identification of Antimicrobial Products Used in Poultry Farming
The questionnaire developed in this research as a tool for investigating respondents’ behavior regarding the use of antimicrobial substances in poultry farming reflected both their knowledge about poultry rearing and care, as well as the antimicrobial products employed for disease prevention and treatment.
The questionnaire was administered online via the Google Forms platform [
62] and physically in veterinary pharmacies in Bacău County, with a total of 50 respondents participating. The survey is not statistically representative and was mainly used to highlight how the antimicrobial substances used in the study were selected.
Analysis of the responses revealed that antibiotics are most frequently used both for disease treatment and prevention during chick rearing. Among the products available on the market at the time of the survey, the most commonly used were as follows: Avianprotect Ten 22% (Romvac Company S.A., Ilfov, Romania), Baytril 18% (Bayer, Berlin, Germany), Levomycetin 13% (Gonane Pharma, Ahmedabad, India), Dithrim 13% (ZAO-NitaFarrn, Saratov, Russia), Enroflon 10% (VIC Group, Moscow, Russia), Doreen 6% (Janssen Pharmaceutica, Beerse, Belgium), Amoxicillin 9% (S.C. Antibiotice S.A., Iasi, Romania), and Doxycycline 9% (S.C. Antibiotice S.A., Iasi, Romania). These findings indicate that the Romanian market offers a wide range of international products from which respondents make their choices.
Furthermore, the evaluation confirmed that the use of antibiotics in poultry farms generally occurs under veterinary prescription (32 respondents). However, there are cases where these products are purchased directly from specialized stores by small-scale farmers, primarily for preventive purposes (18 respondents). The selection of Avianprotect Ten used in this research was supported not only by the online questionnaire, but also by on-site evaluation conducted in 2025 of products marketed in specialized stores in Bacău city and through direct consultations with veterinarians (
Figure 2). It was found that among the best-selling products, particularly to households (small farms), Avianprotect Ten was predominant.
The choice of chloramine as the hygiene product used in the experiments was based on the questionnaire results, as well as the scientific literature confirming the effectiveness of such products in inhibiting bacterial growth and spread, and on previously conducted studies [
25,
26,
27,
28,
29,
30]. For cleaning spaces and equipment, various commercial chlorine-based products were used. Chloramine was identified by 45 respondents as the preferred option due to its accessibility, cost-effectiveness, and ease of handling, as the tablets dissolve in water and quantities can be easily adjusted by the user [
45,
46,
47,
62].
3.3. Cultural Characteristics of B1–B4 Bacterial Strains
The bacterial strains isolated from additionally treated wastewater samples exhibited the cultural characteristics on solid PCA medium, as presented in
Table 4. The morphological and biochemical aspects of these isolates were further analyzed to highlight their diversity and potential functional roles in wastewater environments.
Catalase activity was confirmed through visual observation, as illustrated in
Figure 3, where B2 and B3 strains exhibited positive reactions (a), while B1 and B4 strains were catalase-negative (b). The presence or absence of the catalase enzyme reflects how well bacteria resist oxidative stress. Also, the presence of pigments in the bacterial culture gives the bacteria an advantage in survival, as they act like antioxidants and can protect the bacterial cell from oxidative stress. In our case, the catalase-positive strain B2 may be more resistant than B3 due to the higher degree of pigmentation [
63,
64].
The regrowth of B1–B4 bacterial strains occurs in a differentiated manner, depending on the physicochemical parameters of the wastewater, ambient temperature, and the mode of action of the antimicrobial substances (Clorom/Avianprotect Ten) added after the wastewater treatment process, as well as on the bacterial metabolism.
The bacterial regrowth ratio (R) of each strain (B1–B4) (
Table 5) may impact the environment and human health in different ways and intensities. The regrowth ratio parameter expressed by the N
i/N
f ratio reflects the evolution of the bacterial population in the given context: if
R > 1, the population is in an upward growth rate and a very good recovery rate, if
R = 1, the population is in a plateau of stability and in the situation
R < 1 the bacterial population is in decline.
The use of antibacterial substances (AB) or antiseptic substances (AS) has proven effects on B1–B4 strains, which can be extrapolated into the efficiency of treatments applied to additional poultry wastewater.
A particularly serious risk, both in the short and long term, is represented by the bacterial regrowth ratio that has survived the action of antibacterial treatments, as these strains acquire antibiotic resistance [
52]. Such antibiotic-resistant strains can be transmitted to humans or pets through direct consumption of contaminated products. The health risk can also be caused by the technological circuit, which entails increased costs and attention for neutralization during food production processes. Additionally, there is an environmental risk associated with the dissemination of these bacteria through residues, wastewater, or manure.
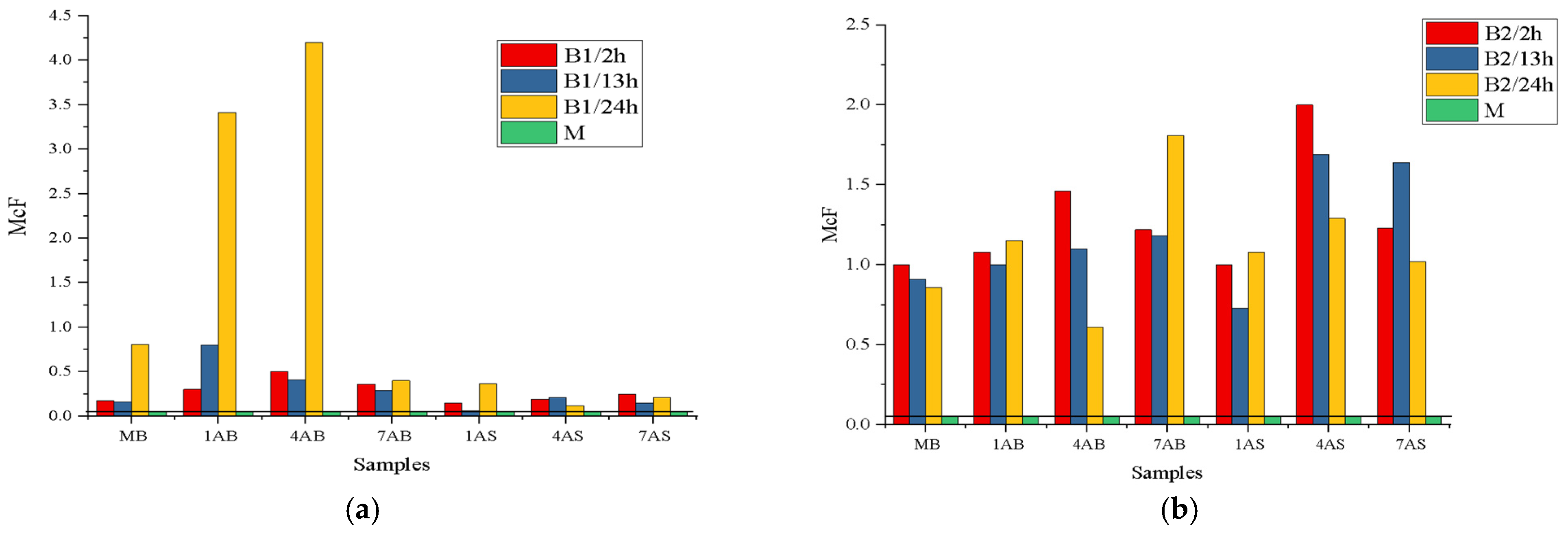
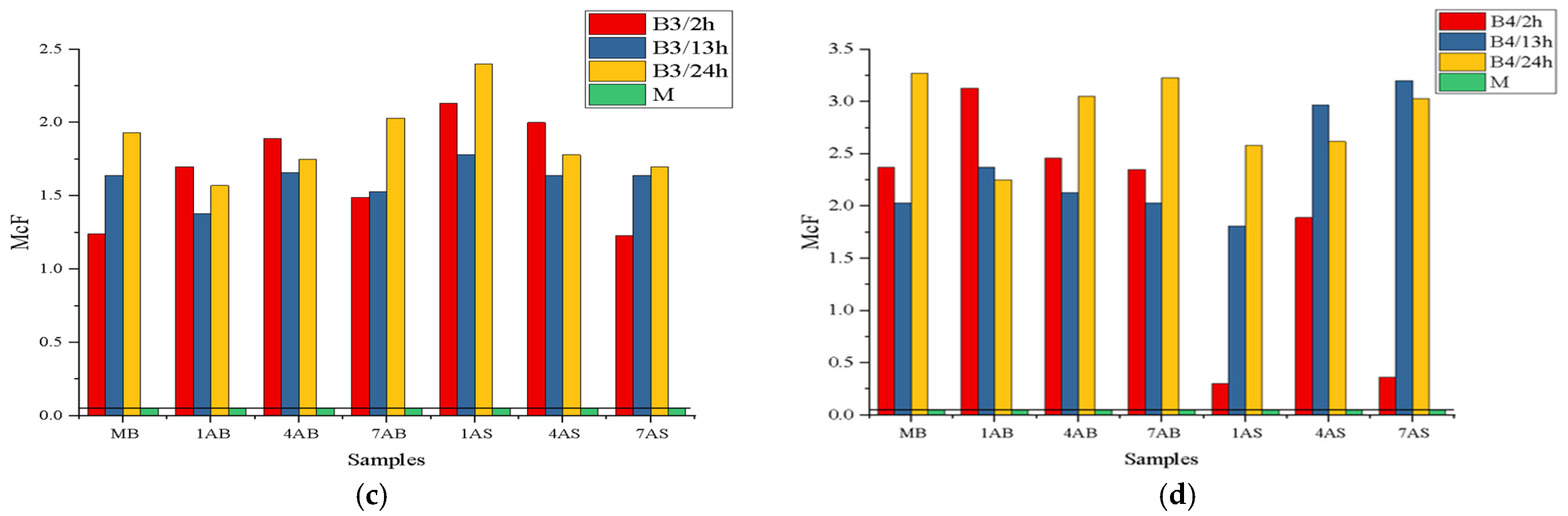
The quantitative consistency of B1–B4 bacterial strains after exposure to AB or AS substances is presented in
Figure 4.
3.4. Mathematical Modeling
This research evaluated eight response functions, represented by the optical densities of B1–B4 bacterial strains isolated in pure culture, developed following treatments applied to poultry slaughterhouse wastewater (
Table 3).
Table 6 and
Table 7 present the reduced variable values (in parentheses) corresponding to the two parameters considered in the 3
2 factorial design. Furthermore, three replicates at the central point (0, 0) were conducted to assess the reliability of the design, and the corresponding data are provided in these tables.
The polynomial coefficients were calculated according to the literature [
69,
70,
71]. The mathematical models that describe the response functions of the optimizing criterion are:
The t-Student test results for the eight response functions are presented in
Table 8.
For the response functions obtained, the first-order partial derivatives with respect to variables x1 and x2 were calculated. By equating these derivatives to zero, the resulting linear systems were solved to identify the optimal points. For the first response function, the optimal point in dimensionless coordinates was (0.076; −0.197), which lies within the initially assumed domain limits (−1, 1). When converted to real values, these coordinates correspond to a chloramine concentration of 3.19 mg·L−1 and an interaction time of approximately 10.83 h, representing the optimal conditions for the optical density of AS-B1.
Similarly, for AS-B2, the optimal point was (0.167; −0.875), which translates into a chloramine concentration of 4.5 mg·L−1 and an interaction time of 3.38 h. For AS-B3, the optimal point (0.076; 0.294) corresponds to a chloramine concentration of 4.23 mg·L−1 and an interaction time of 16.23 h, while for AS-B4, the point (0.281; 0.556) indicates optimal conditions of 4.84 mg·L−1 chloramine and 19.12 h of interaction. Regarding the antibiotic-based response functions, the optimal point for AB-B1 was (−0.125; −0.489), resulting in an antibiotic concentration of 4.84 mg·L−1 and an interaction time of 7.6 h. For AB-B2, the point (−0.51; 0.295) corresponds to 2.47 mg·L−1 and 16.25 h, while AB-B3, with an optimal point of (0.147; −0.146), translates into 4.44 mg·L−1 and 11.39 h. Finally, for AB-B4, the point (−0.786; 0.123) indicates an antibiotic concentration of 1.64 mg·L−1 and an interaction time of 14.35 h. The optimized values obtained from the experimental design are in agreement with the values of parameters initially explored.
In this research, the optical densities of B1-B4 bacteria, which were tested with antimicrobial substances, were established according to two factors: chloramine or antibiotic concentrations and the action times between chloramine or antibiotic and the four bacterial strains—a dependence that can be illustrated using the curves presented in
Figure 5 and
Figure 6.
The graphs presented in
Figure 5 and
Figure 6 reflect the combined influence of the concentration of antimicrobial substances and the duration of their action on bacterial strains isolated in pure culture (B1–B4).
4. Discussion
The cultural and microscopic characterization of the bacterial strains isolated from additionally treated wastewater samples revealed significant morphological and biochemical diversity. Strain B1 exhibited poor colony development and translucent appearance, correlating with its coccoid morphology and catalase-negative reaction, suggesting limited oxidative metabolism. In contrast, strain B2 formed dense, well-developed colonies with a distinctive yellow-orange pigmentation and reticulated margins, features typically associated with high adaptability and enzymatic activity, confirmed by its catalase-positive reaction. Strain B3 displayed staphylococcal clustering and an unpleasant odor, indicating potential production of volatile compounds, while strain B4 showed coccobacillary morphology and opalescent colonies, characteristics often linked to transitional forms between cocci and rods. These findings are consistent with previous studies reporting morphological heterogeneity and enzymatic variability among wastewater isolates. Furthermore, the presence of catalase-positive strains suggests oxidative stress resistance, a trait frequently observed in bacteria adapted to wastewater environments [
44,
72,
73]. Such diversity underlines the importance of integrating cultural, microscopic, and biochemical tests with molecular approaches for accurate identification and risk assessment in wastewater treatment systems.
The regression analysis of the eight response functions provided valuable insights into the influence of chloramine or antibiotic concentrations, as well as contact time, on bacterial optical density. To validate the regression models, the t-Student test was applied to the estimated coefficients using the significance level α = 0.05. The intercept (a0) represents the predicted response at the center of the experimental domain, serving as a reference rather than the maximum observed value. Linear coefficients (a1, a2) quantify the main effects of chloramine or antibiotic concentration (x1) and contact time (x2), with positive values indicating a favorable contribution and negative values suggesting inhibition.
Interaction terms (a12), when statistically significant, describe the combined effect of both variables, where positive signs denote synergistic interactions, while negative signs indicate antagonism. In the case of the response function Y2, the interaction coefficient X1X2 was considered insignificant, following the t-Student test. Thus, the interaction term a12 was, therefore, removed in order to obtain a simplified and statistically robust final model.
Quadratic coefficients (a11, a22) determine the curvature of the response surface. Identical signs indicate the presence of an extremum (maximum or minimum), whereas opposite signs reveal an inflection point, resulting in a saddle-shaped surface. The magnitude of these coefficients reflects the degree of curvature along each axis, with higher absolute values corresponding to stronger curvature effects.
Overall, chloramine-based models (AS-B1 to AS-B4) generally exhibited positive main effects and, in most cases, favorable interactions, leading to response surfaces characterized by maxima or minima depending on the quadratic term signs. Antibiotic-based models showed greater variability, including saddle-shaped surfaces for AB-B1 and AB-B3 due to opposite quadratic signs, highlighting the complexity of factor interactions in these systems. These findings align with previous studies employing RSM for microbial process optimization, where curvature and interaction effects were essential for identifying true optima rather than relying on experimental maxima. Furthermore, the variability observed in antibiotic-based models, including saddle-shaped surfaces for AB-B1 and AB-B3, reflects the complexity of antimicrobial interactions, which is consistent with reports on non-linear dose–response relationships in microbial systems [
55,
56,
57,
58,
59,
60,
61].
The practical implications are significant; chloramine-based treatments generally exhibited favorable main effects and synergistic interactions, supporting their role in sanitation protocols. Conversely, antibiotic-based treatments demonstrated greater variability, reinforcing the need for precise optimization to avoid overuse and mitigate antimicrobial resistance, a concern highlighted globally by WHO and recent reviews [
8,
15]. Improper antibiotic use in poultry farming accelerates resistance development, posing risks to public health and the environment [
13,
14,
15,
18,
22,
23,
27,
44].
Avianprotect Ten exhibits systemic antibacterial action in poultry and, although not designed for effluent disinfection, upstream use can influence downstream wastewater microbiota via selection pressures; testing its optimized dose–time effects on isolates B1–B4, therefore, provides process-level insight relevant to the studied system. In contrast, Clorom is an inexpensive, widely available disinfectant with simple dosing and low residual toxicity in treated wastewater within the tested windows. In our UF–UV–RO context, the antimicrobial benefits outweigh the limited formation of disinfection byproducts.
Assessed independently, these two agents illustrate that factorial design and RSM can deliver precise, agent-specific dosing recommendations for chemically distinct antimicrobial substances. Finally, optical density as a proxy for bacterial concentration must be interpreted cautiously due to instrument variability and calibration requirements, as emphasized in scientific literature [
66,
67]. Integrating robust calibration protocols with factorial modeling enhances reliability and reproducibility in microbial optimization studies.
The treatments applied to wastewater resulting from poultry farming led to a reduction in the microbiota present in the water by approximately 10 times in the case of UF–UV–RO treatments.
The addition of antibiotics/antiseptics for all bacteria that are resistant to wastewater treatments makes them much more vulnerable. These aspects were evaluated by the metabolic rate of development, as well as by the morphological, cultural, microscopic, and biochemical aspects.
The use of antibiotic therapy, both for curative and preventive purposes in poultry farming, must be applied judiciously, given the immediate consequences and environmental impacts. The primary risks associated with improper use of antibacterial substances, particularly antibiotics, lies in the development and transfer of antibiotic resistance among bacteria, as well as environmental contamination through residual water and contaminated waste. Antibiotic-resistant bacteria and their dissemination in the environment represent a threat not only to the healthcare system but also constitute priority topics in current research.
The rate of bacterial growth reflects the efficiency of the treatment applied to the wastewater resulting from poultry farming. The bacterial strains found in the water treated by the techniques applied in this research have an unpredictable behavior towards the tested substances. In this situation, the impact of antiseptic substances (with
R > 1) reflects the rapid recovery of the bacterial population compared to the impact of antibacterial substances (with
R < 1).
Table 6 shows the behavior of bacterial strains B1-B4 upon contact with the studied antimicrobial substances.
From the perspective of the growth rate, in the case of antibiotics, the most effective treatment applied to water was ultrafiltration, UV disinfection, and reverse osmosis, and for antiseptics, the most effective treatment was reverse osmosis.
Bacterial behavior is directly influenced by the additional treatment applied to wastewater [
25,
28], as well as by the quantities and duration of action of antimicrobial substances [
44].
From an industrial standpoint, the implementation of optimal parameters and mathematical modeling is essential to avoid the overuse of antiseptic products during the sanitation of spaces, equipment, and processing lines. This approach not only ensures effective hygiene but also minimizes chemical residues and environmental impact. In intensive poultry farming, mathematical modeling helps to prevent mass outbreaks of disease. RSM facilitates the establishment of conditions that do not compromise human health and do not generate environmental risks.
A comprehensive molecular-level analysis of bacteria that exhibit resistance to conventional treatments is necessary to understand their survival mechanisms and to develop targeted interventions. The creation of a dedicated database containing molecular and phenotypic profiles of resistant strains would serve as a valuable resource. This database should be used in conjunction with advanced spectroscopic techniques, enabling rapid identification and characterization of resistant microorganisms. Such integration would enhance diagnostic accuracy and support the development of more effective antimicrobial strategies [
44,
73,
74,
75].
The effectiveness of UV disinfection, RO, and UF treatments in reducing the quantity of bacteria has been proven, but their association with the use of substances with antimicrobial effects is much more effective. It was found that there is a synergistic effect between the various parameters, time, and concentration, both in the case of the use of the antibiotic and the antiseptic. Good efficiency in the case of B1 strain was obtained at 3.19 mg·L−1 Clorom for 10.83 h and 4.84 mg·L−1 Avianprotect Ten for 7.6 h. In the case of strain B2, it was 4.5 mg·L−1 Clorom for 3.38 h and 2.47 mg·L−1 Avianprotect Ten for 16.25 h. In the case of strain B3, it was 4.23 mg·L−1 Clorom for 16.23 h and 4.44 mg·L−1 Avianprotect Ten for 11.39 h. In the case of strain B4, it was 4.84 mg·L−1 Clorom for 19.12 h and 1.64 mg·L−1 Avianprotect Ten for 14.35 h.
The mathematical model developed in this research can constitute a reference for the use of other substances with antimicrobial effect, respectively, and the adaptation of quantities and time of action depending on the volume of resulting wastewater.
Limitations
This work has several limitations. First, B1–B4 strain characterization relied on cultural and biochemical traits; the lack of molecular identification constrains taxonomic resolution and resistance contextualization. Second, all assays were performed under laboratory conditions with real effluent but not at pilot or plant scale; therefore, matrix variability and operational disturbances typical of full-scale systems were not captured. Third, only two antimicrobial agents (AS, AB) were assessed as locally relevant exemplars rather than an exhaustive screen, which limits generalizability across broader agent classes. Fourth, optical density (McF) was used as a proxy for biomass. Although measurements were performed in triplicate and calibrated, estimates of bacterial cell density may still be influenced by strain-specific light scattering and instrument variability. These limitations will be addressed in a follow-up pilot-farm study by integrating molecular identification and resistome profiling, validating the models under pilot UF–UV–RO operation, and expanding the antibacterial agent panel.