Review on Treatment Pathways and Adsorptive Approaches for Dye-Contaminated Wastewater
Abstract
1. Introduction
2. Dyes Categories
2.1. Natural Dyes
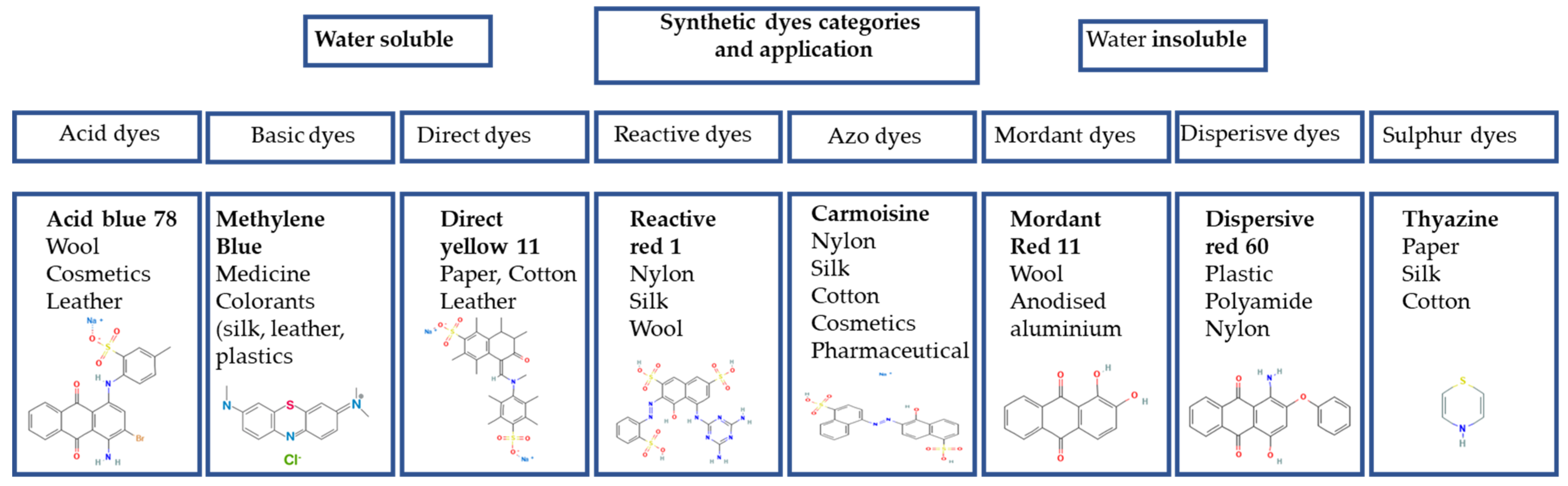
2.2. Synthetic Dyes
3. Methods Explored to Treat Dye Wastewater
3.1. Biological Treatments for Dye Removal from Water Streams
3.2. Chemical Treatments for Dye Removal from Water Streams
3.3. Physical and Physicochemical Treatments for Dyes Removal from Water
3.3.1. Adsorption as a Way to Remove Dyes from Aqueous Phase
Nanoparticles Used to Treat Dye Wastewater
Clays as Dyes Adsorbents
Zeolites and Silicas as Dye Adsorbents
Biopolymers as Dye Adsorbents
Biomaterials as Dye Adsorbents
Biochar as Dye Adsorbents
| Precursor Production Conditions | Dyes | Adsorbent Dosage (g/L) | Initial Concentration (mg/L) | pH | Contact Time (min.) | Temperature (°C) | Amount of Dye Adsorbed (mg g−1) or (%) * | Reference |
|---|---|---|---|---|---|---|---|---|
| Acacia leucophloea, under N2; (250–400 °C); (t = 120 min.) | Reactive red 2 | 0.05 | 100 | 2 | -- | 27 | 98% | [73] |
| Acacia leucophloea N2; (250–400 °C); (t = 120 min.) | Reactive red 29 | 0.05 | 100 | 10 | -- | 27 | 98% | [73] |
| Sunflower seed shells Peanut shells N2; 300, 600 900 °C; 3 h | Remazol Brilliant Blue | 0.5 0.5 | -- | 6.5 6.5 | 5 30 | 25 | 89 100 | [74] |
| Sunflower seed shells Peanut shells N2; 300, 600 900 °C; 3 h | Congo Red | 0.5 0.5 | -- | 6.5 6.5 | 5 30 | 25 | 26 60 | [74] |
| Sheep manure | MO | 0.6 | 20 | 4 | 250 | 25 | 92.55% | [76] |
| Sludge/rice husk 1:1 500 °C; T = 120 min. | Direct Red 4BS Acid Orange II React Blue 19 MB | 0.1 | 300 | -- | 1440 | 25 | 59.77 38.36 42.12 22.59 | [77] |
| Wood Pinus caribaea HTC; 240 °C, 24 h, acidic or basic | MB | 1.0 | 300 | 11 | 360 | 25 | 149.0 (4.7% | [78] |
| Poultry manure (PM); 650 °C, 3 h PC + FeSO4·7H2O; 180 °C; 6 h | MO | 0.7 | 200 | acidic | 1440 | 25 | 98.23 136.25 | [79] |
Activated Carbon as Dye Adsorbent
| Precursor Production Conditions | Dyes | Adsorbent Dosage (g/L) | Initial Concentration (mg/L) | pH | Contact Time (min.) | Temperature (°C) | Amount of Dye Adsorbed (mg g−1) or (%) * | Reference |
|---|---|---|---|---|---|---|---|---|
| Coconut leaves (microwave-induced; NaOH activation); (600 W, 20 min.) | MB | 1.5 | 100 | 8 | 90 | 50 | 87.72 | [98] |
| Ficus carica bast (1 g of biomass, mixed with 800 mL of H2SO4 for 6 h; under reflux for 12 h, at 150 °C) | MB | 0.5 | 50 mg/100 mL | 8 | 90 | 50 | 47.62 | [101] |
| Pea shells ZnCl2; 500 °C; 1 h | MB | 0.1 | 350 | 6.5 | 180 | 25 | 246.9 | [102] |
| Quercus Branti AC (heated at 450–750 °C, t = 30–120 min.) | MB | 2 | 100 | 6 | 180 | 50 | 55 to 60 91.08% | [103] |
| Phosphorus-doped microporous carbon from olive mill wastewater | MB | 0.3–1 | 914 | 8 | 25 | 1010 | [104] | |
| Almond seed shells (H3PO4—ratio of 1:2, soaked 24 h; 500 °C; 2 h) | MB | 1 | 200 | 10 | 120 | 55 | 130.4 | [105] |
| Bamboa; KOH; (1:1); 700 °C | MB | 1 | 250–400 | 7 | 1440 | 25 | 156.9 | [106] |
| Teak; KOH; ratio of 1:1; 600 °C | MB | 1 | 250–400 | 7 | 1440 | 25 | 82.7 | [106] |
| Teak; KOH; ratio of 1:1; 700 °C | MB | 1 | 250–400 | 7 | 1440 | 25 | 159.7 | [106] |
| Teak; K2CO3; ratio of 1:1; 700 °C | MB | 1 | 250–400 | 7 | 1440 | 25 | 248.4 | [106] |
| Waste coffee grounds + FeCl3 700–900 °C; 8 min. | MB | -- | 653.6 | [95] | ||||
| Argan shell waste H3PO4; 900 °C; 2 h | MB | 0.5 | 100 | 10 | 180 | 25 | 70 | [97] |
| Palm tree fiber (H3PO4); 400 °C; 30 min | Rhodamine B (RB) | 1 | 5–50 | 2–12 | 120 | -- | 26.5 99.86% | [100] |
| Coconut Shells biochar 2 N NaOH; 600 °C, 2 h | R6G | 1 | 5–1200 | 7 | 1440 | 40 | 478 | [98] |
| Waste coffee grounds + FeCl3 700–900 °C; 8 min. | MO Rodamine B | 1 | 1440 | -- | 465.8 366.1 | [95] | ||
| Oak cupules; H3PO4 (3:1, acid_OC); 450 °C, 1 h | Crystal violet | 0.01–0.04 | -- | 10 | 90 | 40 | 658 | [99] |
| Argan shell waste H3PO4; 900 °C; 2 h | MO | 0.5 | 100 | 2 | 240 | 25 | 31 | [97] |
| Coconut Shells biochar 2 M H2SO4; 600 °C; 2 h | Anionic dye IC | 1 | 5–1200 | 7 | 1440 | 40 | 306 | [98] |
| Palm tree fiber (H3PO4); 400 °C; 30 min | Congo red (RG) | 1 | 5–50 | 2–12 | 120 | -- | 10.4 98.24% | [100] |
| Oak cupules (OC); H3PO4 (3:1, acid_OC); 450 °C, 1 h | naphthol blue black (NBB) | 0.02–0.08 | -- | 2 | 180 | 40 | 208 | [99] |
| Almond seed shells; H3PO4 (1:2); soaked 24 h; 500 °C; 2 h | MO | 1 | 200 | 150 | -- | 118.6 | [105] | |
| Jamoya fruit seeds HTC 250 °C, 4 h; CO2—400 mL/min.; 850 °C; 2 h | Carmoisine B | 1 | 5 | -- | 45 | 269 | [72] | |
| Activated carbon cloth | IC; RC; Evans Blue | Continuous flux | 7 | -- | 25 | 506; 187; 27 | [107] |
Natural Biomass Used Directly as Dye Adsorbents
3.4. Carbon Material Modifications Process
| Precursor Production Conditions | Dye | Adsorbent Dosage (g/L) | Initial Concentration (mg/L) | pH | Contact Time (min.) | Temperature (°C) | Amount of Dye Adsorbed (mg g−1) | Reference |
|---|---|---|---|---|---|---|---|---|
| Barley (Hordeum vulgare) bran Enset (Ensete ventricosum midrib leaf) | MB | 2.5 | 10 | 5.7 | 1440 | -- | 63.2 35.5 | [117] |
| Citrus limetta peel waste | MB | 2 | 4 | 180 | 25 | 227.3 | [118] | |
| Subble Tectona Grandis Adansonia digitata L. Bamboo flowers | MB | 1 | 400 | 6 | 1440 | 25 | 63.7 27.9 156.8 42.8 | [106] |
| Laminaria digitata; Horse chestnut husk Hazelnut husk Rapeseed residue | MB | 2 | 200 | 5 | 1440 | 30 | 500 137 120 85 | [119] |
| Spent mushroom waste | Direct Red 5B Direct Black 22 Direct Black 71 Reactive Black 5 | -- | 200 | 2 | 240 | -- | 18 15.46 20.19 14.62 | [120] |
| Date stones | Congo Red | 100–800 | 4 | 90 | 50 | 45.08 | [121] | |
| Jujube shells | Congo Red | 100–800 | 4 | 90 | 50 | 59.55 | [121] | |
| Yeast stain of Wickerhamomyces anomalus | Acid Red 1 | 1.25 | 3–4 | 25 | 71.37 | [122] |
Adsorbents Regeneration Processes
4. Conclusions
5. Critical Perspective/Future Challenges
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kang, L.L.; Zeng, Y.N.; Wang, Y.T.; Li, J.G.; Wang, F.P.; Wang, Y.J.; Yu, Q.; Wang, X.-M.; Ji, R.; Gao, D.; et al. Removal of pollutants from wastewater using coffee waste as adsorbent: A review. J. Water Process Eng. 2022, 49, 103178. [Google Scholar] [CrossRef]
- Cansado IPda, P.; Mourão, P.A.M.; Castanheiro, J.E.; Geraldo, P.F.; Suhas Suero, S.R.; Cano, B.L. A Review of the Biomass Valorization Hierarchy. Sustainability 2025, 17, 335. [Google Scholar] [CrossRef]
- Wang, X.; Zhuang, Y.; Shah, K.J.; Sun, Y. Application Progress of Magnetic Chitosan in Heavy Metal Wastewater Treatment. Magnetochemistry 2025, 11, 71. [Google Scholar] [CrossRef]
- Kausar, A.; Iqbal, M.; Javed, A.; Aftab, K.; Nazli, Z.; Bhatti, H.N.; Nouren, S. Dyes adsorption using clay and modified clay: A review. J. Mol. Liq. 2018, 256, 395–407. [Google Scholar] [CrossRef]
- Peng, H.; Guo, J. Removal of chromium from wastewater by membrane filtration, chemical precipitation, ion exchange, adsorption electrocoagulation, electrochemical reduction, electrodialysis, electrodeionization, photocatalysis and nanotechnology: A review. Environ. Chem. Lett. 2020, 18, 2055–2068. [Google Scholar] [CrossRef]
- Zamora-Ledezma, C.; Negrete-Bolagay, D.; Figueroa, F.; Zamora-Ledezma, E.; Ni, M.; Alexis, F.; Guerrero, V.H. Heavy metal water pollution: A fresh look about hazards, novel and conventional remediation methods. Environ. Technol. Innov. 2021, 22, 101504. [Google Scholar] [CrossRef]
- Silva-Gálvez, A.L.; López-Sánchez, A.; Camargo-Valero, M.A.; Prosenc, F.; González-López, M.E.; Gradilla-Hernández, M.S. Strategies for livestock wastewater treatment and optimised nutrient recovery using microalgal-based technologies. J. Environ. Manag. 2024, 354, 120258. [Google Scholar] [CrossRef] [PubMed]
- Revellame, E.; Fortella, D.L.; Sharp, W.; Hernandez, R.; Zappi, M.E. Adsorption kinetic modeling using pseudo-first order and pseudo-second order rate laws. Clean. Eng. Technol. 2020, 1, 100032. [Google Scholar] [CrossRef]
- Ben Slama, H.; Bouket, A.C.; Pourhassan, Z.; Alenezi, F.N.; Silini, A.; Cherif-Silini, H.; Oszako, T.; Luptakova, L.; Golińska, P.; Belbahri, L. Diversity of synthetic dyes from textile industries, discharge impacts and treatment methods. Appl. Sci. 2021, 11, 6255. [Google Scholar] [CrossRef]
- The Ellen MacArthur Foundation_Designing Waste and Pollution out of Fashion—The Earthshot Prize. 2021. Available online: https://earthshotprize.org/news/the-ellen-macarthur-foundation-designing-waste-and-pollution-out-of-fashion/ (accessed on 3 December 2025).
- Jorge, A.M.S.; Athira, K.K.; Alves, M.B.; Gardas, R.L.; Pereira, J.F.B. Textile dyes effluents: A current scenario and the use of aqueous biphasic systems for the recovery of dyes. J. Water Process Eng. 2023, 55, 104125. [Google Scholar] [CrossRef]
- Al-Tohamy, R.; Ali, S.S.; Li, F.; Okasha, K.M.; Mahmoud, Y.A.G.; Elsamahy, T.; Jiao, H.; Fu, Y.; Sun, J. A critical review on the treatment of dye-containing wastewater: Ecotoxicological and health concerns of textile dyes and possible remediation approaches for environmental safety. Ecotoxicol. Environ. Saf. 2022, 231, 113160. [Google Scholar] [CrossRef] [PubMed]
- Holmes, H.E.; Kim, J.; Realff, M.J. Process systems engineering: A key enabler of adsorption-based direct air capture. Curr. Opin. Chem. Eng. 2026, 51, 101202. [Google Scholar] [CrossRef]
- Ganjoo, R.; Verma, C.; Kumar, A.; Quraishi, M.A. Colloidal and interface aqueous chemistry of dyes: Past, present and future scenarios in corrosion mitigation. Adv. Colloid Interface Sci. 2023, 311, 102832. [Google Scholar] [CrossRef] [PubMed]
- Alegbe, E.O.; Uthman, T.O. A review of history, properties, classification, applications and challenges of natural and synthetic dyes. Heliyon 2024, 10, e33646. [Google Scholar] [CrossRef] [PubMed]
- de Farias, N.O.; Pires, M.S.G.; Moreira, B.d.J.; dos Santos, A.; Freeman, H.S.; Toukola, P.; de Albuquerque, A.F.; Räisänen, R.; Umbuzeiro, G.d.A. Natural indigo toxicity for aquatic and terrestrial organisms. Ecotoxicol. Environ. Saf. 2025, 290, 117606. [Google Scholar] [CrossRef] [PubMed]
- Pizzicato, B.; Pacifico, S.; Cayuela, D.; Mijas, G.; Riba-Moliner, M. Advancements in Sustainable Natural Dyes for Textile Applications: A Review. Molecules 2023, 28, 5954. [Google Scholar] [CrossRef] [PubMed]
- Affat, S.S. Classifications, Advantages, Disadvantages, Toxicity Effects of Natural and Synthetic Dyes: A Review Report. Univ. Thi-Qar J. Sci. (UTsci) 2021, 8, 130. [Google Scholar]
- Eren, E. Investigation of a basic dye removal from aqueous solution onto chemically modified Unye bentonite. J. Hazard. Mater. 2009, 166, 88–93. [Google Scholar] [CrossRef] [PubMed]
- El Qada, E.N.; Allen, S.J.; Walker, G.M. Adsorption of basic dyes from aqueous solution onto activated carbons. Chem. Eng. J. 2008, 135, 174–184. [Google Scholar] [CrossRef]
- Notten, P. Sustainability and Circularity in the Textile Value Chain Global Stocktaking; Report; United Nations Environment Programme: Nairobi, Kenya, 2020. [Google Scholar]
- Piaskowski, K.; Świderska-Dąbrowska, R.; Zarzycki, P.K. Dye removal from water and wastewater using various physical, chemical, and biological processes. J. AOAC Int. 2018, 101, 1371–1384. [Google Scholar] [CrossRef] [PubMed]
- Sahoo, A.K.; Dahiya, A.; Patel, B.K. Biological methods for textile dyes removal from wastewaters. In Development in Wastewater Treatment Research and Processes: Innovative Microbe-Based Applications for Removal of Chemicals and Metals in Wastewater Treatment Plants; Elsevier: Amsterdam, The Netherlands, 2021; pp. 127–151. [Google Scholar] [CrossRef]
- Jamee, R.; Siddique, R. Biodegradation of synthetic dyes of textile effluent by microorganisms: An environmentally and economically sustainable approach. Eur. J. Microbiol. Immunol. 2019, 9, 114–118. [Google Scholar] [CrossRef]
- Mocek-Płóciniak, A.; Mencel, J.; Zakrzewski, W.; Roszkowski, S. Phytoremediation as an Effective Remedy for Removing Trace Elements from Ecosystems. Plants 2023, 12, 1653. [Google Scholar] [CrossRef]
- Khan, R.; Bhawana, P.; Fulekar, M.H. Microbial decolorization and degradation of synthetic dyes: A review. Rev. Environ. Sci. Biotechnol. 2013, 12, 75–97. [Google Scholar] [CrossRef]
- Danouche, M.; ELArroussi, H.; El Ghachtouli, N. Mycoremediation of synthetic dyes by yeast cells: A sustainable biodegradation approach. Environ. Sustain. 2021, 4, 5–22. [Google Scholar] [CrossRef]
- Tabish, M.; Tabinda, A.B.; Mazhar, Z.; Yasar, A.; Ansar, J.; Wasif, I. Physical, chemical and biological treatment of textile wastewater for removal of dyes and heavy metals. Desalination Water Treat. 2024, 320, 100842. [Google Scholar] [CrossRef]
- Muda, K.; Aris, A.; Razman, M.; Ibrahim, Z. Sequential Anaerobic-Aerobic Phase Strategy Using Microbial Granular Sludge for Textile Wastewater Treatment. In Biomass Now—Sustainable Growth and Use; InTech: Incheon, Republic of Korea, 2013. [Google Scholar] [CrossRef]
- Kurade, M.B.; Waghmode, T.R.; Khandare, R.V.; Jeon, B.H.; Govindwar, S.P. Biodegradation and detoxification of textile dye Disperse Red 54 by Brevibacillus laterosporus and determination of its metabolic fate. J. Biosci. Bioeng. 2016, 121, 442–449. [Google Scholar] [CrossRef] [PubMed]
- Bożęcka, A.; Orlof-Naturalna, M.; Kopeć, M. Methods of Dyes Removal from Aqueous Environment. J. Ecol. Eng. 2021, 22, 111–118. [Google Scholar] [CrossRef]
- Dutta, S.; Gupta, B.; Srivastava, S.K.; Gupta, A.K. Recent advances on the removal of dyes from wastewater using various adsorbents: A critical review. Mater. Adv. 2021, 2, 4497–4531. [Google Scholar] [CrossRef]
- Faroon, M.; ALSaad, Z.; Albadran, F.; Ahmed, L. Review on Technology-Based on Reverse Osmosis. Anbar J. Eng. Sci. 2023, 14, 89–97. [Google Scholar] [CrossRef]
- Onukwuli, C.J.; Obiora-Okafo, I.A. Removal of Dyes from Synthetic Waste Water by Coagulation Technique using Natural Coagulant. Equat. J. Eng. 2019, 1–5. [Google Scholar]
- Ravele, M.P.; Olatunde, O.C.; Oyewo, O.A.; Makgato, S.S.; Onwudiwe, D.C. Plant-based coagulants for wastewater treatment: Recent advances and applications. Clean Water 2025, 4, 100147. [Google Scholar] [CrossRef]
- Elgarahy, A.M.; Eloffy, M.G.; Guibal, E.; Alghamdi, H.M.; Elwakeel, K.Z. Use of biopolymers in wastewater treatment: A brief review of current trends and prospects. Chin. J. Chem. Eng. 2023, 64, 292–320. [Google Scholar] [CrossRef]
- Tran, H.N.; You, S.J.; Nguyen, T.V.; Chao, H.P. Insight into the adsorption mechanism of cationic dye onto biosorbents derived from agricultural wastes. Chem. Eng. Commun. 2017, 204, 1020–1036. [Google Scholar] [CrossRef]
- N’diaye, A.D. Modeling of adsorption isotherms of dyes onto various adsorbents A Short Review. Arab. J. Chem. Environ. Res. 2018, 5, 42–51. [Google Scholar]
- Serafin, J.; Dziejarski, B. Application of isotherms models and error functions in activated carbon CO2 sorption processes. Microporous Mesoporous Mater. 2023, 354, 112513. [Google Scholar] [CrossRef]
- Pandey, J. Biopolymers and Their Application in Wastewater Treatment (chapter 11). In Emerging Eco-Friendly Green Technologies for Wastewater Treatment; Springer Nature: Singapore, 2020. [Google Scholar] [CrossRef]
- Othman, N.H.; Alias, N.H.; Shahruddin, M.Z.; Abu Bakar, N.F.; Nik Him, N.R.; Lau, W.J. Adsorption kinetics of methylene blue dyes onto magnetic graphene oxide. J. Environ. Chem. Eng. 2018, 6, 2803–2811. [Google Scholar] [CrossRef]
- Zhao, M.; Liu, P. Adsorption of methylene blue from aqueous solutions by modified expanded graphite powder. Desalination 2009, 249, 331–336. [Google Scholar] [CrossRef]
- Albayati, T.M. Removal of anionic azo dye from wastewater using Fe3O4 magnetic nanoparticles adsorbents in a batch system. Desalination Water Treat. 2024, 317, 100033. [Google Scholar] [CrossRef]
- Attallah, O.A.; Al-Ghobashy, M.A.; Nebsen, M.; Salem, M.Y. Removal of cationic and anionic dyes from aqueous solution with magnetite/pectin and magnetite/silica/pectin hybrid nanocomposites: Kinetic, isotherm and mechanism analysis. RSC Adv. 2016, 6, 11461–11480. [Google Scholar] [CrossRef]
- Tehrani-Bagha, A.R.; Nikkar, H.; Mahmoodi, N.M.; Markazi, M.; Menger, F.M. The sorption of cationic dyes onto kaolin: Kinetic, isotherm and thermodynamic studies. Desalination 2011, 266, 274–280. [Google Scholar] [CrossRef]
- Chaari, I.; Fakhfakh, E.; Medhioub, M.; Jamoussi, F. Comparative study on adsorption of cationic and anionic dyes by smectite rich natural clays. J. Mol. Struct. 2019, 1179, 672–677. [Google Scholar] [CrossRef]
- Hajjaji, W.; Pullar, R.C.; Labrincha, J.A.; Rocha, F. Aqueous Acid Orange 7 dye removal by clay and red mud mixes. Appl. Clay Sci. 2016, 126, 197–206. [Google Scholar] [CrossRef]
- Santos, S.C.R.; Boaventura, R.A.R. Adsorption of cationic and anionic azo dyes on sepiolite clay: Equilibrium and kinetic studies in batch mode. J. Environ. Chem. Eng. 2016, 4, 1473–1483. [Google Scholar] [CrossRef]
- El-habacha, M.; Miyah, Y.; Lagdali, S.; Mahmoudy, G.; Dabagh, A.; Chiban, M.; Sinan, F.; Iaich, S.; Zerbet, M. General overview to understand the adsorption mechanism of textile dyes and heavy metals on the surface of different clay materials. Arab. J. Chem. 2023, 16, 105248. [Google Scholar] [CrossRef]
- Adeyemo, A.A.; Adeoye, I.O.; Bello, O.S. Adsorption of dyes using different types of clay: A review. Appl. Water Sci. 2017, 7, 543–568. [Google Scholar] [CrossRef]
- Bahmanzadegan, F.; Ghaemi, A. A comprehensive review on novel zeolite-based adsorbents for environmental pollutant. J. Hazard. Mater. Adv. 2025, 17, 100617. [Google Scholar] [CrossRef]
- Ahmed, K.R.; Mohammed, D.S.; Ali, F.M. SYNTHESIS OF A NEW ADSORBENT BASED ON THE MILD DEALUMINATED BINDER FREE GRANULAR Y ZEOLITE. Sci. J. Univ. Zakho 2023, 11, 333–345. [Google Scholar] [CrossRef]
- Gollakota, A.R.K.; Volli, V.; Munagapati, V.S.; Wen, J.C.; Shu, C.M. Synthesis of novel ZSM-22 zeolite from Taiwanese coal fly ash for the selective separation of Rhodamine 6G. J. Mater. Res. Technol. 2020, 9, 15381–15393. [Google Scholar] [CrossRef]
- Wang, S.; Li, H.; Xie, S.; Liu, S.; Xu, L. Physical and chemical regeneration of zeolitic adsorbents for dye removal in wastewater treatment. Chemosphere 2006, 65, 82–87. [Google Scholar] [CrossRef] [PubMed]
- Kumari, S.; Chowdhry, J.; Kumar, M.; Chandra Garg, M. Zeolites in wastewater treatment: A comprehensive review on scientometric analysis, adsorption mechanisms, and future prospects. Environ. Res. 2024, 260, 119782. [Google Scholar] [CrossRef] [PubMed]
- Saiyad, M.; Shah, N.; Joshipura, M.; Dwivedi, A.; Pillai, S. Modified biopolymers in wastewater treatment: A review. Mater. Today Proc. 2024, in press. [Google Scholar] [CrossRef]
- Elzahar, M.M.H.; Bassyouni, M. Removal of direct dyes from wastewater using chitosan and polyacrylamide blends. Sci. Rep. 2023, 13, 15750. [Google Scholar] [CrossRef] [PubMed]
- Kaczorowska, M.A.; Bożejewicz, D. The Application of Chitosan-Based Adsorbents for the Removal of Hazardous Pollutants from Aqueous Solutions—A Review. Sustainability 2024, 16, 2615. [Google Scholar] [CrossRef]
- Hevira, L.; Ighalo, J.O.; Sondari, D. Chitosan-based polysaccharides for effective synthetic dye adsorption. J. Mol. Liq. 2024, 393, 123604. [Google Scholar] [CrossRef]
- Zubair, M.; Ullah, A. Biopolymers in environmental applications: Industrial wastewater treatment (chapter 14). In Biopolymers and their Industrial Applications; Elsevier: Amsterdam, The Netherlands, 2021; pp. 331–349. [Google Scholar] [CrossRef]
- Wang, G.; Chen, Y.; Xu, G.; Pei, Y. Effective removing of methylene blue from aqueous solution by tannins immobilized on cellulose microfibers. Int. J. Biol. Macromol. 2019, 129, 198–206. [Google Scholar] [CrossRef] [PubMed]
- Raut, E.R.; Thakur, M.A.B.; Chaudhari, A.R. A review on activated carbon preparation from natural and eco-friendly raw materials. In AIP Conference Proceedings; American Institute of Physics Inc.: College Park, MD, USA, 2021. [Google Scholar] [CrossRef]
- Zielińska, M.; Bułkows, K. Agricultural Wastes and Their By-Products for the Energy Market. Energies 2024, 17, 2099. [Google Scholar] [CrossRef]
- Elhassan, M.; Abdullah, R.; Kooh, M.R.R.; Chou Chau, Y.F. Hydrothermal liquefaction: A technological review on reactor design and operating parameters. Bioresour. Technol. Rep. 2023, 21, 101314. [Google Scholar] [CrossRef]
- Alonso, M.; Ledesma, B.; Román, S.; Olivares-Marín, M. Insights about the formation of secondary char during HTC processes. Sustain. Chem. Pharm. 2024, 37, 101420. [Google Scholar] [CrossRef]
- Soffian, M.S.; Abdul Halim, F.Z.; Aziz, F.A.; Rahman, M.; Mohamed Amin, M.A.; Awang Chee, D.N. Carbon-based material derived from biomass waste for wastewater treatment. Environ. Adv. 2022, 9, 100259. [Google Scholar] [CrossRef]
- Sadok, I.; Krzyszczak-Turczyn, A.; Czech, B.; Parlakidis, P.; Vryzas, Z. Advancements in biochar-based materials for decontamination and analytical detection of pesticides and mycotoxins in food. Food Chem. 2025, 492, 145467. [Google Scholar] [CrossRef] [PubMed]
- Enaime, G.; Baçaoui, A.; Yaacoubi, A.; Lübken, M. Biochar for Wastewater Treatment—Conversion. Appl. Sci. 2020, 10, 3492. [Google Scholar] [CrossRef]
- Phiri, Z.; Moja, N.T.; Nkambule, T.T.I.; de Kock, L.A. Utilization of biochar for remediation of heavy metals in aqueous environments: A review and bibliometric analysis. Heliyon 2024, 10, e25785. [Google Scholar] [CrossRef]
- Tang, G.; Mo, H.; Gao, L.; Chen, Y.; Zhou, X. Adsorption of crystal violet from wastewater using alkaline-modified pomelo peel-derived biochar. J. Water Process Eng. 2024, 68, 106334. [Google Scholar] [CrossRef]
- Peerakiatkhajohn, P.; Wongburi, P.; Nakason, K.; Panyapinyopol, B.; Nueangnoraj, K.; Sakulaue, P.; Poggio, D.; Nimmo, W.; Phanthuwongpakdee, J. Sustainable Removal of Methylene Blue Using Minimally Modified Hydrochar from Durian Peels with Experimental Adsorption and Density Functional Theory Studies. Chem. Eng. J. Adv. 2025, 25, 101011. [Google Scholar] [CrossRef]
- Chaudhary, S.; Chaudhary, M.; Tyagi, V.; Chaubey, S.; Suhas Gupta, V.; Cansado, I.P.d.P.; Ahmed, J. Sustainable Production of Porous Activated Carbon from Hydrothermally Carbonized Jamoya Fruit Seeds and Its Potential for Adsorbing the Azo Dye Carmoisine B. Processes 2025, 13, 385. [Google Scholar] [CrossRef]
- Nirmaladevi, S.; Palanisamy, N. A comparative study of the removal of cationic and anionic dyes from aqueous solutions using biochar as an adsorbent. Desalination Water Treat. 2020, 175, 282–292. [Google Scholar] [CrossRef]
- Sutradhar, S.; Mondal, A.; Kuehne, F.; Krueger, O.; Rakshit, S.K.; Kang, K. Comparison of Oil-Seed Shell Biomass-Based Biochar for the Removal of Anionic Dyes—Characterization and Adsorption Efficiency Studies. Plants 2024, 13, 820. [Google Scholar] [CrossRef]
- Kataya, G.; El Badan, D.; Cornu, D.; Mousawi, A.A.; Bechelany, M.; Hijazi, A. Evaluating Biochar’s Role in Dye Adsorption and Wheat. Materials 2025, 18, 4678. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Chen, J.; Bai, Y.; Gao, J.; Peng, M. Adsorption properties of methyl orange in water by sheep manure biochar. Pol. J. Environ. Stud. 2019, 28, 3791–3797. [Google Scholar] [CrossRef]
- Chen, S.; Qin, C.; Wang, T.; Chen, F.; Li, X.; Hou, H.; Zhou, M. Study on the adsorption of dyestuffs with different properties by sludge-rice husk biochar: Adsorption capacity, isotherm, kinetic, thermodynamics and mechanism. J. Mol. Liq. 2019, 285, 62–74. [Google Scholar] [CrossRef]
- Nithyalakshmi, B.; Saraswathi, R. Removal of colorants from wastewater using biochar derived from leaf waste. Biomass Convers. Biorefin. 2023, 13, 1311–1327. [Google Scholar] [CrossRef]
- Alasmary, Z.; Akanji, M.A. Iron-Modified Biochar Derived from Poultry Manure for Efficient Removal of Methyl Orange Dye from Aqueous Solution. Sustainability 2025, 17, 6008. [Google Scholar] [CrossRef]
- Cansado, I.P.P.; Gonçalves, F.A.M.M.; Nabais, J.M.V.; Ribeiro Carrott, M.M.L.; Carrott, P.J.M. PEEK: An excellent precursor for activated carbon production for high temperature application. Fuel Process. Technol. 2009, 90, 232–236. [Google Scholar] [CrossRef]
- Choma, J.; Osuchowski, L.; Dziura, A.; Marszewski, M.; Jaroniec, M. Benzene and Methane Adsorption on Ultrahigh Surface Area Carbons. Adsorpt. Sci. Technol. 2015, 33, 6–8. [Google Scholar] [CrossRef]
- Carrott, P.J.M.; Cansado, I.P.P.; Carrott, M.M.L.R. Carbon molecular sieves from PET for separations involving CH4, CO2, O2 and N2. Appl. Surf. Sci. 2006, 252, 5948–5952. [Google Scholar] [CrossRef]
- Malini, K.; Selvakumar, D.; Kumar, N.S. Activated carbon from biomass: Preparation, factors improving basicity and surface properties for enhanced CO2 capture capacity—A review. J. CO2 Util. 2023, 67, 102318. [Google Scholar] [CrossRef]
- Sathishkumar, R.; Prakash, R. A review on activated carbons (AC) for CO2 capture applications: Preparation, characterisation and surface modification methods. Fuel 2026, 67, 102318. [Google Scholar] [CrossRef]
- Ali Abd, A.; Roslee Othman, M.; Kim, J. A review on application of activated carbons for carbon dioxide capture: Present performance, preparation, and surface modification for further improvement. Environ. Sci. Pollut. Res. 2021, 28, 43329–43364. [Google Scholar] [CrossRef]
- Belo, C.R.; Cansado IPda, P.; Mourão, P.A.M. Synthetic polymers blend used in the production of high activated carbon for pesticides removals from liquid phase. Environ. Technol. 2017, 38, 285–296. [Google Scholar] [CrossRef] [PubMed]
- da Paixão Cansado, I.P.; Belo, C.R.; Mourão, P.A.M. Pesticides abatement using activated carbon produced from a mixture of synthetic polymers by chemical activation with KOH and K2CO3. Environ. Nanotechnol. Monit. Manag. 2019, 12, 100261. [Google Scholar] [CrossRef][Green Version]
- Tarikuzzaman, M. A Review on Activated Carbon: Synthesis, Properties, and Applications. Eur. J. Adv. Eng. Technol. 2023, 10, 114–123. [Google Scholar]
- Kumawat, L.; Sen, R. A Review On Activated Carbon Production Methods From Coconut Shell And Its Applications. In Proceedings of the International Conference on “Emerging Trends in Engineering, Science & Technology “ETEST-2022”, Jaipur, India, 6–7 May 2022. [Google Scholar]
- Heidarinejad, Z.; Dehghani, M.H.; Heidari, M.; Javedan, G.; Ali, I.; Sillanpää, M. Methods for preparation and activation of activated carbon: A review. Environ. Chem. Lett. 2020, 18, 393–415. [Google Scholar] [CrossRef]
- Chew, T.W.; H’Ng, P.S.; Abdullah, B.C.T.G.L.C.; Chin, K.L.; Lee, C.L.; Hafizuddin, B.M.S.M.N.; TaungMai, L. A Review of Bio-Based Activated Carbon Properties Produced. Materials 2023, 16, 7365. [Google Scholar] [CrossRef]
- Lewoyehu, M. Comprehensive review on synthesis and application of activated carbon from agricultural residues for the remediation of venomous pollutants in wastewater. J. Anal. Appl. Pyrolysis 2021, 159, 105279. [Google Scholar] [CrossRef]
- Kumar Mishra, R.; Singh, B.; Acharya, B. A comprehensive review on activated carbon from pyrolysis of lignocellulosic biomass: An application for energy and the environment. Carbon Resour. Convers. 2024, 7, 100228. [Google Scholar] [CrossRef]
- Teimouri, Z.; Salem, A.; Salem, S. Clean and new strategy for catalytic conversion of agriculture waste shells to activated carbon via microwave-assisted impregnation: Applied and eco-friendly aspect for decoloration of industrial corn syrup and process identifications. J. Environ. Chem. Eng. 2019, 7, 103161. [Google Scholar] [CrossRef]
- Wen, X.; Liu, H.; Zhang, L.; Zhang, J.; Fu, C.; Shi, X.; Chen, X.; Mijowska, E.; Chen, M.-J.; Wang, D.-Y. Large-scale converting waste coffee grounds into functional carbon materials as high-efficient adsorbent for organic dyes. Bioresour. Technol. 2019, 272, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Saleem, J.; Moghal, Z.K.B.; Pradhan, S.; McKay, G. High-performance activated carbon from coconut shells for dye removal: Study of isotherm and thermodynamics. RSC Adv. 2024, 14, 33797–33808. [Google Scholar] [CrossRef]
- Ouedrhiri, A.; Ennabely, M.; Lghazi, Y.; Chafi, M.; Alougayl, S.; Youbi, B.; Halabi, A.K.; Khoukhi, M.; Bimaghra, I. Adsorption of anionic and cationic dyes in aqueous solution by a sustainable and low-cost activated carbon based on argan solid waste treated with H3PO4. Environ. Sci. Pollut. Res. 2024, 31, 62010–62021. [Google Scholar] [CrossRef] [PubMed]
- Jawad, A.H.; Ishak, M.A.M.; Farhan, A.M.; Ismail, K. Response surface methodology approach for optimization of color removal and COD reduction of methylene blue using microwave-induced NaOH activated carbon from biomass waste. Desalination Water Treat. 2017, 62, 208–220. [Google Scholar] [CrossRef]
- Alkhabbas, M.; Al-Ma’abreh, A.M.; Edris, G.; Saleh, T.; Alhmood, H. Adsorption of Anionic and Cationic Dyes on Activated Carbon Prepared from Oak Cupules: Kinetics and Thermodynamics Studies. Int. J. Environ. Res. Public Health 2023, 20, 3280. [Google Scholar] [CrossRef] [PubMed]
- Alhogbi, B.; Altayeb, S.; Bahaidarah, E.; Zawrah, M.F. Removal of Anionic and Cationic Dyes from Wastewater Using Activated Carbon from Palm Tree Fiber Waste. Processes 2021, 9, 416. [Google Scholar] [CrossRef]
- Pathania, D.; Sharma, S.; Singh, P. Removal of methylene blue by adsorption onto activated carbon developed from Ficus carica bast. Arab. J. Chem. 2017, 10, S1445–S1451. [Google Scholar] [CrossRef]
- Geçgel, Ü.; Özcan, G.; Gürpnar, G.Ç. Removal of methylene blue from aqueous solution by activated carbon prepared from pea shells (Pisum sativum). J. Chem. 2013, 2013, 614083. [Google Scholar] [CrossRef]
- Khalil, A.; Mangwandi, C.; Salem, M.A.; Ragab, S.; El Nemr, A. Orange peel magnetic activated carbon for removal of acid orange 7 dye from water. Sci. Rep. 2024, 14, 119. [Google Scholar] [CrossRef] [PubMed]
- Mechnou, I.; Meskini, S.; Mourtah, I.; Lebrun, L.; Hlaibi, M. Use of phosphorus-doped microporous carbon from olive mill wastewater for effective removal of Crystal violet and Methylene blue. J. Clean. Prod. 2023, 393, 136333. [Google Scholar] [CrossRef]
- Husaini, M. Efficient adsorption of cationic and anionic dyes using agricultural waste–based activated carbon. Afr. Res. Rep. 2026, 2, 12–20. [Google Scholar] [CrossRef]
- Cansado, I.P.P.; Geraldo, P.F.; Mourão, P.A.M.; Castanheiro, J.E.; Carreiro, E.P.; Suhas. Utilization of Biomass Waste at Water Treatment. Resources 2024, 13, 37. [Google Scholar] [CrossRef]
- Aguirre-Contreras, S.; López-Ramón, M.V.; Velo-Gala, I.; Álvarez-Merino, M.Á.; Aguilar-Aguilar, A.; Ocampo-Pérez, R. A Comparative Study of the Adsorption of Industrial Anionic Dyes with Bone Char and Activated Carbon Cloth. Water 2025, 17, 3422. [Google Scholar] [CrossRef]
- Activated Carbon Price—Current & Forecasts. Available online: https://www.intratec.us/solutions/primary-commodity-prices/commodity/activated-carbon-prices (accessed on 3 March 2026).
- Giannakoudakis, D.A.; Hosseini-Bandegharaei, A.; Tsafrakidou, P.; Triantafyllidis, K.S.; Kornaros, M.; Anastopoulos, I. Aloe vera waste biomass-based adsorbents for the removal of aquatic pollutants: A review. J. Environ. Manag. 2018, 227, 354–364. [Google Scholar] [CrossRef] [PubMed]
- Sridhar, A.; Ponnuchamy, M.; Kapoor, A.; Prabhakar, S. Valorization of food waste as adsorbents for toxic dye removal from contaminated waters: A review. J. Hazard. Mater. 2022, 424, 127432. [Google Scholar] [CrossRef] [PubMed]
- Paradelo, R.; Vecino, X.; Moldes, A.B.; Barral, M.T. Potential use of composts and vermicomposts as low-cost adsorbents for 1 dye removal: An overlooked application. Environ. Sci. Pollut. Res. 2019, 26, 21085–21097. [Google Scholar] [CrossRef]
- Aranda-Figueroa, M.G.; Romero, R.J.; Rodríguez, M.; Rodríguez-Torres, A.; Rodríguez, A.; Bolio-López, G.I.; Arias-Ataide, D.M.; Torres-Islas, Á.; Valladares-Cisneros, M.G. Removal of Azo Dyes from Water on a Large Scale Using a Low-Cost and Eco-Friendly Adsorbent. Sustainability 2025, 17, 4816. [Google Scholar] [CrossRef]
- Momina Shahadat, M.; Isamil, S. Regeneration performance of clay-based adsorbents for the removal of industrial dyes: A review. RSC Adv. 2018, 8, 24571–24587. [Google Scholar] [CrossRef]
- Haque, A.N.M.A.; Sultana, N.; Sayem, A.S.M.; Smriti, S.A. Sustainable Adsorbents from Plant-Derived Agricultural Wastes for Anionic Dye Removal: A Review. Sustainability 2022, 14, 11098. [Google Scholar] [CrossRef]
- Baskar, A.V.; Bolan, N.; Hoang, S.A.; Sooriyakumar, P.; Kumar, M.; Singh, L.; Jasemizad, T.; Padhye, L.P.; Singh, G.; Vinu, A.; et al. Recovery, regeneration and sustainable management of spent adsorbents from wastewater treatment streams: A review. Sci. Total Environ. 2022, 822, 153555. [Google Scholar] [CrossRef] [PubMed]
- Attia, A.A.; Rashwan, W.E.; Khedr, S.A. Capacity of activated carbon in the removal of acid dyes subsequent to its thermal treatment. Dye. Pigment. 2006, 69, 128–136. [Google Scholar] [CrossRef]
- Mekuria, D.; Diro, A.; Melak, F.; Asere, T.G. Adsorptive Removal of Methylene Blue Dye Using Biowaste Materials: Barley Bran and Enset Midrib Leaf. J. Chem. 2022, 2022, 4849758. [Google Scholar] [CrossRef]
- Shakoor, S.; Nasar, A. Removal of methylene blue dye from artificially contaminated water using citrus limetta peel waste as a very low cost adsorbent. J. Taiwan Inst. Chem. Eng. 2016, 66, 154–163. [Google Scholar] [CrossRef]
- Güleç, F.; Williams, O.; Samson, A.; Kostas, E.T.; Stevens, L.A.; Lester, E. Exploring the Utilisation of Natural Biosorbents for Effective Methylene Blue Removal. Appl. Sci. 2024, 14, 81. [Google Scholar] [CrossRef]
- Alhujaily, A.; Yu, H.; Zhang, X.; Ma, F. Adsorptive removal of anionic dyes from aqueous solutions using spent mushroom waste. Appl. Water Sci. 2020, 10, 183. [Google Scholar] [CrossRef]
- El Messaoudi, N.; Dbik, A.; El Khomri, M.; Sabour, A.; Bentahar, S.; Lacherai, A. Date stones of Phoenix dactylifera and jujube shells of Ziziphus lotus as potential biosorbents for anionic dye removal. Int. J. Phytoremediat. 2017, 19, 1047–1052. [Google Scholar] [CrossRef] [PubMed]
- Danouche, M.; El Arroussi, H.; El Ghachtouli, N. Bioremoval of Acid Red 14 dye by Wickerhamomyces anomalus biomass: Kinetic and thermodynamic study, characterization of physicochemical interactions, and statistical optimization of the biosorption process. Biomass Convers. Biorefin. 2024, 14, 2829. [Google Scholar] [CrossRef]
- Cansado, I.P.P.; Mourão, P.A.M. Impact of the use of co-adjuvants agents during chemical activation on the performance of activated carbons in the removal of 4-chloro-2-methyl-phenoxyacetic acid. Environ. Technol Innov. 2021, 24, 102058. [Google Scholar] [CrossRef]
- Khataee, A.; Kayan, B.; Kalderis, D.; Karimi, A.; Akay, S.; Konsolakis, M. Ultrasound-assisted removal of Acid Red 17 using nanosized Fe3O4-loaded coffee waste hydrochar. Ultrason. Sonochem. 2017, 35, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Chen, Z.; Du, W.; Liu, P.; Zhang, L.; Shi, F. Treatment of wastewater containing methyl orange dye by fluidized three dimensional electrochemical oxidation process integrated with chemical oxidation and adsorption. J. Environ. Manag. 2022, 311, 114775. [Google Scholar] [CrossRef] [PubMed]
- Jevremovic, A.; Rankovic, M.; Ležajic, A.J.; Uskokovic-Markovic, S.; Vasiljevic, B.N.; Gavrilov, N.; Bajuk-Bogdanović, D.; Milojević-Rakić, M. Regeneration or Repurposing of Spent Pollutant Adsorbents in energy-related applications: A sustainable choice? Sustain. Chem. 2025, 6, 28. [Google Scholar] [CrossRef]
- El Messaoudi, N.; El Khomri, M.; Bentahar, S.; Dbik, A.; Lacherai, A.; Bakiz, B. Evaluation of performance of chemically treated date stones: Application for the removal of cationic dyes from aqueous solutions. J. Taiwan Inst. Chem. Eng. 2016, 67, 244–253. [Google Scholar] [CrossRef]
- Eren, S.; Türk, F.N.; Arslanoğlu, H. Synthesis of zeolite from industrial wastes: A review on characterization and heavy metal and dye removal. Environ. Sci. Pollut. Res. 2024, 31, 41791–41823. [Google Scholar] [CrossRef] [PubMed]
- Hammood, Z.A.; Chyad, T.F.; Al-Saedi, R. Adsorption Performance of Dyes over Zeolite for Textile Wastewater Treatment. Ecol. Chem. Eng. S 2021, 28, 329–337. [Google Scholar] [CrossRef]
- Sultakhan, S.; Kunarbekova, M.; Khalkhabai, B.; Kakimov, U.; Kuldeyev, E.; Berndtsson, R.; Lee, J.; Azat, S. Performance of a Zeolite-Filled Slow Filter for Dye Removal and Turbidity Reduction. Water 2025, 17, 3557. [Google Scholar] [CrossRef]
- Chen, C.W.; Yang, T.L.; Chen, Y.C. Using magnetic micelles as adsorbents to remove dyes from aqueous solutions. J. Environ. Chem. Eng. 2023, 11, 109457. [Google Scholar] [CrossRef]
- Hegazi, H.A. Removal of heavy metals from wastewater using agricultural and industrial wastes as adsorbents. HBRC J. 2013, 9, 276–282. [Google Scholar] [CrossRef]
- Han, R.; Ding, D.; Xu, Y.; Zou, W.; Wang, Y.; Li, Y.; Zou, L. Use of rice husk for the adsorption of congo red from aqueous solution in column mode. Bioresour. Technol. 2008, 99, 2938–2946. [Google Scholar] [CrossRef] [PubMed]
- Batista, T.; Cansado, I.P.P.; Tita, B.; Mourão, P.A.M.; Nabais, J.M.V.; Castanheiro, J.E.; Borges, C.; Matos, G. Dealing with PlasticWaste from Agriculture Activity. Agronomy 2022, 12, 134. [Google Scholar] [CrossRef]
- Hotan Alsohaimi, I.; Alhumaimess, M.S.; Abdullah Alqadami, A.; Tharwi Alshammari, G.; Fawzy Al-Olaimi, R.; Abdeltawab, A.A.; El-Sayed, M.Y.; Hassan, H.M. Adsorptive performance of aminonaphthalenesulfonic acid modified magnetic-graphene oxide for methylene blue dye: Mechanism, isotherm and thermodynamic studies. Inorg. Chem. Commun. 2023, 147, 110261. [Google Scholar] [CrossRef]
- Bal, G.; Thakur, A. Distinct approaches of removal of dyes from wastewater: A review. Mater. Today Proc. 2022, 50, 1575–1579. [Google Scholar] [CrossRef]
- Rahmoun, H.B.; Boumediene, M.; Ghenim, A.N.; Da Silva, E.F.; Labrincha, J. Coupling Coagulation–Flocculation–Sedimentation with Adsorption on Biosorbent (Corncob) for the Removal of Textile Dyes from Aqueous Solutions. Environments 2025, 12, 201. [Google Scholar] [CrossRef]

| Comparison Between Biological, Chemical and Physicochemical Process | |||
|---|---|---|---|
| Parameter | Biological | Chemical | Physicochemical |
| Main advantages | Environmentally friendly and sustainable | Fast and effective for a wide range of dyes | Easy to operate, effective in a wide range of dyes |
| Drawback | Ineffective for many synthetic or toxic dyes | Risk of forming toxic by-products | Cost of adsorbents |
| Mechanism | Microbial degradation | Chemical oxidation | Adsorption |
| Effectiveness on synthetic dyes | Low (mainly with recalcitrant dyes | Effective for a dye diversity degradation | High (could remove non-biodegradable dyes) |
| Treatment time | Slow (hours to days) | Relatively fast | Fast (minutes to hours) |
| Sensitive to conditions | Highly sensitive to pH, temperature, and dyes toxicity | pH and chemical dosages must be controlled | High (pH, concentration, T must be controlled) |
| Toxicity handling | Toxic dyes may inhibit or kill microbes | Degrade recalcitrant and non-biodegradable dyes | Handles toxics dyes well |
| Sludge production | Produce less amount of wastes (biomass/sludge needs disposal) | Production of chemical sludge, which must be treated and disposed | Minimal to moderate chemical waste (spent adsorbents must be treated or disposed |
| Cost | Often low operational cost, high maintenance cost | High chemical cost and maintenance | The cost depends on the adsorbent used |
| Regeneration/reuse | Difficult (sludge cannot be reused | Feasible (with suitable adsorbent) | |
| By-products | May produce toxic intermediate metabolites | Risk of forming toxic by-products | Generally, none or non-toxic products are produced |
| Scalability | It requires a large installation area, better suited for large-scale | Requires precise control of reaction conditions (pH, dosage, etc.) | Easy for small and medium-scale setups |
| Factors Affecting the Adsorption Performance | |||
|---|---|---|---|
| Adsorbents | Solution | Adsorbate | Removal Rate |
| Surface area | Temperature | Cationic or anionic | Adsorption site available |
| Porous volume | Other pollutants | Water solubility | Functional groups |
| Mean pore size | Organic matter | Concentration | Competitie adsorption |
| Surface chemistry | pH | Molecular size | Agitation speed |
| Particle size | Contact time | ||
| Adsorbents | Raw Materials Used | Advantages | Drawbacks | References |
|---|---|---|---|---|
| Clays | bentonite, kaolinite, montmorillonite | Local available, low cost, non-toxic and biodegradable | Low adsorption capacity, mainly for anionic dyes. Regeneration maybe costly or inefficient | [4] |
| Zeolites | MCM—22 | Ion-exchange capacity Potential application for removing dyes and dissolved inorganic contaminants Production of synthetic zeolites | Lower adsorption capacity Chemical regeneration is not very successful Molecular sieve effect for larger molecules | [128] [129] [130] |
| Magnetic adsorbents | Magnetic micelles | Small size particles, low cost, high surface area, amount of active sites High efficiency | Production of magnetic adsorbent is time-consuming and involve complicated synthesis steps | [131] |
| Industrial wastes | Sludge, red mud, metal hydroxide sludge and fly ash | High amount of low-cost materials High surface area and porous volume | Moderate to high cost for processing Not recommended for industrial scale | [132] |
| Agricultural wastes | Fruit peels, bagasse, coir pith, maize cob, bark materials from the cleaning of soils and trees, barley bran. | High amount of low-cost materials available locally. Strong affinity for metallic ions and dyes. Environmental sustainability | Performance depends on pH and temperature. Not recommended for industrial scale. Release of soluble organic compounds into the water body. | [133] [134] [117] |
| Composite adsorbents | Lignocellulosic wastes with magnetic particles Graphene with metal oxide particles | High amount of active sites, adsorption capacity and efficiency | Performance depends on pH and temperature, not adequate for column setup, promotes secondary pollution and needs additional cost for processing | [135] |
| Biochar and Activated carbon | Industrial, agricultural, manure, sludge, and urban solid by-products | Low-cost precursors. High surface area and porous volume Chemical and physical treatments available Reuse and regeneration treatments available | Moderate-high cost to obtain ACs in high amount. Generate secondary pollution Need replacement and disposal of spent adsorbents High cost to regenerate | [65] [4] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Cansado, I.P.d.P.; Mourão, P.A.M.; Castanheiro, J.E.F.; Geraldo, P.F.; Suhas; Suero, S.R.; Cano, B.L. Review on Treatment Pathways and Adsorptive Approaches for Dye-Contaminated Wastewater. Processes 2026, 14, 898. https://doi.org/10.3390/pr14060898
Cansado IPdP, Mourão PAM, Castanheiro JEF, Geraldo PF, Suhas, Suero SR, Cano BL. Review on Treatment Pathways and Adsorptive Approaches for Dye-Contaminated Wastewater. Processes. 2026; 14(6):898. https://doi.org/10.3390/pr14060898
Chicago/Turabian StyleCansado, Isabel Pestana da Paixão, Paulo Alexandre Mira Mourão, José Eduardo Felix Castanheiro, Pedro Francisco Geraldo, Suhas, Silvia Román Suero, and Beatriz Ledesmas Cano. 2026. "Review on Treatment Pathways and Adsorptive Approaches for Dye-Contaminated Wastewater" Processes 14, no. 6: 898. https://doi.org/10.3390/pr14060898
APA StyleCansado, I. P. d. P., Mourão, P. A. M., Castanheiro, J. E. F., Geraldo, P. F., Suhas, Suero, S. R., & Cano, B. L. (2026). Review on Treatment Pathways and Adsorptive Approaches for Dye-Contaminated Wastewater. Processes, 14(6), 898. https://doi.org/10.3390/pr14060898











